Tintinnid Ciliate Communities in Pre- and Post-Winter Conditions in the Southern Adriatic Sea (NE Mediterranean)
Abstract
:1. Introduction
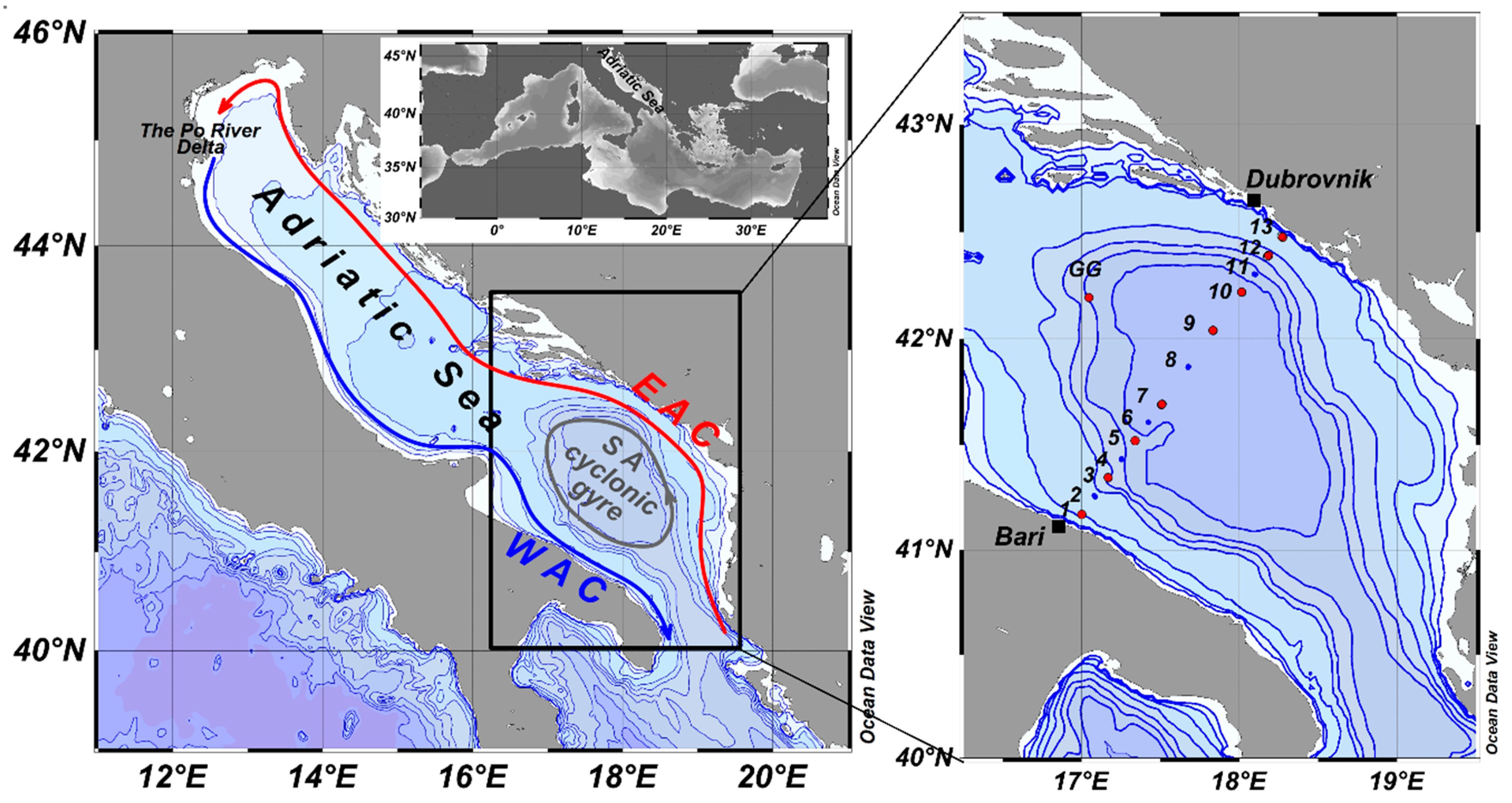
2. Material and Methods
3. Results
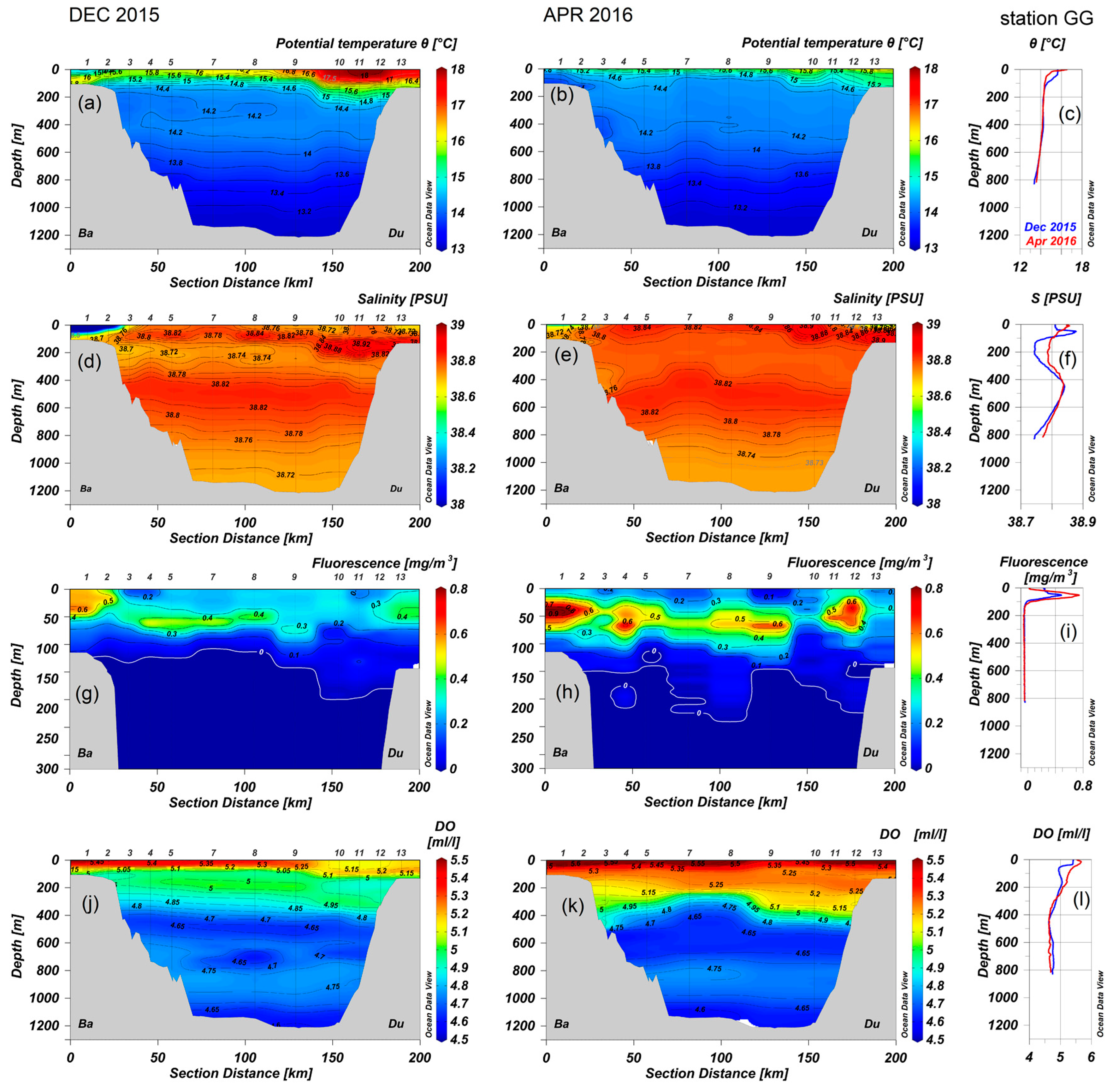
3.1. Hydrography, Dissolved Oxygen and Chlorophyll A
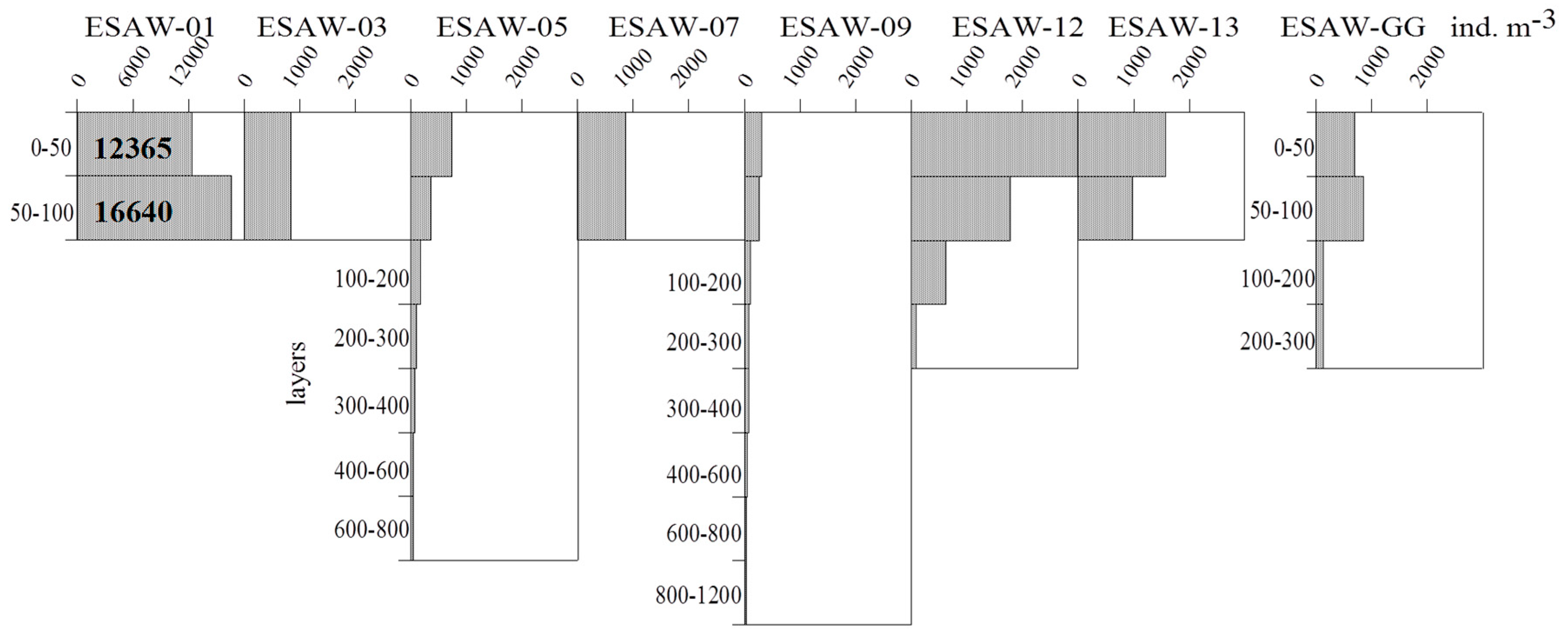
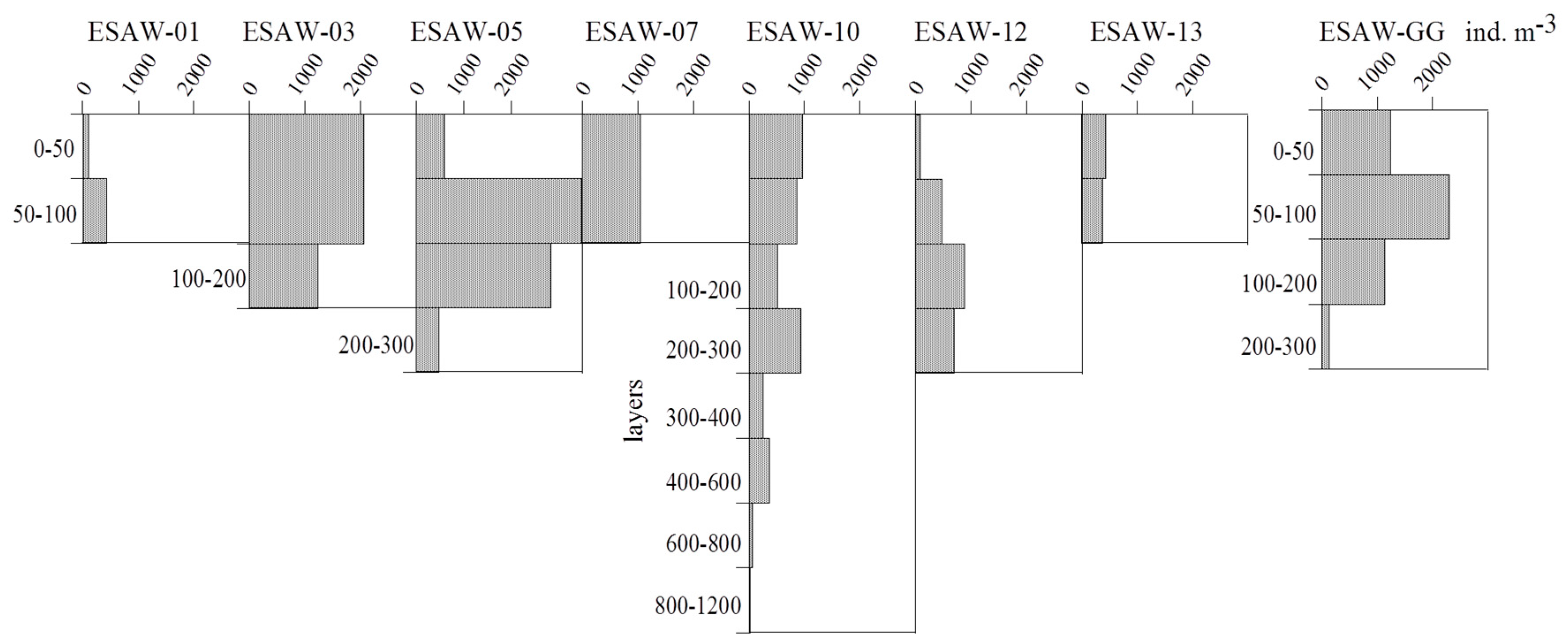
3.2. Vertical and Horizontal Distribution of Total Tintinnid Abundance
3.3. Tintinnid Species Composition and Distribution
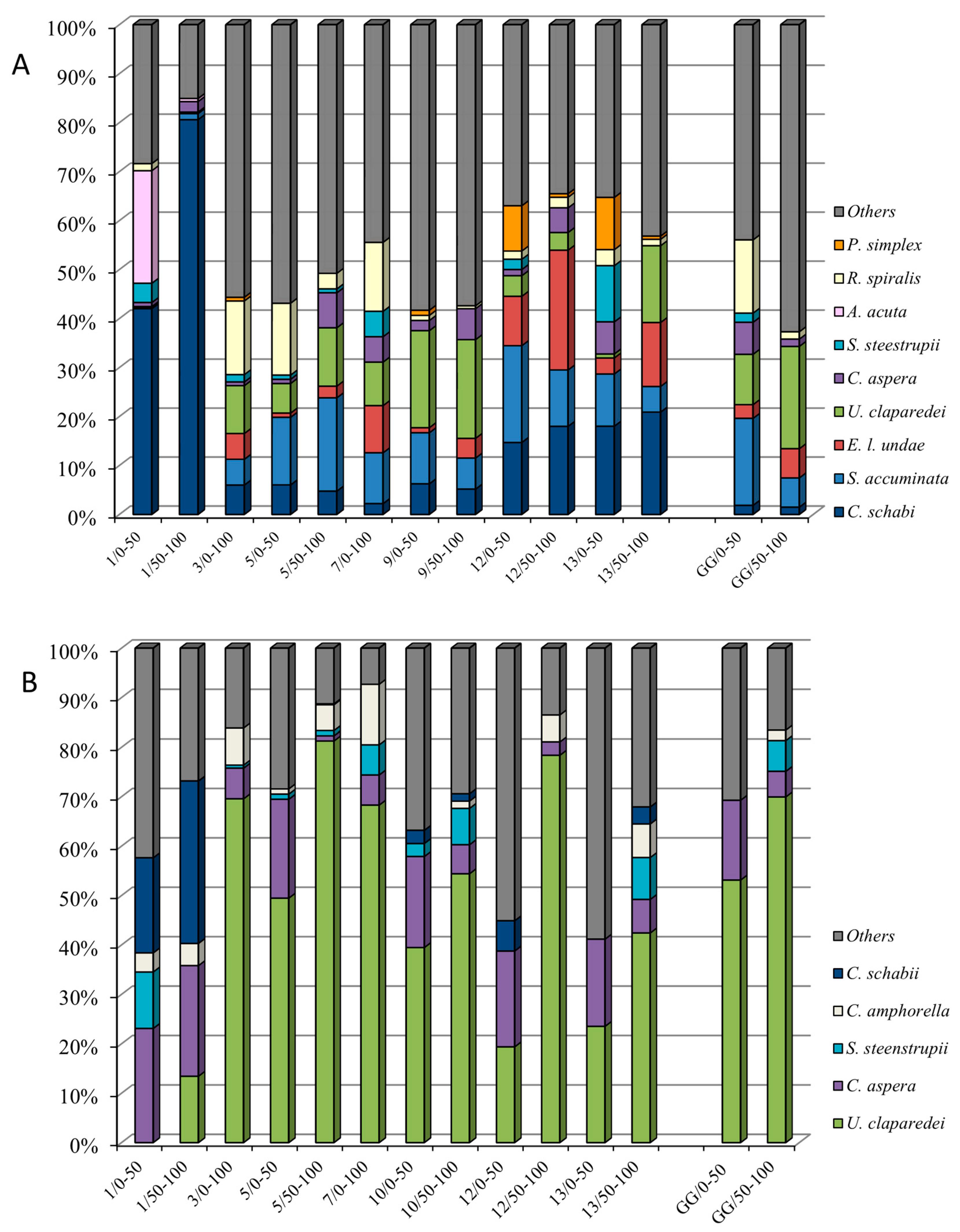
3.4. Dominant Species in the Surface and Subsurface Layers (0–100 m)
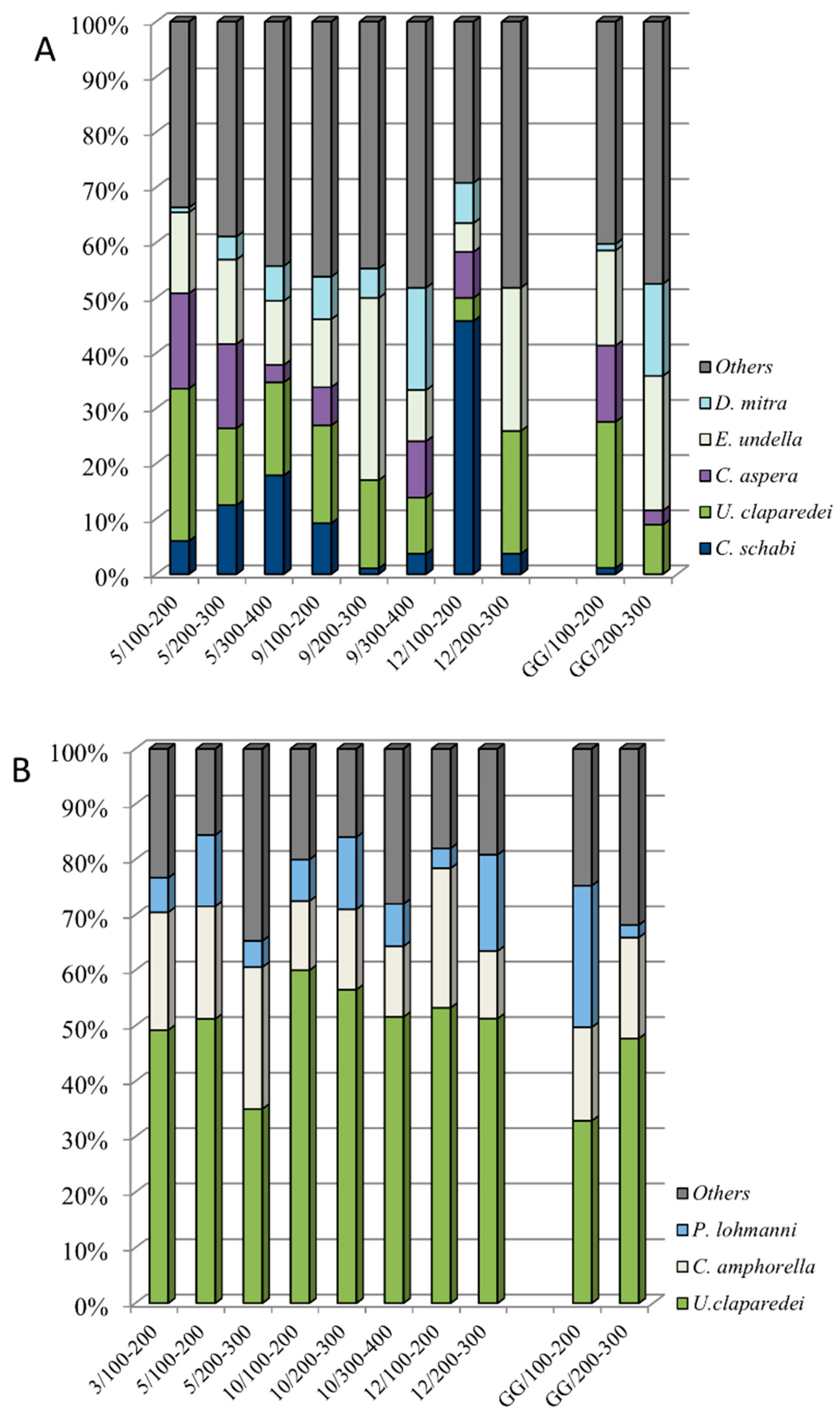
3.5. Dominant Species in the Mesopelagic Layers (100–300/400 m)
3.6. Dominant Species in the Deep Sea Layers (400–1200 m)
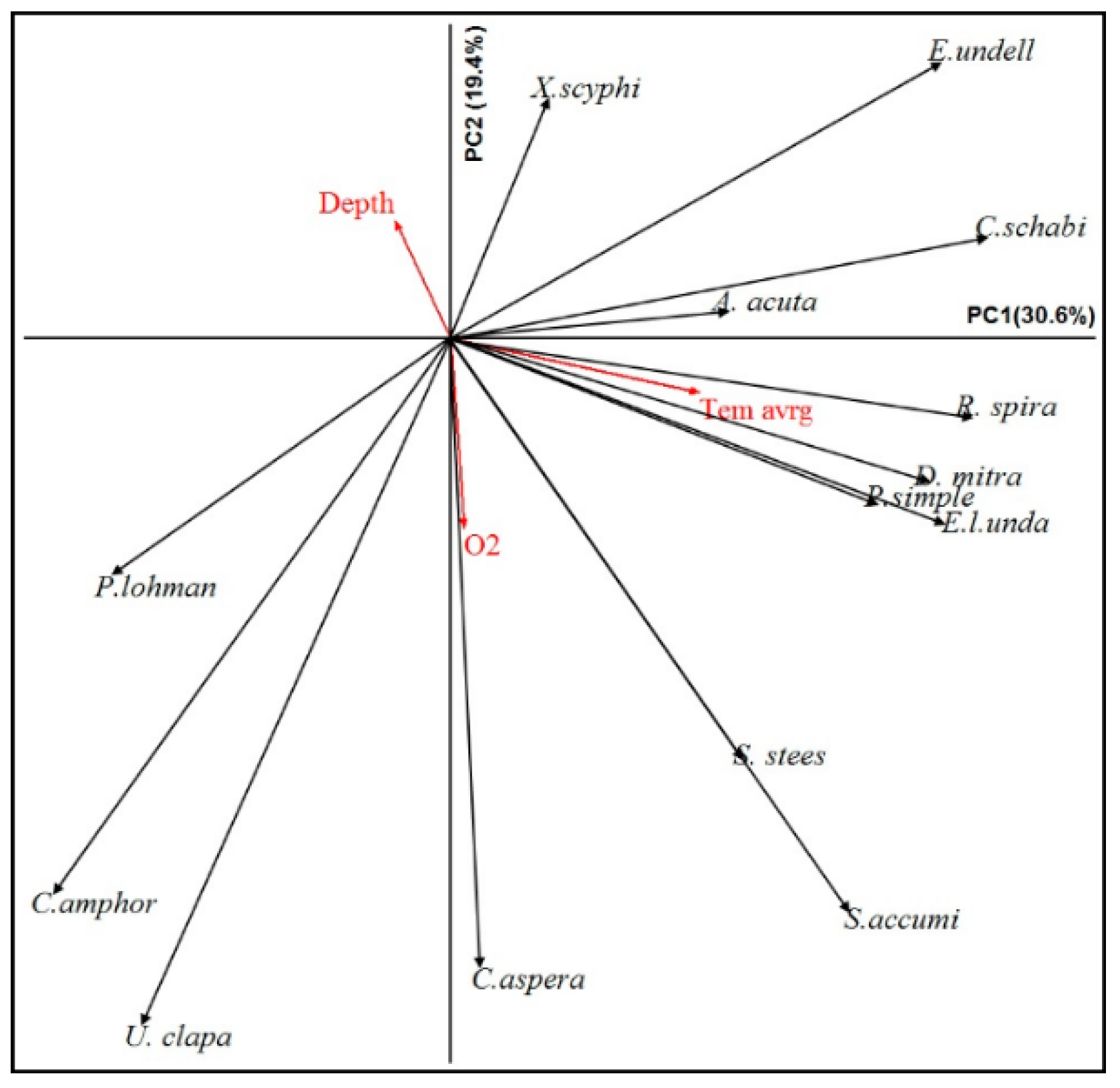
3.7. Relationship between Dominant Species and Environmental Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pierce, R.W.; Turner, J.T. Ecology of planktonic ciliates in marine food webs. Rev. Aquat. Sci. 1992, 6, 139–181. [Google Scholar]
- Pierce, R.; Turner, J. Global biogeography of marine tintinnids. Mar. Ecol. Prog. Ser. 1993, 94, 11–26. [Google Scholar] [CrossRef]
- Rassoulzadegan, F.; Laval-Peuto, M.; Sheldon, R.W. Partitioning of the food ration of marine ciliates between pico- and nanoplankton. Hydrobiologia 1988, 159, 75–88. [Google Scholar] [CrossRef]
- Bernard, C.; Rassoulzadegan, F. The role of picoplankton (cyanobacteria and plastidic picoflagellates) in the diet of tintinnids. J. Plankton Res. 1993, 15, 361–373. [Google Scholar] [CrossRef]
- Stoecker, D.K.; Sanders, N.K. Differential grazing by Acartia tonsa on a dinoflagellate and a tintinnid. J. Plankton Res. 1985, 7, 85–100. [Google Scholar] [CrossRef]
- Sherr, E.B.; Sherr, B.F.; Fallon, R.D.; Newell, S.Y. Small, aloricate ciliates as a major component of the marine heterotrophic nanoplankton. Limnol. Oceanogr. 1986, 31, 177–183. [Google Scholar] [CrossRef]
- Paffenhöfer, G.-A. On the relation of structure, perception and activity in marine planktonic copepods. J. Mar. Syst. 1998, 15, 457–473. [Google Scholar] [CrossRef]
- Thompson, G.A.; Alder, V.A.; Boltovskoy, D.; Brandini, F. Abundance and biogeography of tintinnids (Ciliophora) and associated microzooplankton in the Southwestern Atlantic Ocean. J. Plankton Res. 1999, 21, 1265–1298. [Google Scholar] [CrossRef] [Green Version]
- Dolan, J.R. Tintinnid ciliate diversity in the Mediterranean Sea: Longitudinal patterns related to water column structure in late spring-early summer. Aquat. Microb. Ecol. 2000, 22, 69–78. [Google Scholar] [CrossRef]
- Kršinić, F. Summer distributions of Tintinnids in open waters of the Adriatic Sea. Rapp. Procès-Verbaux La Réun. Comm. Int. Pour Explor. Sci. Mer Mediterr. 1979, 25/26, 107–108. [Google Scholar]
- Kršinić, F. On vertical distribution of tintinnines (Ciliata, Oligotrichida, Tintinnina) in the open waters of the South Adriatic. Mar. Biol. 1982, 68, 83–90. [Google Scholar] [CrossRef]
- Kršinić, F. Vertical distribution of protozoan and microcopepod communities in the South Adriatic Pit. J. Plankton Res. 1998, 20, 1033–1060. [Google Scholar] [CrossRef]
- Kršinić, F.; Grbec, B. Some distributional characteristics of small zooplankton at two stations in the Otranto Strait (Eastern Mediterranean). Hydrobiologia 2002, 482, 119–136. [Google Scholar] [CrossRef]
- Kršinić, F.; Grbec, B. Horizontal distribution of tintinnids in the open waters of the South Adriatic (Eastern Mediterranean). Sci. Mar. 2006, 70, 77–88. [Google Scholar] [CrossRef]
- Dolan, J.R.; Claustre, H.; Carlotti, F.; Plounevez, S.; Moutin, T. Microzooplankton diversity: Relationships of tintinnid ciliates with resources, competitors and predators from the Atlantic Coast of Morocco to the Eastern Mediterranean. Deep Sea Res. Part I: Oceanogr. Res. Pap. 2002, 49, 1217–1232. [Google Scholar] [CrossRef]
- Dolan, J.R.; Ciobanu, M.; Marro, S.; Coppola, L. An exploratory study of heterotrophic protists of the mesopelagic Mediterranean Sea. ICES J. Mar. Sci. 2019, 76, 616–625. [Google Scholar] [CrossRef]
- Manca, B.B.; Kovačević, V.; Gačić, M.; Viezzoli, D. Dense water formation in the Southern Adriatic Sea and spreading into the Ionian Sea in the period 1997–1999. MATER MAss Transf. Ecosyst. Response 2002, 33–34, 133–154. [Google Scholar] [CrossRef]
- Williams, R.G.; Follows, M.J. The Ekman transfer of nutrients and maintenance of new production over the North Atlantic. Deep Sea Res. Part Oceanogr. Res. Pap. 1998, 45, 461–489. [Google Scholar] [CrossRef]
- Marra, J.; Houghton, R.W.; Garside, C. Phytoplankton growth at the shelf-break front in the Middle Atlantic Bight. J. Mar. Res. 1990, 48, 851–868. [Google Scholar] [CrossRef]
- Civitarese, G.; Gačić, M. Had the Eastern Mediterranean Transient an impact on the new production in the southern Adriatic? Geophys. Res. Lett. 2001, 28, 1627–1630. [Google Scholar] [CrossRef]
- Gačić, M.; Civitarese, G.; Miserocchi, S.; Cardin, V.; Crise, A.; Mauri, E. The open-ocean convection in the Southern Adriatic: A controlling mechanism of the spring phytoplankton bloom. Cont. Shelf Res. 2002, 22, 1897–1908. [Google Scholar] [CrossRef]
- Bernardi Aubry, F.; Falcieri, F.M.; Chiggiato, J.; Boldrin, A.; Luna, G.M.; Finotto, S.; Camatti, E.; Acri, F.; Sclavo, M.; Carniel, S.; et al. Massive shelf dense water flow influences plankton community structure and particle transport over long distance. Sci. Rep. 2018, 8, 4554. [Google Scholar] [CrossRef]
- Zore, M. On the gradient currents in the Adriatic Sea. Acta Adriat. 1956, 8, 1–38. [Google Scholar]
- Grbec, B.; Dulčić, J.; Morović, M. Long-Term changes in landings of small pelagic fish in the eastern Adriatic-Possible influence of climate oscillations over the Northern Hemisphere. Clim. Res. 2002, 20, 241–252. [Google Scholar]
- Gačić, M.; Marullo, S.; Santoleri, R.; Bergamasco, A. Analysis of the seasonal and interannual variability of the sea surface temperature field in the Adriatic Sea from AVHRR data (1984–1992). J. Geophys. Res. Oceans 1997, 102, 22937–22946. [Google Scholar] [CrossRef]
- Hopkins, T.; Artegiani, A.; Kinder, C.; Pariante, R. A discussion of the northern Adriatic circulation and flushing as determined from the ELNA hydrography. In The Adriatic Sea, Ecosystem Research Report; Hopkins, T.S., Artegiani, A., Cauwet, G., Degobbis, D., Malej, A., Eds.; European Commission: Brussels, Belgium, 1999; Volume 32, pp. 85–106. [Google Scholar]
- Civitarese, G.; Gačić, M.; Lipizer, M.; Eusebi Borzelli, G.L. On the impact of the Bimodal Oscillating System (BiOS) on the biogeochemistry and biology of the Adriatic and Ionian Seas (Eastern Mediterranean). Biogeosciences 2010, 7, 3987–3997. [Google Scholar] [CrossRef] [Green Version]
- Batistić, M.; Viličić, D.; Kovačević, V.; Jasprica, N.; Garić, R.; Lavigne, H.; Carić, M. Occurrence of winter phytoplankton bloom in the open southern Adriatic: Relationship with hydroclimatic events in the Eastern Mediterranean. Cont. Shelf Res. 2019, 174, 12–25. [Google Scholar] [CrossRef]
- Gačić, M.; Borzelli, G.L.E.; Civitarese, G.; Cardin, V.; Yari, S. Can internal processes sustain reversals of the ocean upper circulation? The Ionian Sea example. Geophys. Res. Lett. 2010, 37. [Google Scholar] [CrossRef]
- Vilibić, I.; Matijević, S.; Šepić, J.; Kušpilić, G. Changes in the Adriatic oceanographic properties induced by the Eastern Mediterranean Transient. Biogeosciences 2012, 9, 2085–2097. [Google Scholar] [CrossRef] [Green Version]
- Batistić, M.; Jasprica, N.; Carić, M.; Čalić, M.; Kovačević, V.; Garić, R.; Njire, J.; Mikuš, J.; Bobanović-Ćolić, S. Biological evidence of a winter convection event in the South Adriatic: A phytoplankton maximum in the aphotic zone. South. Adriat. Oceanogr. 2012, 44, 57–71. [Google Scholar] [CrossRef]
- Batistić, M.; Garić, R.; Molinero, J.C. Interannual variations in Adriatic Sea zooplankton mirror shifts in circulation regimes in the Ionian Sea. Clim. Res. 2014, 61, 231–240. [Google Scholar] [CrossRef]
- Ljubimir, S.; Jasprica, N.; Čalić, M.; Hrustić, E.; Dupčić Radić, I.; Car, A.; Batistić, M. Interannual (2009–2013) variability of winter-spring phytoplankton in the open South Adriatic Sea: Effects of deep convection and lateral advection. Cont. Shelf Res. 2017, 143, 311–321. [Google Scholar] [CrossRef]
- Lučić, D.; Ljubešić, Z.; Babić, I.; Bosak, S.; Cetinić, I.; Vilibić, I.; Mihanović, H.; Hure, M.; Njire, J.; Lučić, P.; et al. Unusual winter zooplankton bloom in the open southern Adriatic Sea. Turk. J. Zool. 2017, 41, 1024–1035. [Google Scholar] [CrossRef]
- Čalić, M.; Ljubimir, S.; Bosak, S.; Car, A. First records of two planktonic Indo-Pacific diatoms: Chaetoceros bacteriastroides and C. pseudosymmetricus in the Adriatic Sea. Oceanologia 2018, 60, 101–105. [Google Scholar] [CrossRef]
- Hure, M.; Mihanović, H.; Lučić, D.; Ljubešić, Z.; Kružić, P. Mesozooplankton spatial distribution and community structure in the South Adriatic Sea during two winters (2015, 2016). Mar. Ecol. 2018, 39, e12488. [Google Scholar] [CrossRef]
- Kršinić, F. A new type of zooplankton sampler. J. Plankton Res. 1990, 12, 337–343. [Google Scholar] [CrossRef]
- Lynn, D. The Ciliated Protozoa. Characterization, Classification and Guide to the Literature, 3rd ed.; Springer Netherlands: Dordrecht, The Netherlands, 2008. [Google Scholar]
- Kršinić, F. Tintinnids (Tintinnida, Choreotrichia, Ciliata) in the Adriatic Sea, Mediterranean. Part, I. Taxonomy; Institute of Oceanography and Fisheries: Split, Croatia, 2010. [Google Scholar]
- Vilibić, I.; Orlić, M. Adriatic water masses, their rates of formation and transport through the Otranto Strait. Deep Sea Res. Part Oceanogr. Res. Pap. 2002, 49, 1321–1340. [Google Scholar] [CrossRef]
- Abbound-Abi Saab, M. Distribution and ecology of tintinnids in the plankton of Lebanese coastal waters (eastern Mediterranean). J. Plankton Res. 1989, 11, 203–222. [Google Scholar] [CrossRef]
- Sitran, R.; Bergamasco, A.; Decembrini, F.; Guglielmo, L. Microzooplankton (tintinnid ciliates) diversity: Coastal community structure and driving mechanisms in the southern Tyrrhenian Sea (Western Mediterranean). J. Plankton Res. 2008, 31, 153–170. [Google Scholar] [CrossRef]
- Kršinić, F. Tintinnids (Tintinnida, Choreotrichia, Ciliata) in the Adriatic Sea, Mediterranean. Part II. Ecology; Institute of Oceanography and Fisheries: Split, Croatia, 2010. [Google Scholar]
- Kokkini, Z.; Mauri, E.; Gerin, R.; Poulain, P.M.; Simoncelli, S.; Notarstefano, G. On the salinity structure in the South Adriatic as derived from float and glider observations in 2013–2016. Deep Sea Res. Part II Top. Stud. Oceanogr. 2019, in press. [Google Scholar] [CrossRef]
- Mihanović, H.; Vilibić, I.; Carniel, S.; Tudor, M.; Russo, A.; Bergamasco, A.; Bubić, N.; Ljubešić, Z.; Viličić, D.; Boldrin, A.; et al. Exceptional dense water formation on the Adriatic shelf in the winter of 2012. Ocean Sci. 2013, 9, 561–572. [Google Scholar] [CrossRef] [Green Version]
- Bensi, M.; Cardin, V.; Rubino, A.; Notarstefano, G.; Poulain, P.M. Effects of winter convection on the deep layer of the Southern Adriatic Sea in 2012. J. Geophys. Res. Oceans 2013, 118, 6064–6075. [Google Scholar] [CrossRef] [Green Version]
- Gačić, M.; Civitarese, G.; Eusebi Borzelli, G.L.; Kovačević, V.; Poulain, P.-M.; Theocharis, A.; Menna, M.; Catucci, A.; Zarokanellos, N. On the relationship between the decadal oscillations of the northern Ionian Sea and the salinity distributions in the eastern Mediterranean. J. Geophys. Res. Oceans 2011, 116, C12002. [Google Scholar] [CrossRef]
- Mann, K.H.; Lazier, J.R.N. Dynamics of Marine Ecosystems: Biological-Physical Interactions in the Oceans, 3rd ed.; Blackwell Publishing: Oxford, UK, 2006. [Google Scholar]
- Šantić, D.; Kovačević, V.; Bensi, M.; Giani, M.; Vrdoljak Tomaš, A.; Ordulj, M.; Santinelli, C.; Šestanović, S.; Šolić, M.; Grbec, B. Picoplankton Distribution and Activity in the Deep Waters of the Southern Adriatic Sea. Water 2019, 11, 1655. [Google Scholar] [CrossRef]
- Viličić, D.; Vučak, Z.; Škrivanić, A.; Gržetić, Z. Phytoplankton blooms in the oligotrophic open South Adriatic waters. Int. Symp. Chem. Mediterr. 1989, 28, 89–107. [Google Scholar] [CrossRef]
- Kršinić, F. The family Xystonellidae (Ciliophora, Tintinnina) in the Adriatic Sea. J. Plankton Res. 1988, 10, 413–429. [Google Scholar] [CrossRef]

| Stations | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Depth Layers | 1 | 3 | 5 | 7 | 9 * | 10 ** | 12 | 13 | GG |
| 0–50 | X | X | X * | X ** | X | X | X | ||
| 50–100 | X | X | X * | X ** | X | X | X | ||
| 0–100 | X | X | |||||||
| 100–200 | X | X | X * | X ** | X | X | |||
| 200–300 | X | X * | X ** | X | X | ||||
| 300–400 | X * | X * | X ** | X | |||||
| 400–600 | X * | X * | X ** | ||||||
| 600–800 | X * | X * | X ** | ||||||
| 1200–800 | X * | X ** | |||||||
| Species | Station | Layers (m) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 3 | 5 | 7 | 9 | 10 | 12 | 13 | GG | I | II | III | IV | |
| 1. Codonella amphorella (OSS) | A | A | + | + | D | A | + | + | + | + | + | + | + |
| 2. Codonella apicata (OS, OSS) | . | . | D | D | D | . | D | D | D | D | D | D | . |
| 3. Codonella aspera (OS, OSS) | + | + | + | A | D | A | + | + | + | + | + | + | + |
| 4. Codonella galea (OSS) | . | + | + | . | D | A | + | D | + | + | + | + | A |
| 5. Codonaria cistellula (OS, OSS) | . | + | A | D | . | A | + | + | + | + | + | A | A |
| 6. Poroecus apiculatus (OS) | . | . | . | . | D | . | . | . | . | . | . | D | . |
| 7. Tintinnopsis angulata (OS) | . | . | D | . | . | . | . | D | . | D | . | . | D |
| 8. Tintinnopsis campanula (NE) | D | . | D | . | D | . | D | + | . | + | D | D | D |
| 9. Tintinnopsis radix (NE) | D | . | D | D | D | . | D | D | . | D | D | D | D |
| 10. Codonellopsis orthoceras (OS, OSS) | . | . | D | . | D | . | . | . | D | . | . | D | D |
| 11. Codonellopsis schabi (NE) | D | + | + | D | D | A | + | + | + | + | + | + | + |
| 12. Stenosemella nivalis (NE) | + | . | . | . | . | . | . | . | D | + | + | . | . |
| 13. Stenosemella ventricosa (NE) | D | . | D | . | . | . | D | D | . | D | D | D | D |
| 14. Helicostomella subulata (NE) | D | . | . | . | . | . | D | . | . | D | . | D | . |
| 15. Cyttarocylis cassis (OSS, OMP) | . | D | + | . | D | A | . | D | A | D | + | + | D |
| 16. Cyttarocylis eucecryphalus (OS) | A | A | A | . | . | . | D | . | A | . | + | A | . |
| 17. Petalotricha ampulla (OSS) | A | + | + | . | . | . | . | . | D | D | + | + | . |
| 18. Favella azorica (OS) | . | . | D | . | . | . | . | D | D | D | D | D | . |
| 19. Epiplocylis acuminata (OS) | D | D | D | D | D | . | D | D | D | D | D | D | D |
| 20. Epiplocylis undella (OS) | D | D | D | D | D | . | D | D | D | D | D | D | D |
| 21. Protorhabdonella simplex (OSS) | . | D | . | . | D | . | D | D | . | D | D | . | . |
| 22. Rhabdonella elegans (OS, OSS) | . | . | D | . | D | . | . | . | . | D | . | D | . |
| 23. Rhabdonella spiralis (OS) | + | D | + | D | D | A | D | D | + | D | + | + | + |
| 24. Parundella aculeata (OS, OSS) | . | . | A | . | . | . | . | D | . | . | D | A | . |
| 25. Parundella lohmanni (OMP) | A | + | + | . | D | A | + | D | + | D | + | + | + |
| 26. Parundella messinensis (OMP) | . | D | + | . | D | A | . | . | + | . | D | + | D |
| 27. Xystonella lohmanni (OS, OSS) | . | . | . | . | . | . | . | . | D | D | . | D | . |
| 28. Xystonella longicauda (OS) | . | . | . | . | . | . | + | . | . | A | D | D | . |
| 29. Xystonella treforti (OSS) | . | + | A | . | D | . | . | . | . | D | + | D | . |
| 30. Xystonellopsis cymatica (OMP) | . | . | + | . | D | A | A | D | . | . | D | + | . |
| 31. Xystonellopsis paradoxa (OSS) | D | A | + | . | . | A | + | D | + | . | + | + | . |
| 32. Xystonellopsis scyphium (ODS) | A | D | + | D | D | A | A | . | D | D | D | + | + |
| 33. Amplectella collaria (OSS) | . | . | . | . | D | . | . | D | . | D | D | . | . |
| 34. Undella biangulata (OS, OSS) | . | . | . | . | D | A | D | . | A | . | . | + | + |
| 35. Undella claparedei (OSS) | A | + | + | + | D | A | + | + | + | + | + | + | + |
| 36. Undella pentagona (OS) | . | . | . | . | D | . | . | . | . | D | . | . | . |
| 37. Undella subcaudata acuta (OS) | A | + | + | D | D | A | + | + | + | + | + | + | D |
| 38. Undella subcaudata subcaudata (OSS) | . | . | A | . | D | A | D | . | A | + | + | A | . |
| 39. Undella clevei (OS) | . | D | D | D | . | . | D | . | D | D | D | D | . |
| 40. Undella hyalina (OS) | . | . | + | . | D | A | D | D | . | + | . | D | + |
| 41. Undellopsis marsupialis (OSS) | . | A | + | + | D | A | + | A | A | + | + | + | . |
| 42. Undellopsis subangulata (OS, OSS) | . | . | D | . | D | A | D | . | . | . | A | D | D |
| 43. Dictyocysta elegans (OS, OSS) | D | A | + | . | D | A | + | A | + | + | + | + | . |
| 44. Dictyocysta entzi (OSS) | . | . | A | . | . | . | A | . | A | . | A | A | . |
| 45. Dictyocysta lepida (OS, OSS) | D | + | A | . | . | A | A | . | + | . | D | + | A |
| 46. Dictyocysta mitra (OS, OSS) | + | + | + | + | D | A | + | + | + | + | + | + | D |
| 47. Dictyocysta muelleri (OS, OSS) | . | A | . | . | D | . | . | . | A | . | + | + | . |
| 48. Amphorides quadrilineata (OS, OSS) | D | + | + | A | D | A | + | + | + | + | + | + | . |
| 49. Amphorellopsis acuta (OS) | D | . | . | . | . | . | . | . | . | D | D | . | . |
| 50. Dadayiella ganymedes (OS) | . | D | + | D | . | A | A | D | + | + | + | + | . |
| 51. Dadayiella pachytoecus (OS) | . | . | . | . | D | . | . | . | . | D | D | . | . |
| 52. Steenstrupiella steenstrupii (OS) | + | + | + | + | . | A | + | D | + | + | + | A | D |
| 53. Steenstrupiella intumescens (OS, OSS) | . | . | D | . | . | . | D | D | . | D | . | . | . |
| 54. Daturella striata (OMP) | . | D | A | . | D | . | . | . | D | . | + | + | . |
| 55. Eutintinnus lusus-undae (OS) | + | D | + | D | D | A | + | + | + | + | + | + | . |
| 56. Eutintinnus latus (OS, OSS) | A | . | D | . | D | . | D | A | D | + | + | D | D |
| 57. Eutintinnus elegans (OS) | . | . | . | . | . | . | D | . | . | D | . | . | . |
| 58. Eutintinnus fraknoi (OS) | D | D | D | D | D | A | D | + | D | + | + | + | + |
| 59. Eutintinnus stramentus (OSS) | . | . | D | . | . | . | D | D | D | D | D | . | . |
| 60. Salpingella acuminata (OS) | D | + | + | + | D | A | + | + | + | + | + | + | A |
| 61. Salpingella glockentoegeri (OS, OSS) | . | . | A | . | D | A | . | A | + | D | + | A | . |
| 62. Salpingella rotundata (OS) | . | . | + | D | D | . | D | . | . | D | D | A | . |
| 63. Salpingella subconica (OS) | . | D | . | . | D | . | . | D | . | D | D | . | . |
| TOTAL December | 20 | 27 | 40 | 20 | 42 | . | 38 | 34 | 34 | 49 | 50 | 44 | 24 |
| TOTAL April | 14 | 20 | 32 | 8 | . | 29 | 22 | 16 | 27 | 22 | 31 | 34 | 14 |
| Species | December 2015 n = 27 | April 2016 n = 29 | ||||||
|---|---|---|---|---|---|---|---|---|
| Temp | Sal | Chl-a | DO | Temp | Sal | Chl-a | DO | |
| Amphorellopsis acuta | 0.127 | −0.340 * | 0.349 * | 0.327 | ||||
| Codonella aspera | 0.537 ** | −0.184 | 0.480 * | 0.606 *** | 0.279 | 0.342 * | 0.241 | 0.412 * |
| Codonella amphorella | −0.279 | −0.157 | −0.394 * | −0.313 | ||||
| Codonellopsis schabi | 0.775 *** | −0.016 | 0.557 ** | 0.665 *** | ||||
| Dictyocysta mitra | 0.511 ** | 0.182 | 0.575 ** | 0.480 * | ||||
| Eutintinnus lusus-undae | 0.859 *** | 0.083 | 0.593 *** | 0.651 *** | ||||
| Parundella lohmanni | −0.427 * | −0.346 * | −0.607 *** | −0.456 * | ||||
| Rhabdonella spiralis | 0.499 ** | −0.140 | 0.638 *** | 0.652 *** | ||||
| Salpingella acuminata | 0.865 *** | 0.051 | 0.738 *** | 0.740 *** | ||||
| Steenstrupiella steenstrupii | 0.420 * | −0.093 | 0.672 *** | 0.521 ** | 0.454 * | 0.321 | 0.616 *** | 0.536 ** |
| Undella claparedei | 0.626 *** | 0.328 | 0.294 | 0.414 * | 0.213 | 0.443 ** | 0.125 | 0.188 |
| Protorhabdonella simplex | 0.588 *** | 0.069 | 0.138 | 0.302 | ||||
| Epiplocylis undella | 0.537 ** | 0.228 | 0.244 | 0.364 * | ||||
| Xystonellopsis scyphium | −0.348 * | 0.194 | −0.127 | −0.341 * | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Njire, J.; Batistić, M.; Kovačević, V.; Garić, R.; Bensi, M. Tintinnid Ciliate Communities in Pre- and Post-Winter Conditions in the Southern Adriatic Sea (NE Mediterranean). Water 2019, 11, 2329. https://doi.org/10.3390/w11112329
Njire J, Batistić M, Kovačević V, Garić R, Bensi M. Tintinnid Ciliate Communities in Pre- and Post-Winter Conditions in the Southern Adriatic Sea (NE Mediterranean). Water. 2019; 11(11):2329. https://doi.org/10.3390/w11112329
Chicago/Turabian StyleNjire, Jakica, Mirna Batistić, Vedrana Kovačević, Rade Garić, and Manuel Bensi. 2019. "Tintinnid Ciliate Communities in Pre- and Post-Winter Conditions in the Southern Adriatic Sea (NE Mediterranean)" Water 11, no. 11: 2329. https://doi.org/10.3390/w11112329
APA StyleNjire, J., Batistić, M., Kovačević, V., Garić, R., & Bensi, M. (2019). Tintinnid Ciliate Communities in Pre- and Post-Winter Conditions in the Southern Adriatic Sea (NE Mediterranean). Water, 11(11), 2329. https://doi.org/10.3390/w11112329






