Activated Carbon, Biochar and Charcoal: Linkages and Synergies across Pyrogenic Carbon’s ABCs
Abstract
:1. Introduction
2. Pyrogenic Carbonaceous Materials (PCM): Brief History and Definitions
2.1. Natural Pyrogenic Organic Matter, Black Carbon and Char
2.2. Charcoal
2.3. Activated Carbon
2.4. Biochar
2.5. Other Solid Products Obtained from Pyrolysis
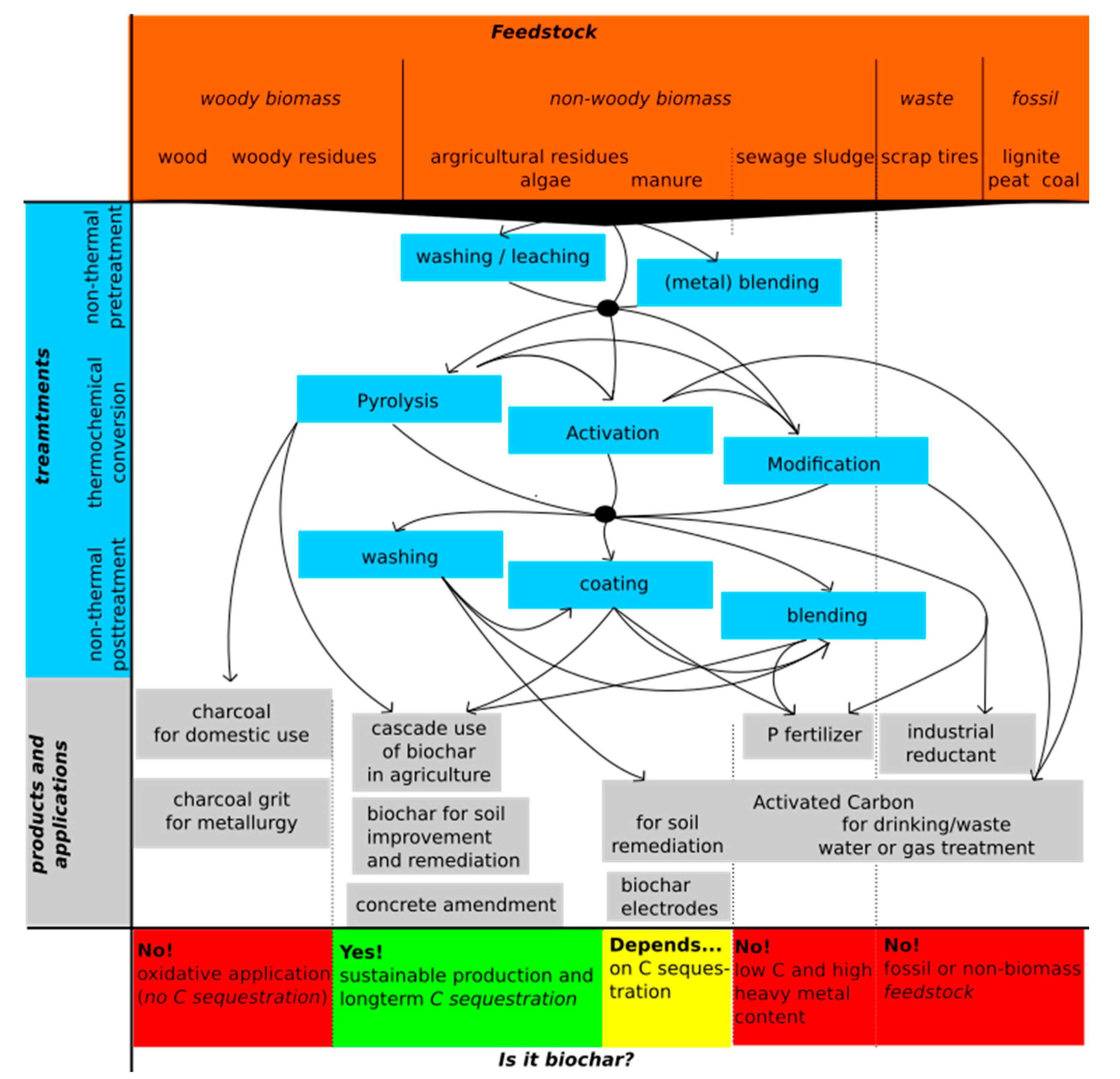
3. Producing Pyrogenic Carbonaceous Materials: Thermal Conversions and Additional Treatments
3.1. Pyrolysis and Gasification
3.2. Chemical Activation
3.3. Physical Activation
3.4. Modification
3.5. Non-Thermal Pre- and Post-Treatments
4. Conclusions and Future Research
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lehmann, J.; Joseph, S. Biochar for environmental management: An introduction In Biochar for Environmental Management: Science, Technology and Implementation; Lehmann, J., Joseph, S., Eds.; Routledge: Oxford, UK, 2015. [Google Scholar]
- European Biochar Certificate (EBC). European Biochar Foundation—European Biochar Certificate—Guidelines for a Sustainable Production of Biochar; Version 6.3E of August 14, 2017; European Biochar Certificate (EBC): Arbaz, Switzerland, 2017; pp. 1–24. [Google Scholar]
- Marsh, H.; Reinoso, F.R. Activated Carbon; Elsevier: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Smisek, M.; Cerny, S. Activated carbon. In Topics in Organic and General Chemistry; Elsevier Co.: New York, NY, USA, 1970. [Google Scholar]
- Hilber, I.; Bucheli, T. Activated carbon amendment to remediate contaminated sediments and soils: A review. Glob. Nest J. 2010, 12, 305–307. [Google Scholar]
- Hale, S.E.; Cornelissen, G.; Werner, D. Sorption and remediation of organic compounds in soils and sediments by (activated) biochar. In Biochar for Environmental Management: Science, Technology and Implementation; Lehmann, J., Joseph, S., Eds.; Routledge: Oxford, UK, 2015. [Google Scholar]
- International Biochar Initiative (IBI). International Biochar Initiative—Standardized Product Definition and Product Testing Guidelines for Biochar That is Used in Soil (aka IBI Biochar Standards) Version 2.0; International Biochar Initiative: Westerville, OH, USA, 2014. [Google Scholar]
- Devi, P.; Saroha, A.K. Utilization of sludge based adsorbents for the removal of various pollutants: A review. Sci. Total Environ. 2017, 578, 16–33. [Google Scholar] [CrossRef] [PubMed]
- Shimabuku, K.K.; Kearns, J.P.; Martinez, J.E.; Mahoney, R.B.; Moreno-Vasquez, L.; Summers, R.S. Biochar sorbents for sulfamethoxazole removal from surface water, stormwater, and wastewater effluent. Water Res. 2016, 96, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Román, S.; Libra, J.; Berge, N.; Sabio, E.; Ro, K.; Li, L.; Ledesma, B.; Álvarez, A.; Bae, S. Hydrothermal carbonization: Modeling, final properties design and applications: A review. Energies 2018, 11, 216. [Google Scholar] [CrossRef]
- Cao, X.; Ro, K.S.; Chappell, M.; Li, Y.; Mao, J. Chemical structures of swine-manure chars produced under different carbonization conditions investigated by advanced solid-state 13C nuclear magnetic resonance (NMR) spectroscopy. Energy Fuels 2011, 25, 388–397. [Google Scholar] [CrossRef]
- Knicker, H. Pyrogenic organic matter in soil: Its origin and occurrence, its chemistry and survival in soil environments. Quat. Int. 2011, 243, 251–263. [Google Scholar] [CrossRef]
- Reisser, M.; Purves, R.S.; Schmidt, M.W.I.; Abiven, S. Pyrogenic carbon in soils: A literature-based inventory and a global estimation of its content in soil organic carbon and stocks. Front. Earth Sci. 2016, 4. [Google Scholar] [CrossRef]
- Sun, T.; Levin, B.D.A.; Guzman, J.J.L.; Enders, A.; Muller, D.A.; Angenent, L.T.; Lehmann, J. Rapid electron transfer by the carbon matrix in natural pyrogenic carbon. Nat. Commun. 2017, 8, 14873. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.; del Campo, B.; Boateng, A.A.; Garcia-Perez, M.; Masek, O. Fundamentals of biochar production. In Biochar for Environmental Management: Science, Technology and Implementation; Lehmann, J., Joseph, S., Eds.; Routledge: Oxford, UK, 2015. [Google Scholar]
- Goldberg, E. Black Carbon in the Environment; John Wiley: New York, NY, USA, 1985. [Google Scholar]
- Gray, E.; Marsh, H.; McLaren, M. A short history of gunpowder and the role of charcoal in its manufacture. J. Mater. Sci. 1982, 17, 3385–3400. [Google Scholar] [CrossRef]
- Dorfer, L.; Moser, M.; Bahr, F.; Spindler, K.; Egarter-Vigl, E.; Giullén, S.; Dohr, G.; Kenner, T. A medical report from the stone age? Lancet 1999, 354, 1023–1025. [Google Scholar] [CrossRef]
- Spindler, K. The Man in the Ice; Hachette UK: London, UK, 2013. [Google Scholar]
- Food and Agriculture Organization of the United Nations (FAO). Faostat—Forestry Production and Trade. 2017. Available online: http://fenixservices.fao.org/faostat/static/bulkdownloads/Forestry_E_All_Data.zip: (accessed on 6 December 2017).
- Food and Agriculture Organization of the United Nations (FAO). Simple Technologies for Charcoal Making. Rome, 1987; Vol. 41. Available online: http://www.fao.org/docrep/x5328e/x5328e00.htm (accessed on 7 December 2017).
- WorldAtlas. st.Laurent, qc Canada 2017. Available online: http://www.Worldatlas.Com/articles/top-wood-charcoal-exporting-and-importing-countries.html (accessed on 6 December 2017).
- United Nations Security Council (UNSC). United Nations Security Council Resolution 2385; UNSC: New York, NY, USA, 2017. [Google Scholar]
- Destatis; Federal Statistical Office of Germany: Wiesbaden, Germany, 28 June 2016.
- Kaltschmitt, M.; Hartmann, H.; Hofbauer, H. Energie Aus Biomasse-Grundlagen, Techniken und Verfahren; Springer: Heidelberg, Germany; Dordrecht, The Netherlands; London, UK; NewYork, NY, USA, 2009. [Google Scholar]
- Food and Agriculture Organization of the United Nations (FAO). Industrial Charcoal Making; FAO: Rome, Italy, 1985; Volume 63, Available online: http://www.fao.org/docrep/x5555e/x5555e00.htm (accessed on 7 December 2017).
- Schanz, J.J.; Parry, R.H. The activated carbon industry. Ind. Eng. Chem. 1962, 54, 24–28. [Google Scholar] [CrossRef]
- Smith, A. On the absorption of gases by charcoal.—No. I. Proc. R. Soc. Lond. 1862, 12, 424–426. [Google Scholar] [CrossRef]
- Dewar, J. The absorption and thermal evolution of gases occluded in charcoal at low temperatures. Proc. R. Soc. Lond. 1904, 74, 122–127. [Google Scholar] [CrossRef]
- Chaney, N. The activation of carbon. Trans. Am. Electron. Chem. Soc. 3_6 1919, 91. [Google Scholar]
- Chaney, N.K. Adsorptive Carbon and Process of Making the Same. U.S. Patent 1497543, 10 June 1924. [Google Scholar]
- Metalbank-Co. Zinc Chloride for Activating Charcoal. British Patent 238,889, 1924. [Google Scholar]
- Bayer-&-Co. Manufacture of Active Charcoal by Briquetting Charcoal Fines and Chemicals under Pressure. British Patents 195,390, 1923. [Google Scholar]
- Sutcliffe, E.R. Absorbent and decolorising carbons. J. Soc. Chem. Ind. 1924, 43, 635–637. [Google Scholar] [CrossRef]
- Lewis, W.K.; Metzner, A.B. Engineering, design and process development section: Activation of carbons. Ind. Eng. Chem. 1954, 46, 849–858. [Google Scholar] [CrossRef]
- Helbig, W.A. Activated carbon. J. Chem. Educ. 1946, 23, 98. [Google Scholar] [CrossRef]
- Wright, W. Oxidations on charcoal. In Mathematical Proceedings of the Cambridge Philosophical Society; Cambridge University Press: Cambridge, UK, 1926; pp. 187–190. [Google Scholar]
- Lowry, H.H. On the nature of active carbon. J. Phys. Chem. 1929, 34, 63–73. [Google Scholar] [CrossRef]
- Ruff, O.; Schmidt, G. Amorpher Kohlenstoff und Graphit. Zeitschrift für Anorganische und Allgemeine Chemie 1925, 148, 313–331. [Google Scholar] [CrossRef]
- Sheldon, H.H. Charcoal activation. Phys. Rev. 1920, 16, 165–172. [Google Scholar] [CrossRef]
- Weiser, H.B.; Sherrick, J. Adsorption by precipitates. I. J. Phys. Chem. 1919, 23, 205–252. [Google Scholar] [CrossRef]
- Mezohegyi, G.; van der Zee, F.P.; Font, J.; Fortuny, A.; Fabregat, A. Towards advanced aqueous dye removal processes: A short review on the versatile role of activated carbon. J. Environ. Manag. 2012, 102, 148–164. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Rahunen, N.; Varcoe, J.R.; Chandra, A.; Avignone-Rossa, C.; Thumser, A.E.; Slade, R.C. Activated carbon cloth as anode for sulfate removal in a microbial fuel cell. Environ. Sci. Technol. 2008, 42, 4971–4976. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Cheng, S.; Pant, D.; Van Bogaert, G.; Logan, B.E. Power generation using an activated carbon and metal mesh cathode in a microbial fuel cell. Electrochem. Commun. 2009, 11, 2177–2179. [Google Scholar] [CrossRef]
- Grand-View-Research-Group. Activated Carbon Market Analysis by Product (Powdered Activated Carbon (pac), Granular Activated Carbon (gac)), by Application (Liquid Phase, Gas Phase), by End-Use (Water Treatment, Food & Beverages, Pharmaceutical & Medical, Automotive, Air Purification) and Segment Forecasts to 2024, 100p, Report ID: 978-1-68038-073-6. Available online: https://www.Grandviewresearch.Com/industry-analysis/activated-carbon-market (accessed on 6 December 2017).
- Glaser, B.; Haumaier, L.; Guggenberger, G.; Zech, W. The ‘terra preta’ phenomenon: A model for sustainable agriculture in the humid tropics. Naturwissenschaften 2001, 88, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Glaser, B.; Balashov, E.; Haumaier, L.; Guggenberger, G.; Zech, W. Black carbon in density fractions of anthropogenic soils of the Brazilian Amazon region. Org. Geochem. 2000, 31, 669–678. [Google Scholar] [CrossRef]
- Glaser, B.; Lehmann, J.; Zech, W. Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal-a review. Biol. Fertil. Soils 2002, 35, 219–230. [Google Scholar] [CrossRef]
- O’Toole, A.; Andersson, D.; Gerlach, A.; Glaser, B.; Kammann, C.; Kern, J.; Kuoppamäki, K.; Mumme, J.; Schmidt, H.-P.; Schulze, M. Current and future applications for biochar. In Biochar in European Soils and Agriculture—Science and Practice; Routledge: Abingdon-on-Thames, UK; Taylor & Francis Group: Abingdon, UK, 2016; pp. 253–280. [Google Scholar]
- Schmidt, H.-P.; Wilson, K. 55 uses of biochar. Ithaka J. 2012, 29, 286–289. [Google Scholar]
- Gerlach, H.; Schmidt, H.-P. Biochar in poultry farming. Ithaka J. 2012, 2012, 262–264. [Google Scholar]
- Joseph, S.; Doug, P.; Dawson, K.; Mitchell, D.R.; Rawal, A.; James, H.; Taherymoosavi, S.; Van Zwieten, L.; Joshua, R.; Donne, S. Feeding biochar to cows: An innovative solution for improving soil fertility and farm productivity. Pedosphere 2015, 25, 666–679. [Google Scholar] [CrossRef]
- Akhtar, A.; Sarmah, A.K. Novel biochar-concrete composites: Manufacturing, characterization and evaluation of the mechanical properties. Sci. Total Environ. 2018, 616–617, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Kua, H.W. Factors determining the potential of biochar as a carbon capturing and sequestering construction material: Critical review. J. Mater. Civ. Eng. 2017, 29, 04017086. [Google Scholar] [CrossRef]
- Huggins, T.; Wang, H.; Kearns, J.; Jenkins, P.; Ren, Z.J. Biochar as a sustainable electrode material for electricity production in microbial fuel cells. Bioresour. Technol. 2014, 157, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Wang, X.; Gu, Z.; Polin, J. Carbon materials from high ash biochar for supercapacitor and improvement of capacitance with HNO3 surface oxidation. J. Power Sources 2013, 236, 285–292. [Google Scholar] [CrossRef]
- Kuzyakov, Y.; Bogomolova, I.; Glaser, B. Biochar stability in soil: Decomposition during eight years and transformation as assessed by compound-specific 14C analysis. Soil Biol. Biochem. 2014, 70, 229–236. [Google Scholar] [CrossRef]
- Lehmann, J.; Abiven, S.; Kleber, M.; Pan, G.; Singh, B.P.; Sohi, S.; Zimmerman, A.R. Persistence of biochar in soil. In Biochar for Environmental Management: Science, Technology and Implementation; Lehmann, J., Joseph, S., Eds.; Routledge: Oxford, UK, 2015. [Google Scholar]
- Smith, P. Soil carbon sequestration and biochar as negative emission technologies. Glob. Chang. Biol. 2016, 22, 1315–1324. [Google Scholar] [CrossRef] [PubMed]
- Woolf, D.; Lehmann, J.; Lee, D.R. Optimal bioenergy power generation for climate change mitigation with or without carbon sequestration. Nat. Commun. 2016, 7, 13160. [Google Scholar] [CrossRef] [PubMed]
- Werner, C.; Kammann, C.I.; Schmidt, H.-P.; Gerten, D.; Lucht, W. Potential of Pyrolysis-ccs as Negative Emission Technology: Chances and Planetary Limits, “4 per 1000 Initiative Day”, Bonn. Available online: https://4per1000day.sciencesconf.org/resource/page?id=6&forward-action=page&forward-controller=resource&lang=es (accessed on 29 December 2017).
- Rockström, J.; Gaffney, O.; Rogelj, J.; Meinshausen, M.; Nakicenovic, N.; Schellnhuber, H.J. A roadmap for rapid decarbonization. Science 2017, 355, 1269–1271. [Google Scholar]
- Korcak, R. Bone char made in Central Park, New York. The Biochar Journal 2015. Available online: https://www.biochar-journal.org/en/ct/70 (accessed on 29 December 2017).
- Iriarte-Velasco, U.; Ayastuy, J.L.; Zudaire, L.; Sierra, I. An insight into the reactions occurring during the chemical activation of bone char. Chem. Eng. J. 2014, 251, 217–227. [Google Scholar] [CrossRef]
- Chen, Y.-N.; Chai, L.-Y.; Shu, Y.-D. Study of arsenic (V) adsorption on bone char from aqueous solution. J. Hazard. Mater. 2008, 160, 168–172. [Google Scholar] [CrossRef] [PubMed]
- Cheung, C.W.; Porter, J.F.; McKay, G. Sorption kinetic analysis for the removal of cadmium ions from effluents using bone char. Water Res. 2001, 35, 605–612. [Google Scholar] [CrossRef]
- Zwetsloot, M.J.; Lehmann, J.; Bauerle, T.; Vanek, S.; Hestrin, R.; Nigussie, A. Phosphorus availability from bone char in a P-fixing soil influenced by root-mycorrhizae-biochar interactions. Plant Soil 2016, 408, 95–105. [Google Scholar] [CrossRef]
- UN-HABITAT. Global Atlas of Excreta, Wastewater Sludge, and Biosolids Management: Moving Forward the Sustainable and Welcome Uses of a Global Resource. Available online: https://esa.Un.Org/iys/docs/san_lib_docs/habitat2008.Pdf (accessed on 28 December 2017).
- Khan, S.; Chao, C.; Waqas, M.; Arp, H.P.H.; Zhu, Y.-G. Sewage sludge biochar influence upon rice (Oryza sativa L.) yield, metal bioaccumulation and greenhouse gas emissions from acidic paddy soil. Environ. Sci. Technol. 2013, 47, 8624–8632. [Google Scholar] [CrossRef] [PubMed]
- Dias, J.M.; Alvim-Ferraz, M.C.M.; Almeida, M.F.; Rivera-Utrilla, J.; Sanchez-Polo, M. Waste materials for activated carbon preparation and its use in aqueous-phase treatment: A review. J. Environ. Manag. 2007, 85, 833–846. [Google Scholar] [CrossRef] [PubMed]
- Rio, S.; Faur-Brasquet, C.; Le Coq, L.; Courcoux, P.; Le Cloirec, P. Experimental design methodology for the preparation of carbonaceous sorbents from sewage sludge by chemical activation—Application to air and water treatments. Chemosphere 2005, 58, 423–437. [Google Scholar] [CrossRef] [PubMed]
- Rio, S.; Le Coq, L.; Faur, C.; Lecomte, D.; Le Cloirec, P. Preparation of adsorbents from sewage sludge by steam activation for industrial emission treatment. Process Saf. Environ. Prot. 2006, 84, 258–264. [Google Scholar] [CrossRef]
- Juma, M.; Koreňová, Z.; Markoš, J.; Annus, J.; Jelemenský, Ľ. Pyrolysis and combustion of scrap tire. Pet. Coal 2006, 48, 15–26. [Google Scholar]
- Jouhara, H.; Czajczyńska, D.; Krzyżyńska, R.; Spencer, N. Use of pyrolytic gas from waste tire as a fuel: A review. Energy 2017, 134, 1121–1131. [Google Scholar]
- Hita, I.; Arabiourrutia, M.; Olazar, M.; Bilbao, J.; Arandes, J.M.; Castaño, P. Opportunities and barriers for producing high quality fuels from the pyrolysis of scrap tires. Renew. Sustain. Energy Rev. 2016, 56, 745–759. [Google Scholar] [CrossRef]
- Rivera-Utrilla, J.; Sánchez-Polo, M.; Gómez-Serrano, V.; Alvarez, P.; Alvim-Ferraz, M.; Dias, J. Activated carbon modifications to enhance its water treatment applications. An overview. J. Hazard. Mater. 2011, 187, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Torri, C.; Adamiano, A.; Fabbri, D.; Lindfors, C.; Monti, A.; Oasmaa, A. Comparative analysis of pyrolysate from herbaceous and woody energy crops by py-gc with atomic emission and mass spectrometric detection. J. Anal. Appl. Pyrolysis 2010, 88, 175–180. [Google Scholar] [CrossRef]
- Boateng, A.A.; Garcia-Perez, M.; Masek, O.; Brown, R.; del Campo, B. Biochar production technology. In Biochar for Environmental Management: Science, Technology and Implementation; Lehmann, J., Joseph, S., Eds.; Routledge: Oxford, UK, 2015. [Google Scholar]
- Novak, J.; Busscher, W. Selection and use of designer biochars to improve characteristics of southeastern USA coastal plain degraded soils. In Advanced Biofuels and Bioproducts; Springer: Berlin, Germany, 2013; pp. 69–96. [Google Scholar]
- Demirbas, A.; Arin, G. An overview of biomass pyrolysis. Energy Sources 2002, 24, 471–482. [Google Scholar] [CrossRef]
- Mohan, D.; Pittman, C.U.; Steele, P.H. Pyrolysis of wood/biomass for bio-oil: A critical review. Energy Fuels 2006, 20, 848–889. [Google Scholar] [CrossRef]
- Brown, R. Biochar production technology. In Biochar for Environmental Management: Science and Technology; Lehmann, J., Joseph, S., Eds.; Routledge: Abingdon-on-Thames, UK, 2009; pp. 127–146. [Google Scholar]
- Sohi, S.; Krull, E.; Lopez-Capel, E.; Bol, R. A review of biochar and its use and function in soil. Adv. Agron. 2010, 105, 47–82. [Google Scholar]
- Bridgwater, A.V. Review of fast pyrolysis of biomass and product upgrading. Biomass Bioenergy 2012, 38, 68–94. [Google Scholar] [CrossRef]
- Spokas, K.A.; Cantrell, K.B.; Novak, J.M.; Archer, D.W.; Ippolito, J.A.; Collins, H.P.; Boateng, A.A.; Lima, I.M.; Lamb, M.C.; McAloon, A.J. Biochar: A synthesis of its agronomic impact beyond carbon sequestration. J. Environ. Qual. 2012, 41, 973–989. [Google Scholar] [CrossRef] [PubMed]
- Budi, E.; Umiatin; Nasbey, H.; Bintoro, R.A.; Wulandari, F.; Erlina. Activated coconut shell charcoal carbon using chemical-physical activation. In AIP Conference Proceedings; AIP Publishing: Melville, NY, USA, 2016; p. 050003. [Google Scholar]
- Martin, M.J.; Balaguer, M.D.; Rigola, M. Feasibility of activated carbon production from biological sludge by chemical activation with ZnCl2 and H2SO4. Environ. Technol. 1996, 17, 667–672. [Google Scholar] [CrossRef]
- Bouguettoucha, A.; Reffas, A.; Chebli, D.; Mekhalif, T.; Amrane, A. Novel activated carbon prepared from an agricultural waste, Stipa tenacissima, based on ZnCl2 activation—Characterization and application to theremoval of methylene blue. Desalin. Water Treat. 2016, 57, 24056–24069. [Google Scholar] [CrossRef]
- Caturla, F.; Molina-Sabio, M.; Rodriguez-Reinoso, F. Preparation of activated carbon by chemical activation with ZnCl2. Carbon 1991, 29, 999–1007. [Google Scholar] [CrossRef]
- Mahapatra, K.; Ramteke, D.S.; Paliwal, L.J. Production of activated carbon from sludge of food processing industry under controlled pyrolysis and its application for methylene blue removal. J. Anal. Appl. Pyrolysis 2012, 95, 79–86. [Google Scholar] [CrossRef]
- Theydan, S.K.; Ahmed, M.J. Adsorption of methylene blue onto biomass-based activated carbon by FeCl3 activation: Equilibrium, kinetics, and thermodynamic studies. J. Anal. Appl. Pyrolysis 2012, 97, 116–122. [Google Scholar] [CrossRef]
- Yang, X.; Xu, G.; Yu, H.; Zhang, Z. Preparation of ferric-activated sludge-based adsorbent from biological sludge for tetracycline removal. Bioresour. Technol. 2016, 211, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Bagreev, A.; Bandosz, T.J. H2S adsorption/oxidation on materials obtained using sulfuric acid activation of sewage sludge-derived fertilizer. J. Colloid Interface Sci. 2002, 252, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Legrouri, K.; Khouya, E.; Ezzine, M.; Hannache, H.; Denoyel, R.; Pallier, R.; Naslain, R. Production of activated carbon from a new precursor molasses by activation with sulphuric acid. J. Hazard. Mater. 2005, 118, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Belaroui, K.; Seghier, A.; Hadjel, M. Synthesis of activated carbon based on apricot stones for wastewater treatment. Desalin. Water Treat. 2014, 52, 1422–1433. [Google Scholar] [CrossRef]
- Ahmed, M.B.; Zhou, J.L.; Ngo, H.H.; Guo, W.; Chen, M. Progress in the preparation and application of modified biochar for improved contaminant removal from water and wastewater. Bioresour. Technol. 2016, 214, 836–851. [Google Scholar] [CrossRef] [PubMed]
- Budinova, T.; Ekinci, E.; Yardim, F.; Grimm, A.; Bjornbom, E.; Minkova, V.; Goranova, M. Characterization and application of activated carbon produced by H3PO4 and water vapor activation. Fuel Proc. Technol. 2006, 87, 899–905. [Google Scholar] [CrossRef]
- Girgis, B.S.; Attia, A.A.; Fathy, N.A. Modification in adsorption characteristics of activated carbon produced by H3PO4 under flowing gases. Colloids Surf. A-Physicochem. Eng. Asp. 2007, 299, 79–87. [Google Scholar] [CrossRef]
- Kan, Y.; Yue, Q.; Kong, J.; Gao, B.; Li, Q. The application of activated carbon produced from waste printed circuit boards (PCBs) by H3PO4 and steam activation for the removal of malachite green. Chem. Eng. J. 2015, 260, 541–549. [Google Scholar] [CrossRef]
- Kuppireddy, S.K.R.; Rashid, K.; Al Shoaibi, A.; Srinivasakannan, C. Production and characterization of porous carbon from date palm seeds by chemical activation with H3PO4: Process optimization for maximizing adsorption of methylene blue. Chem. Eng. Commun. 2014, 201, 1021–1040. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Deliyanni, E.A.; Matis, K.A. Activated carbons produced by pyrolysis of waste potato peels: Cobalt ions removal by adsorption. Colloids Surf. A-Physicochem. Eng. Asp. 2016, 490, 74–83. [Google Scholar] [CrossRef]
- Niandou, M.A.S.; Novak, J.M.; Bansode, R.R.; Yu, J.; Rehrah, D.; Ahmedna, M. Selection of pecan shell-based activated carbons for removal of organic and inorganic impurities from water. J. Environ. Qual. 2013, 42, 902–911. [Google Scholar] [CrossRef] [PubMed]
- Romero-Anaya, A.; Lillo-Rodenas, M.; de Lecea, C.S.-M.; Linares-Solano, A. Hydrothermal and conventional H3PO4 activation of two natural bio-fibers. Carbon 2012, 50, 3158–3169. [Google Scholar] [CrossRef]
- Singh, K.P.; Malik, A.; Sinha, S.; Ojha, P. Liquid-phase adsorption of phenols using activated carbons derived from agricultural waste material. J. Hazard. Mater. 2008, 150, 626–641. [Google Scholar] [CrossRef] [PubMed]
- Toles, C.A.; Marshall, W.E.; Johns, M.M.; Wartelle, L.H.; McAloon, A. Acid-activated carbons from almond shells: Physical, chemical and adsorptive properties and estimated cost of production. Bioresour. Technol. 2000, 71, 87–92. [Google Scholar] [CrossRef]
- Veksha, A.; Bhuiyan, T.I.; Hill, J.M. Activation of aspen wood with carbon dioxide and phosphoric acid for removal of total organic carbon from oil sands produced water: Increasing the yield with bio-oil recycling. Materials 2016, 9, 20. [Google Scholar] [CrossRef] [PubMed]
- Tong, Y.; Mayer, B.K.; McNamara, P.J. Triclosan adsorption using wastewater biosolids-derived biochar. Environ. Sci. Water Res. Technol. 2016, 2, 761–768. [Google Scholar] [CrossRef]
- Ramaraju, B.; Manoj Kumar Reddy, P.; Subrahmanyam, C. Low cost adsorbents from agricultural waste for removal of dyes. Environ. Prog. Sustain. Energy 2014, 33, 38–46. [Google Scholar] [CrossRef]
- Anfruns, A.; Canals-Batlle, C.; Ros, A.; Lillo-Ródenas, M.; Linares-Solano, A.; Fuente, E.; Montes-Morán, M.A.; Martín, M.J. Removal of odour-causing compounds using carbonaceous adsorbents/catalysts prepared from sewage sludge. Water Sci. Technol. 2009, 59, 1371–1376. [Google Scholar] [CrossRef] [PubMed]
- Angeles Lillo-Rodenas, M.; Ros, A.; Fuente, E.; Montes-Moran, M.A.; Martin, M.J.; Linares-Solano, A. Further insights into the activation process of sewage sludge-based precursors by alkaline hydroxides. Chem. Eng. J. 2008, 142, 168–174. [Google Scholar] [CrossRef]
- Cansado, I.P.P.; Mourao, P.A.M.; Falcao, A.I.; Ribeiro Carrott, M.M.L.; Carrott, P.J.M. The influence of the activated carbon post-treatment on the phenolic compounds removal. Fuel Process. Technol. 2012, 103, 64–70. [Google Scholar] [CrossRef]
- Duranoglu, D.; Beker, U. Steam and koh activated carbons from peach stones. Energy Sources Part A-Recovery Util. Environ. Effects 2012, 34, 1004–1015. [Google Scholar] [CrossRef]
- Fuente, E.; Gil, R.; Giron, R.; Lillo-Rodenas, M.; Montes-Moran, M.; Martin, M.; Linares-Solano, A. Evidence for the presence of cyanide during carbon activation by KOH. Carbon 2010, 48, 1032–1037. [Google Scholar] [CrossRef]
- Jung, C.; Park, J.; Lim, K.H.; Park, S.; Heo, J.; Her, N.; Oh, J.; Yun, S.; Yoon, Y. Adsorption of selected endocrine disrupting compounds and pharmaceuticals on activated biochars. J. Hazard. Mater. 2013, 263, 702–710. [Google Scholar] [CrossRef] [PubMed]
- Kyzas, G.Z.; Deliyanni, E.A. Modified activated carbons from potato peels as green environmental-friendly adsorbents for the treatment of pharmaceutical effluents. Chem. Eng. Res. Des. 2015, 97, 135–144. [Google Scholar] [CrossRef]
- Lillo-Ródenas, M.; Juan-Juan, J.; Cazorla-Amorós, D.; Linares-Solano, A. About reactions occurring during chemical activation with hydroxides. Carbon 2004, 42, 1371–1375. [Google Scholar] [CrossRef]
- Linares-Solano, A.; Lozano-Castello, D.; Lillo-Rodenas, M.; Cazorla-Amoros, D. Carbon activation by alkaline hydroxides. In Chemistry & Physics of Carbon: Volume 30; CRC Press: Boca Raton, FL, USA, 2007; pp. 1–62. [Google Scholar]
- Mendoza-Carrasco, R.; Cuerda-Correa, E.M.; Alexandre-Franco, M.F.; Fernandez-Gonzalez, C.; Gomez-Serrano, V. Preparation of high-quality activated carbon from polyethyleneterephthalate (PET) bottle waste. Its use in the removal of pollutants in aqueous solution. J. Environ. Manag. 2016, 181, 522–535. [Google Scholar] [CrossRef] [PubMed]
- Okada, K.; Yamamoto, N.; Kameshima, Y.; Yasumori, A. Porous properties of activated carbons from waste newspaper prepared by chemical and physical activation. J. Colloid Interface Sci. 2003, 262, 179–193. [Google Scholar] [CrossRef]
- Song, M.; Jin, B.; Xiao, R.; Yang, L.; Wu, Y.; Zhong, Z.; Huang, Y. The comparison of two activation techniques to prepare activated carbon from corn cob. Biomass Bioenergy 2013, 48, 250–256. [Google Scholar] [CrossRef]
- Sudaryanto, Y.; Hartono, S.; Irawaty, W.; Hindarso, H.; Ismadji, S. High surface area activated carbon prepared from cassava peel by chemical activation. Bioresour. Technol. 2006, 97, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Liu, S. Activated carbon from extracted sawdust waste with alkaline activation by physical mixing. For. Prod. J. 2010, 60, 276–281. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, N.; Yin, B. Preparation of sludge-based activated carbon and its application in dye wastewater treatment. J. Hazard. Mater. 2008, 153, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.C.; Tseng, R.L.; Juang, R.S. Comparisons of porous and adsorption properties of carbons activated by steam and koh. J. Colloid Interface Sci. 2005, 283, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Mestre, A.S.; Pires, J.; Nogueira, J.M.F.; Carvalho, A.P. Activated carbons for the adsorption of ibuprofen. Carbon 2007, 45, 1979–1988. [Google Scholar] [CrossRef]
- Ucar, S.; Erdem, M.; Tay, T.; Karagoz, S. Removal of lead (II) and nickel (II) ions from aqueous solution using activated carbon prepared from rapeseed oil cake by Na2CO3 activation. Clean Technol. Environ. Policy 2015, 17, 747–756. [Google Scholar] [CrossRef]
- Daud, W.M.A.W.; Houshamnd, A.H. Textural characteristics, surface chemistry and oxidation of activated carbon. J. Nat. Gas Chem. 2010, 19, 267–279. [Google Scholar] [CrossRef]
- Molina-Sabio, M.; Gonzalez, M.; Rodriguez-Reinoso, F.; Sepúlveda-Escribano, A. Effect of steam and carbon dioxide activation in the micropore size distribution of activated carbon. Carbon 1996, 34, 505–509. [Google Scholar] [CrossRef]
- Aworn, A.; Thiravetyan, P.; Nakbanpote, W. Preparation and characteristics of agricultural waste activated carbon by physical activation having micro- and mesopores. J. Anal. Appl. Pyrolysis 2008, 82, 279–285. [Google Scholar] [CrossRef]
- Bacaoui, A.; Yaacoubi, A.; Dahbi, A.; Bennouna, C.; Luu, R.P.T.; Maldonado-Hodar, F.J.; Rivera-Utrilla, J.; Moreno-Castilla, C. Optimization of conditions for the preparation of activated carbons from olive-waste cakes. Carbon 2001, 39, 425–432. [Google Scholar] [CrossRef]
- Bae, W.; Kim, J.; Chung, J. Production of granular activated carbon from food-processing wastes (walnut shells and jujube seeds) and its adsorptive properties. J. Air Waste Manag. Assoc. 2014, 64, 879–886. [Google Scholar] [CrossRef] [PubMed]
- Bouchelta, C.; Medjram, M.S.; Bertrand, O.; Bellat, J.-P. Preparation and characterization of activated carbon from date stones by physical activation with steam. J. Anal. Appl. Pyrolysis 2008, 82, 70–77. [Google Scholar] [CrossRef]
- Chang, C.-F.; Chang, C.-Y.; Tsai, W.-T. Effects of burn-off and activation temperature on preparation of activated carbon from corn cob agrowaste by CO2 and steam. J. Colloid Interface Sci. 2000, 232, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Elsayed, Y.; Khamis, M.; Samara, F.; Alqaydi, M.; Sara, Z.; Al Zubaidi, I.; Mortula, M.M. Novel method for water purification using activated adsorbents developed from sewage sludge. Desalin. Water Treat. 2016, 57, 15649–15659. [Google Scholar] [CrossRef]
- Fu, K.; Yue, Q.; Gao, B.; Sun, Y.; Zhu, L. Preparation, characterization and application of lignin-based activated carbon from black liquor lignin by steam activation. Chem. Eng. J. 2013, 228, 1074–1082. [Google Scholar] [CrossRef]
- Galiatsatou, P.; Metaxas, M.; Arapoglou, D.; Kasselouri-Rigopoulou, V. Treatment of olive mill waste water with activated carbons from agricultural by-products. Waste Manag. 2002, 22, 803–812. [Google Scholar] [CrossRef]
- Guo, M.; Song, W. Converting poultry litter to activated carbon: Optimal carbonization conditions and product sorption for benzene. Environ. Technol. 2011, 32, 1789–1798. [Google Scholar] [CrossRef]
- Ioannidou, O.A.; Zabaniotou, A.A.; Stavropoulos, G.G.; Islam, M.A.; Albanis, T.A. Preparation of activated carbons from agricultural residues for pesticide adsorption. Chemosphere 2010, 80, 1328–1336. [Google Scholar] [CrossRef] [PubMed]
- Johns, M.M.; Marshall, W.E.; Toles, C.A. Agricultural by-products as granular activated carbons for adsorbing dissolved metals and organics. J. Chem. Technol. Biotechnol. 1998, 71, 131–140. [Google Scholar] [CrossRef]
- Johns, M.M.; Marshall, W.E.; Toles, C.A. The effect of activation method on the properties of pecan shell-activated carbons. J. Chem. Technol. Biotechnol. 1999, 74, 1037–1044. [Google Scholar] [CrossRef]
- Li, W.-H.; Yue, Q.-Y.; Gao, B.-Y.; Wang, X.-J.; Qi, Y.-F.; Zhao, Y.-Q.; Li, Y.-J. Preparation of sludge-based activated carbon made from paper mill sewage sludge by steam activation for dye wastewater treatment. Desalination 2011, 278, 179–185. [Google Scholar] [CrossRef]
- Li, W.-H.; Yue, Q.-Y.; Ma, Z.-H.; Gao, B.-Y.; Li, Y.-J.; Zhao, H.-X. Effect of preparation conditions and washing of activated carbon from paper mill sewage sludge on its adsorptive properties. Water Sci. Technol. 2013, 67, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Lima, I.M.; Boykin, D.L.; Klasson, K.T.; Uchimiya, M. Influence of post-treatment strategies on the properties of activated chars from broiler manure. Chemosphere 2014, 95, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Lorenc-Grabowska, E.; Rutkowski, P. High adsorption capacity carbons from biomass and synthetic polymers for the removal of organic compounds from water. Water Air Soil Pollut. 2014, 225, 2082. [Google Scholar] [CrossRef]
- Marquez-Montesinos, F.; Cordero, T.; Rodriguez-Mirasol, J.; Rodriguez, J.J. Powdered activated carbons from pinus caribaea sawdust. Sep. Sci. Technol. 2001, 36, 3191–3206. [Google Scholar] [CrossRef]
- Ntuli, V.; Hapazari, I. Sustainable waste management by production of activated carbon from agroforestry residues. S. Afr. J. Sci. 2013, 109, 1–6. [Google Scholar] [CrossRef]
- Olorundare, O.F.; Msagati, T.A.M.; Krause, R.W.M.; Okonkwo, J.O.; Mamba, B.B. Steam activation, characterisation and adsorption studies of activated carbon from maize tassels. Chem. Ecol. 2014, 30, 473–490. [Google Scholar] [CrossRef]
- Qiu, G.; Guo, M. Quality of poultry litter-derived granular activated carbon. Bioresour. Technol. 2010, 101, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Rajapaksha, A.U.; Vithanage, M.; Lee, S.S.; Seo, D.-C.; Tsang, D.C.W.; Ok, Y.S. Steam activation of biochars facilitates kinetics and pH-resilience of sulfamethazine sorption. J. Soils Sediments 2016, 16, 889–895. [Google Scholar] [CrossRef]
- Rombaldo, C.F.S.; Lisboa, A.C.L.; Mendez, M.O.A.; Coutinho, A.R. Brazilian natural fiber (jute) as raw material for activated carbon production. Anais Da Academia Brasileira De Ciencias 2014, 86, 2137–2144. [Google Scholar] [CrossRef]
- Selvaraju, G.; Abu Bakar, N.K. Production of a new industrially viable green-activated carbon from artocarpus integer fruit processing waste and evaluation of its chemical, morphological and adsorption properties. J. Clean. Prod. 2017, 141, 989–999. [Google Scholar] [CrossRef]
- Şentorun-Shalaby, Ç.D.; Uçak-Astarlıoglu, M.G.; Artok, L.; Sarıcı, Ç. Preparation and characterization of activated carbons by one-step steam pyrolysis/activation from apricot stones. Microporous Mesoporous Mater. 2006, 88, 126–134. [Google Scholar] [CrossRef]
- Toles, C.A.; Marshall, W.E.; Wartelle, L.H.; McAloon, A. Steam- or carbon dioxide-activated carbons from almond shells: Physical, chemical and adsorptive properties and estimated cost of production. Bioresour. Technol. 2000, 75, 197–203. [Google Scholar] [CrossRef]
- Torres-Perez, J.; Soria-Serna, L.A.; Solache-Rios, M.; McKay, G. One step carbonization/activation process for carbonaceous material preparation from pecan shells for tartrazine removal and regeneration after saturation. Adsorpt. Sci. Technol. 2015, 33, 895–913. [Google Scholar] [CrossRef]
- Uchimiya, M.; Wartelle, L.H.; Lima, I.M.; Klasson, K.T. Sorption of deisopropylatrazine on broiler litter biochars. J. Agric. Food Chem. 2010, 58, 12350–12356. [Google Scholar] [CrossRef] [PubMed]
- Vanreppelen, K.; Vanderheyden, S.; Kuppens, T.; Schreurs, S.; Yperman, J.; Carleer, R. Activated carbon from pyrolysis of brewer’s spent grain: Production and adsorption properties. Waste Manag. Res. 2014, 32, 634–645. [Google Scholar] [CrossRef] [PubMed]
- Warhurst, A.M.; McConnachie, G.L.; Pollard, S.J.T. Characterisation and applications of activated carbon produced from moringa oleifera seed husks by single-step steam pyrolysis. Water Res. 1997, 31, 759–766. [Google Scholar] [CrossRef]
- Wilson, K.; Yang, H.; Seo, C.W.; Marshall, W.E. Select metal adsorption by activated carbon made from peanut shells. Bioresour. Technol. 2006, 97, 2266–2270. [Google Scholar] [CrossRef] [PubMed]
- Betancur, M.; Daniel Martinez, J.; Murillo, R. Production of activated carbon by waste tire thermochemical degradation with CO2. J. Hazard. Mater. 2009, 168, 882–887. [Google Scholar] [CrossRef] [PubMed]
- Jaramillo, J.; Gomez-Serrano, V.; Alvarez, P.M. Enhanced adsorption of metal ions onto functionalized granular activated carbons prepared from cherry stones. J. Hazard. Mater. 2009, 161, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Quek, A.; Balasubramanian, R. Preparation and characterization of low energy post-pyrolysis oxygenated tire char. Chem. Eng. J. 2011, 170, 194–201. [Google Scholar] [CrossRef]
- Mendez, A.; Gasco, G.; Freitas, M.M.A.; Siebielec, G.; Stuczynski, T.; Figueiredo, J.L. Preparation of carbon-based adsorbents from pyrolysis and air activation of sewage sludges. Chem. Eng. J. 2005, 108, 169–177. [Google Scholar] [CrossRef]
- Plaza, M.G.; Gonzalez, A.S.; Pis, J.J.; Rubiera, F.; Pevida, C. Production of microporous biochars by single-step oxidation: Effect of activation conditions on CO2 capture. Appl. Energy 2014, 114, 551–562. [Google Scholar] [CrossRef]
- Suliman, W.; Harsh, J.B.; Abu-Lail, N.I.; Fortuna, A.-M.; Dallmeyer, I.; Garcia-Pérez, M. The role of biochar porosity and surface functionality in augmenting hydrologic properties of a sandy soil. Sci. Total Environ. 2017, 574, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Xiao, F.; Pignatello, J.J. Effects of post-pyrolysis air oxidation of biomass chars on adsorption of neutral and ionizable compounds. Environ. Sci. Technol. 2016, 50, 6276–6283. [Google Scholar] [CrossRef] [PubMed]
- Calahorro, C.V.; Garcia, A.M.; Garcia, A.B.; Serrano, V.G. Study of sulfur introduction in activated carbon. Carbon 1990, 28, 321–335. [Google Scholar] [CrossRef]
- Figueiredo, J.L.; Pereira, M.F.R. The role of surface chemistry in catalysis with carbons. Catal. Today 2010, 150, 2–7. [Google Scholar] [CrossRef]
- Yang, G.-X.; Jiang, H. Amino modification of biochar for enhanced adsorption of copper ions from synthetic wastewater. Water Res. 2014, 48, 396–405. [Google Scholar] [CrossRef] [PubMed]
- Ro, K.S.; Cantrell, K.B.; Hunt, P.G. High-temperature pyrolysis of blended animal manures for producing renewable energy and value-added biochar. Ind. Eng. Chem. Res. 2010, 49, 10125–10131. [Google Scholar] [CrossRef]
- Agrafioti, E.; Kalderis, D.; Diamadopoulos, E. Ca and fe modified biochars as adsorbents of arsenic and chromium in aqueous solutions. J. Environ. Manag. 2014, 146, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Frišták, V.; Micháleková-Richveisová, B.; Víglašová, E.; Ďuriška, L.; Galamboš, M.; Moreno-Jimenéz, E.; Pipíška, M.; Soja, G. Sorption separation of Eu and As from single-component systems by Fe-modified biochar: Kinetic and equilibrium study. J. Iran. Chem. Soc. 2017, 14, 521–530. [Google Scholar] [CrossRef]
- Hagemann, N.; Joseph, S.; Schmidt, H.-P.; Kammann, C.I.; Harter, J.; Borch, T.; Young, R.B.; Varga, K.; Taherymoosavi, S.; Elliott, K.W.; et al. Organic coating on biochar explains its nutrient retention and stimulation of soil fertility. Nat. Commun. 2017, 8, 1089. [Google Scholar] [CrossRef] [PubMed]
- Conte, P.; Laudicina, V.A. Mechanisms of organic coating on the surface of a poplar biochar. Curr. Org. Chem. 2017, 21, 1–7. [Google Scholar] [CrossRef]
- Kammann, C.I.; Schmidt, H.P.; Messerschmidt, N.; Linsel, S.; Steffens, D.; Muller, C.; Koyro, H.W.; Conte, P.; Stephen, J. Plant growth improvement mediated by nitrate capture in co-composted biochar. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef]
- Gray, M.R.; Corcoran, W.H.; Gavalas, G.R. Pyrolysis of a wood-derived material. Effects of moisture and ash content. Ind. Eng. Chem. Process Des. Dev. 1985, 24, 646–651. [Google Scholar] [CrossRef]
| Process | Temperature Range [°C] | Heating Rate [°C s−1] | Vapor Residence Time | Primary Product |
|---|---|---|---|---|
| Slow pyrolysis | 550–950 [80] 600 [81] 500 [82] “low-moderate” [83] 400 [84] 350–700 [85] | 0.1–1 [80] “low” [81] 1–100 [85] | 5–30 min [81,82] “long” [83] Hours–days [84] Hours [85] | 35% Char [82,83,84] 15–40% Solid, 20–55% Liquid, 20–60% Gas [85] |
| Fast and intermediate pyrolysis | 850–1250 [80] 650 [81] 500 [82,83,84] 450–550 [85] | 10–200 [80] “very high” [81] >1000 [85] | 0.5–5 s [81] 1 s [82] <2 s [83] <1 min [85] | bio-oil [81] 75% liquid [82,83,84] 50–70% liquid [85] |
| Gasification | >750 [82] >800 [83] 750–900 [84] >800 [85] | Variable [85] | 10–20 s [82] “long” [83] Seconds–minutes [85] | 85% Gas [82,83,84] 90–100% Gas [85] |
| Activation Agent | References |
|---|---|
| ZnCl | [87,88,89,90] |
| FeCl3 | [91,92] |
| H2SO4 | [71,87,93,94,95] |
| H3PO4 | [96,97,98,99,100,101,102,103,104,105,106] |
| HCl | [107] |
| HNO3 | [108] |
| NaOH/KOH | [107,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124] |
| Na2CO3/K2CO3 | [102,119,125,126] |
| Urea | [111] |
| Oxidant | Idealized Reaction [3,4] | Energy Balance [3] | References |
|---|---|---|---|
| Steam/H2O | C + H2O → CO + H2 2C + H2 → 2C(H) 1 | endothermic | [72,95,96,97,112,118,120,124,125,129,130,131,132,133,134,135,136,137,138,139,140,141,142,143,144,145,146,147,148,149,150,151,152,153,154,155,156,157,158] |
| CO2 | C + CO2 → C(O) +CO 1 C(O) → CO sum: C + CO2 → 2CO | endothermic | [106,129,133,139,140,145,153,159] |
| Air/O2 | C + O2 → CO2 | exothermic | [104,105,114,119,139,158,160,161,162,163,164,165] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hagemann, N.; Spokas, K.; Schmidt, H.-P.; Kägi, R.; Böhler, M.A.; Bucheli, T.D. Activated Carbon, Biochar and Charcoal: Linkages and Synergies across Pyrogenic Carbon’s ABCs. Water 2018, 10, 182. https://doi.org/10.3390/w10020182
Hagemann N, Spokas K, Schmidt H-P, Kägi R, Böhler MA, Bucheli TD. Activated Carbon, Biochar and Charcoal: Linkages and Synergies across Pyrogenic Carbon’s ABCs. Water. 2018; 10(2):182. https://doi.org/10.3390/w10020182
Chicago/Turabian StyleHagemann, Nikolas, Kurt Spokas, Hans-Peter Schmidt, Ralf Kägi, Marc Anton Böhler, and Thomas D. Bucheli. 2018. "Activated Carbon, Biochar and Charcoal: Linkages and Synergies across Pyrogenic Carbon’s ABCs" Water 10, no. 2: 182. https://doi.org/10.3390/w10020182
APA StyleHagemann, N., Spokas, K., Schmidt, H.-P., Kägi, R., Böhler, M. A., & Bucheli, T. D. (2018). Activated Carbon, Biochar and Charcoal: Linkages and Synergies across Pyrogenic Carbon’s ABCs. Water, 10(2), 182. https://doi.org/10.3390/w10020182






