Effects of a Large Irrigation Reservoir on Aquatic and Riparian Plants: A History of Survival and Loss
Abstract
1. Introduction
2. Materials and Methods
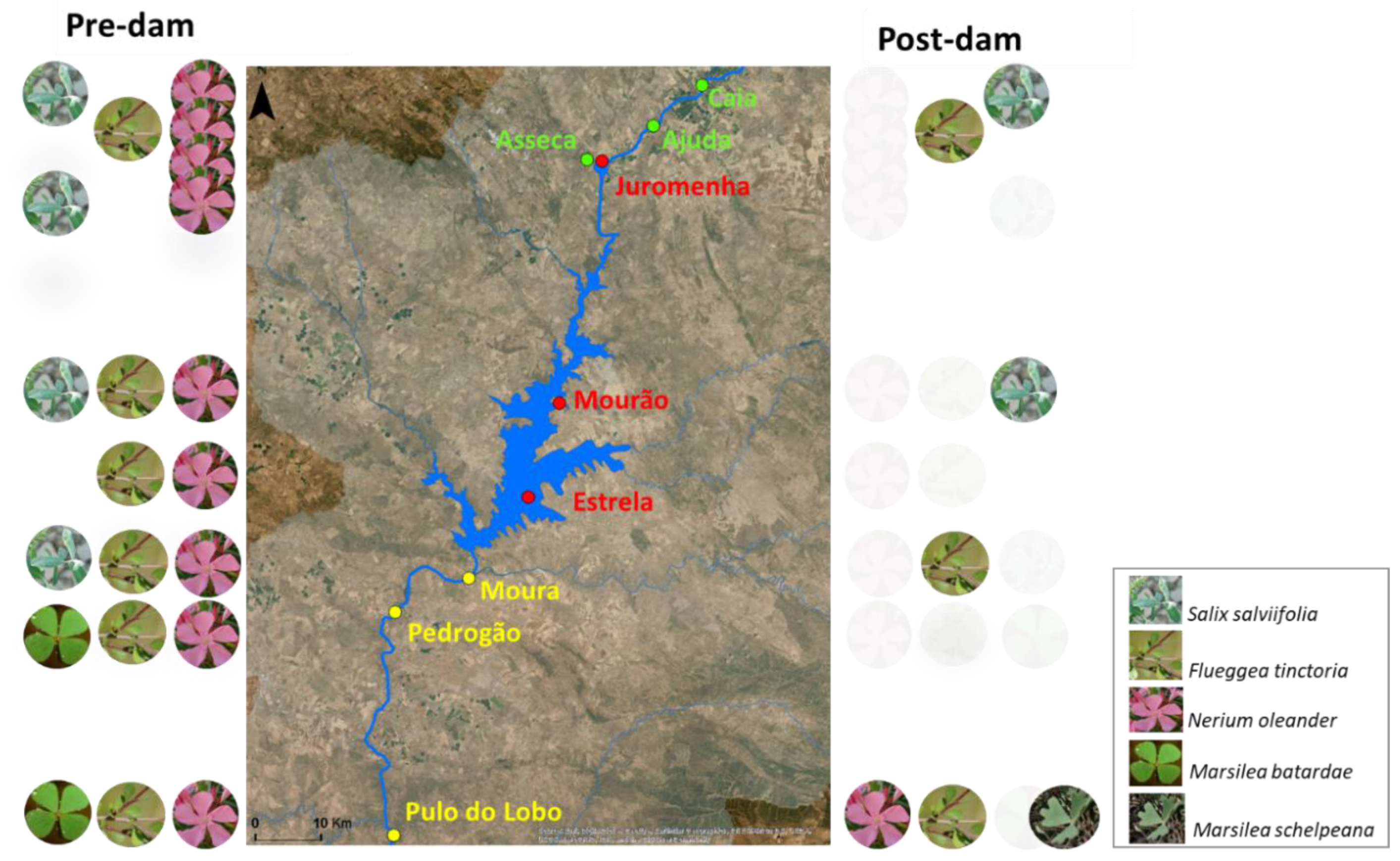
2.1. Study Area
2.2. Sampling Design and Data
2.2.1. Floristic Data
2.2.2. Spatial Data
2.2.3. Statistical Analysis
3. Results
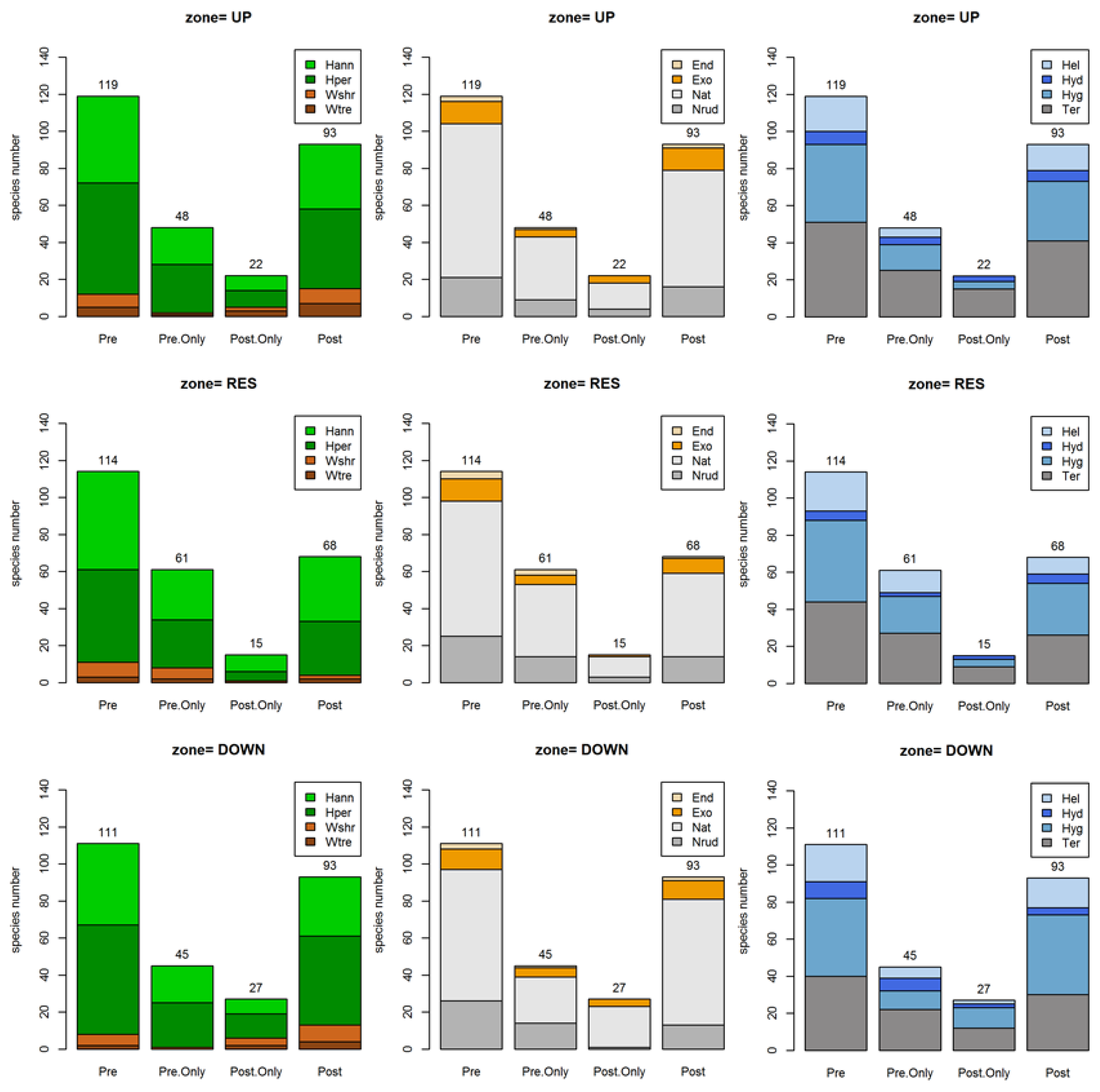
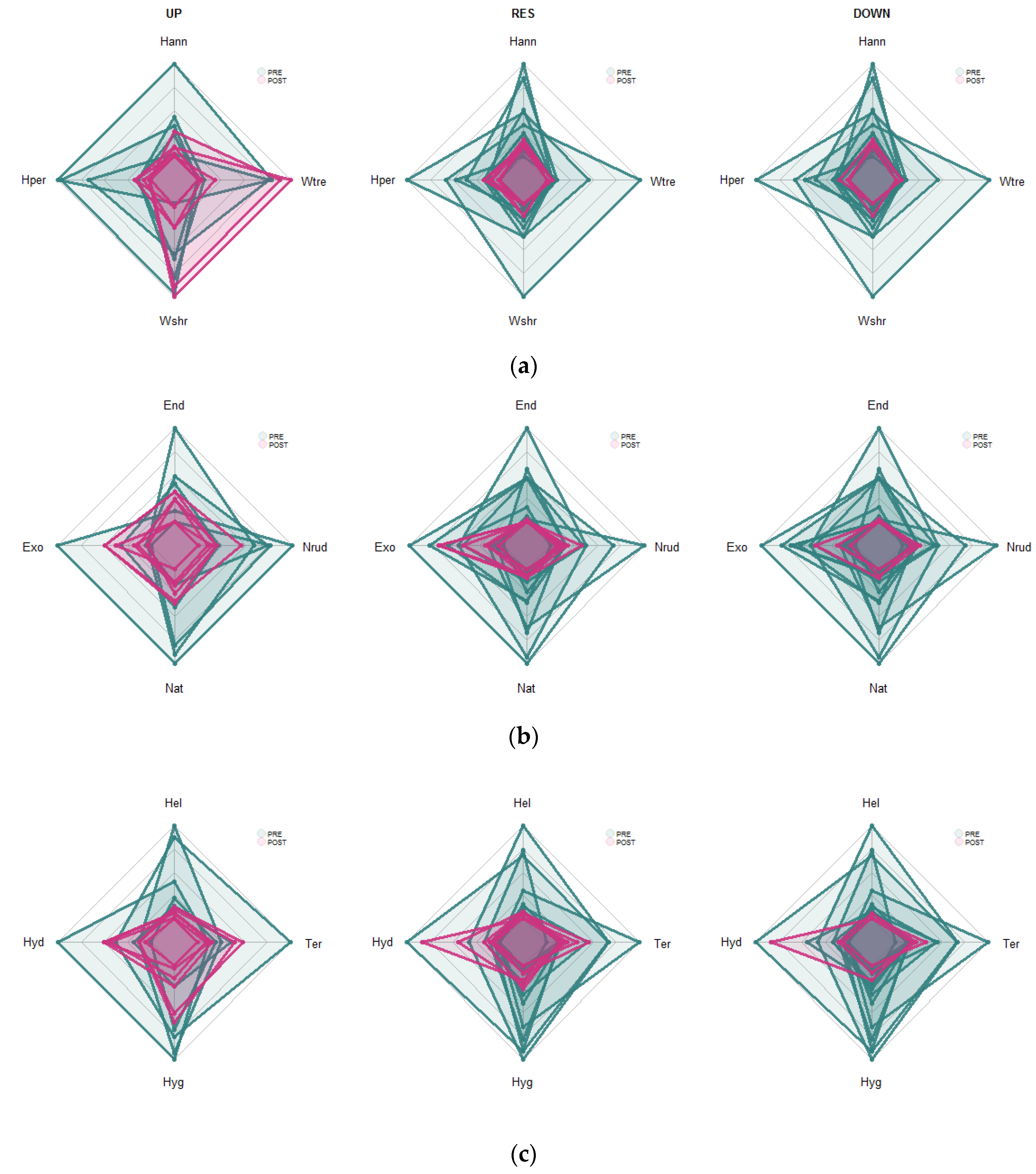
3.1. Species Richness and Floristic Composition Changes
3.2. Functional Changes
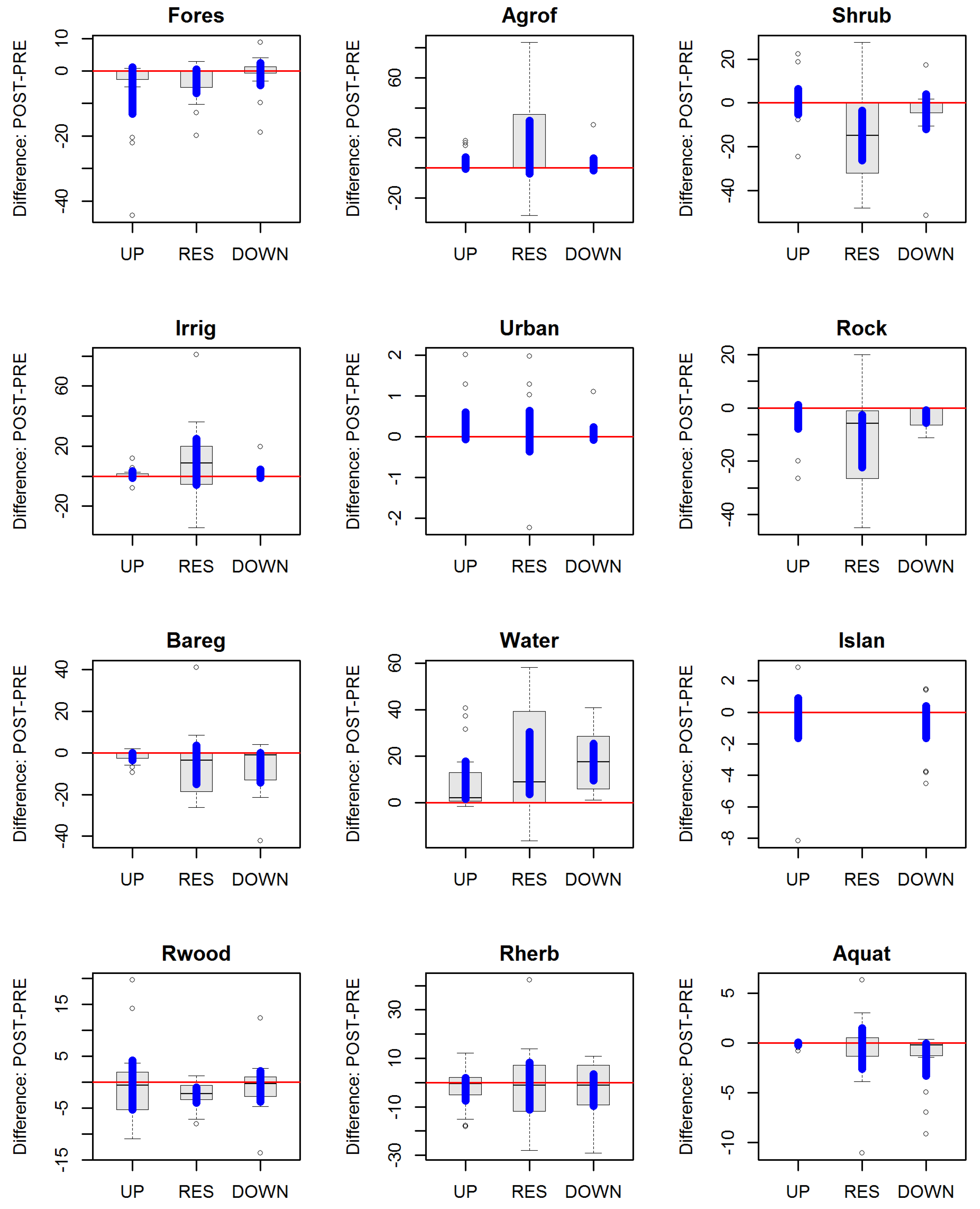
3.3. Riverine Land Use and Land Cover Changes
3.4. Compositional and Landscape Diversity
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Angeler, D.G.; Allen, C.R.; Barichievy, C.; Eason, T.; Garmestani, A.S.; Graham, N.A.; Granholm, D.; Gunderson, L.H.; Knutson, M.; Nash, K.L.; et al. Management applications of discontinuity theory. J. Appl. Ecol. 2016, 53, 688–698. [Google Scholar] [CrossRef]
- Grill, G.; Lehner, B.; Thieme, M.; Geenen, B.; Tickner, D.; Antonelli, F.; Babu, S.; Borrelli, P.; Cheng, L.; Crochetiere, H.; et al. Mapping the world’s free-flowing rivers. Nature 2019, 569, 215–221. [Google Scholar] [CrossRef]
- Karjalainen, T.P.; Järvikoski, T. Negotiating river ecosystems: Impact assessment and conflict mediation in the cases of hydro-power construction. Environ. Impact Assess. Rev. 2010, 30, 319–327. [Google Scholar] [CrossRef]
- Petts, G.E. Water engineering and landscape: Prospect for environmentally sound development. In Water, Engineering and Landscape: Water Control and Landscape Transformation in the Modern Period; Cosgrove, D., Petts, G.E., Eds.; Bellhaven: London, UK, 1990; pp. 188–208. [Google Scholar]
- Poff, N.L.; Olden, J.D.; Merritt, D.M.; Pepin, D.M. Homogenization of regional river dynamics by dams and global biodiversity implications. Proc. Natl. Acad. Sci. USA 2007, 104, 5732–5737. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, C.; Berggren, K. Alterations of Riparian Ecosystems Caused by River Regulation: Dam operations have caused global-scale ecological changes in riparian ecosystems. BioScience 2000, 50, 783–792. [Google Scholar] [CrossRef]
- Corenblit, D.; Steiger, J.; Tabacchi, E. Biogeomorphologic succession dynamics in a Mediterranean river system. Ecography 2010, 33, 1136–1148. [Google Scholar] [CrossRef]
- Del Tánago, M.G.; Gurnell, A.M.; Belletti, B.; De Jalón, D.G. Indicators of river system hydromorphological character and dynamics: Understanding current conditions and guiding sustainable river management. Aquat. Sci. 2016, 78, 35–55. [Google Scholar] [CrossRef]
- Garófano-Gómez, V.; Martínez-Capel, F.; Bertoldi, W.; Gurnell, A.; Estornell, J.; Segura-Beltrán, F. Six decades of changes in the riparian corridor of a Mediterranean river: A synthetic analysis based on historical data sources. Ecohydrology 2013, 6, 536–553. [Google Scholar] [CrossRef]
- Baxter, R.M. Environmental effects of dams and impoundments. Ann. Rev. Ecol. Syst. 1977, 8, 255–283. [Google Scholar] [CrossRef]
- Godinho, F.N.; Pinheiro, P.J.; Oliveira, J.M.; Azedo, R. Responses of intermittent stream fish assemblages to irrigation development. River Res. Appl. 2014, 30, 1248–1256. [Google Scholar] [CrossRef]
- Kondolf, G.M. PROFILE: Hungry water: Effects of dams and gravel mining on river channels. Environ. Manag. 1997, 21, 533–551. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, F.C.; Segurado, P.; Martins, M.J.; Bejarano, M.D.; Nilsson, C.; Portela, M.M.; Merritt, D. The abundance and distribution of guilds of riparian woody plants change in response to land use and flow regulation. J. Appl. Ecol. 2018, 55, 2227–2240. [Google Scholar] [CrossRef]
- Bejarano, M.D.; Nilsson, C.; Aguiar, F.C. Riparian plant guilds become simpler and most likely fewer following flow regulation. J. Appl. Ecol. 2018, 55, 365–376. [Google Scholar] [CrossRef]
- Petts, G.E.; Morales, Y.; Sadler, J.P. Linking hydrology and biology in assessing water needs for riverine ecosystems. Hydrol. Process. 2006, 20, 2247–2251. [Google Scholar] [CrossRef]
- Aguiar, F.C.; Martins, M.J.; Silva, P.C.; Fernandes, M.R. Riverscapes downstream of hydropower dams: Effects of altered flows and historical land-use change. Landsc. Urban Plan. 2016, 153, 83–98. [Google Scholar] [CrossRef]
- Rivaes, R.; Pinheiro, A.N.; Egger, G.; Ferreira, M.T. The Role of River Morphodynamic Disturbance and Groundwater Hydrology as Driving Factors of Riparian Landscape Patterns in Mediterranean Rivers. Front. Plant Sci. 2017, 8, 1612. [Google Scholar] [CrossRef]
- Fraaije, R.G.; Braak, C.J.; Verduyn, B.; Verhoeven, J.T.; Soons, M.B. Dispersal versus environmental filtering in a dynamic system: Drivers of vegetation patterns and diversity along stream riparian gradients. J. Ecol. 2015, 103, 1634–1646. [Google Scholar] [CrossRef]
- Liro, M. Dam reservoir backwater as a field-scale laboratory of human-induced changes in river biogeomorphology: A review focused on gravel-bed rivers. Sci. Total Environ. 2019, 651, 2899–2912. [Google Scholar] [CrossRef]
- Grabowski, R.C.; Surian, N.; Gurnell, A.M. Characterizing geomorphological change to support sustainable river restoration and management. Water 2014, 1, 483–512. [Google Scholar] [CrossRef]
- Brierley, G.J.; Fryirs, K.A. Geomorphology and River Management: Applications of the River Styles Framework; John Wiley & Sons: Chichester, UK, 2013. [Google Scholar]
- Arvela, A.S.; Panagopoulos, T.; Cakula, A.; Ferreira, V.; Azevedo, J.C. Analysis of Landscape Change Following the Construction of the Alqueva Dam, southern Portugal: Approach and Methods. In Recent Researches in Environmental Science and Landscaping; Energy, Environmental and Structural Engineering Series 2; Burley, J., Loures, L., Panagopoulos, T., Eds.; WSEAS Press: Faro, Portugal, 2012; pp. 42–47. [Google Scholar]
- Lehner, B.; Liermann, C.R.; Revenga, C.; Vörösmarty, C.; Fekete, B.; Crouzet, P.; Döll, P.; Endejan, M.; Frenken, K.; Magome, J.K.; et al. High-resolution mapping of the world’s reservoirs and dams for sustainable river-flow management. Front. Ecol. Environ. 2011, 9, 494–502. [Google Scholar] [CrossRef]
- EDIA—Empresa de Desenvolvimento e Infra-Estruturas do Alqueva, S. EDIA, SA. Available online: http://www.edia.pt/en/ (accessed on 3 July 2019).
- Radke, J.; Pinto, P.; Lachwani, K.; Kondlof, G.M.; Rocha, J.; Serra Lloet, A. Alqueva. Changing Ecologies of the Montado Landscape. Alentejo, Portugal; The Department of Landscape Architecture & Environmental Planning and The Portuguese Studies Program; University of California: Berkeley, CA, USA, 2015. [Google Scholar]
- Santos, M.J.; Pedroso, N.M.; Ferreira, J.P.; Matos, H.M.; Sales-Luís, T.; Pereira, Í.; Baltazar, C.; Grilo, C.; Cândido, A.T.; Sousa, I.; et al. Assessing dam implementation impact on threatened carnivores: The case of Alqueva in SE Portugal. Environ. Monit. Assess. 2008, 142, 47–64. [Google Scholar] [CrossRef] [PubMed]
- Ballester-Hernández, S.; Rosselló-Graell, A.; Draper, D.; Correia, A.I.D. Monitorização de plantas prioritárias na área da albufeira do Alqueva. Linhas metodológicas. Port. Acta Biol. 2000, 19, 201–218. [Google Scholar]
- Pereira, M.; Figueiredo, D. Effects of the Alqueva Dam Reservoir on the Distribution of Steppe Birds. Phys. Geogr. 2009, 30, 43–63. [Google Scholar] [CrossRef]
- Fulan, J.A.; Raimundo, R.; Figueiredo, D.; Correia, M. Abundance and diversity of dragonflies four years after the construction of a reservoir. Limnetica 2010, 29, 279–286. [Google Scholar]
- Morais, P. Review on the major ecosystem impacts caused by damming and watershed development in an Iberian basin (SW-Europe): Focus on the Guadiana estuary. Ann. Limnol. Int. J. Limnol. 2008, 44, 105–117. [Google Scholar] [CrossRef]
- Chícharo, M.A.; Chícharo, L.; Morais, P. Inter-annual differences of ichthyofauna structure of the Guadiana estuary and adjacent coastal area (SE Portugal/SW Spain): Before and after Alqueva dam construction. Estuar. Coast. Shelf Sci. 2006, 70, 39–51. [Google Scholar] [CrossRef]
- Morais, M.M.; Serafim, A.M.; Pinto, P.; Ilhéu, A.; Ruivo, M. Reservoir and River Basin Management. Exchange of Experiences from Brazil, Portugal and Germany. In Monitoring the Water Quality in Alqueva Reservoir, Guadiana River, Southern Portugal; Gunkel, G., Sobral, M., Eds.; Technical University of Berlin: Berlin, Germany, 2007; pp. 96–113. [Google Scholar]
- Palma, P.; Alvarenga, P.; Palma, V.; Matos, C.; Fernandes, R.M.; Soares, A.; Barbosa, I.R. Evaluation of surface water quality using an ecotoxicological approach: A case study of the Alqueva Reservoir (Portugal). Environ. Sci. Pollut. Res. 2010, 17, 703–716. [Google Scholar] [CrossRef]
- Ferreira, T.; Monteiro, A. Estudo das comunidades de macrófitos aquáticos do rio Guadiana. Actas Col. Ecol. Bacias Hidrogr. Rec. Zoológicos 1988, 129–137. [Google Scholar]
- Capelo, J. Esboço da paisagem vegetal da bacia portuguesa do rio Guadiana. Silva Lusit. 1996, 4, 13–64. [Google Scholar]
- Ferreira, M.T.; Godinho, F.N.; Cortes, R.M. Macrophytes in a southern Iberian river. Int. Ver. Theor. Angew. Limnol. Verh. 1998, 26, 1835–1841. [Google Scholar] [CrossRef]
- Rosselló-Graell, A.; Draper, D.; Tauleigne Gomes, C.; Correia, A.I.D. Marsilea batardae Launert em Portugal e determinação do seu estatuto de ameaça. Port. Acta Biol. 2000, 19, 219–224. [Google Scholar]
- Ferreira, M.T.; Albuquerque, A.; Aguiar, F.C.; Catarino, L.F. Seasonal and yearly variations of macrophytes in a Southern Iberian River. Int. Ver. Theor. Angew. Limnol. Verh. 2001, 27, 3833–3837. [Google Scholar] [CrossRef]
- Ferreira, M.T.; Albuquerque, A.; Aguiar, F.C.; Sidorkewicz, N. Assessing reference sites and ecological quality of river plant assemblages from an Iberian basin using a multivariate approach. Arch. Hydrobiol. 2002, 155, 121–145. [Google Scholar] [CrossRef]
- Aguiar, F.C.; Ferreira, M.T.; Albuquerque, A. Patterns of exotic and native plant species richness and over along a semi-arid Iberian river and across its floodplain. Plant Ecol. 2006, 184, 189–202. [Google Scholar] [CrossRef]
- Draper, D.; Marques, I.; Iriondo, J.M. Species distribution models with field validation, a key approach for successful selection of receptor sites in conservation translocations. Glob. Ecol. Conserv. 2019, 19, e00653. [Google Scholar] [CrossRef]
- Portuguese Environment Agency (APA). Plano de Gestão de Região Hidrográfica. Parte 2—Caracterização e Diagnóstico. Região Hidrográfica do Guadiana (RH7); APA: Amadora, Portugal, 2016. [Google Scholar]
- Neto, S. Alqueva dam in the Guadiana River Basin—Past and future of illusive water abundance. Water Policy 2019, 5, 71–84. [Google Scholar] [CrossRef]
- Aguiar, F.C.; Ferreira, M.T.; Albuquerque, A.; Moreira, I. Alien and endemic flora on reference and non-reference sites from Mediterranean type-streams of Portugal. Aquat. Conserv. Mar. Freshw. Ecosysyst. 2007, 17, 335–347. [Google Scholar] [CrossRef]
- Franco, J.A.; Rocha-Afonso, M.L. Nova Flora de Portugal (Continente e Açores). 3(1) Alismataceae-Iridaceae; 3(2) Gramineae; 3(3) Juncaceae-Orchidaceae; Escolar Editora: Lisbon, Portugal, 1994. [Google Scholar]
- Christenhusz, M.; Lansdown, R.V.; Bento Elias, R.; Dyer, R.; Ivanenko, Y.; Rouhan, G.; Rumsey, F.; Väre, H. Marsilea batardae. The IUCN Red List of Threatened Species 2017; E.T161966A85434785; International Union for Conservation of Nature and Natural Resources: Cambridge, UK, 2019. [Google Scholar]
- Victor, J.E.; Dold, A.P. Marsilea schelpeana. The IUCN Red List of Threatened Species 2010; E.T185228A8371113; International Union for Conservation of Nature and Natural Resources: Cambridge, UK, 2019. [Google Scholar]
- Lavorel, S. Ecological diversity and resilience of Mediterranean vegetation to disturbance. Divers. Distrib. 1999, 5, 3–13. [Google Scholar] [CrossRef]
- Fraaije, R.G.; Braak, C.J.; Verduyn, B.; Breeman, L.B.; Verhoeven, J.T.; Soons, M.B. Early plant recruitment stages set the template for the development of vegetation patterns along a hydrological gradient. Funct. Ecol. 2015, 29, 971–980. [Google Scholar] [CrossRef]
- Su, X.; Polvi, L.E.; Lind, L.; Pilotto, F.; Nilsson, C. Importance of landscape context for post-restoration recovery of riparian vegetation. Freshw. Biol. 2019, 64, 1015–1028. [Google Scholar] [CrossRef]
- Guerreiro, S.B.; Birkinshaw, S.; Kilsby, C.; Fowler, H.J.; Lewis, E. Dry getting drier—The future of transnational river basins in Iberia. J. Hydrol. Reg. Stud. 2017, 12, 238–252. [Google Scholar] [CrossRef]
- Canuto, N.; Ramos, T.B.; Oliveira, A.R.; Simionesei, L.; Basso, M.; Neves, R. Influence of reservoir management on Guadiana streamflow regime. J. Hydrol. Reg. Stud. 2019, 25, 100628. [Google Scholar] [CrossRef]
- Brandt, S.A. Classification of geomorphological effects downstream of dams. Catena 2000, 40, 375–401. [Google Scholar] [CrossRef]
- Reynolds, C.; Miranda, N.A.; Cumming, G.S. The role of waterbirds in the dispersal of aquatic alien and invasive species. Divers. Distrib. 2015, 21, 744–754. [Google Scholar] [CrossRef]
- Braatne, J.H.; Rood, S.B.; Goater, L.A.; Blair, C.L. Analyzing the impacts of dams on riparian ecosystems: A review of research strategies and their relevance to the Snake River through Hells Canyon. Environ. Manag. 2008, 41, 267–281. [Google Scholar] [CrossRef]
- Rood, S.B.; Braatne, J.H.; Goater, L.A. Favorable fragmentation: River reservoirs can impede downstream expansion of riparian weeds. Ecol. Appl. 2010, 20, 1664–1677. [Google Scholar] [CrossRef]
- Larson, J.E.; Funk, J.L. Regeneration: An overlooked aspect of trait-based plant community assembly models. J. Ecol. 2016, 104, 1284–1298. [Google Scholar] [CrossRef]
- Parody, J.M.; Cuthbert, F.J.; Decker, E. The effect of 50 years of landscape change on species richness and community composition. Glob. Ecol. Biogeogr. 2001, 10, 305–313. [Google Scholar] [CrossRef]
- Godinho, C.; Rabaça, J.E.; Segurado, P. Breeding bird assemblages in riparian galleries of the Guadiana River basin Portugal—The effect of habitat structure and habitat variables. Ecol. Res. 2010, 25, 283–294. [Google Scholar] [CrossRef]
- Rebelo, H.; Rainho, A. Bat conservation and large dams: Spatial changes in habitat use caused by Europe’s largest reservoir. Endanger. Species Res. 2009, 8, 61–68. [Google Scholar] [CrossRef]
- Daveau, S. Bases geográficas do problema da barragem de Alqueva. Finisterra 1977, 12, 342–351. [Google Scholar] [CrossRef]
- Stallins, J.A.; Corenblit, D. Interdependence of geomorphic and ecologic resilience properties in a geographic context. Geomorphology 2018, 305, 76–93. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aguiar, F.C.; Fernandes, M.R.; Martins, M.J.; Ferreira, M.T. Effects of a Large Irrigation Reservoir on Aquatic and Riparian Plants: A History of Survival and Loss. Water 2019, 11, 2379. https://doi.org/10.3390/w11112379
Aguiar FC, Fernandes MR, Martins MJ, Ferreira MT. Effects of a Large Irrigation Reservoir on Aquatic and Riparian Plants: A History of Survival and Loss. Water. 2019; 11(11):2379. https://doi.org/10.3390/w11112379
Chicago/Turabian StyleAguiar, Francisca C., Maria Rosário Fernandes, Maria João Martins, and Maria Teresa Ferreira. 2019. "Effects of a Large Irrigation Reservoir on Aquatic and Riparian Plants: A History of Survival and Loss" Water 11, no. 11: 2379. https://doi.org/10.3390/w11112379
APA StyleAguiar, F. C., Fernandes, M. R., Martins, M. J., & Ferreira, M. T. (2019). Effects of a Large Irrigation Reservoir on Aquatic and Riparian Plants: A History of Survival and Loss. Water, 11(11), 2379. https://doi.org/10.3390/w11112379






