Activated Carbon from Yam Peels Modified with Fe3O4 for Removal of 2,4-Dichlorophenoxyacetic Acid in Aqueous Solution
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Activated Carbon (AC) and Magnetic Activated Carbon (MAC)
2.2.1. Synthesis of Magnetite (Fe3O4) Nanoparticles
2.2.2. Preparation of Activated Carbon (AC) from D. Rotundata Peels
2.2.3. Preparation of MAC Using Magnetite Fe3O4 Nanoparticles
2.3. Characterization of Materials
2.4. Batch Adsorption Study
2.4.1. Effect of Solution pH
2.4.2. Effect of Initial Concentration, Temperature, and Contact Time
Adsorption Kinetics
2.4.3. Adsorption Isotherms
2.5. Regeneration Study
3. Results and Discussion
3.1. Characterization of AC and MAC
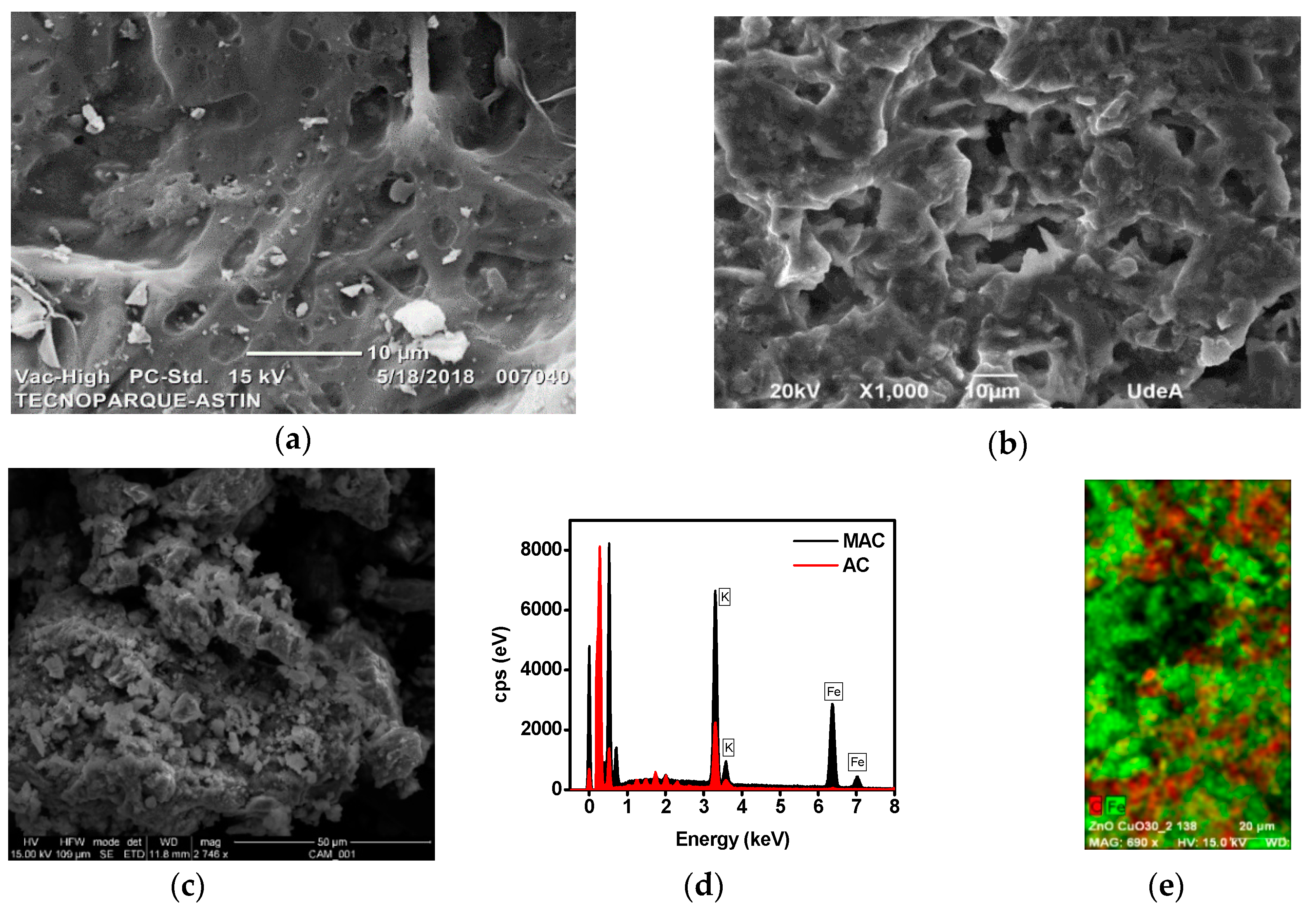
3.1.1. SEM-EDS
3.1.2. BET (Brunauer, Emmett and Teller) Surface Area
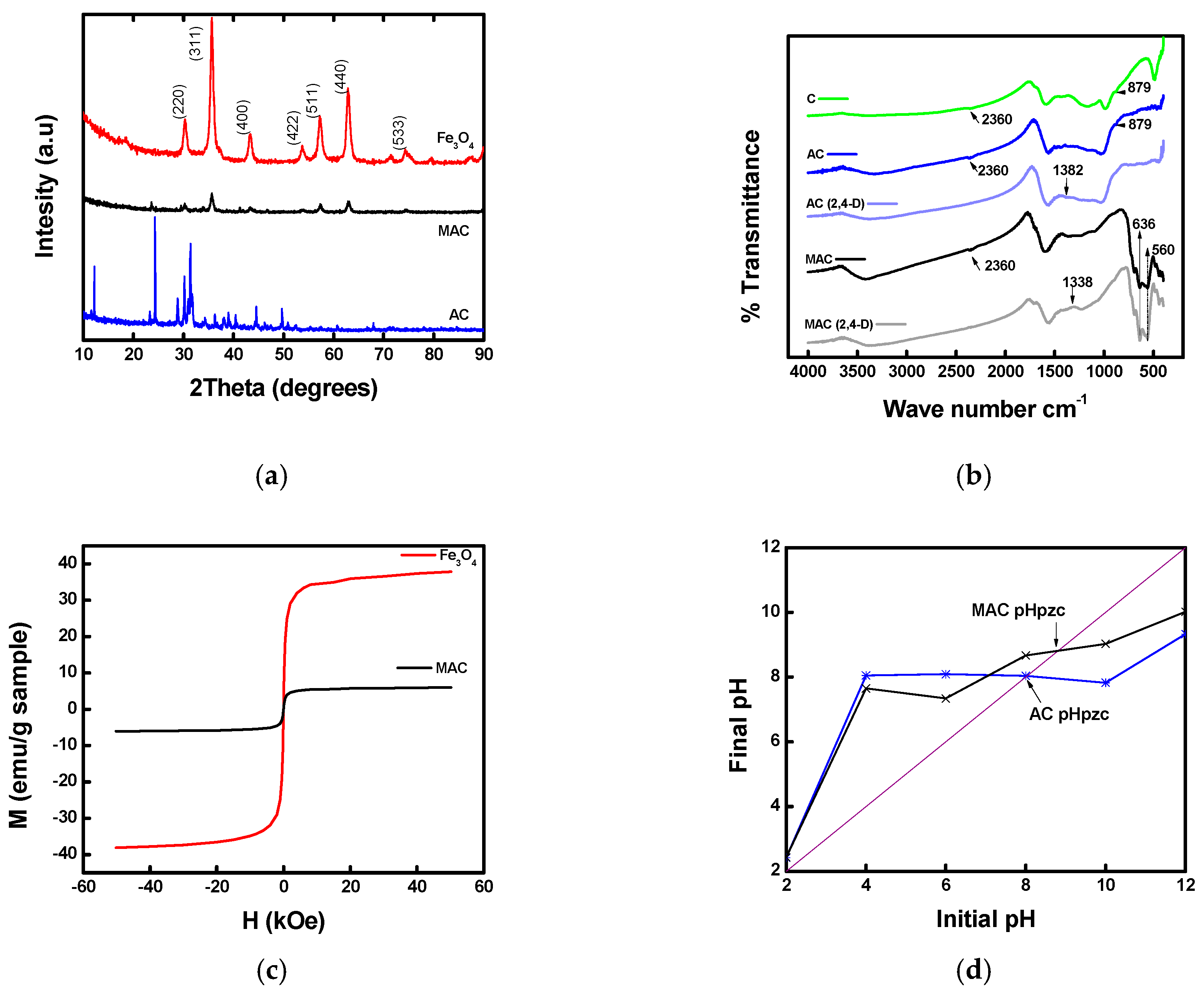
3.1.3. XRD
3.1.4. FTIR
3.1.5. VSM
3.1.6. Point of Zero Charge (pHpzc)
3.2. Adsorption Study
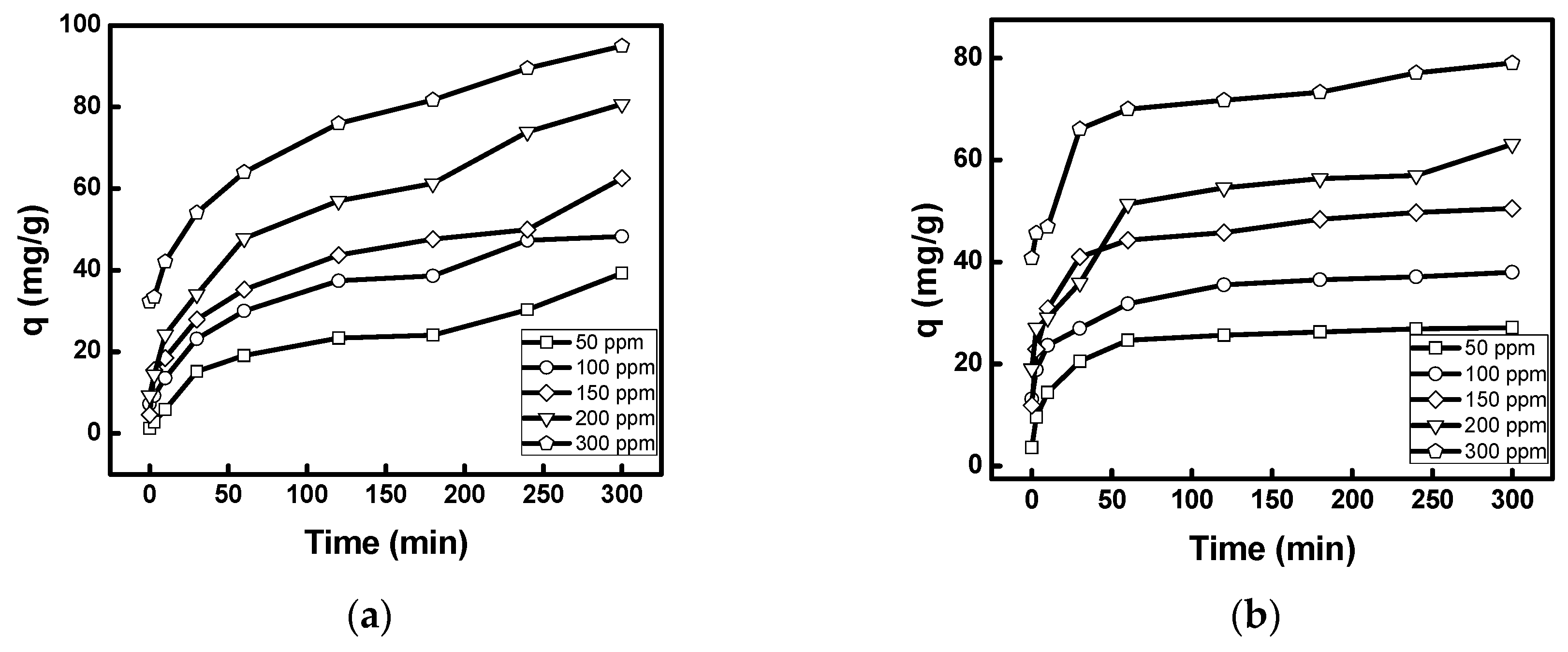
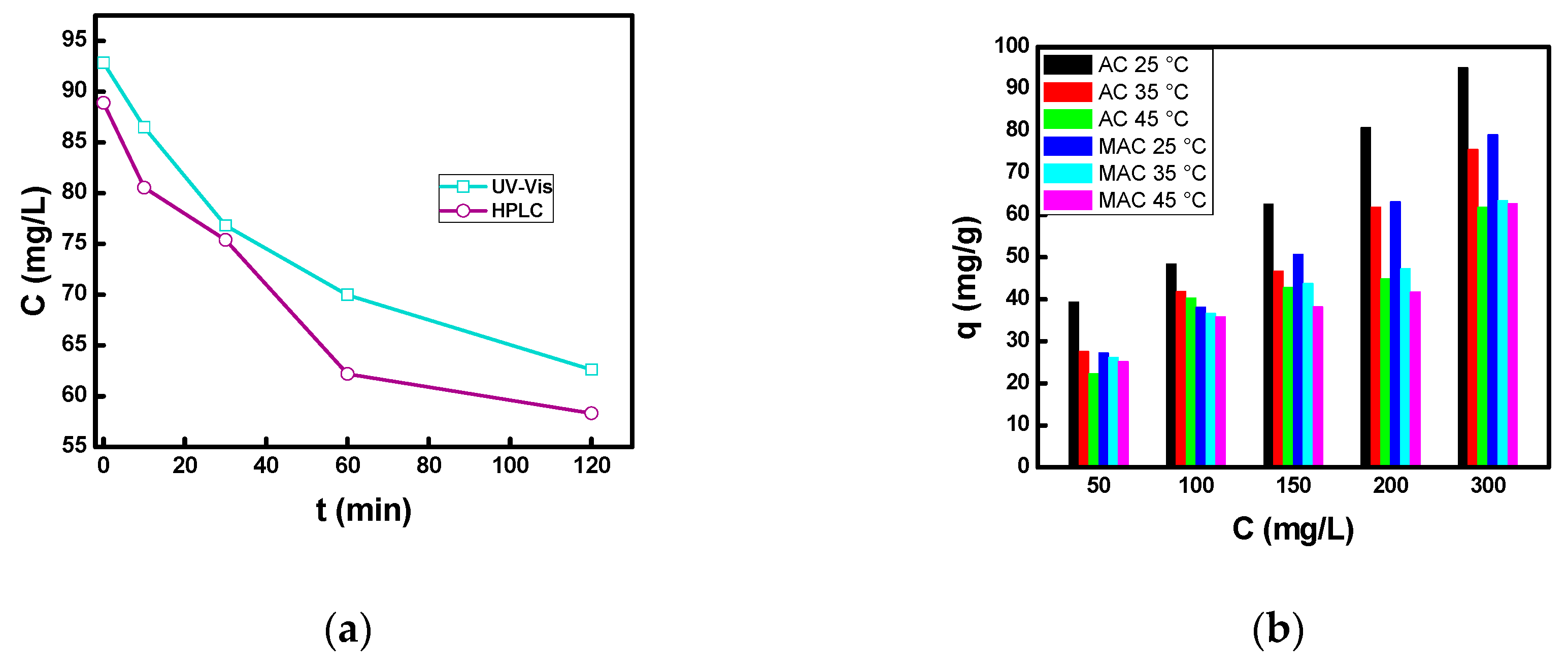
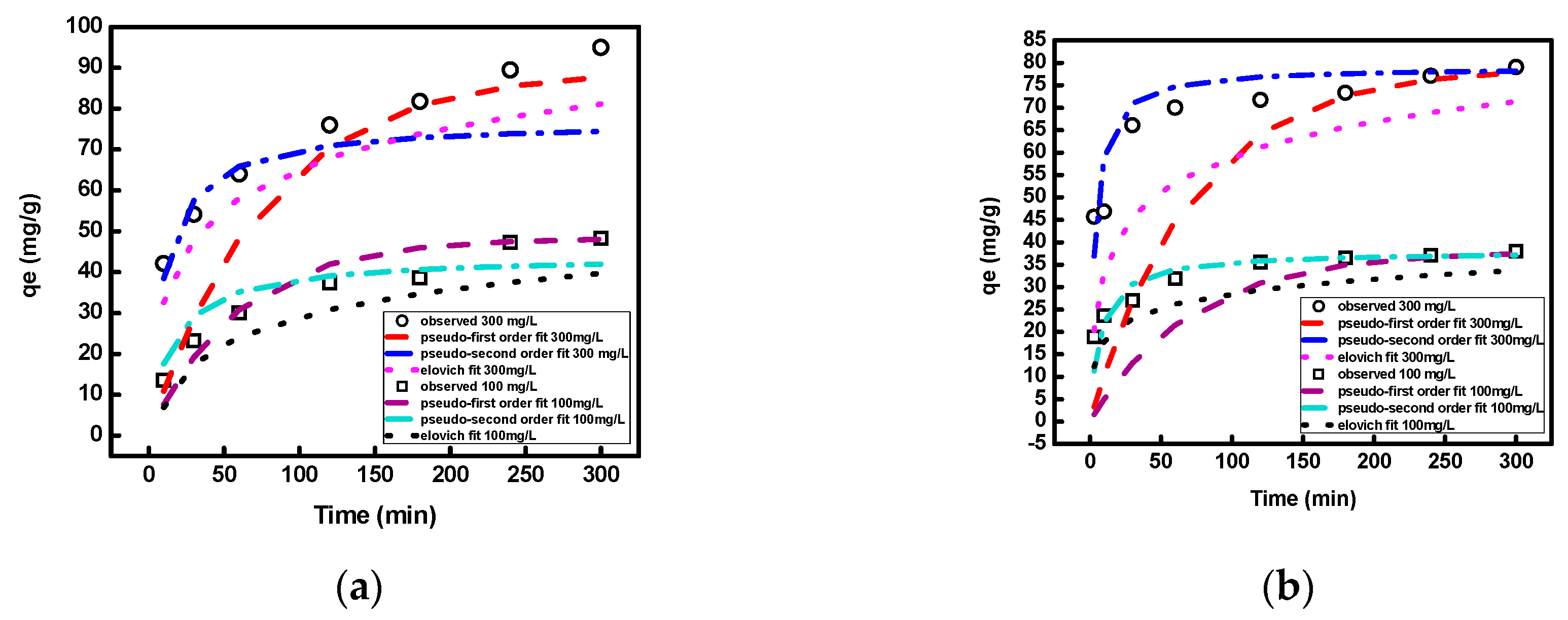
3.2.1. Effect of Contact Time, Initial Concentration, and Temperature
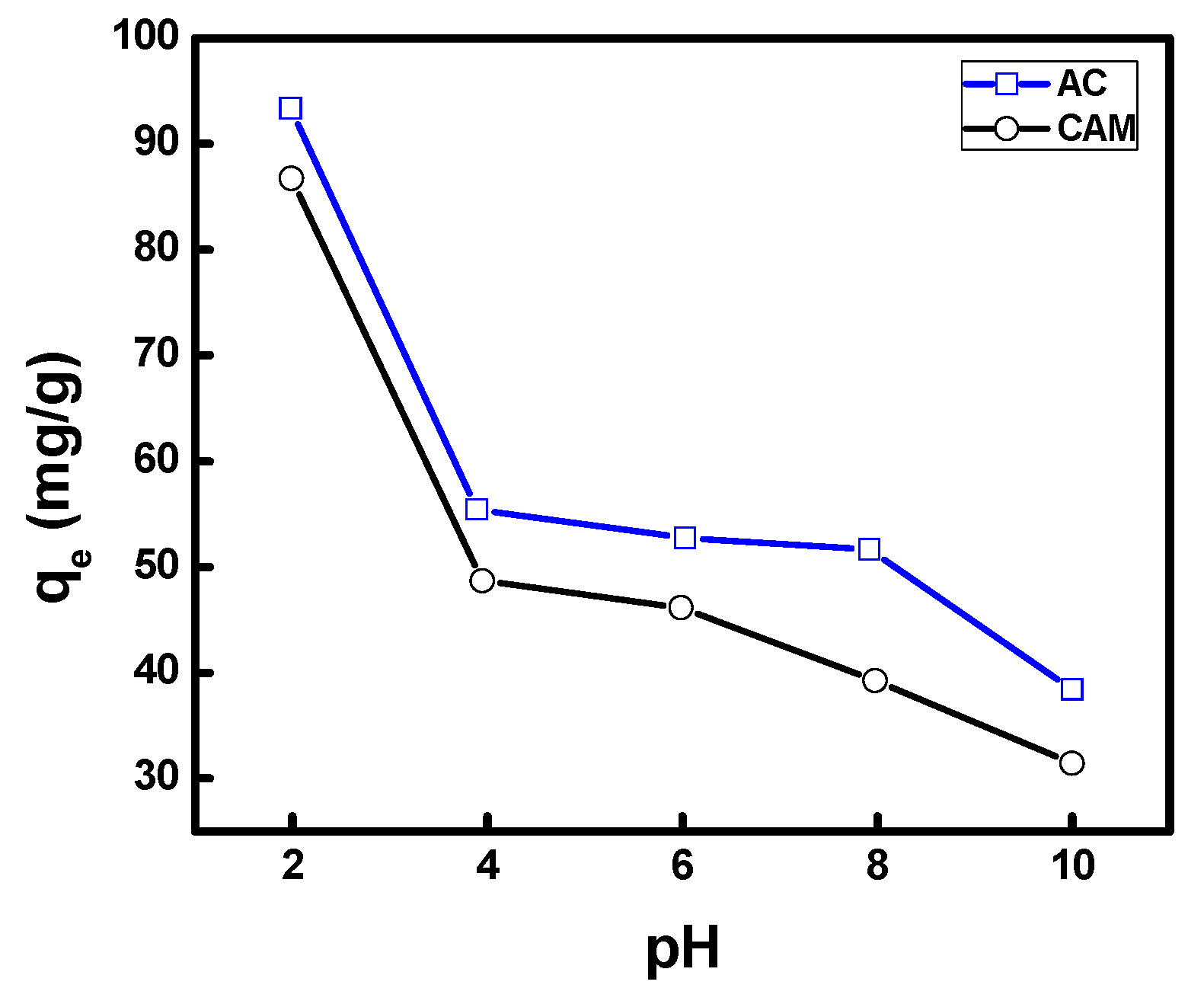
3.2.2. Effect of pH
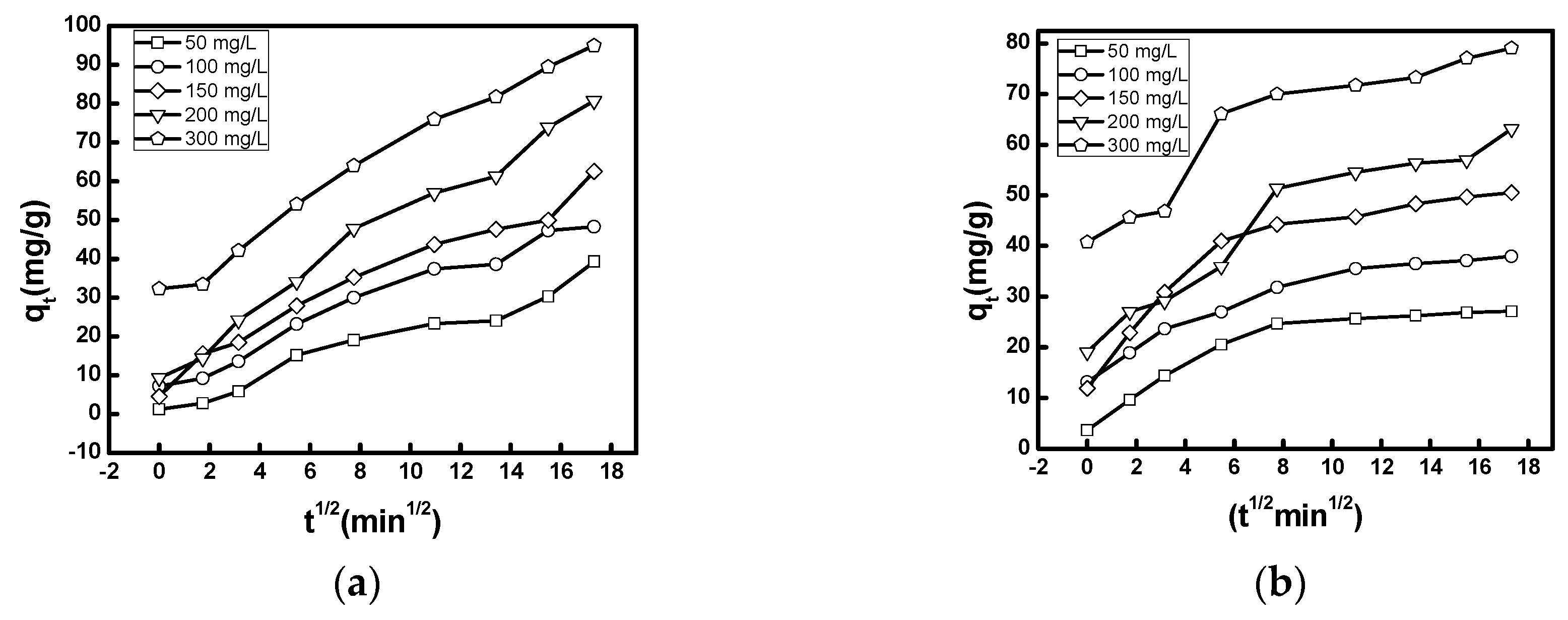
3.2.3. Kinetic Study
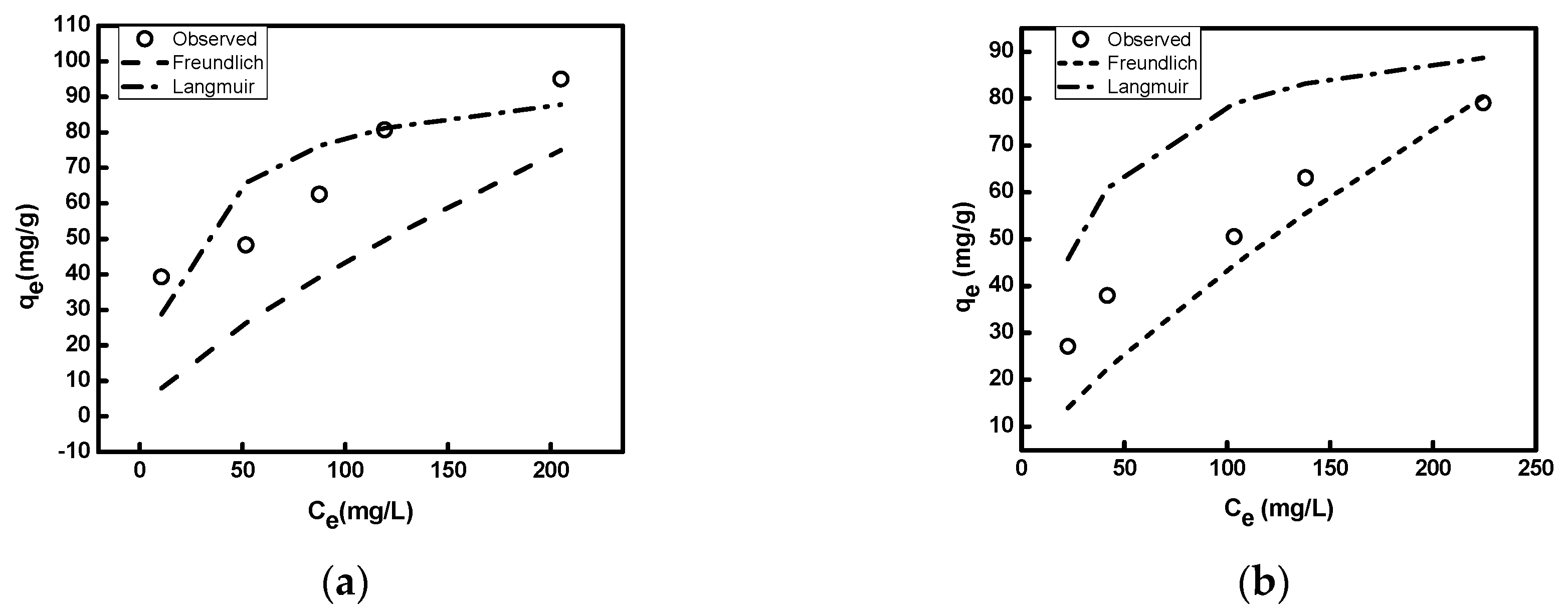
3.2.4. Adsorption Isotherms
3.2.5. Thermodynamic Study
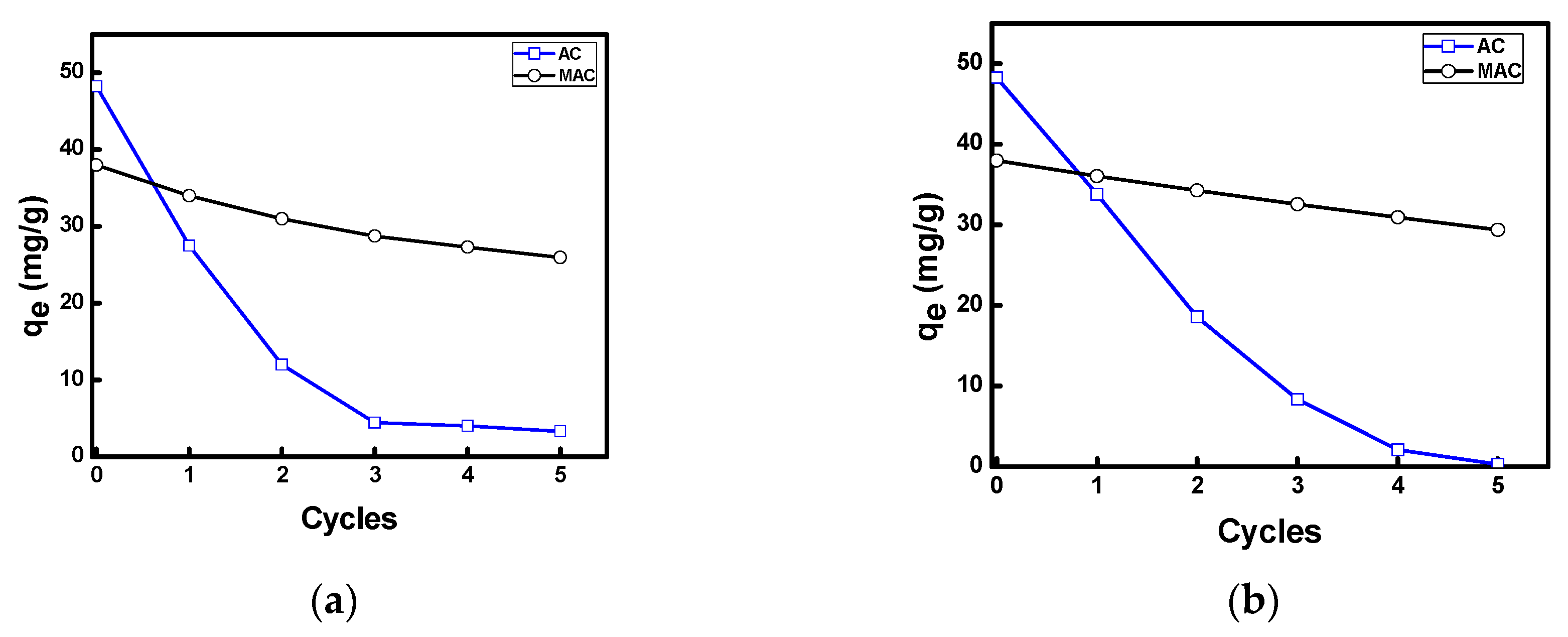
3.2.6. Regeneration Capacity of AC and MAC
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kim, K.H.; Kabir, E.; Jahan, S.A. Exposure to pesticides and the associated human health effects. Sci. Total Environ. 2017, 575, 525–535. [Google Scholar] [CrossRef] [PubMed]
- WHO. Preventing Disease Through Healthy Environments: Exposure To Highly Hazardous Pesticides-A Major Public Health Concern; WHO: Geneva, Switzerland, 2010. [Google Scholar]
- Arias-Estévez, M.; López-Periago, E.; Martínez-Carballo, E.; Simal-Gándara, J.; Mejuto, J.C.; García-Río, L. The mobility and degradation of pesticides in soils and the pollution of groundwater resources. Agric. Ecosyst. Environ. 2008, 123, 247–260. [Google Scholar] [CrossRef]
- Ghormade, V.; Deshpande, M.V.; Paknikar, K.M. Perspectives for nano-biotechnology enabled protection and nutrition of plants. Biotechnol. Adv. 2011, 29, 792–803. [Google Scholar] [CrossRef] [PubMed]
- Salman, J.M.; Hameed, B.H. Adsorption of 2,4-dichlorophenoxyacetic acid and carbofuran pesticides onto granular activated carbon. Desalination 2010, 256, 129–135. [Google Scholar] [CrossRef]
- Trivedi, N.S.; Kharkar, R.A.; Mandavgane, S.A. 2, 4-Dichlorophenoxyacetic acid adsorption on adsorbent prepared from groundnut shell: Effect of preparation conditions on equilibrium adsorption capacity. Arab. J. Chem. 2016, in press. [Google Scholar] [CrossRef]
- Trivedi, N.S.; Mandavgane, S.A.; Kulkarni, B.D. Mustard plant ash: A source of micronutrient and an adsorbent for removal of 2,4-dichlorophenoxyacetic acid. Environ. Sci. Pollut. Res. 2016, 23, 20087–20099. [Google Scholar] [CrossRef]
- Lozano, V.L.; Miranda, C.E.; Vinocur, A.L.; González, C.; Unrein, F.; Wolansky, M.J.; Pizarro, H.N. Turbidity matters: Differential effect of a 2,4-D formulation on the structure of microbial communities from clear and turbid freshwater systems. Heliyon 2019, 5, e02221. [Google Scholar] [CrossRef]
- Echeverry, G.; Zapata, A.M.; Paéz, M.I.; Méndez, F.; Peña, M.R. Valoración del riesgo en salud para un grupo poblacional de la ciudad de Cali-Colombia por exposición a Pb, Cd, Hg, 2-4D y Diuron debido al consumo de agua potable y alimentos. Biomédica 2015, 35, 110–119. [Google Scholar] [CrossRef]
- Qurratu, A.; Reehan, A. A review of 2,4-Dichlorophenoxyacetic acid (2,4-D) derivatives: 2,4-D dimethylamine salt and 2,4-D butyl ester. Int. J. Appl. Eng. Res. 2016, 11, 9946–9955. [Google Scholar]
- Sun, S.; Yu, Q.; Li, M.; Zhao, H.; Wu, C. Preparation of coffee-shell activated carbon and its application for water vapor adsorption. Renew. Energy 2019, 142, 11–19. [Google Scholar] [CrossRef]
- Mierzejewska, E.; Baran, A.; Tankiewicz, M.; Urbaniak, M. Removal and Ecotoxicity of 2,4-D and MCPA in Microbial Cultures Enriched with Structurally-Similar Plant Secondary Metabolites. Water 2019, 11, 1451. [Google Scholar] [CrossRef]
- Heydari, G.; Hollman, J.; Achari, G.; Langford, C.H. Comparative study of four TiO2-based photocatalysts to degrade 2,4-D in a semi-passive system. Water 2019, 11, 621. [Google Scholar] [CrossRef]
- Lota, K.; Acznik, I.; Sierczynska, A.; Lota, G. The capacitance properties of activated carbon obtained from chitosan as the electrode material for electrochemical capacitors. Mater. Lett. 2016, 173, 72–75. [Google Scholar] [CrossRef]
- Zhu, G.Z.; Deng, X.L.; Hou, M.; Sun, K.; Zhang, Y.P.; Li, P.; Liang, F.M. Comparative study on characterization and adsorption properties of activated carbons by phosphoric acid activation from corncob and its acid and alkaline hydrolysis residues. Fuel Process. Technol. 2016, 144, 255–261. [Google Scholar] [CrossRef]
- Shaarani, F.W.; Hameed, B.H. Batch adsorption of 2,4-dichlorophenol onto activated carbon derived from agricultural waste. Desalination 2010, 255, 159–164. [Google Scholar] [CrossRef]
- Vo, A.T.; Nguyen, V.P.; Ouakouak, A.; Nieva, A. Efficient Removal of Cr (VI) from Water by Biochar and Activated Carbon Prepared through Hydrothermal Carbonization and Pyrolysis: Adsorption-Coupled Reduction Mechanism. Water 2019, 11, 1164. [Google Scholar] [CrossRef]
- Njoku, V.; Azharul, M.; Asif, M.; Hameed, B. Adsorption of 2,4-dichlorophenoxyacetic acid by mesoporous activated carbon prepared from H3PO4-Activated langsat empty fruit. J. Environ. Manag. 2015, 154, 138–144. [Google Scholar] [CrossRef]
- Hameed, B.H.; Salman, J.M.; Ahmad, A.L. Adsorption isotherm and kinetic modeling of 2,4-D pesticide on activated carbon derived from date stones. J. Hazard. Mater. 2009, 163, 121–126. [Google Scholar] [CrossRef]
- Zhu, L.; Zhao, N.; Tong, L.; Lv, Y.; Li, G. Characterization and evaluation of surface modified materials based on porous biochar and its adsorption properties for 2,4-dichlorophenoxyacetic acid. Chemosphere 2018, 210, 734–744. [Google Scholar] [CrossRef]
- Kakavandi, B.; Jafari, A.J.; Kalantary, R.R.; Nasseri, S.; Ameri, A.; Esrafily, A. Synthesis and properties of Fe3O4-activated carbon magnetic nanoparticles for removal of aniline from aqueous solution: Equilibrium, kinetic and thermodynamic studies. Iran. J. Environ. Heal. Sci. Eng. 2013, 10, 2–10. [Google Scholar] [CrossRef]
- Yegane Badi, M.; Azari, A.; Pasalari, H.; Esrafili, A.; Farzadkia, M. Modification of activated carbon with magnetic Fe3O4 nanoparticle composite for removal of ceftriaxone from aquatic solutions. J. Mol. Liq. 2018, 261, 146–154. [Google Scholar] [CrossRef]
- Zhang, G.; Qu, J.; Liu, H.; Cooper, A.T.; Wu, R. CuFe2O4/activated carbon composite: A novel magnetic adsorbent for the removal of acid orange II and catalytic regeneration. Chemosphere 2007, 68, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Patricia, L.; Benítez, T.; Tovar, C.T.; Ortiz, A.V.; Dunoyer, A.T. Aprovechamiento del ñame espino (Dioscorea rotundata) en la producción de bioplásticos. Prospectiva 2008, 6, 68–74. [Google Scholar]
- Reina-Aranza, Y.C. El cultivo de ñame en el Caribe colombiano. Documentos de Trabajo Sobre Economía Regional Y Urbana; Banco de la Republica de Colombia: Bogotá, Colombia, 2012. [Google Scholar]
- Patiño-Ruiz, D.; Sanchez-Botero, L.; Hinestroza, J.; Herrera, A. Modification of Cotton Fibers with Magnetite and Magnetic Core-Shell Mesoporous Silica Nanoparticles. Phys. Status Solidi Appl. Mater. Sci. 2018, 215, 1–7. [Google Scholar] [CrossRef]
- Macías-Martínez, B.I.; Cortés-Hernández, D.A.; Zugasti-Cruz, A.; Cruz-Ortíz, B.R.; Múzquiz-Ramos, E.M. Heating ability and hemolysis test of magnetite nanoparticles obtained by a simple co-precipitation method. J. Appl. Res. Technol. 2016, 14, 239–244. [Google Scholar] [CrossRef]
- Zailani, R.; Ghafar, H.; So’aib, M.S. The influence of oxygen in the carbonization of oil palm shell on bio- char yield and properties. Appl. Mech. Mater. 2013, 393, 499–504. [Google Scholar] [CrossRef]
- Correia, R.; Gonçalves, M.; Nobre, C.; Mendes, B. Impact of torrefaction and low-temperature carbonization on the properties of biomass wastes from Arundo donax L. and Phoenix canariensis. Bioresour. Technol. 2017, 223, 210–218. [Google Scholar] [CrossRef]
- Bastami, T.R.; Entezari, M.H. Activated carbon from carrot dross combined with magnetite nanoparticles for the efficient removal of p-nitrophenol from aqueous solution. Chem. Eng. J. 2012, 210, 510–519. [Google Scholar] [CrossRef]
- Lagergren, S.; Sven, K. Zur Theorie der Sogenannten Adsorption Gelöster Stoffe, Kungliga Svenska Vetenskapsakademiens. Handlingar 1898, 24, 1–39. [Google Scholar]
- Ho, Y.S.; McKay, G. Sorption of dye from aqueous solution by peat. Chem. Eng. J. 1998, 70, 115–124. [Google Scholar] [CrossRef]
- Weber, W.J.; Morris, J.C.S. Advances in water pollution research: Removal of biologically resistant pollutant from waste water by adsorption. Proc. Int. Conf. Water Pollut. Symp. 1962, 2, 231–266. [Google Scholar]
- Li, Y.; Zhang, P.; Du, Q.; Peng, X.; Liu, T.; Wang, Z.; Xia, Y.; Zhang, W.; Wang, K.; Zhu, H.; et al. Adsorption of fluoride from aqueous solution by graphene. J. Colloid Interface Sci. 2011, 363, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Chen, X. Modeling of experimental adsorption isotherm data. Information 2015, 6, 14–22. [Google Scholar] [CrossRef]
- Freundlich, H. Adsorption in Solution. Z. für Phys. Chem. 1906, 57, 384–470. [Google Scholar]
- Farouq, R.; Yousef, N.S. Equilibrium and Kinetics Studies of adsorption of Copper (II) Ions on Natural Biosorbent. Int. J. Chem. Eng. Appl. 2015, 6, 319–324. [Google Scholar] [CrossRef]
- Salman, J.M.; Njoku, V.O.; Hameed, B.H. Adsorption of pesticides from aqueous solution onto banana stalk activated carbon. Chem. Eng. J. 2011, 174, 41–48. [Google Scholar] [CrossRef]
- Fu, Z.; Yan, L.; Li, K.; Ge, B.; Pu, L.; Zhang, X. The performance and mechanism of modified activated carbon air cathode by non-stoichiometric nano Fe in the microbial fuel cell. Biosens. Bioelectron. 2015, 74, 989–995. [Google Scholar] [CrossRef]
- Rai, P.; Gautam, R.K.; Banerjee, S.; Rawat, V.; Chattopadhyaya, M.C. Synthesis and characterization of a novel SnFe@activated carbon magnetic nanocomposite and its effectiveness in the removal of crystal violet from aqueous solution. J. Environ. Chem. Eng. 2015, 3, 2281–2291. [Google Scholar] [CrossRef]
- Mohan, D.; Sarswat, A.; Singh, V.K.; Alexandre-Franco, M.; Pittman, C.U. Development of magnetic activated carbon from almond shells for trinitrophenol removal from water. Chem. Eng. J. 2011, 172, 1111–1125. [Google Scholar] [CrossRef]
- Benz, M. Superparamagnetism: Theory and Applications. In Unpublished Manuscript; Libraries: Boston, MA, USA, 2012. [Google Scholar]
- Derylo-Marczewska, A.; Blachnio, M.; Marczewski, A.W.; Swiatkowski, A.; Tarasiuk, B. Adsorption of selected herbicides from aqueous solutions on activated carbon. J. Therm. Anal. Calorim. 2010, 101, 785–794. [Google Scholar] [CrossRef]
- Diaz-Flores, P.E.; Leyva-Ramos, R.; Guerrero-Coronado, R.M.; Mendoza-Barron, J. Adsorption of pentachlorophenol from aqueous solution onto activated carbon fiber. Ind. Eng. Chem. Res. 2006, 45, 330–336. [Google Scholar] [CrossRef]
- Goyne, K.W.; Chorover, J.; Zimmerman, A.R.; Komarneni, S.; Brantley, S.L. Influence of mesoporosity on the sorption of 2,4-dichlorophenoxyacetic acid onto alumina and silica. J. Colloid Interface Sci. 2004, 272, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Kuśmierek, K.; Zarȩbska, K.; Świątkowski, A. Hard coal as a potential low-cost adsorbent for removal of 4-chlorophenol from water. Water Sci. Technol. 2016, 73, 2025–2030. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.S.; Mckay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Vargas, M.; Diosa, J.E.; Mosquera, E. Data on study of hematite nanoparticles obtained from Iron (III) oxide by the Pechini method. Data Br. 2019, 25, 104183. [Google Scholar] [CrossRef]
- Ayranci, E.; Duman, O. Adsorption behaviors of some phenolic compounds onto high specific area activated carbon cloth. J. Hazard. Mater. 2005, 124, 125–132. [Google Scholar] [CrossRef]
- Bedin, K.C.; Souza, I.P.A.F.; Cazetta, A.L.; Ronix, A.; Almeida, V.C. CO2-spherical activated carbon as a new adsorbent for Methylene Blue removal: Kinetic, equilibrium and thermodynamic studies. J. Mol. Liq. 2018, 269, 132–139. [Google Scholar] [CrossRef]
- Fierro, V.; Torne, V. Adsorption of phenol onto activated carbons having different textural and surface properties. Microporous Mesoporous Mater. 2008, 111, 276–284. [Google Scholar] [CrossRef]
- Peng, Z.; Fang, Z.; Li, T.; Young, S. Adsorption isotherm, thermodynamics and kinetics studies of polyphenols separation from kiwifruit juice using adsorbent resin. J. Food Eng. 2013, 116, 195–201. [Google Scholar]
- Tauetsile, P.J.; Oraby, E.A.; Eksteen, J.J. Activated carbon adsorption of gold from cyanide-starved glycine solutions containing copper. Part 1: Isotherms. Sep. Purif. Technol. 2018, 211, 594–601. [Google Scholar] [CrossRef]
- Huang, J.; Deng, R.; Huang, K. Equilibria and kinetics of phenol adsorption on a toluene-modified hyper-cross-linked poly(styrene-co-divinylbenzene) resin. Chem. Eng. J. 2011, 171, 951–957. [Google Scholar] [CrossRef]
| Model | Lineal Form | |
|---|---|---|
| I | (8) | |
| II | (9) | |
| III | (10) | |
| IV | (11) | |
| Co (mg/L) | qe,exp (mg/g) | Pseudo-First Order | Pseudo-Second Order | ||||
| K1 (1/min) | qe | R2 | K2 (mg/g min) | qe | R2 | ||
| 50 | 27.50 | 0.012 | 27.51 | 0.94 | 0.0010 | 29.85 | 0.98 |
| 100 | 41.78 | 0.017 | 41.79 | 0.80 | 0.0015 | 44.05 | 0.99 |
| 150 | 46.63 | 0.013 | 46.64 | 0.97 | 0.0013 | 48.31 | 0.99 |
| 200 | 61.88 | 0.015 | 61.89 | 0.94 | 0.0008 | 65.36 | 0.99 |
| 300 | 75.51 | 0.013 | 75.50 | 0.99 | 0.0013 | 76.92 | 0.99 |
| Co (mg/L) | qe,exp (mg/g) | Elovich | Intraparticular Diffusion Model | ||||
| β (g/mg) | α (mg/g min) | R2 | Kp (mg/g min1/2) | Ci | R2 | ||
| 50 | 27.5 | 0.193 | 0.382 | 0.96 | 1.664 | 1.651 | 0.94 |
| 100 | 41.78 | 0.144 | 1.382 | 0.96 | 2.230 | 9.085 | 0.93 |
| 150 | 46.63 | 0.121 | 0.914 | 0.99 | 2.565 | 8.168 | 0.91 |
| 200 | 61.88 | 0.093 | 0.983 | 0.97 | 3.430 | 10.391 | 0.94 |
| 300 | 75.51 | 0.107 | 9.739 | 0.97 | 3.009 | 29.646 | 0.95 |
| Co (mg/L) | qe,exp (mg/g) | Pseudo-First Order | Pseudo-Second Order | ||||
| K1 (1/min) | qe | R2 | K2 (mg/g min) | qe | R2 | ||
| 50 | 27.14 | 0.012 | 27.143 | 0.78 | 0.0054 | 27.55 | 0.99 |
| 100 | 37.99 | 0.011 | 37.991 | 0.92 | 0.0037 | 38.31 | 0.99 |
| 150 | 50.55 | 0.012 | 50.552 | 0.92 | 0.0032 | 50.76 | 0.99 |
| 200 | 63.11 | 0.001 | 63.113 | 0.91 | 0.0014 | 62.11 | 0.99 |
| 300 | 79.05 | 0.011 | 79.058 | 0.94 | 0.0022 | 78.74 | 0.99 |
| Co (mg/L) | qe,exp (mg/g) | Elovich | Intraparticular Diffusion Model | ||||
| β (g/mg) | α (mg/g min) | R2 | Kp (mg/g min1/2) | Ci | R2 | ||
| 50 | 27.14 | 0.2381 | 2.99 | 0.98 | 1.245 | 9.47 | 0.81 |
| 100 | 37.99 | 0.2281 | 21.36 | 0.99 | 1.357 | 17.73 | 0.90 |
| 150 | 50.55 | 0.1519 | 9.48 | 0.98 | 1.953 | 22.03 | 0.81 |
| 200 | 63.11 | 0.1304 | 8.38 | 0.95 | 2.464 | 23.11 | 0.93 |
| 300 | 79.05 | 0.1411 | 21.78 | 0.95 | 2.207 | 44.94 | 0.87 |
| Isotherm Model | AC | MAC | |||||
|---|---|---|---|---|---|---|---|
| T (°C) | 25 | 35 | 45 | 25 | 35 | 45 | |
| Langmuir | KL (L/mg) | 0.0382 | 0.0266 | 0.0291 | 0.0223 | 0.0309 | 0.0281 |
| qm (mg/g) | 99.0099 | 80.0000 | 63.2911 | 86.2069 | 64.5161 | 60.9756 | |
| R2 | 0.9213 | 0.9042 | 0.9187 | 0.8801 | 0.9279 | 0.8755 | |
| Freundlich | Kf (mg/g)/(mg/L)1/n | 1.3067 | 1.1202 | 1.0970 | 1.1021 | 1.1257 | 1.2108 |
| nf | 1.3144 | 1.2469 | 1.2341 | 1.3519 | 1.3215 | 1.2296 | |
| R2 | 0.8761 | 0.9596 | 0.9633 | 0.9694 | 0.9518 | 0.9493 | |
| Elovich | KE | 0.1801 | 0.0513 | 0.0491 | 0.0333 | 0.0930 | 0.0550 |
| qm (mg/g) | 34.6021 | 39.6825 | 32.6797 | 49.0196 | 26.1780 | 29.9401 | |
| R2 | 0.6927 | 0.8477 | 0.7408 | 0.8469 | 0.8672 | 0.6321 | |
| Langmuir II | KLII (L/mg) | 0.0936 | 0.0251 | 0.0162 | 0.0221 | 0.0316 | 0.0342 |
| qm (mg/g) | 75.7576 | 74.6269 | 74.6269 | 78.1250 | 59.5238 | 53.7634 | |
| R2 | 0.7309 | 0.9426 | 0.9519 | 0.9197 | 0.9371 | 0.8743 | |
| Langmuir III | KLIII (L/mg) | 0.0808 | 0.0221 | 0.0199 | 0.0189 | 0.0281 | 0.0304 |
| qm (mg/g) | 81.0900 | 79.4410 | 66.7290 | 84.6980 | 62.3777 | 56.7530 | |
| R2 | 0.5192 | 0.7622 | 0.7600 | 0.7192 | 0.7625 | 0.5790 | |
| Langmuir IV | KLIV (L/mg) | 0.0420 | 0.0168 | 0.0147 | 0.0136 | 0.0214 | 0.0176 |
| qm (mg/g) | 95.7786 | 88.5714 | 75.3605 | 97.4853 | 67.9206 | 68.4943 | |
| R2 | 0.5192 | 0.7622 | 0.0147 | 0.7192 | 0.7625 | 0.5790 | |
| AC | MAC | |||||
|---|---|---|---|---|---|---|
| Co (mg/L) | 100 | 200 | 300 | 100 | 200 | 300 |
| ∆H (J/mol) | −6837.49 | −10.402.47 | −712.28 | −1379.12 | −5760.18 | −2849.45 |
| R2 | 0.7775 | 0.9979 | 0.9897 | 0.9872 | 0.9293 | 0.7977 |
| AC | MAC | |||||
|---|---|---|---|---|---|---|
| T (°C) | 25 | 35 | 45 | 25 | 35 | 45 |
| ∆G (J/mol) | −491.75 | −362.83 | −256.51 | −505.78 | −384.55 | −255.56 |
| ∆S (J/mol K) | ||||||
|---|---|---|---|---|---|---|
| Co (mg/L) | AC | MAC | ||||
| 25 (°C) | 35 (°C) | 45 (°C) | 25 (°C) | 35 (°C) | 45 (°C) | |
| 100 | −21.2587 | −20.9876 | −20.6624 | −2.9258 | −3.1227 | −3.2239 |
| 200 | −33.2017 | −32.5434 | −31.8555 | −16.8780 | −17.4251 | −17.6027 |
| 300 | −0.7388 | −1.1327 | −1.4310 | −7.8515 | −7.9900 | −7.991 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herrera-García, U.; Castillo, J.; Patiño-Ruiz, D.; Solano, R.; Herrera, A. Activated Carbon from Yam Peels Modified with Fe3O4 for Removal of 2,4-Dichlorophenoxyacetic Acid in Aqueous Solution. Water 2019, 11, 2342. https://doi.org/10.3390/w11112342
Herrera-García U, Castillo J, Patiño-Ruiz D, Solano R, Herrera A. Activated Carbon from Yam Peels Modified with Fe3O4 for Removal of 2,4-Dichlorophenoxyacetic Acid in Aqueous Solution. Water. 2019; 11(11):2342. https://doi.org/10.3390/w11112342
Chicago/Turabian StyleHerrera-García, Udualdo, Jefferson Castillo, David Patiño-Ruiz, Ricardo Solano, and Adriana Herrera. 2019. "Activated Carbon from Yam Peels Modified with Fe3O4 for Removal of 2,4-Dichlorophenoxyacetic Acid in Aqueous Solution" Water 11, no. 11: 2342. https://doi.org/10.3390/w11112342
APA StyleHerrera-García, U., Castillo, J., Patiño-Ruiz, D., Solano, R., & Herrera, A. (2019). Activated Carbon from Yam Peels Modified with Fe3O4 for Removal of 2,4-Dichlorophenoxyacetic Acid in Aqueous Solution. Water, 11(11), 2342. https://doi.org/10.3390/w11112342





