The Toxicity and Uptake of As, Cr and Zn in a Stygobitic Syncarid (Syncarida: Bathynellidae)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Test Species
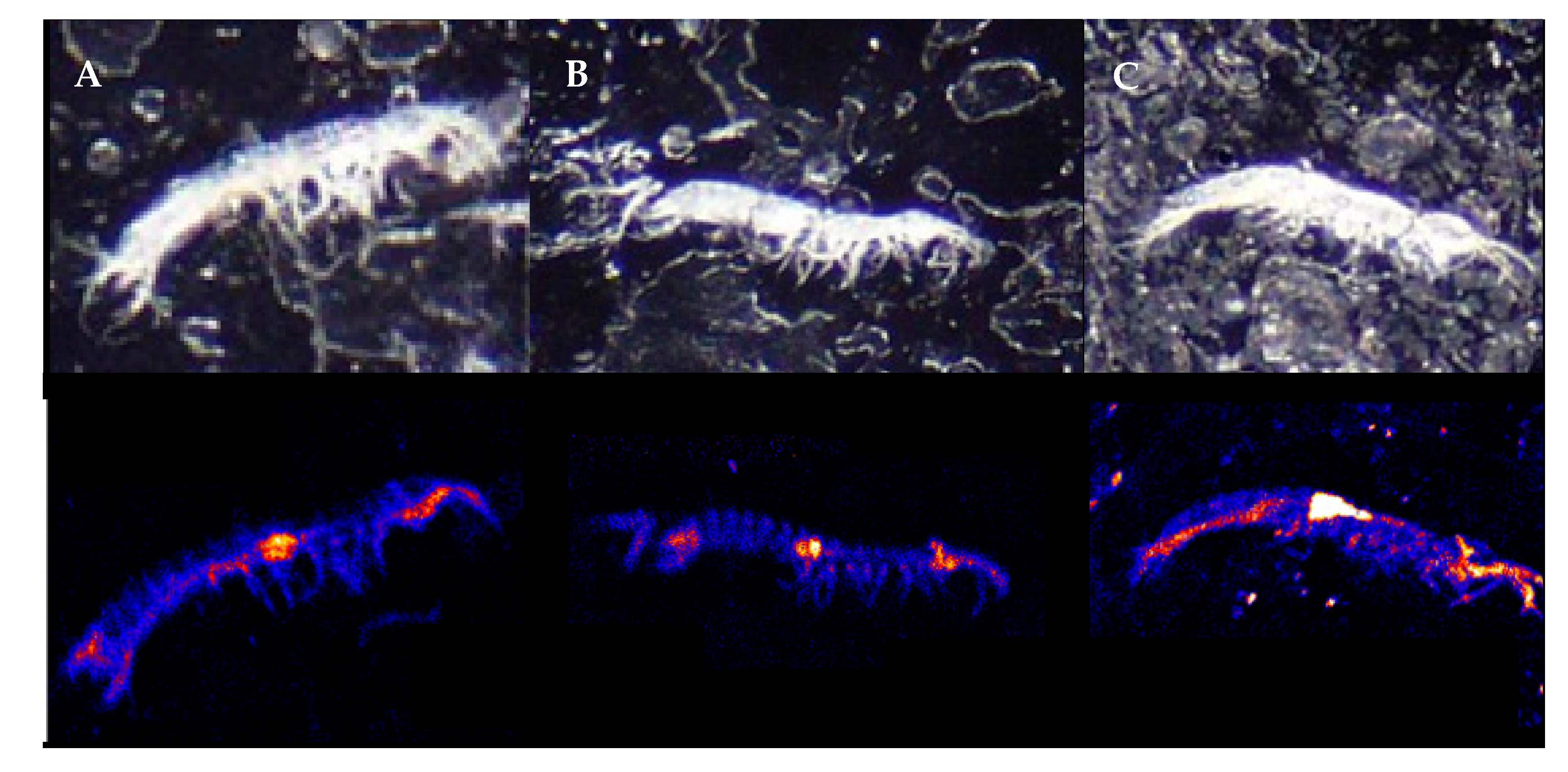
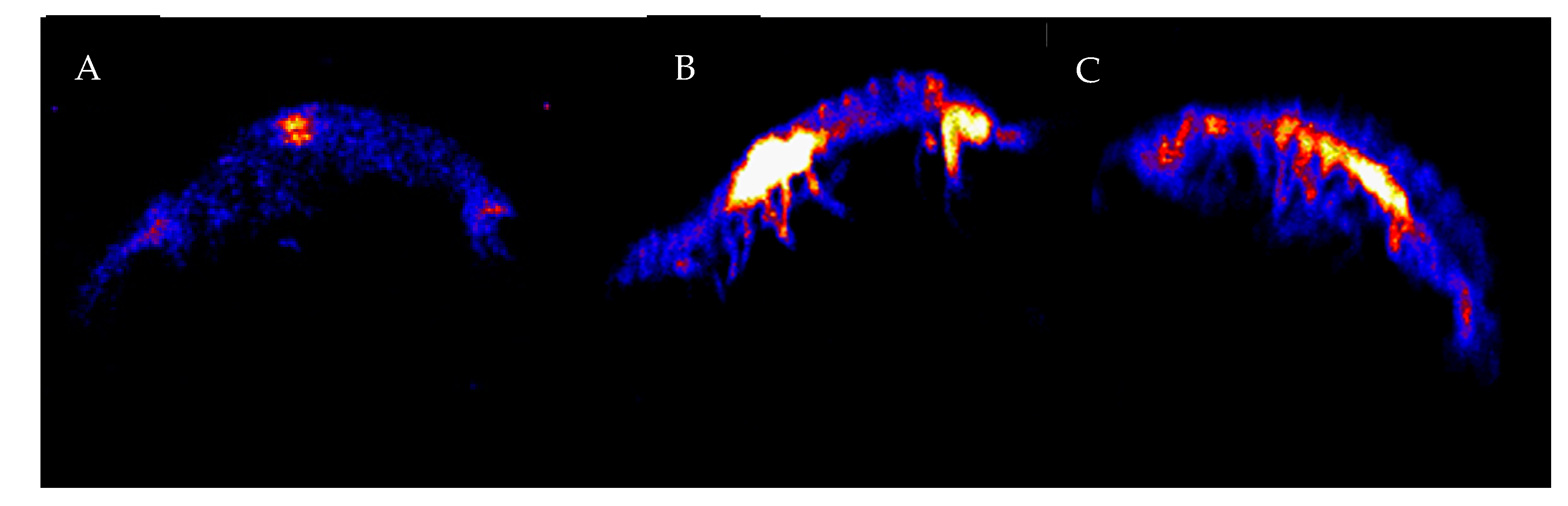
2.2. PIXE Elemental Analysis
2.3. Statistical Analysis
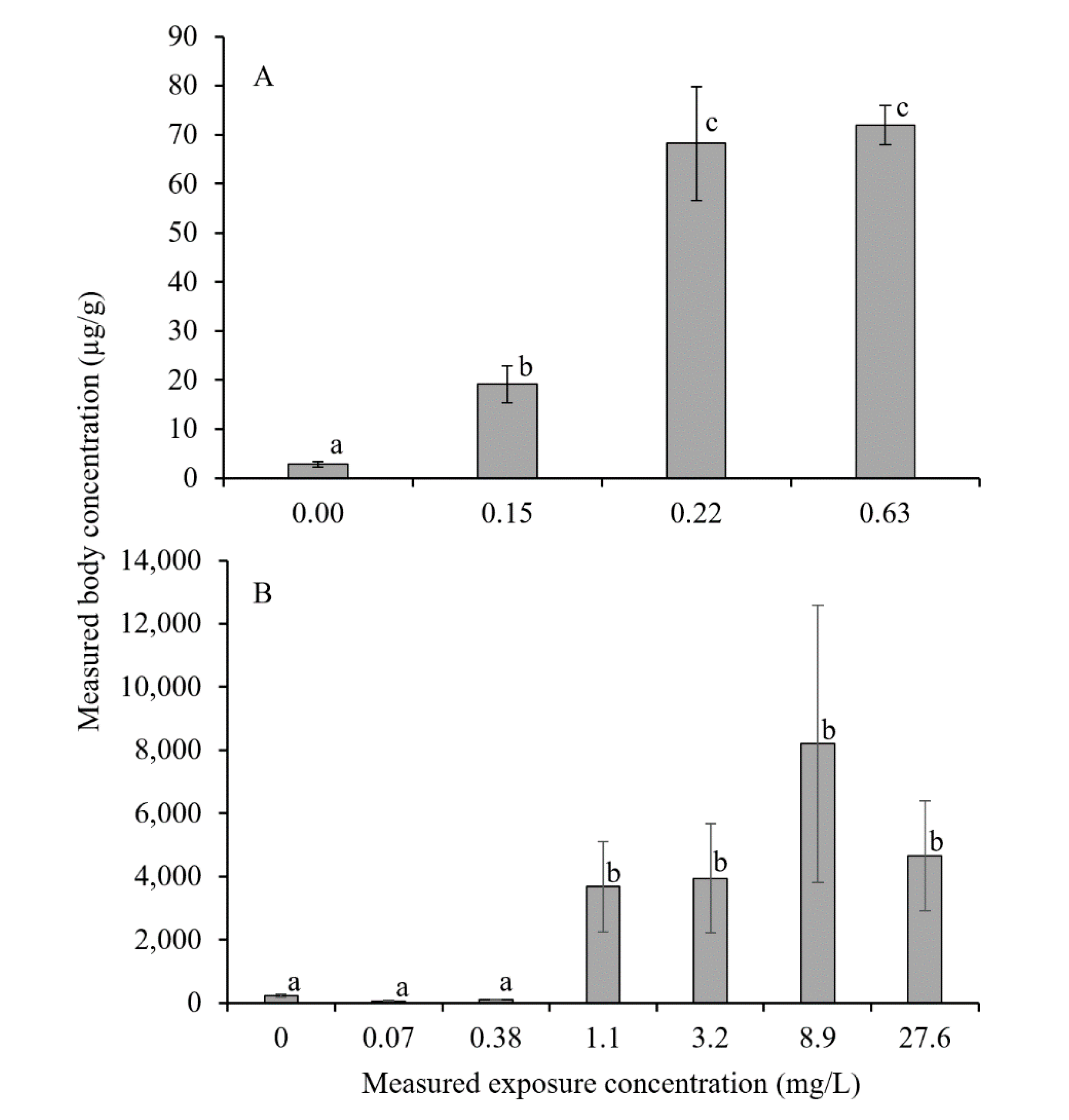
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alley, W.M.; Healy, R.W.; LaBaugh, J.W.; Reilly, T.E. Flow and storage in groundwater systems. Science 2002, 296, 1985. [Google Scholar] [CrossRef] [PubMed]
- Morris, B.L.; Lawrence, A.R.L.; Chilton, P.J.C.; Adams, B.; Calow, R.C.; Klinck, B.A. Groundwater and Its Susceptibility to Degradation: A Global Assessment of the Problem and Options for Management; United Nations Environment Programme: Nairobi, Kenya, 2003; p. 126. [Google Scholar]
- Humphreys, W.F. Aquifers: The ultimate groundwater-dependent ecosystems. Aust. J. Bot. 2006, 54, 115–132. [Google Scholar] [CrossRef]
- Camacho, A.I.; Valdecasas, A.G. Global diversity of syncarids (Syncarida; Crustacea) in freshwater. In Freshwater Animal Diversity Assessment; Balian, E.V., Lévêque, C., Segers, H., Martens, K., Eds.; Springer: Dordrecht, The Netherlands, 2008; pp. 257–266. [Google Scholar]
- Hose, G.C. Response to Humphreys’ (2007) Comments on Hose GC (2005) Assessing the Need for Groundwater Quality Guidelines for Pesticides Using the Species Sensitivity Distribution Approach. Hum. Ecol. Risk Assess. 2007, 13, 241–246. [Google Scholar] [CrossRef]
- Griebler, C.; Avramov, M.; Hose, G. Groundwater ecosystems and their services—Current status and potential risks. In Atlas of Ecosystem Services—Drivers, Risks, and Societal Responses; Schröter, M., Bonn, A., Klotz, S., Seppelt, R., Baessler, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2019; pp. 197–203. [Google Scholar]
- Hose, G.C.; Asmyhr, M.G.; Cooper, S.J.B.; Humphreys, W.F. Down under Down Under: Austral groundwater life. In Austral Ark: The State of Wildlife in Australia and New Zealand; Stow, A., Maclean, N., Holwell, G.I., Eds.; Cambridge University Press: Cambridge, UK, 2015; pp. 512–536. [Google Scholar]
- Castaño-Sánchez, A.; Hose, G.; Reboleira, A. Ecotoxicological effects of anthropogenic stressors in subterranean organisms: A review. Chemosphere 2019, in press. [Google Scholar] [CrossRef]
- Hose, G.C. Assessing the Need for Groundwater Quality Guidelines for Pesticides Using the Species Sensitivity Distribution Approach. Hum. Ecol. Risk Assess. 2005, 11, 951–966. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Di Marzio, W.D.; Fiasca, B.; Galassi, D.M.P.; Korbel, K.; Iepure, S.; Pereira, J.L.; Reboleira, A.S.P.S.; Schmidt, S.I.; Hose, G.C. Recommendations for ecotoxicity testing with stygobiotic species in the framework of groundwater environmental risk assessment. Sci. Total Environ. 2019, 681, 292–304. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Di Marzio, W.D.; Spigoli, D.; Baratti, M.; Messana, G.; Cannicci, S.; Galassi, D.M.P. Metabolic rates of a hypogean and an epigean species of copepod in an alluvial aquifer. Freshw. Biol. 2015, 60, 426–435. [Google Scholar] [CrossRef]
- Little, J.; Schmidt, D.J.; Cook, B.D.; Page, T.J.; Hughes, J.M. Diversity and phylogeny of south-east Queensland Bathynellacea. J. Aust. J. Zool. 2016, 64, 36–47. [Google Scholar] [CrossRef]
- Perina, G.; Camacho, A.I.; Huey, J.; Horwitz, P.; Koenders, A. New Bathynellidae (Crustacea) taxa and their relationships in the Fortescue catchment aquifers of the Pilbara region, Western Australia. Syst. Biodivers. 2019, 17, 148–164. [Google Scholar] [CrossRef]
- Camacho, A.I.; Mas-Peinado, P.; Dorda, B.A.; Casado, A.; Brancelj, A.; Knight, L.R.F.D.; Hutchins, B.; Bou, C.; Perina, G.; Rey, I. Molecular tools unveil an underestimated diversity in a stygofauna family: A preliminary world phylogeny and an updated morphology of Bathynellidae (Crustacea: Bathynellacea). Zool. J. Linn. Soc. 2017, 183, 70–96. [Google Scholar] [CrossRef]
- Korbel, K.L.; Hose, G.C. Habitat, water quality, seasonality, or site? Identifying environmental correlates of the distribution of groundwater biota. Freshw. Sci. 2015, 34, 329–343. [Google Scholar] [CrossRef]
- Hose, G.C.; Symington, K.; Lott, M.J.; Lategan, M.J. The toxicity of arsenic(III), chromium(VI) and zinc to groundwater copepods. Environ. Sci. Pollut. Res. 2016, 23, 18704–18713. [Google Scholar] [CrossRef] [PubMed]
- Fruchter, J. Peer Reviewed: In-Situ Treatment of Chromium-Contaminated Groundwater. Environ. Sci. Technol. 2002, 36, 464A–472A. [Google Scholar] [CrossRef] [PubMed]
- Scheeren, P.J.H.; Koch, R.O.; Buisman, C.J.N.; Barnes, L.J.; Versteegh, J.H. New biological treatment plant for heavy metal contaminated groundwater. In EMC ’91: Non-Ferrous Metallurgy—Present and Future; Springer: Dordrecht, The Netherlands, 1991; pp. 403–416. [Google Scholar]
- Smedley, P.L.; Kinniburgh, D.G. A review of the source, behaviour and distribution of arsenic in natural waters. Appl. Geochem. 2002, 17, 517–568. [Google Scholar] [CrossRef]
- Edmunds, W.M.; Shand, P. Groundwater Baseline Quality. In Natural Groundwater Quality; Edmunds, W.M., Shand, P., Eds.; Blackwell Publishing: Oxford, UK, 2009; pp. 1–21. [Google Scholar]
- Klockenkämper, R.; von Bohlen, A. Elemental Analysis of Environmental Samples by Total Reflection X-Ray Fluorescence: A Review. X-ray Spectrom. 1996, 25, 156–162. [Google Scholar] [CrossRef]
- Farías, S.S.; Casa, V.A.; Vázquez, C.; Ferpozzi, L.; Pucci, G.N.; Cohen, I.M. Natural contamination with arsenic and other trace elements in ground waters of Argentine Pampean Plain. Sci. Total Environ. 2003, 309, 187–199. [Google Scholar] [CrossRef]
- Siegele, R.; Cohen, D.D.; Dytlewski, N. The ANSTO high energy heavy ion microprobe. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms 1999, 158, 31–38. [Google Scholar] [CrossRef]
- Ryan, C.G. Developments in Dynamic Analysis for quantitative PIXE true elemental imaging. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms 2001, 181, 170–179. [Google Scholar] [CrossRef]
- Ryan, C.G.; Jamieson, D.N.; Churms, C.L.; Pilcher, J.V. A new method for on-line true-elemental imaging using PIXE and the proton microprobe. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms 1995, 104, 157–165. [Google Scholar] [CrossRef]
- Ritz, C.; Streibig, J.C. Bioassay analysis using R. J. Stat. Softw. 2005, 12, 1–22. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Hyne, R.V.; Pablo, F.; Julli, M.; Markich, S.J. Influence of water chemistry on the acute toxicity of copper and zinc to the cladoceran Ceriodaphnia cf dubia. Environ. Toxicol. Chem. 2005, 24, 1667–1675. [Google Scholar] [CrossRef] [PubMed]
- Hahn, H.J. The GW-Fauna-Index: A first approach to a quantitative ecological assessment of groundwater habitats. Limnol. Ecol. Manag. Inland Waters 2006, 36, 119–137. [Google Scholar] [CrossRef]
- Notenboom, J.; Cruys, K.; Hoekstra, J.; Vanbeelen, P. Effect of Ambient Oxygen Concentration Upon the Acute Toxicity of Chlorophenols and Heavy-Metals to the Groundwater Copepod Parastenocaris-Germanica (Crustacea). Ecotoxicol. Environ. Saf. 1992, 24, 131–143. [Google Scholar] [CrossRef]
- Lima, A.; Curtis, C.; Hammermeister, D.; Markee, T.; Northcott, C.E.; Brooke, L.T. Acute and chronic toxicities of arsenic(III) to fathead minnows, flagfish, daphnids, and an amphipod. Arch. Environ. Contam. Toxicol. 1984, 13, 595–601. [Google Scholar] [CrossRef]
- Canivet, V.; Chambon, P.; Gibert, J. Toxicity and bioaccumulation of arsenic and chromium in epigean and hypogean freshwater macroinvertebrates. Arch. Environ. Contam. Toxicol. 2001, 40, 345–354. [Google Scholar] [CrossRef]
- Di Marzio, W.D.; Castaldo, D.; Pantani, C.; Di Cioccio, A.; Di Lorenzo, T.; Saenz, M.E.; Galassi, D.M.P. Relative Sensitivity of Hyporheic Copepods to Chemicals. Bull. Environ. Contam. Toxicol. 2009, 82, 488–491. [Google Scholar] [CrossRef]
- Reboleira, A.S.; Abrantes, N.; Oromí, P.; Gonçalves, F. Acute Toxicity of Copper Sulfate and Potassium Dichromate on Stygobiont Proasellus: General Aspects of Groundwater Ecotoxicology and Future Perspectives. Water Air Soil Pollut. 2013, 224, 1–9. [Google Scholar] [CrossRef]
- Fargasova, A. Toxicity of Metals on Daphnia magna and Tubifex tubifex. Ecotoxicol. Environ. Saf. 1994, 27, 210–213. [Google Scholar] [CrossRef]
- Ewell, W.S.; Gorsuch, J.W.; Kringle, R.O.; Robillard, K.A.; Spiegel, R.C. Simultaneous evaluation of the acute effects of chemicals on seven aquatic species. Environ. Toxicol. Chem. 1986, 5, 831–840. [Google Scholar] [CrossRef]
- Meinel, W.; Krause, R. Zur Korrelation zwischen Zink und verschiedenen pH–Werten in in her toxischen Wirkung auf einige Grundwasser–Organismen. Z. Für Angew. Zool. 1988, 75, 159–182. [Google Scholar]
- Coppellotti Krupa, O.; Toniello, V.; Guidolin, L. Niphargus and Gammarus from karst waters: First data on heavy metal (Cd, Cu, Zn) exposure in a biospeleology laboratory. Subterr. Biol. 2004, 2, 33–41. [Google Scholar]
- Lazorchak, J.M.; Smith, M.E.; Haring, H.J. Development and validation of a Daphnia magna four-day survival and growth test method. Environ. Toxicol. Chem. 2009, 28, 1028–1034. [Google Scholar] [CrossRef] [PubMed]
- Daam, M.A.; Silva, E.; Leitao, S.; Trindade, M.J.; Cerejeira, M.J. Does the actual standard of 0.1 mu g/L overestimate or underestimate the risk of plant protection products to groundwater ecosystems? Ecotoxicol. Environ. Saf. 2010, 73, 750–756. [Google Scholar] [CrossRef] [PubMed]
- Paulauskis, J.D.; Winner, R.W. Effects of water hardness and humic acid on zinc toxicity to Daphnia magna Straus. Aquat. Toxicol. 1988, 12, 273–290. [Google Scholar] [CrossRef]
- Pawlisz, A.V.; Kent, R.A.; Schneider, U.A.; Jefferson, C. Canadian water quality guidelines for chromium. Environ. Toxicol. Water Qual. 1997, 12, 123–183. [Google Scholar] [CrossRef]
- Park, E.J.; Jo, H.J.; Jung, J. Combined effects of pH, hardness and dissolved organic carbon on acute metal toxicity to Daphnia magna. J. Ind. Eng. Chem. 2009, 15, 82–85. [Google Scholar] [CrossRef]
- Warne, M.; Batley, G.; van Dam, R.; Chapman, J.; Fox, D.; Hickey, C.; Stauber, J. Revised Method for Deriving Australian and New Zealand Water Quality Guideline Values for Toxicants—Update of 2015 Version; Australian and New Zealand Governments and Australian State and Territory Governments: Canberra, Australia, 2018; p. 48.
- Sprague, J.B. Measurement of pollutant toxicity to fish I. Bioassay methods for acute toxicity. Water Res. 1969, 3, 793–821. [Google Scholar] [CrossRef]
- Avramov, M.; Schmidt, S.I.; Griebler, C. A new bioassay for the ecotoxicological testing of VOCs on groundwater invertebrates and the effects of toluene on Niphargus inopinatus. Aquat. Toxicol. 2013, 130, 1–8. [Google Scholar] [CrossRef]
- Williams, G.; West, J.M.; Snow, E.T. Total arsenic accumulation in yabbies (Cherax destructor clark) exposed to elevated arsenic levels in Victorian gold mining areas, Australia. Environ. Toxicol. Chem. 2008, 27, 1332–1342. [Google Scholar] [CrossRef]
- White, S.L.; Rainbow, P.S. Regulation of zinc concentration by Palaemon elegans (Crustacea: Decapoda): Zinc flux and effects of temperature, zinc concentration and moulting. Mar. Ecol. Prog. Ser. 1984, 16, 135–147. [Google Scholar] [CrossRef]
- Cresswell, T.; Simpson, S.L.; Mazumder, D.; Callaghan, P.D.; Nguyen, A.P. Bioaccumulation Kinetics and Organ Distribution of Cadmium and Zinc in the Freshwater Decapod Crustacean Macrobrachium australiense. Environ. Sci. Technol. 2015, 49, 1182–1189. [Google Scholar] [CrossRef] [PubMed]
- Steenken, B.; Schminke, H.K. Ultrastructure of maxillary gland of Antrobathynella stammeri (Syncarida, Malacostraca). J. Morphol. 1996, 228, 107–117. [Google Scholar] [CrossRef]
- Smith, G. On the Anaspidacea, Living and Fossil. J. Cell Sci. 1909, s2-53, 489–578. [Google Scholar]
- Fox, H.M. Anal and Oral Intake of Water by Crustacea. J. Exp. Biol. 1952, 29, 583. [Google Scholar] [CrossRef]
- White, S.L.; Rainbow, P.S. Zinc flux in Palaemon elegans (Crustacea: Decapoda): Moulting, individual variation and tissue distribution. Mar. Ecol. Prog. Ser. 1984, 19, 153–166. [Google Scholar] [CrossRef]
- Smith, K.S.; Huyck, H. An overview of the abundance, relative mobility, bioavailabilty, and human toxicity of metals. In The Environmental Geochemistry of Mineral Deposits: Part, A; Processes, Techniques, and Health Issues; Plumlee, G., Logsdon, J., Eds.; Society for Economic Geologists: Littleton, CO, USA, 1999; Volume 6A, pp. 29–70. [Google Scholar]
- Spehar, R.L.; Fiandt, J.T.; Anderson, R.L.; DeFoe, D. Comparative toxicity of arsenic compounds and their accumulation in invertebrates and fish. Arch. Environ. Contam. Toxicol. 1980, 9, 53–63. [Google Scholar] [CrossRef]
- Winner, R.W.; Gauss, J.D. Relationship between chronic toxicity and bioaccumulation of copper, cadmium and zinc as affected by water hardness and humic acid. Aquat. Toxicol. 1986, 8, 149–161. [Google Scholar] [CrossRef]
- Markich, S.; Brown, P.; Batley, G.; Apte, S.; Stauber, J. Incorporating metal speciation and bioavailability into water quality guidelines for protecting aquatic ecosystems. Aust. J. Ecotoxicol. 2001, 7, 109–122. [Google Scholar]
- Rainbow, P.S.; Luoma, S.N. Metal toxicity, uptake and bioaccumulation in aquatic invertebrates—Modelling zinc in crustaceans. Aquat. Toxicol. 2011, 105, 455–465. [Google Scholar] [CrossRef]
- Rainbow, P.S. Trace metal bioaccumulation: Models, metabolic availability and toxicity. Environ. Int. 2007, 33, 576–582. [Google Scholar] [CrossRef]
- Vijayram, K.; Geraldine, P. Toxicology. Regulation of Essential Heavy Metals (Cu, Cr, and Zn) by the Freshwater Prawn Macrobrachium malcolmsonii (Milne Edwards). Bull. Environ. Contam. Toxicol. 1996, 56, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Koenemann, S.; Jenner, R.A.; Hoenemann, M.; Stemme, T.; von Reumont, B.M. Arthropod phylogeny revisited, with a focus on crustacean relationships. Arthropod Struct. Dev. 2010, 39, 88–110. [Google Scholar] [CrossRef] [PubMed]
- Luoma, S.N.; Rainbow, P.S. Metal. Contamination in Aquatic Environments: Science and Lateral Management; Cambridge University Press: Cambridge, UK, 2008. [Google Scholar] [CrossRef]
- Kuhn, A.; Munns, W.R., Jr.; Serbst, J.; Edwards, P.; Cantwell, M.G.; Gleason, T.; Pelletier, M.C.; Berry, W. Evaluating the ecological significance of laboratory response data to predict population-level effects for the estruarine amphipod Ampelisca abdita. Environ. Toxicol. Chem. 2002, 21, 865–874. [Google Scholar] [CrossRef] [PubMed]
- Maltby, L.; Naylor, C. Preliminary Observations on the Ecological Relevance of the Gammarus ‘Scope for Growth’ Assay: Effect of Zinc on Reproduction. Funct. Ecol. 1990, 4, 393–397. [Google Scholar] [CrossRef]
- Kammenga, J.E.; Van Gestel, C.A.M.; Bakker, J. Toxicology. Patterns of sensitivity to cadmium and pentachlorophenol among nematode species from different taxonomic and ecological groups. Arch. Environ. Contam. Toxicol. 1994, 27, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Wellborn, G.A.; Witt, J.D.S.; Cothran, R.D. Chapter 31—Class Malacostraca, Superorders Peracarida and Syncarida. In Thorp and Covich’s Freshwater Invertebrates, 4th ed.; Thorp, J.H., Rogers, D.C., Eds.; Academic Press: Cambridge, MA, USA, 2015; pp. 781–796. [Google Scholar] [CrossRef]
- Lategan, M.J.; Klare, W.; Kidd, S.; Hose, G.C.; Nevalainen, H. The unicellular fungal tool RhoTox for risk assessments in groundwater systems. Ecotoxicol. Environ. Saf. 2016, 132, 18–25. [Google Scholar] [CrossRef]
- Hose, G.C.; Stumpp, C. Architects of the underworld: Bioturbation by groundwater invertebrates influences aquifer hydraulic properties. Aquat. Sci. 2019, 81, 20. [Google Scholar] [CrossRef]
| Water Quality Variable | Units | Concentration |
|---|---|---|
| pH | 4.2–5.6 | |
| Conductivity | µS/cm | 131–195 |
| Dissolved Oxygen | % Saturation | 59–83 |
| Hardness | mg/L as CaCO3 | 25–44 |
| Total Organic Carbon | mg/L | 3–13 |
| As | mg/L | <0.01 |
| Cr | mg/L | 0.03 |
| Zn | mg/L | 0.02 |
| As(III) | Cr(VI) | Zn(II) | |||||
|---|---|---|---|---|---|---|---|
| Test Duration | ECx | Estimate | 95% Confidence Interval | Estimate | 95% Confidence Interval | Estimate | 95% Confidence Interval |
| N = 74 | N = 71 | N = 190 | |||||
| 96 h | 10 | 0.17 | (−0.04–0.38) | 0.11 | (−0.07–0.28) | 0.78 | (0.30–1.26) |
| 50 | 1.99 | (0.79–3.19) | 1.83 | (−0.10–3.77) | 3.61 | (2.56–4.66) | |
| 14 days | 10 | 0.01 | (−0.02–0.04) | 0.004 | (−0.010–0.018) | 0.21 | (0.02–0.39) |
| 50 | 0.25 | (0.07–0.44) | 0.51 | (−0.07–1.09) | 1.77 | (1.11–2.43) | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hose, G.C.; Symington, K.; Lategan, M.J.; Siegele, R. The Toxicity and Uptake of As, Cr and Zn in a Stygobitic Syncarid (Syncarida: Bathynellidae). Water 2019, 11, 2508. https://doi.org/10.3390/w11122508
Hose GC, Symington K, Lategan MJ, Siegele R. The Toxicity and Uptake of As, Cr and Zn in a Stygobitic Syncarid (Syncarida: Bathynellidae). Water. 2019; 11(12):2508. https://doi.org/10.3390/w11122508
Chicago/Turabian StyleHose, Grant C., Katelyn Symington, Maria J. Lategan, and Rainer Siegele. 2019. "The Toxicity and Uptake of As, Cr and Zn in a Stygobitic Syncarid (Syncarida: Bathynellidae)" Water 11, no. 12: 2508. https://doi.org/10.3390/w11122508
APA StyleHose, G. C., Symington, K., Lategan, M. J., & Siegele, R. (2019). The Toxicity and Uptake of As, Cr and Zn in a Stygobitic Syncarid (Syncarida: Bathynellidae). Water, 11(12), 2508. https://doi.org/10.3390/w11122508






