Transport of Microplastic Particles in Saturated Porous Media
Abstract
1. Introduction
2. Materials and Methods
2.1. MPs and Porous Media
2.2. Column Transport Experiments
2.3. Column Dissection Experiments
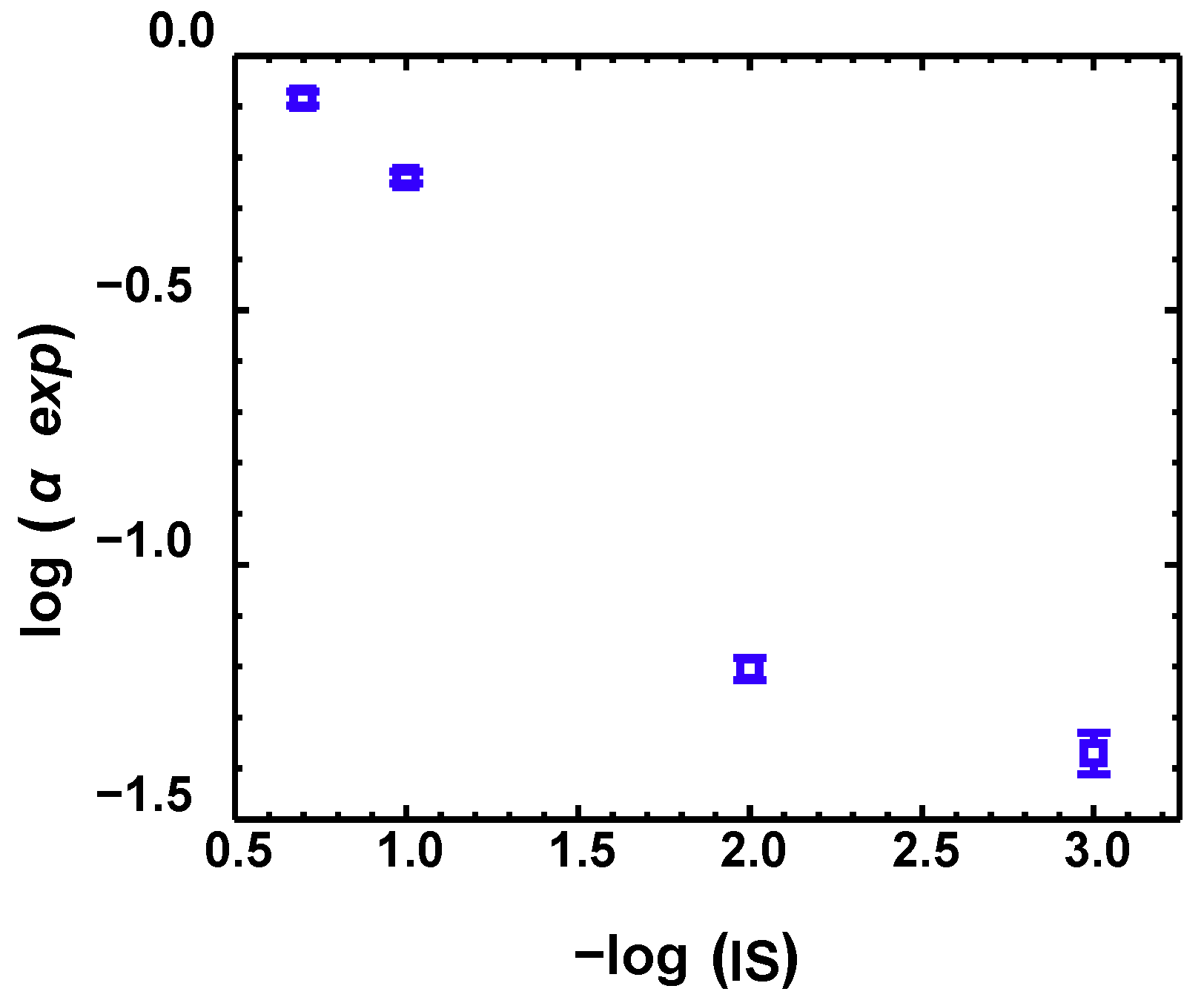
2.4. Calculation of Attachment Efficiency
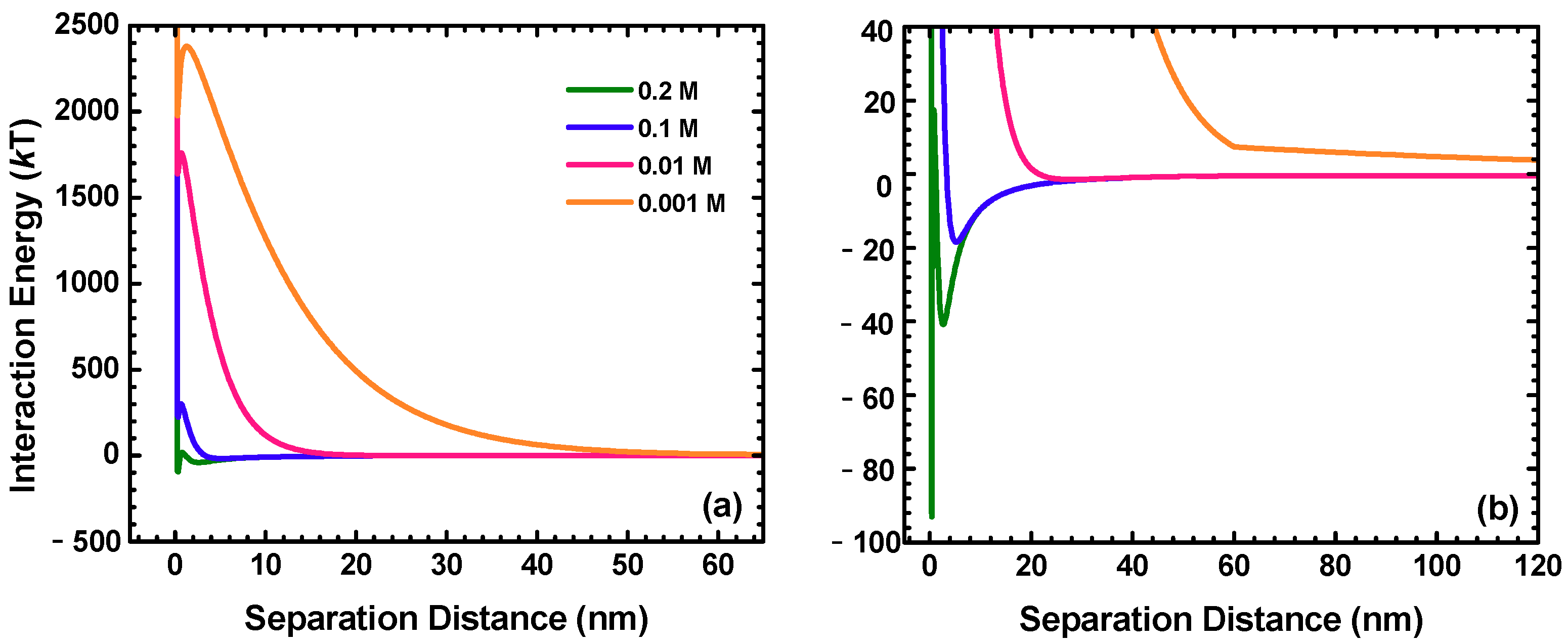
2.5. DLVO Interaction Energy Calculations
2.6. Transport Model
3. Results and Discussion
3.1. Electrokinetic Properties of MPs and Collectors
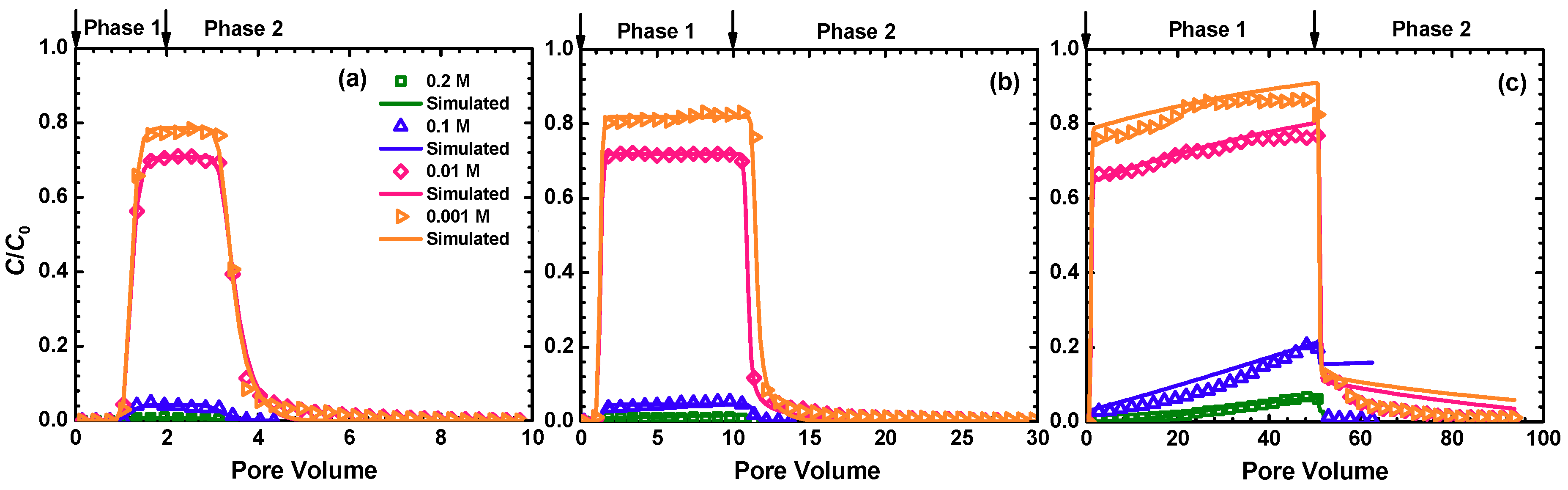
3.2. BTCs of MPs
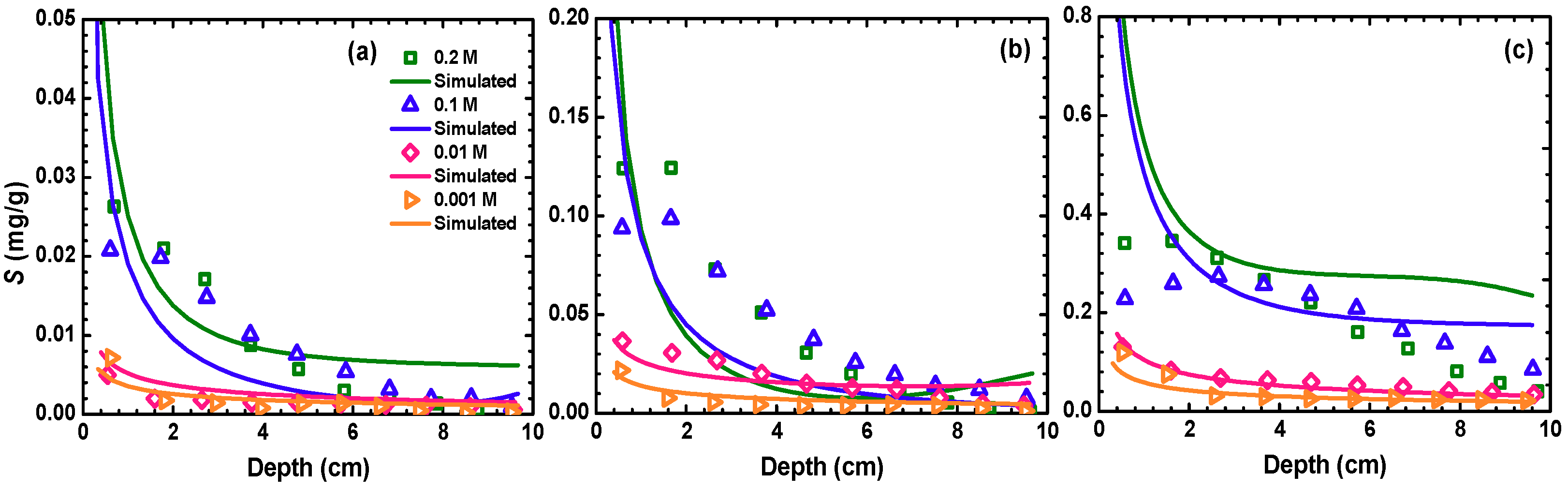
3.3. Profiles of Retained Particles
3.4. Modeling BTCs and RPs
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- John, G.; Nagarajan, S.; Vemula, P.K.; Silverman, J.R.; Pillai, C.K.S. Natural Monomers: A mine for functional and sustainable materials-occurrence, chemical modification and polymerization. Prog. Polym. Sci. 2019, 92, 158–209. [Google Scholar] [CrossRef]
- Alimi, O.S.; Budarz, J.F.; Hernandez, L.M.; Tufenkji, N. Microplastics and nanoplastics in aquatic environments: Aggregation, deposition, and enhanced contaminant transport. Environ. Sci. Technol. 2018, 5252, 1704–1724. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, L.M.; Xu, E.G.; Larsson, H.C.; Tahara, R.; Maisuria, V.B.; Tufenkji, N. Plastic teabags release billions of microparticles and nanoparticles into tea. Environ. Sci. Technol. 2019. [Google Scholar] [CrossRef] [PubMed]
- McCormick, A.; Hoellein, T.J.; Mason, S.A.; Schluep, J.; Kelly, J.J. Microplastic is an abundant and distinct microbial habitat in an urban river. Environ. Sci. Technol. 2014, 4848, 11863–11871. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.C.; Olsen, Y.; Mitchell, R.P.; Davis, A.; Rowland, S.J.; John, A.W.; Russell, A.E. Lost at sea: Where is all the plastic? Science 2004, 304, 838. [Google Scholar] [CrossRef] [PubMed]
- Rillig, M.C. Microplastic in terrestrial ecosystems and the soil? Environ. Sci. Technol. 2012, 4646, 6453–6454. [Google Scholar] [CrossRef]
- Rillig, M.C.; Ingraffia, R.; de Souza Machado, A.A. Microplastic incorporation into soil in agroecosystems. Front. Plant Sci. 2017, 8, 1805. [Google Scholar] [CrossRef]
- Yao, K.M.; Habibian, M.T.; O’Melia, C.R. Water and waste water filtration. Concepts and applications. Environ. Sci. Technol. 1971, 55, 1105–1112. [Google Scholar] [CrossRef]
- Bradford, S.A.; Yates, S.R.; Bettahar, M.; Simunek, J. Physical factors affecting the transport and fate of colloids in saturated porous media. Water Resour. Res. 2002, 3838. [Google Scholar] [CrossRef]
- Shen, C.; Jin, Y.; Zhuang, J.; Li, T.; Xing, B. Role and importance of surface heterogeneities in transport of particles in saturated porous media. Crit. Rev. Environ. Sci. Technol. 2019. [Google Scholar] [CrossRef]
- Ryan, J.N.; Elimelech, M. Colloid mobilization and transport in groundwater. Colloid Surf. A 1996, 107, 1–56. [Google Scholar] [CrossRef]
- Ng, W.K.; Jegatheesan, V.; Lee, S.H. Influence of particle concentration on initial collection efficiency and surface coverage in porous media filtration. Korean J. Chem. Eng. 2006, 2323, 333–342. [Google Scholar] [CrossRef]
- Molnar, I.L.; Johnson, W.P.; Gerhard, J.I.; Willson, C.S.; O’Carroll, D.M. Predicting colloid transport through saturated porous media: A critical review. Water Resour. Res. 2015, 5151, 6804–6845. [Google Scholar] [CrossRef]
- Tufenkji, N.; Redman, J.A.; Elimelech, M. Interpreting deposition patterns of microbial particles in laboratory-scale column experiments. Environ. Sci. Technol. 2003, 3737, 616–623. [Google Scholar] [CrossRef]
- Tufenkji, N.; Elimelech, M. Correlation equation for predicting single-collector efficiency in physicochemical filtration in saturated porous media. Environ. Sci. Technol. 2004, 3838, 529–536. [Google Scholar] [CrossRef]
- Tufenkji, N.; Elimelech, M. Breakdown of colloid filtration theory: Role of the secondary energy minimum and surface charge heterogeneities. Langmuir 2005, 21, 841–852. [Google Scholar] [CrossRef]
- Hahn, M.W.; O’Melia, C.R. Deposition and reentrainment of Brownian particles in porous media under unfavorable chemical conditions: Some concepts and applications. Environ. Sci. Technol. 2004, 38, 210–220. [Google Scholar] [CrossRef]
- Li, X.; Scheibe, T.D.; Johnson, W.P. Apparent decreases in colloid deposition rate coefficients with distance of transport under unfavorable deposition conditions: A general phenomenon. Environ. Sci. Technol. 2004, 38, 5616–5625. [Google Scholar] [CrossRef]
- Shen, C.; Li, B.; Huang, Y.; Jin, Y. Kinetics of coupled primary-and secondary-minimum deposition of colloids under unfavorable chemical conditions. Environ. Sci. Technol. 2007, 41, 6976–6982. [Google Scholar] [CrossRef]
- Kasel, D.; Bradford, S.A.; Simunek, J.; Heggen, M.; Vereecken, H.; Klumpp, E. Transport and retention of multi-walled carbon nanotubes in saturated porous media: Effects of input concentration and grain size. Water Res. 2013, 47, 933–944. [Google Scholar] [CrossRef]
- Liang, Y.; Bradford, S.A.; Simunek, J.; Vereecken, H.; Klumpp, E. Sensitivity of the transport and retention of stabilized silver nanoparticles to physicochemical factors. Water Res. 2013, 47, 2572–2582. [Google Scholar] [CrossRef]
- Wang, D.; Ge, L.; He, J.; Zhang, W.; Jaisi, D.P.; Zhou, D. Hyperexponential and non-monotonic retention of polyvinylpyrrolidone-coated silver nanoparticles in an Ultisol. J. Contam. Hydrol. 2014, 164, 35–48. [Google Scholar] [CrossRef]
- Chequer, L.; Russell, T.; Behr, A.; Genolet, L.; Kowollik, P.; Badalyan, A.; Bedrikovetsky, P. Non-monotonic permeability variation during colloidal transport: Governing equations and analytical model. J. Hydrol. 2018, 557, 547–560. [Google Scholar] [CrossRef]
- Hilpert, M.; Johnson, W.P. A binomial modeling approach for upscaling colloid transport under unfavorable attachment conditions: Emergent prediction of non-monotonic retention profiles. Water Resour. Res. 2018, 54, 46–60. [Google Scholar] [CrossRef]
- He, J.; Wang, D.; Zhou, D. Transport and retention of silver nanoparticles in soil: Effects of input concentration, particle size and surface coating. Sci. Total Environ. 2019, 648, 102–108. [Google Scholar] [CrossRef]
- Bradford, S.A.; Bettahar, M. Straining, attachment, and detachment of Cryptosporidium oocysts in saturated porous media. J. Environ. Qual. 2005, 34, 469–478. [Google Scholar] [CrossRef]
- Tong, M.; Johnson, W.P. Colloid population heterogeneity drives hyperexponential deviation from classic filtration theory. Environ. Sci. Technol. 2007, 41, 493–499. [Google Scholar] [CrossRef]
- Schijven, J.F.; Hoogenboezem, W.; Hassanizadeh, M.; Peters, J.H. Modeling removal of bacteriophages MS2 and PRD1 by dune recharge at Castricum, Netherlands. Water Resour. Res. 1999, 35, 1101–1111. [Google Scholar] [CrossRef]
- Harter, T.; Wagner, S.; Atwill, E.R. Colloid transport and filtration of Cryptosporidium parvum in sandy soils and aquifer sediments. Environ. Sci. Technol. 2000, 34, 62–70. [Google Scholar] [CrossRef]
- Redman, J.A.; Estes, M.K.; Grant, S.B. Resolving macroscale and microscale heterogeneity in virus filtration. Colloid Surf. A 2001, 191, 57–70. [Google Scholar] [CrossRef]
- Bradford, S.A.; Simunek, J.; Bettahar, M.; van Genuchten, M.T.; Yates, S.R. Modeling colloid attachment, straining, and exclusion in saturated porous media. Environ. Sci. Technol. 2003, 37, 2242–2250. [Google Scholar] [CrossRef] [PubMed]
- Bradford, S.A.; Bettahar, M.; Simunek, J.; van Genuchten, M.T. Straining and attachment of colloids in physically heterogeneous porous media. Vadose Zone J. 2004, 3, 384–394. [Google Scholar] [CrossRef]
- Li, X.; Johnson, W.P. Non-monotonic variations in deposition rate coefficients of microspheres in porous media under unfavorable deposition conditions. Environ. Sci. Technol. 2005, 39, 1658–1665. [Google Scholar] [CrossRef] [PubMed]
- Tong, M.; Li, X.; Brow, C.N.; Johnson, W.P. Detachment-influenced transport of an adhesion-deficient bacterial strain within water-reactive porous media. Environ. Sci. Technol. 2005, 39, 2500–2508. [Google Scholar] [CrossRef]
- Li, Y.; Wang, Y.; Pennell, K.D.; Abriola, L.M. Investigation of the transport and deposition of fullerene (C60) nanoparticles in quartz sands under varying flow conditions. Environ. Sci. Technol. 2008, 42, 7174–7180. [Google Scholar] [CrossRef]
- Liu, Y.; Janjaroen, D.; Kuhlenschmidt, M.S.; Kuhlenschmidt, T.B.; Nguyen, T.H. Deposition of Cryptosporidium parvum oocysts on natural organic matter surfaces: Microscopic evidence for secondary minimum deposition in a radial stagnation point flow cell. Langmuir 2009, 25, 1594–1605. [Google Scholar] [CrossRef]
- Li, X.; Zhang, P.; Lin, C.L.; Johnson, W.P. Role of hydrodynamic drag on microsphere deposition and re-entrainment in porous media under unfavorable conditions. Environ. Sci. Technol. 2005, 39, 4012–4020. [Google Scholar] [CrossRef]
- Rasmuson, A.; Pazmino, E.; Assemi, S.; Johnson, W.P. Contribution of nano-to microscale roughness to heterogeneity: Closing the gap between unfavorable and favorable colloid attachment conditions. Environ. Sci. Technol. 2017, 51, 2151–2160. [Google Scholar] [CrossRef]
- Johnson, W.P.; Rasmuson, A.; Pazmiño, E.; Hilpert, M. Why variant colloid transport behaviors emerge among identical individuals in porous media when colloid–surface repulsion exists. Environ. Sci. Technol. 2018, 52, 7230–7239. [Google Scholar] [CrossRef]
- Elimelech, M.; O’Melia, C.R. Kinetics of deposition of colloidal particles in porous media. Environ. Sci. Technol. 1990, 24, 1528–1536. [Google Scholar] [CrossRef]
- Tufenkji, N.; Elimelech, M. Deviation from the classical colloid filtration theory in the presence of repulsive DLVO interactions. Langmuir 2004, 20, 10818–10828. [Google Scholar] [CrossRef] [PubMed]
- Redman, J.A.; Walker, S.L.; Elimelech, M. Bacterial adhesion and transport in porous media: Role of the secondary energy minimum. Environ. Sci. Technol. 2004, 38, 1777–1785. [Google Scholar] [CrossRef] [PubMed]
- Bradford, S.A.; Simunek, J.; Walker, S.L. Transport and straining of E. coli O157: H7 in saturated porous media. Water Resour. Res. 2006, 42. [Google Scholar] [CrossRef]
- Hoek, E.M.; Agarwal, G.K. Extended DLVO interactions between spherical particles and rough surfaces. J. Colloid Interface Sci. 2006, 298, 50–58. [Google Scholar] [CrossRef]
- Shen, C.; Zhang, M.; Zhang, S.; Wang, Z.; Zhang, H.; Li, B.; Huang, Y. Influence of surface heterogeneities on reversibility of fullerene (nC60) nanoparticle attachment in saturated porous media. J. Hazard. Mater. 2015, 290, 60–68. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, D.; Li, B.; Wang, J.; Li, T.; Zhang, M.; Shen, C. Detachment of fullerene nC60 nanoparticles in saturated porous media under flow/stop-flow conditions: Column experiments and mechanistic explanations. Environ. Pollut. 2016, 213, 698–709. [Google Scholar] [CrossRef]
- Li, T.; Jin, Y.; Huang, Y.; Li, B.; Shen, C. Observed dependence of colloid detachment on the concentration of initially attached colloids and collector surface heterogeneity in porous media. Environ. Sci. Technol. 2017, 51, 2811–2820. [Google Scholar] [CrossRef]
- Gregory, J. Approximate expressions for retarded van der Waals interaction. J. Colloid Interface Sci. 1981, 83, 138–145. [Google Scholar] [CrossRef]
- Ruckenstein, E.; Prieve, D.C. Adsorption and desorption of particles and their chromatographic separation. Aiche J. 1976, 22, 276–283. [Google Scholar] [CrossRef]
- Hogg, R.; Healy, T.W.; Fuerstenau, D.W. Mutual coagulation of colloidal dispersions. Trans. Faraday Soc. 1966, 62, 1638–1651. [Google Scholar] [CrossRef]
- Elimelech, M.; O’Melia, C.R. Effect of particle size on collision efficiency in the deposition of Brownian particles with electrostatic energy barriers. Langmuir 1990, 6, 1153–1163. [Google Scholar] [CrossRef]
- Shen, C.; Bradford, S.A.; Li, T.; Huang, Y. Can nanoscale surface charge heterogeneity really explain colloid detachment from primary minima upon reduction of solution ionic strength? J. Nanopart. Res. 2018, 20, 165. [Google Scholar] [CrossRef]
- Adamczyk, Z.; Siwek, B.; Zembala, M.; Belouschek, P. Kinetics of localized adsorption of colloid particles. Adv. Colloid Interface Sci. 1994, 48, 151–280. [Google Scholar] [CrossRef]
- Haque, M.E.; Shen, C.; Li, T.; Chu, H.; Wang, H.; Li, Z.; Huang, Y. Influence of biochar on deposition and release of clay colloids in saturated porous media. J. Environ. Qual. 2017, 46, 1480–1488. [Google Scholar] [CrossRef]
- Torkzaban, S.; Tazehkand, S.S.; Walker, S.L.; Bradford, S.A. Transport and fate of bacteria in porous media: Coupled effects of chemical conditions and pore space geometry. Water Resour. Res. 2008, 44, W04403. [Google Scholar] [CrossRef]
- Bradford, S.A.; Torkzaban, S.; Kim, H.; Simunek, J. Modeling colloid and microorganism transport and release with transients in solution ionic strength. Water Resour. Res. 2012, 48. [Google Scholar] [CrossRef]
- Wu, J.; Shen, C.; Wang, C.; Yan, A.; Zhang, H. The failure of using equilibrium adsorption of fosthiazate onto montmorillonite clay particles to predict their cotransport in porous media as revealed by batch and column studies. J. Soil Sediment. 2019, 19, 917–928. [Google Scholar] [CrossRef]
- Zhang, P.; Simunek, J.; Bowman, R.S. Nonideal transport of solute and colloidal tracers through reactive zeolite/iron pellets. Water Resour. Res. 2004, 40, W04207. [Google Scholar] [CrossRef]
- Simunek, J.; van Genuchten, M.T.; Sejna, M. The Hydrus-1D software package for simulating the one-dimentional movement of water. In Heat, and Multiple Solutes in Variably-Saturated Media; Version 3.0; Department of Environmental Science, University of California Riverside: Riverside, CA, USA, 2005. [Google Scholar]
- Shen, C.; Lazouskaya, V.; Zhang, H.; Li, B.; Jin, Y.; Huang, Y. Influence of surface chemical heterogeneity on attachment and detachment of microparticles. Colloid Surf. A 2013, 433, 14–29. [Google Scholar] [CrossRef]
- Zhang, C.; Yan, A.; Wang, G.; Jin, C.; Chen, Y.; Shen, C. Impact of flow velocity on transport of graphene oxide nanoparticles in saturated porous media. Vadose Zone J. 2018, 17, 180019. [Google Scholar] [CrossRef]
- Zhao, K.; Chen, C.; Cheng, T.; Shang, J. Graphene oxide-facilitated uranium transport and release in saturated medium: Effect of ionic strength and medium structure. Environ. Pollut. 2019, 247, 668–677. [Google Scholar] [CrossRef]
- Hahn, M.W.; Abadzic, D.; O’Melia, C.R. Aquasols: on the role of secondary minima. Environ. Sci. Technol. 2004, 38, 5915–5924. [Google Scholar] [CrossRef]
- Franchi, A.; O’Melia, C.R. Effects of natural organic matter and solution chemistry on the deposition and reentrainment of colloids in porous media. Environ. Sci. Technol. 2003, 37, 1122–1129. [Google Scholar] [CrossRef]
- Walker, S.L.; Redman, J.A.; Elimelech, M. Role of cell surface lipopolysaccharides in Escherichia coli k12 adhesion and transport. Langmuir 2004, 20, 7736–7746. [Google Scholar] [CrossRef]
- Liang, Y.; Bradford, S.A.; Simunek, J.; Klumpp, E. Mechanisms of graphene oxide aggregation, retention, and release in quartz sand. Sci. Total Environ. 2019, 656, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Babakhani, P.; Bridge, J.; Doong, R.; Phenrat, T. Continuum-based models and concepts for the transport of nanoparticles in saturated porous media: A state-of-the-science review. Adv. Colloid Interface Sci. 2017, 246, 75–104. [Google Scholar] [CrossRef] [PubMed]
| IS (M) | Zeta Potential (mV) | MP Size (nm) | |
|---|---|---|---|
| Glass Beads | MPs | ||
| 0.2 | −26.1 | −34.1 | 997.2 |
| 0.1 | −31.7 | −43.9 | 982.4 |
| 0.01 | −50.4 | −67.3 | 943.1 |
| 0.001 | −53.6 | −75.7 | 910.4 |
| IS (M) | Umax (kT) | ΔU (kT) | −Usec | |
|---|---|---|---|---|
| Depth (kT) | Distance (nm) | |||
| 0.2 | 17.3 | 110.4 | 40.8 | 2.7 |
| 0.1 | 301.6 | 77.9 | 18.4 | 5.3 |
| 0.01 | 1761.3 | 123.8 | 1.4 | 28.0 |
| 0.001 | 2379.8 | 402.1 | 0.1 | 625.1 |
| PV of Injected MP Suspension | IS (M) | M12 (%) | MS (%) | MT (%) |
|---|---|---|---|---|
| 2 | 0.2 | 0.49 | 103.14 | 103.63 |
| 2 | 0.1 | 4.03 | 101.18 | 105.21 |
| 2 | 0.01 | 82.51 | 23.58 | 106.09 |
| 2 | 0.001 | 90.37 | 19.65 | 110.02 |
| 10 | 0.2 | 0.73 | 101.38 | 102.1 |
| 10 | 0.1 | 3.93 | 99.21 | 103.14 |
| 10 | 0.01 | 72.3 | 37.52 | 109.82 |
| 10 | 0.001 | 93.91 | 13.36 | 107.27 |
| 50 | 0.2 | 2.36 | 90.22 | 92.57 |
| 50 | 0.1 | 8.29 | 88.06 | 96.35 |
| 50 | 0.01 | 77.52 | 27.47 | 104.99 |
| 50 | 0.001 | 89.9 | 17.96 | 107.86 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chu, X.; Li, T.; Li, Z.; Yan, A.; Shen, C. Transport of Microplastic Particles in Saturated Porous Media. Water 2019, 11, 2474. https://doi.org/10.3390/w11122474
Chu X, Li T, Li Z, Yan A, Shen C. Transport of Microplastic Particles in Saturated Porous Media. Water. 2019; 11(12):2474. https://doi.org/10.3390/w11122474
Chicago/Turabian StyleChu, Xianxian, Tiantian Li, Zhen Li, An Yan, and Chongyang Shen. 2019. "Transport of Microplastic Particles in Saturated Porous Media" Water 11, no. 12: 2474. https://doi.org/10.3390/w11122474
APA StyleChu, X., Li, T., Li, Z., Yan, A., & Shen, C. (2019). Transport of Microplastic Particles in Saturated Porous Media. Water, 11(12), 2474. https://doi.org/10.3390/w11122474





