Treatment of Effluents from the Textile Industry through Polyethersulfone Membranes
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Membranes Preparation
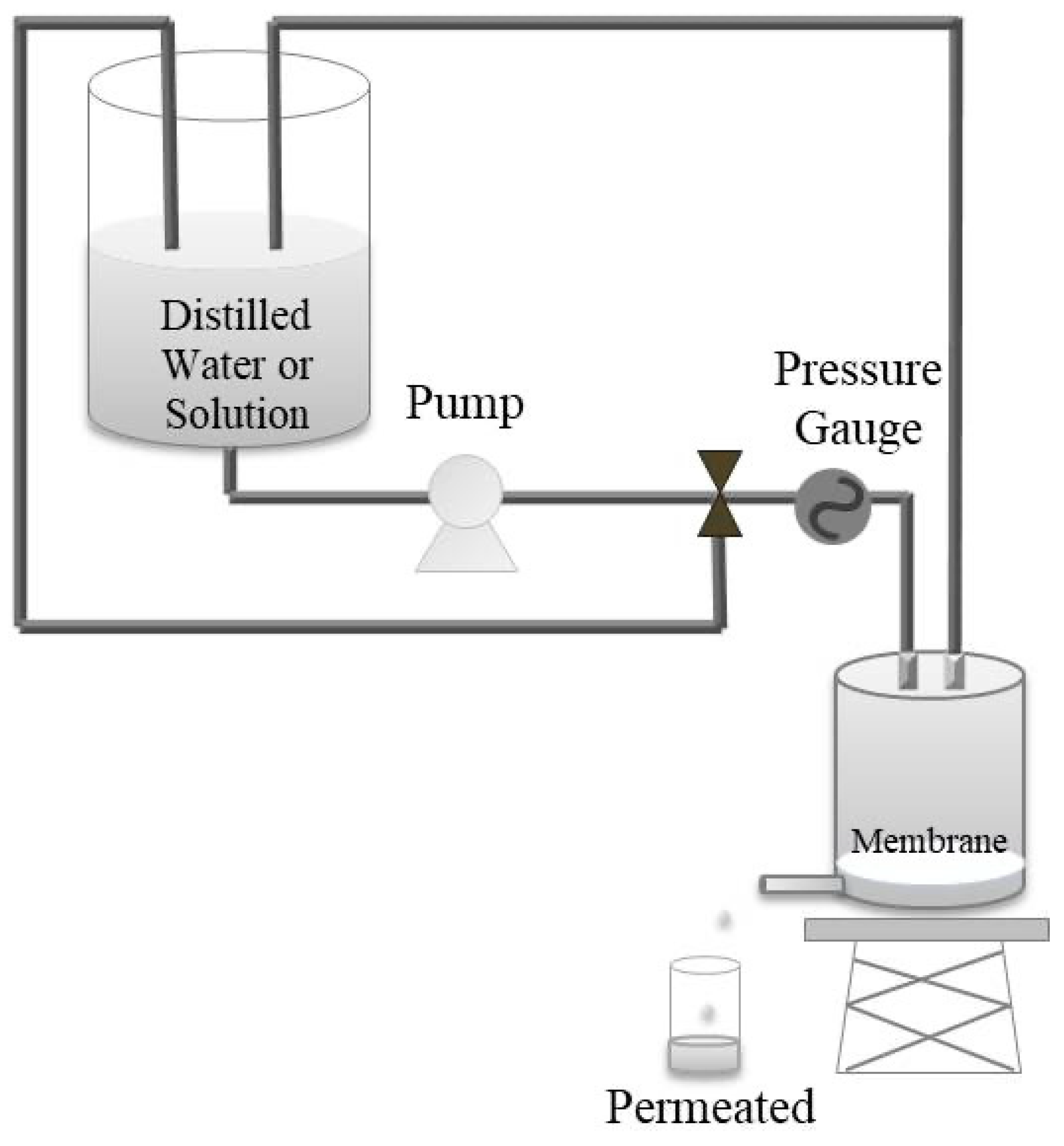
2.3. Characterizations
3. Results and Discussion
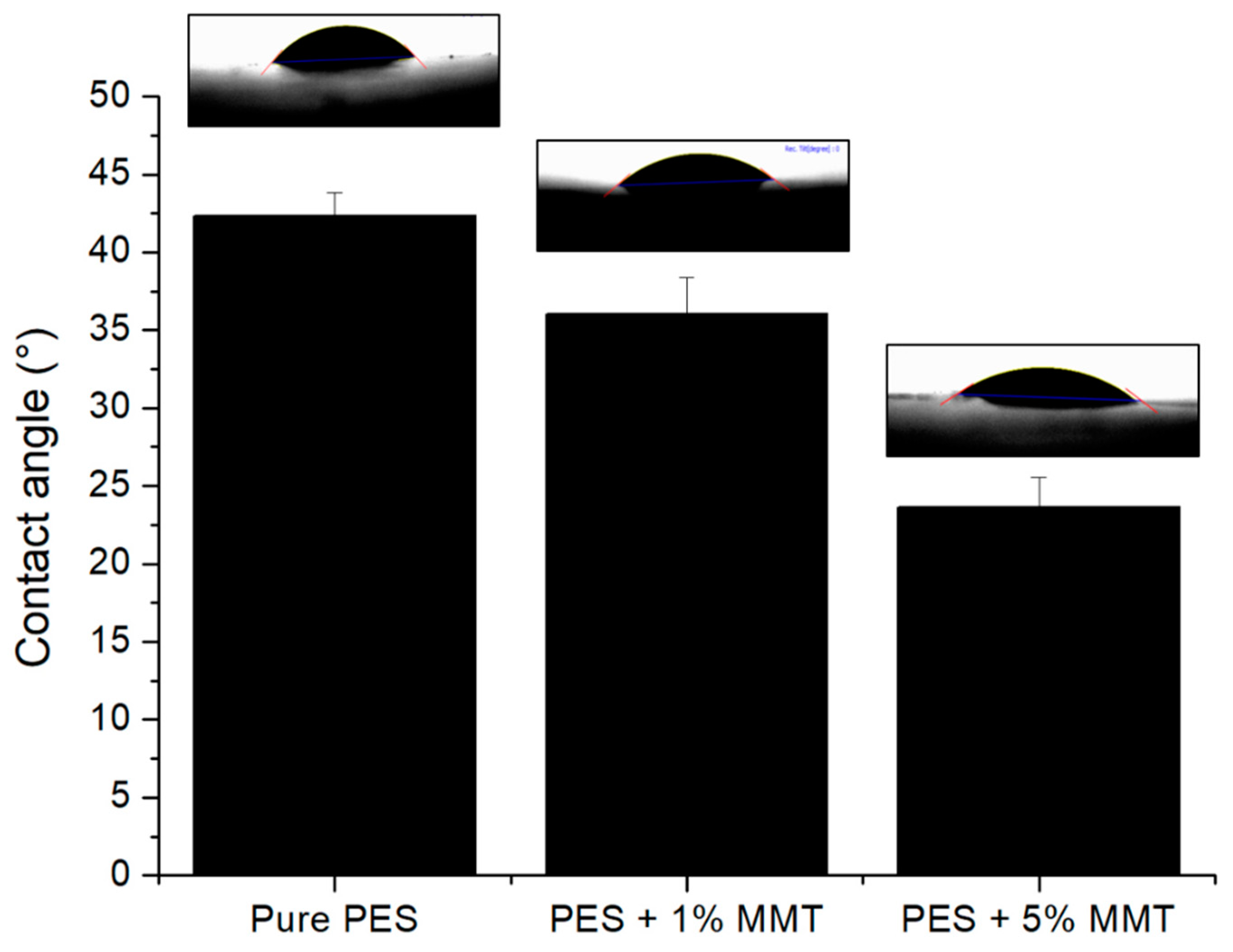
3.1. Effect of Clay on Membrane Hydrophilicity
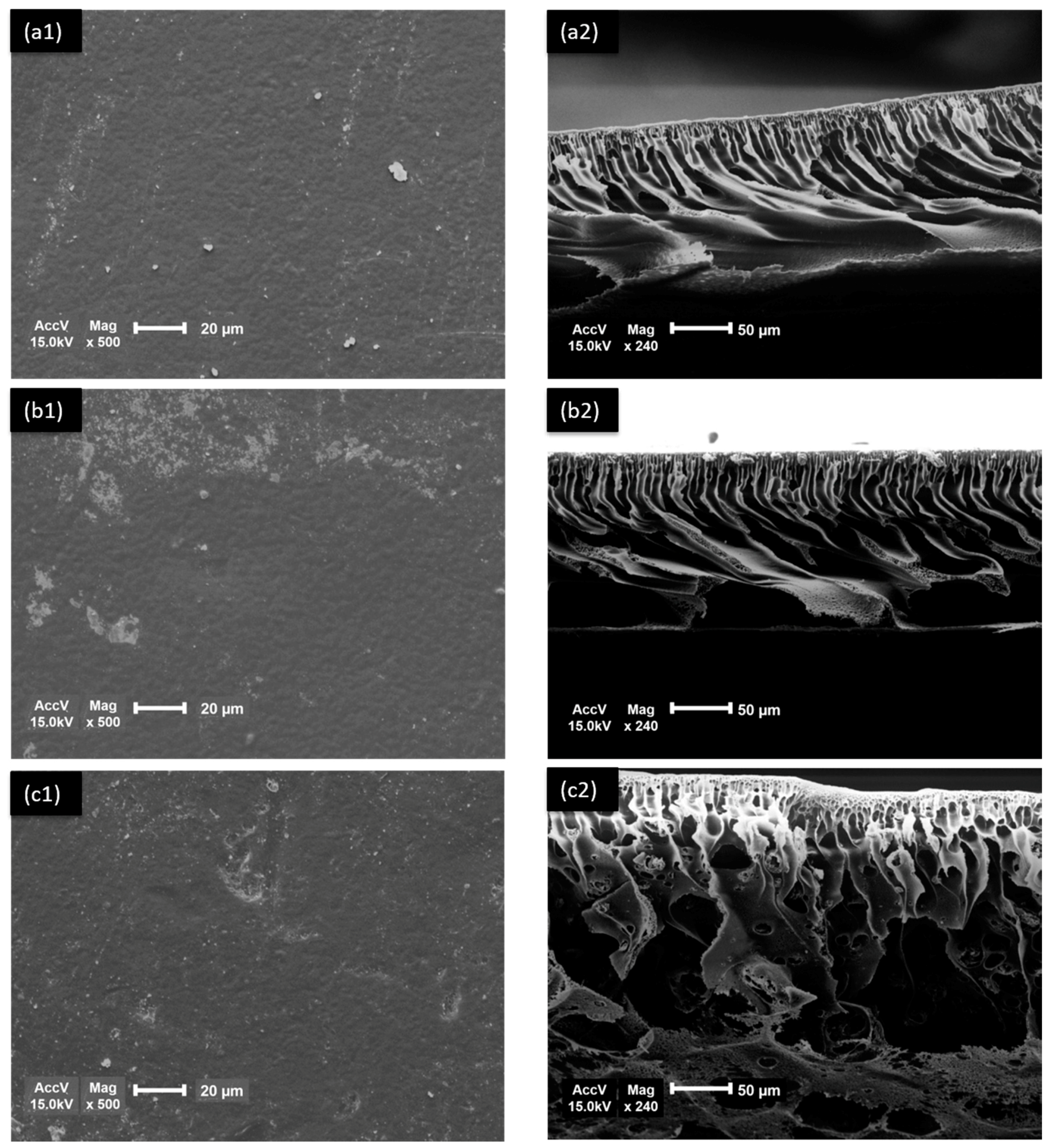
3.2. Membrane Morphology
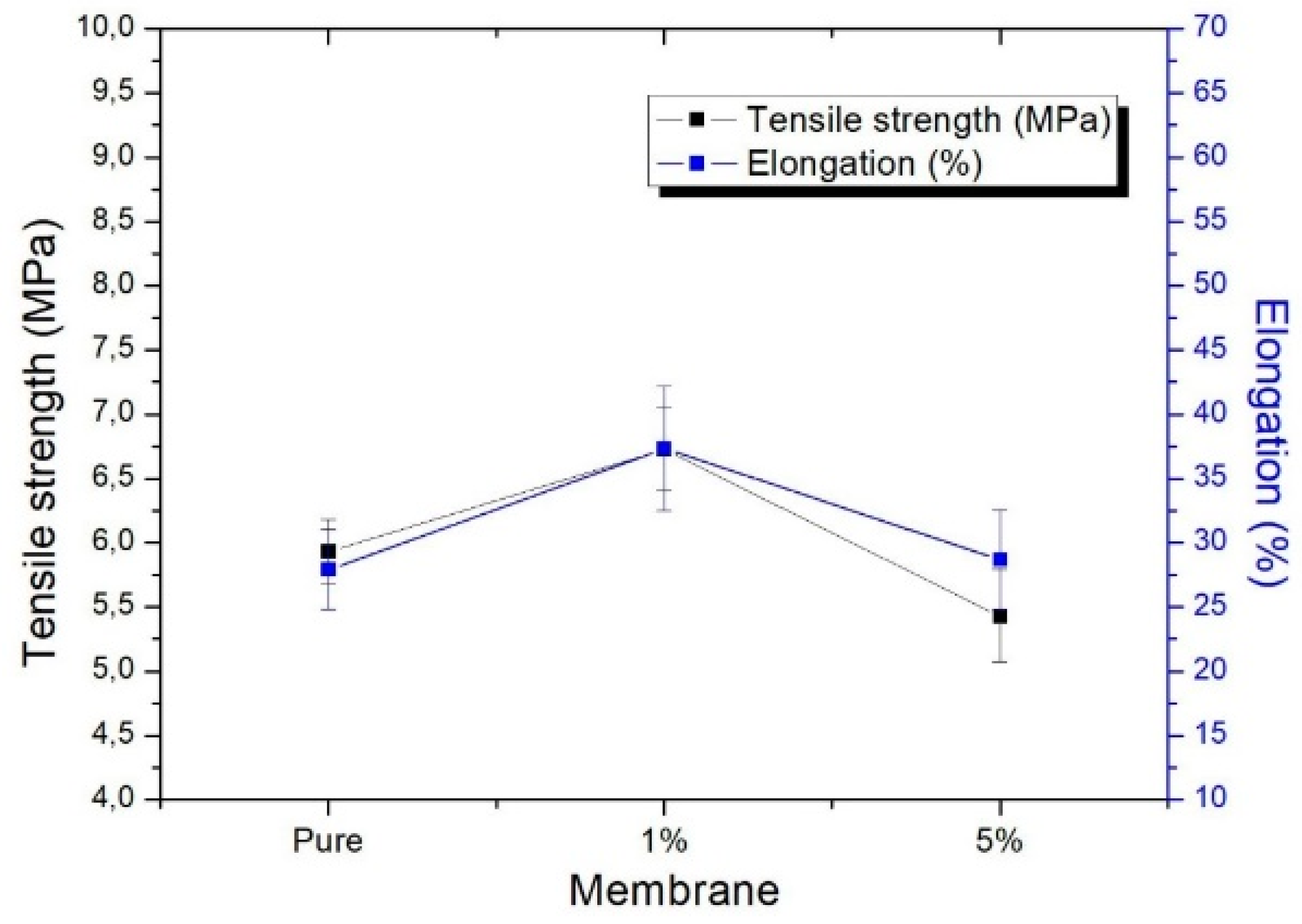
3.3. Tensile Strength
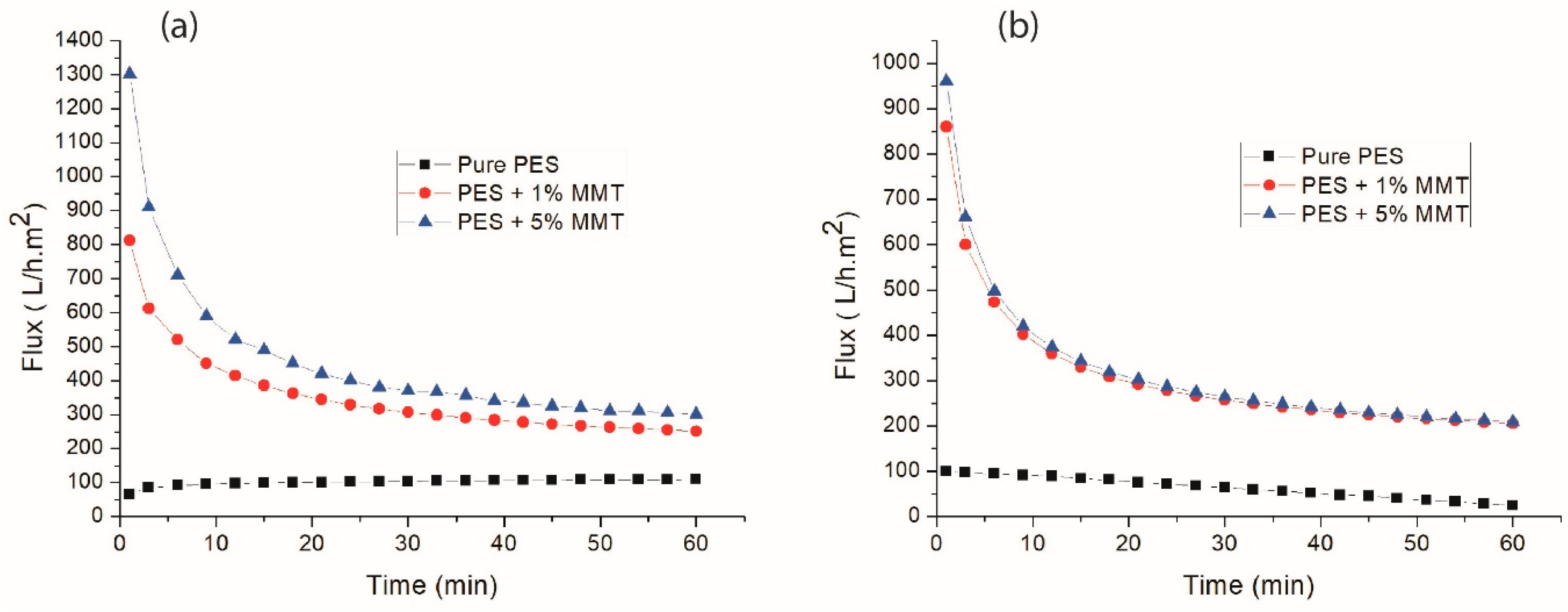
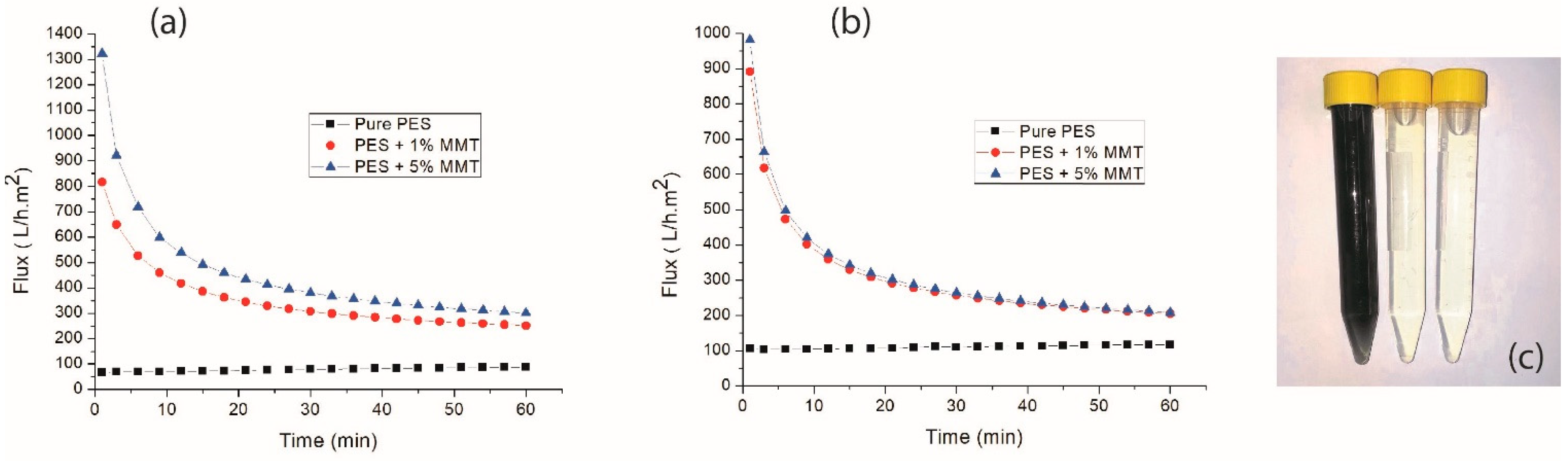
3.4. Membrane Flow Properties and Efficiency
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Xia, Q.C.; Liu, M.L.; Cao, X.L.; Wang, Y.; Xing, W.; Sun, S.P. Structure design and applications of dual-layer polymeric membranes. J. Membr. Sci. 2018, 562, 85–111. [Google Scholar] [CrossRef]
- Farahani, M.H.D.A.; Rabiee, H.; Vatanpour, V. Comparing the effect of incorporation of various nanoparticulate on the performance and antifouling properties of polyethersulfone nanocomposite membranes. J. Water Process Eng. 2019, 27, 47–57. [Google Scholar] [CrossRef]
- Liu, F.; Hashim, N.A.; Liu, Y.; Abed, M.R.M.; Li, K. Progress in the production and modification of PVDF membranes. J. Membr. Sci. 2011, 375, 1–27. [Google Scholar] [CrossRef]
- Amaral, M.C.S.; Neta, L.S.F.; Souza, M.; Cerqueira, N.; Carvalho, R.B.D. Evaluation of operational parameters from a microfiltration system for indigo blue dye recovery from textile dye effluent. Desalin. Water Treat. 2014, 52, 257–266. [Google Scholar] [CrossRef]
- Li, L.; Yan, G.; Wu, J.; Yu, X.; Guo, Q. Surface-initiated atom-transfer radical polymerization from polyethersulfone membranes and their use in antifouling. e-Polymers 2009, 9. [Google Scholar] [CrossRef][Green Version]
- Xu, W.; Ge, Q. Synthetic polymer materials for forward osmosis (FO) membranes and FO applications: A review. Res. Chem. Eng. 2019, 35, 191–209. [Google Scholar] [CrossRef]
- Goh, P.S.; Ong, C.S.; Ng, B.C.; Ismail, A.F. Nanotechnology in Water and Wastewater Treatment: Applications of Emerging Nanomaterials for Oily Wastewater Treatment; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Wu, G.; Gan, S.; Cui, L.; Xu, Y. Preparation and characterization of PES/TiO2 composite membranes. Appl. Surf. Sci. 2008, 254, 7080–7086. [Google Scholar] [CrossRef]
- Shen, L.; Bian, X.; Lu, X.; Shi, L.; Liu, Z.; Chen, L.; Hou, Z.; Fan, K. Preparation and characterization of ZnO/polyethersulfone (PES) hybrid membranes. Desalination 2012, 293, 21–29. [Google Scholar] [CrossRef]
- Razmjou, A.; Mansouri, J.; Chen, V. The effects of mechanical and chemical modification of TiO2 nanoparticles on the surface chemistry, structure and fouling performance of PES ultrafiltration membranes. J. Membr. Sci. 2011, 378, 73–84. [Google Scholar] [CrossRef]
- Hosseini, S.A.; Vossoughi, M.; Mahmoodi, N.M.; Sadrzadeh, M. Clay-based electrospun nanofibrous membranes for colored wastewater treatment. Appl. Clay Sci. 2019, 168, 77–86. [Google Scholar] [CrossRef]
- Simona, C.; Raluca, I.; Anita-Laura, R.; Andrei, S.; Raluca, S.; Bogdan, T.; Elvira, A.; Catalin-Ilie, S.; Claudiu, F.R.; Daniela, I.-E.; et al. Synthesis, characterization and efficiency of new organically modified montmorillonite polyethersulfone membranes for removal of zinc ions from wastewasters. Appl. Clay Sci. 2017, 137, 135–142. [Google Scholar] [CrossRef]
- Mierzwa, J.C.; Arieta, V.; Verlage, M.; Carvalho, J.; Vecitis, C.D. Effect of clay nanoparticles on the structure and performance of polyethersulfone ultrafiltration membranes. Desalination 2013, 314, 147–158. [Google Scholar] [CrossRef]
- ASTM. Standard Test Methods for Tensile Properties of Thin Plastic Sheeting. D 882. Annual Book of ASTM.; American Society for Testing and Materials: Philadelphia, PA, USA, 1992. [Google Scholar]
- Ahmad, A.L.; Abdulkrim, A.A.; Shafie, Z.M.H.M.; Ooi, B.S. Fouling evaluation of PES/ZnO mixed matrix hollow fiber membrane. Desalination 2017, 403, 53–63. [Google Scholar] [CrossRef]
- Shen, J.N.; Ruan, H.M.; Wu, L.G.; Gao, C.J. Preparation and characterization of PES–SiO2 organic–inorganic composite ultrafiltration membrane for raw water pretreatment. Chem. Eng. J. 2011, 168, 1272–1278. [Google Scholar] [CrossRef]
- Leo, C.P.; Lee, W.C.; Ahmad, A.L.; Mohammad, A.W. Polysulfone membranes blended with ZnO nanoparticles for reducing fouling by oleic acid. Sep. Purif. Technol. 2012, 89, 51–56. [Google Scholar] [CrossRef]
- Junaidi, N.F.D.; Othman, N.H.; Shahruddin, M.Z.; Alias, N.H.; Lau, W.J.; Ismail, A.F. Effect of graphene oxide (GO) and polyvinylpyrollidone (PVP) additives on the hydrophilicity of composite polyethersulfone (PES) membrane. Malays. J. Appl. Sci. 2019, 15, 361–366. [Google Scholar]
- Ghaemi, N.; Madaeni, S.S.; Alizadeh, A.; Rajabi, H.; Daraei, P. Preparation, characterization and performance of polyethersulfone/organically modified montmorillonite nanocomposite membranes in removal of pesticides. J. Membr. Sci. 2011, 382, 135–147. [Google Scholar] [CrossRef]
- Ferreira, R.S.B.; Pereira, C.H.Ó.; Paz, R.A.; Leite, A.M.D.; Araújo, E.M.; Lira, H.L. Influence of Processing Type in the Morphology of Membranes Obtained from PA6/MMT nanocomposites. Adv. Mater. Sci. Eng. 2014, 2014, 659148. [Google Scholar] [CrossRef]
- Rekik, S.B.; Gassara, S.; Bouaziz, J.; Deratani, A.; Baklouti, S. Enhancing hydrophilicity and permeation flux of chitosan/kaolin composite membranes by using polyethylene glycol as porogen. Appl. Clay Sci. 2019, 168, 312–323. [Google Scholar] [CrossRef]
- Daraei, P.; Ghaemi, N. Synergistic effect of Cloisite 15A and 30B nanofillers on the characteristics of nanocomposite polyethersulfone membrane. Appl. Clay Sci. 2019, 172, 96–105. [Google Scholar] [CrossRef]
- Rambabu, K.; Bharath, G.; Monash, P.; Velu, S.; Banat, F.; Naushad, M.; Arthanareeswaran, G.; Show, P.L. Effective treatment of dye polluted wastewater using nanoporous CaCl2 modified polyethersulfone membrane. Process Saf. Environ. 2019, 124, 266–278. [Google Scholar] [CrossRef]
- Wang, X.L.; Qian, H.J.; Chen, L.J.; Lu, Z.Y.; Li, Z.S. Dissipative particle dynamics simulation on the polymer membrane formation by immersion precipitation. J. Membr. Sci. 2008, 311, 251–258. [Google Scholar] [CrossRef]
- Ahmad, A.L.; Abdulkarim, A.A.; Ismail, S.; Ooi, B.S. Preparation and characterisation of PES-ZnO mixed matrix membranes for humic acid removal. Desalin. Water Treat. 2015, 54, 3257–3268. [Google Scholar] [CrossRef]
- Rezaei, M.; Ismail, A.F.; Hashemifard, S.A.; Bakeri, G.; Matsuura, T. Experimental study on the performance and long-term stability of PVDF/montmorillonite hollow fiber mixed matrix membranes for CO2 separation process. Int. J. Greenh. Gas Con. 2014, 26, 147–157. [Google Scholar] [CrossRef]
- Sotto, A.; Rashed, A.; Zhang, R.X.; Martínez, A.; Braken, L.; Luis, P.; Van der Bruggen, B. Improved membrane structures for seawater desalination by studying the influence of sublayers. Desalination 2012, 287, 317–325. [Google Scholar] [CrossRef]
- Yeh, J.M.; Yu, M.Y.; Liou, S.J. Dehydration of water–alcohol mixtures by vapor permeation through PVA/clay nanocomposite membrane. J. Appl. Polym. Sci. 2003, 89, 3632–3638. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, R. Novel method for incorporating hydrophobic silica nanoparticles on polyetherimide hollow fiber membranes for CO2 absorption in a gas–liquid membrane contactor. J. Membr. Sci. 2014, 452, 379–389. [Google Scholar] [CrossRef]
- Ghalamchi, L.; Aber, S.; Vatanpour, V.; Kian, M. A novel antibacterial mixed matrixed PES membrane fabricated from embedding aminated Ag3PO4/g-C3N4 nanocomposite for use in the membrane bioreactor. J. Ind. Eng. Chem. 2019, 70, 412–426. [Google Scholar] [CrossRef]
- Ismail, N.M.; Ismail, A.F.; Mustafa, A.; Matsuura, T.; Soga, T.; Nagata, K.; Asaka, T. Qualitative and quantitative analysis of intercalated and exfoliated silicate layers in asymmetric polyethersulfone/cloisite15A® mixed matrix membrane for CO2/CH4 separation. Chem. Eng. J. 2015, 268, 371–383. [Google Scholar] [CrossRef]
- Azeez, A.A.; Rhee, K.Y.; Park, S.J.; Hui, D. Epoxy clay nanocomposites–processing, properties and applications: A review. Compos. Part B Eng. 2013, 45, 308–320. [Google Scholar] [CrossRef]
- Muhamad, M.S.; Salim, M.R.; Lau, W.J. Surface modification of SiO2 nanoparticles and its impact on the properties of PES-based hollow fiber membrane. RSC Adv. 2015, 5, 58644–58654. [Google Scholar] [CrossRef]
- Ferreira, R.S.B.; Oliveira, S.S.L.; Salviano, A.F.; Araújo, E.M.; Leite, A.M.D.; Lira, H.L. Polyethersulfone Hollow Fiber Membranes Developed for Oily Emulsion Treatment. Mater. Res. 2019, 22, 1–8. [Google Scholar] [CrossRef]
- Gao, J.; Thong, Z.; Wang, K.Y.; Chung, T.S. Fabrication of loose inner-selective polyethersulfone (PES) hollow fibers by one-step spinning process for nanofiltration (NF) of textile dyes. J. Membr. Sci. 2017, 541, 413–424. [Google Scholar] [CrossRef]
- Lima, R.C.O.; Lira, H.L.; Neves, G.A.; Silva, M.C.; França, K.B. Use of ceramic membrane for indigo separation in effluent from textile industry. Mater. Sci. Forum 2014, 789, 537–541. [Google Scholar] [CrossRef]

| Sample | Solvent (%) | Polymer (%) | Clay (%) | PVP (%) |
|---|---|---|---|---|
| Pure PES | 80 | 15 | 0 | 5 |
| PES + MMT 1% | 80 | 14 | 1 | 5 |
| PES + MMT 5% | 80 | 10 | 5 | 5 |
| Membrane | Ra (nm) | Rz (nm) | Rzjis (nm) |
|---|---|---|---|
| Pure PES | 3.219 | 28.126 | 13.742 |
| PES + MMT 1% | 4.108 | 40.77 | 20.191 |
| PES + MMT 5% | 9.401 | 63.593 | 31.533 |
| Membrane | Flux (L/h·m2) 1.0 bar | Rejection (%) | Flux (L/h·m2) 1.5 bar | Rejection (%) |
|---|---|---|---|---|
| Pure PES | 88.0 ± 1.15 | 83.0 ± 1.22 | 114.0 ± 0.84 | 89.0 ± 0.95 |
| PES + MMT 1% | 251.0 ± 0.92 | 89.0 ± 1.10 | 203.5 ± 1.75 | 94.0 ± 1.50 |
| PES + MMT 5% | 298.5 ± 1.03 | 89.0 ± 1.90 | 203.5 ± 1.60 | 83.0 ± 2.05 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Silva Barbosa Ferreira, R.; Florindo Salviano, A.; Sonaly Lima Oliveira, S.; Maria Araújo, E.; da Nóbrega Medeiros, V.; de Lucena Lira, H. Treatment of Effluents from the Textile Industry through Polyethersulfone Membranes. Water 2019, 11, 2540. https://doi.org/10.3390/w11122540
da Silva Barbosa Ferreira R, Florindo Salviano A, Sonaly Lima Oliveira S, Maria Araújo E, da Nóbrega Medeiros V, de Lucena Lira H. Treatment of Effluents from the Textile Industry through Polyethersulfone Membranes. Water. 2019; 11(12):2540. https://doi.org/10.3390/w11122540
Chicago/Turabian Styleda Silva Barbosa Ferreira, Rodholfo, Aline Florindo Salviano, Sandriely Sonaly Lima Oliveira, Edcleide Maria Araújo, Vanessa da Nóbrega Medeiros, and Hélio de Lucena Lira. 2019. "Treatment of Effluents from the Textile Industry through Polyethersulfone Membranes" Water 11, no. 12: 2540. https://doi.org/10.3390/w11122540
APA Styleda Silva Barbosa Ferreira, R., Florindo Salviano, A., Sonaly Lima Oliveira, S., Maria Araújo, E., da Nóbrega Medeiros, V., & de Lucena Lira, H. (2019). Treatment of Effluents from the Textile Industry through Polyethersulfone Membranes. Water, 11(12), 2540. https://doi.org/10.3390/w11122540






