Potential Impacts of Induced Bank Filtration on Surface Water Quality: A Conceptual Framework for Future Research
Abstract
1. Introduction
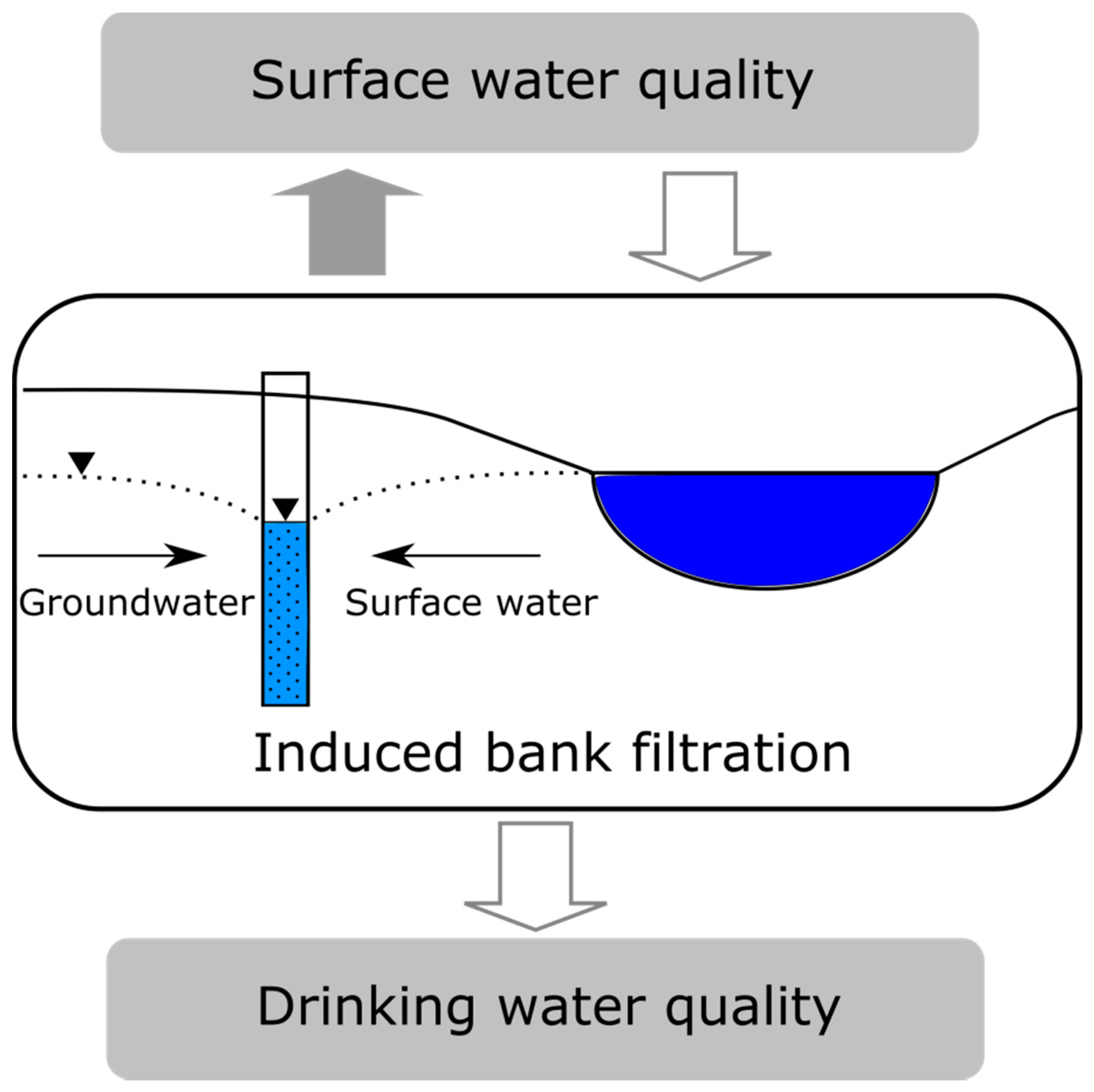
2. Use of Induced Bank Filtration (IBF) and Source Surface Water Bodies
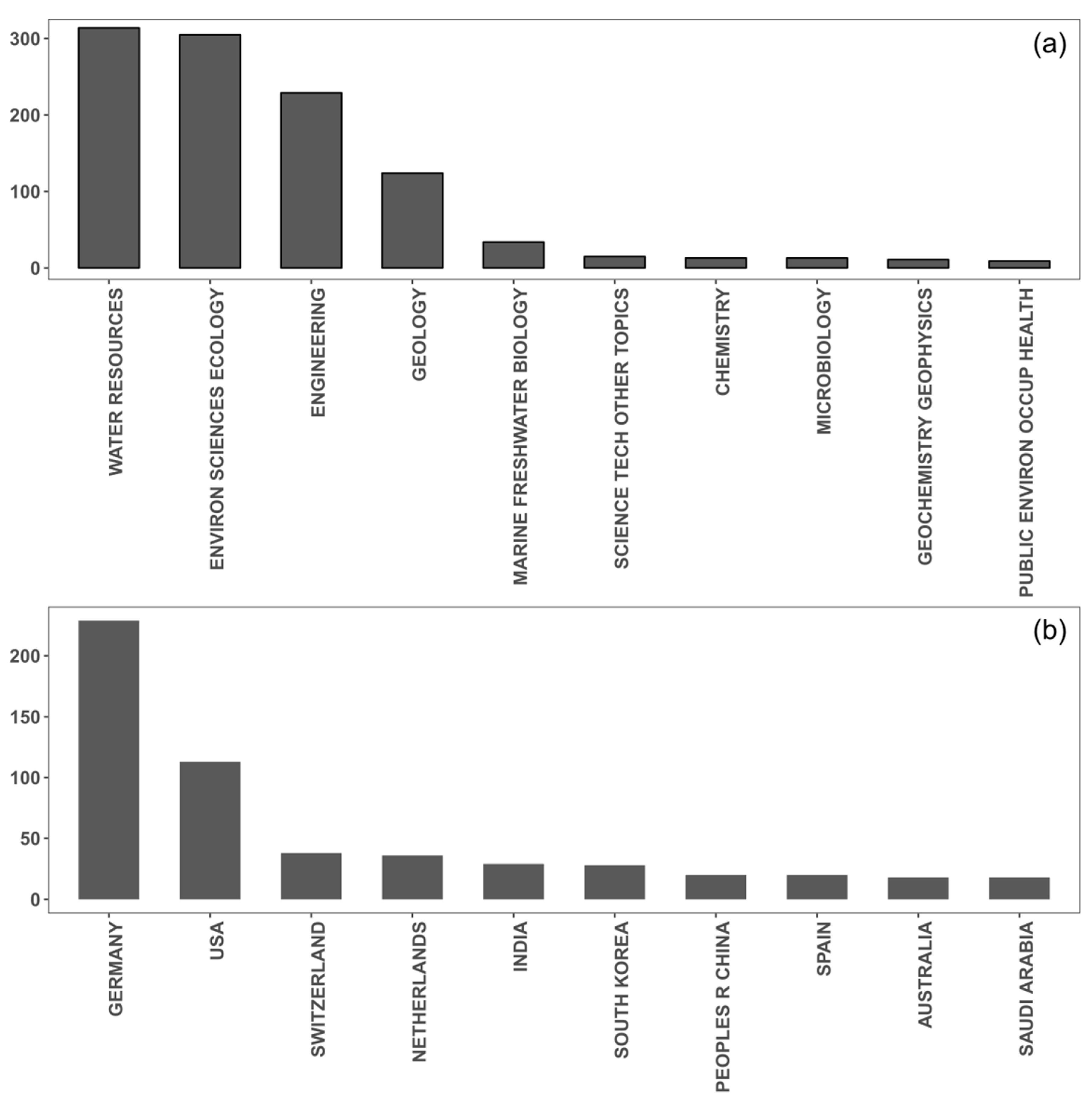
2.1. Worldwide Use of IBF and Affected Surface Waters
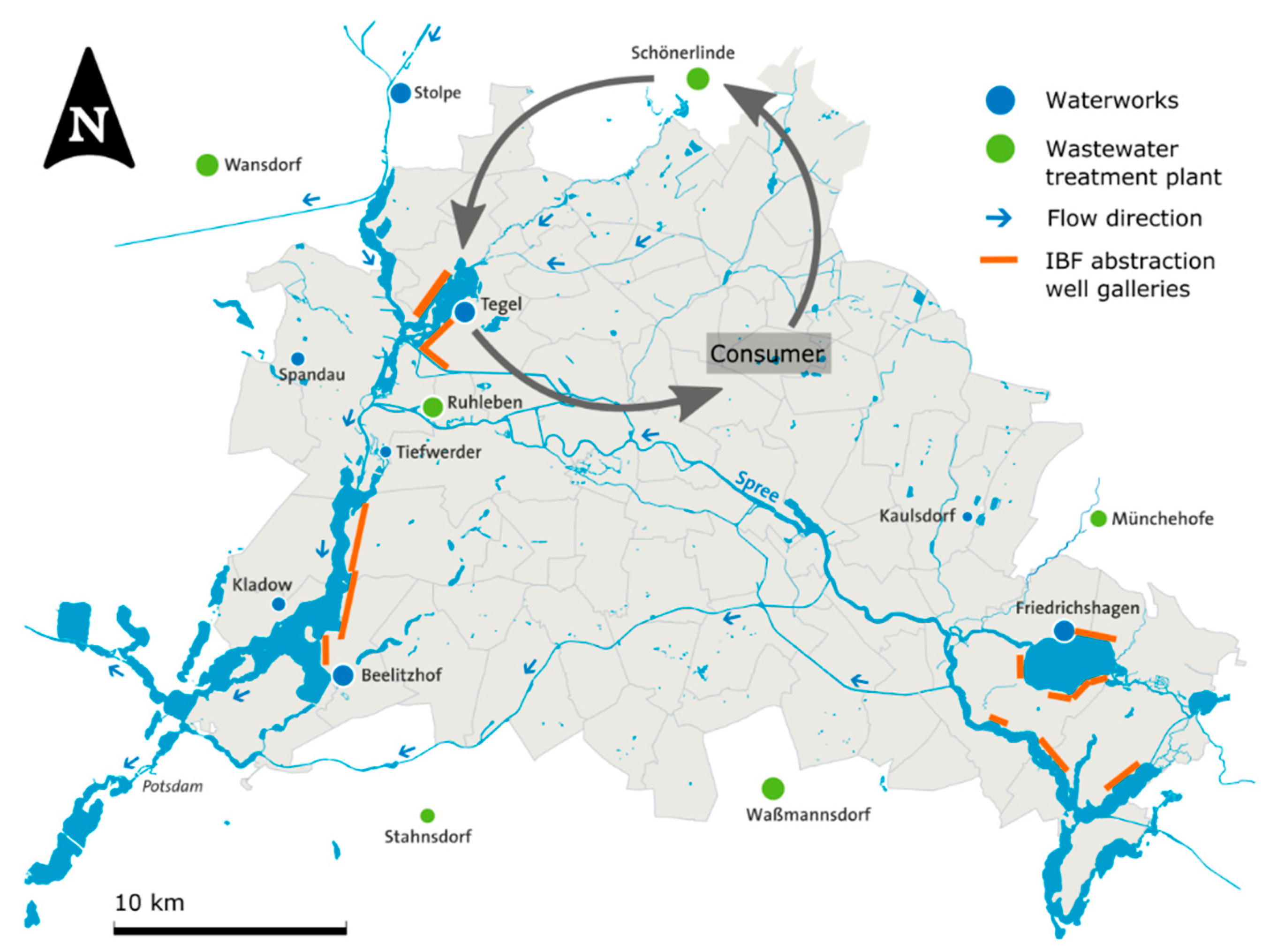
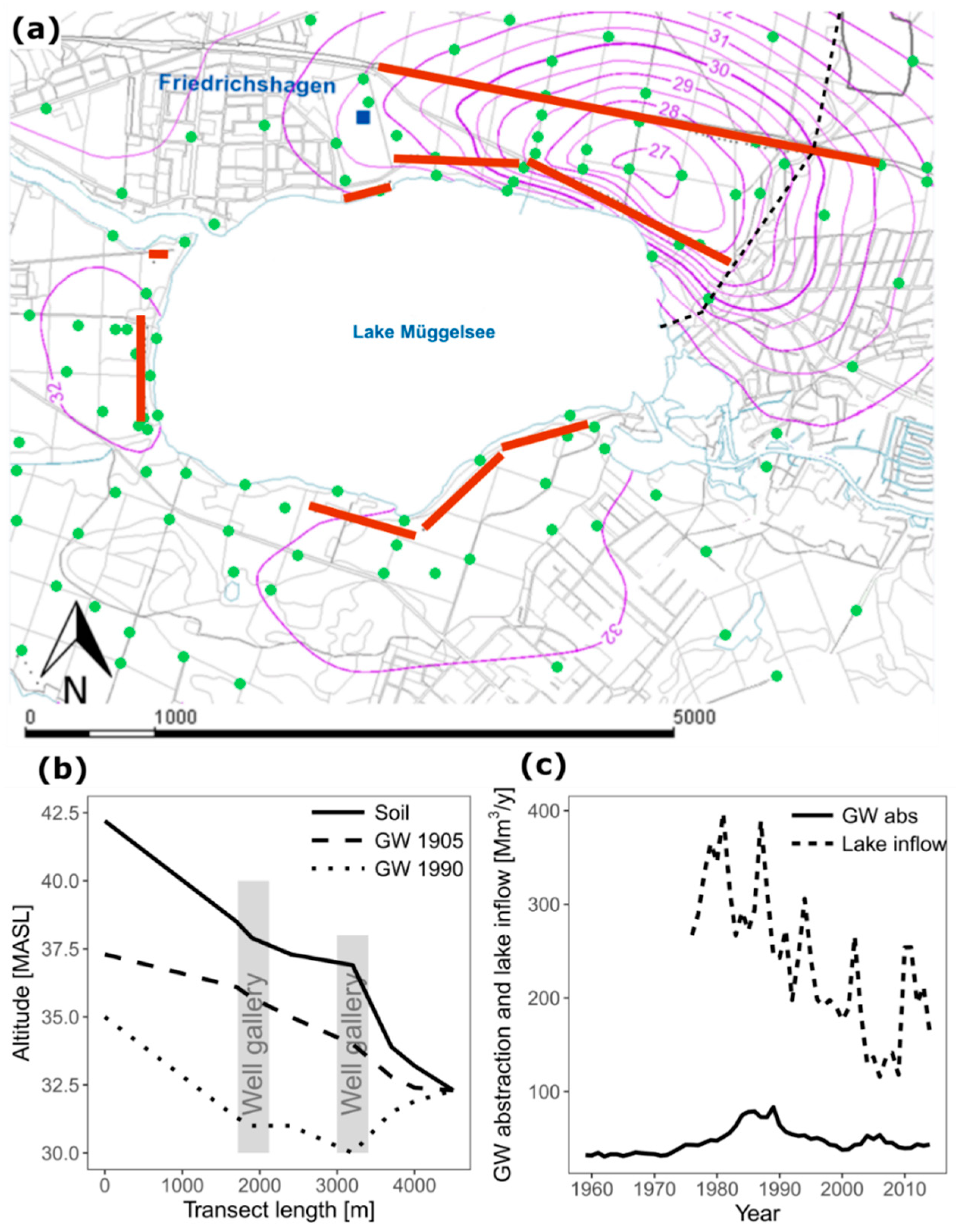
2.2. Example of IBF Application in Berlin (Germany)
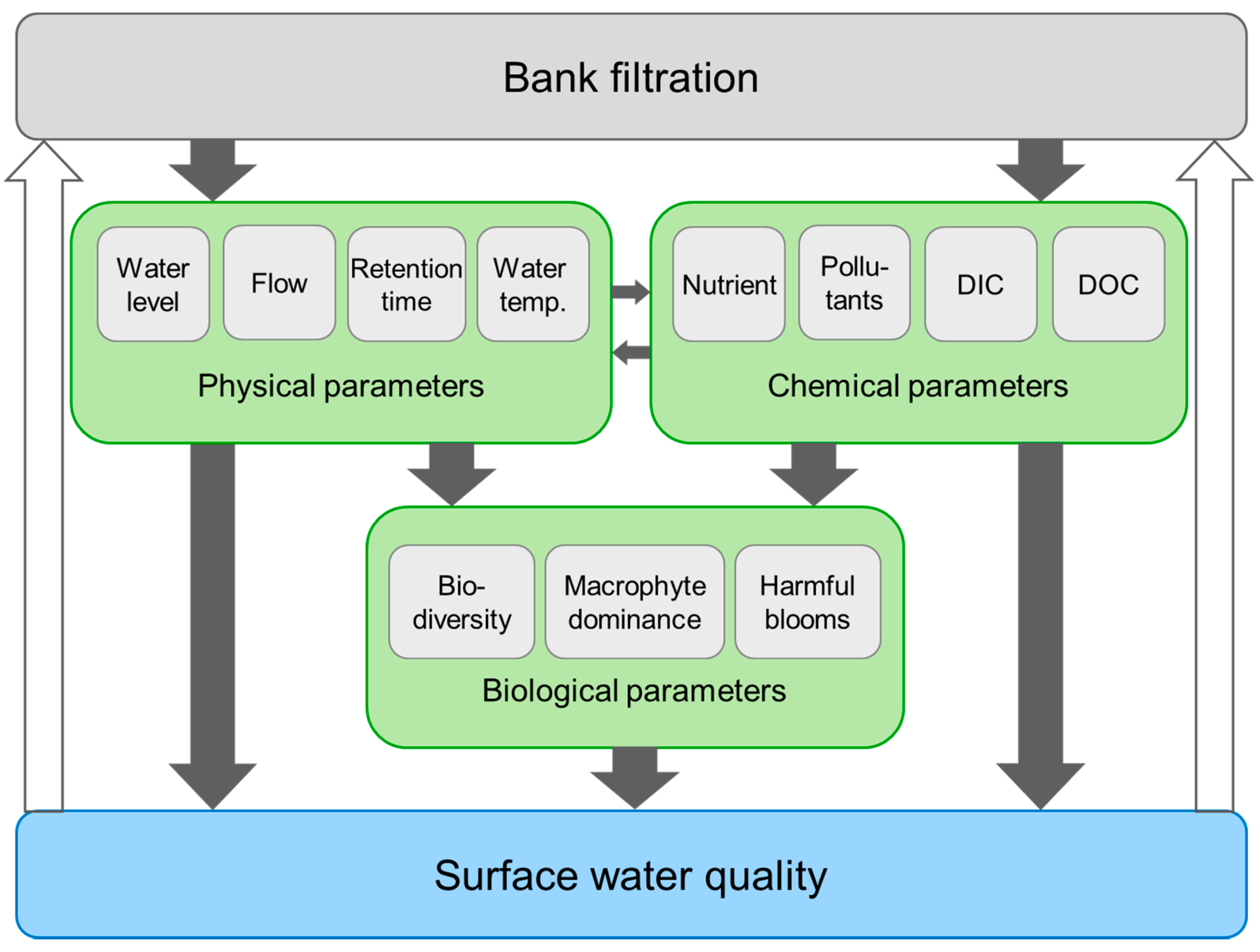
3. IBF Effects on Surface Water Quality
3.1. Discharge and Retention Time
3.2. Water Level Fluctuation
3.3. Sediment Characteristics
3.4. Water Temperature
3.5. Nutrient Availability
3.6. Pollutants
3.7. Dissolved Inorganic Carbon (DIC) Availability
3.8. Dissolved Organic Carbon (DOC)
4. Summary on IBF Effects on Surface Water Quality
4.1. Biodiversity
4.2. Macrophyte Abundance
4.3. Harmful Cyanobacteria Blooms
5. Conclusions
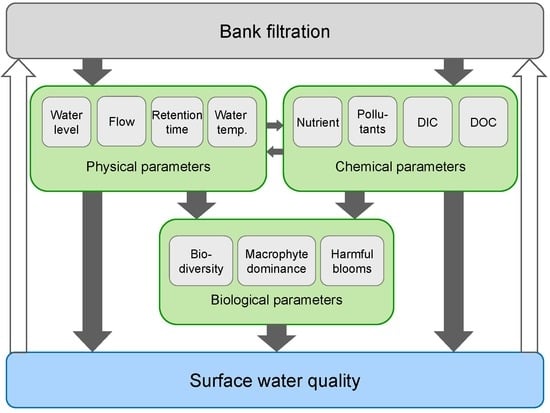
- We suggest that IBF directly and indirectly affects physical, chemical and biological processes in surface water that may have both negative and positive effects on their water quality (Figure 5). Potential adverse effects would in turn negatively affect the quality of the water abstracted for drinking water production via IBF (Figure 1 and Figure 5). We predict that IBF-induced changes in water temperature, CO2 availability and water retention times in lakes can lead to macrophyte disappearance, phytoplankton dominance and more suitable conditions for cyanobacteria blooms, among other consequences.
- Effects of IBF on surface water bodies are assumed to be highest in cases where discharge or lake volumes are small relative to the amount of water abstracted by IBF.
- Our conceptual impact assessment indicates the need for specific research on IBF effects on source aquatic ecosystems. While field and laboratory experiments may be suitable to test for selected processes, whole ecosystem experiments, monitoring, long-term data sets on aquatic ecosystems before and after the onset of IBF, and modelling are needed to understand the joint impact of IBF.
- Global change and urbanization are expected to increase the number of surface water bodies being used for IBF. Research on how to minimize potential negative impacts of IBF on their source surface water is thus urgently needed to ensure a sustainable use of this valuable technology.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Winter, T.C. Ground Water and Surface Water: A Single Resource; U.S. Geological Survey: Denver, CO, USA, 1998; ISBN 978-0-607-89339-7.
- Ray, C.; Melin, G.; Linsky, R.B. (Eds.) Riverbank Filtration: Improving Source-Water Quality; Water Science and Technology Library; Kluwer Acad. Publ: Dordrecht, The Netherlands, 2002; ISBN 978-1-4020-1133-7. [Google Scholar]
- Bouwer, H. Artificial recharge of groundwater: Hydrogeology and engineering. Hydrogeol. J. 2002, 10, 121–142. [Google Scholar] [CrossRef]
- Hiscock, K.M.; Grischek, T. Attenuation of groundwater pollution by bank filtration. J. Hydrol. 2002, 266, 139–144. [Google Scholar] [CrossRef]
- Chorus, I.; Klein, G.; Fastner, J.; Rotard, W. Off-flavors in surface waters—How efficient is bank filtration for their abatement in drinking-water. Water Sci. Technol. 1992, 25, 251–258. [Google Scholar] [CrossRef]
- Jacobson, R.A.; Warner, G.S.; Parasiewicz, P.; Bagtzoglou, A.C.; Ogden, F.L. An interdisciplinary study of the effects of groundwater extraction on freshwater fishes. Int. J. Ecol. Econ. Stat. 2008, 12, 7–25. [Google Scholar]
- Schubert, J. Hydraulic aspects of riverbank filtration—Field studies. J. Hydrol. 2002, 266, 145–161. [Google Scholar] [CrossRef]
- Umar, D.A.; Ramli, M.F.; Aris, A.Z.; Sulaiman, W.N.A.; Kura, N.U.; Tukur, A.I. An overview assessment of the effectiveness and global popularity of some methods used in measuring riverbank filtration. J. Hydrol. 2017, 550, 497–515. [Google Scholar] [CrossRef]
- Lahti, K.; Rapala, J.; Kivimaki, A.L.; Kukkonen, J.; Niemela, M.; Sivonen, K. Occurrence of microcystins in raw water sources and treated drinking water of Finnish waterworks. Water Sci. Technol. 2001, 43, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Pazouki, P.; Prévost, M.; McQuaid, N.; Barbeau, B.; de Boutray, M.-L.; Zamyadi, A.; Dorner, S. Breakthrough of cyanobacteria in bank filtration. Water Res. 2016, 102, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Zamyadi, A.; MacLeod, S.L.; Fan, Y.; McQuaid, N.; Dorner, S.; Sauve, S.; Prevost, M. Toxic cyanobacterial breakthrough and accumulation in a drinking water plant: A monitoring and treatment challenge. Water Res. 2012, 46, 1511–1523. [Google Scholar] [CrossRef] [PubMed]
- Massmann, G.; Nogeitzig, A.; Taute, T.; Pekdeger, A. Seasonal and spatial distribution of redox zones during lake bank filtration in Berlin, Germany. Environ. Geol. 2008, 54, 53–65. [Google Scholar] [CrossRef]
- Sprenger, C.; Hartog, N.; Hernández, M.; Vilanova, E.; Grützmacher, G.; Scheibler, F.; Hannappel, S. Inventory of managed aquifer recharge sites in Europe: Historical development, current situation and perspectives. Hydrogeol. J. 2017, 25, 1909–1922. [Google Scholar] [CrossRef]
- Tufenkji, N.; Ryan, J.N.; Elimelech, M. The promise of bank filtration. Environ. Sci. Technol. 2002, 36, 422A–428A. [Google Scholar] [CrossRef] [PubMed]
- Ray, C.; Schubert, J.; Linsky, R.B.; Melin, G. Introduction. In Riverbank Filtration: Improving Source-Water Quality; Ray, C., Melin, G., Linsky, R.B., Eds.; Water Science and Technology Library; Kluwer Acad. Publ: Dordrecht, The Netherlands, 2002; pp. 1–15. ISBN 978-1-4020-1133-7. [Google Scholar]
- Sharma, S.K.; Amy, G.L. Bank filtration: A sustainable water treatment technology for developing countries. In Water, Sanitation and Hygiene: Sustainable Development and Multisectoral Approaches; Water, Engineering and Development Centre, Loughborough University of Technology, WEDC: Addis Ababa, Ethopia, 2009. [Google Scholar]
- Wett, B.; Jarosch, H.; Ingerle, K. Flood induced infiltration affecting a bank filtrate well at the River Enns, Austria. J. Hydrol. 2002, 266, 222–234. [Google Scholar] [CrossRef]
- Schön, M. Systematic Comparison of Riverbank Filtration Sites in Austria and India; Leopold Franzens Universität Innsbruck: Innsbruck, Austria, 2014. [Google Scholar]
- ICPDR. Groundwater Guidance; International Commission for the Protection of the Danube River: Vienna, Austria, 2016. [Google Scholar]
- Miettinen, I.; Martikainen, P.; Vartiainen, T. Humus transformation at the bank filtration water-plant. Water Sci. Technol. 1994, 30, 179–187. [Google Scholar] [CrossRef]
- Miettinen, I.T.; Vartiainen, T.; Martikainen, P.J. Bacterial enzyme activities in ground water during bank filtration of lake water. Water Res. 1996, 30, 2495–2501. [Google Scholar] [CrossRef]
- Doussan, C.; Ledoux, E.; Detay, M. River-groundwater exchanges, bank filtration, and groundwater quality: Ammonium behavior. J. Environ. Qual. 1998, 27, 1418–1427. [Google Scholar] [CrossRef]
- FSO. Public Water Supply; Public Water Supply and Wastewater Treatment; Federal Statistical Office: Wiesbaden, Germany, 2013; p. 18. [Google Scholar]
- Schmidt, C.F.; Lange, F.T.; Brauch, H.-J. Assessing the impact of different redox conditions and residence times on the fate of organic micropollutants during riverbank filtration. In Proceedings of the 4th International Conference on Pharmaceuticals and Endocrine Disrupting Chemicals in Water, Minneapolis, MN, USA, 13–15 October 2004; National Ground Water Association: Minneapolis, MN, USA, 2004; pp. 195–205. [Google Scholar]
- Hoffmann, A.; Gunkel, G. Carbon input, production and turnover in the interstices of a Lake Tegel bank filtration site, Berlin, Germany. Limnologica 2011, 41, 151–159. [Google Scholar] [CrossRef]
- Hoffmann, A.; Gunkel, G. Bank filtration in the sandy littoral zone of Lake Tegel (Berlin): Structure and dynamics of the biological active filter zone and clogging processes. Limnologica 2011, 41, 10–19. [Google Scholar] [CrossRef]
- Heberer, T.; Massmann, G.; Fanck, B.; Taute, T.; Duennbier, U. Behaviour and redox sensitivity of antimicrobial residues during bank filtration. Chemosphere 2008, 73, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Massmann, G.; Dünnbier, U.; Heberer, T.; Taute, T. Behaviour and redox sensitivity of pharmaceutical residues during bank filtration—Investigation of residues of phenazone-type analgesics. Chemosphere 2008, 71, 1476–1485. [Google Scholar] [CrossRef] [PubMed]
- Maeng, S.K.; Ameda, E.; Sharma, S.K.; Grützmacher, G.; Amy, G.L. Organic micropollutant removal from wastewater effluent-impacted drinking water sources during bank filtration and artificial recharge. Water Res. 2010, 44, 4003–4014. [Google Scholar] [CrossRef] [PubMed]
- Wiese, B.; Massmann, G.; Jekel, M.; Heberer, T.; Duennbier, U.; Orlikowski, D.; Gruetzmacher, G. Removal kinetics of organic compounds and sum parameters under field conditions for managed aquifer recharge. Water Res. 2011, 45, 4939–4950. [Google Scholar] [CrossRef] [PubMed]
- Henzler, A.F.; Greskowiak, J.; Massmann, G. Modeling the fate of organic micropollutants during river bank filtration (Berlin, Germany). J. Contam. Hydrol. 2014, 156, 78–92. [Google Scholar] [CrossRef] [PubMed]
- Henzler, A.F.; Greskowiak, J.; Massmann, G. Seasonality of temperatures and redox zonations during bank filtration—A modeling approach. J. Hydrol. 2016, 535, 282–292. [Google Scholar] [CrossRef]
- Burke, V.; Greskowiak, J.; Asmuss, T.; Bremermann, R.; Taute, T.; Massmann, G. Temperature dependent redox zonation and attenuation of wastewater-derived organic micropollutants in the hyporheic zone. Sci. Total Environ. 2014, 482, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Grützmacher, G.; Böttcher, G.; Chorus, I.; Knappe, A.; Pekdeger, A. Cyanobacterial toxins in bank filtered water from Lake Wannsee, Berlin. In Management of Aquifer Recharge for Sustainability; Dillon, P., Ed.; Swets and Zeitlinger: Lisse, The Netherlands, 2002; pp. 175–179. [Google Scholar]
- Heberer, T.; Mechlinski, A.; Fanck, B.; Knappe, A.; Massmann, G.; Pekdeger, A.; Fritz, B. Field Studies on the Fate and Transport of Pharmaceutical Residues in Bank Filtration. Groundw. Monit. Remediat. 2004, 24, 70–77. [Google Scholar] [CrossRef]
- Massmann, G.; Sueltenfuss, J.; Duennbier, U.; Knappe, A.; Taute, T.; Pekdeger, A. Investigation of groundwater a residence times during bank filtration in Berlin: Multi-tracer approach. Hydrol. Process. 2008, 22, 788–801. [Google Scholar] [CrossRef]
- Kohfahl, C.; Massmann, G.; Pekdeger, A. Sources of oxygen flux in groundwater during induced bank filtration at a site in Berlin, Germany. Hydrogeol. J. 2009, 17, 571–578. [Google Scholar] [CrossRef]
- Chorus, I. (Ed.) Cyanotoxins: Occurrence, Causes, Consequences; Springer: Berlin, Germany; New York, NY, USA, 2012; ISBN 978-3-642-59514-1. [Google Scholar]
- Achten, C.; Kolb, A.; Puttmann, W. Occurrence of methyl tert-butyl ether (MTBE) in riverbank filtered water and drinking water produced by riverbank filtration 2. Environ. Sci. Technol. 2002, 36, 3662–3670. [Google Scholar] [CrossRef] [PubMed]
- Ray, C.; Soong, T.W.; Lian, Y.Q.; Roadcap, G.S. Effect of flood-induced chemical load on filtrate quality at bank filtration sites. J. Hydrol. 2002, 266, 235–258. [Google Scholar] [CrossRef]
- Homonnay, Z. Use of Bank Filtration in Hungary. In Riverbank Filtration: Understanding Contaminant Biogeochemistry and Pathogen Removal; Ray, C., Ed.; Springer: Dordrecht, The Netherlands, 2002; pp. 221–228. ISBN 978-1-4020-0955-6. [Google Scholar]
- Rossetto, R.; Barbagli, A.; Borsi, I.; Mazzanti, G.; Vienken, T.; Bonari, E. Site investigation and design of the monitoring system at the Sant’Alessio Induced RiverBank Filtration plant (Lucca, Italy). Rendiconti online della Società Geologica Italiana 2015, 35, 248–251. [Google Scholar] [CrossRef]
- Eynard, F.; Mez, K.; Walther, J.-L. Risk of cyanobacterial toxins in Riga waters (Latvia). Water Res. 2000, 34, 2979–2988. [Google Scholar] [CrossRef]
- Medema, G.J.; Stuyfzand, P.J. Removal of micro-organisms upon basin recharge, deep well injection and river bank filtration in the Netherlands. In Management of Aquifer Recharge for Sustainability; Dillon, P., Ed.; Swets and Zeitlinger: Lisse, The Netherlands, 2002; pp. 125–131. ISBN 90 5809 527 4. [Google Scholar]
- Hamann, E.; Stuyfzand, P.J.; Greskowiak, J.; Timmer, H.; Massmann, G. The fate of organic micropollutants during long-term/long-distance river bank filtration. Sci. Total Environ. 2016, 545, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Mollema, P.N.; Stuyfzand, P.J.; Juhasz-Holterman, M.H.A.; Van Diepenbeek, P.M.J.A.; Antonellini, M. Metal accumulation in an artificially recharged gravel pit lake used for drinking water supply. J. Geochem. Explor. 2015, 150, 35–51. [Google Scholar] [CrossRef]
- Mollema, P.N.; Antonellini, M.; Hubeek, A.; Van Diepenbeek, P.M.J.A. The effect of artificial recharge on hydrochemistry: A comparison of two fluvial gravel pit lakes with different post-excavation uses in the netherlands. Water 2016, 8, 409. [Google Scholar] [CrossRef]
- Kvitsand, H.M.L.; Myrmel, M.; Fiksdal, L.; Osterhus, S.W. Evaluation of bank filtration as a pretreatment method for the provision of hygienically safe drinking water in Norway: Results from monitoring at two full-scale sites. Hydrogeol. J. 2017, 25, 1257–1269. [Google Scholar] [CrossRef]
- Przybylek, J.; Dragon, K.; Kaczmarek, P.M.J. Hydrogeological investigations of river bed clogging at a river bank filtration site along the River Warta, Poland. Geologos 2017, 23, 201–214. [Google Scholar] [CrossRef]
- Rojanschi, V.; Mlenajek, L.; Stanciulesc, M. Riverbank filtration in water supply in Romania—Old solutions, new problems. In Riverbank Filtration: Understanding Contaminant Biogeochemistry and Pathogen Removal; Ray, C., Ed.; Springer Science & Business Media: Berlin, Germany, 2002; pp. 235–245. ISBN 978-1-4020-0954-9. [Google Scholar]
- Vogt, T.; Hoehn, E.; Schneider, P.; Cirpka, O.A. Untersuchung der Flusswasserinfiltration in voralpinen Schottern mittels Zeitreihenanalyse. Grundwasser 2009, 14, 179–194. [Google Scholar] [CrossRef]
- Castella, E.; Bickerton, M.; Armitage, P.D.; Petts, G.E. The effects of water abstractions on invertebrate communities in U.K. streams. Hydrobiologia 1995, 308, 167–182. [Google Scholar] [CrossRef]
- Ahmed, A.K.A.; Marhaba, T.F. Review on river bank filtration as an in situ water treatment process. Clean Technol. Environ. Policy 2017, 19, 349–359. [Google Scholar] [CrossRef]
- Weiss, W.J.; Bouwer, E.J.; Aboytes, R.; LeChevallier, M.W.; O’Melia, C.R.; Le, B.T.; Schwab, K.J. Riverbank filtration for control of microorganisms: Results from field monitoring. Water Res. 2005, 39, 1990–2001. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, G.B.; Ray, C.; Wang, J.Z.; Hubbs, S.A.; Song, R.; Jasperse, J.; Seymour, D. Use of artificial neural networks to evaluate the effectiveness of riverbank filtration. Water Res. 2005, 39, 2505–2516. [Google Scholar] [CrossRef] [PubMed]
- Ray, C. (Ed.) Riverbank Filtration: Understanding Contaminant Biogeochemistry and Pathogen Removal; Springer Science & Business Media: Berlin, Germany, 2002; ISBN 978-1-4020-0954-9. [Google Scholar]
- Harvey, R.W.; Metge, D.W.; LeBlanc, D.R.; Underwood, J.; Aiken, G.R.; Butler, K.; McCobb, T.D.; Jasperse, J. Importance of the Colmation Layer in the Transport and Removal of Cyanobacteria, Viruses, and Dissolved Organic Carbon during Natural Lake-Bank Filtration. J. Environ. Qual. 2015, 44, 1413–1423. [Google Scholar] [CrossRef] [PubMed]
- Freitas, D.A.; Cabral, J.J.S.P.; Rocha, F.J.S.; Paiva, A.L.R.; Sens, M.L.; Veras, T.B. Cryptosporidium spp. and Giardia spp. removal by bank filtration at Beberibe River, Brazil. River Res. Appl. 2017, 33, 1079–1087. [Google Scholar] [CrossRef]
- Romero, L.G.; Mondardo, R.I.; Sens, M.L.; Grischek, T. Removal of cyanobacteria and cyanotoxins during lake bank filtration at Lagoa do Peri, Brazil. Clean Technol. Environ. Policy 2014, 16, 1133–1143. [Google Scholar] [CrossRef]
- Romero-Esquivel, L.G.; Pizzolatti, B.S.; Sens, M.L. Potential application of bank filtration in Santa Catarina, Brazil. Interciencia 2016, 41, 740–747. [Google Scholar]
- Hu, B.; Teng, Y.; Zhai, Y.; Zuo, R.; Li, J.; Chen, H. Riverbank filtration in China: A review and perspective. J. Hydrol. 2016, 541, 914–927. [Google Scholar] [CrossRef]
- Sandhu, C.; Grischek, T.; Kumar, P.; Ray, C. Potential for riverbank filtration in India. Clean Technol. Environ. Policy 2011, 13, 295–316. [Google Scholar] [CrossRef]
- Lorenzen, G.; Sprenger, C.; Taute, T.; Pekdeger, A.; Mittal, A.; Massmann, G. Assessment of the potential for bank filtration in a water-stressed megacity (Delhi, India). Environ. Earth Sci. 2010, 61, 1419–1434. [Google Scholar] [CrossRef]
- Sharma, B.; Uniyal, D.P.; Dobhal, R.; Kimothi, P.C.; Grischek, T. A sustainable solution for safe drinking water through bank filtration technology in Uttarakhand, India. Curr. Sci. 2014, 107, 1118–1124. [Google Scholar]
- Thakur, A.K.; Ojha, C.S.P.; Singh, V.P.; Gurjar, B.R.; Sandhu, C. Removal of pathogens by river bank filtration at Haridwar, India. Hydrol. Process. 2013, 27, 1535–1542. [Google Scholar] [CrossRef]
- Dash, R.R.; Mehrotra, I.; Kumar, P.; Grischek, T. Lake bank filtration at Nainital, India: Water-quality evaluation. Hydrogeol. J. 2008, 16, 1089–1099. [Google Scholar] [CrossRef]
- Othman, S.Z.; Adlan, M.N.; Selamat, M.R. A study on the potential of riverbank filtration for the removal of color, iron, turbidy and E.coli in Sungai Perak, Kota Lama Kiri, Kuala Kangsar, Perak, Malaysia. Jurnal Teknologi 2015, 74, 83–91. [Google Scholar] [CrossRef]
- Bork, J.; Berkhoff, S.E.; Bork, S.; Hahn, H.J. Using subsurface metazoan fauna to indicate groundwater-surface water interactions in the Nakdong River floodplain, South Korea. Hydrogeol. J. 2009, 17, 61–75. [Google Scholar] [CrossRef]
- Pholkern, K.; Srisuk, K.; Grischek, T.; Soares, M.; Schäfer, S.; Archwichai, L.; Saraphirom, P.; Pavelic, P.; Wirojanagud, W. Riverbed clogging experiments at potential river bank filtration sites along the Ping River, Chiang Mai, Thailand. Environ. Earth Sci. 2015, 73, 7699–7709. [Google Scholar] [CrossRef]
- Ghodeif, K.; Grischek, T.; Bartak, R.; Wahaab, R.; Herlitzius, J. Potential of river bank filtration (RBF) in Egypt. Environ. Earth Sci. 2016, 75, 671. [Google Scholar] [CrossRef]
- Shamrukh, M.; Abdel-Wahab, A. Riverbank filtration for sustainable water supply: Application to a large-scale facility on the Nile River. Clean Technol. Environ. Policy 2008, 10, 351–358. [Google Scholar] [CrossRef]
- Hamdan, A.M.; Sensoy, M.M.; Mansour, M.S. Evaluating the effectiveness of bank infiltration process in new Aswan City, Egypt. Arab. J. Geosci. 2013, 6, 4155–4165. [Google Scholar] [CrossRef]
- Stadtwerke Düsseldorf. Clearly Drinking Water—Element of Life; Stadtwerke Düsseldorf: Düsseldorf, Germany, 2011. [Google Scholar]
- Zippel, M.; Hannappel, S. Evaluation of the groundwater yield of Berlin water works using regional numerical groundwater flow models. Grundwasser 2008, 13, 195–207. [Google Scholar] [CrossRef]
- Berlin Water Utilities. Groundwater Extraction. Available online: http://www.bwb.de/content/language1/html/961.php (accessed on 1 June 2018).
- Berlin Water Utilities. Wasserschutzgebiete auf Einen Blick; Berlin Water Utilities: Berlin, Germany, 2018. [Google Scholar]
- Jekel, M.; Ruhl, A.S.; Meinel, F.; Zietzschmann, F.; Lima, S.P.; Baur, N.; Wenzel, M.; Gnirß, R.; Sperlich, A.; Dünnbier, U.; et al. Anthropogenic organic micro-pollutants and pathogens in the urban water cycle: Assessment, barriers and risk communication (ASKURIS). Environ. Sci. Eur. 2013, 25, 20. [Google Scholar] [CrossRef]
- Driescher, E.; Behrendt, H.; Schellenberger, G.; Stellmacher, R. Lake Müggelsee and its environment—Natural conditions and anthropogenic impacts. Int. Revue Gesamten Hydrobiol. Hydrogr. 1993, 78, 327–343. [Google Scholar] [CrossRef]
- Zippel, M. Model-Based Assessment of the Underground Water Resources of Berlin—The Groundwater Situation in the Catchment Area of Lake Müggelsee; Freie Universität: Berlin, Germany, 2006. [Google Scholar]
- Berlin Water Utilities. Pumping Rates from Well Galleries around Lake Müggelsee; Berlin Water Utilities: Berlin, Germany, 2018. [Google Scholar]
- Schröter, W. Groundwater Levels of the Main Aquifer and Panke Valley Aquifer. 2015. Available online: http://www.stadtentwicklung.berlin.de/umwelt/umweltatlas/kq212.htm (accessed on 15 May 2018).
- Frey, W.; Skripalle, J.; Krüger, P. Ecological Consequences of Large-Scale Groundwater Drawdown; Senate of Berlin, Department for the Environment, Transport and Climate Protection: Berlin, Germany, 1992. [Google Scholar]
- Senate of Berlin. Measured and Modelled Water Discharge into Lake Müggelsee; Berlin Senate Department for the Environment, Transport and Climate Protection: Berlin, Germany, 2018. [Google Scholar]
- Schumacher, F.; Storz, P. State of Model 2016 and Transient Calculation of the Hydrological Years 2004 to 2014; Senate of Berlin, Department for the Environment, Transport and Climate Protection: Berlin, Germany, 2016; 12p. [Google Scholar]
- Vollenweider, R.A. Advances in defining critical loading levels for phosphorus in lake eutrophication. Mem. Ist. Ital. Idrobiol. 1976, 33, 53–83. [Google Scholar]
- Shatwell, T.; Köhler, J. Decreased nitrogen loading controls summer cyanobacterial blooms without promoting nitrogen-fixing taxa: Long-term response of a shallow lake. Limnol. Oceanogr. 2018. [Google Scholar] [CrossRef]
- Bunn, S.E.; Arthington, A.H. Basic principles and ecological consequences of altered flow regimes for aquatic biodiversity. Environ. Manag. 2002, 30, 492–507. [Google Scholar] [CrossRef]
- Bickerton, M.; Petts, G.; Armitage, P.; Castella, E. Assessing the ecological effects of groundwater abstraction on chalk streams: Three examples from Eastern England. Regul. Rivers Res. Mgmt. 1993, 8, 121–134. [Google Scholar] [CrossRef]
- Acreman, M.C.; Adams, B.; Birchall, P.; Connorton, B. Does groundwater abstraction cause degradation of rivers and wetlands? Water Environ. J. 2000, 14, 200–206. [Google Scholar] [CrossRef]
- Hilt, S.; Schoenfelder, I.; Rudnickaa, A.; Carls, R.; Nikolaevich, N.; Sukhodolov, A.; Engelhardt, C. Reconstruction of pristine morphology, flow, nutrient conditions and submerged vegetation of lowland river spree (Germany) from palaeomeanders. River Res. Appl. 2008, 24, 310–329. [Google Scholar] [CrossRef]
- Armitage, P.; Petts, G. Biotic score and prediction to assess the effects of water abstractions. Aquat. Conserv. Mar. Freshw. Ecosyst. 1992, 2, 1–17. [Google Scholar] [CrossRef]
- Hilt, S.; Koehler, J.; Kozerski, H.-P.; van Nes, E.H.; Scheffer, M. Abrupt regime shifts in space and time along rivers and connected lake systems. Oikos 2011, 120, 766–775. [Google Scholar] [CrossRef]
- Liu, C.; Liu, J.; Wang, X.-S.; Zheng, C. Analysis of groundwater–lake interaction by distributed temperature sensing in Badain Jaran Desert, Northwest China. Hydrol. Process. 2016, 30, 1330–1341. [Google Scholar] [CrossRef]
- Adrian, R.; Hessen, D.O.; Blenckner, T.; Hillebrand, H.; Hilt, S.; Jeppesen, E.; Livingstone, D.M.; Trolle, D. Environmental impacts—Lake ecosystems. In North Sea Region Climate Change Assessment; Quante, M., Colijn, F., Eds.; Springer International Publ.: Cham, Switzerland, 2016; pp. 315–340. ISBN 978-3-319-39743-6. [Google Scholar]
- Mooij, W.M.; Janse, J.H.; Domis, L.N.D.S.; Huelsmann, S.; Ibelings, B.W. Predicting the effect of climate change on temperate shallow lakes with the ecosystem model PCLake. Hydrobiologia 2007, 584, 443–454. [Google Scholar] [CrossRef]
- Cieśliński, R.; Piekarz, J.; Zieliński, M. Groundwater supply of lakes: The case of Lake Raduńskie Górne (northern Poland, Kashubian Lake District). Hydrol. Sci. J. 2016, 61, 2427–2434. [Google Scholar] [CrossRef]
- Hellsten, S. Environmental Factors and Aquatic Macrophytes in the Littoral Zone of Regulated Lakes; University of Oulo: Oulo, Finland, 2000. [Google Scholar]
- Hilt, S.; Brothers, S.; Jeppesen, E.; Veraart, A.J.; Kosten, S. Translating Regime Shifts in Shallow Lakes into Changes in Ecosystem Functions and Services. BioScience 2017, 67, 928–936. [Google Scholar] [CrossRef]
- Bakker, E.S.; Hilt, S. Impact of water-level fluctuations on cyanobacterial blooms: Options for management. Aquat. Ecol. 2016, 50, 485–498. [Google Scholar] [CrossRef]
- Rorslett, B.; Johansen, S.W. Remedial measures connected with aquatic macrophytes in Norwegian regulated rivers and reservoirs. Regul. Rivers Res. Manag. 1996, 12, 509–522. [Google Scholar] [CrossRef]
- Mitrovic, S.M.; Hardwick, L.; Dorani, F. Use of flow management to mitigate cyanobacterial blooms in the Lower Darling River, Australia. J. Plankton Res. 2011, 33, 229–241. [Google Scholar] [CrossRef]
- Barko, J.W.; Gunnison, D.; Carpenter, S.R. Sediment interactions with submersed macrophyte growth and community dynamics. Aquat. Bot. 1991, 41, 41–65. [Google Scholar] [CrossRef]
- Vandel, E.; Vaasma, T.; Terasmaa, J.; Koff, T.; Vainu, M. Effect of human induced drastic water-level changes to ecologically sensitive small lakes. In Proceedings of 2nd International Conference—Water Resources and Wetlands; Romanian Limnogeographical Association: Târgoviște, Romania, 2014; pp. 204–211. [Google Scholar]
- Leyer, I. Predicting plant species’ responses to river regulation: The role of water level fluctuations. J. Appl. Ecol. 2005, 42, 239–250. [Google Scholar] [CrossRef]
- Rorslett, B. Environmental-factors and aquatic macrophyte response in regulated lakes—A statistical approach. Aquat. Bot. 1984, 19, 199–220. [Google Scholar] [CrossRef]
- Stets, E.G.; Striegl, R.G.; Aiken, G.R.; Rosenberry, D.O.; Winter, T.C. Hydrologic support of carbon dioxide flux revealed by whole-lake carbon budgets. J. Geophys. Res. 2009, 114, G01008. [Google Scholar] [CrossRef]
- Périllon, C.; Hilt, S. Groundwater influence differentially affects periphyton and macrophyte production in lakes. Hydrobiologia 2015, 1–13. [Google Scholar] [CrossRef]
- O’Neil, J.M.; Davis, T.W.; Burford, M.A.; Gobler, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Jeppesen, E.; Meerhoff, M.; Davidson, T.A.; Trolle, D.; Sondergaard, M.; Lauridsen, T.L.; Beklioglu, M.; Brucet, S.; Volta, P.; Gonzalez-Bergonzoni, I.; et al. Climate change impacts on lakes: An integrated ecological perspective based on a multi-faceted approach, with special focus on shallow lakes. J. Limnol. 2014, 73, 88–111. [Google Scholar] [CrossRef]
- Phillips, G.L.; Eminson, D.; Moss, B. A mechanism to account for macrophyte decline in progressively eutrophicated freshwaters. Aquat. Bot. 1978, 4, 103–126. [Google Scholar] [CrossRef]
- Williamson, C.E.; Overholt, E.P.; Pilla, R.M.; Leach, T.H.; Brentrup, J.A.; Knoll, L.B.; Mette, E.M.; Moeller, R.E. Ecological consequences of long-term browning in lakes. Sci. Rep. 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Solomon, C.T.; Jones, S.E.; Weidel, B.C.; Buffam, I.; Fork, M.L.; Karlsson, J.; Larsen, S.; Lennon, J.T.; Read, J.S.; Sadro, S.; et al. Ecosystem Consequences of Changing Inputs of Terrestrial Dissolved Organic Matter to Lakes: Current Knowledge and Future Challenges. Ecosystems 2015, 18, 376–389. [Google Scholar] [CrossRef]
- Vasconcelos, F.R.; Diehl, S.; Rodriguez, P.; Hedstrom, P.; Karlsson, J.; Bystrom, P. Asymmetrical competition between aquatic primary producers in a warmer and browner world. Ecology 2016, 97, 2580–2592. [Google Scholar] [CrossRef] [PubMed]
- Brothers, S.; Koehler, J.; Attermeyer, K.; Grossart, H.P.; Mehner, T.; Meyer, N.; Scharnweber, K.; Hilt, S. A feedback loop links brownification and anoxia in a temperate, shallow lake. Limnol. Oceanogr. 2014, 59, 1388–1398. [Google Scholar] [CrossRef]
- Relyea, R.A. The impact of insecticides and herbicides on the biodiversity and productivity of aquatic communities. Ecol. Appl. 2005, 15, 618–627. [Google Scholar] [CrossRef]
- Coops, H.; Beklioglu, M.; Crisman, T.L. The role of water-level fluctuations in shallow lake ecosystems—Workshop conclusions. Hydrobiologia 2003, 506–509, 23–27. [Google Scholar] [CrossRef]
- Leira, M.; Cantonati, M. Effects of water-level fluctuations on lakes: An annotated bibliography. Hydrobiologia 2008, 613, 171–184. [Google Scholar] [CrossRef]
- Scheffer, M. Ecology of Shallow Lakes; Springer: Dordrecht, The Netherlands, 2004; ISBN 978-1-4020-2306-4. [Google Scholar]
- Sachse, R.; Petzoldt, T.; Blumstock, M.; Moreira, S.; Paetzig, M.; Ruecker, J.; Janse, J.H.; Mooij, W.M.; Hilt, S. Extending one-dimensional models for deep lakes to simulate the impact of submerged macrophytes on water quality. Environ. Model. Softw. 2014, 61, 410–423. [Google Scholar] [CrossRef]
- Wiese, B.; Nützmann, G. Transient leakance and infiltration characteristics during lake bank filtration. Groundwater 2009, 47, 57–68. [Google Scholar] [CrossRef] [PubMed]
- WASY. Hydrogeological Report on the Impact of Fundamental Changes in the Operation of Groundwater Enrichment at Tegel Waterworks; WASY Institute for Water Resources Planning and Systems Research Ltd.: Berlin, Germany, 2004. [Google Scholar]
- Gunkel, G.; Beulker, C.; Hoffmann, A.; Kosmol, J. Fine particulate organic matter (FPOM) transport and processing in littoral interstices—Use of fluorescent markers. Limnologica 2009, 39, 185–199. [Google Scholar] [CrossRef]
- Van Zuidam, B.G.; Cazemier, M.M.; van Geest, G.J.; Roijackers, R.M.M.; Peeters, E.T.H.M. Relationship between redox potential and the emergence of three submerged macrophytes. Aquat. Bot. 2014, 113, 56–62. [Google Scholar] [CrossRef]
- Ding, S.; Wang, Y.; Wang, D.; Li, Y.Y.; Gong, M.; Zhang, C. In situ, high-resolution evidence for iron-coupled mobilization of phosphorus in sediments. Sci. Rep. 2016, 6, 24341. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, S.M.; Rinklebe, J.; Rupp, H.; Meissner, R. Temporal dynamics of pore water concentrations of Cd, Co, Cu, Ni, and Zn and their controlling factors in a contaminated floodplain soil assessed by undisturbed groundwater lysimeters. Environ. Pollut. 2014, 191, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Uhlmann, W.; Zimmermann, K. Case Analysis of the Sulphate Load in the River Spree 2014/15; Senate of Berlin, Department for the Environment, Transport and Climate Protection: Dresden, Germany, 2015; 96p. [Google Scholar]
- Koch, M.S.; Mendelssohn, I.A.; McKee, K.L. Mechanism for the hydrogen sulfide-induced growth limitation in wetland macrophytes. Limnol. Oceanogr. 1990, 35, 399–408. [Google Scholar] [CrossRef]
- Bayarsaikhan, U.; Filter, J.; Gernert, U.; Jekel, M.; Ruhl, A.S. Fate of leaf litter deposits and impacts on oxygen availability in bank filtration column studies. Environ. Res. 2018, 164, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Hannah, D.M.; Webb, B.W.; Nobilis, F. River and stream temperature: Dynamics, processes, models and implications—Preface. Hydrol. Process. 2008, 22, 899–901. [Google Scholar] [CrossRef]
- Boisneau, C.; Moatar, F.; Bodin, M.; Boisneau, P. Does global warming impact on migration patterns and recruitment of Allis shad (Alosa alosa L.) young of the year in the Loire River, France? Hydrobiologia 2008, 602, 179–186. [Google Scholar] [CrossRef]
- Yvon-Durocher, G.; Montoya, J.M.; Trimmer, M.; Woodward, G. Warming alters the size spectrum and shifts the distribution of biomass in freshwater ecosystems. Glob. Chang. Biol. 2011, 17, 1681–1694. [Google Scholar] [CrossRef]
- Hussner, A.; van Dam, H.; Vermaat, J.E.; Hitt, S. Comparison of native and neophytic aquatic macrophyte developments in a geothermally warmed river and thermally normal channels. Fundam. Appl. Limnol. 2014, 185, 155–165. [Google Scholar] [CrossRef]
- Boscarino, B.T.; Rudstam, L.G.; Mata, S.; Gal, G.; Johannsson, O.E.; Mills, E.L. The effects of temperature and predator—Prey interactions on the migration behavior and vertical distribution of Mysis relicta. Limnol. Oceanogr. 2007, 52, 1599–1613. [Google Scholar] [CrossRef]
- Wehrly, K.E.; Wang, L.; Mitro, M. Field-based estimates of thermal tolerance limits for trout: Incorporating exposure time and temperature fluctuation. Trans. Am. Fish. Soc. 2007, 136, 365–374. [Google Scholar] [CrossRef]
- Power, G.; Brown, R.S.; Imhof, J.G. Groundwater and fish—Insights from northern North America. Hydrol. Process. 1999, 13, 401–422. [Google Scholar] [CrossRef]
- Imholt, C.; Gibbins, C.N.; Malcolm, I.A.; Langan, S.; Soulsby, C. Influence of riparian cover on stream temperatures and the growth of the mayfly Baetis rhodani in an upland stream. Aquat. Ecol. 2010, 44, 669–678. [Google Scholar] [CrossRef]
- Jensen, A.J. Atlantic salmon (Salmo salar) in the regulated River Alta: Effects of altered water temperature on parr growth. River Res. Appl. 2003, 19, 733–747. [Google Scholar] [CrossRef]
- Whitledge, G.W.; Rabeni, C.F.; Annis, G.; Sowa, S.P. Riparian Shading and Groundwater Enhance Growth Potential for Smallmouth Bass in Ozark Streams. Ecol. Appl. 2006, 16, 1461–1473. [Google Scholar] [CrossRef]
- Gerten, D.; Adrian, R. Species-specific changes in the phenology and peak abundance of freshwater copepods in response to warm summers. Freshw. Biol. 2002, 47, 2163–2173. [Google Scholar] [CrossRef]
- Harper, M.P.; Peckarsky, B.L. Emergence cues of a mayfly in a high-altitude stream ecosystem: Potential response to climate change. Ecol. Appl. 2006, 16, 612–621. [Google Scholar] [CrossRef]
- Alvarez, D.; Nicieza, A.G. Compensatory response “defends” energy levels but not growth trajectories in brown trout, Salmo trutta L. Proc. R. Soc. B Biol. Sci. 2005, 272, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Yvon-Durocher, G.; Jones, J.I.; Trimmer, M.; Woodward, G.; Montoya, J.M. Warming alters the metabolic balance of ecosystems. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 2117–2126. [Google Scholar] [CrossRef] [PubMed]
- McKee, D.; Atkinson, D.; Collings, S.; Eaton, J.; Harvey, I.; Heyes, T.; Hatton, K.; Wilson, D.; Moss, B. Macro-zooplankter responses to simulated climate warming in experimental freshwater microcosms. Freshw. Biol. 2002, 47, 1557–1570. [Google Scholar] [CrossRef]
- Mahdy, A.; Hilt, S.; Filiz, N.; Beklioğlu, M.; Hejzlar, J.; Özkundakci, D.; Papastergiadou, E.; Scharfenberger, U.; Šorf, M.; Stefanidis, K.; et al. Effects of water temperature on summer periphyton biomass in shallow lakes: A pan-European mesocosm experiment. Aquat. Sci. 2015, 77, 499–510. [Google Scholar] [CrossRef]
- Velthuis, M.; Domis, L.N.; Frenken, T.; Stephan, S.; Kazanjian, G.; Aben, R.; Hilt, S.; Kosten, S.; van Donk, E.; Van de Waal, D.B. Warming advances top-down control and reduces producer biomass in a freshwater plankton community. Ecosphere 2017, 8, e01651. [Google Scholar] [CrossRef]
- Kazanjian, G.; Velthuis, M.; Aben, R.; Stephan, S.; Peeters, E.T.H.M.; Frenken, T.; Touwen, J.; Xue, F.; Kosten, S.; Van de Waal, D.B.; et al. Impacts of warming on top-down and bottom-up controls of periphyton production. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W.; Huisman, J. Climate change: A catalyst for global expansion of harmful cyanobacterial blooms. Environ. Microbiol. Rep. 2009, 1, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Huber, V.; Wagner, C.; Gerten, D.; Adrian, R. To bloom or not to bloom: Contrasting responses of cyanobacteria to recent heat waves explained by critical thresholds of abiotic drivers. Oecologia 2012, 169, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Kosten, S.; Huszar, V.L.M.; Bécares, E.; Costa, L.S.; van Donk, E.; Hansson, L.-A.; Jeppesen, E.; Kruk, C.; Lacerot, G.; Mazzeo, N.; et al. Warmer climates boost cyanobacterial dominance in shallow lakes. Glob. Chang. Biol. 2012, 18, 118–126. [Google Scholar] [CrossRef]
- Jensen, H.S.; Andersen, F.O. Importance of temperature, nitrate, and pH for phosphate release from aerobic sediments of four shallow, eutrophic lakes. Limnol. Oceanogr. 1992, 37, 577–589. [Google Scholar] [CrossRef]
- Bannick, C.; Engelmann, B.; Fendler, J.; Frauenstein, J.; Ginzky, H.; Hornemann, C.; Ilvonen, O.; Kirschbaum, B.; Penn-Bressel, G.; Rechenberg, J.; et al. Groundwater in Germany; Federal Ministry for the Environment, Nature Conservation, Building and Nuclear Safety: Bonn, Germany, 2008; p. 72. [Google Scholar]
- Schuetz, T.; Weiler, M. Quantification of localized groundwater inflow into streams using ground-based infrared thermography. Geophys. Res. Lett. 2011, 38, L03401. [Google Scholar] [CrossRef]
- Sebok, E.; Duque, C.; Kazmierczak, J.; Engesgaard, P.; Nilsson, B.; Karan, S.; Frandsen, M. High-resolution distributed temperature sensing to detect seasonal groundwater discharge into Lake Vaeng, Denmark. Water Resour. Res. 2013, 49, 5355–5368. [Google Scholar] [CrossRef]
- Whitehead, P.G.; Wilby, R.L.; Battarbee, R.W.; Kernan, M.; Wade, A.J. A review of the potential impacts of climate change on surface water quality. Hydrol. Sci. J. 2009, 54, 101–123. [Google Scholar] [CrossRef]
- Vanek, V. Riparian Zone as a Source of Phosphorus for a Groundwater-Dominated Lake. Water Res. 1991, 25, 409–418. [Google Scholar] [CrossRef]
- Meinikmann, K.; Hupfer, M.; Lewandowski, J. Phosphorus in groundwater discharge—A potential source for lake eutrophication. J. Hydrol. 2015, 524, 214–226. [Google Scholar] [CrossRef]
- Ouyang, Y. Estimation of shallow groundwater discharge and nutrient load into a river. Ecol. Eng. 2012, 38, 101–104. [Google Scholar] [CrossRef]
- Perillon, C.; Poeschke, F.; Lewandowski, J.; Hupfer, M.; Hilt, S. Stimulation of epiphyton growth by lacustrine groundwater discharge to an oligo-mesotrophic hard-water lake. Freshw. Sci. 2017, 36, 555–570. [Google Scholar] [CrossRef]
- Phillips, G.; Willby, N.; Moss, B. Submerged macrophyte decline in shallow lakes: What have we learnt in the last forty years? Aquat. Bot. 2016, 135, 37–45. [Google Scholar] [CrossRef]
- Scheffer, M.; Hosper, S.; Meijer, M.; Moss, B.; Jeppesen, E. Alternative Equilibria in Shallow Lakes. Trends Ecol. Evol. 1993, 8, 275–279. [Google Scholar] [CrossRef]
- Hilt, S.; Koehler, J.; Adrian, R.; Monaghan, M.T.; Sayer, C.D. Clear, crashing, turbid and back—Long-term changes in macrophyte assemblages in a shallow lake. Freshw. Biol. 2013, 58, 2027–2036. [Google Scholar] [CrossRef]
- Hilt, S. Regime shifts between macrophytes and phytoplankton—Concepts beyond shallow lakes, unravelling stabilizing mechanisms and practical consequences. Limnetica 2015, 34, 467–479. [Google Scholar]
- Bradley, P.M.; Barber, L.B.; Duris, J.W.; Foreman, W.T.; Furlong, E.T.; Hubbard, L.E.; Hutchinson, K.J.; Keefe, S.H.; Kolpin, D.W. Riverbank filtration potential of pharmaceuticals in a wastewater-impacted stream. Environ. Pollut. 2014, 193, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Haack, S.K.; Metge, D.W.; Fogarty, L.R.; Meyer, M.T.; Barber, L.B.; Harvey, R.W.; LeBlanc, D.R.; Kolpin, D.W. Effects on groundwater microbial communities of an engineered 30-Day in situ exposure to the antibiotic sulfamethoxazole. Environ. Sci. Technol. 2012, 46, 7478–7486. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.-L.; Zhang, Q.-Q.; Chen, F.; Wang, L.; Ying, G.-G.; Liu, Y.-S.; Yang, B.; Zhou, L.-J.; Liu, S.; Su, H.-C.; et al. Evaluation of triclosan and triclocarban at river basin scale using monitoring and modeling tools: Implications for controlling of urban domestic sewage discharge. Water Res. 2013, 47, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Eerkes-Medrano, D.; Thompson, R.C.; Aldridge, D.C. Microplastics in freshwater systems: A review of the emerging threats, identification of knowledge gaps and prioritisation of research needs. Water Res. 2015, 75, 63–82. [Google Scholar] [CrossRef] [PubMed]
- Conkle, J.L.; Gan, J.; Anderson, M.A. Degradation and sorption of commonly detected PPCPs in wetland sediments under aerobic and anaerobic conditions. J. Soils Sediments 2012, 12, 1164–1173. [Google Scholar] [CrossRef]
- Ebele, A.J.; Abou-Elwafa Abdallah, M.; Harrad, S. Pharmaceuticals and personal care products (PPCPs) in the freshwater aquatic environment. Emerg. Contam. 2017, 3, 1–16. [Google Scholar] [CrossRef]
- Drury, B.; Rosi-Marshall, E.; Kelly, J.J. Wastewater treatment effluent reduces the abundance and diversity of benthic bacterial communities in urban and suburban rivers. Appl. Environ. Microbiol. 2013, 79, 1897–1905. [Google Scholar] [CrossRef] [PubMed]
- Roy, J.W.; Malenica, A. Nutrients and toxic contaminants in shallow groundwater along Lake Simcoe urban shorelines. Inland Waters 2013, 3, 125–138. [Google Scholar] [CrossRef]
- Sand-Jensen, K. Environmental variables and their effect on photosynthesis of aquatic plant communities. Aquat. Bot. 1989, 34, 5–25. [Google Scholar] [CrossRef]
- Maberly, S.C.; Madsen, T.V. Affinity for CO2 in relation to the ability of freshwater macrophytes to use HCO−3. Funct. Ecol. 1998, 12, 99–106. [Google Scholar] [CrossRef]
- Demars, B.O.L.; Tremolieres, M. Aquatic macrophytes as bioindicators of carbon dioxide in groundwater fed rivers. Sci. Total Environ. 2009, 407, 4752–4763. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.J.; Caraco, N.F.; Kling, G.W.; Kratz, T.K. Carbon Dioxide Supersaturation in the Surface Waters of Lakes. Science 1994, 265, 1568–1570. [Google Scholar] [CrossRef] [PubMed]
- Weyhenmeyer, G.A.; Kosten, S.; Wallin, M.B.; Tranvik, L.J.; Jeppesen, E.; Roland, F. Significant fraction of CO2 emissions from boreal lakes derived from hydrologic inorganic carbon inputs. Nat. Geosci. 2015, 8, 933–936. [Google Scholar] [CrossRef]
- Marce, R.; Obrador, B.; Morgui, J.-A.; Lluis Riera, J.; Lopez, P.; Armengol, J. Carbonate weathering as a driver of CO2 supersaturation in lakes. Nat. Geosci. 2015, 8, 107–111. [Google Scholar] [CrossRef]
- Sand-Jensen, K.; Borum, J. Interactions among phytoplankton, periphyton, and macrophytes in temperate freshwaters and estuaries. Aquat. Bot. 1991, 41, 137–175. [Google Scholar] [CrossRef]
- Sand-Jensen, K.; Staehr, P.A. CO2 dynamics along Danish lowland streams: Water-air gradients, piston velocities and evasion rates. Biogeochemistry 2012, 111, 615–628. [Google Scholar] [CrossRef]
- Macpherson, G.L. CO2 distribution in groundwater and the impact of groundwater extraction on the global C cycle. Chem. Geol. 2009, 264, 328–336. [Google Scholar] [CrossRef]
- Vesper, D.J.; Edenborn, H.M. Determination of free CO2 in emergent groundwaters using a commercial beverage carbonation meter. J. Hydrol. 2012, 438–439, 148–155. [Google Scholar] [CrossRef]
- Hutchinson, G.E. A Treatise on Limnology. 3: Limnological Botany; Wiley: New York, NY, USA, 1975; ISBN 978-0-471-42574-8. [Google Scholar]
- Moss, B. Ecology of Fresh Waters: Man and Medium, 2nd ed.; Blackwell Science: Oxford, UK, 1996; ISBN 978-0-632-01642-6. [Google Scholar]
- Jones, J.I. The metabolic cost of bicarbonate use in the submerged plant Elodea nuttallii. Aquat. Bot. 2005, 83, 71–81. [Google Scholar] [CrossRef]
- Olesen, B.; Madsen, T.V. Growth and physiological acclimation to temperature and inorganic carbon availability by two submerged aquatic macrophyte species, Callitriche cophocarpa and Elodea canadensis. Funct. Ecol. 2000, 14, 252–260. [Google Scholar] [CrossRef]
- Maberly, S.C. Photosynthesis by Fontinalis antipyretica. I. New Phytol. 1985, 100, 127–140. [Google Scholar] [CrossRef]
- Maberly, S.C. Photosynthesis by Fontinalis antipyretica. II. New Phytol. 1985, 100, 141–155. [Google Scholar] [CrossRef]
- Maberly, S.C.; Berthelot, S.A.; Stott, A.W.; Gontero, B. Adaptation by macrophytes to inorganic carbon down a river with naturally variable concentrations of CO2. J. Plant Physiol. 2015, 172, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Frandsen, M.; Nilsson, B.; Engesgaard, P.; Pedersen, O. Groundwater seepage stimulates the growth of aquatic macrophytes. Freshw. Biol. 2012, 57, 907–921. [Google Scholar] [CrossRef]
- Takahashi, K.; Asaeda, T. The effect of spring water on the growth of a submerged macrophyte Egeria densa. Landsc. Ecol. Eng. 2012, 10, 99–107. [Google Scholar] [CrossRef]
- Korner, S. Development of submerged macrophytes in shallow Lake Muggelsee (Berlin, Germany) before and after its switch to the phytoplankton-dominated state. Arch. Hydrobiol. 2001, 152, 395–409. [Google Scholar] [CrossRef]
- Maberly, S.C. The fitness of the environments of air and water for photosynthesis, growth, reproduction and dispersal of photoautotrophs: An evolutionary and biogeochemical perspective. Aquat. Bot. 2014, 118, 4–13. [Google Scholar] [CrossRef]
- Gooddy, D.C.; Shand, P.; Kinniburgh, D.G.; Van Riemsdijk, W.H. Field-based partition coefficients for trace elements in soil solutions. Eur. J. Soil Sci. 1995, 46, 265–285. [Google Scholar] [CrossRef]
- Monteith, D.T.; Stoddard, J.L.; Evans, C.D.; de Wit, H.A.; Forsius, M.; Hogasen, T.; Wilander, A.; Skjelkvale, B.L.; Jeffries, D.S.; Vuorenmaa, J.; et al. Dissolved organic carbon trends resulting from changes in atmospheric deposition chemistry. Nature 2007, 450, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Reckhow, D.; Singer, P.; Malcolm, R. Chlorination of humic materials: Byproduct formation and chemical interpretations. Environ. Sci. Technol. 1990, 24, 1655–1664. [Google Scholar] [CrossRef]
- Reyjol, Y.; Argillier, C.; Bonne, W.; Borja, A.; Buijse, A.D.; Cardoso, A.C.; Daufresne, M.; Kernan, M.; Ferreira, M.T.; Poikane, S.; et al. Assessing the ecological status in the context of the European Water Framework Directive: Where do we go now? Sci. Total Environ. 2014, 497, 332–344. [Google Scholar] [CrossRef] [PubMed]
- Birk, S.; Bonne, W.; Borja, A.; Brucet, S.; Courrat, A.; Poikane, S.; Solimini, A.; van de Bund, W.; Zampoukas, N.; Hering, D. Three hundred ways to assess Europe’s surface waters: An almost complete overview of biological methods to implement the Water Framework Directive. Ecol. Indic. 2012, 18, 31–41. [Google Scholar] [CrossRef]
- Jeppesen, E.; Jensen, J.P.; Sondergaard, M.; Lauridsen, T.; Landkildehus, F. Trophic structure, species richness and biodiversity in Danish lakes: Changes along a phosphorus gradient. Freshw. Biol. 2000, 45, 201–218. [Google Scholar] [CrossRef]
- Kuiper, J.J.; Verhofstad, M.J.J.M.; Louwers, E.L.M.; Bakker, E.S.; Brederveld, R.J.; van Gerven, L.P.A.; Janssen, A.B.G.; de Klein, J.J.M.; Mooij, W.M. Mowing submerged macrophytes in shallow lakes with alternative stable states: Battling the good guys? Environ. Manag. 2017, 59, 619–634. [Google Scholar] [CrossRef] [PubMed]
- Hilt, S.; Henschke, I.; Ruecker, J.; Nixdorf, B. Can submerged macrophytes influence turbidity and trophic state in deep lakes? Suggestions from a case study. J. Environ. Qual. 2010, 39, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, R.P.; Sinha, R.P.; Incharoensakdi, A. The cyanotoxin-microcystins: Current overview. Rev. Environ. Sci. Biol. Technol. 2014, 13, 215–249. [Google Scholar] [CrossRef]
- Chorus, I.; Bartram, J. (Eds.) Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring, and Management; E & FN Spon: London, UK; New York, NY, USA, 1999; ISBN 978-0-419-23930-7. [Google Scholar]
- Heisler, J.; Glibert, P.M.; Burkholder, J.M.; Anderson, D.M.; Cochlan, W.; Dennison, W.C.; Dortch, Q.; Gobler, C.J.; Heil, C.A.; Humphries, E.; et al. Eutrophication and harmful algal blooms: A scientific consensus. Harmful Algae 2008, 8, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Grützmacher, G.; Wiese, B.; Hülsoff, I.; Orlikowski, D.; Hoa, E.; Moreau-Le Golvan, Y. Bank Filtration and Aquifer Recharge for Drinking Water Production: Application, Efficiency and Perspectives—An Integration of NASRI Outcomes and International Experiences; Kompetenzzentrum Wasser Berlin gGmbH: Berlin, Germany, 2011. [Google Scholar]
| Country | City | Percentage of Drinking Water Provided by IBF | Source Water Bodies | River Discharge, Lake Volume/Size | Reference |
|---|---|---|---|---|---|
| Austria | * | * | River Enns | 65–206 m3/s | [17] |
| ” | Innsbruck | * | River Inn | 730 m3/s | [18] |
| ” | Vienna, Linz | * | Danube | 1900 m3/s | [19] |
| Bulgaria | * | * | * | - | [19] |
| Finland | Kuopio | * | Lake Kallavesi | 4730 million m3 | [20,21] |
| ” | * | * | Not mentioned | * | [9] |
| France | Paris region | * | Seine River | 450 m3/s | [22] |
| Germany | - | 9 to 16 | - | - | [23,24] |
| ” | Berlin | 60 | - | - | [4] |
| ” | ” | - | Lake Müggelsee | 36 million m3 | [5] |
| ” | ” | - | Lake Tegel | 26 million m3 | [5,25,26,27,28,29,30,31,32] |
| ” | ” | - | Lake Wannsee | 15 million m3 | [5,12,27,33,34,35,36,37] |
| ” | Radeburg | - | Radeburg Reservoir | 0.35 km2 Max depth: 3 m | [38] |
| ” | Düsseldorf | ~100 | River Rhine | 2300 m3/s | [7] |
| ” | Frankfurt am Main | * | River Rhine | 2300 m3/s | [39] |
| ” | ” | * | Lower River Main | 193 m3/s | [39] |
| ” | Torgau and Mockritz | * | Elbe River | 700 m3/s | [40] |
| Hungary | - | 45 | - | - | [7] |
| ” | Budapest | - | Danube | 6460 m3/s | [40] |
| ” | * | - | Rivers Raba, Drava, Ipoly, Sajo, Hernád | 17, 500, 21, 67, 27 m3/s | [41] |
| Italy | Lucca, Pisa, Livorno | (300,000 inhabitants) | River Serchio | 46 m3/s | [42] |
| Latvia | Riga | * | Lake Mazais Baltezers Lake Lielais Baltezers | 10 million m3 18 million m3 | [43] ” |
| The Netherlands | - | 5 | - | - | [7] |
| ” | Remmerden | * | River Rhine | 2300 m3/s | [44] |
| ” | Zwijndrecht | * | River Rhine | 2300 m3/s | ” |
| ” | Roosteren | * | River Meuse | 276 m3/s | ” |
| ” | Roermond | * | Gravel pit lake De Lange Vlieter | 1.2 km2 Max depth: 35 m | [45,46,47] |
| Norway ” | Hemne ” | * * | Lake Rovatnet River Buga | 8 km2 * | [48] |
| Poland | Poznań | * | River Warta | 60 m3/s | [49] |
| Romania | Iasi | * | Moldova River | 143 m3/s | [50] |
| Slovak Republic | - | 50 | - | - | [7] |
| Slovenia | Maribor | - | Drava River | 500 m3/s | [40] |
| Switzerland | - | 10–30 | - River Thur | - 40–50 m3/s | [13,51] |
| UK | * | * | Streams Wissey, Rhee and Pang | 1.9, 1.25, 0.64 m3/s | [52] |
| Canada | * | * | Lake A and B (artificial) | * | [10] |
| USA | Jeffersonville | * | Ohio River | 3512 m3/s | [53,54] |
| ” | Santa Rosa | * | Russian River | 66 m3/s | [55] |
| ” | Cincinnati | * | Great Miami River | 109 m3/s | [56] |
| ” | Columbus | * | Scioto/Big Walnut Creek | 6 m3/s | ” |
| ” | Galesburg | * | Mississippi River | 16,792 m3/s | ” |
| ” | Independence Kansas City Parkville | * | Missouri River | 2158 m3/s | ” ” [54] |
| ” | Jacksonville | * | Illinois River | 659 m3/s | [56] |
| ” | Kalama | * | Kalama River | 40 m3/s | ” |
| ” | Kennewick | * | Columbia River | 7500 m3/s | ” |
| ” | Lincoln | * | Platte River | 203 m3/s | ” |
| ” | Mt. Carmel Terre Haute | * | Wabash River | 837 m3/s | ” [54] |
| ” | Sacramento | * | Sacramento River | 660 m3/s | ” |
| ” | Cape Cod | * | Ashumet Pond | 6 million m3 | [57] |
| Brazil | * | * | Beberibe River | 0.3–0.4 m water depth | [58] |
| ” | * | * | Lake Lagoa do Peri | 36 million m3 | [59,60] |
| China | Matan | 96 | Yellow River | 1839 m3/s | [61] |
| ” | Baisha Town | 82.1 | Yangtze River | 31,100 m3/s | ” |
| ” | Jiuwutan | 82.6 | Yellow River | 1839 m3/s | ” |
| ” | Qingpu district | 70–80 | Taipu River | 300 m3/s | ” |
| ” | Xuzhou | >80 | Kui River | * | ” |
| ” | Chengdu | 80 | Yinma River | 30 m3/s | ” |
| India | - | * | - | - | [62] |
| ” | Delhi | * | Yamuna River | 100–1300 m3/s | [63] |
| ” | Satpuli Srinagar | * * | East Nayar River River Alaknanda | - 507 m3/s | [64] |
| ” | Haridwar | * | River Ganga | 1455 m3/s | [65] |
| ” | Nainital | * | Lake Nainital | 6 million m3 | [66] |
| Malaysia | Kuala Kangsar | * | Sungai Perak (river) | 57 m3/s | [67] |
| South Korea | * | * | Nakdong River | 37–3462 m3/s | [68] |
| Thailand | Chiang Mai | * | Ping River | 287 m3/s | [69] |
| Egypt | - | 0.1 (increasing) | Upper Nile | 1548 m3/s | [70] |
| ” | Sidfa | * | Nile | 2830 m3/s | [71] |
| ” | Aswan | * | ” | ” | [72] |
| Parameter | Predicted Effect | Mechanism | References | Affected Biological Parameter | Effect | References (Example) |
|---|---|---|---|---|---|---|
| Physical | Higher summer WT | I | [93] | Biodiversity Macrophyte dominance Harmful blooms | ± − + | [94] [95] [95] |
| Lower winter WT | I | [96] | Biodiversity Macrophyte dominance Harmful blooms | ? − ? | [97] | |
| Higher RT | D, I | Biodiversity Macrophyte presence Harmful blooms | ± ± ± | [98] [92] [99] | ||
| Lower flow | D, I | [89] | Biodiversity Macrophyte presence Harmful blooms | ± ± + | [87] [100] [101] | |
| Sediment clogging | D | [26] | Biodiversity Macrophyte dominance Harmful blooms | ? ± ? | [102] | |
| Lower WL | D, I | [103] | Biodiversity Macrophyte presence Harmful blooms | − (?) − + | [103] [99] | |
| Stronger WL fluctuations | D, I | [103] | Biodiversity Macrophyte presence Harmful blooms | ± − ? | [104] [105] | |
| Chemical | Lower DIC | I | [106] | Biodiversity Macrophyte dominance Harmful blooms | ± − ± | [98] [107] [108] |
| Lower external nutrient load | I | Biodiversity Macrophyte dominance Harmful blooms | + + − | [109] [110] [108] | ||
| Lower DOC | I | [106] | Biodiversity Macrophyte dominance Harmful blooms | + (?) + (?) +/− | [111] [112] [113]/[114] | |
| Lower pollutant load | I | Biodiversity Macrophyte dominance Harmful blooms | + + (?) ? | [115] | ||
| Higher pollutant load in the littoral | I | Biodiversity Macrophyte dominance Harmful blooms | − − (?) ? | [115] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gillefalk, M.; Massmann, G.; Nützmann, G.; Hilt, S. Potential Impacts of Induced Bank Filtration on Surface Water Quality: A Conceptual Framework for Future Research. Water 2018, 10, 1240. https://doi.org/10.3390/w10091240
Gillefalk M, Massmann G, Nützmann G, Hilt S. Potential Impacts of Induced Bank Filtration on Surface Water Quality: A Conceptual Framework for Future Research. Water. 2018; 10(9):1240. https://doi.org/10.3390/w10091240
Chicago/Turabian StyleGillefalk, Mikael, Gudrun Massmann, Gunnar Nützmann, and Sabine Hilt. 2018. "Potential Impacts of Induced Bank Filtration on Surface Water Quality: A Conceptual Framework for Future Research" Water 10, no. 9: 1240. https://doi.org/10.3390/w10091240
APA StyleGillefalk, M., Massmann, G., Nützmann, G., & Hilt, S. (2018). Potential Impacts of Induced Bank Filtration on Surface Water Quality: A Conceptual Framework for Future Research. Water, 10(9), 1240. https://doi.org/10.3390/w10091240