Removal of Enteric Pathogens from Real Wastewater Using Single and Catalytic Ozonation
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Volcanic Rock
2.2. Experimental Procedures for Single and Catalytic Ozonation
2.3. Municipal Wastewater Sampling
2.4. Quantification of Culturable Bacteria—E. coli
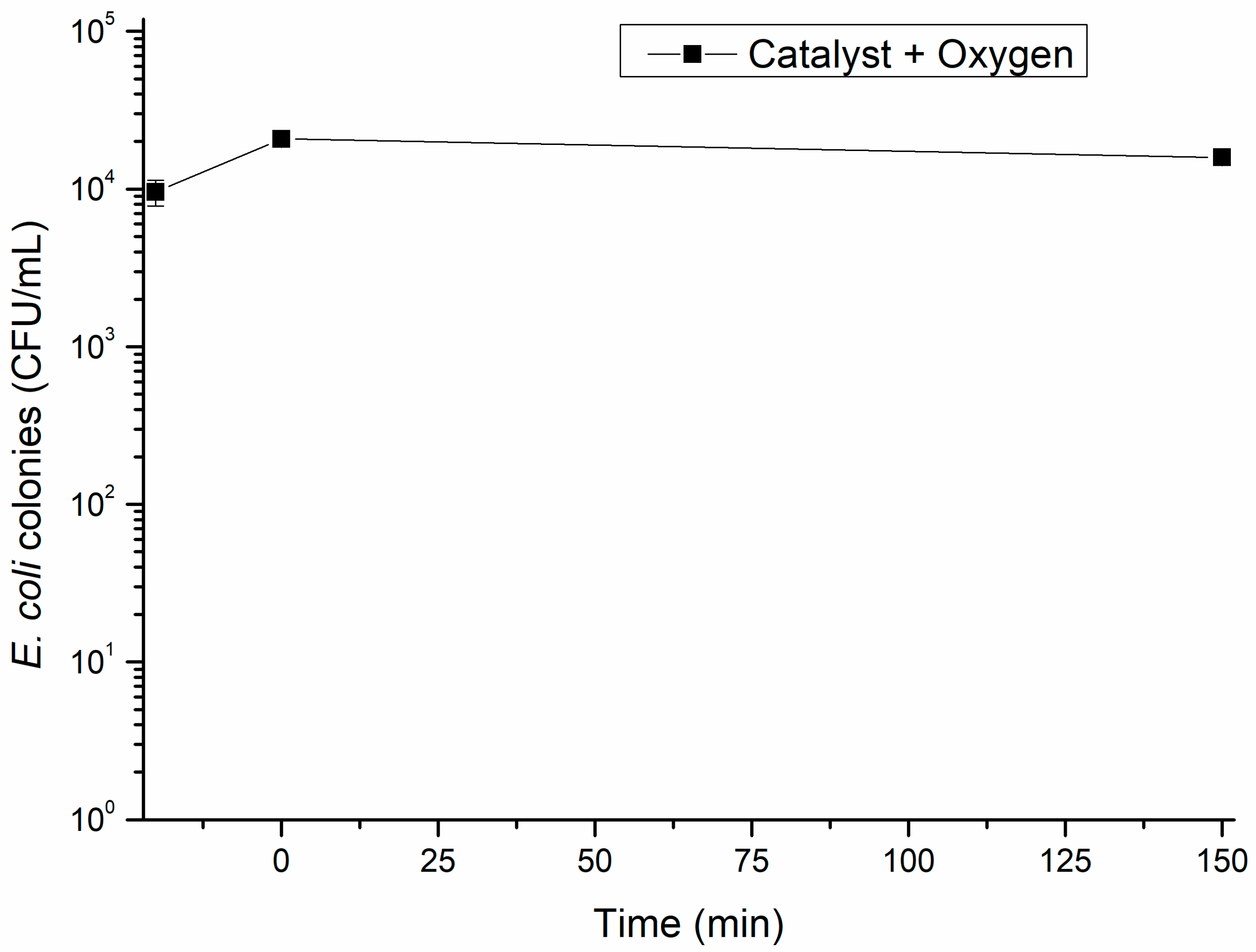
2.5. Regrowth
2.6. Viruses Analysis
3. Results
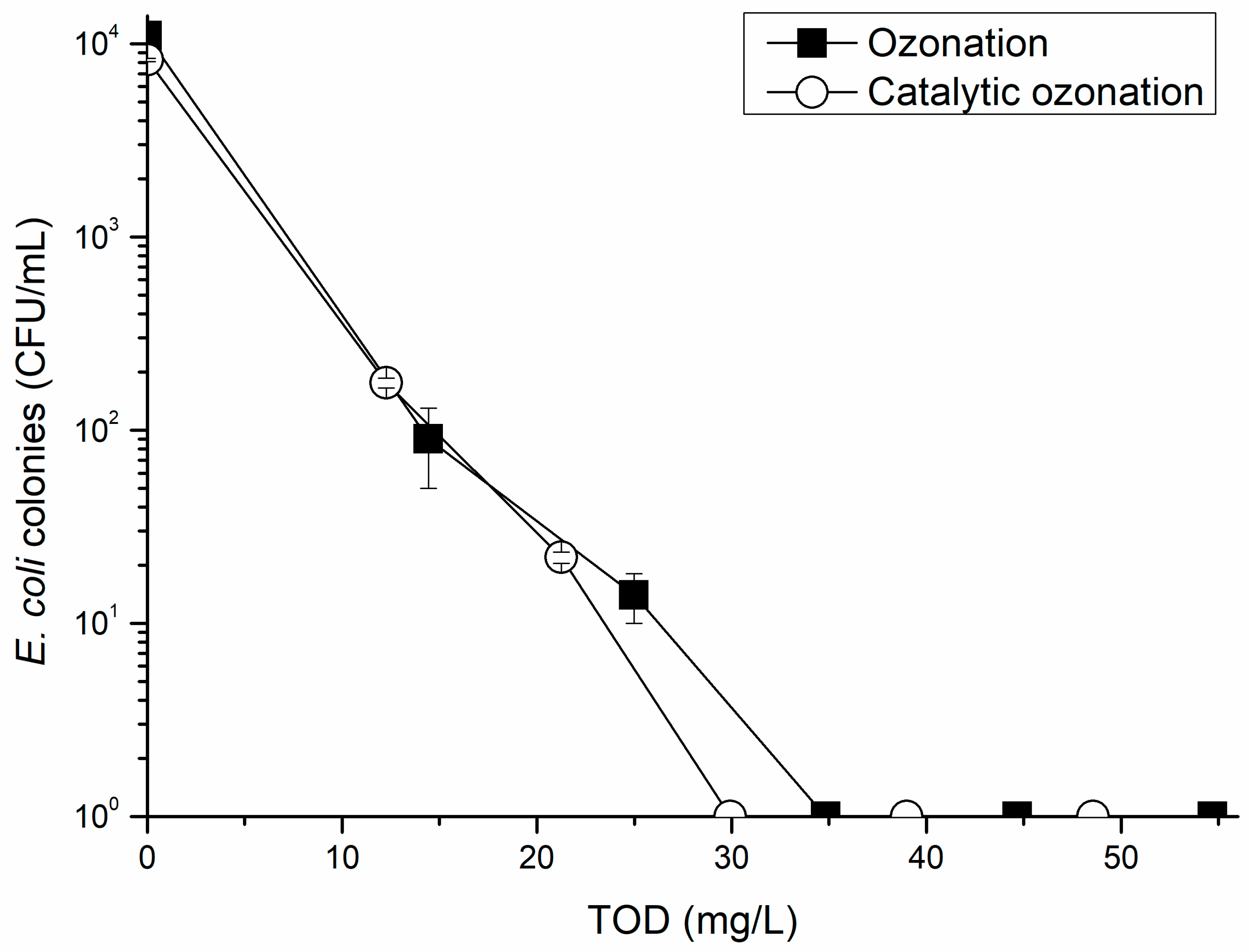
3.1. E. coli Removal from Municipal Wastewater
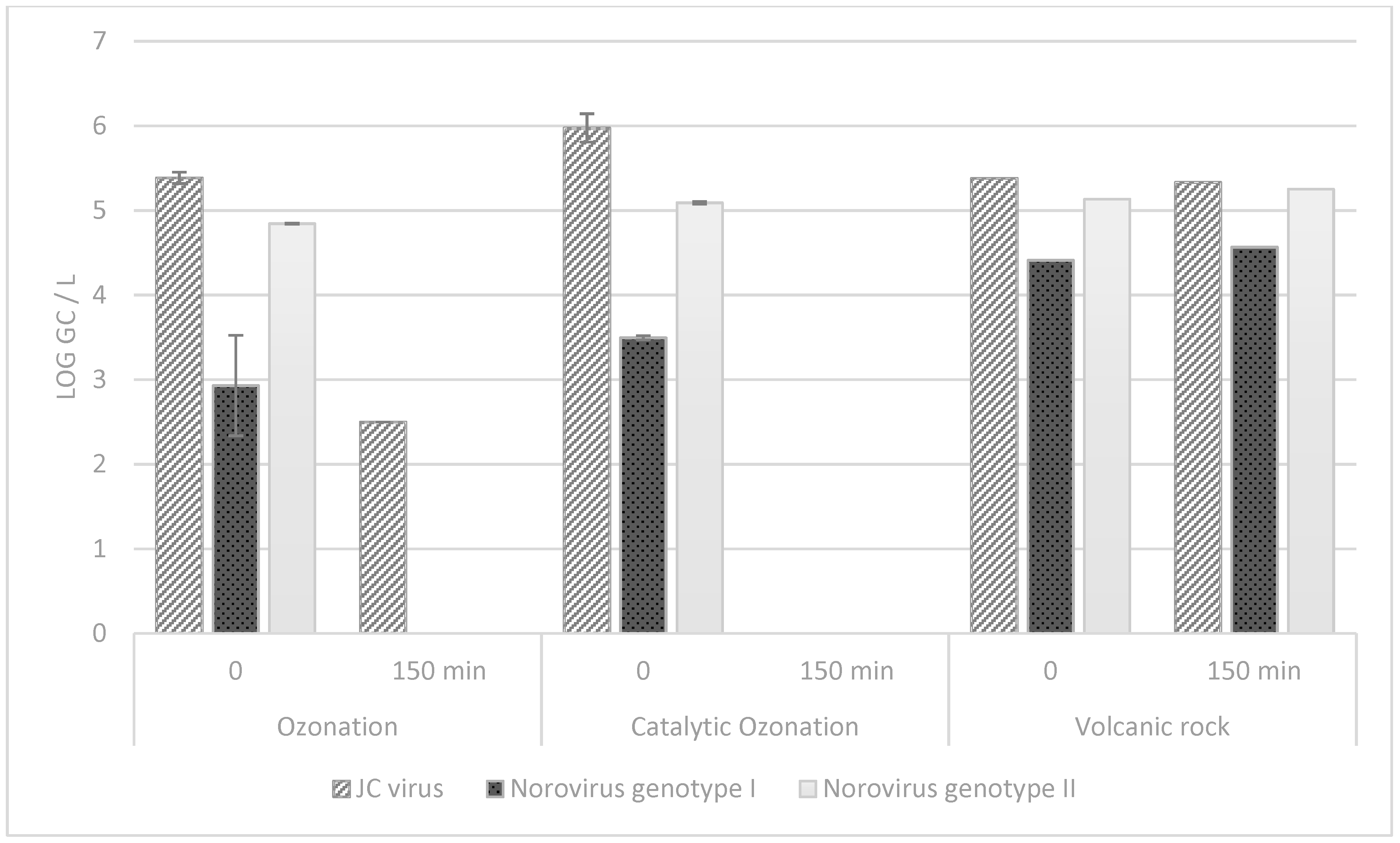
3.2. Virus Removal from Municipal Effluent
3.3. Economic Implications
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Nabeel, F.; Rasheed, T.; Bilal, M.; Li, C.; Yu, C.; Iqbal, H.M.N. Bio-Inspired Supramolecular Membranes: A Pathway to Separation and Purification of Emerging Pollutants. Sep. Purif. Rev. 2018, 1–17. [Google Scholar] [CrossRef]
- Hernandez-Vargas, G.; Sosa-Hernández, J.E.; Saldarriaga-Hernandez, S.; Villalba-Rodríguez, A.M.; Parra-Saldivar, R.; Iqbal, H.M.N. Electrochemical Biosensors: A Solution to Pollution Detection with Reference to Environmental Contaminants. Biosensors 2018, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Rasheed, T.; Bilal, M.; Nabeel, F.; Adeel, M.; Iqbal, H.M.N. Environmentally-related contaminants of high concern: Potential sources and analytical modalities for detection, quantification, and treatment. Environ. Int. 2019, 122, 52–66. [Google Scholar] [CrossRef] [PubMed]
- Diamond, J.; Munkittrick, K.; Kapo, K.E.; Flippin, J. A Framework for Screening Sites at Risk from Contaminants of Emerging Concern. Environ. Toxicol. Chem. 2015, 34, 2671–2681. [Google Scholar] [CrossRef] [PubMed]
- Gomes, I.B.; Simões, L.C.; Simões, M. The effects of emerging environmental contaminants on Stenotrophomonas maltophilia isolated from drinking water in planktonic and sessile states. Sci. Total Environ. 2018, 643, 1348–1356. [Google Scholar] [CrossRef] [PubMed]
- Rasheed, T.; Bilal, M.; Iqbal, H.M.N.; Hu, H.; Zhang, X. Reaction Mechanism and Degradation Pathway of Rhodamine 6G by Photocatalytic Treatment. Water Air Soil Pollut. 2017, 228, 291. [Google Scholar] [CrossRef]
- Bilal, M.; Rasheed, T.; Iqbal, H.M.N.; Hu, H.; Wang, W.; Zhang, X. Toxicological Assessment and UV/TiO2-Based Induced Degradation Profile of Reactive Black 5 Dye. Environ. Manag. 2018, 61, 171–180. [Google Scholar] [CrossRef]
- Tondera, K.; Klaer, K.; Gebhardt, J.; Wingender, J.; Koch, C.; Horstkott, M.; Strathmann, M.; Jurzik, L.; Hamza, I.A.; Pinnekamp, J. Reducing pathogens in combined sewer overflows using ozonation or UV irradiation. Int. J. Hyg. Environ. Health 2015, 218, 731–741. [Google Scholar] [CrossRef]
- Campos, C.J.A.; Avant, J.; Lowther, J.; Till, D.; Lees, D.N. Human norovirus in untreated sewage and effluents from primary, secondary and tertiary treatment processes. Water Res. 2016, 103, 224–232. [Google Scholar] [CrossRef]
- Molins-Delgado, D.; Díaz-Cruz, M.S.; Barceló, D. Ecological risk assessment associated to the removal of endocrine-disrupting parabens and benzophenone-4 in wastewater treatment. J. Hazard. Mater. 2016, 310, 143–151. [Google Scholar] [CrossRef]
- Gomes, J.; Costa, R.; Quinta-Ferreira, R.; Martins, R. Application of ozonation for pharmaceuticals and personal care products removal from water. Sci. Total Environ. 2017, 586, 265–283. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Sikora, P.; Rutgersson, C.; Lindh, M.; Brodin, T.; Björlenius, B.; Larsson, D.G.J.; Norder, H. Differential removal of human pathogenic viruses from sewage by conventional and ozone treatments. Int. J. Hyg. Environ. Health 2018, 221, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Osuolale, O.; Okoh, A. Human enteric bacteria and viruses in five wastewater treatment plants in the Eastern Cape, South Africa. J. Infect. Public Health 2017, 10, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Jacangelo, J.G.; Trussell, R.R. International report: Water and wastewater disinfection-trends, issues and practices. Water Sci. Technol. Water Supply 2002, 2, 147–157. [Google Scholar] [CrossRef]
- Nguyen, Y.; Sperandio, V. Enterohemorrhagic E. coli (EHEC) pathogenesis. Front. Cell. Infect. Microbiol. 2012, 2, 90. [Google Scholar] [CrossRef] [PubMed]
- US EPA. National Primary Drinking Water Regulations | US EPA. 2018. Available online: https://www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulations (accessed on 27 April 2018).
- Okoh, A.I.; Sibanda, T.; Gusha, S.S. Inadequately treated wastewater as a source of human enteric viruses in the environment. Int. J. Environ. Res. Public Health 2010, 7, 2620–2637. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, M.; Iker, B.C.; Pepper, I.L.; Gerba, C.P. Relative abundance and treatment reduction of viruses during wastewater treatment processes—Identification of potential viral indicators. Sci. Total Environ. 2014, 488–489, 290–296. [Google Scholar] [CrossRef]
- Gomes, J.; Matos, A.; Quinta-Ferreira, R.; Martins, R.C. Environmentally applications of invasive bivalves for water and wastewater decontamination. Sci. Total Environ. 2018, 630, 1016–1027. [Google Scholar] [CrossRef]
- Gomes, J.; Matos, A.; Gmurek, M.; Quinta-Ferreira, R.; Martins, R.C. Ozone and Photocatalytic Processes for Pathogens Removal from Water: A Review. Catalysts 2019, 9, 46. [Google Scholar] [CrossRef]
- Percival, S.; Yates, M.; Williams, D.; Chalmers, R.; Gray, N. Microbiology of Waterborne Diseases, 2nd ed.; Elsevier: Oxford, UK, 2014; pp. 537–615. [Google Scholar]
- Kasprzyk-Hordern, B.; Ziólek, M.; Nawrocki, J. Catalytic ozonation and methods of enhancing molecular ozone reactions in water treatment. Appl. Catal. B Environ. 2003, 46, 639–669. [Google Scholar] [CrossRef]
- Gomes, J.; Bednarczyk, K.; Gmurek, M.; Stelmachowski, M.; Zaleska-Medynska, A.; Bastos, F.; Quinta-Ferreira, M.; Costa, R.; Quinta-Ferreira, R.; Martins, R. Noble metal–TiO2 supported catalysts for the catalytic ozonation of parabens mixtures. Proc. Saf. Environ. Prot. 2017, 111, 148–159. [Google Scholar] [CrossRef]
- Demeestere, K.; Gago-Ferrero, P.; Van Langenhove, H.; Díaz-Cruz, M.; Barceló, D. Ozonation as an Advanced Treatment Technique for the Degradation of Personal Care Products in Water. Handb. Environ. Chem. 2014, 36, 375–397. [Google Scholar]
- Lazarova, V.; Liechti, P.; Savoye, P.; Hausler, R. Ozone disinfection: Main parameters for process design in wastewater treatment and reuse. J. Water Reuse Desalin. 2013, 3, 337. [Google Scholar] [CrossRef]
- Gomes, J.F.; Lopes, A.; Gonçalves, D.; Luxo, C.; Gmurek, M.; Costa, R.; Quinta-Ferreira, R.M.; Martins, R.C.; Matos, A. Biofiltration using C. fluminea for E.coli removal from water: Comparison with ozonation and photocatalytic oxidation. Chemosphere 2018, 208, 674–681. [Google Scholar] [CrossRef] [PubMed]
- Sousa, J.M.; Macedo, G.; Pedrosa, M.; Becerra-Castro, C.; Castro-Silva, S.; Pereira, M.F.R.; Silva, A.M.T.; Nunes, O.C.; Manaia, C.M. Ozonation and UV254 nm radiation for the removal of microorganisms and antibiotic resistance genes from urban wastewater. J. Hazard. Mater. 2017, 323, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Martins, R.C.; Quinta-Ferreira, R.M. Catalytic ozonation of phenolic acids over a Mn–Ce–O catalyst. Appl. Catal. B Environ. 2009, 90, 268–277. [Google Scholar] [CrossRef]
- Gomes, J.; Quinta-Ferreira, M.E.; Costa, R.; Quinta-Ferreira, R.M.; Martins, R.C. Parabens degradation using catalytic ozonation over volcanic rocks. Environ. Sci. Pollut. Res. 2018, 25, 7346–7357. [Google Scholar] [CrossRef] [PubMed]
- Shin, G.; Sobsey, M. Reduction of Norwalk Virus, Poliovirus 1, and Bacteriophage MS2 by Ozone Disinfection of Water. Appl. Environ. Microbiol. 2003, 69, 3975–3978. [Google Scholar] [CrossRef]
- Schaar, H.; Sommer, R.; Schürhagl, R.; Yillia, P.; Kreuzinger, N. Microorganism inactivation by an ozonation step optimized for micropollutant removal from tertiary effluent. Water Sci. Technol. 2013, 68, 311. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, B.; Liu, Y.; Cai, X.; Liu, X.; Dai, R. Removal and inactivation of virus by drinking water treatment in the presence of bromide or iodide. J. Water Chem. Technol. 2015, 37, 96–101. [Google Scholar] [CrossRef]
- Gomes, J.F.; Leal, I.; Bednarczyk, K.; Gmurek, M.; Stelmachowski, M.; Zaleska-Medynska, A.; Quinta-Ferreira, M.E.; Costa, R.; Quinta-Ferreira, R.M.; Martins, R.C. Detoxification of parabens using UV-A enhanced by noble metals—TiO2 supported catalysts. J. Environ. Chem. Eng. 2017, 5, 3065–3074. [Google Scholar] [CrossRef]
- Gomes, J.; Lopes, A.; Bednarczyk, K.; Gmurek, M.; Stelmachowski, M.; Zaleska-Medynska, A.; Quinta-Ferreira, M.; Costa, R.; Quinta-Ferreira, R.; Martins, R.; et al. Effect of Noble Metals (Ag, Pd, Pt) Loading over the Efficiency of TiO2 during Photocatalytic Ozonation on the Toxicity of Parabens. ChemEngineering 2018, 2, 4. [Google Scholar] [CrossRef]
- Giannakis, S.; Darakas, E.; Escalas-Cañellas, A.; Pulgarin, C. Solar disinfection modeling and post-irradiation response of Escherichia coli in wastewater. Chem. Eng. J. 2015, 281, 588–598. [Google Scholar] [CrossRef]
- Rafique, A.; Jiang, S.C. Genetic diversity of human polyomavirus JCPyV in Southern California wastewater. J. Water Health 2008, 6, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Ryschkewitsch, C.; Jensen, P.; Hou, J.; Fahle, G.; Fischer, S.; Major, E.O. Comparison of PCR-southern hybridization and quantitative real-time PCR for the detection of JC and BK viral nucleotide sequences in urine and cerebrospinal fluid. J. Virol. Methods 2004, 121, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, A.K.; Le Saux, J.C.; Parnaudeau, S.; Pommepuy, M.; Elimelech, M.; Le Guyader, F.S. Evaluation of removal of noroviruses during wastewater treatment, using real-time reverse transcription-PCR: Different behaviors of genogroups I and II. Appl. Environ. Microbiol. 2007, 73, 7891–7897. [Google Scholar] [CrossRef] [PubMed]
- Svraka, S.; Duizer, E.; Vennema, H.; de Bruin, E.; van der Veer, B.; Dorresteijn, B.; Koopmans, M. Etiological role of viruses in outbreaks of acute gastroenteritis in The Netherlands from 1994 through 2005. J. Clin. Microbiol. 2007, 45, 1389–1394. [Google Scholar] [CrossRef]
- Loisy, F.; Atmar, R.L.; Guillon, P.; Le Cann, P.; Pommepuy, M.; Le Guyader, F.S. Real-time RT-PCR for norovirus screening in shellfish. J. Virol. Methods 2005, 123, 1–7. [Google Scholar] [CrossRef]
- Kageyama, T.; Shinohara, M.; Uchida, K.; Fukushi, S.; Hoshino, F.B.; Kojima, S.; Takai, R.; Oka, T.; Takeda, N.; Katayama, K. Coexistence of multiple genotypes, including newly identified genotypes, in outbreaks of gastroenteritis due to Norovirus in Japan. J. Clin. Microbiol. 2004, 42, 2988–2995. [Google Scholar] [CrossRef]
- Costafreda, M.I.; Bosch, A.; Pintó, R.M. Development, evaluation, and standardization of a real-time TaqMan reverse transcription-PCR assay for quantification of hepatitis A virus in clinical and shellfish samples. Appl. Environ. Microbiol. 2006, 72, 3846–3855. [Google Scholar] [CrossRef]
- Rolfe, K.J.; Curran, M.D.; Mangrolia, N.; Gelson, W.; Alexander, G.J.; L’estrange, M.; Vivek, R.; Tedder, R.; Ijaz, S. First case of genotype 4 human hepatitis E virus infection acquired in India. J. Clin. Virol. 2010, 48, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Domenjoud, B.; Tatari, C.; Esplugas, S.; Baig, S. Ozone-based processes applied to municipal secondary effluents. Ozone Sci. Eng. 2011, 33, 243–249. [Google Scholar] [CrossRef]
- Hunt, N.; Mariñas, B. Kinetics of Escherichia coli inactivation with ozone. Water Res. 1997, 31, 1355–1362. [Google Scholar] [CrossRef]
- Hunt, N.K.; Mariñas, B.J. Inactivation of Escherichia coli with ozone: Chemical and inactivation kinetics. Water Res. 1999, 33, 2633–2641. [Google Scholar] [CrossRef]
- Bofill-Mas, S.; Girones, R. Role of the environment in the transmission of JC virus. J. Neurovirol. 2003, 91, 54–58. [Google Scholar] [CrossRef]
- Comerlato, J.; Souza-Campos, F.; Souza-Arantes, T.; Roos-Kulmann, M.I.; Trindade-Oliveira, M.; Rosado-Spilki, F.; Guedes-Frazzon, A.P.; Roehe, P.M.; Franco, A.C. Distribution and genetic diversity of the human polyomaviruses JC and BK in surface water and sewage treatment plant during 2009 in Porto Alegre, Southern Brazil. Braz. J. Biol. 2017, 77, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Lee, B.E.; Neumann, N.; Ashbolt, N.; Craik, S.; Maal-Bared, R.; Pang, X.L. Assessment of human virus removal during municipal wastewater treatment in Edmonton, Canada. J. Appl. Microbiol. 2015, 119, 1729–1739. [Google Scholar] [CrossRef]
- Albinana-Gimenez, N.; Clemente-Casares, P.; Bofill-Mas, S.; Hundesa, A.; Ribas, F.; Girones, R. Distribution of Human Polyoma-viruses, Adenoviruses, and Hepatitis E Virus in the Environment and in a Drinking-Water Treatment Plant. Environ. Sci. Technol. 2006, 40, 7416–7422. [Google Scholar] [CrossRef]
- Hollender, J.; Zimmermann, S.; Koepke, S.; Krauss, M.; McArdell, C.; Ort, C.; Singer, H.; von Gunten, U.; Siegrist, H. Elimination of Organic Micropollutants in a Municipal Wastewater Treatment Plant Upgraded with a Full-Scale Post-Ozonation Followed by Sand Filtration. Environ. Sci. Technol. 2009, 43, 7862–7869. [Google Scholar] [CrossRef]
| Ozonation | Catalytic Ozonation | |
|---|---|---|
| Time (min) | TOD (mgO3/L) | TOD (mgO3/L) |
| 0 | 0.00 | 0.00 |
| 30 | 14.4 | 12.3 |
| 60 | 25.0 | 21.3 |
| 90 | 34.8 | 29.9 |
| 120 | 44.7 | 39.0 |
| 150 | 54.7 | 48.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomes, J.; Frasson, D.; Quinta-Ferreira, R.M.; Matos, A.; Martins, R.C. Removal of Enteric Pathogens from Real Wastewater Using Single and Catalytic Ozonation. Water 2019, 11, 127. https://doi.org/10.3390/w11010127
Gomes J, Frasson D, Quinta-Ferreira RM, Matos A, Martins RC. Removal of Enteric Pathogens from Real Wastewater Using Single and Catalytic Ozonation. Water. 2019; 11(1):127. https://doi.org/10.3390/w11010127
Chicago/Turabian StyleGomes, João, Danilo Frasson, Rosa M. Quinta-Ferreira, Ana Matos, and Rui C. Martins. 2019. "Removal of Enteric Pathogens from Real Wastewater Using Single and Catalytic Ozonation" Water 11, no. 1: 127. https://doi.org/10.3390/w11010127
APA StyleGomes, J., Frasson, D., Quinta-Ferreira, R. M., Matos, A., & Martins, R. C. (2019). Removal of Enteric Pathogens from Real Wastewater Using Single and Catalytic Ozonation. Water, 11(1), 127. https://doi.org/10.3390/w11010127








