Treatment of Organics Contaminated Wastewater by Ozone Micro-Nano-Bubbles
Abstract
1. Introduction
2. Materials and Methods
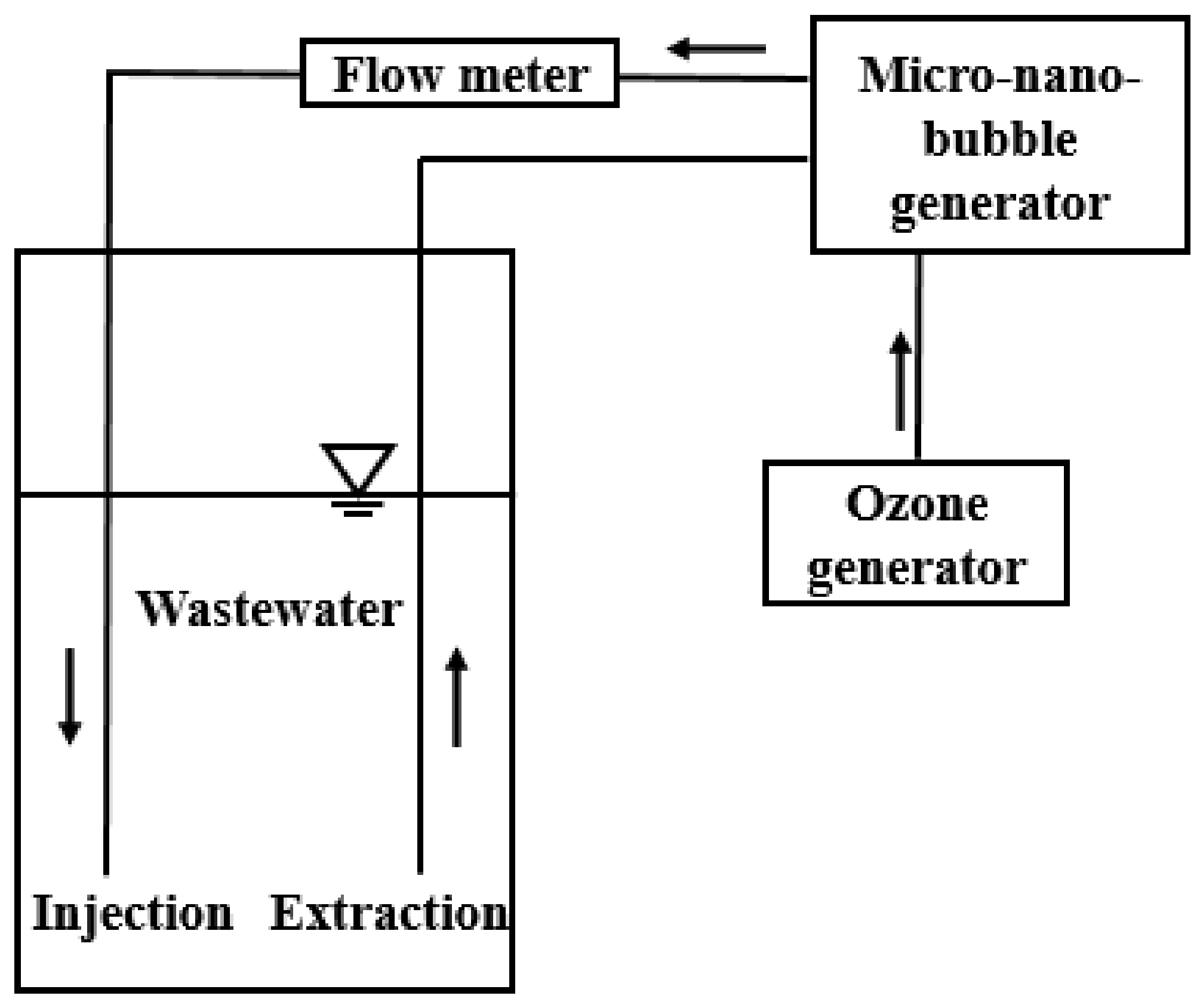
2.1. Experimental Facilities
2.1.1. Micro-Nano-Bubble Generator
2.1.2. Ozone Generator
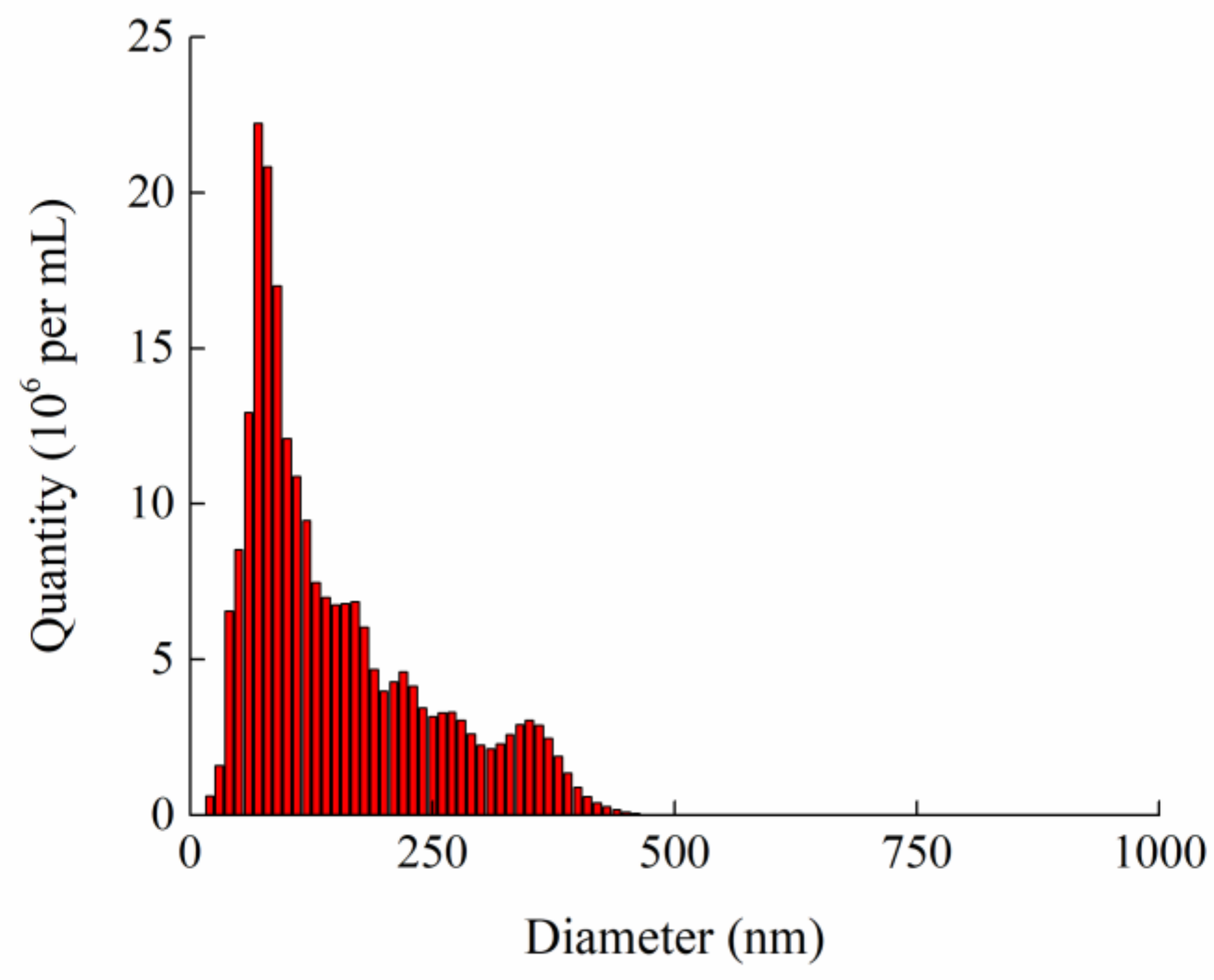
2.1.3. Size Distribution Analyzer
2.1.4. UV Spectrophotometer
2.1.5. Gas Chromatograph
2.1.6. Chemical Oxygen Demand (COD) Measurement Devices
2.1.7. Reaction Tank
2.2. Experimental Setup
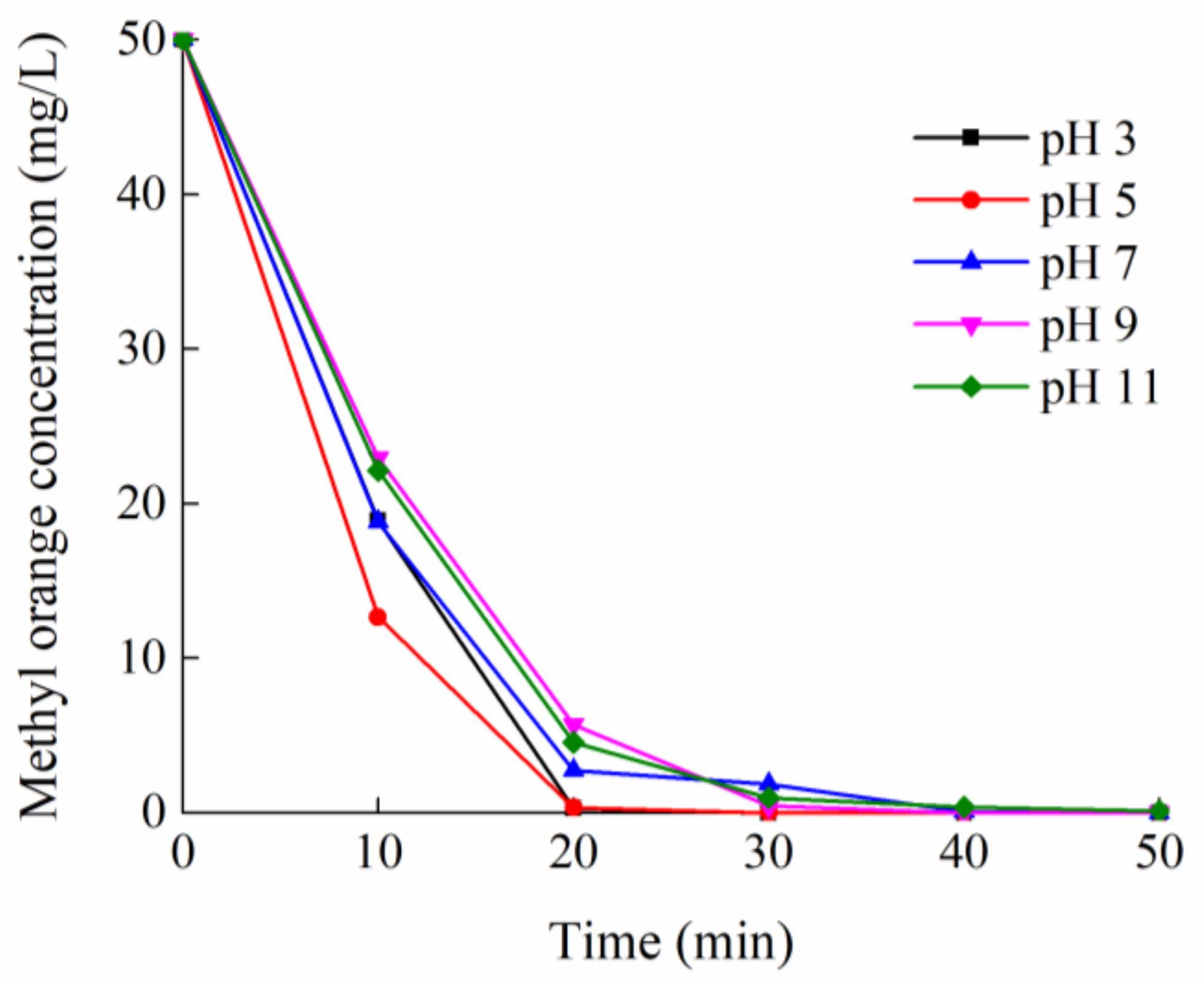
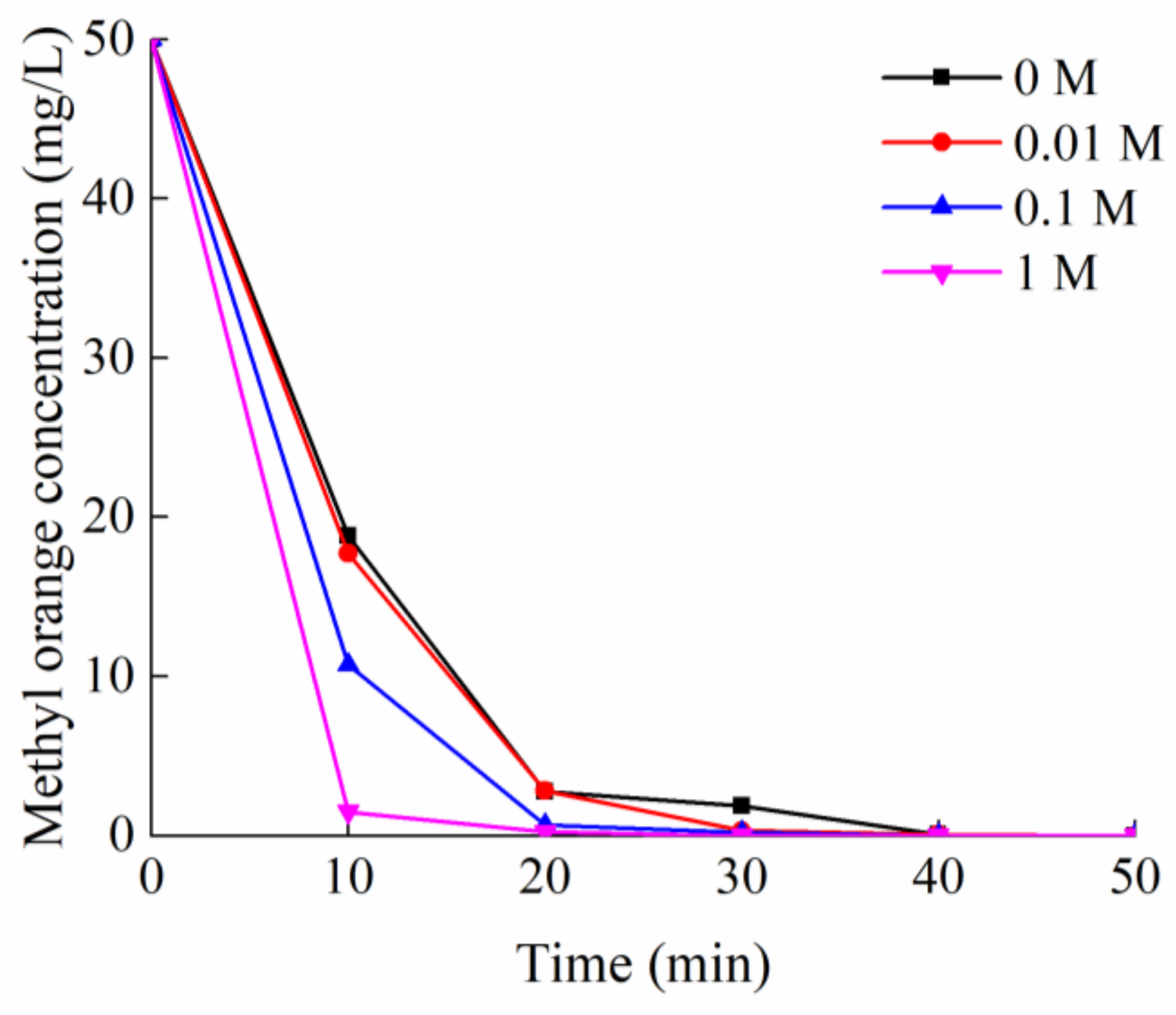
2.2.1. Effects of pH and Salinity
2.2.2. Treatment of Industrial Wastewater with High Salinity
2.2.3. Treatment of Contaminated Groundwater with Complex Organic Components
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kolpin, D.W.; Furlong, E.T.; Meyer, M.T.; Thurman, E.M.; Zaugg, S.D.; Barber, L.B.; Buxton, H.T. Pharmaceuticals, hormones, and other organic wastewater contaminants in U.S. streams, 1999–2000: A national reconnaissance. Environ. Sci. Technol. 2002, 36, 1202–1211. [Google Scholar] [CrossRef] [PubMed]
- Lapworth, D.J.; Baran, N.; Stuart, M.E.; Ward, R.S. Emerging organic contaminants in groundwater: A review of sources, fate and occurrence. Environ. Pollut. 2012, 163, 287–303. [Google Scholar] [CrossRef] [PubMed]
- Ali, I.; Asim, M.; Khan, T.A. Low cost adsorbents for the removal of organic pollutants from wastewater. J. Environ. Manag. 2012, 113, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.C.; De Voogt, P. Persistent organic pollutants (POPs): State of the science. Environ. Pollut. 1999, 100, 209–221. [Google Scholar] [CrossRef]
- Liu, Z.; Kanjo, Y.; Mizutani, S. Removal mechanisms for endocrine disrupting compounds (EDCs) in wastewater treatment- physical means, biodegradation, and chemical advanced oxidation: A review. Sci. Total Environ. 2009, 407, 731–748. [Google Scholar] [CrossRef] [PubMed]
- Ikehata, K.; Gamal EI-Din, M.; Snyder, S.A. Ozonation and advanced oxidation treatment of emerging organic pollutants in water and wastewater. Ozone-Sci. Eng. 2008, 30, 21–26. [Google Scholar] [CrossRef]
- Lefebvre, O.; Moletta, R. Treatment of organic pollution in industrial saline wastewater: A literature review. Water Res. 2006, 40, 3671–3682. [Google Scholar] [CrossRef]
- Sun, L.; Li, Y.; Li, A.M. Treatment of actual chemical wastewater by a heterogeneous fenton process using natural pyrite. Int. J. Environ. Res. Public Health 2015, 12, 13762–13778. [Google Scholar] [CrossRef]
- Gogate, P.R.; Pandit, A.B. A review of imperative technologies for wastewater treatment I: Oxidation technologies at ambient conditions. Adv. Environ. Res. 2004, 8, 501–551. [Google Scholar] [CrossRef]
- Asghar, A.; Raman, A.A.A.; Daud, W.M.A.W. Advanced oxidation processes for in-sit production of hydrogen peroxide/hydroxyl radicals for textile wastewater treatment: A review. J. Clean. Prod. 2015, 87, 826–838. [Google Scholar] [CrossRef]
- Oturan, M.A.; Aaron, J.J. Advanced oxidation processes in water/wastewater treatment: Principles and applications. A review. Crit. Rev. Env. Sci. Technol. 2014, 44, 2577–2641. [Google Scholar] [CrossRef]
- Mitani, M.M.; Keller, A.A.; Bunton, C.A.; Rinker, R.G.; Sandall, O.C. Kinetics and products of reactions of MTBE with ozone and ozone/hydrogen peroxide in water. J. Hazard. Mater. 2002, 89, 197–212. [Google Scholar] [CrossRef]
- Llyas, H.; Masih, L.; van der Hoek, J.P. Disinfection methods for swimming pool water: Byproduct formation and control. Water 2018, 10, 797. [Google Scholar]
- Gomes, J.; Costa, R.; Quinta-Ferreira, R.M.; Martins, R.C. Application of ozonation for pharmaceuticals and personal care products removal from water. Sci. Total Environ. 2017, 586, 265–283. [Google Scholar] [CrossRef] [PubMed]
- Oller, I.; Malato, S.; Sánchez-Pérez, J.A. Combination of advanced oxidation processes and biological treatments for wastewater decomtamination—A review. Sci. Total Environ. 2011, 409, 4141–4166. [Google Scholar] [CrossRef] [PubMed]
- Attard, P.; Moody, M.P.; Tyrrell, J.W.G. Nanobubbles, The big picture. Phys. A Stat. Mech. Appl. 2002, 314, 696–705. [Google Scholar] [CrossRef]
- Chu, L.B.; Xing, X.H.; Yu, A.F.; Zhou, Y.N.; Sun, X.L.; Jurcik, B. Enhanced ozonation of simulated dyestuff wastewater by microbubbles. Chemosphere 2007, 68, 1854–1860. [Google Scholar] [CrossRef]
- Hu, L.M.; Xia, Z.R. Application of ozone micro-nano-bubbles to groundwater remediation. J. Hazard. Mater. 2018, 342, 446–453. [Google Scholar] [CrossRef]
- Seddon, J.R.T.; Lohse, D.; Ducker, W.A.; Craig, V.S.J. A deliberation on nanobubbles at surfaces and in bulk. ChemPhysChem 2012, 13, 2179–2187. [Google Scholar] [CrossRef]
- Azevedo, A.; Etchepare, R.; Calgaroto, S.; Rubio, J. Aqueous dispersion of nanobubbles: Generation, properties and features. Min. Eng. 2016, 94, 29–37. [Google Scholar] [CrossRef]
- Li, H.; Hu, L.; Song, D.; Lin, F. Characteristics of micro-nano bubbles and potential application in groundwater bioremediation. Water Environ. Res. 2014, 86, 844–851. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Ng, W.J.; Liu, Y. Principle and applications of microbubble and nanobubble technology for water treatment. Chemosphere 2011, 84, 1175–1180. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.C. Effects of factors and interacted factors on the optimal decolorization process of methyl orange by ozone. Water Res. 2000, 34, 974–982. [Google Scholar] [CrossRef]
- Xia, Z.R.; Hu, L.M. Remediation of organics contaminated groundwater by ozone micro-nano bubble. In Proceedings of the 15th Asian Regional Conference on Soil Mechanics and Geotechnical Engineering, Fukuoka, Japan, 9–13 November 2015; pp. 1978–1981. [Google Scholar]
- Sevimli, M.F.; Sarikaya, H.Z. Ozone treatment of textile effluents and dyes: Effect of applied ozone dose, pH and dye concentration. J. Chem. Technol. Biotechnol. 2002, 77, 842–850. [Google Scholar] [CrossRef]
- Bilińska, L.; Gmurek, M.; Ledakowicz, S. Comparison between industrial and simulated textile wastewater treatment by AOPs—Biodegradability, toxicity and cost assessment. Chem. Eng. J. 2016, 306, 550–559. [Google Scholar] [CrossRef]
- Zhang, H.; Duan, L.J.; Zhang, D.B. Decolorization of methyl orange by ozonation in combination with ultrasonic irradiation. J. Hazard. Mater. 2006, 138, 53–59. [Google Scholar] [CrossRef]
- Staehelin, J.; Hoigne, J. Decomposition of ozone in water in the presence of organic solutes as promoters and inhibitors of radical chain reactions. Environ. Sci. Technol. 1985, 19, 1206–1213. [Google Scholar] [CrossRef] [PubMed]
- Zaghouane-Boudiaf, H.; Boutahala, M.; Arab, L. Removal of methyl orange from aqueous solution by uncalcined and calcined MgNiAl layered double hydroxides (LDHs). Chem. Eng. J. 2012, 187, 142–149. [Google Scholar] [CrossRef]
- He, P.T.; Wang, L.; Xue, J.J.; Cao, Z.B. Electrolytic treatment of methyl orange in aqueous solution using three-dimensional electrode reactor coupling ultrasonics. Environ. Technol. 2010, 31, 417–422. [Google Scholar] [CrossRef]
- Gül, Ş.; Özcan, Ö.; Erbatur, O. Ozonation of Cl Reactive Red 194 and Cl Reactive Yellow 145 in aqueous solution in the presence of granular activated carbon. Dyes Pigment. 2007, 75, 426–431. [Google Scholar] [CrossRef]
- Constapel, M.; Schellenträger, M.; Marzinkowski, J.M.; Gäb, S. Degradation of reactive dyes in wastewater from the textile industry by ozone: Analysis of the products by accurate masses. Water. Res. 2009, 43, 733–743. [Google Scholar] [CrossRef] [PubMed]
- Yuan, R.X.; Ramjaun, S.N.; Wang, Z.H.; Liu, J.S. Effects of chloride ion on degradation of Acid Orange 7 by sulfate radical-based advanced oxidation process: Implications for formation of chlorinated aromatic compounds. J. Hazard. Mater. 2011, 196, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.H.; Yuan, R.X.; Guo, Y.G.; Xu, L.; Liu, J.S. Effect of chloride ions on bleaching of azo dyes by Co2+/oxone regent: Kinetic analysis. J. Hazard. Mater. 2011, 190, 1083–1087. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Ray, W.A. The surface tension of solutions of electrolytes as a function of the concentration. III. Sodium chloride. J. Am. Chem. Soc. 1941, 63, 3262–3263. [Google Scholar] [CrossRef]
- Li, H.Z.; Hu, L.M.; Song, D.J.; Al-Tabbaa, A. Subsurface transport behavior of micro-nano bubbles and potential applications for groundwater remediation. Int. J. Environ. Res. Public Health 2014, 11, 473–486. [Google Scholar] [CrossRef] [PubMed]
- Bilińska, L.; Gmurek, M.; Ledakowicz, S. Textile wastewater treatment by AOPs for brine reuse. Process Saf. Environ. 2017, 109, 420–428. [Google Scholar] [CrossRef]
- Liu, Z.H.; Guo, W.L.; Han, X.M.; Li, X.H.; Zhang, K. In situ remediation of ortho-nitrochlorobenzene in soil by dual oxidants (hydrogen peroxide/persulfate). Environ. Sci. Pollut. Res. 2016, 23, 19707–19712. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.R.; Hu, L.M.; Kusaba, S.; Song, D.J. Remediation of TCE contaminated site by ozone micro-nano-bubbles. In Proceedings of the International Congress on Environmental Geotechnics, Hangzhou, China, 28 October–1 November 2018; pp. 796–803. [Google Scholar]
- Petrella, A.; Mascolo, G.; Murgolo, S.; Petruzzelli, V.; Ranieri, E.; Spasiano, D.; Petruzzelli, D. Photocatalytic oxidation of organic micro-pollutants: Pilot plant investigation and mechanistic aspects of the degradation reaction. Chem. Eng. Commun. 2016, 203, 1298–1307. [Google Scholar] [CrossRef]
- Devi, L.G.; Kumar, S.G.; Reddy, K.M.; Munikrishnappa, C. Photo degradation of methyl orange an azo dye by advanced Fenton process using zero valent metallic iron: Influence of various reaction parameters and its degradation mechanism. J. Hazard. Mater. 2009, 164, 459–467. [Google Scholar] [CrossRef]
- Ge, D.M.; Zeng, Z.Q.; Arowo, M.; Zou, H.K.; Chen, J.F.; Shao, L. Degradation of methyl orange by ozone in the presence of ferrous and persulfate ions in a rotating packed bed. Chemosphere 2016, 146, 413–418. [Google Scholar] [CrossRef]
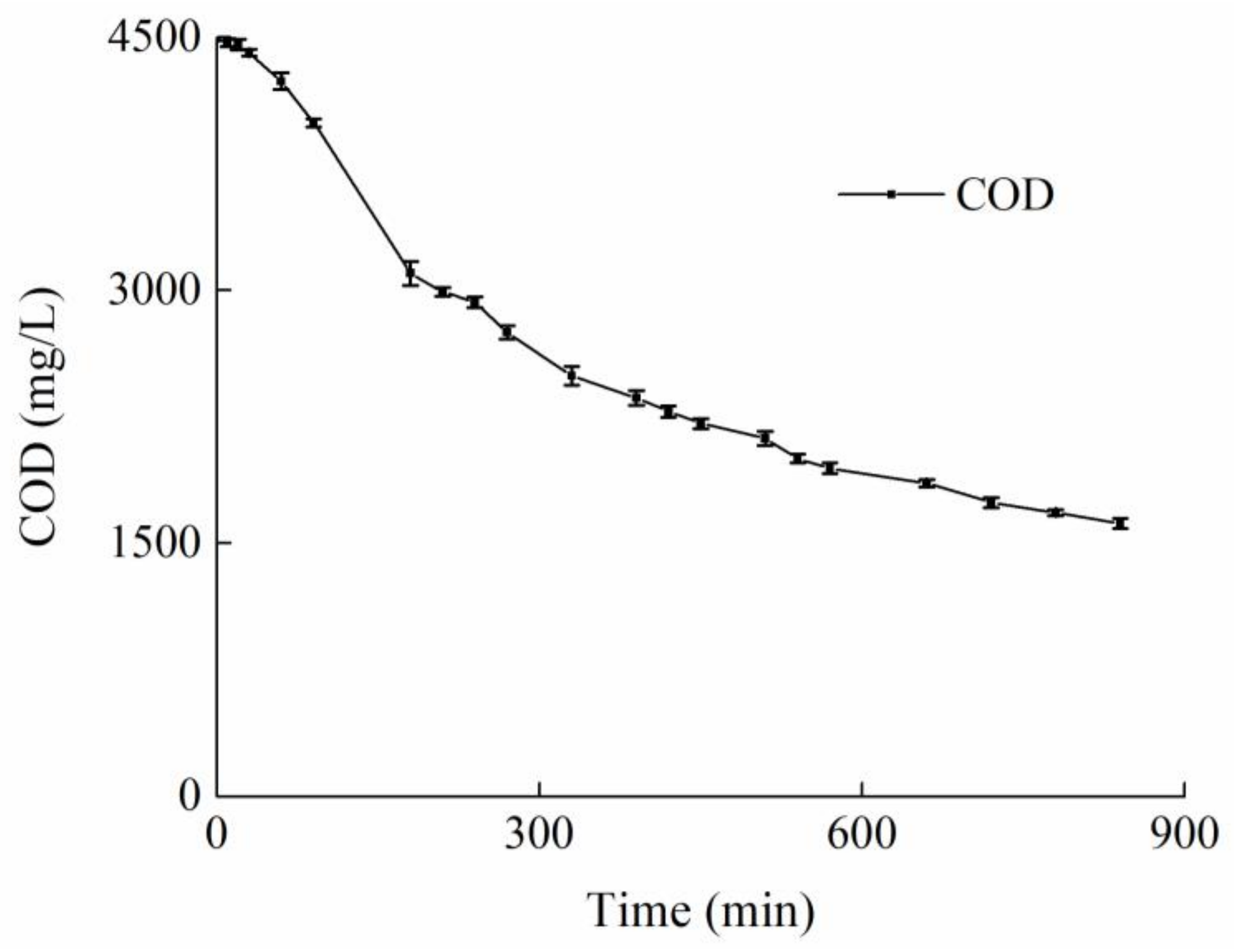
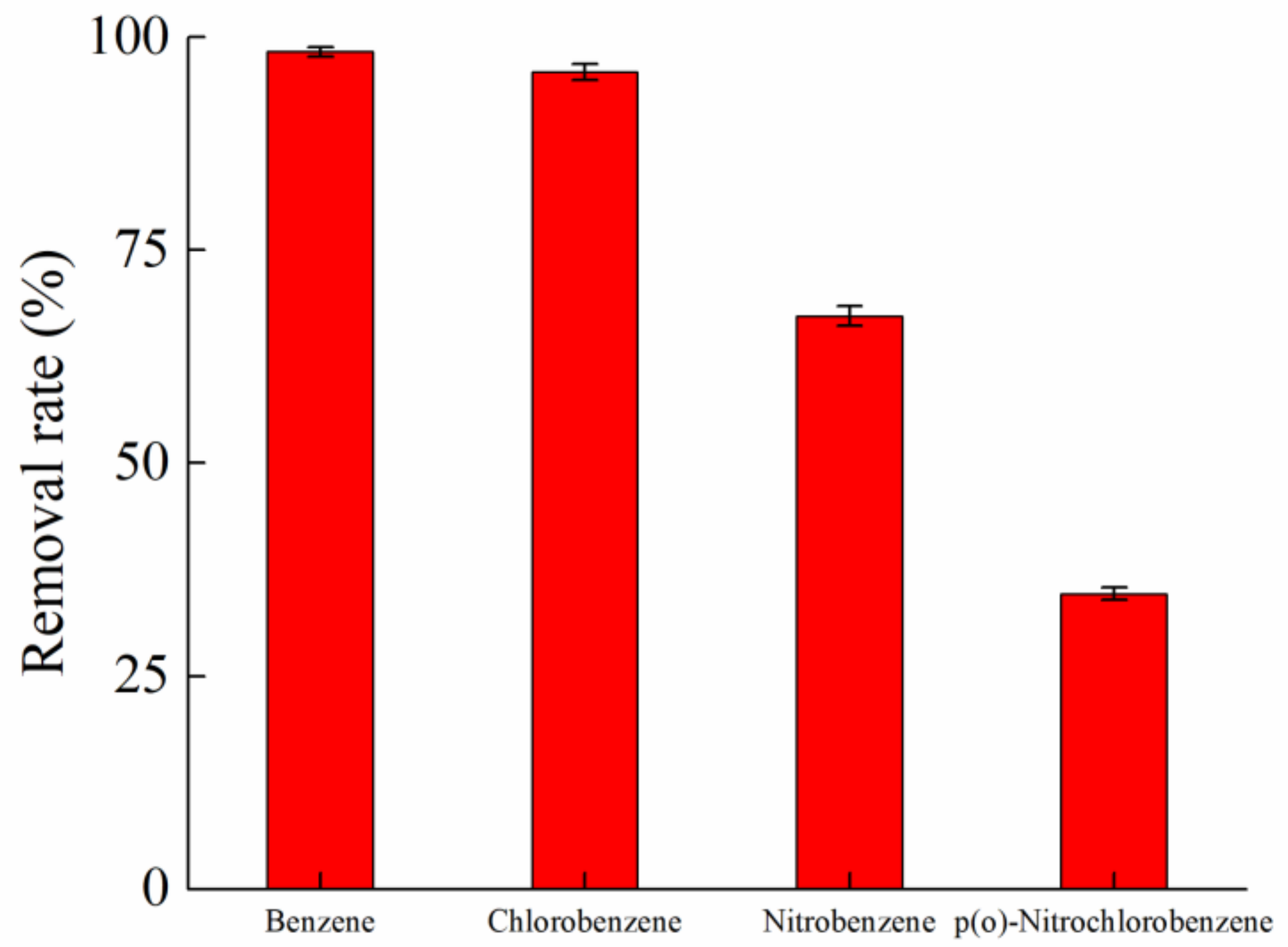
| Ion | K+ | Na+ | Cl− | SO42− | NO3− |
|---|---|---|---|---|---|
| Concentration (mg/L) | 1710 ± 0.02 | 114000 ± 0.02 | 123000 ± 0.02 | 58500 ± 0.09 | 23300 ± 0.08 |
| Contaminant | Benzene | Chlorobenzene | Nitrobenzene | p(o)-nitrochlorobenzene |
|---|---|---|---|---|
| Initial concentration (mg/L) | 30.5 ± 5.1 | 14.8 ± 2.5 | 502.8 ± 57.5 | 9.5 ± 0.6 |
| Initial pH | 3.0 ± 0.1 | 5.0 ± 0.1 | 7.0 ± 0.1 | 9.0 ± 0.1 | 11.0 ± 0.1 |
| Final pH | 2.5 ± 0.1 | 3.9 ± 0.1 | 6.8 ± 0.1 | 8.2 ± 0.1 | 9.0 ± 0.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xia, Z.; Hu, L. Treatment of Organics Contaminated Wastewater by Ozone Micro-Nano-Bubbles. Water 2019, 11, 55. https://doi.org/10.3390/w11010055
Xia Z, Hu L. Treatment of Organics Contaminated Wastewater by Ozone Micro-Nano-Bubbles. Water. 2019; 11(1):55. https://doi.org/10.3390/w11010055
Chicago/Turabian StyleXia, Zhiran, and Liming Hu. 2019. "Treatment of Organics Contaminated Wastewater by Ozone Micro-Nano-Bubbles" Water 11, no. 1: 55. https://doi.org/10.3390/w11010055
APA StyleXia, Z., & Hu, L. (2019). Treatment of Organics Contaminated Wastewater by Ozone Micro-Nano-Bubbles. Water, 11(1), 55. https://doi.org/10.3390/w11010055






