Biosensors in Monitoring Water Quality and Safety: An Example of a Miniaturizable Whole-Cell Based Sensor for Hg2+ Optical Detection in Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
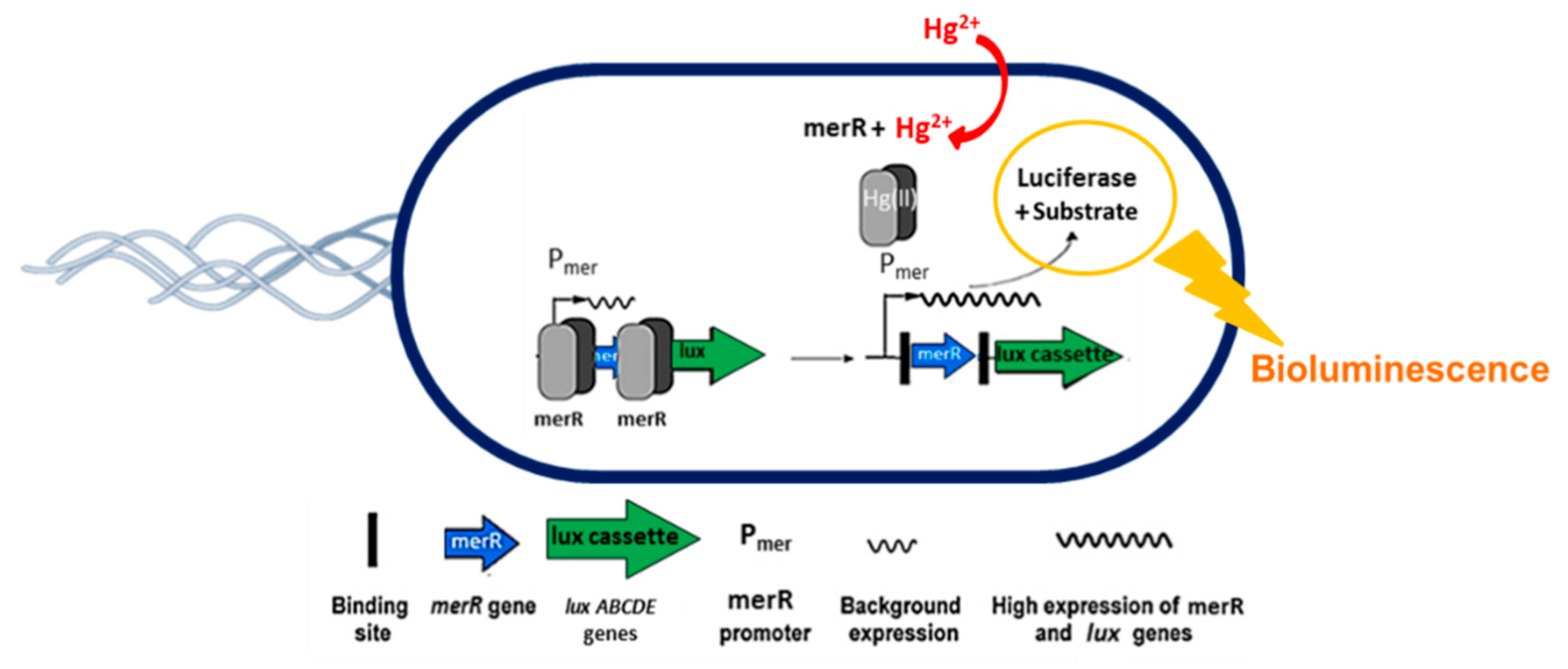
2.2. Hg2+ Reporter Cell Preparation
2.3. Hg Reporter Assay in Water
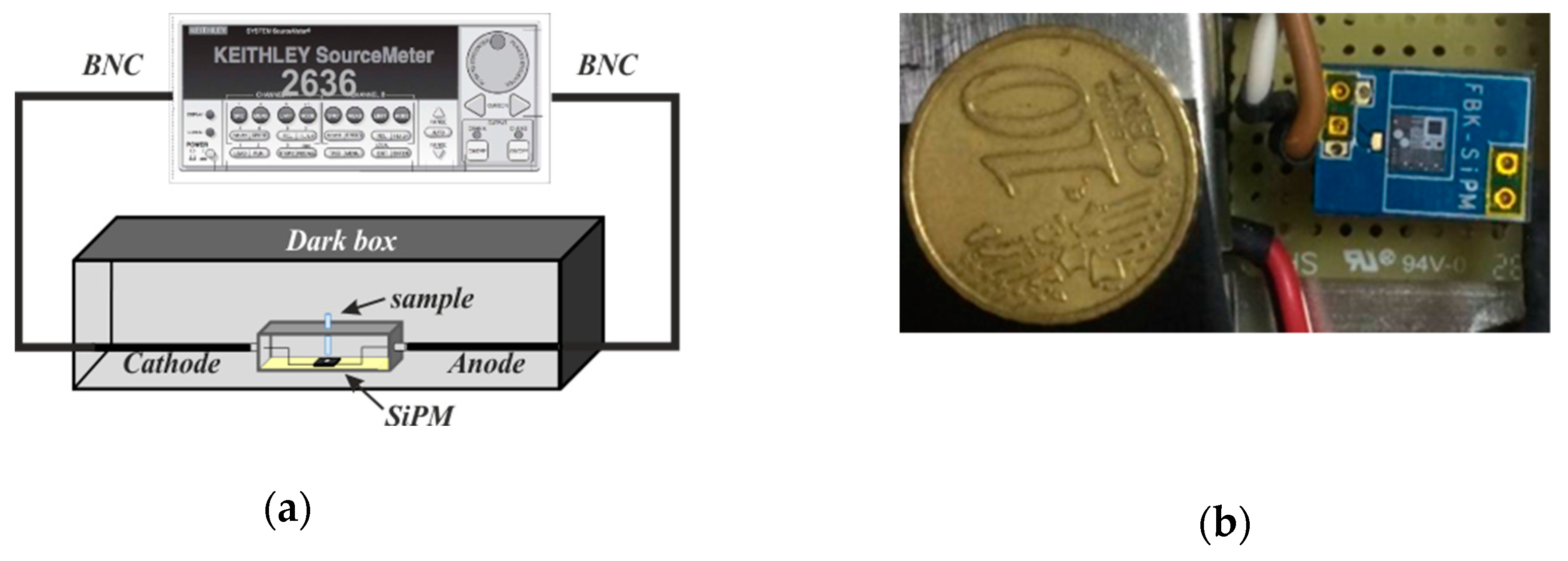
2.4. Optical Sensor for the Hg Sensing in Water
3. Results
3.1. First Optical Check of Hg2+ Reporter-Induced Bioluminescence
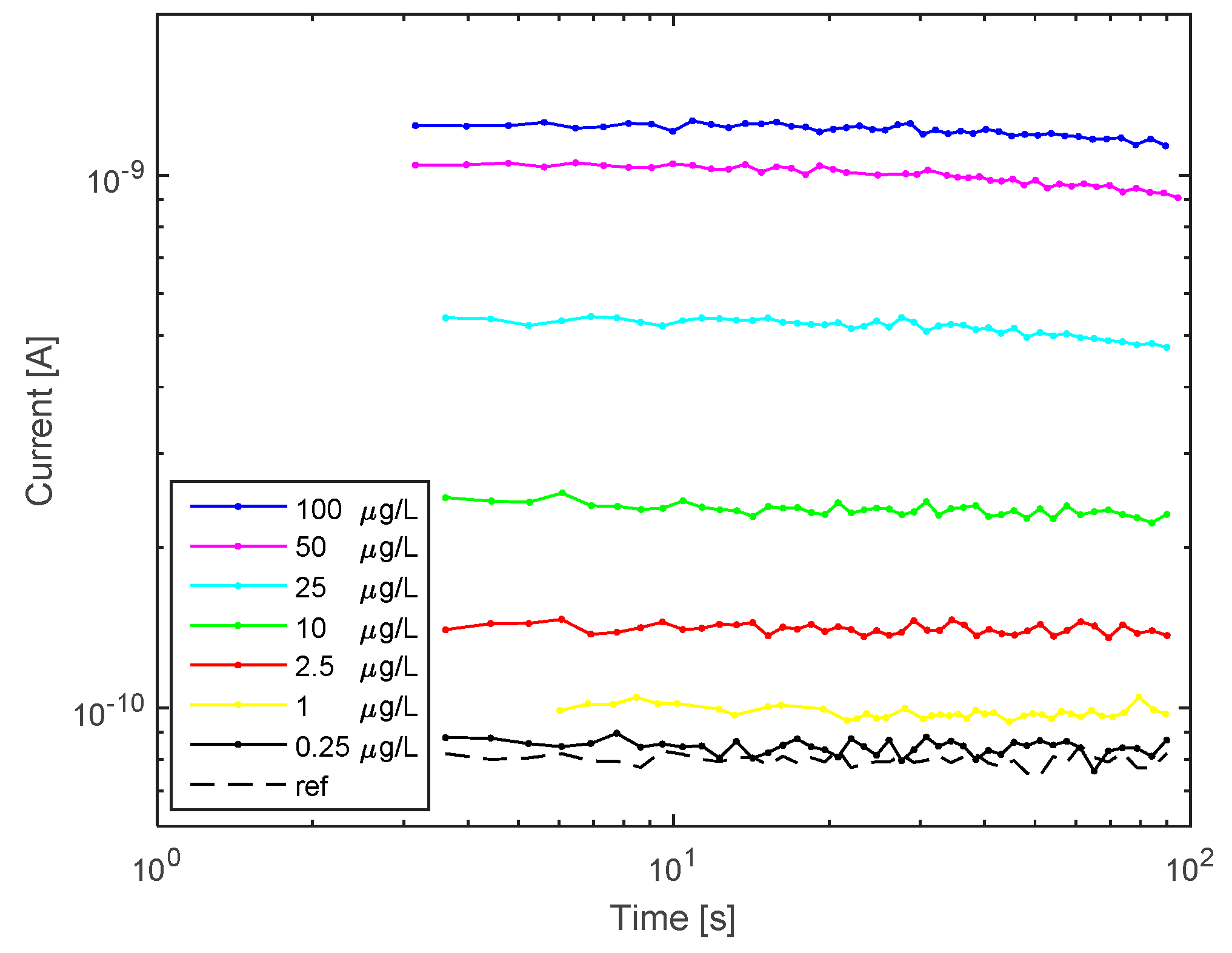
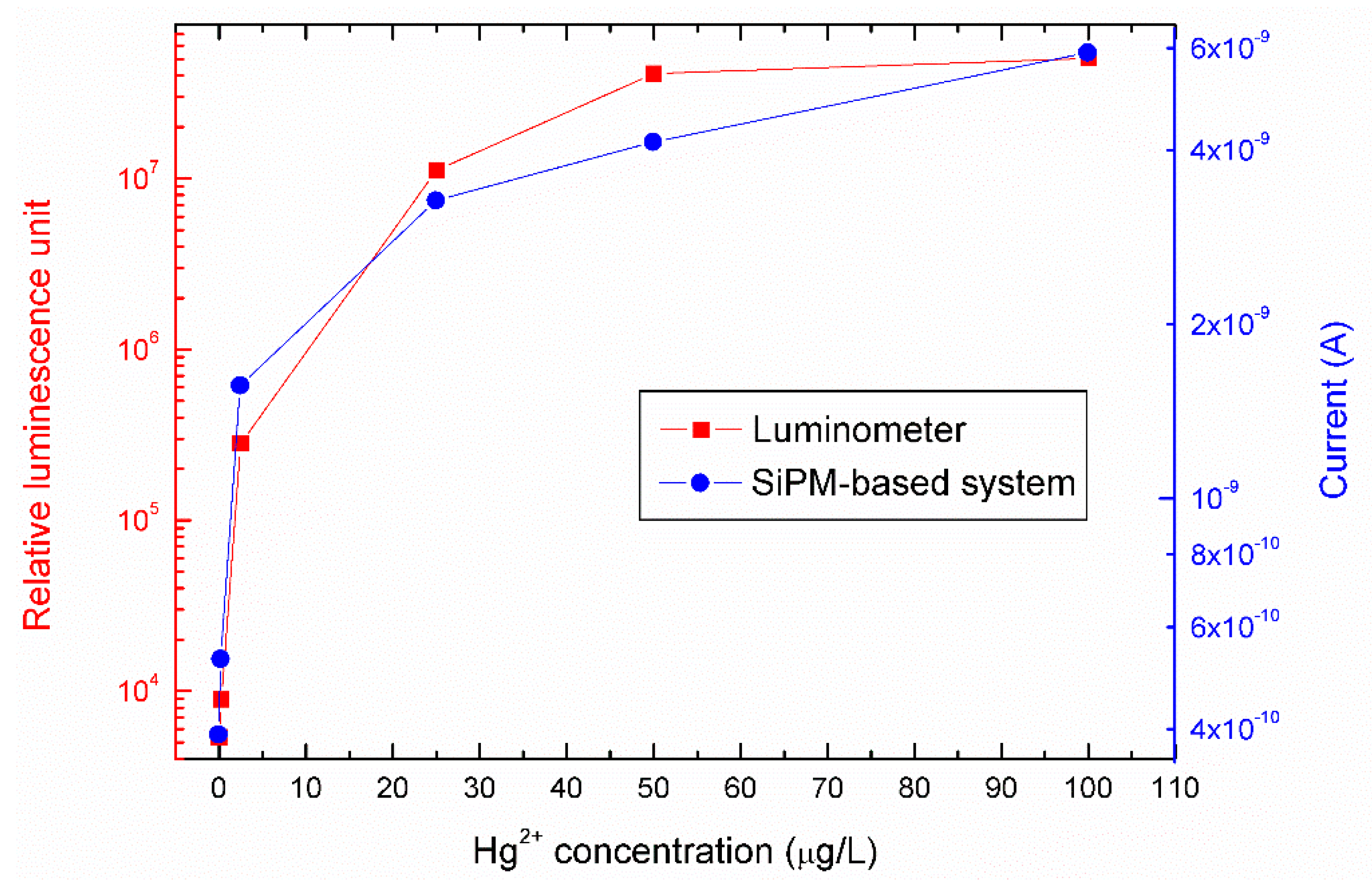
3.2. Hg2+ Reporter Activity Validation by Silicon Photomultiplier (SiPM)-Based Sensing System
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Texas Water Quality Standards. Criteria for Recreation; TCEQ Staff: Austin, TX, USA, 2007.
- World Health Organization. Coastal and Fresh Waters. In Guidelines for Safe Recreational Water Environments; WHO: Geneva, Switzerland, 2003; Volume 1, ISBN 92 4 154580. [Google Scholar]
- Valeriani, F.; Protano, C.; Vitali, M.; Romano Spica, V. Swimming attendance during childhood and development of asthma: Meta-analysis. Pediatr. Int. 2017, 59, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Water Quality: Ambient Water Quality Guidelines for Mercury Overview Report—First Update; Ministry of Environment: Victoria, BC, Canada, 1981.
- World Health Organization. Recommendations. In Guidelines for Drinking-Water Quality, 3rd ed.; WHO: Geneva, Switzerland, 2004; Volume 1, ISBN 92 4 154638 7. [Google Scholar]
- Lepak, J.M.; Shayler, H.A.; Kraft, C.E.; Knuth, B.A. Mercury contamination in sport fish in the Northeastern United States: Considerations for future data collection. BioScience 2009, 59, 174–181. [Google Scholar] [CrossRef]
- Taylor, D.R.; Williamson, P.R. Mercury contamination in Southern New England coastal fisheries and dietary habits of recreational anglers and their families: Implications to human health and issuance of consumption advisories. Mar. Pollut. Bull. 2017, 114, 144–156. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Elemental Mercury and Inorganic Mercury Compounds: Human Health Aspects. 2003. Available online: https://www.who.int/ipcs/publications/cicad/en/cicad50.pdf (accessed on 4 June 2019).
- CDC. Environmental Health. Mercury; 2009. Available online: https://www.cdc.gov/biomonitoring/pdf/Mercury_FactSheet.pdf (accessed on 12 June 2019).
- Environmental Protection Agency. Mercury: Basic Information. Available online: http://www.epa.gov/mercury/about.htm (accessed on 12 June 2019).
- National Research Council. Toxicological Effects of Methylmercury; National Academy Press: Washington, DC, USA, 2000. [Google Scholar]
- Mergler, D.; Anderson, H.A.; Chan, L.H.M.; Mahaffey, K.R.; Murray, M.; Sakamoto, M.; Stern, A.H. Methylmercury exposure and health effects in humans: A worldwide concern. Ambio 2007, 36, 3–11. [Google Scholar] [CrossRef]
- Yu, Y.L.; Wang, J.H. Recent advances in flow-based sample pretreatment for the determination of metal species by atomic spectrometry. Chin. Sci. Bull. 2013, 58, 1992–2002. [Google Scholar] [CrossRef]
- Suvarapu, L.N.; Seo, Y.-K.; Baek, S.-O. Speciation and determination of mercury by various analytical techniques. Rev. Anal. Chem. 2013, 32, 225–245. [Google Scholar] [CrossRef]
- Popp, M.; Hann, S.; Koellensperger, G. Environmental application of elemental speciation analysis based on liquid or gas chromatography hyphenated to inductively coupled plasma mass spectrometry—A review. Anal. Chim. Acta 2010, 668, 114–129. [Google Scholar] [CrossRef] [PubMed]
- Turker, A.R. Separation, preconcentration and speciation of metal ions by solid phase extraction. Sep. Purif. Rev. 2012, 41, 3169–3206. [Google Scholar] [CrossRef]
- Ferreira, S.L.C.; Lemos, V.A.; Silva, L.O.B.; Queiroz, A.F.S.; Souza, A.S.; da Silva, E.G.P.; dos Santos, W.N.L.; das Virgens, C.F. Analytical strategies of sample preparation for the determination of mercury in food matrices—A review. Microchem. J. 2015, 121, 227–236. [Google Scholar] [CrossRef]
- Gao, Y.; Shi, Z.; Long, Z.; Wu, P.; Zheng, C.; Hou, X. Determination and speciation of mercury in environmental and biological samples by analytical atomic spectrometry. Microchem. J. 2012, 103, 1–14. [Google Scholar] [CrossRef]
- Chung, S.W.; Chan, B.T. A reliable method to determine methylmercury and ethylmercury simultaneously in foods by gas chromatography with inductively coupled plasma mass spectrometry after enzymatic and acid digestion. J. Chrom. A 2011, 9, 1260–1265. [Google Scholar] [CrossRef] [PubMed]
- Kodamatani, H.; Kanzaki, R.; Tomiyasu, T.; Saito, K.; Kono, Y. Determination of organic and inorganic mercury species as emetine dithiocarbamate complexes by high-performance liquid chromatography with electrogeneratedtris (2,20-bipyridine) ruthenium(iii) chemiluminescence detection. Anal. Lett. 2011, 44, 2769–2779. [Google Scholar] [CrossRef]
- Hashemi-Moghaddam, H.; Saber-Tehrani, M. Sensitive mercury speciation by reversed phase column high-performance liquid chromatography with UV-visible detection after solid-phase extraction using 6-mercaptopurine and dithizone. J. AOAC Int. 2008, 6, 1453–1458. [Google Scholar]
- Rodrigues, J.L.; de Souza, S.S.; de Oliveira Souza, V.C.; Barbosa, F., Jr. Methylmercury and inorganic mercury determination in blood by using liquid chromatography with inductively coupled plasma mass spectrometry and a fast sample preparation procedure. Talanta 2010, 3, 1158–1163. [Google Scholar] [CrossRef] [PubMed]
- Dressler, V.L.; Santos, C.M.M.; Antes, F.G.; Bentlin, F.R.S.; Pozebon, D.; Flores, E.M.M. Total mercury, inorganic mercury and methyl mercury determination in red wine. Food Anal. Methods 2012, 5, 505–511. [Google Scholar] [CrossRef]
- Zhu, Z.Q.; Su, Y.; Li, J.; Li, D.; Zhang, J.; Song, S.; Zhao, Y.; Li, G.; Fan, C. Highly sensitive electrochemical sensor for mercury(II) ions by using a mercury-specific oligonucleotide probe and gold nanoparticle-based Amplification. Anal. Chem. 2009, 81, 7660–7666. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.C.; Chen, X.; Guo, Z.; Xie, C.G.; Kong, L.T.; Liu, J.H.; Huang, X.J. Stripping voltammetric detection of mercury(II) based on a surface ion imprinting strategy in electropolymerized microporous poly(2-mercaptobenzothiazole) films modified glassy carbon electrode. Anal. Chim. Acta 2011, 685, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.Q.; Chen, X.; Yang, Q.; Liu, J.H.; Huang, X.J. Selective adsorption toward toxic metal ions results in selective response: Electrochemical studies on a polypyrrole/reduced graphene oxide nanocomposite. Chem. Comm. 2012, 48, 2180–2182. [Google Scholar] [CrossRef]
- Fu, X.C.; Wu, J.; Nie, L.; Xie, C.G.; Liu, J.H.; Huang, X.J. Electropolymerized surface ion imprinting films on a gold nanoparticles/single-wall carbon nanotube nanohybrids modified glassy carbon electrode for electrochemical detection of trace mercury(II) in water. Anal. Chim. Act. 2012, 720, 29–37. [Google Scholar] [CrossRef]
- Gao, C.; Huang, X.J. Voltammetric determination of mercury(II). TrAC Trends Anal. Chem. 2013, 51, 1–12. [Google Scholar] [CrossRef]
- Guerrini, L.; Rodriguez-Loureiro, I.; Correa-Duarte, M.A.; Lee, Y.H.; Ling, X.Y.; García de Abajo, F.J.; Alvarez-Puebla, R.A. Chemical speciation of heavy metals by surface-enhanced Raman scattering spectroscopy: Identification and quantification of inorganic- and methyl-mercury in water. Nanoscale 2014, 6, 8368–8375. [Google Scholar] [CrossRef] [PubMed]
- Pujol, L.; Evrard, D.; Groenen-Serrano, K.; Freyssinier, M.; Ruffien-Cizsak, A.; Gros, P. Electrochemical sensors and devices for heavy metals assay in water: The French groups’ contribution. Front. Chem. 2014, 2, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Preedy, V.R.; Patel, V.B. Biosensors and Environmental Health; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Dzantiev, B.B.; Zherdev, A.V. Antibody-Based Biosensors. In Portable Biosensing of Food Toxicants and Environmental Pollutants; CRC Press: Boca Raton, FL, USA, 2013; pp. 161–196. [Google Scholar]
- Gu, M.B.; Mitchell, R.J.; Kim, B.C. Whole-cell-based biosensors for environmental biomonitoring and application. Adv. Biochem. Eng. Biotechnol. 2004, 87, 269–305. [Google Scholar] [PubMed]
- Gui, Q.; Lawson, T.; Shan, S.; Yan, L.; Liu, Y. The application of whole cell-based biosensors for use in environmental analysis and in medical diagnostics. Sensors 2017, 17, 1623. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, K.H.; Giovangrandi, L.; Whittington, R.H.; Kovacs, G.T. Sensitivity of cell-based biosensors to environmental variables. Biosens. Bioelectr. 2005, 20, 1397–1406. [Google Scholar] [CrossRef] [PubMed]
- Hakkila, K.; Green, T.; Leskinen, P.; Ivask, A.; Marks, R.; Virta, M. Detection of bioavailable heavy metals in EILA Tox-Oregon samples using whole-cell luminescent bacterial sensors in suspension or immobilized onto fibre-optic tips. J. App. Toxicol. 2004, 24, 333–342. [Google Scholar] [CrossRef]
- Van der Meer, J.R.; Belkin, S. Where microbiology meets microengineering: Design and applications of reporter bacteria. Nat. Rev. Microbiol. 2010, 8, 511–522. [Google Scholar] [CrossRef]
- Schaefer, J.K.; Letowski, J.; Barkay, T. Mer-Mediated Resistance and Volatilization of Hg(II) Under Anaerobic Conditions. Geomicrobiol. J. 2002, 19, 87–102. [Google Scholar] [CrossRef]
- Barkay, T.; Miller, S.M.; Summers, A.O. Bacterial mercury resistance from atoms to ecosystems. FEMS Microbiol. Rev. 2003, 27, 355–384. [Google Scholar] [CrossRef]
- Brown, N.L.; Stoyanov, J.V.; Kidd, S.P.; Hobman, J.L. The MerR family of transcriptional regulators. FEMS Microbiol. Rev. 2003, 27, 145–163. [Google Scholar] [CrossRef]
- Condee, C.W.; Summers, A.O. A mer-lux Transcriptional Fusion for Real-Time Examination of In Vivo Gene Expression Kinetics and Promoter Response to Altered Superhelicity. J. Bacteriol. 1992, 174, 8094–8101. [Google Scholar] [CrossRef][Green Version]
- Tinikul, R.; Chaiyen, P. Structure, Mechanism, and Mutation of Bacterial Luciferase. In Bioluminescence: Fundamentals and Applications in Biotechnology—Volume 3. Advances in Biochemical Engineering/Biotechnology; Thouand, G., Marks, R., Eds.; Springer: Berlin, Germany, 2014; Volume 154. [Google Scholar]
- Cortés-Salazar, F.; Beggah, S.; van der Meer, J.R.; Girault, H.H. Electrochemical As(III) whole-cell based biochip sensor. Biosens. Bioelectr. 2013, 47, 237–242. [Google Scholar] [CrossRef]
- Mazzillo, M.; Ronzhin, A.; Los, S.; Abisso, S.; Sanfilippo, D.; Valvo, G.; Carbone, B.; Piana, A.; Fallica, G.; Albrow, M.; et al. Electro-optical performances of p on n and n on p Silicon Photomultipliers. IEEE Trans. Elctr. Dev. 2012, 59, 3419–3425. [Google Scholar] [CrossRef]
- Pagano, R.; Valvo, G.; Sanfilippo, D.; Libertino, S.; Corso, D.; Fallica, P.G.; Lombardo, S. Silicon photomultiplier device architecture with dark current improved to the ultimate physical limit. Appl. Phys. Lett. 2013, 102, 183502. [Google Scholar] [CrossRef]
- Acerbi, F.; Paternoster, G.; Gola, A.; Regazzoni, V.; Zorzi, N.; Piemonte, C. High-Density Silicon Photomultipliers: Performance and Linearity Evaluation for High Efficiency and Dynamic-Range Applications. IEEE J. Quantum Electron. 2018, 54, 4700107. [Google Scholar] [CrossRef]
- Council of the European Union. Council Directive 98/83/EC of 3 November 1998 on the quality of water intended for human consumption. Off. J. Eur. Communities 1998, 41, 32–54. [Google Scholar]
- Mercury and Air Toxics Standards (MATS). Available online: https://www.epa.gov/mats (accessed on 24 May 2019).
- Maranhao, T.A.; Silva, J.S.A.; de Andrade, R.M.; Bascunan, A.F.; de Oliveira, F.J.S.; Curtius, A.J. Determination of As and Hg in acetic acid extract by vapor generation coupled to atomic spectrometry for solid waste classification. Microchem. J. 2013, 106, 139. [Google Scholar] [CrossRef]
- Santos, E.J.; Herrmann, A.; Vieira, A.M.; Frescura, A.V.L.; Curtius, A.J. Evaluation of slurry preparation procedures for the determination of mercury by axial view inductively coupled plasma optical emission spectrometry using on-line cold vapor generation. Spectrochim. Acta B At. Spectrosc. 2005, 60, 659–665. [Google Scholar] [CrossRef]
- Leopold, K.; Foulkes, M.; Worsfold, P. Methods for the determination and speciation of mercury in natural waters. A review. Anal. Chim. Acta 2010, 663, 127. [Google Scholar] [CrossRef]
- Dinu, C.; Vasile, G.G.; Cruceru, L. Advanced analytical methods for mercury determination in slightly contaminated water samples. J. Environ. Prot. Ecol. 2013, 14, 1515–1524. [Google Scholar]
- Sciuto, E.L.; Villaggio, G.; Santangelo, M.F.; Laudani, S.; Federico, C.; Saccone, S.; Sinatra, F.; Libertino, S. Study of a Miniaturizable System for Optical Sensing Application to Human Cells. Appl. Sci. 2019, 9, 975. [Google Scholar] [CrossRef]
- Libertino, S.; Conoci, S.; Santangelo, M.F.; Pagano, R.; Sciuto, E.L.; Sinatra, F.; Sanfilippo, D.; Fallica, G.; Lombardo, S. Optical and electrical Si-based biosensors: Fabrication and trasduction issues. J. Anal. Bioanal. Tech. 2014, 12, 1. [Google Scholar]
- Santangelo, M.F.; Sciuto, E.L.; Lombardo, S.A.; Busacca, A.C.; Petralia, S.; Conoci, S.; Libertino, S. Si photomultipliers for bio-sensing applications. IEEE J. Sel. Top. Quant. Electron. 2015, 22, 335–341. [Google Scholar] [CrossRef]
- Santangelo, M.F.; Libertino, S.; Turner, A.P.F.; Filippini, D.; Mak, W.C. Integrating printed microfluidics with silicon photomultipliers for miniaturised and highly sensitive ATP bioluminescence detection. Biosens. Bioelectron. 2018, 99, 464–470. [Google Scholar] [CrossRef]
- Petralia, S.; Sciuto, E.L.; Santangelo, M.; Libertino, S.; Messina, M.A.; Conoci, S. Sulfide species optical monitoring by a miniaturized silicon photomultiplier. Sensors 2018, 18, 727. [Google Scholar] [CrossRef]
- Buffi, N.; Beggah, S.; Truffer, F.; Geiser, M.; van Lintel, H.; Renauda, P.; van der Meer, J.R. An automated microreactor for semi-continuous biosensor measurements. Lab Chip 2016, 16, 1383–1392. [Google Scholar] [CrossRef][Green Version]
- Durand, M.J.; Hua, A.; Jouanneau, S.; Cregut, M.; Thouand, G. Detection of Metal and Organometallic Compounds with Bioluminescent Bacterial Bioassays. In Bioluminescence: Fundamentals and Applications in Biotechnology—Volume 3. Advances in Biochemical Engineering/Biotechnology; Thouand, G., Marks, R., Eds.; Springer: Berlin, Germany, 2015; Volume 154. [Google Scholar]
- Giampaoli, S.; Garrec, N.; Donzé, G.; Valeriani, F.; Erdinger, L.; Romano Spica, V. Regulations concerning natural swimming ponds in Europe: Considerations on public health issues. J. Water Health 2014, 12, 564–572. [Google Scholar] [CrossRef]



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sciuto, E.L.; Coniglio, M.A.; Corso, D.; van der Meer, J.R.; Acerbi, F.; Gola, A.; Libertino, S. Biosensors in Monitoring Water Quality and Safety: An Example of a Miniaturizable Whole-Cell Based Sensor for Hg2+ Optical Detection in Water. Water 2019, 11, 1986. https://doi.org/10.3390/w11101986
Sciuto EL, Coniglio MA, Corso D, van der Meer JR, Acerbi F, Gola A, Libertino S. Biosensors in Monitoring Water Quality and Safety: An Example of a Miniaturizable Whole-Cell Based Sensor for Hg2+ Optical Detection in Water. Water. 2019; 11(10):1986. https://doi.org/10.3390/w11101986
Chicago/Turabian StyleSciuto, Emanuele Luigi, Maria Anna Coniglio, Domenico Corso, Jan Roelof van der Meer, Fabio Acerbi, Alberto Gola, and Sebania Libertino. 2019. "Biosensors in Monitoring Water Quality and Safety: An Example of a Miniaturizable Whole-Cell Based Sensor for Hg2+ Optical Detection in Water" Water 11, no. 10: 1986. https://doi.org/10.3390/w11101986
APA StyleSciuto, E. L., Coniglio, M. A., Corso, D., van der Meer, J. R., Acerbi, F., Gola, A., & Libertino, S. (2019). Biosensors in Monitoring Water Quality and Safety: An Example of a Miniaturizable Whole-Cell Based Sensor for Hg2+ Optical Detection in Water. Water, 11(10), 1986. https://doi.org/10.3390/w11101986






