The Influence of Karst Aquifer Mineralogy and Geochemistry on Groundwater Characteristics: West Bank, Palestine
Abstract
:1. Introduction
2. Materials and Methods
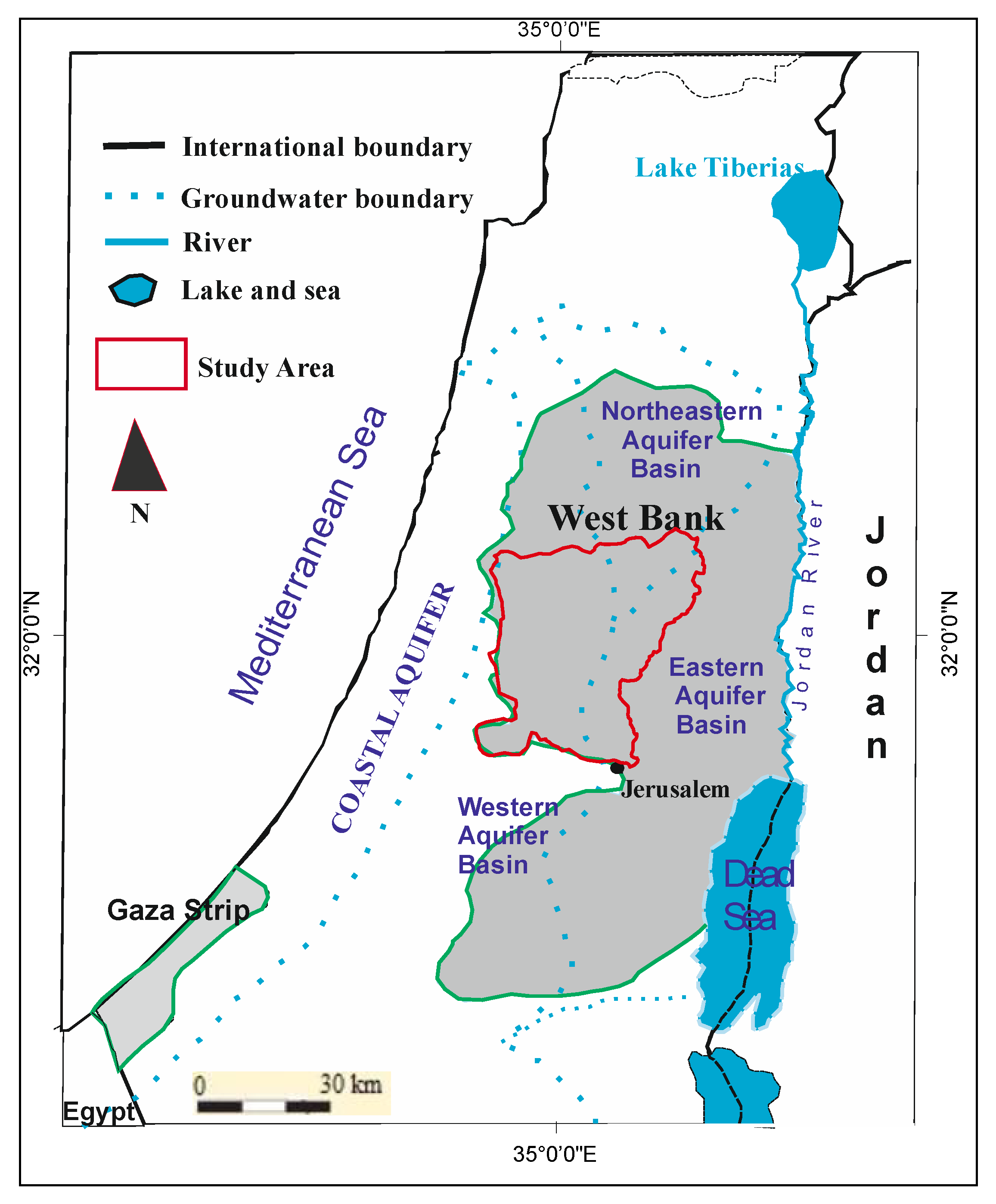
2.1. Study Site Description
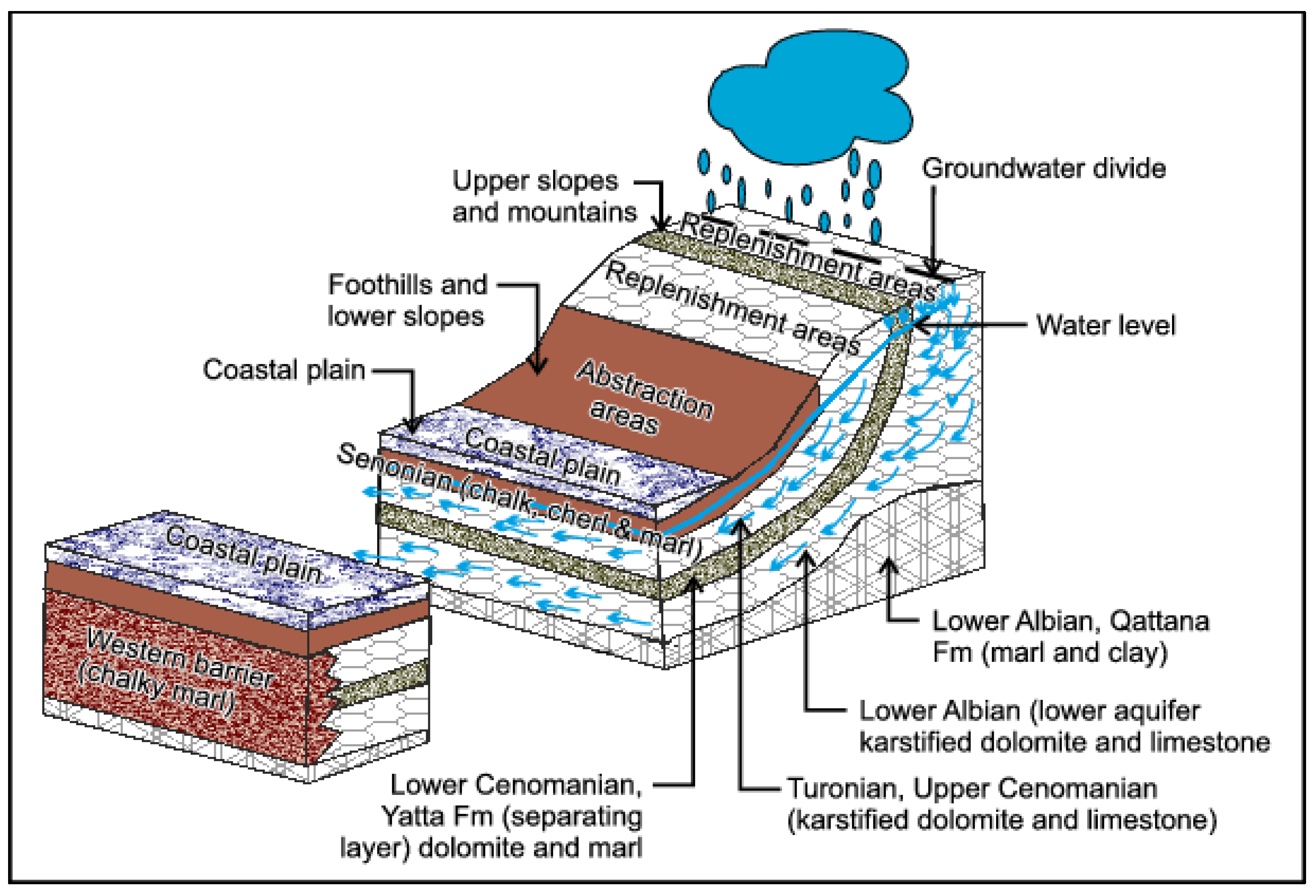
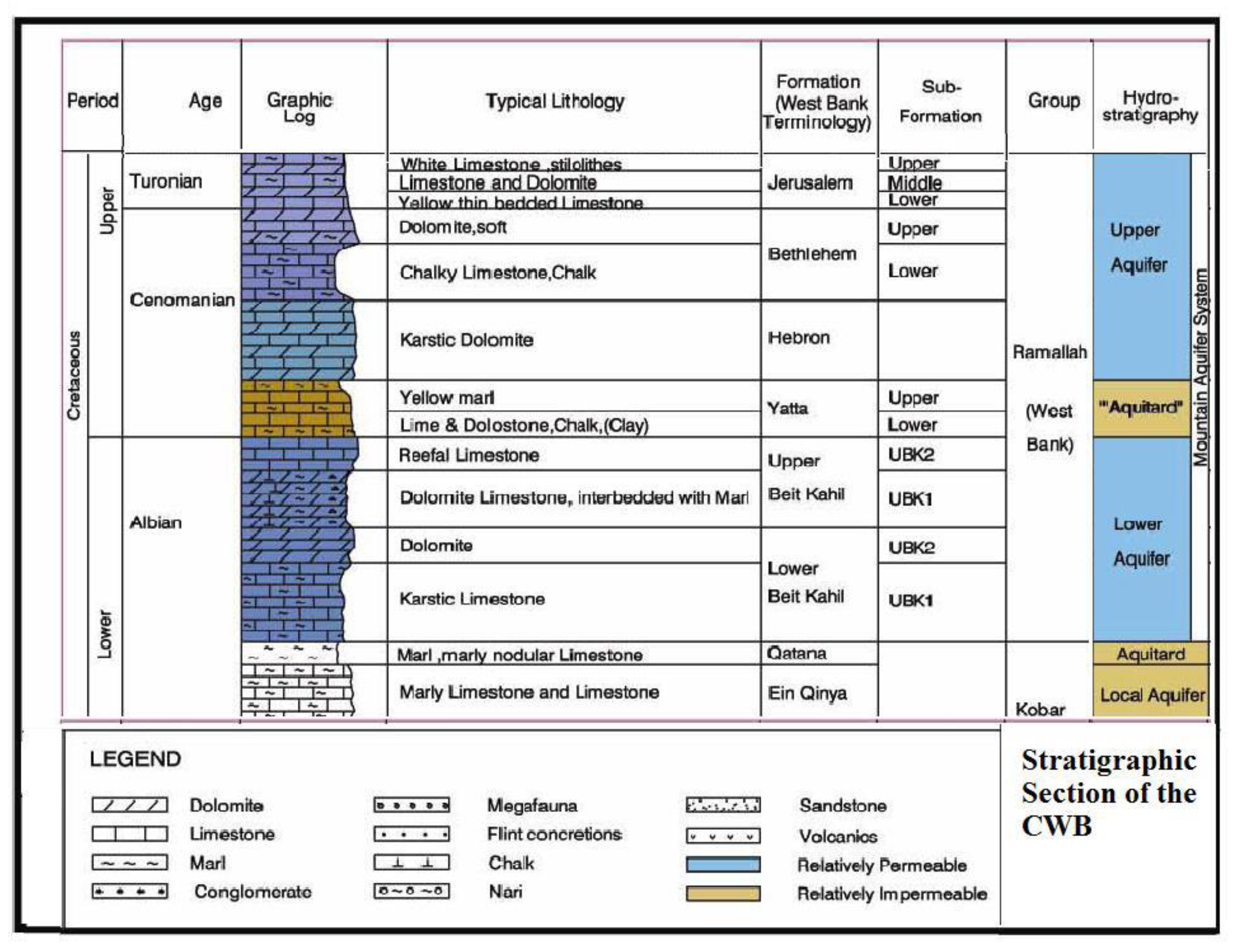
Geological and Hydrogeological Setting
- Lower aquifer: Albian-age Beit Kahil formation with 340-m thickness. This formation is found in many places in the north and the middle of the West Bank.
- Separating layer: Cenomanian-age Yatta formation with an average thickness of about 110 m.
- Upper aquifer: Turonian-age Jerusalem, Bethlehem, and Hebron formations with an average thickness of 150 m.
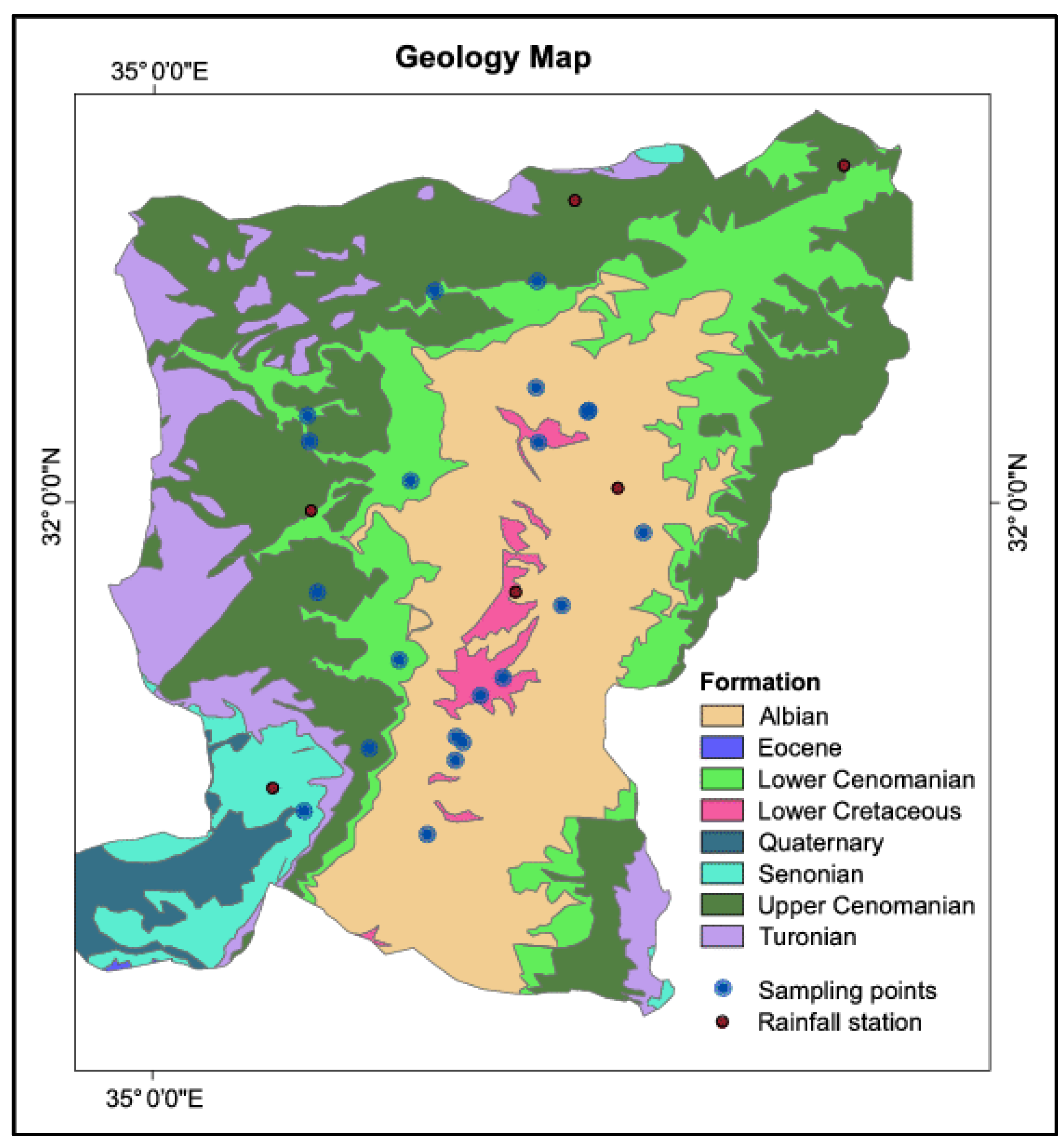
2.2. Sampling and Laboratory Analysis of Sediment
2.3. Hydrochemical Analysis
2.4. Multivariate Statistical Analysis
3. Results and Discussion
3.1. Mineralogical Characterization
3.2. Geochemistry
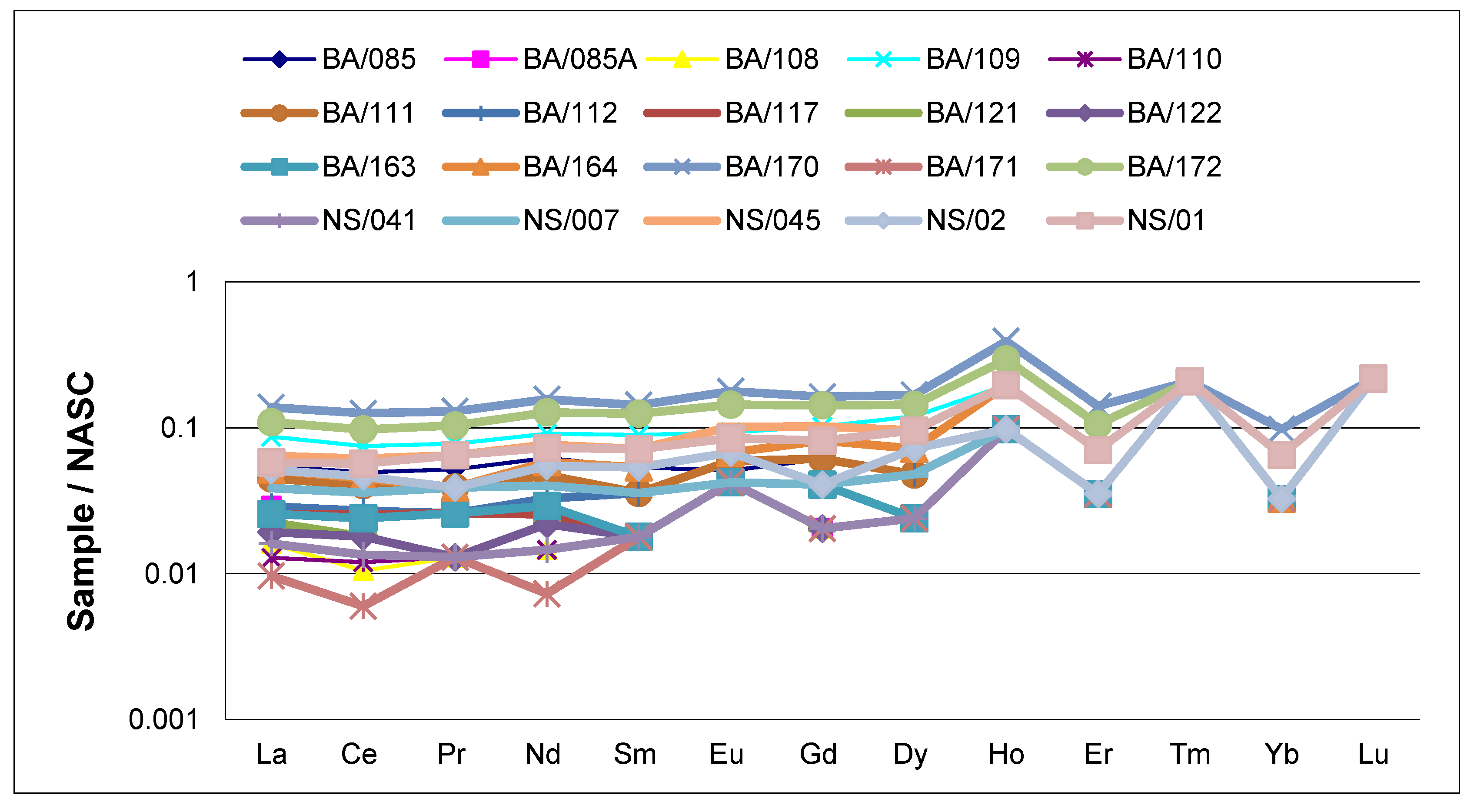
3.3. Rare Earth Elements
3.4. Hydrochemistry
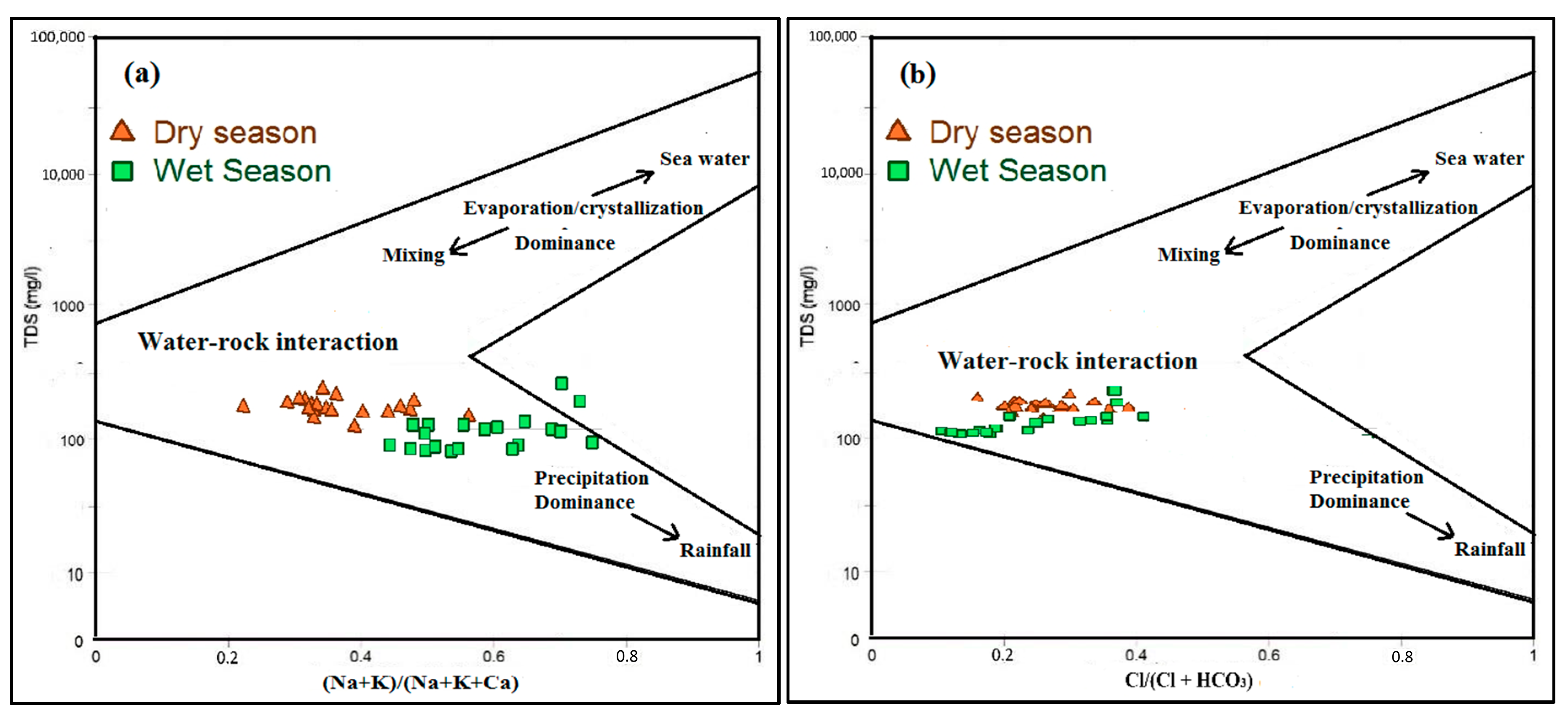
3.4.1. Carbonate Weathering and Dissolution
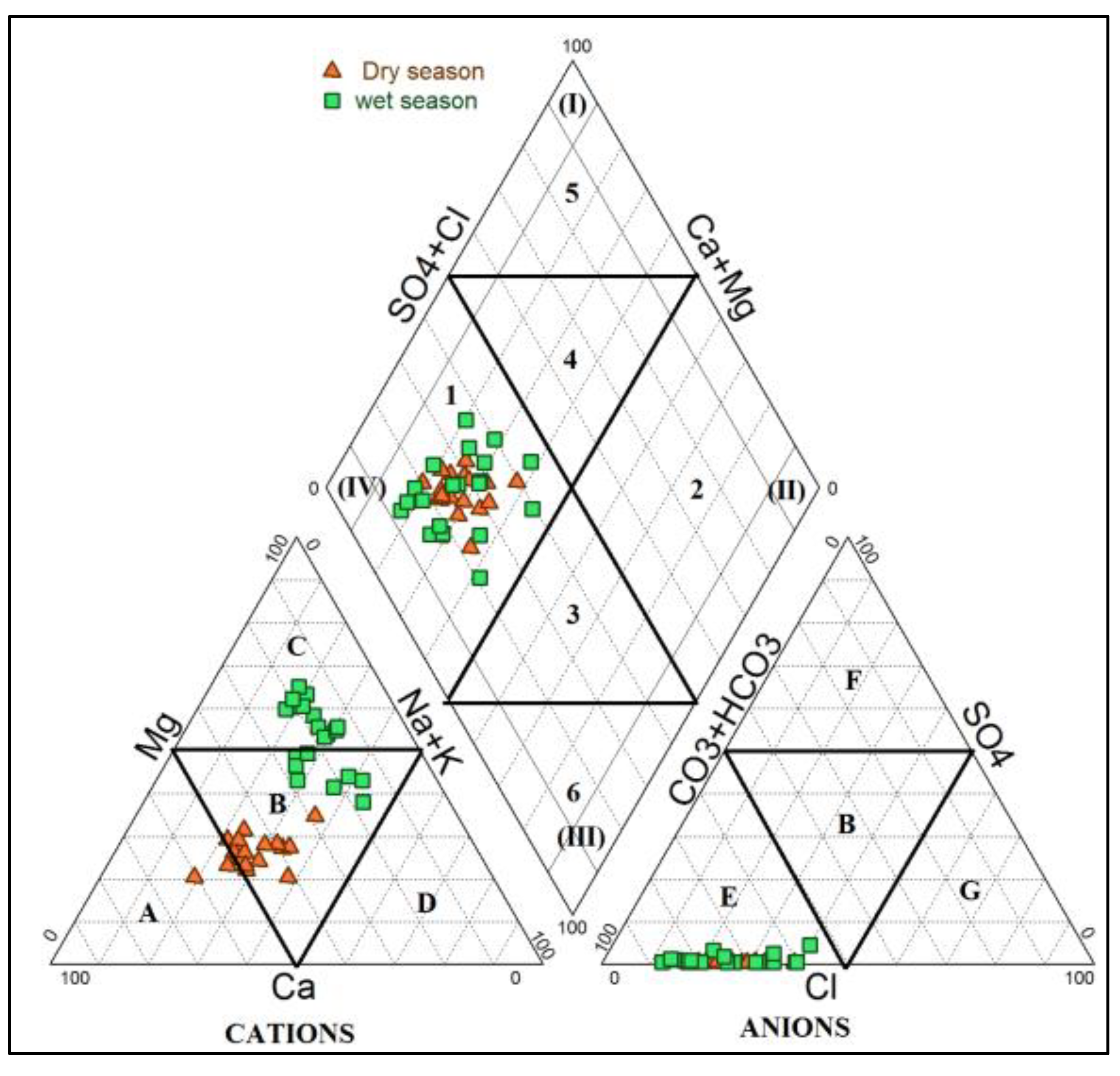
3.4.2. Major Ion Chemistry
3.4.3. Trace Elements in Groundwater
3.5. Statistical Analysis
3.5.1. Correlation Matrix
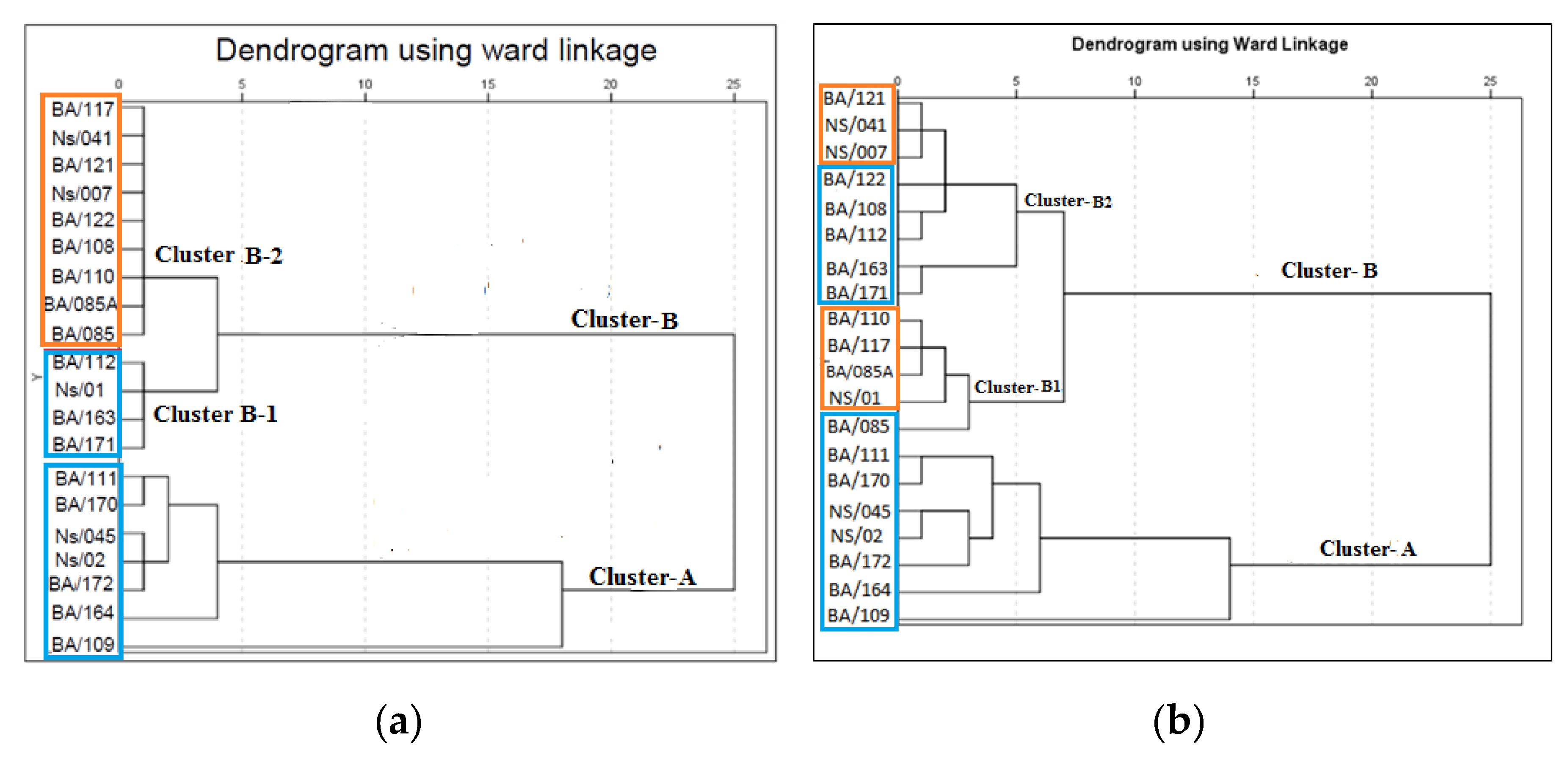
3.5.2. Hierarchical Cluster Analysis (HCA)
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jeannina, P.Y.; Hessenauer, M.; Malard, A.; Chapuis, V. Impact of global change on karst groundwater mineralization in the Jura Mountains. Sci. Total Environ. 2016, 541, 1208–1221. [Google Scholar] [CrossRef] [PubMed]
- Bakalowicz, M. Karst and karst groundwater resources in the Mediterranean. Environ. Earth Sci. 2015, 74, 5–14. [Google Scholar] [CrossRef]
- Galvão, P.; Halihan, T.; Hirata, R. Evaluating karst geotechnical risk in the urbanized area of Sete Lagoas, Minas Gerais, Brazil. Hydrogeol. J. 2015, 23, 1499–1513. [Google Scholar] [CrossRef]
- Jebreen, H.; Wohnlich, S.; Banning, A.; Wisotzky, F.; Niedermayr, A.; Ghanem, M. Recharge, geochemical processes and water quality in karst aquifers: Central West Bank, Palestine. Environ. Earth Sci. 2018, 77, 261. [Google Scholar] [CrossRef]
- Zwahlen, F. Vulnerability and Risk Mapping for the Protection of Carbonate (Karst) Aquifers, Final Report COST Action 620; European Commission: Brussels, Belgium, 2004. [Google Scholar]
- Foster, S.; Hirata, R.; Andreo, B. The aquifer pollution vulnerability concept: Aid or impediment in promoting groundwater protection? Hydrogeol. J. 2013, 21, 1389–1392. [Google Scholar] [CrossRef]
- Siebert, C.; Rosenthal, P.; Möller, P.; Rödiger, T.; Meiler, M. The hydrochemical identification of groundwater flowing to the Bet She’an-Harod multiaquifer system (Lower Jordan Valley) by rare earth elements, yttrium, stable isotopes (H, O) and Tritium. Appl. Geochem. 2012, 27, 703–714. [Google Scholar] [CrossRef]
- Kronfeld, J.; Rosenthal, E. In search of a characteristic signature for groundwater aquifers—A case study from Israel—Comment. J. Hydrol. 1987, 93, 359–372. [Google Scholar] [CrossRef]
- Weinberger, G.; Rosenthal, E.; Ben-Zvi, A.; Zeitoun, D.G. The Yarkon-Taninim groundwater basin, Israel hydrogeology: Case study and critical review. J. Hydrol. 1994, 161, 227–255. [Google Scholar] [CrossRef]
- Rofe and Raffety Consulting Engineers. Nablus District Water Resources Survey; Geological and Hydrological Report; Hashemite Kingdom of Jordan, Central Water Authority, Westminster: London, UK, 1965. [Google Scholar]
- Abed, A.; Wishahi, S. Geology of Palestine: The West Bank and Gaza Strip; Palestinian Hydrology Group: Jerusalem, Israel, 1999. [Google Scholar]
- Jebreen, H.; Wohnlich, S.; Wisotzky, F.; Banning, A.; Niedermayr, A.; Ghanem, M. Recharge estimation in semi-arid karst catchments: Central West Bank, Palestine. Grundwasser J. 2018, 23, 91–101. [Google Scholar] [CrossRef]
- Abusaada, M.; Sauter, M. Studying the flow dynamics of a karst aquifer system with an equivalent porous medium model. Groundwater 2013, 51, 641–650. [Google Scholar] [CrossRef]
- Sheffer, N.A.; Cohen, M.; Morin, E.; Grodek, T.; Gimburg, A.; Magal, E.; Gvirtzman, H.; Nied, M.; Isele, D.; Frumkin, A. Integrated cave drip monitoring for epikarst recharge estimation in a dry Mediterranean area, Sif Cave, Israel. Hydrol. Process. 2011, 25, 2837–2845. [Google Scholar] [CrossRef]
- Dafny, E.; Burg, A.; Gvirtzman, H. Effects of Karst and geological structure on groundwater flow: The case of Yarqon-Taninim Aquifer, Israel. J. Hydrol. 2010, 389, 260–275. [Google Scholar] [CrossRef]
- Weiss, M.; Gvirtzman, H. Estimating ground water recharge using flow models of perched Karstic aquifers. Groundw. J. 2007, 45, 761–773. [Google Scholar] [CrossRef] [PubMed]
- Lange, J.; Greenbaum, N.; Husary, S.; Ghanem, M.; Leibundgut, C.; Schick, A.P. Runoff generation from successive simulated rainfalls on a rocky, semi-arid, Mediterranean hillslope. Hydrol. Process. 2003, 17, 279–296. [Google Scholar] [CrossRef]
- SUSMAQ. Sustainable Management of the West Bank and Gaza Aquifers: Monthly Recharge Estimation in Wadi Natuf, Palestine; Palestinian Water Authority, Palestine and the University of Newcastle upon Tyne: Newcastle, UK, 2004. [Google Scholar]
- Issar, A.S. Recharge and salination processes in the carbonate aquifers in Israel. Environ. Geol. 1993, 21, 152–159. [Google Scholar] [CrossRef]
- Palestinian Water Authority (PWA). Hydrology Department; PWA: Ramallah, Israel, 2016. [Google Scholar]
- Abed Rabbo, A.; Scarpa, D.J.; Qannam, Z.; Abdul Jaber, Q.; Yaunger, P. Springs in the West Bank—Water Quality and Chemistry; Palestinian Hydrology Group and Newcastle University, CMS Ltd.: Palestine, Israel, 1999. [Google Scholar]
- Frumkin, A. Interaction between karst, water and agriculture over the climatic gradient of Israel. Int. J. Speleol. 1999, 26B, 99–110. [Google Scholar] [CrossRef]
- Avisar, D.; Rosenthal, E.; Flexer, A.; Shulman, H.; Ben-Avraham, Z.; Guttman, J. Salinity sources of Kefar Uriya wells in the Judea Group aquifer of Israel. Part 1, conceptual hydrogelogical model. J. Hydrol. 2003, 207, 27–38. [Google Scholar] [CrossRef]
- Gromet, L.P.; Dymek, R.F.; Haskin, L.A.; Korotev, R.L. The “North American shale composite”: Its compilation, major and trace element characteristics. Geochim. Cosmochim. Acta 1984, 48, 2469–2482. [Google Scholar] [CrossRef]
- Malassa, H.; Al-Rimawi, F.; Al-Khatib, M.; Al-Qutob, M. Determination of trace heavy metals in harvested rainwater used for drinking in Hebron (south West Bank, Palestine) by ICP-MS. Environ. Monit. Assess. J. 2014, 186, 6985–6992. [Google Scholar] [CrossRef]
- Clark, E.V.; Daniels, W.L.; Zipper, C.E.; Eriksson, K. Mineralogical influences on water quality from weathering of surface coal mine spoils. Appl. Geochem. 2018, 91, 97–106. [Google Scholar] [CrossRef]
- Taylor, S.R.; McLennan, S.M. The Continental Crust: Its Composition and Evolution; Blackwell Scientific Publication: Carlton, Australia, 1985; 312p. [Google Scholar]
- Yang, X.; Mei, Q.; Wang, X.; Dong, Z.; Li, Y.; Huo, F. Indication of rare earth element characteristics to dolomite petrogenesis—A case study of the fifth member of Ordovician Majiagou Formation in the Ordos Basin, central China. Mar. Pet. Geol. 2018, 92, 1028–1040. [Google Scholar] [CrossRef]
- Worrall, F.; Pearson, D.G. Water–rock interaction in an acidic mine discharge as indicated by rare earth element patterns. Geochim. Cosmochim. Acta 2001, 65, 3027–3040. [Google Scholar] [CrossRef]
- Ford, D.C.; Williams, P.W. Karst Hydrogeology and Geomorphology; Wiley: Chichester, UK, 2007. [Google Scholar]
- Piper, A.M. A graphic procedure in the geochemical interpretation of water analyses. Am. Geophys. Union Trans. 1944, 25, 914–928. [Google Scholar] [CrossRef]
- Nekouei, E.; Zarei, M.; Raeisi, E. The influence of diapir brine on groundwater quality of surrounding aquifers, Larestan, Iran. Environ. Earth Sci. 2016, 75, 571. [Google Scholar] [CrossRef]
- Ma, R.; Wang, Y.; Sun, Z.; Zheng, C.; Ma, T.; Prommer, H. Geochemical evolution of groundwater in carbonate aquifers in Taiyuan, northern China. Appl. Geochem. 2011, 26, 884–897. [Google Scholar] [CrossRef]
- Gascoyne, M. Trace-element partition coefficients in the calcite—Water system and their paleoclimatic significance in cave studies. J. Hydrol. 1983, 61, 213–222. [Google Scholar] [CrossRef]
- Drew, L.; Grunsky, E.; Sutphin, D.; Woodruff, L. Multivariate analysis of the geochemistry and mineralogy of soils along two continental-scale transects in North America. Sci. Total Environ. 2010, 409, 218–227. [Google Scholar] [CrossRef]
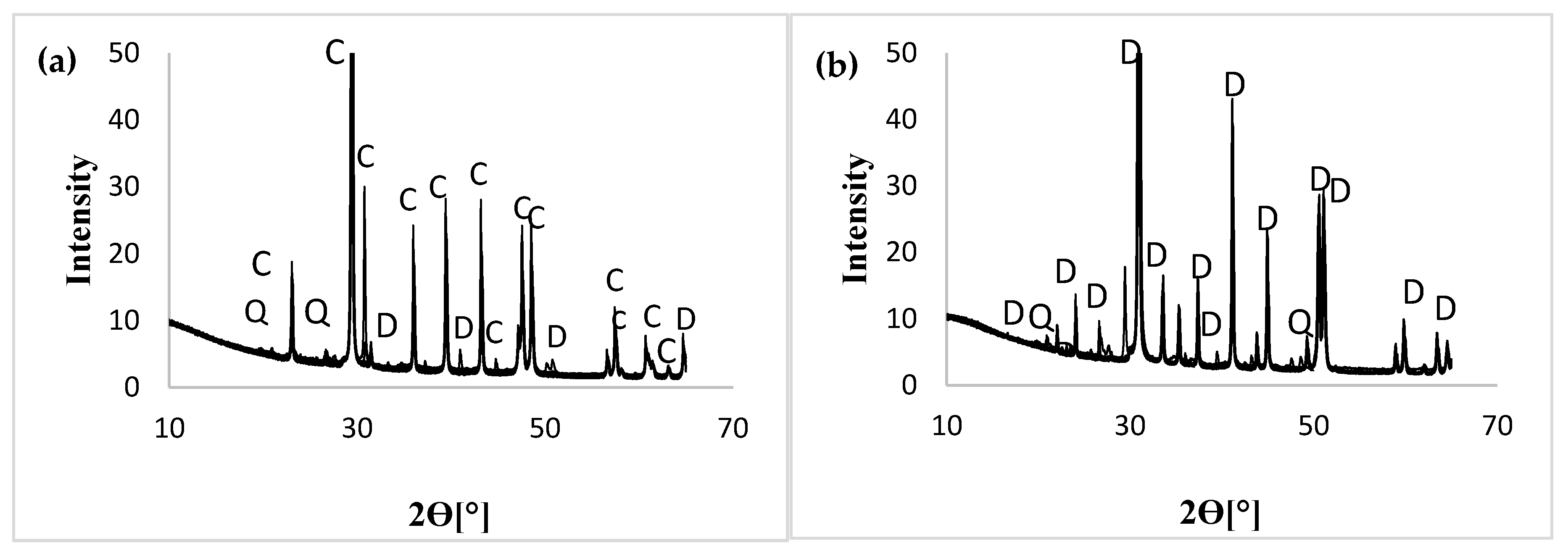
| Sample (ID) | Calcite (C) ≈ wt. % | Dolomite (D) ≈ wt. % | Quartz (Q) ≈ wt. % | Feldspar ≈ (Kf) | Spyrite wt. % | Stotal wt. % | Cinorg wt. % | Ctotal wt. % | Aquifer | Formation | Sub-Formation |
|---|---|---|---|---|---|---|---|---|---|---|---|
| BA/085 | 94 | 4 | 2 | n.d. | <0.01 | 0.04 | 10.8 | 11.4 | Lower Cenomanian | Bethlehem | Lower |
| BA/085A | n.d. | 100 | <1 | n.d. | <0.01 | 0.02 | 12.1 | 12.8 | Lower Cenomanian | Yatta | Lower |
| BA/108 | <1 | 100 | <<1 | n.d. | <0.01 | 0.02 | 12.5 | 12.9 | Al bain | Lower Beit Kahil | UBK2 |
| BA/109 | n.d. | 97 | 1 | 2 | <0.01 | 0.02 | 11.1 | 11.5 | Al bain | Lower Beit Kahil | UBK2 |
| BA/110 | n.d. | 100 | 1 | n.d. | <0.01 | 0.02 | 12.5 | 13.1 | Al bain | Lower Beit Kahil | UBK2 |
| BA/111 | 99 | n.d. | 1 | n.d. | <0.01 | 0.04 | 11.9 | 12.1 | Al bain | Lower Beit Kahil | UBK1 |
| BA/112 | 100 | <1 | n.d. | n.d. | <0.01 | 0.05 | 11.1 | 12.1 | Al bain | Lower Beit Kahil | UBK1 |
| BA/117 | n.d. | 98 | 2 | n.d. | <0.01 | 0.02 | 12.9 | 14.0 | Lower Cenomanian | Yatta | Lower |
| BA/121 | n.d. | 100 | 1 | n.d. | <0.01 | 0.04 | 12.1 | 13.0 | Lower Cenomanian | Yatta | Lower |
| BA/122 | 100 | n.d. | 1 | n.d. | <0.01 | 0.02 | 11.7 | 12.1 | Lower Cenomanian | Bethlehem | Lower |
| BA/163 | 82 | 18 | n.d. | n.d. | <0.01 | 0.04 | 11.8 | 12.3 | Al bain | Lower Beit Kahil | UBK1 |
| BA/164 | 100 | n.d. | 1 | n.d. | <0.01 | 0.04 | 11.7 | 12.1 | Al bain | Lower Beit Kahil | UBK1 |
| BA/170 | 99 | n.d. | 1 | n.d. | <0.01 | 0.06 | 9.70 | 10.5 | Al bain | Lower Beit Kahil | UBK1 |
| BA/171 | 100 | n.d. | n.d. | n.d. | <0.01 | 0.04 | 11.5 | 12.2 | Al bain | Lower Beit Kahil | UBK2 |
| BA/172 | 98 | n.d. | 2 | 1 | <0.01 | 0.08 | 10.9 | 11.4 | Al bain | Ein Qinya | - |
| NS/041 | 5 | 95 | n.d. | n.d. | <0.01 | 0.02 | 12.1 | 13.0 | Lower Cenomanian | Yatta | Lower |
| NS/007 | n.d. | 98 | 2 | n.d. | <0.01 | 0.02 | 10.8 | 10.9 | Lower Cenomanian | Bethlehem | Upper |
| NS/045 | 100 | n.d. | 1 | n.d. | <0.01 | 0.05 | 11.3 | 11.6 | Al bain | Ein Qinya | - |
| Ns/02 | 100 | n.d. | 1 | n.d. | <0.01 | 0.02 | 11.4 | 12.1 | Upper Cenomanian | Bethlehem | Upper |
| Ns/01 | n.d. | 96 | 4 | n.d. | <0.01 | 0.05 | 11.8 | 12.3 | Upper Cenomanian | Bethlehem | Upper |
| Elements (ppm) | Unit | Dolomite Aquifers (n = 9) | Calcite Aquifers (n = 11) | Upper Crust | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Min | Max | Mean | St dev | Min | Max | Mean | St dev | [27] | ||
| Au | ppb | 2.0 | 2.0 | 2.0 | 0 | 2.0 | 2.0 | 2.0 | 0 | 1.8 |
| Ag | ppm | 0.05 | 0.05 | 0.05 | 0 | 0.06 | 0.05 | 0.051 | 0.002 | 0.05 |
| Cu | ppm | 0.90 | 8.10 | 3.80 | 2.4 | 0.8 | 53.9 | 8.78 | 15.1 | 25 |
| Cd | ppm | 0.10 | 0.10 | 0.10 | 0 | 0.1 | 0.40 | 0.18 | 0.11 | 0.05 |
| Mo | ppm | 1.0 | 7.0 | 1.70 | 1.90 | 1 | 1.60 | 1.06 | 0.18 | 1.50 |
| Pb | ppm | 0.50 | 1.20 | 0.70 | 0.20 | 0.50 | 1.90 | 0.82 | 0.42 | 20.0 |
| Ni | ppm | 0.50 | 10.3 | 2.90 | 3.20 | 0.50 | 9.90 | 3.08 | 3.11 | 20.0 |
| Zn | ppm | 2.30 | 7.90 | 4.34 | 2.10 | 1.70 | 28.4 | 9.37 | 9.27 | 71.0 |
| Al | wt. % | 0.06 | 0.76 | 0.20 | 0.22 | 0.03 | 0.9 | 0.35 | 0.25 | 8.04 |
| As | ppm | 0.5 | 5.9 | 1.66 | 1.89 | 0.5 | 3 | 1.77 | 0.93 | 1.5 |
| Ba | ppm | 3 | 32 | 8.55 | 9.05 | 2 | 12 | 6.81 | 3.42 | 550 |
| Be | ppm | 0.1 | 0.1 | 0.1 | 0 | 0.1 | 0.2 | 0.124 | 0.03 | 3.0 |
| Bi | ppm | 0.1 | 0.16 | 0.10 | 0.02 | 0.1 | 0.17 | 0.108 | 0.021 | 0.127 |
| Ca | wt. % | 16.3 | 19.4 | 17.9 | 0.83 | 28.1 | 31.3 | 29.7 | 1.05 | 3.0 |
| Co | ppm | 2.2 | 6 | 3.55 | 1.13 | 1.3 | 7.7 | 3.47 | 2.28 | 16.0 |
| Cr | ppm | 3 | 10 | 5.11 | 2.71 | 4 | 23 | 10.09 | 6.23 | 80.3 |
| Cs | ppm | 0.05 | 0.53 | 0.17 | 0.15 | 0.05 | 0.5 | 0.20 | 0.146 | 3.7 |
| Fe | wt. % | 0.01 | 0.99 | 0.22 | 0.30 | 0.01 | 0.86 | 0.34 | 0.24 | 3.5 |
| Hf | ppm | 0.1 | 0.6 | 0.166 | 0.164 | 0.1 | 0.3 | 0.15 | 0.09 | 5.8 |
| Ga | ppm | 0.2 | 1.3 | 0.53 | 0.38 | 0.2 | 2.5 | 0.92 | 0.68 | 17 |
| Ge | ppm | 0.1 | 0.16 | 0.106 | 0.02 | 0.1 | 0.18 | 0.11 | 0.02 | 1.6 |
| In | ppm | 0.1 | 0.11 | 0.10 | 0.003 | 0.1 | 0.19 | 0.11 | 0.03 | 0.05 |
| Ir | ppb | 5 | 5.1 | 5.01 | 0.03 | 5 | 5.9 | 5.19 | 0.33 | 0. 2 |
| K | wt. % | 0.02 | 0.71 | 0.15 | 0.22 | 0.02 | 0.83 | 0.26 | 0.24 | 2.80 |
| Li | ppm | 1.6 | 14.9 | 4.67 | 3.96 | 0.5 | 7.2 | 2.73 | 2.07 | 20.0 |
| Mg | wt. % | 9.66 | 11.7 | 10.9 | 0.64 | 0.21 | 1.96 | 0.53 | 0.48 | 1.33 |
| Mn | ppm | 31 | 367 | 85 | 106.3 | 17 | 134 | 82.2 | 40.6 | 600 |
| Nb | ppm | 0.1 | 1.7 | 0.4 | 0.50 | 0.2 | 1.9 | 0.85 | 0.59 | 25.0 |
| Na | wt. % | 0.01 | 0.04 | 0.02 | 0.019 | 0.01 | 0.03 | 0.01 | 0.006 | 2.89 |
| P | wt. % | 0.002 | 0.055 | 0.011 | 0.116 | 0.002 | 0.014 | 0.006 | 0.003 | 0.07 |
| Rb | ppm | 0.7 | 9.4 | 2.9 | 2.74 | 0.5 | 12.5 | 4.49 | 3.65 | 112 |
| Sb | ppm | 0.1 | 0.3 | 0.14 | 0.07 | 0.1 | 0.3 | 0.16 | 0.08 | 0.2 |
| Se | ppm | 0.1 | 0.2 | 0.12 | 0.04 | 0.1 | 0.4 | 0.30 | 0.09 | 50.0 |
| Sr | ppm | 53.3 | 112 | 76.1 | 15.7 | 16.9 | 330 | 160.5 | 87.1 | 350 |
| V | ppm | 5 | 21 | 10.1 | 5.39 | 3 | 23 | 12.4 | 6.20 | 60.0 |
| U | ppm | 0.9 | 2.3 | 1.58 | 0.53 | 0.2 | 3.3 | 1.66 | 0.92 | 2.8 |
| W | ppm | 12 | 35 | 18.6 | 8.87 | 5 | 37 | 10.8 | 8.82 | 2.0 |
| Y | ppm | 0.4 | 1.9 | 0.81 | 0.57 | 0.3 | 3.6 | 1.50 | 0.98 | 22 |
| Zr | ppm | 1 | 22 | 4.89 | 6.64 | 3 | 16 | 7.36 | 4.90 | 190 |
| Elements (ppm) | BA /085 | BA /085A | BA /108 | BA /109 | BA /110 | BA /111 | BA /112 | BA /117 | BA /121 | BA /122 | BA /163 | BA /164 | BA /170 | BA /171 | BA /172 | NS /041 | NS /007 | NS /045 | NS /02 | NS /01 | Upper Crust [27] |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| La | 1.70 | 0.90 | 0.50 | 2.70 | 0.40 | 1.40 | 0.90 | 0.80 | 0.70 | 0.60 | 0.80 | 1.60 | 4.30 | 0.30 | 3.40 | 0.50 | 1.20 | 2.0 | 1.60 | 1.80 | 30.0 |
| Ce | 3.30 | 1.60 | 0.70 | 5.0 | 0.80 | 2.70 | 1.80 | 1.70 | 1.20 | 1.20 | 1.60 | 3.0 | 8.40 | 0.40 | 6.50 | 0.90 | 2.40 | 4.10 | 3.10 | 3.80 | 64.0 |
| Pr | 0.40 | 0.20 | 0.10 | 0.60 | 0.10 | 0.30 | 0.20 | 0.20 | 0.10 | 0.10 | 0.20 | 0.30 | 1.0 | 0.10 | 0.80 | 0.10 | 0.30 | 0.50 | 0.30 | 0.50 | 7.10 |
| Nd | 1.70 | 0.70 | 0.40 | 2.50 | 0.40 | 1.30 | 0.90 | 0.70 | 0.60 | 0.60 | 0.80 | 1.60 | 4.30 | 0.20 | 3.50 | 0.40 | 1.10 | 2.10 | 1.50 | 2.0 | 26.0 |
| Sm | 0.30 | 0.10 | 0.10 | 0.50 | 0.10 | 0.20 | 0.20 | 0.10 | 0.10 | 0.10 | 0.10 | 0.30 | 0.80 | 0.10 | 0.70 | 0.10 | 0.20 | 0.40 | 0.30 | 0.40 | 4.50 |
| Eu | 0.06 | 0.05 | 0.05 | 0.11 | 0.05 | 0.07 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 | 0.08 | 0.21 | 0.05 | 0.17 | 0.05 | 0.05 | 0.12 | 0.08 | 0.10 | 0.88 |
| Gd | 0.30 | 0.10 | 0.10 | 0.50 | 0.10 | 0.30 | 0.20 | 0.10 | 0.10 | 0.10 | 0.20 | 0.40 | 0.80 | 0.10 | 0.70 | 0.10 | 0.20 | 0.50 | 0.20 | 0.40 | 3.80 |
| Dy | 0.20 | 0.10 | 0.10 | 0.50 | 0.10 | 0.20 | 0.20 | 0.10 | 0.10 | 0.10 | 0.10 | 0.30 | 0.70 | 0.10 | 0.60 | 0.10 | 0.20 | 0.40 | 0.30 | 0.40 | 3.50 |
| Ho | 0.10 | 0.10 | 0.10 | 0.20 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.20 | 0.40 | 0.10 | 0.30 | 0.10 | 0.10 | 0.20 | 0.10 | 0.20 | 0.80 |
| Er | 0.10 | 0.10 | 0.10 | 0.20 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.20 | 0.40 | 0.10 | 0.30 | 0.10 | 0.10 | 0.20 | 0.10 | 0.20 | 2.30 |
| Tm | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.33 |
| Yb | 0.10 | 0.10 | 0.10 | 0.20 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.30 | 0.10 | 0.20 | 0.10 | 0.10 | 0.20 | 0.10 | 0.20 | 2.20 |
| Lu | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.32 |
| ΣREE | 8.46 | 4.25 | 2.55 | 13.21 | 2.55 | 6.97 | 4.95 | 4.25 | 3.45 | 3.35 | 4.35 | 8.28 | 21.81 | 1.85 | 17.37 | 2.75 | 6.15 | 10.92 | 7.88 | 10.2 | - |
| ΣLREE | 7.76 | 3.65 | 1.95 | 11.91 | 1.95 | 6.27 | 4.25 | 3.65 | 2.85 | 2.75 | 3.75 | 7.28 | 19.81 | 1.25 | 15.77 | 2.15 | 5.45 | 9.72 | 7.08 | 9.0 | - |
| ΣHREE | 0.70 | 0.60 | 0.60 | 1.30 | 0.60 | 0.70 | 0.70 | 0.60 | 0.60 | 0.60 | 0.60 | 1.0 | 2.0 | 0.60 | 1.60 | 0.60 | 0.70 | 1.20 | 0.80 | 1.20 | - |
| LREE/HREE | 11.0 | 6.08 | 3.25 | 9.16 | 3.25 | 8.95 | 6.07 | 6.08 | 4.75 | 4.58 | 6.25 | 7.28 | 9.90 | 2.08 | 9.85 | 3.58 | 7.78 | 8.1 | 8.85 | 7.5 | - |
| ΣREE | 8.46 | 4.25 | 2.55 | 13.21 | 2.55 | 6.97 | 4.95 | 4.25 | 3.45 | 3.35 | 4.35 | 8.28 | 21.81 | 1.85 | 17.37 | 2.75 | 6.15 | 10.92 | 7.88 | 10.2 | - |
| Ce/Ce∗ | 1.04 | 1.08 | 0.84 | 1.03 | 1.07 | 1.07 | 1.07 | 1.21 | 0.99 | 1.07 | 1.07 | 1.0 | 1.04 | 0.87 | 1.01 | 1.08 | 1.12 | 1.07 | 1.07 | 1.07 | - |
| Eu/Eu∗ | 0.60 | 1.52 | 1.52 | 0.67 | 1.52 | 0.87 | 0.76 | 1.52 | 1.52 | 1.52 | 1.07 | 0.70 | 0.80 | 1.52 | 0.74 | 1.52 | 0.76 | 0.81 | 0.99 | 0.76 | - |
| (La/Yb)N | 11.5 | 6.11 | 3.39 | 9.17 | 2.71 | 9.51 | 6.11 | 5.43 | 4.75 | 4.07 | 5.43 | 10.8 | 9.73 | 2.03 | 11.5 | 3.39 | 8.15 | 6.79 | 10.8 | 6.11 | - |
| (La/Gd)N | 4.75 | 7.55 | 4.19 | 4.53 | 3.35 | 3.91 | 3.77 | 6.71 | 5.87 | 5.03 | 3.35 | 3.35 | 4.51 | 2.51 | 4.07 | 4.14 | 5.03 | 3.35 | 6.71 | 3.77 | - |
| Group | Major Composition | Common Impurities | Members |
|---|---|---|---|
| Calcite | CaCO3 | Mn, Fe, Zn, Co, Sr, Pb, Mg, Cu, Al, Ni, V, Cr, Mo | Gaspeite [Ni,Mg,Fe)CO3], magnesite [MgCO3], otavite [CdCO3], rhodochrosite [MnCO3], siderite [FeCO3], smithsonite [ZnCO3], spherocobaltite [CoCO3] |
| Dolomite | CaMg(CO3)2 | Fe, Mn, Co, Pb, Zn | Ankerite [Ca(Fe,Mg)(CO3)2], kutnohorite [Ca(Mn,Mg,Fe)(CO3)2], minrecordite [CaZn(CO3)2], norsethite [BaMg(CO3)2] |
| Spring ID | Li | B | Al | Ba | TI | Pb | Bi | Cr | Mn | Fe | Co | Ni | Cu | Zn | Sr | Mo | Ag | Cd |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BA/085 | 1.37 | 60.8 | 6.17 | 36.3 | 0.032 | 0.21 | 0.08 | 0.51 | 0.23 | 176.1 | 0.13 | 2.26 | 1.3 | 18.28 | 124.4 | 0.20 | 0.04 | 0.008 |
| BA/085A | 1.48 | 30.4 | 0.69 | 32.5 | 0.032 | 0.1 | 0.08 | 0.41 | 0.04 | 179.9 | 0.07 | 0.95 | 1.98 | 4.50 | 166.7 | 0.20 | 0.04 | 0.007 |
| BA/108 | 0.76 | 26.3 | 5.51 | 13.3 | 0.032 | 0.31 | 0.08 | 0.35 | 0.2 | 97.2 | 0.06 | 0.99 | 0.71 | 3.30 | 84.40 | 0.20 | 0.04 | 0.01 |
| BA/109 | 1.25 | 32.6 | 2.48 | 19.1 | 0.01 | 0.23 | 0.08 | 2.41 | 0.66 | 129.5 | 0.17 | 3.9 | 1.32 | 12.2 | 94.21 | 0.20 | 0.04 | 0.01 |
| BA/110 | 1.41 | 30.8 | 2.06 | 29.07 | 0.03 | 0.08 | 0.08 | 0.56 | 0.14 | 106.8 | 0.09 | 2.07 | 0.36 | 2.11 | 100.7 | 0.20 | 0.04 | 0.008 |
| BA/111 | 1.55 | 43.3 | 1.59 | 39.6 | 0.04 | 0.09 | 0.08 | 0.98 | 0.14 | 222.7 | 0.27 | 2.8 | 0.83 | 3.28 | 167.1 | 0.20 | 0.04 | 0.013 |
| BA/112 | 0.91 | 27.2 | 0.69 | 13.4 | 0.03 | 0.14 | 0.08 | 0.55 | 0.22 | 105.1 | 0.06 | 5.82 | 0.54 | 4.33 | 85.44 | 0.20 | 0.04 | 0.01 |
| BA/117 | 0.43 | 23.6 | 22.3 | 15.07 | 0.04 | 0.11 | 0.08 | 0.58 | 0.43 | 144.4 | 0.09 | 1.89 | 0.51 | 2.31 | 83.44 | 0.20 | 0.04 | 0.01 |
| BA/121 | 0.93 | 28.5 | 11.1 | 20.07 | 0.14 | 0.46 | 0.08 | 1.78 | 0.64 | 140.1 | 0.11 | 2.42 | 0.82 | 3.50 | 131.6 | 0.20 | 0.04 | 0.03 |
| BA/122 | 0.32 | 12.2 | 6.91 | 9.68 | 0.05 | 0.07 | 0.08 | 0.18 | 0.06 | 60.6 | 0.04 | 0.09 | 0.11 | 0.02 | 65.60 | 0.20 | 0.04 | 0.01 |
| BA/163 | 0.27 | 10.5 | 6.52 | 6.81 | 0.04 | 0.18 | 0.08 | 0.19 | 0.13 | 70.0 | 0.03 | 1.28 | 0.11 | 0.58 | 47.40 | 0.20 | 0.04 | 0.01 |
| BA/164 | 1.61 | 26.5 | 17.3 | 14.8 | 0.03 | 0.51 | 0.08 | 1.33 | 26.7 | 104.6 | 0.28 | 1.48 | 0.45 | 3.19 | 124.0 | 1.01 | 0.05 | 0.03 |
| BA/170 | 0.72 | 14.5 | 6.46 | 7.75 | 0.03 | 0.51 | 0.08 | 1.33 | 0.23 | 60.9 | 0.05 | 0.65 | 0.35 | 2.32 | 49.80 | 0.66 | 0.10 | 0.03 |
| BA/171 | 1.18 | 34.8 | 22.07 | 14.5 | 0.03 | 0.51 | 0.08 | 1.44 | 19.6 | 89.7 | 0.18 | 6.07 | 0.66 | 3.07 | 63.90 | 1.13 | 0.16 | 0.03 |
| BA/172 | 0.75 | 17.2 | 3.34 | 7.82 | 0.03 | 0.51 | 0.08 | 1.29 | 0.21 | 72.02 | 0.03 | 0.81 | 0.34 | 2.88 | 57.10 | 0.45 | 0.12 | 0.03 |
| NS/041 | 0.81 | 19.7 | 17.04 | 12.2 | 0.03 | 0.51 | 0.08 | 1.24 | 0.25 | 63.2 | 0.06 | 0.9 | 0.34 | 3.006 | 59.70 | 0.34 | 0.13 | 0.03 |
| NS/007 | 1.09 | 20.1 | 110.8 | 15.04 | 0.03 | 1.07 | 0.08 | 2.97 | 3.29 | 109.7 | 0.19 | 1.37 | 0.53 | 5.48 | 80.40 | 0.54 | 0.46 | 0.12 |
| NS/045 | 0.71 | 13.6 | 75.4 | 8.83 | 0.03 | 0.74 | 0.08 | 2.78 | 0.50 | 78.8 | 0.12 | 1.16 | 0.38 | 5.27 | 54.90 | 0.78 | 0.57 | 0.10 |
| Ns/02 | 0.87 | 25.0 | 22.9 | 24.8 | 0.03 | 0.51 | 0.08 | 1.67 | 0.30 | 112.7 | 0.11 | 1.21 | 0.8 | 3.71 | 101.8 | 0.42 | 0.45 | 0.03 |
| Ns/01 | 1.03 | 31.5 | 11.1 | 18.4 | 0.04 | 0.51 | 0.08 | 1.44 | 0.28 | 98.4 | 0.16 | 1.82 | 0.98 | 3.38 | 81.17 | 0.37 | 0.54 | 0.03 |
| MIN | 0.27 | 10.5 | 0.69 | 6.81 | 0.01 | 0.07 | 0.08 | 0.18 | 0:04 | 60.6 | 0.03 | 0.09 | 0.11 | 0.02 | 47.39 | 0.2 | 0.04 | 0.007 |
| MAX | 1.61 | 60.8 | 110.8 | 39.6 | 0.14 | 1.07 | 0.08 | 2.97 | 26.7 | 222.7 | 0.28 | 6.07 | 1.98 | 18.28 | 167.13 | 1.13 | 0.57 | 0.12 |
| AV | 0.97 | 26.4 | 17.6 | 17.9 | 0.03 | 0.36 | 0.08 | 1.19 | 2.7 | 111 | 0.11 | 1.99 | 0.67 | 4.33 | 91.2 | 0.39 | 0.15 | 0.02 |
| Mg | Ca | Li | B | Al | Ba | TI | Pb | Cr | Mn | Fe | Co | Cu | Zn | Sr | Mo | Ag | Cd | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mg | 1 | |||||||||||||||||
| Ca | 0.80 | 1 | ||||||||||||||||
| Li | 0.35 | 0.51 | 1 | |||||||||||||||
| B | 0.63 | 0.71 | 0.70 | 1 | ||||||||||||||
| Al | −0.40 | −0.35 | −0.07 | −0.27 | 1 | |||||||||||||
| Ba | 0.68 | 0.86 | 0.72 | 0.82 | −0.23 | 1 | ||||||||||||
| TI | −0.05 | 0.17 | −0.14 | −0.01 | −0.07 | 0.03 | 1 | |||||||||||
| Pb | −0.56 | −0.59 | −0.01 | −0.28 | 0.79 | −0.38 | 0.01 | 1 | ||||||||||
| Cr | −0.26 | −0.32 | 0.16 | −0.13 | 0.71 | −0.16 | −0.01 | 0.80 | 1 | |||||||||
| Mn | −0.34 | −0.22 | 0.39 | 0.09 | 0.10 | −0.12 | −0.10 | 0.25 | 0.13 | 1 | ||||||||
| Fe | 0.75 | 0.95 | 0.61 | 0.75 | −0.15 | 0.88 | 0.14 | −0.36 | −0.09 | −0.09 | 1 | |||||||
| Co | 0.22 | 0.28 | 0.70 | 0.47 | 0.26 | 0.42 | −0.06 | 0.24 | 0.43 | 0.59 | 0.48 | 1 | ||||||
| Cu | 0.44 | 0.66 | 0.59 | 0.64 | −0.20 | 0.67 | −0.02 | −0.20 | 0.02 | −0.10 | 0.70 | 0.23 | 1 | |||||
| Zn | 0.31 | 0.37 | 0.42 | 0.70 | 0.03 | 0.45 | −0.19 | 0.0 | 0.20 | −0.08 | 0.43 | 0.24 | 0.57 | 1 | ||||
| Sr | 0.57 | 0.87 | 0.73 | 0.65 | −0.24 | 0.86 | 0.24 | −0.34 | −0.14 | 0.06 | 0.89 | 0.49 | 0.69 | 0.28 | 1 | |||
| Mo | −0.55 | −0.53 | 0.18 | −0.15 | 0.40 | −0.34 | −0.20 | 0.61 | 0.45 | 0.79 | −0.35 | 0.42 | −0.24 | −0.13 | −0.27 | 1 | ||
| Ag | −0.24 | −0.41 | −0.08 | −0.21 | 0.69 | −0.15 | −0.13 | 0.72 | 0.66 | −0.06 | −0.23 | 0.18 | −0.05 | −0.02 | −0.27 | 0.35 | 1 | |
| Cd | −0.47 | −0.44 | −0.04 | −0.32 | 0.95 | −0.29 | −0.02 | 0.89 | 0.80 | 0.11 | −0.24 | 0.25 | −0.22 | −0.01 | −0.28 | 0.48 | 0.75 | 1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jebreen, H.; Banning, A.; Wohnlich, S.; Niedermayr, A.; Ghanem, M.; Wisotzky, F. The Influence of Karst Aquifer Mineralogy and Geochemistry on Groundwater Characteristics: West Bank, Palestine. Water 2018, 10, 1829. https://doi.org/10.3390/w10121829
Jebreen H, Banning A, Wohnlich S, Niedermayr A, Ghanem M, Wisotzky F. The Influence of Karst Aquifer Mineralogy and Geochemistry on Groundwater Characteristics: West Bank, Palestine. Water. 2018; 10(12):1829. https://doi.org/10.3390/w10121829
Chicago/Turabian StyleJebreen, Hassan, Andre Banning, Stefan Wohnlich, Andrea Niedermayr, Marwan Ghanem, and Frank Wisotzky. 2018. "The Influence of Karst Aquifer Mineralogy and Geochemistry on Groundwater Characteristics: West Bank, Palestine" Water 10, no. 12: 1829. https://doi.org/10.3390/w10121829
APA StyleJebreen, H., Banning, A., Wohnlich, S., Niedermayr, A., Ghanem, M., & Wisotzky, F. (2018). The Influence of Karst Aquifer Mineralogy and Geochemistry on Groundwater Characteristics: West Bank, Palestine. Water, 10(12), 1829. https://doi.org/10.3390/w10121829






