Adsorption of Reactive Brilliant Red X-3B in Aqueous Solutions on Clay–Biochar Composites from Bagasse and Natural Attapulgite
Abstract
:1. Introduction
2. Materials and Methods
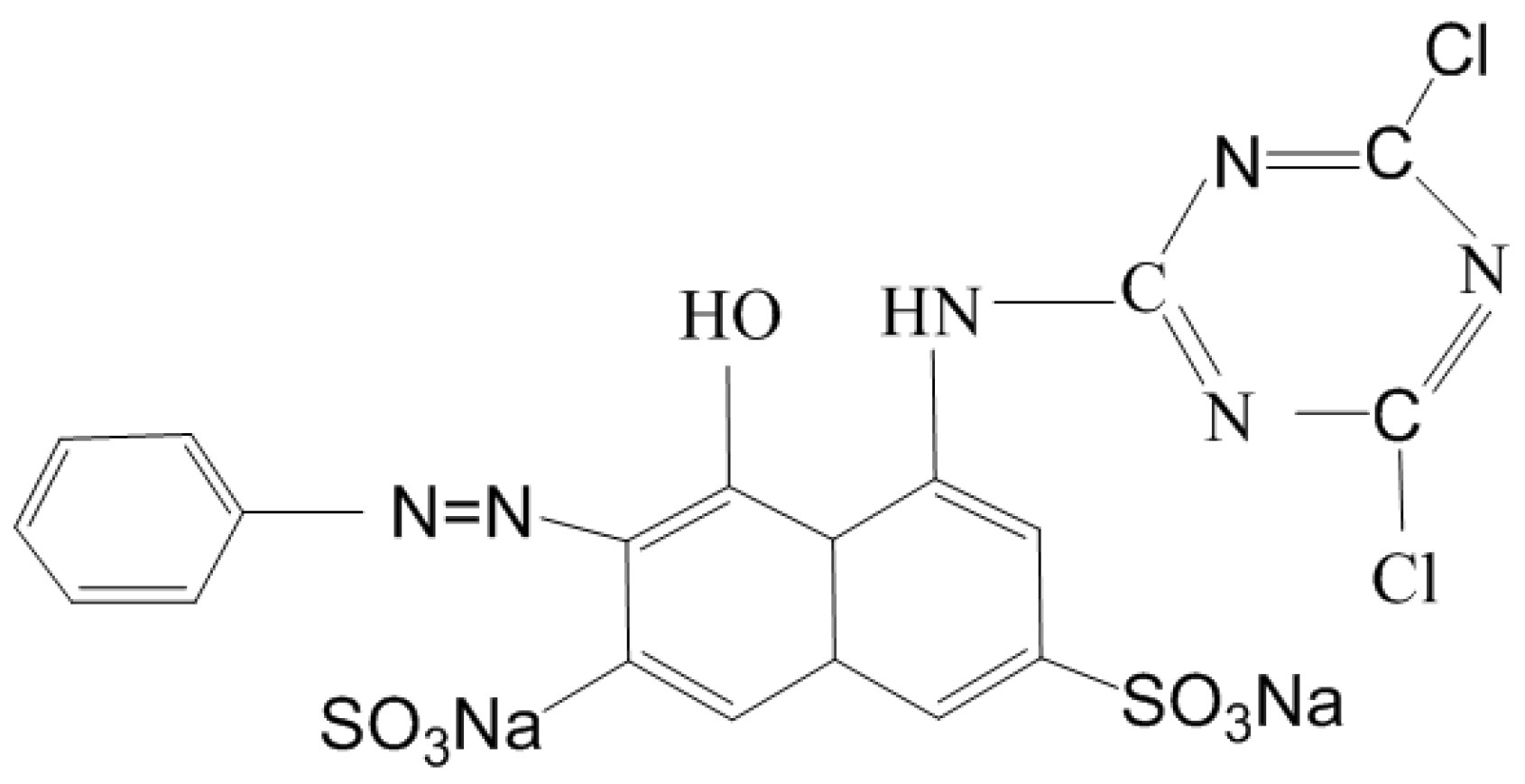
2.1. Reagents
2.2. Preparation of Biochar
2.3. Physical and Chemical Properties of Biochar Samples
2.4. Adsorption Experiments
3. Results and Discussion
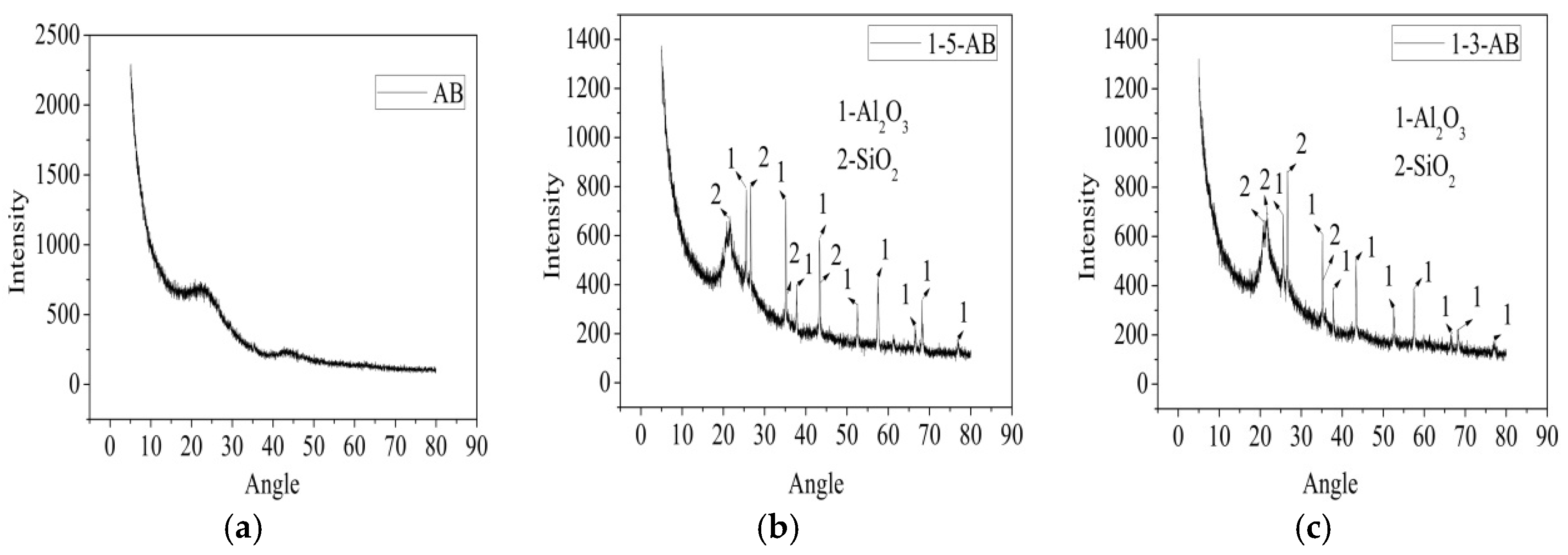
3.1. Biochar Properties
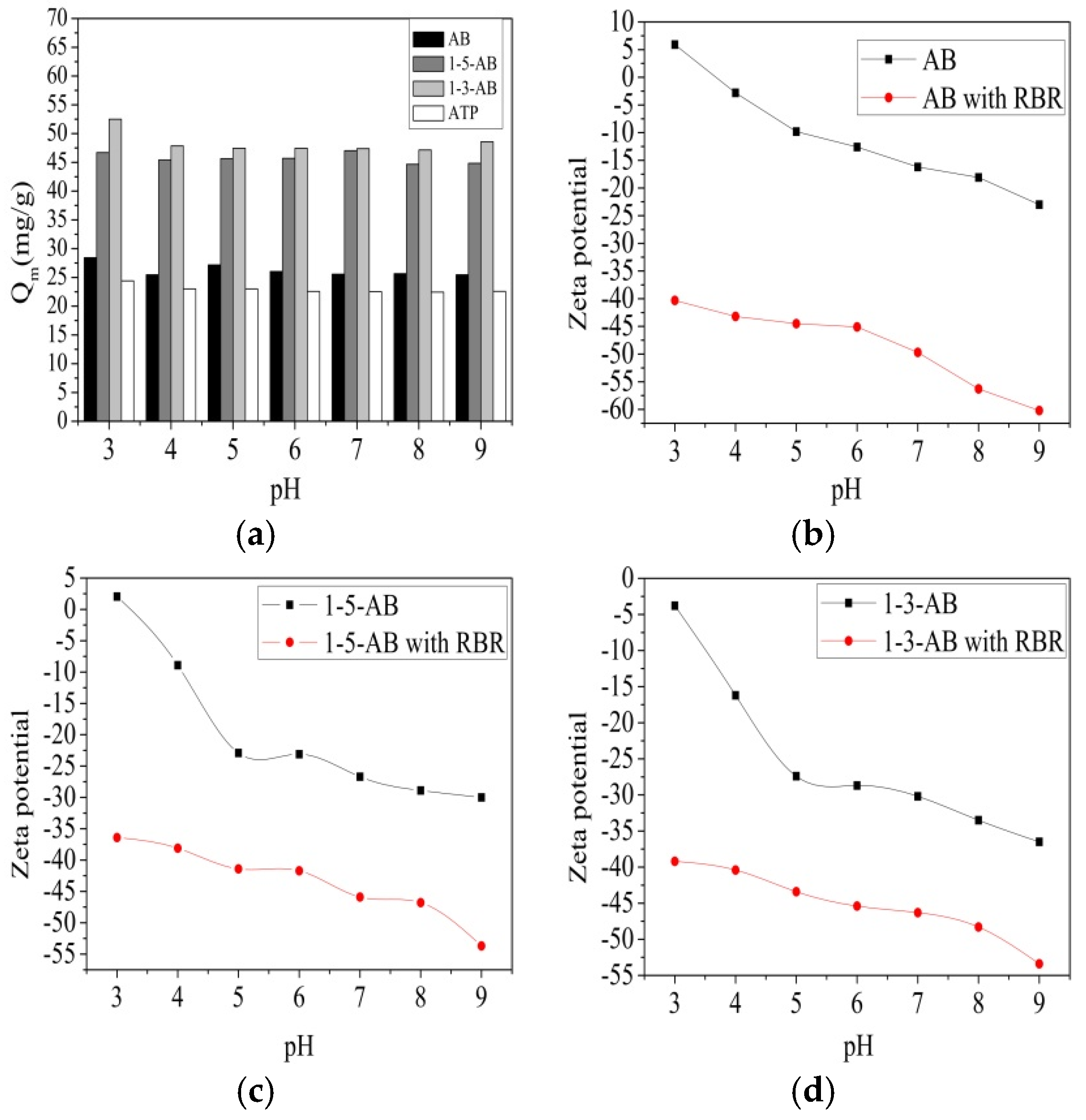
3.2.Effects of pH on Zeta Potential and the Adsorption of RBR by the Biochar Samples
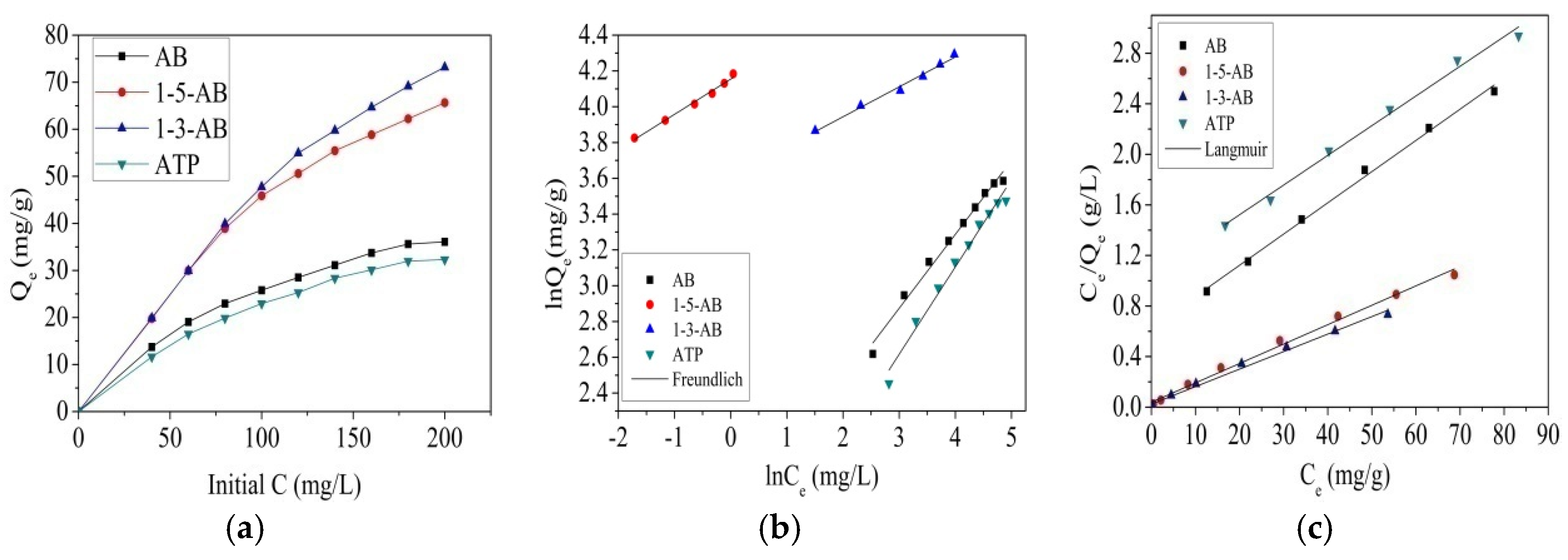
3.3. Adsorption Isotherms of RBR by the Biochar Samples
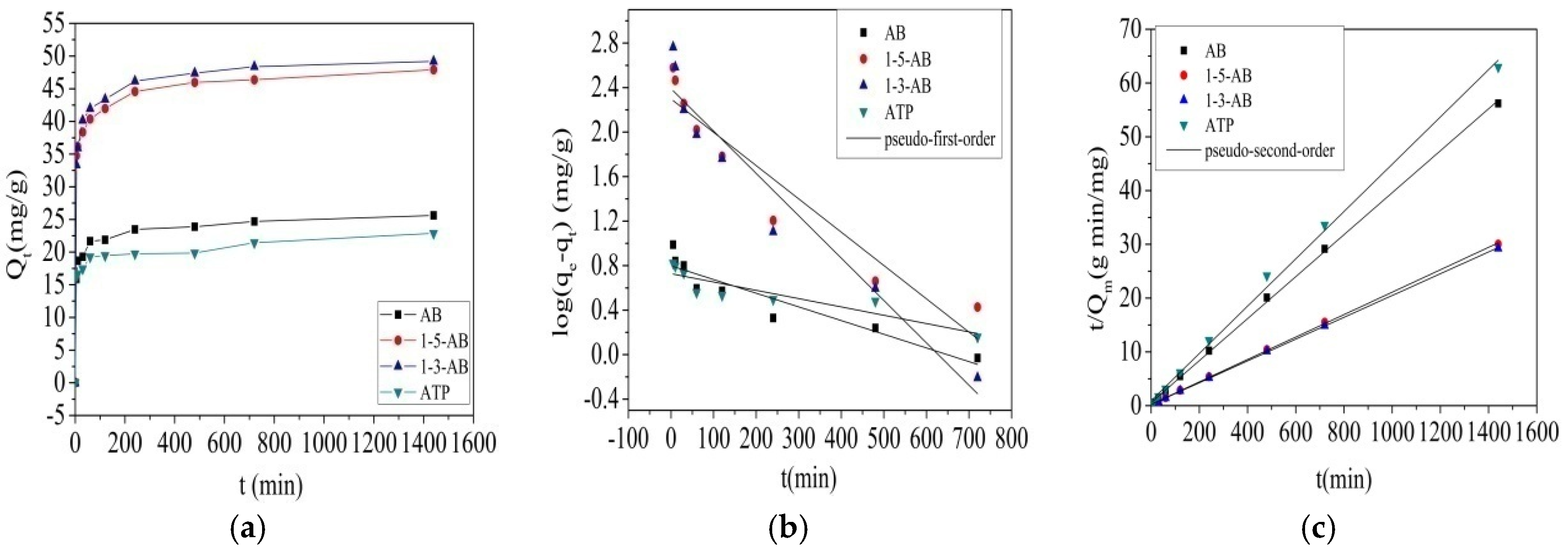
3.4. Adsorption Kinetics
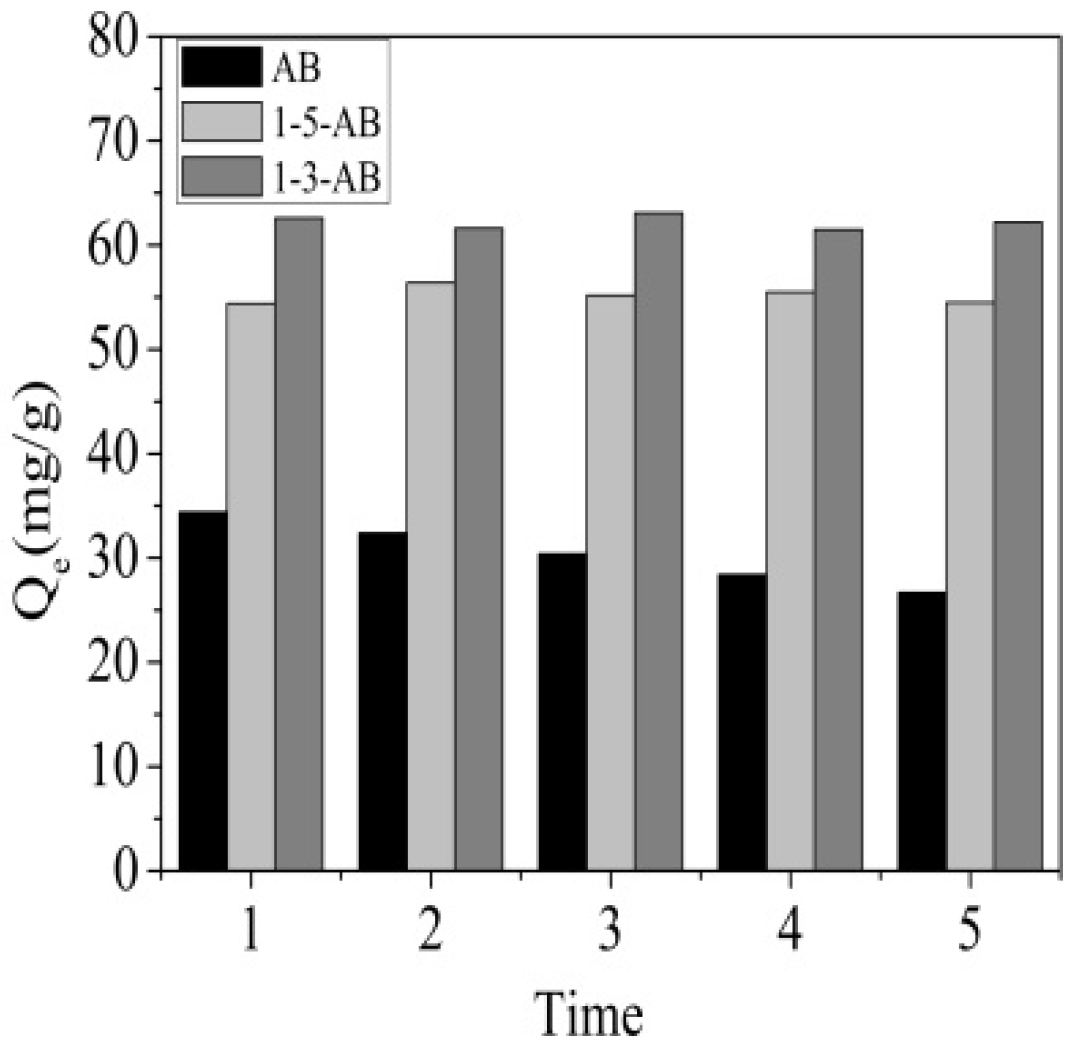
3.5. Regeneration of Adsorbents
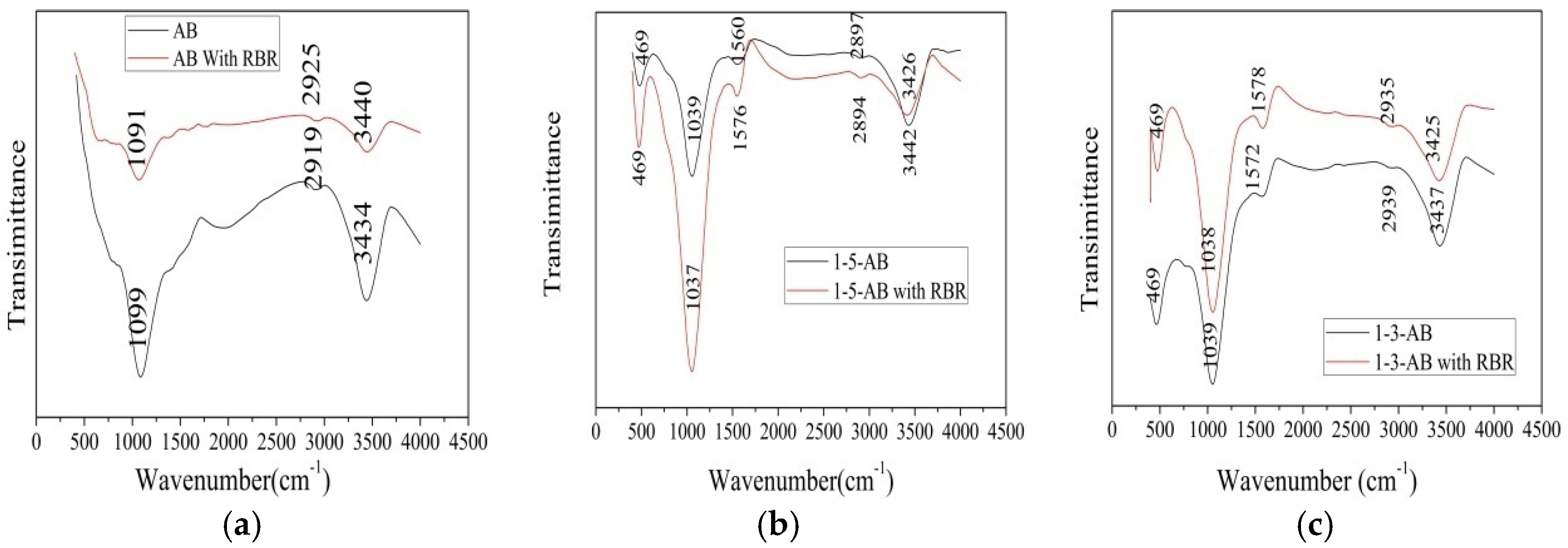
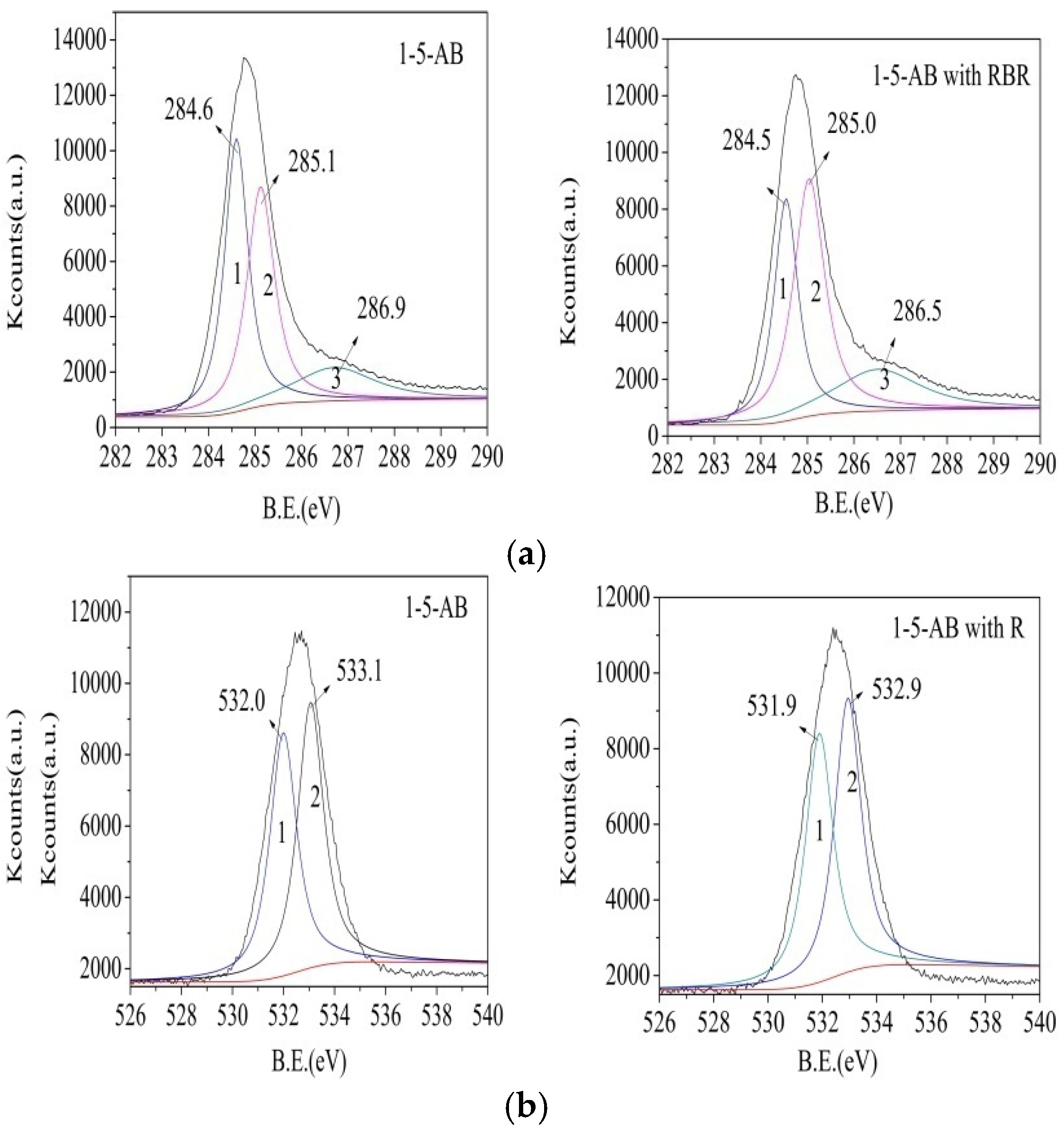
3.6. Adsorption Mechanisms
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carneiro, P.A.; Umbuzeiro, G.A.; Oliveira, D.P.; Zanoni, M.V. Assessment of water contamination caused by a mutagenic textile effluent/dyehouse effluent bearing disperse dyes. J. Hazard. Mater. 2016, 174, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Dubey, S.K.; Yadav, R.; Chaturvedi, R.K.; Yadav, R.K.; Sharma, V.K.; Minhas, P.S. Contamination of ground water as a consequence of land disposal of dye waste mixed sewage effluents: A case study of Panipat district of Haryana, India. Bull. Environ. Contam. Toxicol. 2010, 85, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.X.; Wang, M.E. Adsorption–desorption characteristics and pollution behavior of reactive X-3B red dye in four Chinese typical soils. J. Soils Sediments 2010, 10, 1324–1334. [Google Scholar] [CrossRef]
- Topac, F.O.; Dindar, E.; Ucaroglu, S.; Baskaya, H.S. Effect of a sulfonated azo dye and sulfanilic acid on nitrogen transformation processes in soil. J. Hazard. Mater. 2009, 170, 1006–1013. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Zhang, J.; Zhang, J.; Zhang, H. Degradation pathway and mechanism of Reactive Brilliant Red X-3B in electro-assisted microbial system under anaerobic condition. J. Hazard. Mater. 2017, 329, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Faria, P.C.; Orfao, J.J.; Pereira, M.F. Adsorption of anionic and cationic dyes on activated carbons with different surface chemistries. Water Res. 2004, 38, 2043–2052. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhang, J.; Zhang, C.; Yue, Q.; Li, Y.; Li, C. Equilibrium and kinetic studies of methyl orange and methyl violet adsorption on activated carbon derived from Phragmites australis. Desalination 2010, 252, 149–156. [Google Scholar] [CrossRef]
- Soldatkina, L.M.; Sagaidak, E.V. Kinetics of adsorption of water-soluble dyes on activated carbons. J. Water Chem. Technol. 2010, 32, 212–217. [Google Scholar] [CrossRef]
- Liu, R.; Zhang, B.; Mei, D.; Zhang, H.; Liu, J. Adsorption of methyl violet from aqueous solution by halloysite nanotubes. Desalination 2011, 268, 111–116. [Google Scholar] [CrossRef]
- Zhang, M.; Gao, B.; Varnoosfaderani, S.; Hebard, A.; Yao, Y.; Inyang, M. Preparation and characterization of a novel magnetic biochar for arsenic removal. Bioresour. Technol. 2013, 130, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Su, Y.J.; Wang, Y.; Liu, B.; Sun, L.M. Bioadsorption of methyl violet from aqueous solution onto Pu-erh tea powder. J. Hazard. Mater. 2010, 179, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Ofomaja, A.E.; Ho, Y.S. Effect of temperatures and pH on methyl violet biosorption by Mansonia wood sawdust. Bioresour. Technol. 2008, 99, 5411–5417. [Google Scholar] [CrossRef] [PubMed]
- Hameed, B.H. Equilibrium and kinetic studies of methyl violet sorption by agricultural waste. J. Hazard. Mater. 2008, 154, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Ali, I.; Saini, V.K.; Gerven, T.V.; der Bruggen, B.V.; Vandecasteele, C. Removal of Dyes from Wastewater Using Bottom Ash. Ind. Eng. Chem. Res. 2005, 44, 3655–3664. [Google Scholar] [CrossRef]
- Solpan, D.; Duran, S.; Saraydin, D.; Güven, O. Adsorption of methyl violet in aqueous solutions by poly(acrylamide-co-acrylic acid) hydrogels. Radiat. Phys. Chem. 2003, 66, 117–127. [Google Scholar] [CrossRef]
- Doğan, M.; Alkan, M. Removal of methyl violet from aqueous solution by perlite. J. Colloid Interface Sci. 2003, 267, 32–41. [Google Scholar] [CrossRef]
- Tong, X.-J.; Li, J.-Y.; Yuan, J.-H.; Xu, R.-K. Adsorption of Cu(II) by biochars generated from three crop straws. Chem. Eng. J. 2011, 172, 828–834. [Google Scholar] [CrossRef]
- Hossain, M.K.; Strezov, V.; Chan, K.Y.; Nelson, P.F. Agronomic properties of wastewater sludge biochar and bioavailability of metals in production of cherry tomato (Lycopersicon esculentum). Chemosphere 2010, 78, 1167–1171. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Gao, B.; Inyang, M.; Zimmerman, A.R.; Cao, X.; Pullammanappallil, P.; Yang, L. Biochar derived from anaerobically digested sugar beet tailings: Characterization and phosphate removal potential. Bioresour. Technol. 2011, 102, 6273–6278. [Google Scholar] [CrossRef] [PubMed]
- Enders, A.; Hanley, K.; Whitman, T.; Joseph, S.; Lehmann, J. Characterization of biochars to evaluate recalcitrance and agronomic performance. Bioresour. Technol. 2012, 114, 644–653. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Hu, X.; Zimmerman, A.R.; Gao, B. Sorption and cosorption of lead (II) and methylene blue on chemically modified biomass. Bioresour. Technol. 2014, 167, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Agrafioti, E.; Kalderis, D.; Diamadopoulos, E. Ca and Fe modified biochars as adsorbents of arsenic and chromium in aqueous solutions. J. Environ. Manag. 2014, 146, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Zuo, X.; Liu, Z.; Chen, M. Effect of H2O2 concentrations on copper removal using the modified hydrothermal biochar. Bioresour. Technol. 2016, 207, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.X.; Jiang, H. Amino modification of biochar for enhanced adsorption of copper ions from synthetic wastewater. Water Res. 2014, 48, 396–405. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Xu, R.-K.; Jiang, T.-Y.; Li, Z. Immobilization of Cu(II), Pb(II) and Cd(II) by the addition of rice straw derived biochar to a simulated polluted Ultisol. J. Hazard. Mater. 2012, 229–230, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Chen, Z.; Lv, S. A novel magnetic biochar efficiently sorbs organic pollutants and phosphate. Bioresour. Technol. 2011, 102, 716–723. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Charrua, A.B.; Weng, C.-H.; Yuan, X.; Ding, F. Characterization of biochars derived from agriculture wastes and their adsorptive removal of atrazine from aqueous solution: A comparative study. Bioresour. Technol. 2015, 198, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, M.E.; Nabil, G.M.; EI-Mallah, N.M.; Bassiluny, H.I.; Kumar, S.; Abdel-Fattah, T.M. Kinetics, isotherm, and thermodynamic studies of the adsorption of reactive red 195 A dye from water by modified switchgrass Biocha radsorbent. J. Ind. Eng. Chem. 2016, 37, 156–167. [Google Scholar] [CrossRef]
- Angin, D.; Kose, T.E.; Selengil, U. production and characterization of activated carbon prepared from safflower seed cake biochar and its ability to absorb reactive dyestuff. Appl. Surface Sci. 2013, 280, 705–710. [Google Scholar] [CrossRef]
- Khataee, A.; Kayan, B.; Gholami, P.; Kalderis, D.; Akay, S.; Dinpazhoh, L. Sonocatalytic degradation of Reactive Yellow 39 using synthesized ZrO2 nanoparticles on biochar. Ultrason. Sono Chem. 2017, 39, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, Z.; Xie, X.; Zhu, J.; Li, R.; Qin, T. Removal of Norfloxacin from aqueous solution by clay–biochar composite prepared from potato stem and natural attapulgite. Colloids Surf. A Physicochem. Eng. Aspects 2017, 514, 126–136. [Google Scholar] [CrossRef]
- Fan, Q.-H.; Tan, X.-L.; Li, J.-X.; Wang, X.-K.; Wu, W.-S.; Montavon, G. Sorption of Eu(III) on attapulgite studied by batch, XPS, and EXAFS techniques. Environ. Sci. Technol. 2009, 43, 5776–5782. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, D.K.; Salleh, M.A.M.; Karim, W.A.; Idris, A.; Abidin, Z.Z. Batch adsorption of basic dye using acid treated kenaf fibre char: Equilibrium, kinetic and thermodynamic studies. Chem. Eng. J. 2012, 181–182, 449–457. [Google Scholar] [CrossRef]
- Xue, Y.; Gao, B.; Yao, Y.; Inyang, M.; Zhang, M.; Zimmerman, A.R.; Ro, K.S. Hydrogen peroxide modification enhances the ability of biochar (hydrochar) produced from hydrothermal carbonization of peanut hull to remove aqueous heavy metals: Batch and column tests. Chem. Eng. J. 2012, 200–202, 673–680. [Google Scholar] [CrossRef]
- Mohan, D.; Sarswat, A.; Ok, Y.S.; Pittman, C.U., Jr. Organic and inorganic contaminants removal from water with biochar, a renewable, low cost and sustainable adsorbent—A critical review. Bioresour. Technol. 2014, 160, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Gao, B.; Yao, Y.; Xue, Y.; Inyang, M. Synthesis of porous MgO-biochar nanocomposites for removal of phosphate and nitrate from aqueous solutions. Chem. Eng. J. 2012, 210, 26–32. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, B.; Fang, J.; Zhang, M.; Chen, H.; Zhou, Y.-M.; Creamer, A.E.; Sun, Y.-N.; Yang, L.-Y. Characterization and environmental applications of clay–biochar composites. Chem. Eng. J. 2014, 242, 136–143. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Keiluweit, M.; Nico, P.S.; Johnson, M.G.; Kleber, M. Dynamic molecular structure of plant biomass-derived black carbon (biochar). Enriron. Sci. Technol. 2010, 44, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, Q.; Wang, A. Synthesis and characterization of chitosan-g-poly(acrylic acid)/attapulgite superabsorbent composites. Carbohydr. Polym. 2007, 68, 367–374. [Google Scholar] [CrossRef]
- Lammers, K.; Arbuckle-Keil, G.; Dighton, J. FT-IR study of the changes in carbohydrate chemistry of three New Jersey pine barrens leaf litters during simulated control burning. Soil Biol. Biochem. 2009, 41, 340–347. [Google Scholar] [CrossRef]
- Özçimen, D.; Ersoy-Meriçboyu, A. Characterization of biochar and bio-oil samples obtained from carbonization of various biomass materials. Renew. Energy 2010, 35, 1319–1324. [Google Scholar] [CrossRef]
- Xu, R.-K.; Xiao, S.-C.; Yuan, J.-H.; Zhao, A.-Z. Adsorption of methyl violet from aqueous solutions by the biochars derived from crop residues. Bioresour. Technol. 2011, 102, 10293–10298. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Ok, Y.S.; Kim, S.H.; Cho, J.S.; Heo, J.S.; Delaune, R.D.; Seo, D.C. Competitive adsorption of heavy metals onto sesame straw biochar in aqueous solutions. Chemosphere 2016, 142, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Bazrafshan, A.A.; Hajati, S.; Ghaedi, M. Synthesis of regenerable Zn(OH)2 nanoparticle-loaded activated carbon for the ultrasound-assisted removal of malachite green: Optimization, isotherm and kinetics. RSC Adv. 2015, 5, 79119–79128. [Google Scholar] [CrossRef]
- Li, X.; Xie, X.; Luo, H.; Li, L.; Li, Z.; Xue, Z.; Li, W. Adsorption of reactive yellow X-RG and reactive brilliant red X-3B onto cucurbit[8]uril and cucurbit[6]uril: Effect factors, adsorption behavior and mechanism study. J. Colloid Interface Sci. 2017, 498, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Li, G.; Mu, L.; Tao, S. Pyridine-functionalized mesoporous silica as an efficient adsorbent for the removal of acid dyestuffs. J. Mater. Chem. 2006, 16, 1717–1725. [Google Scholar] [CrossRef]
- Cao, C.; Xiao, L.; Chen, C.; Shi, X.; Cao, Q.; Gao, L. In situ preparation of magnetic Fe3O4/chitosan nanoparticles via a novel reduction–precipitation method and their application in adsorption of reactive azo dye. Powder Technol. 2014, 260, 90–97. [Google Scholar] [CrossRef]
- Song, Z.; Chen, L.; Hu, J.; Richards, R. NiO(111) nanosheets as efficient and recyclable adsorbents for dye pollutant removal from wastewater. Nanotechnology 2009, 20, 275707. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Song, Z.; Chen, L.; Yang, H.; Li, J.; Richards, R. Adsorption properties of MgO(111) nanoplates for the dye pollutants from wastewater. J. Chem. Eng. Data 2010, 55, 3742–3748. [Google Scholar] [CrossRef]
- Wu, X.; Wu, D.; Fu, R. Studies on the adsorption of reactive brilliant red X-3B dye on organic and carbon aerogels. J. Hazard. Mater. 2007, 147, 1028–1036. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yang, S.; Chen, H.; He, H.; Sun, C. Adsorption behavior and mechanism of reactive brilliant red X-3B in aqueous solution over three kinds of hydrotalcite-like LDHs. Appl. Surf. Sci. 2014, 301, 329–337. [Google Scholar] [CrossRef]
- Liu, W.-J.; Zeng, F.-X.; Jiang, H.; Zhang, X.-S. Preparation of high adsorption capacity bio-chars from waste biomass. Bioresour. Technol. 2011, 102, 8247–8252. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Liu, Q.; Guo, L.; Zhang, Y.; Lou, Z.; Wang, Y.; Qian, G. Cu(II) removal from aqueous solution by Spartina alterniflora derived biochar. Bioresour. Technol. 2013, 141, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Valdes, H.; sanchez-Polo, M.; Rivera-Utrilla, J.; Zaror, C.A. Effect of ozone treatment on surface properties of activated carbon. Langmuir 2002, 18, 2111–2116. [Google Scholar] [CrossRef]
- Zhu, X.; Tsang, D.C.; Chen, F.; Li, S.; Yang, X. Ciprofloxacin adsorption on graphene and granular activated carbon: Kinetics, isotherms, and effects of solution chemistry. Environ. Technol. 2015, 36, 3094–3102. [Google Scholar] [CrossRef] [PubMed]


| Experimental Item | Biochar Weight (g) | RBR Solution Concentrations (mg/L) | Sampling Time (min) | Temp. (°C) | Vibration Rate (rpm) | SamplingMethod |
|---|---|---|---|---|---|---|
| Adsorption isotherms | 0.1 | 40–200 (40,60,80,100,120,140,160,180,200) | 1440 | 25 | 150 | Syringe membrane |
| Adsorption kinetics | 0.1 | 100 | 5–1440 (5,10,30,60,120,240,720,1440) | 25 | 150 | Syringe membrane |
| Effects of pH | 0.1 | 100 | 1440 | 25 | 150 | Syringe membrane |
| Sample | AB | 1-5-AB | 1-3-AB |
|---|---|---|---|
| Pore characteristics | |||
| BET surface area (m2/g) | 219.49 | 178.51 | 63.91 |
| Total pore volume (m3/g) | 0.1270 | 0.1343 | 0.0644 |
| Average pore width (nm) | 2.31 | 3.01 | 4.03 |
| Elemental analysis (%) | |||
| C | 78.27 | 54.76 | 44.96 |
| H | 0.98 | 0.62 | 0.65 |
| O | 5.13 | 8.53 | 8.92 |
| N | 0.46 | 0.18 | 0.14 |
| K | 1.37 | 2.92 | 3.58 |
| Na | 3.25 | 5.58 | 6.57 |
| Mg | 0.71 | 1.80 | 2.69 |
| Al | 0.66 | 2.58 | 3.64 |
| Fe | 0.75 | 1.77 | 3.04 |
| Ca | 2.05 | 3.68 | 3.89 |
| Si | 6.28 | 16.31 | 20.86 |
| Biochar Samples | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| Qm (mg/g) | KL (L/mg) | R2 | KF (mg/g(mg/L)1/n) | n | R2 | |
| AB | 40.50 | 0.039 | 0.995 | 1.512 | 0.413 | 0.986 |
| 1-5-AB | 65.15 | 0.401 | 0.993 | 1.218 | 0.197 | 0.988 |
| 1-3-AB | 72.20 | 0.581 | 0.992 | 1.182 | 0.167 | 0.988 |
| ATP | 42.59 | 0.022 | 0.991 | 1.629 | 0.488 | 0.981 |
| Adsorbent | Qm (mg/g) | Ref. |
|---|---|---|
| AB | 40.5 | This study |
| 1-5-AB | 65.1 | This study |
| 1-3-AB | 72.2 | This study |
| CB [6] | 158.5 | [45] |
| Py-MS | 891.1 | [46] |
| MFe3O4/CsNPS | 476.8 | [47] |
| NiO nanosheets | 30.4 | [48] |
| MgO nanoplates | 303.0 | [49] |
| Organic aerogels | 90.0 | [50] |
| Carbon aerogels | 450.0 | [50] |
| Ni/Al-LDH | 4.9 | [51] |
| Mg/Al-LDH | 3.8 | [51] |
| Co/Al-LDH | 2.3 | [51] |
| Kinetic Models | AB | 1-5-AB | 1-3-AB | ATP |
|---|---|---|---|---|
| qe,exp (mg/g) | 25.619 | 47.911 | 49.195 | 22.876 |
| Pseudo-first-order | ||||
| qe,cal (mg/g) | 2.222 | 9.956 | 10.916 | 2.207 |
| k1( min−1) | 0.001 | 0.002 | 0.004 | 0.001 |
| r2 | 0.858 | 0.895 | 0.931 | 0.792 |
| Pseudo-second-order | ||||
| qe,cal (mg/g) | 25.63 | 47.94 | 49.33 | 22.74 |
| k2 (g/mg min) | 0.04 | 0.002 | 0.002 | 0.002 |
| r2 | 0.999 | 0.999 | 0.999 | 0.996 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, S.; Zhou, M.; Wang, H.-F.; Wang, T.; Wang, X.-S.; Hou, H.-B.; Song, B.-Y. Adsorption of Reactive Brilliant Red X-3B in Aqueous Solutions on Clay–Biochar Composites from Bagasse and Natural Attapulgite. Water 2018, 10, 703. https://doi.org/10.3390/w10060703
Chen S, Zhou M, Wang H-F, Wang T, Wang X-S, Hou H-B, Song B-Y. Adsorption of Reactive Brilliant Red X-3B in Aqueous Solutions on Clay–Biochar Composites from Bagasse and Natural Attapulgite. Water. 2018; 10(6):703. https://doi.org/10.3390/w10060703
Chicago/Turabian StyleChen, Si, Min Zhou, Han-Fei Wang, Teng Wang, Xiao-Shu Wang, Hao-Bo Hou, and Bi-Yu Song. 2018. "Adsorption of Reactive Brilliant Red X-3B in Aqueous Solutions on Clay–Biochar Composites from Bagasse and Natural Attapulgite" Water 10, no. 6: 703. https://doi.org/10.3390/w10060703
APA StyleChen, S., Zhou, M., Wang, H.-F., Wang, T., Wang, X.-S., Hou, H.-B., & Song, B.-Y. (2018). Adsorption of Reactive Brilliant Red X-3B in Aqueous Solutions on Clay–Biochar Composites from Bagasse and Natural Attapulgite. Water, 10(6), 703. https://doi.org/10.3390/w10060703





