Response of Aquatic Plants and Water Quality to Large-Scale Nymphoides peltata Harvest in a Shallow Lake
Abstract
1. Introduction
2. Method
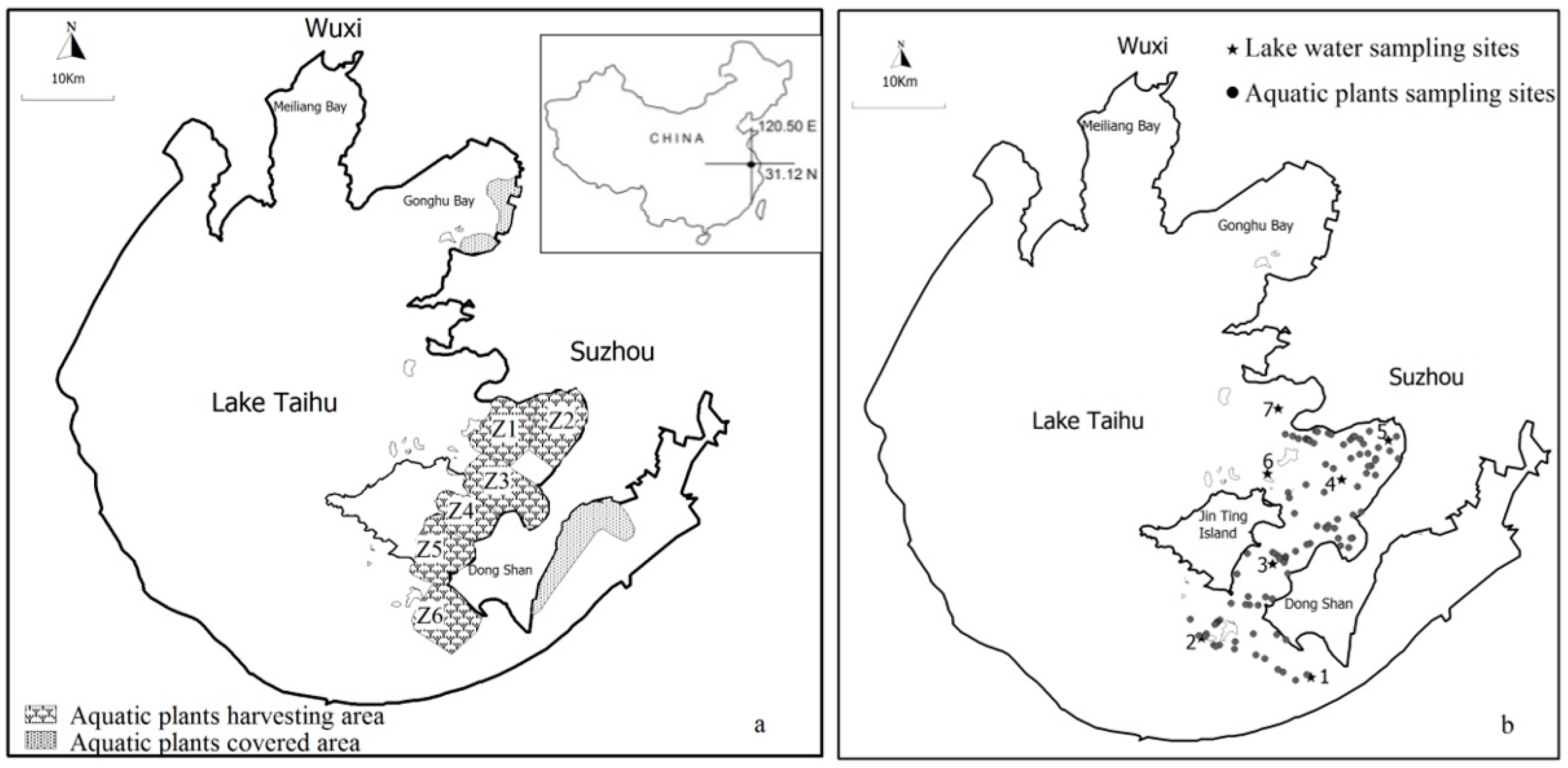
2.1. Study Area
2.2. Study Species and Aquatic Vegetation Harvesting
2.3. Aquatic Vegetation Coverage and Biomass Survey
2.4. Physical and Chemical Properties
2.5. Assessment Method
3. Results
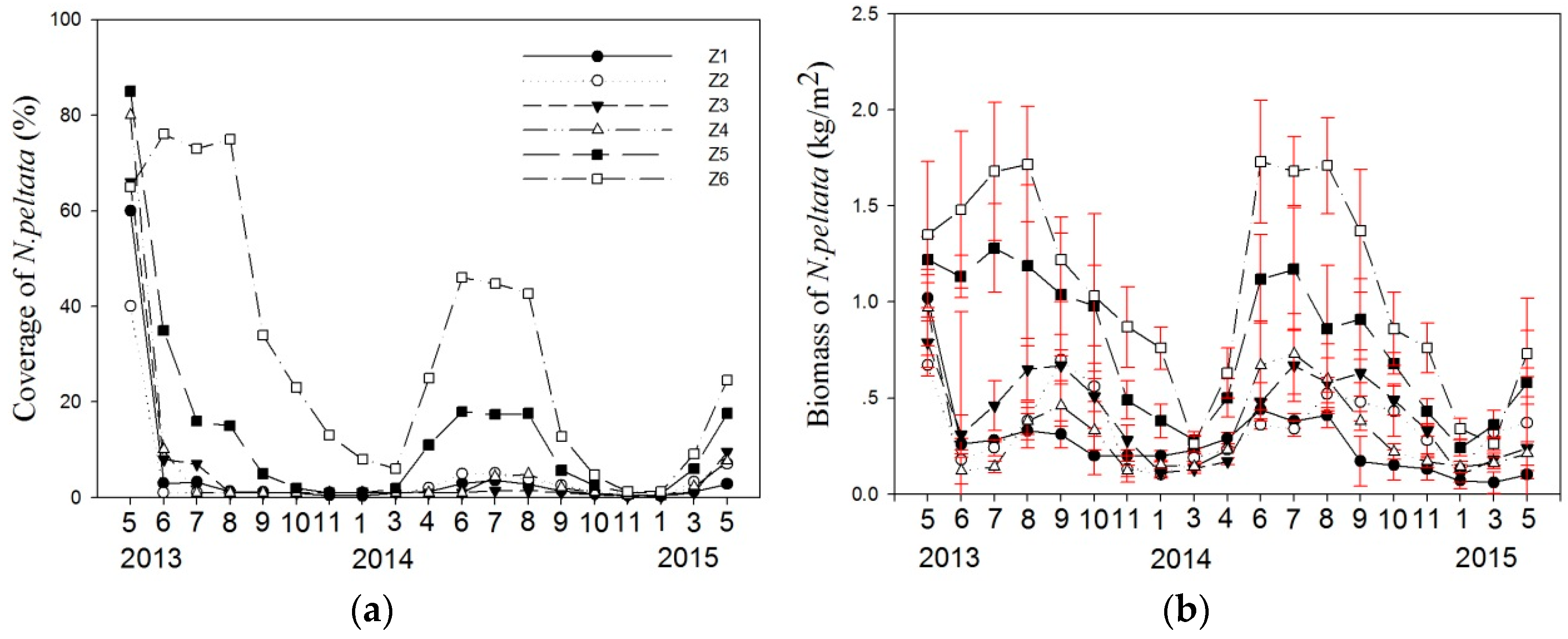
3.1. Variation in Coverage and Biomass of N. peltata
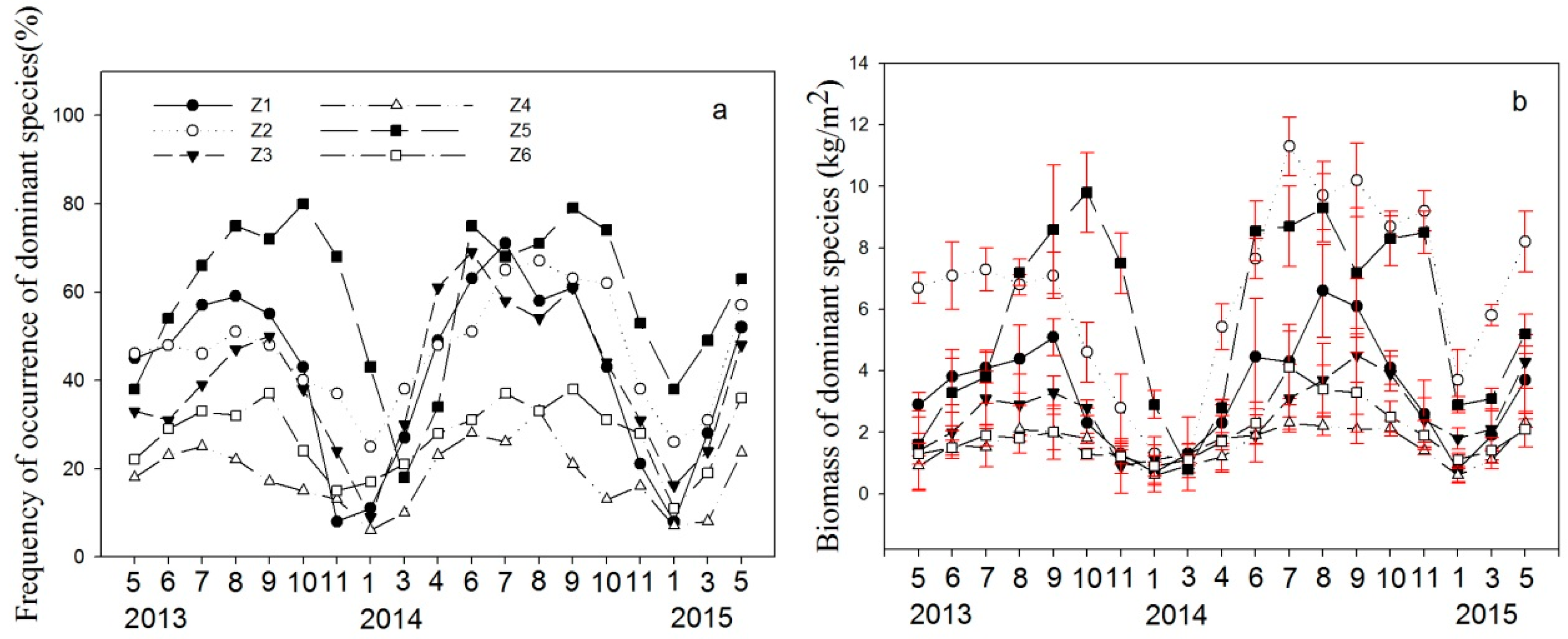
3.2. Aquatic Plant Community
3.3. Plant-N and Plant-P
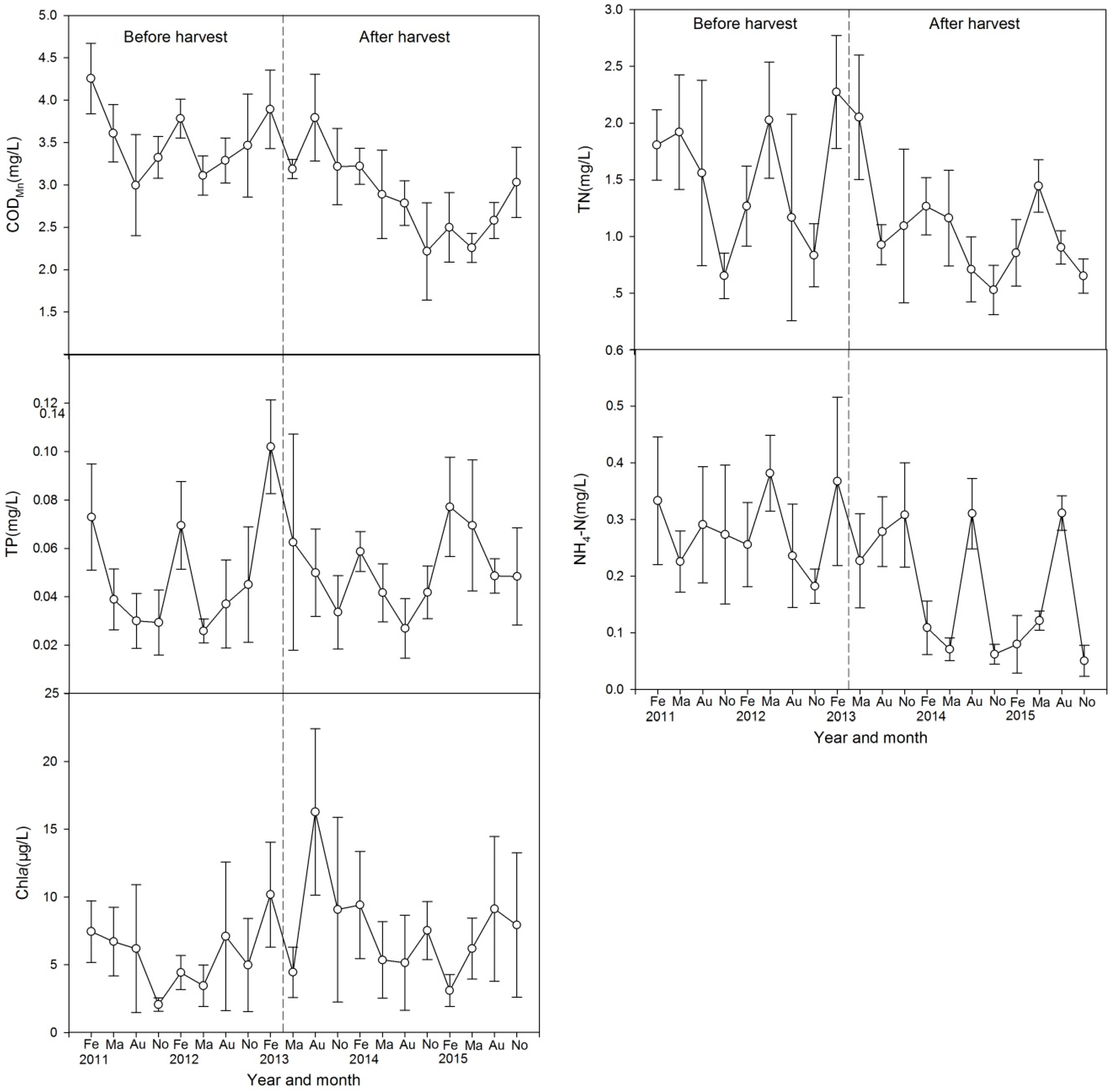
3.4. Water Quality
4. Discussion
4.1. Effect of Harvest on Water Quality
4.2. Effect of Harvest on the Regrowth of N. peltata
4.3. Spatial Differentiation in Aquatic Plant Coverage after Harvesting
4.4. Response of Submerged Plants to Harvesting
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vereecken, H.; Baetens, J.; Viaene, P.; Mostaert, F.; Meire, P. Ecological management of aquatic plants: Effects in lowland streams. Hydrobiologia 2006, 570, 205–210. [Google Scholar] [CrossRef]
- Li, C.; Wang, B.; Ye, C.; Ba, Y. The release of nitrogen and phosphorus during the decomposition process of submerged macrophyte (Hydrilla verticillata Royle) with different biomass levels. Ecol. Eng. 2014, 70, 268–274. [Google Scholar] [CrossRef]
- Han, H.; Weiping, H.; Shuijing, Z. Modelling nitrogen and phosphorus transfer in Potamogeton malaianus Miq. decompostion. Environ. Sci. 2010, 31, 1483–1488. (In Chinese) [Google Scholar]
- Evans, J.M.; Wilkie, A.C. Life cycle assessment of nutrient remediation and bioenergy production potential from the harvest of hydrilla (Hydrilla verticillata). J. Environ. Manag. 2010, 91, 2626–2631. [Google Scholar] [CrossRef] [PubMed]
- Caffrey, J.M.; Barrett, R.R.F.; Ferreira, M.T.; Moreira, T.S.; Murphy, K.J.; Wode, P.M. Integrated submerged aquatic vegetation mamagement in an urban New Zealand rive. Hydrobiologia 1999, 415, 235–241. [Google Scholar]
- Garbey, C.; Thiébaut, G.; Muller, S. Impact of manual spring harvesting on the regrowth of a spreading aquatic plant: Ranunculus peltatus SCHRANK. Arch. Hydrobiol. 2003, 156, 271–286. [Google Scholar] [CrossRef]
- Nino, F.D.; Thiébaut, G.; Muller, S. Response of Elodea Nuttallii (Planch.) H. St. John to manual harvesting in the North-East of France. Hydrobiologia 2005, 551, 147–157. [Google Scholar] [CrossRef]
- Xu, W.; Hu, W.; Deng, J.; Zhu, J.; Li, Q. Effects of harvest management of Trapa bispinosa on an aquatic macrophyte community and water quality in a eutrophic lake. Ecol. Eng. 2014, 64, 120–129. [Google Scholar] [CrossRef]
- Van Nes, E.H.; van den Berg, M.S.; Clayton, J.S.; Coops, H.; Scheffer, M.; van Ierland, E. A simple model for evaluating the costs and benefits of aquatic macrophytes. Hydrobiologia 1999, 415, 335–339. [Google Scholar] [CrossRef]
- Van Nes, E.H.; Scheffer, M.; van den Berg, M.S.; Coops, H. Aquatic macrophytes: Restore, eradicate or is there a compromise? Aquat. Bot. 2002, 72, 387–403. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Day, J.W., Jr. Thinking big with whole-ecosystem studies and ecosystem restoration—A legacy of H.T. Odum. Ecol. Model. 2004, 178, 133–155. [Google Scholar] [CrossRef]
- Chapin, F.S.; Zavaleta, E.S.; Eviner, V.T.; Naylor, R.L.; Vitousek, P.M.; Reynolds, H.L.; Hooper, D.U.; Lavorel, S.; Sala, O.E.I.; Hobbie, S.E.; et al. Consequences of changing biodiversity. Nature 2000, 405, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Dong, B.; Qin, B.; Gao, G.; Cai, X. Submerged macrophyte communities and the controlling factors in large, shallow Lake Taihu (China): Sediment distribution and water depth. J. Great Lakes Res. 2014, 40, 646–655. [Google Scholar] [CrossRef]
- Li, K.-Y.; Liu, Z.-W.; Guan, B.-H. Effects of nutrient levels in surface water and sediment on the growth of the floating-leaved macrophyte Trapa maximowiczii: Implication for management of macrophytes in East Bay of Lake Taihu, China. Limnology 2009, 11, 95–101. [Google Scholar] [CrossRef]
- Huang, W.; Chen, K.; Shi, X.; Ren, K.; Li, W. The contribution of seeds to the recruitment of a Nymphoides peltata population. Limnologica 2014, 44, 1–8. [Google Scholar] [CrossRef]
- Marion, L.; Paillisson, J.-M. A mass balance assessment of the contribution of floating-leaved macrophytes in nutrient stocks in an eutrophic macrophyte-dominated lake. Aquat. Bot. 2003, 73, 249–260. [Google Scholar] [CrossRef]
- Lu, J.; Bunn, S.E.; Burford, M.A. Nutrient release and uptake by littoral macrophytes during water level fluctuations. Sci. Total Environ. 2018, 622–623, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Zhai, S.; Hu, W.; Zhu, Z. Ecological impacts of water transfers on Lake Taihu from the Yangtze River, China. Ecol. Eng. 2010, 36, 406–420. [Google Scholar] [CrossRef]
- Wu, T.; Qin, B.; Zhu, G.; Zhu, M.; Li, W.; Luan, C. Modeling of turbidity dynamics caused by wind-induced waves and current in the Taihu Lake. Int. J. Sediment Res. 2013, 28, 139–148. [Google Scholar] [CrossRef]
- Li, Z.; Xu, J.; Cao, T.; Ni, L.; Xie, P. Adaptive responses of a floating-leaved macrophyte, Nymphoides peltata, to a terrestrial habitat. J. Freshw. Ecol. 2010, 25, 481–486. [Google Scholar] [CrossRef]
- Environment Agency. Threatened Wildlife of Japan-Red Data Book, 2nd ed.; Japan Wild life Research Center: Tokyo, Japan, 2002.
- Larson, D. Growth of three submerged plants below different densities of Nymphoides peltata (S. G. Gmel.) Kuntze. Aquat. Bot. 2007, 86, 280–284. [Google Scholar] [CrossRef]
- Darbyshire, S.J.; Francis, A. The biology of invasive alien plants in Canada. 10. Nymphoides peltata (S. G. Gmel.) Kuntze. Can. J. Plant Sci. 2008, 88, 811–829. [Google Scholar] [CrossRef]
- Child, L.; Brock, J.H.; Brundu, G.; Prach, K.; Pysˇek, K.; Wade, P.M.; Williamson, M. Plant invasions: Ecological threats and management solutions. In the Evaluation and Management of Aquatic Weeds in New Zealand; Champion, P.D., Clayton, J.S., Eds.; Backhuys: Leiden, The Netherlands, 2003; pp. 429–434. [Google Scholar]
- Jin, X.; Tu, Q. Criterion to Lake Eutrophication Survey; China Environmental Science Press: Beijing, China, 1999; p. 317. [Google Scholar]
- Wang, C.-Y.; Sample, D.J.; Day, S.D.; Grizzard, T.J. Floating treatment wetland nutrient removal through vegetation harvest and observations from a field study. Ecol. Eng. 2014, 78, 15–26. [Google Scholar] [CrossRef]
- Hu, L.; Hu, W.; Deng, J.; Li, Q.; Gao, F.; Zhu, J.; Han, T. Nutrient removal in wetlands with different macrophyte structures in eastern Lake Taihu, China. Ecol. Eng. 2010, 36, 1725–1732. [Google Scholar] [CrossRef]
- Zhu, J.; Hu, W.; Hu, L.; Deng, J.; Li, Q.; Gao, F. Variation in the efficiency of nutrient removal in a pilot-scale natural wetland. Wetlands 2012, 32, 311–319. [Google Scholar] [CrossRef]
- Wu, T.; Qin, B.; Brookes, J.D.; Yan, W.; Ji, X.; Feng, J. Spatial distribution of sediment nitrogen and phosphorus in Lake Taihu from a hydrodynamics-induced transport perspective. Sci. Total Environ. 2019, 650, 1554–1565. [Google Scholar] [CrossRef]
- Blindow, I.; Hargeby, A.; Andersson, G. Seasonal changes of mechanisms maintaining clear water in a shallow lake with abundant. Aquat. Bot. 2002, 72, 315–334. [Google Scholar] [CrossRef]
- Langeland, K.A. Hydrilla verticillata (L.f.) Royle (Hydrocharitaceae), “the perfect aquatic weed”. Castanea 1996, 61, 293–304. [Google Scholar]
- Zhu, J.G.; Liu, X.; Deng, J.C.; Peng, J.X.; Zhang, H.T. Pollutant transport rates in the rivers around western Lake Taihu. J. Lake Sci. 2018, 30, 1509–1517. [Google Scholar]
- Carpenter, S.R.; Lodge, D.M. Effects of submersed macrophytes on ecosystem processes. Aquat. Bot. 1986, 3–4, 341–370. [Google Scholar] [CrossRef]
- Rybicki, N.B.; Landwehr, J.M. Long-term changes in abundance and diversity of macrophyte and waterfowl populations in an estuary with exotic macrophytes and improving water quality. Limnol. Oceanogr. 2007, 52, 1195–1207. [Google Scholar] [CrossRef]
- Zhu, J.Y.; Liu, B.; Wang, J.; Gao, Y.L.; Wu, Z.B. Study on the mechanism of allelopathic influence on cyanobacteria and chlorophytes by submerged macrophyte (Myriophyllum spicatum) and its secretion. Aquat. Toxicol. 2010, 98, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Paudel, R.; Grace, K.; Galloway, S.; Zamorano, M.; Jawitz, J. Effect of hydraulic resistance by vegetation on stage dynamics of a stormwater treatment wetland. J. Hydrol. 2013, 484, 74–85. [Google Scholar] [CrossRef]
- Wang, P.F.; Wang, C. Numerical model for flow through submerged vegetation regions in a shallow lake. J. Hydrodyn. 2011, 23, 170–178. [Google Scholar] [CrossRef]
- Xu, G.; Sun, Z.; Fang, W.; Liu, J.; Xu, X.; Lv, C. Release of phosphorus from sediments under wave-induced liquefaction. Water Res. 2018, 144, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.; Ma, R.; Xu, X.; Kong, F.; Zhang, S.; Kong, W.; Hao, J.; Shang, L. Two-decade reconstruction of algal blooms in China’s Lake Taihu. Environ. Sci. Technol. 2009, 43, 3522–3528. [Google Scholar] [CrossRef]
- Newman, J. Nuttall’s Pondweed; Centre for Ecology & Hydrology: Bailrigg, UK, 2001; p. 28. [Google Scholar]
- Boedeltje, G.; Bakker, J.P.; Bekker, R.M.; Groenendael, J.M.V.; Soesbergen, M. Plant dispersal in a lowland stream in relation to occurrence and three specific life-history traits of the species in the species pool. J. Ecol. 2003, 91, 855–866. [Google Scholar] [CrossRef]
- Sand-Jensen, K.; Riis, T.; Vestergaard, O.L.E.; Larsen, S.E. Macrophyte decline in Danish lakes and streams over the past 100 years. J. Ecol. 2000, 88, 1030–1040. [Google Scholar] [CrossRef]
- Sousa, W.T.Z. Hydrilla verticillata (Hydrocharitaceae), a recent invader threatening Brazil’s freshwater environments: A review of the extent of the problem. Hydrobiologia 2011, 669, 1–20. [Google Scholar] [CrossRef]
- Mony, C.; Koschnick, T.J.; Haller, W.T.; Muller, S. Competition between two invasive Hydrocharitaceae (Hydrilla verticillata (L.f.) (Royle) and Egeria densa (Planch)) as influenced by sediment fertility and season. Aquat. Bot. 2007, 86, 236–242. [Google Scholar] [CrossRef]
- Umetsu, C.A.; Evangelista, H.S.B.A.; Thomaz, S.M. The colonization, regeneration, and growth rates of macrophytes from fragments: A comparison between exotic and native submerged aquatic species. Aquat. Ecol. 2012, 46, 443–449. [Google Scholar] [CrossRef]
- Riis, T.; Sand-Jensen, K.A.J. Dispersal of plant fragments in small streams. Freshw. Biol. 2006, 51, 274–286. [Google Scholar] [CrossRef]
- Silveira, M.J.; Thomaz, S.M.; Mormul, R.P.; Camacho, F.P. Effects of desiccation and sediment type on early regeneration of plant fragments of three species of aquatic macrophytes. Int. Rev. Hydrobiol. 2009, 94, 169–178. [Google Scholar] [CrossRef]
| Zones | Area (km2) | Wave Conditions | Harvesting Start and End Date | Dominant Submerged Plant | Secondary Dominant Submerged Plant |
|---|---|---|---|---|---|
| Zone 1 (Z1) | 24.63 | strong | 3–26 May | P. malaianus | M. verticillatum |
| Zone 2 (Z2) | 35.90 | moderate | 3–30 May | P. maackianus | M. verticillatum |
| Zone 3 (Z3) | 36.44 | strong | 5–27 May | P. malaianus | M. verticillatum |
| Zone 4 (Z4) | 26.32 | moderate | 5–22 May | P. malaianus | H. verticillata |
| Zone 5 (Z5) | 33.12 | weak | 5 May–13 June | H. verticillata | P. malaianus |
| Zone 6 (Z6) | 33.81 | moderate | 26 August–14 September | P. malaianus | H. verticillata |
| Zone | Z2 | Z3 | Z4 | Z5 | Z6 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| C | B | C | B | C | B | C | B | C | B | |
| Z1 | F = 0.02 p = 0.89 | F = 1.32 p = 0.26 | F = 0.03 p = 0.86 | F = 0.367 p = 0.06 | F = 0.11 p = 0.74 | F = 0.59 p = 0.45 | F = 2.72 p = 0.11 | F = 27.78 p = 0.01 | F = 14.94 p = 0.01 | F = 39.12 p = 0.01 |
| Z2 | F = 0.12 p = 0.73 | F = 0.90 p = 0.35 | F = 0.24 p = 0.63 | F = 0.05 p = 0.83 | F = 3.74 p = 0.06 | F = 22.02 p = 0.01 | F = 17.65 p = 0.01 | F = 33.75 p = 0.01 | ||
| Z3 | F = 0.03 p = 0.87 | F = 1.02 p = 0.32 | F = 2.09 p = 0.16 | F = 14.66 p = 0.01 | F = 13.34 p = 0.01 | F = 24.74 p = 0.01 | ||||
| Z4 | F = 1.42 p = 0.24 | F = 19.96 p = 0.01 | F = 11.12 p = 0.01 | F = 32.01 p = 0.01 | ||||||
| Z5 | F = 5.23 p = 0.03 | F = 4.12 p = 0.05 | ||||||||
| Parameters | 2012/2013 | 2012/2014 | 2012/2015 | 2013/2014 | 2013/2015 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| F | p | F | p | F | p | F | p | F | p | |
| TP | 4.72 | 0.03 | 0.15 | 0.70 | 7.19 | 0.01 | 7.04 | 0.01 | 0.02 | 0.89 |
| TN | 1.8 | 0.19 | 6.99 | 0.01 | 5.90 | 0.02 | 16.40 | 0.01 | 15.15 | 0.01 |
| NH3-N | 1.28 | 0.26 | 20.20 | 0.01 | 19.64 | 0.01 | 28.93 | 0.01 | 28.35 | 0.01 |
| CODMn | 0.76 | 0.39 | 23.49 | 0.01 | 26.01 | 0.01 | 27.91 | 0.01 | 30.42 | 0.01 |
| Chla | 13.14 | 0.01 | 4.04 | 0.049 | 2.23 | 0.14 | 5.14 | 0.03 | 5.34 | 0.03 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, J.; Peng, Z.; Liu, X.; Deng, J.; Zhang, Y.; Hu, W. Response of Aquatic Plants and Water Quality to Large-Scale Nymphoides peltata Harvest in a Shallow Lake. Water 2019, 11, 77. https://doi.org/10.3390/w11010077
Zhu J, Peng Z, Liu X, Deng J, Zhang Y, Hu W. Response of Aquatic Plants and Water Quality to Large-Scale Nymphoides peltata Harvest in a Shallow Lake. Water. 2019; 11(1):77. https://doi.org/10.3390/w11010077
Chicago/Turabian StyleZhu, Jinge, Zhaoliang Peng, Xin Liu, Jiancai Deng, Yihui Zhang, and Weiping Hu. 2019. "Response of Aquatic Plants and Water Quality to Large-Scale Nymphoides peltata Harvest in a Shallow Lake" Water 11, no. 1: 77. https://doi.org/10.3390/w11010077
APA StyleZhu, J., Peng, Z., Liu, X., Deng, J., Zhang, Y., & Hu, W. (2019). Response of Aquatic Plants and Water Quality to Large-Scale Nymphoides peltata Harvest in a Shallow Lake. Water, 11(1), 77. https://doi.org/10.3390/w11010077




