The Use of a Polymer Inclusion Membrane for Arsenate Determination in Groundwater
Abstract
:1. Introduction
2. Methods
2.1. Reagents and Solutions
2.2. Colorimetric Detection of As(V)
2.3. Polymer Inclusion Membrane (PIM) Preparation
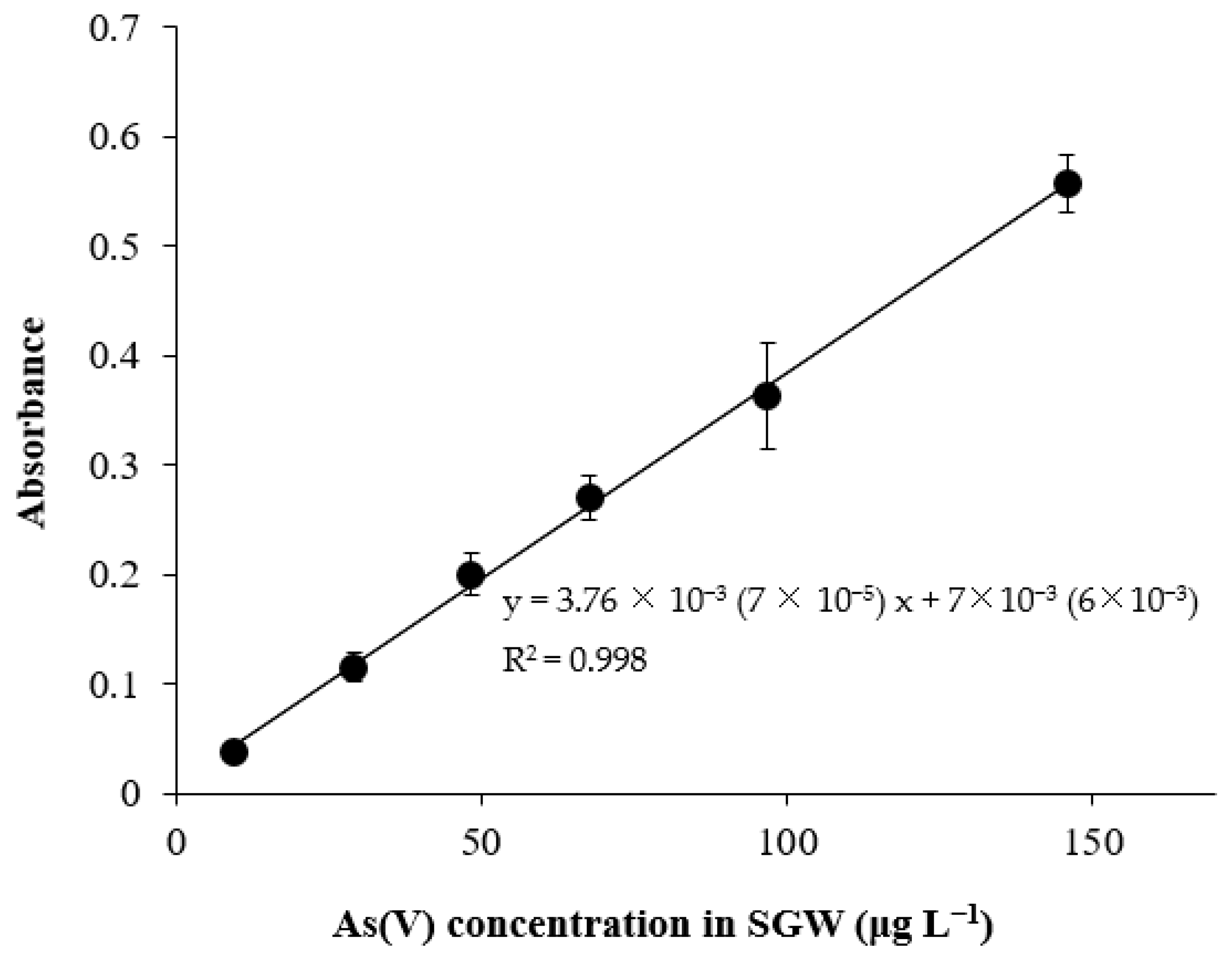
2.4. Preconcentration Experiments and Calibration Curve
2.5. Apparatus
2.6. Water Samples
3. Results and Discussion
3.1. Parameters Affecting the Preconcentration System
3.1.1. Stripping Composition
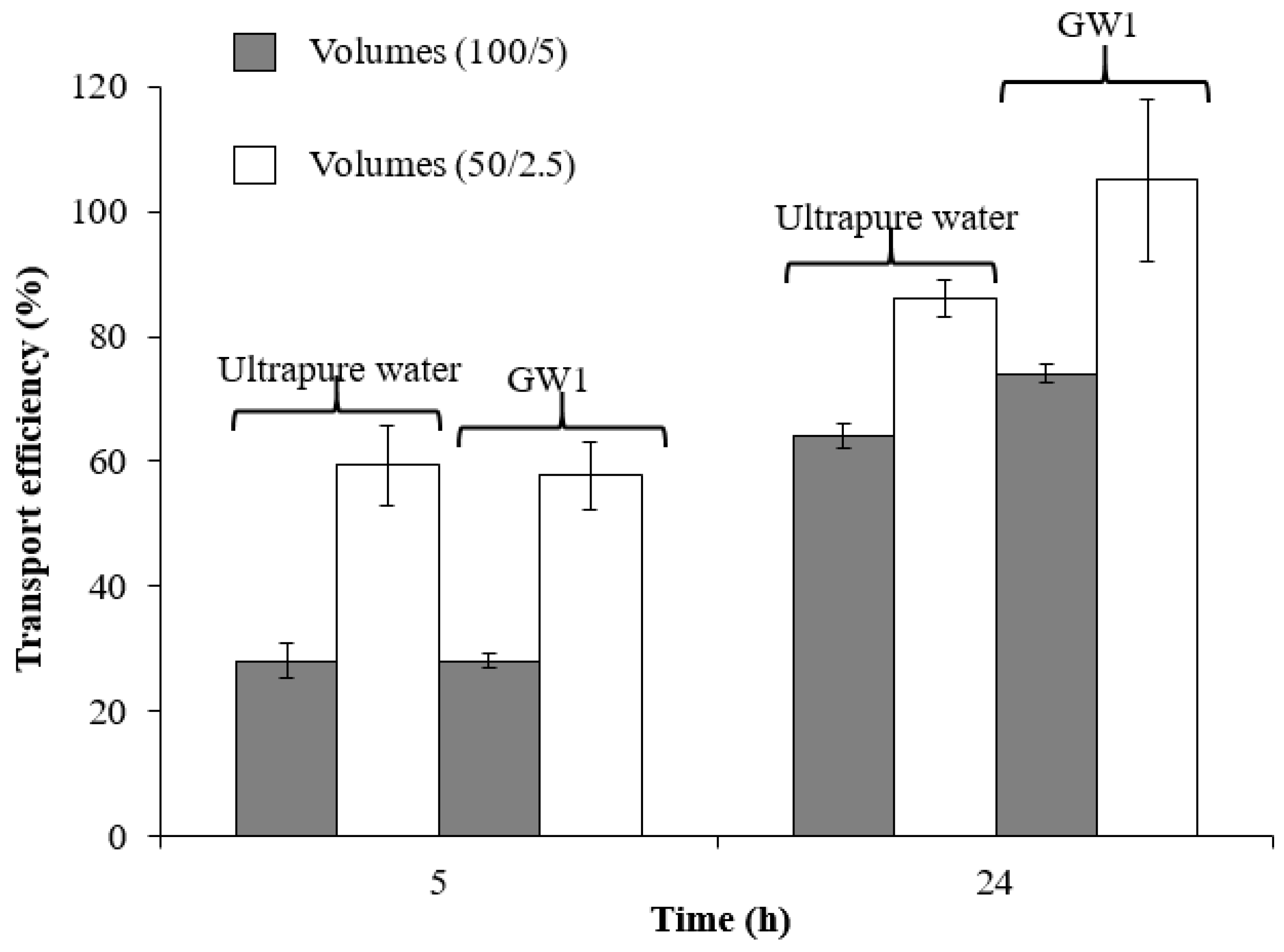
3.1.2. Contact Time and Sample and Stripping Volume
3.1.3. Membrane Characteristics
3.2. Analytical Application of the PIM-device
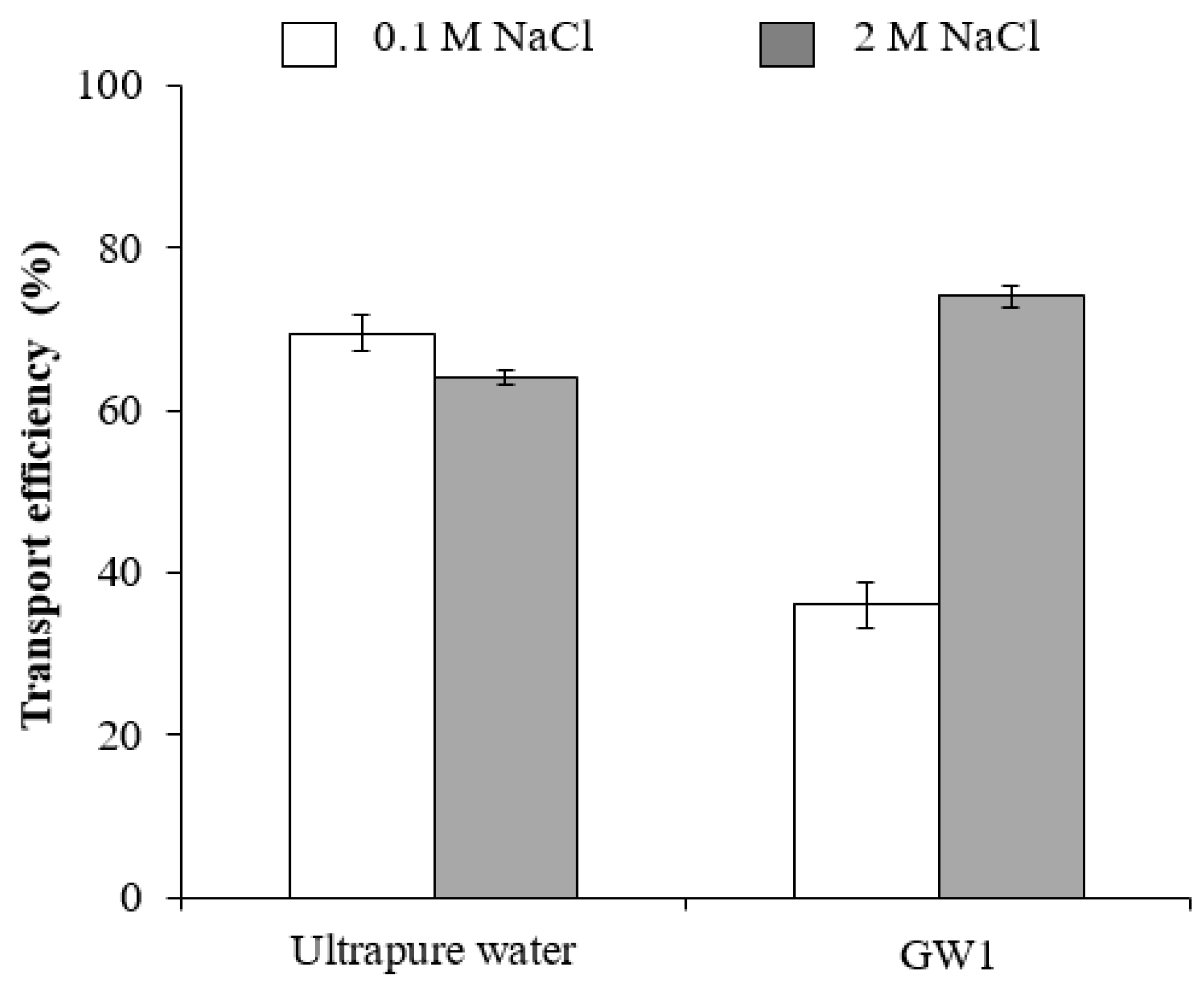
3.2.1. Effect of Water Matrix
3.2.2. Analytical Parameters
3.3. Application to Contaminated Groundwater (GW) Samples
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Villaescusa, I.; Bollinger, J.-C. Arsenic in drinking water: Sources, occurrence and health effects (a review). Rev. Environ. Sci. Bio./Technol. 2008, 7, 307–323. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Guidelines for Drinking-Water Quality, 4th ed.; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Almeida, M.I.G.S.; Cattrall, R.W.; Kolev, S.D. Polymer inclusion membranes (PIMs) in chemical analysis—A review. Anal. Chim. Acta 2017, 987, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.I.G.S.; Chan, C.; Pettigrove, V.J.; Cattrall, R.W.; Kolev, S.D. Development of a passive sampler for Zinc (II) in urban pond waters using a polymer inclusion membrane. Environ. pollut. 2014, 193, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.I.G.S.; Silva, A.M.L.; Coleman, R.A.; Pettigrove, V.J.; Cattrall, R.W.; Kolev, S.D. Development of a passive sampler based on a polymer inclusion membrane for total ammonia monitoring in freshwaters. Anal. Bioanal. Chem. 2016, 408, 3213–3222. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rodríguez, A.; Matamoros, V.; Kolev, S.D.; Fontàs, C. Development of a polymer inclusion membrane (PIM) for the preconcentration of antibiotics in environmental water samples. J. Membr. Sci. 2015, 492, 32–39. [Google Scholar] [CrossRef]
- Pont, N.; Salvadó, V.; Fontàs, C. Selective transport and removal of cd from chloride solutions by polymer inclusion membranes. J. Membr. Sci. 2008, 318, 340–345. [Google Scholar] [CrossRef]
- Güell, R.; Anticó, E.; Kolev, S.D.; Benavente, J.; Salvadó, V.; Fontàs, C. Development and characterization of polymer inclusion membranes for the separation and speciation of inorganic as species. J. Membr. Sci. 2011, 383, 88–95. [Google Scholar] [CrossRef]
- Fontàs, C.; Vera, R.; Batalla, A.; Kolev, S.D.; Anticó, E. A novel low-cost detection method for screening of arsenic in groundwater. Environ. Sci. Pollut. Res. 2014, 21, 11682–11688. [Google Scholar] [CrossRef] [PubMed]
- Tsang, S.; Phu, F.; Baum, M.M.; Poskrebyshev, G.A. Determination of phosphate/arsenate by a modified molybdenum blue method and reduction of arsenate by S2O42−. Talanta 2007, 71, 1560–1568. [Google Scholar] [CrossRef] [PubMed]
- Vera, R.; Fontàs, C.; Galceran, J.; Serra, O.; Anticó, E. Polymer inclusion membrane to access Zn speciation: Comparison with root uptake. Sci. Total Environ. 2018, 622–623, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rodríguez, A.; Fontàs, C.; Matamoros, V.; Almeida, M.I.G.S.; Cattrall, R.W.; Kolev, S.D. Development of a polymer inclusion membrane-based passive sampler for monitoring of sulfamethoxazole in natural waters. Minimizing the effect of the flow pattern of the aquatic system. Microchem. J. 2016, 124, 175–180. [Google Scholar] [CrossRef]
- Vázquez, M.I.; Romero, V.; Fontàs, C.; Anticó, E.; Benavente, J. Polymer inclusion membranes (PIMs) with the ionic liquid (IL) Aliquat 336 as extractant: Effect of base polymer and IL concentration on their physical–chemical and elastic characteristics. J. Membr. Sci. 2014, 455, 312–319. [Google Scholar] [CrossRef]
- Kebiche-Senhadji, O.; Tingry, S.; Seta, P.; Benamor, M. Selective extraction of Cr (VI) over metallic species by polymer inclusion membrane (PIM) using anion (Aliquat 336) as carrier. Desalination 2010, 258, 59–65. [Google Scholar] [CrossRef]
- Konczyk, J.; Kozlowski, C.; Walkowiak, W. Removal of chromium (III) from acidic aqueous solution by polymer inclusion membranes with D2EHPA and Aliquat 336. Desalination 2010, 263, 211–216. [Google Scholar] [CrossRef]
- Mohapatra, P.K.; Lakshmi, D.S.; Bhattacharyya, A.; Manchanda, V.K. Evaluation of polymer inclusion membranes containing crown ethers for selective cesium separation from nuclear waste solution. J. Hazardous Mater. 2009, 169, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Kusumocahyo, S.P.; Kanamori, T.; Sumaru, K.; Aomatsu, S.; Matsuyama, H.; Teramoto, M.; Shinbo, T. Development of polymer inclusion membranes based on cellulose triacetate: Carrier-mediated transport of cerium (III). J. Membr. Sci. 2004, 244, 251–257. [Google Scholar] [CrossRef]
- AOAC Official Methods of analysis. Appendix F: Guidelines for Standard Method Performance Requirements; AOAC International: Rockville, Maryland, 2012. [Google Scholar]
| Samples | Georeferences of Sampling Point (Coordinates) | pH | Conductivity (μS cm−1) | Ions (mg L−1) | Arsenic Concentration (μg L−1) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| [NO3−] | [Cl−] | [SO42−] | [HCO3−] | [Na+] | [Mg+] | [Ca+] | |||||
| GW1 (Pujarnol) | 42°6′16.907″ N Lat., 2°42′34.64″ E Long. | 7.21 | 684 | 1.2 | 15.4 | 63.9 | 269 | 19.0 | 30.1 | 96.1 | n.f. |
| GW2 (Mongai) | 41°47′59.047″ N Lat., 0°57′38.832″ E Long. | 7.76 | 470 | 9.5 | 15 | 38.3 | n.m. | 10.8 | 14.9 | 68.9 | n.f. |
| GW3 (St. Hilari) | 41°53′16.46″ N Lat., 2°31′11.867″ E Long. | 7.98 | 275 | 21.9 | 11.4 | 8.7 | 172 | 16.6 | 9.0 | 50.0 | n.f. |
| GW4 (Cerdanya) | 42°21′16.059″ N Lat., 1°42′17.742″ E Long. | 7.5 | 423 | 0.3 | 4.3 | 2.5 | 349 | 11.4 | 13.2 | 70.7 | n.f. |
| GW5 (Setcases) | 42°22′22.208″ N Lat., 2°18′3.026″ E Long. | 7.56 | 110 | 3.3 | 0.9 | 8.0 | 88 | 3.7 | 2.7 | 23.6 | n.f. |
| GW6 (Cerdanya) | 42°22′16.393″ N Lat., 1°40′41.159″ E Long. | 7.5 | 236 | 2.2 | 2.7 | 12.4 | 140 | n.m. | n.m. | n.m. | 67.1 |
| GW7 (Cerdanya) | 42°22′12.595″ N Lat., 1°40′54.456″ E Long. | 9.69 | 185 | 0.4 | 2.2 | 11.8 | n.m. | n.m. | n.m. | n.m. | 70.4 |
| SGW | - | 7.5 | 459 | n.a. | 71 | 47.0 | 123 | 70.2 | n.a. | 40.3 | n.a. |
| Polymer | PIM Composition (w/w) | Thickness (μm) | [As] Stripping (μg L−1) (± SD) |
|---|---|---|---|
| Poly(vinyl chloride) (PVC) | 69% PVC–31% Aliquat 336 | 60 | 1160 (± 58) |
| 30 | 940 (± 94) | ||
| 50% PVC–50% Aliquat 336 | 96 | 900 (± 45) | |
| 39 | 840 (± 53) | ||
| Cellulose triacetate (CTA) | 70% CTA–30% Aliquat 336 | 38 | 880 (± 18) |
| 52% CTA–48% Aliquat 336 | 45 | 1060 (± 85) | |
| 25 | 940 (± 19) |
| Water Sample | Conductivity (μS cm−1) | Amount of As(V) Added (μg L−1) | TE (%) (± SD) |
|---|---|---|---|
| GW1 | 684 | 100 | 53 (± 8) |
| GW2 | 470 | 100 | 65 (± 6) |
| GW3 | 275 | 100 | 72 (± 6) |
| GW4 | 423 | 100 | 66 (± 23) |
| GW5 | 110 | 100 | 81 (± 11) |
| SGW | 459 | 100 | 63 (± 8) |
| Water Sample | As(V) Recovery (%) (± SD) |
|---|---|
| GW1 | 79 (± 13) |
| GW2 | 98 (± 10) |
| GW3 | 109 (± 9) |
| GW4 | 100 (± 35) |
| GW5 | 124 (± 17) |
| Water Sample | As Concentration Measured (μg L−1) (± SD) | |
|---|---|---|
| ICP-OES | PIM-Based Device | |
| GW6 | 67 (± 2) | 82 (± 2) |
| GW7 | 70 (± 3) | 67.4 (± 0.5) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vera, R.; Anticó, E.; Fontàs, C. The Use of a Polymer Inclusion Membrane for Arsenate Determination in Groundwater. Water 2018, 10, 1093. https://doi.org/10.3390/w10081093
Vera R, Anticó E, Fontàs C. The Use of a Polymer Inclusion Membrane for Arsenate Determination in Groundwater. Water. 2018; 10(8):1093. https://doi.org/10.3390/w10081093
Chicago/Turabian StyleVera, Ruben, Enriqueta Anticó, and Clàudia Fontàs. 2018. "The Use of a Polymer Inclusion Membrane for Arsenate Determination in Groundwater" Water 10, no. 8: 1093. https://doi.org/10.3390/w10081093
APA StyleVera, R., Anticó, E., & Fontàs, C. (2018). The Use of a Polymer Inclusion Membrane for Arsenate Determination in Groundwater. Water, 10(8), 1093. https://doi.org/10.3390/w10081093







