Consequences of the Integration of a Hyperbolic Funnel into a Showerhead for Droplets, Jet Break-Up Lengths, and Physical-Chemical Parameters
Abstract
:1. Introduction
1.1. Motivation
1.2. Hyperbolic Vortices
- the velocity vector field of such structures is quite particular, meaning particles immersed in vortex structures, depending on their size, will be subjected to different tangential, axial, and radial velocities. This varies (considerably) with position and time,
- when considering liquid-based hyperbolic flow structures, there is always a well-defined air-liquid internal interface, which could be eventually used to enhance gas-diffusion in the liquid,
- for liquid structures, there is also, and necessarily, a solid-liquid interface, which would contribute to enhancing shear stresses and would be partially responsible (together with viscous stresses) for the axial velocity gradient and energy losses of the tangential component of the liquid velocity.
2. Materials and Methods
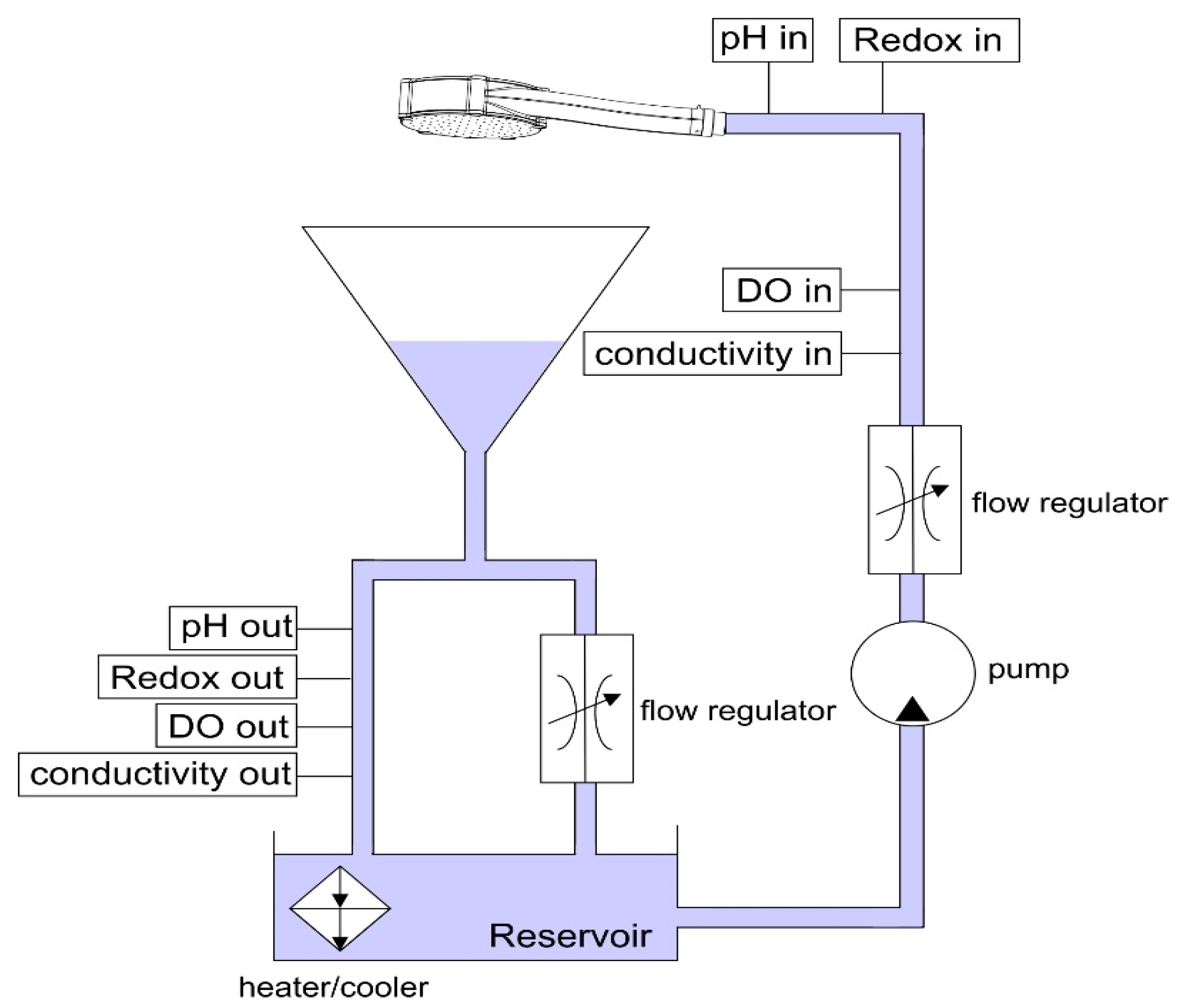
2.1. Showerheads
2.2. Jet Break-Up Length, Jet Velocity, and Droplet Characteristics
2.3. Physical and Chemical Parameters
3. Results and Discussion
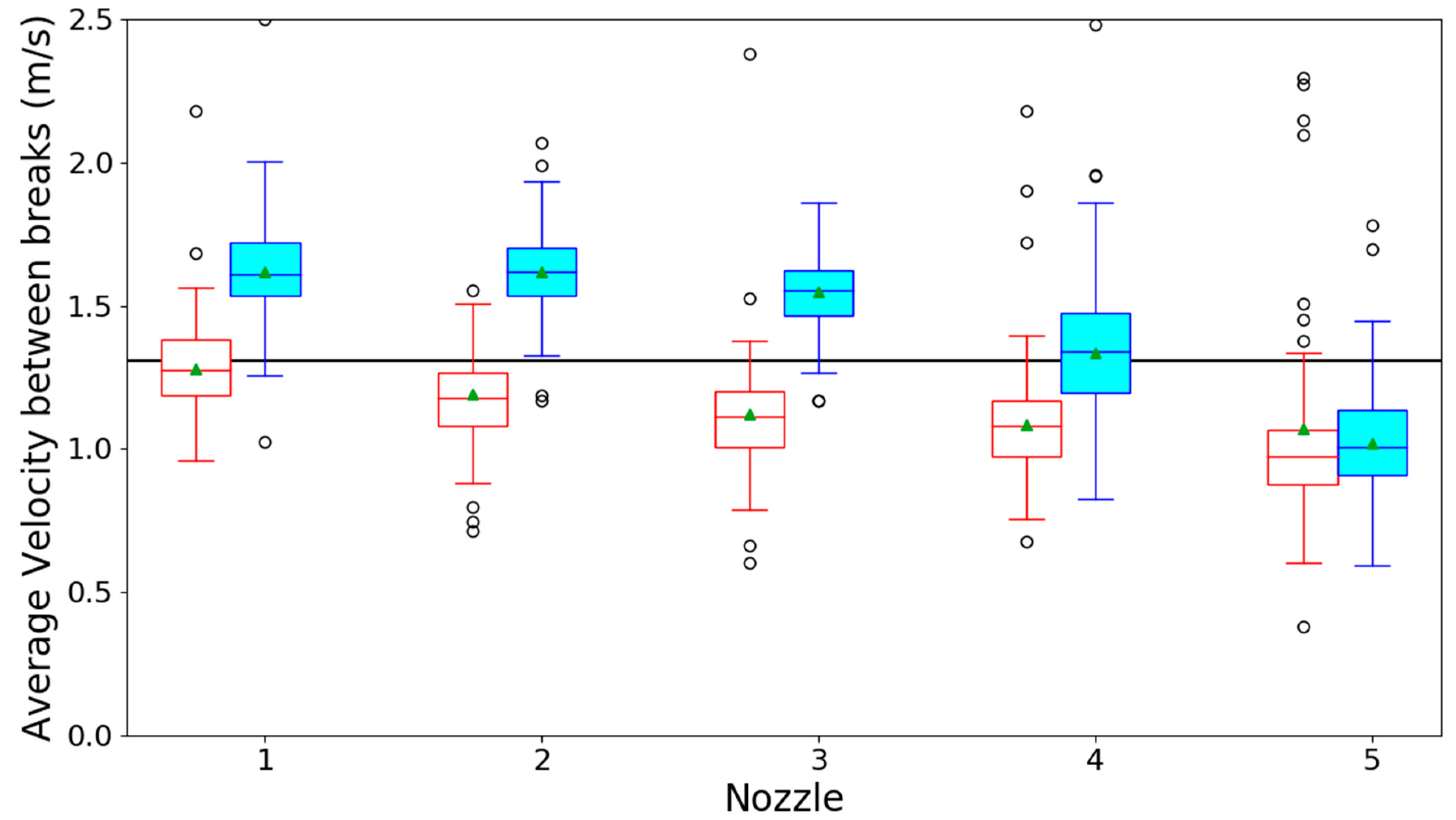
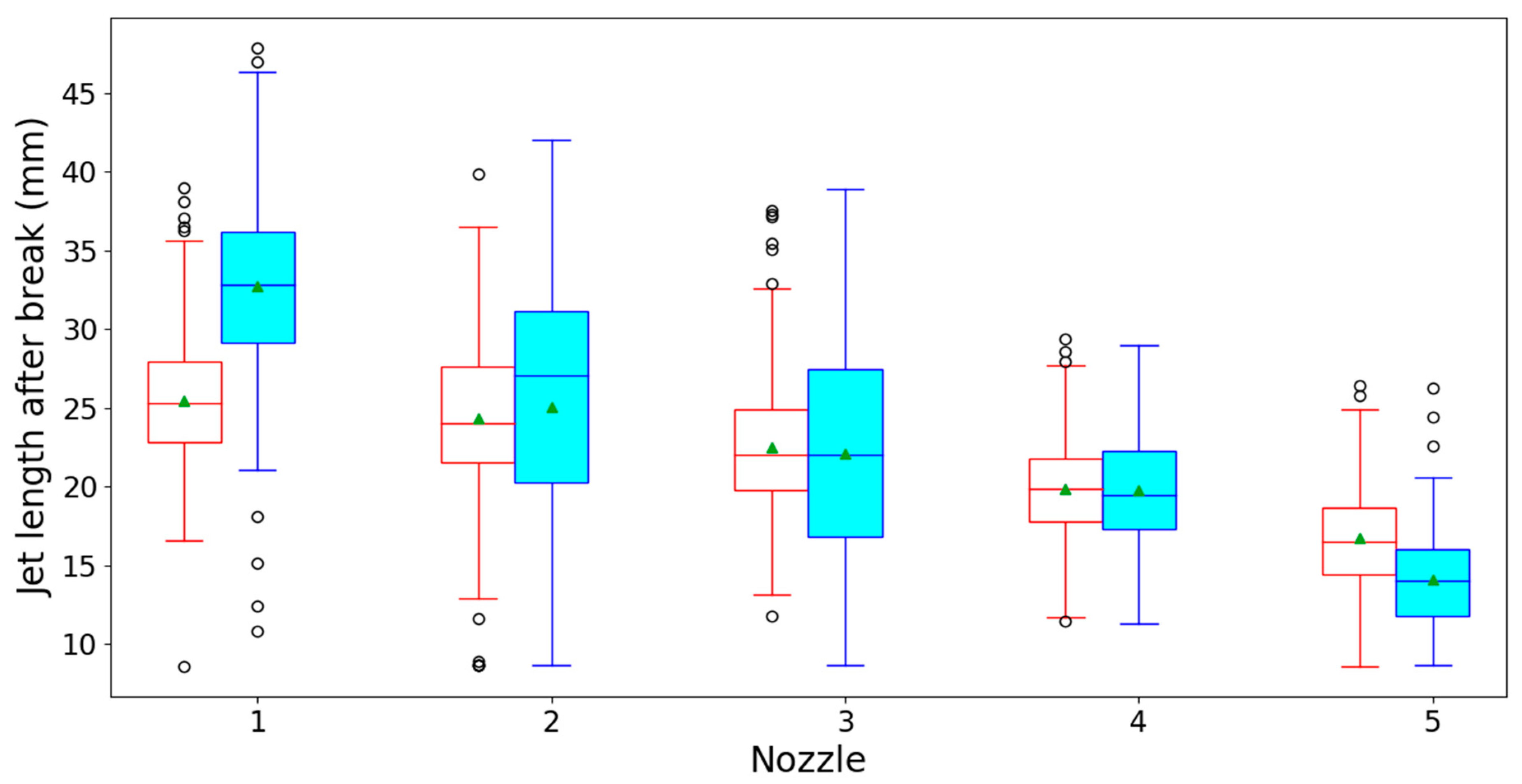
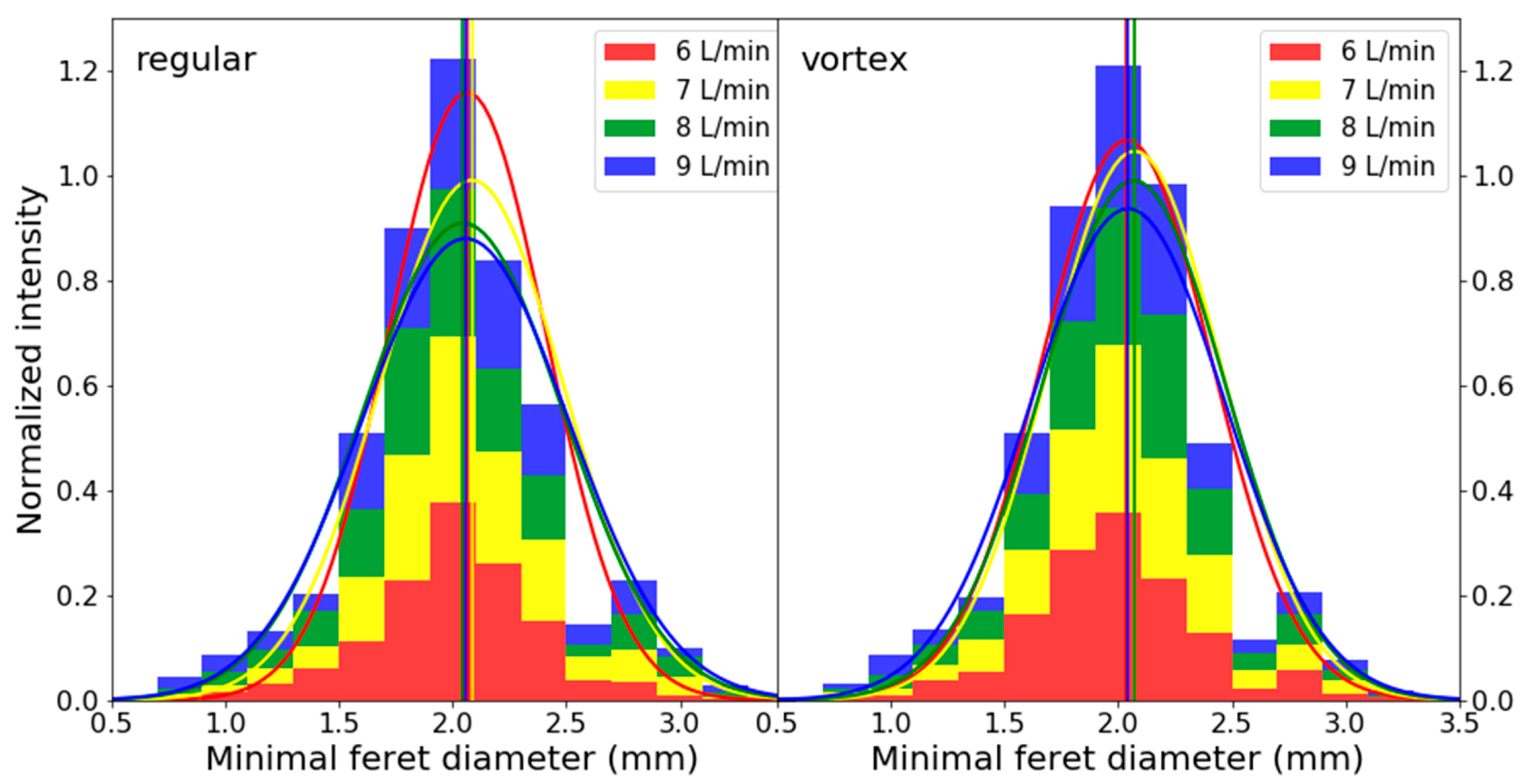
3.1. Optical Spray Analysis
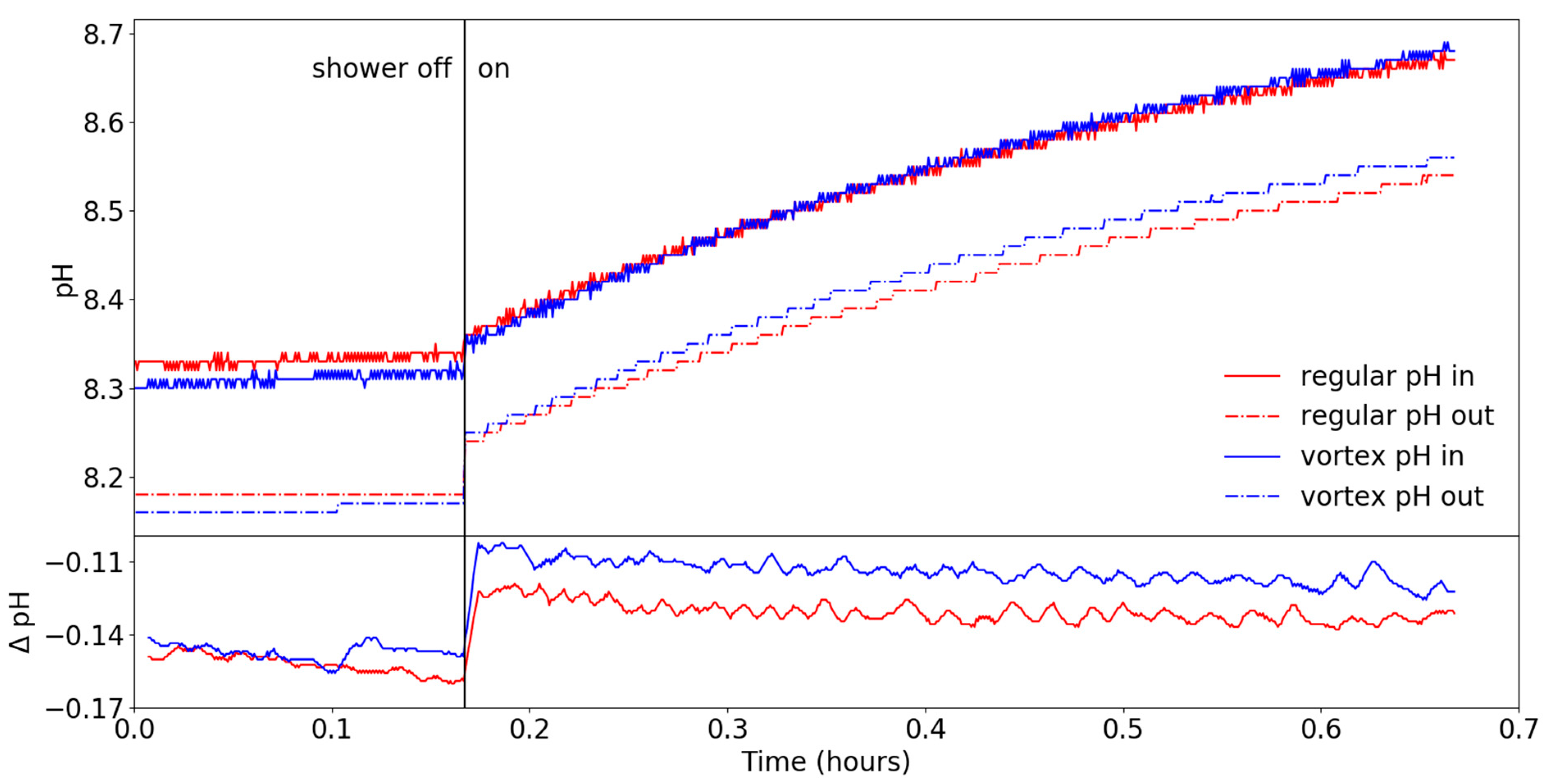
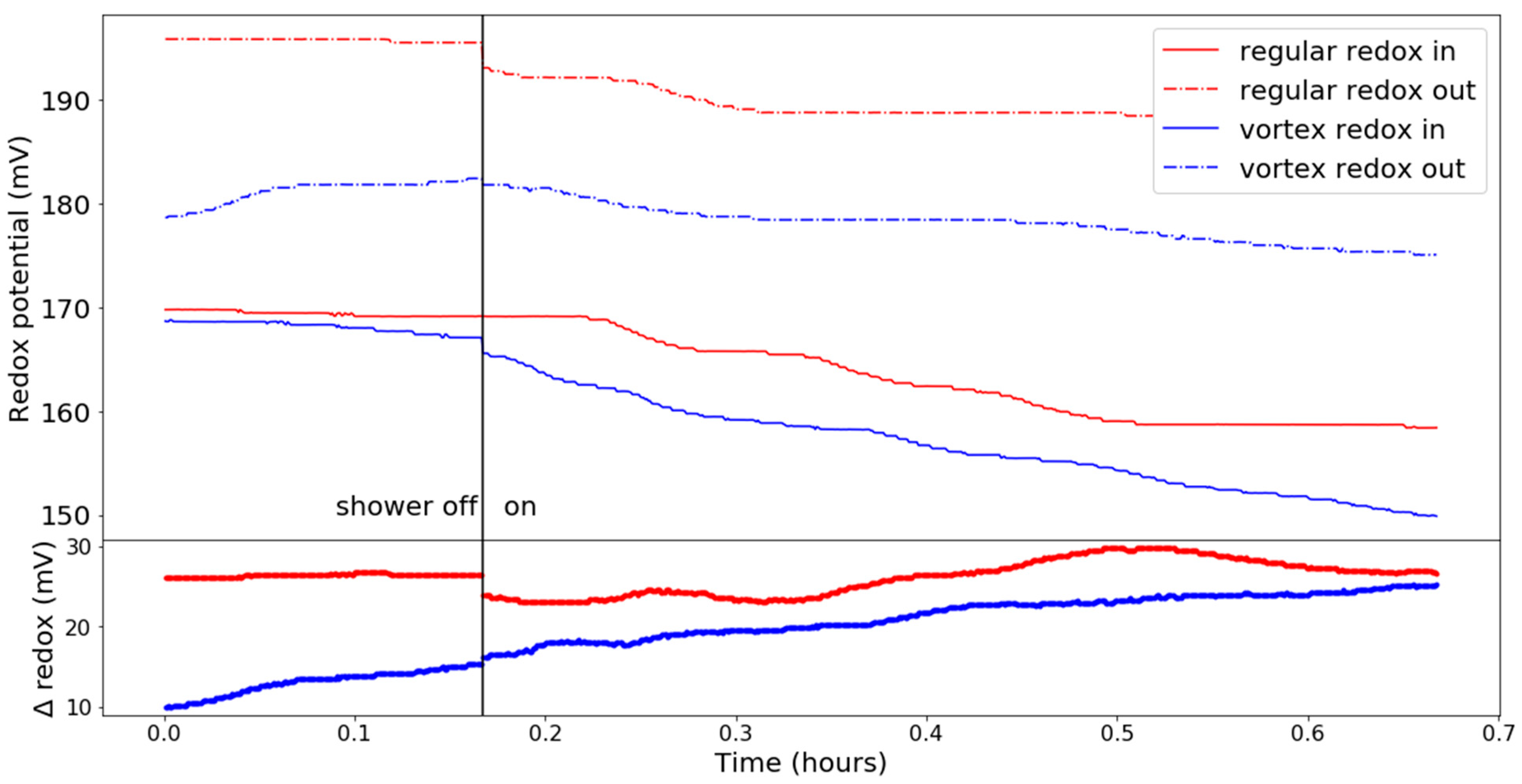
3.2. Chemical Parameters
- the change of pH,
- the (missing) increase in DO.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
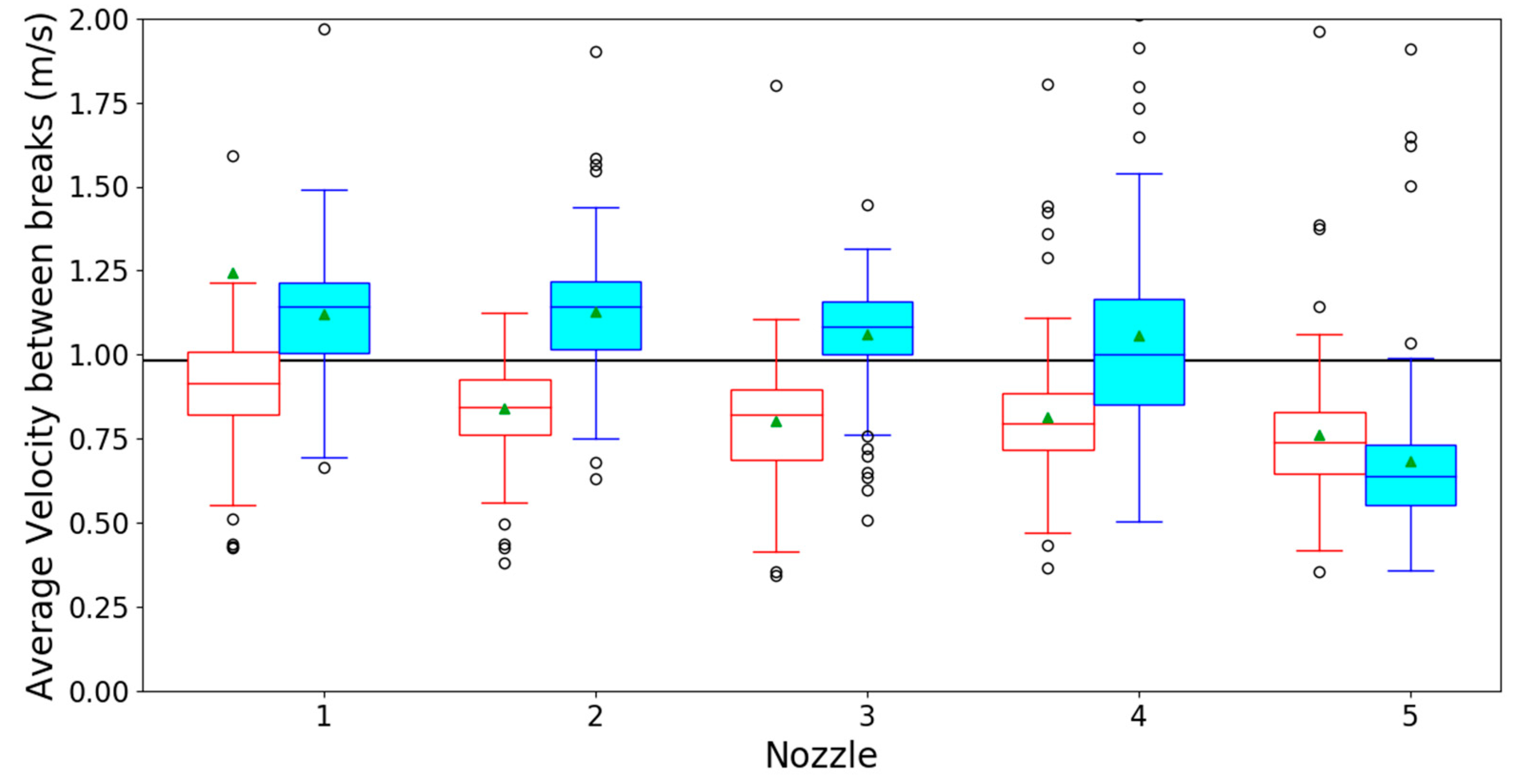
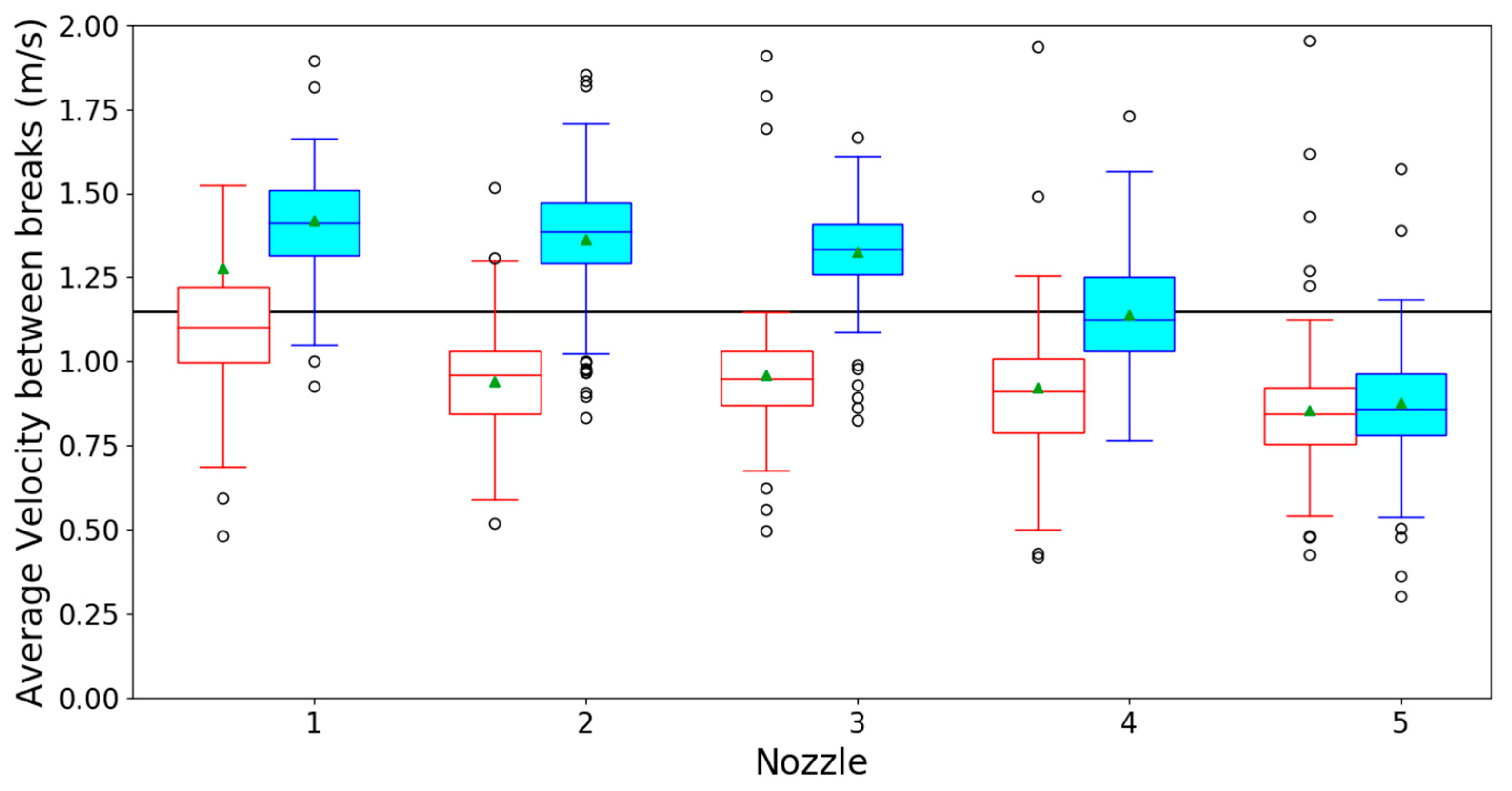
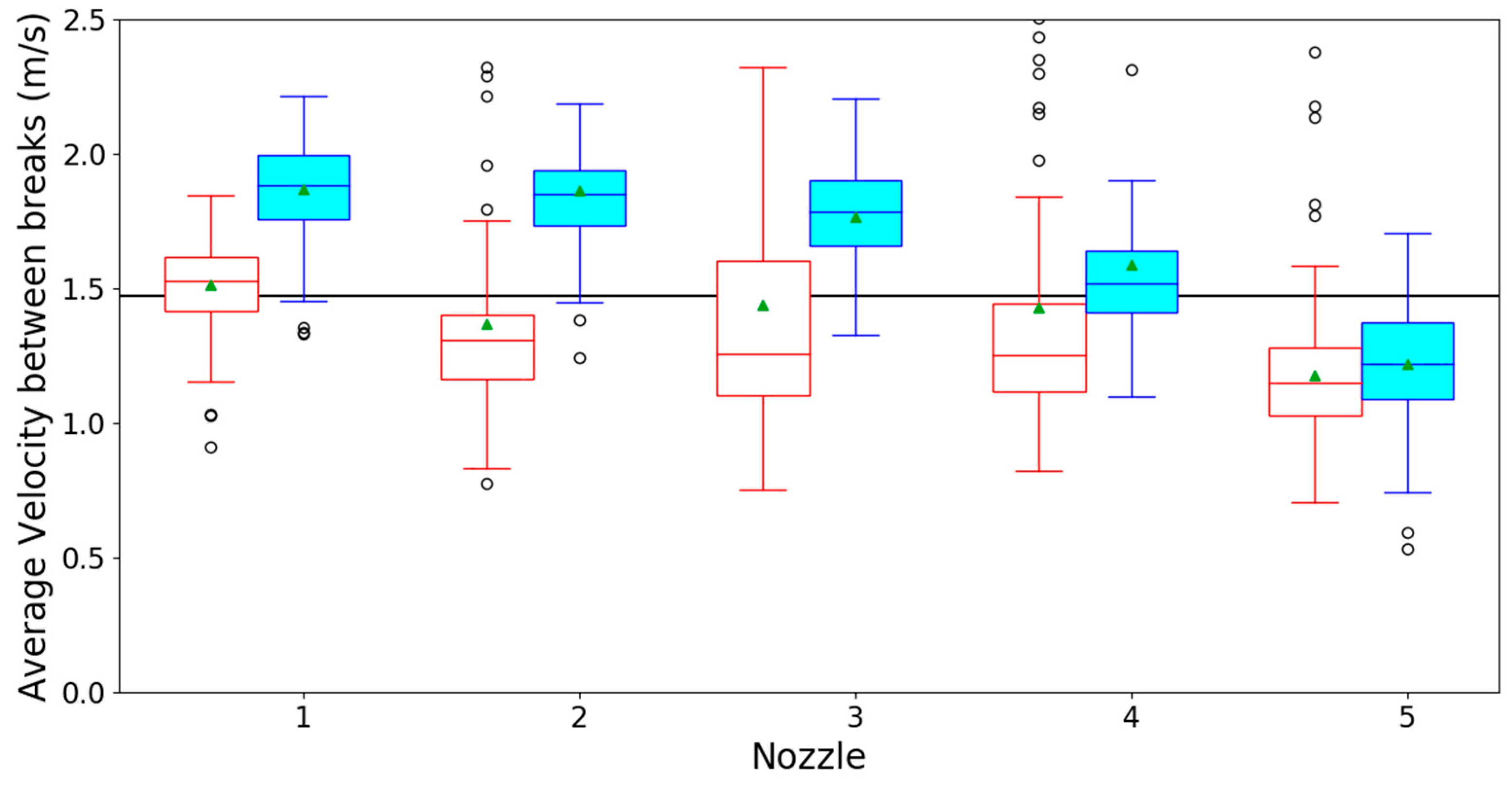
References
- United Nations. Department of Economic and Social Affairs, Population Division; World Population Prospects; United Nations: New York, NY, USA, 2019. [Google Scholar]
- Sokolow, S.; Godwin, H.; Cole, B.L. Impacts of urban water conservation strategies on energy, greenhouse gas emissions, and health: Southern California as a case study. Am. J. Public Health 2016, 106, 941–948. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, Y.; Dejima, S.; Toyosada, K. The CO2 emission factor of water in Japan. Water 2012, 4, 759–769. [Google Scholar] [CrossRef]
- Hakket, M.J.; Gray, N.F. Carbon dioxide emission savings potential of household water user reduction in the UK. J. Sustain. Dev. 2009, 2, 36–43. [Google Scholar]
- Okamoto, M.; Sato, M.; Shodai, Y.; Kamijo, M. Identifying the physical properties of showers that influence user satisfaction to aid in developing water-saving showers. Water 2015, 7, 4054–4062. [Google Scholar] [CrossRef]
- Wood, V.T.; Brown, R.A. Simulated Tornadic Vortex Signatures of Tornado-Like Vortices Having One- and Two-Celled Structures. J. Appl. Meteorol. Climatol. 2011, 50, 2338–2342. [Google Scholar] [CrossRef]
- Schauberger, V. Die Natur als Lehrmeisterin. Implosion 1963, 7, 21–27. [Google Scholar]
- Schauberger, W. Klaus Radlberger, Der Hyperbolische Kegel; PKS Eigenverlag: Bad Ischl, Austria, 2002; ISBN 3950068619. [Google Scholar]
- Drullion, F. Numerical Simulation of tornado-like vortices around complex geometries. Int. J. Comp. Math. 2009, 86, 1947–1955. [Google Scholar] [CrossRef]
- Wan, J.W.L.; Ding, X. Physically-Based Simulation of Tornados. In Workshop on Virtual Reality Interaction and Physical Simulation, Proceedings of the Second International Conference on Virtual Reality Interaction and Physical Simulation, Pisa, Italy, 8–11 November 2005; Ganovelli, F., Mendoza, C., Eds.; ISTI-CNR: Pisa, Italy, 2005. [Google Scholar]
- Trapp, R.; Fiedler, B. Numerical simulation of tornado-like vortices in asymmetric flow. In The Tornado: Its structure, dynamics, prediction, and hazards. Geophys. Monogr. 1993, 79, 49–54. [Google Scholar]
- Rotunno, R. Numerical simulation of a laboratory vortex. J. Atmospheric Sci. 1977, 34, 1942–1956. [Google Scholar] [CrossRef]
- Agostinho, L.L.F. Electrohydrodynamic Atomization in the Simple-Jet Mode: Out-scaling and Application. Ph.D. Thesis, TU Delft, Delft, The Netherlands, 2013. [Google Scholar]
- Merkus, H.G. Particle Size Measurements: Fundamentals, Practice, Quality. Springer: Pijnacker, The Netherlands, 2009; ISBN 978-1-4020-9016-5. [Google Scholar]
- Pedlosky, J. Geophysical Fluid Dynamics, 2nd ed.; Springer: New York, NY, USA, 1987; pp. 10–13, ISBN-13 978-0-387-96387-7. [Google Scholar]
- Qian, Z.; Wu, P.; Guo, Z.; Huai, W. Numerical simulation of air entrainment and suppression in pump sump. Sci. China Technol. Sci. 2016, 59, 1847–1855. [Google Scholar] [CrossRef]
- Blaszczyk, A.; Papierski, A.; Kunicki, R.; Susik, M. Surface Vortices and Pressures in Suction Intakes of Vertical Axial-Flow Pumps. Mech. Mech. Eng. 2012, 16, 51–71. [Google Scholar]
- Lin, S.P.; Reitz, R.D. Drop and spray formation from a liquid jet. Annu. Rev. Fluid Mech. 1998, 30, 85. [Google Scholar] [CrossRef]
- Van Hoeve, W.; Gekle, S.; Snoeijer, J.H.; Versluis, M.; Brenner, M.P.; Lohse, D. Breakup of diminutive Rayleigh Jets. Phys. Fluids 2010, 22, 122003. [Google Scholar] [CrossRef]
- Van Hoeve, W. Fluid Dynamics at a Pinch: Droplet and Bubble Formation in Microfluidic Devices. Ph.D. Thesis, University of Twente, Enschede, The Netherlands, 2011. [Google Scholar]
- Eggers, J.; Villermaux, E. Physics of liquid jets. Rep. Prog. Phys. 2008, 71, 036601. [Google Scholar] [CrossRef]
- Rayleigh, L. On the instability of jets. Proc. R. Soc. Lond. 1879, 10, 4. [Google Scholar] [CrossRef]
- Engineering ToolBox. Water—Density, Specific Weight and Thermal Expansion Coefficient. 2003. Available online: https://www.engineeringtoolbox.com/water-density-specific-weight-d_595.html (accessed on 31 October 2019).
- Vargaftik, N.B.; Volkov, B.N.; Voljak, L.D. International Tables of the Surface Tension of Water. J. Phys. Chem. Ref. Data 1983, 12, 817. Available online: http://twt.mpei.ru/MCS/Worksheets/iapws/Surf-H2O.xmcd (accessed on 31 October 2019). [CrossRef]
- Yin, Z.G.; Cheng, D.S.; Liang, B.C. Oxygen transfer by air injection in horizontal pipe flow. J. Environ. Eng. ASCE 2012, 139, 908–912. [Google Scholar] [CrossRef]
- Matia, L.; Rauret, G.; Rubio, R. Fresenius J. Anal. Chem. 1991, 339, 455–462. [Google Scholar] [CrossRef]
- Wetzel, R.G. Limnology. In Chapter 14—Iron, Sulfur and Silica Cycles, 3rd ed.; Academic Press: Cambridge, MA, USA, 2001; pp. 289–330. ISBN 9780127447605. [Google Scholar]
- Orna, M.V.; Stock, J. Electrochemistry, Past and Present; American Chemical Society: Columbus, OH, USA, 1989; ISBN 978-0-8412-1572-6. [Google Scholar]



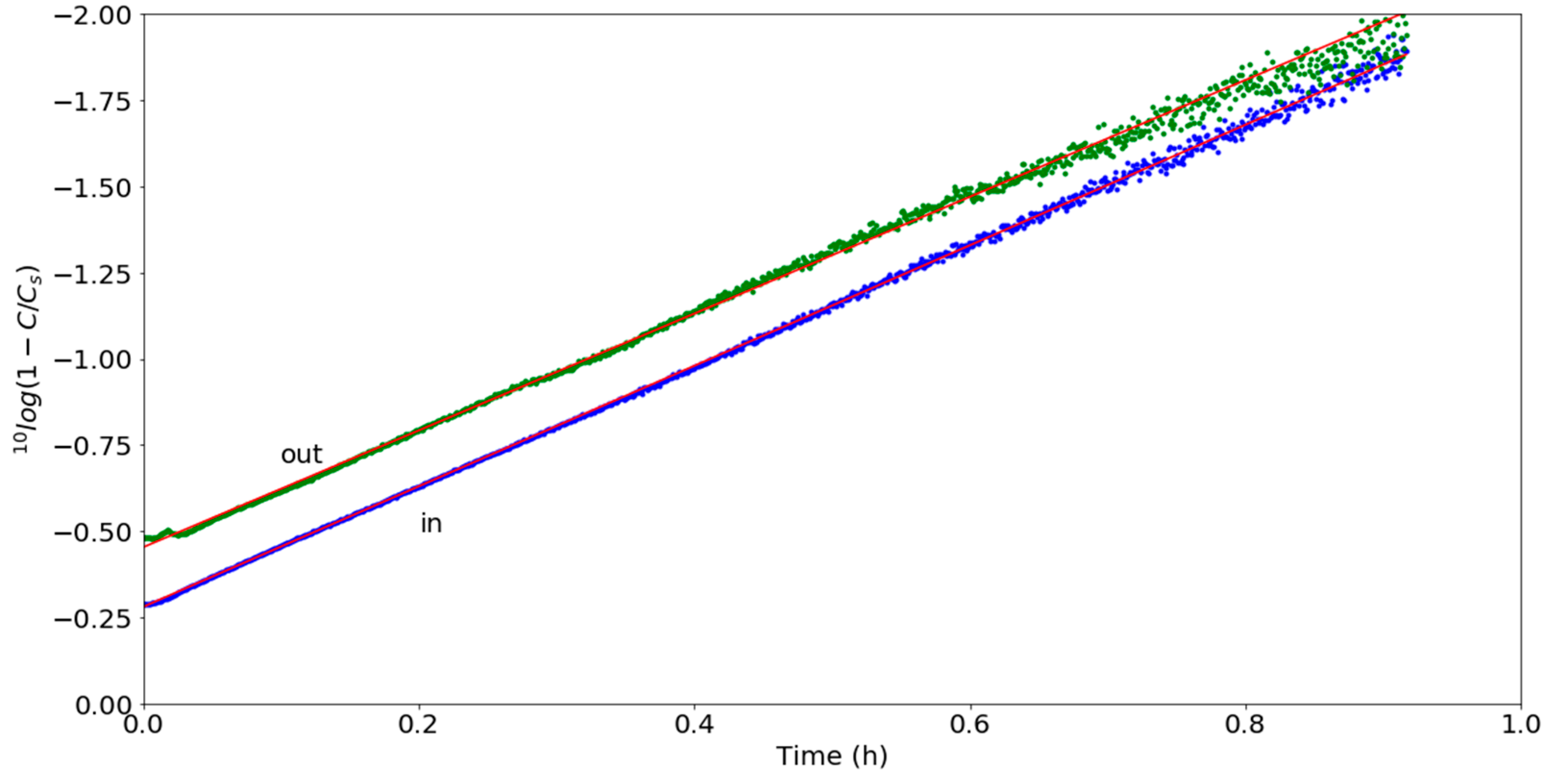
| Experiment | Showerhead | Sensor | Cs − C(t1)/ppm | τ/h | Cs/ppm | R² |
|---|---|---|---|---|---|---|
| 1 | Vortex | In | 3.13 | 0.21 | 6.13 | 0.9992 |
| Out | 2.13 | 0.20 | 6.11 | 0.9972 | ||
| Regular | In | 3.14 | 0.22 | 6.17 | 0.9998 | |
| Out | 2.09 | 0.24 | 6.17 | 0.9987 | ||
| 2 | Vortex | In | 3.23 | 0.25 | 6.17 | 0.9999 |
| Out | 2.17 | 0.26 | 6.16 | 0.9989 | ||
| Regular | In | 3.21 | 0.26 | 6.14 | 0.9998 | |
| Out | 2.20 | 0.25 | 6.12 | 0.9994 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
van de Griend, M.V.; Agostinho, L.L.F.; Fuchs, E.C.; Dyer, N.; Loiskandl, W. Consequences of the Integration of a Hyperbolic Funnel into a Showerhead for Droplets, Jet Break-Up Lengths, and Physical-Chemical Parameters. Water 2019, 11, 2446. https://doi.org/10.3390/w11122446
van de Griend MV, Agostinho LLF, Fuchs EC, Dyer N, Loiskandl W. Consequences of the Integration of a Hyperbolic Funnel into a Showerhead for Droplets, Jet Break-Up Lengths, and Physical-Chemical Parameters. Water. 2019; 11(12):2446. https://doi.org/10.3390/w11122446
Chicago/Turabian Stylevan de Griend, Maarten V., Luewton L. F. Agostinho, Elmar C. Fuchs, Nigel Dyer, and Willibald Loiskandl. 2019. "Consequences of the Integration of a Hyperbolic Funnel into a Showerhead for Droplets, Jet Break-Up Lengths, and Physical-Chemical Parameters" Water 11, no. 12: 2446. https://doi.org/10.3390/w11122446
APA Stylevan de Griend, M. V., Agostinho, L. L. F., Fuchs, E. C., Dyer, N., & Loiskandl, W. (2019). Consequences of the Integration of a Hyperbolic Funnel into a Showerhead for Droplets, Jet Break-Up Lengths, and Physical-Chemical Parameters. Water, 11(12), 2446. https://doi.org/10.3390/w11122446







