Lithologic Control of the Hydrochemistry of a Point-Bar Alluvial Aquifer at the Low Reach of the Nakdong River, South Korea: Implications for the Evaluation of Riverbank Filtration Potential
Abstract
1. Introduction
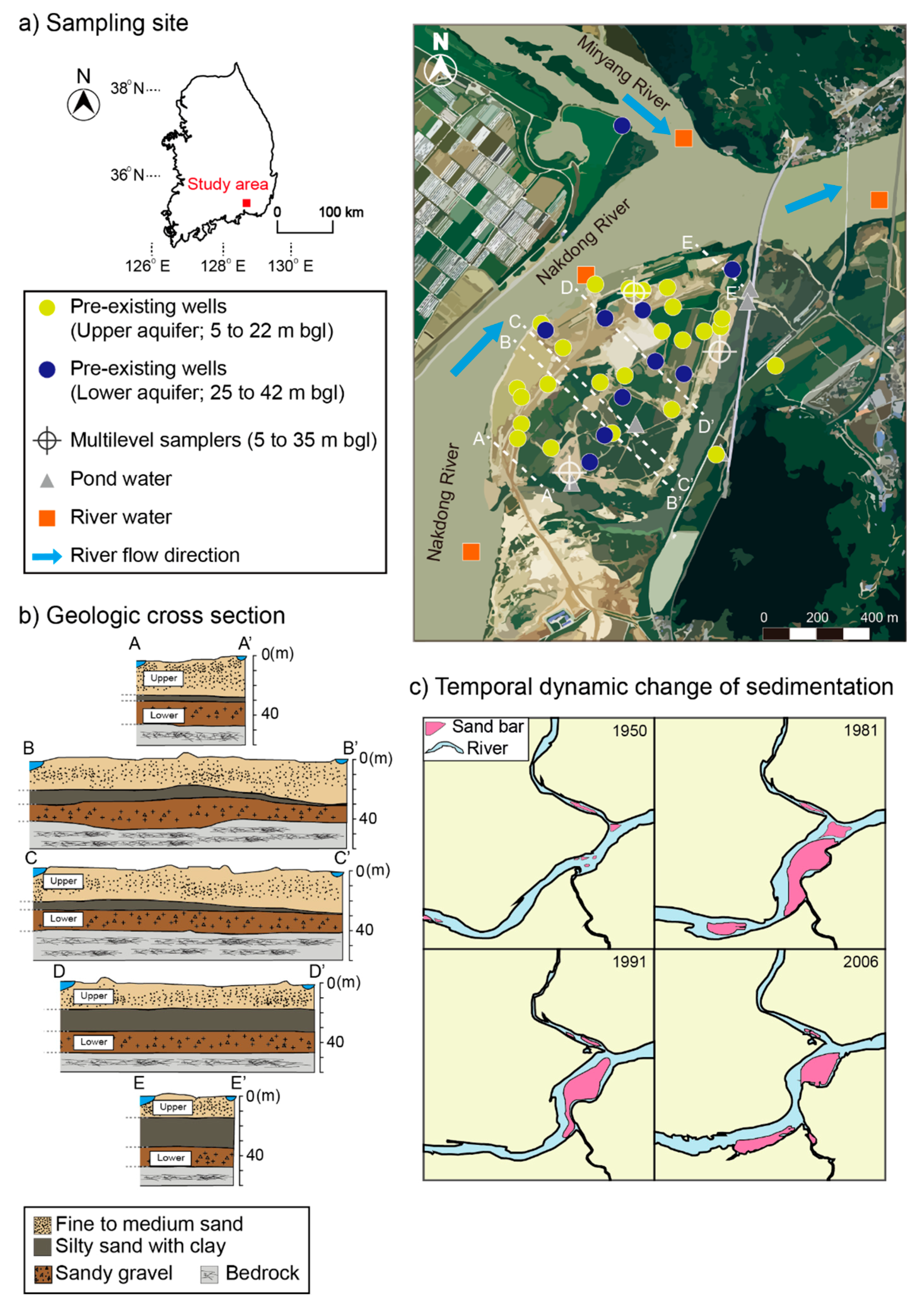
2. Study Area
3. Materials and Methods
4. Results and Discussion
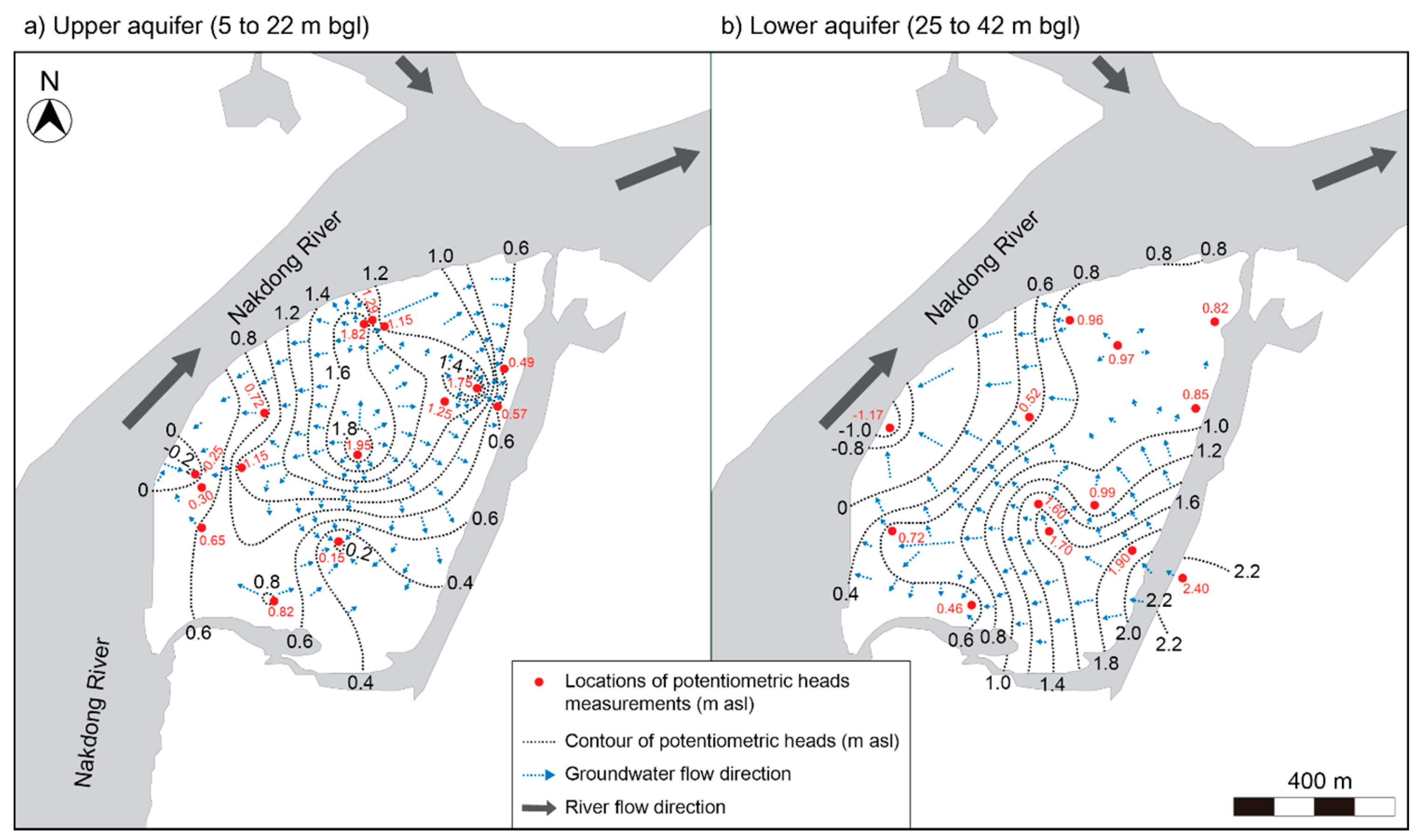
4.1. Subsurface Geology and Hydrogeologic Condition
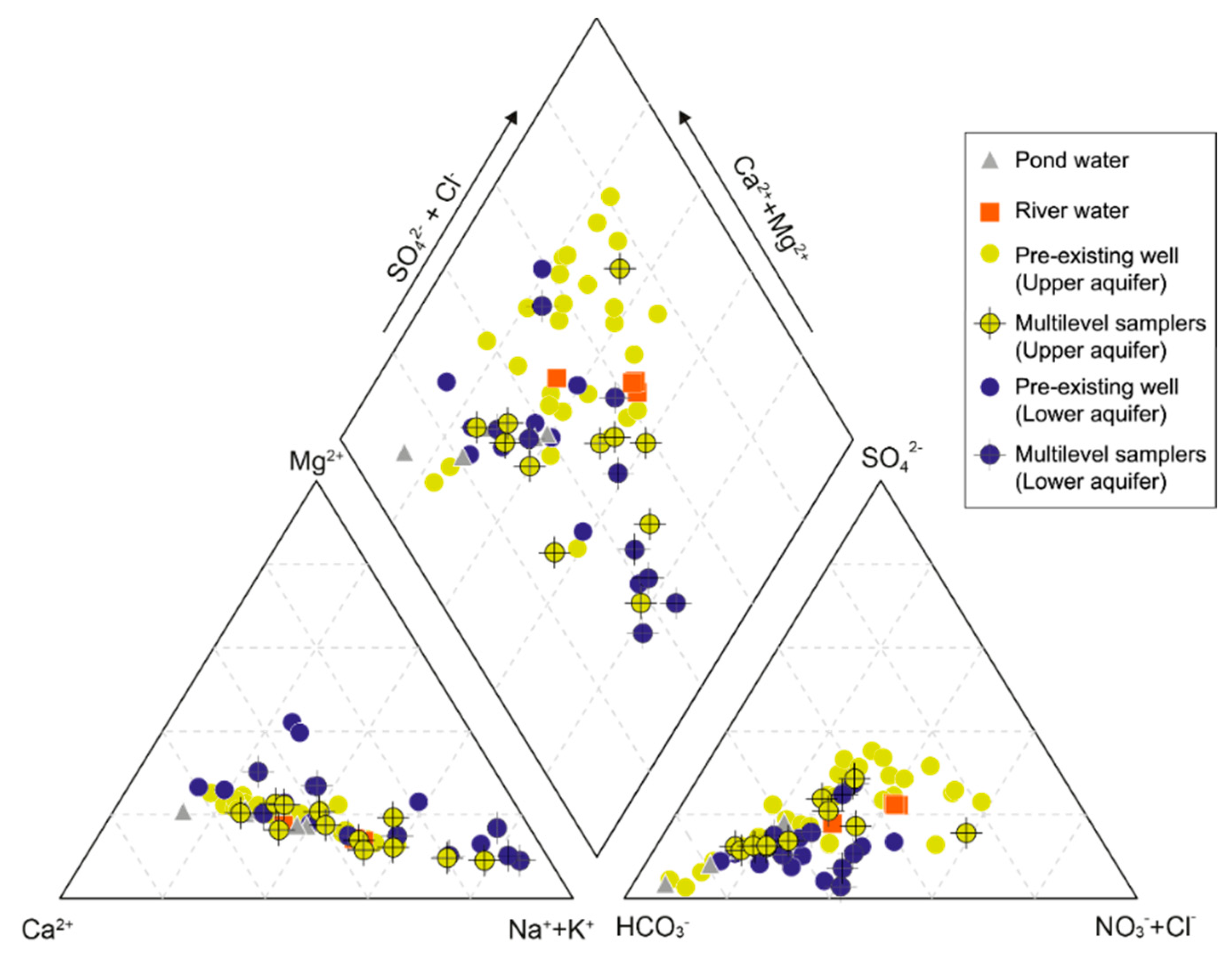
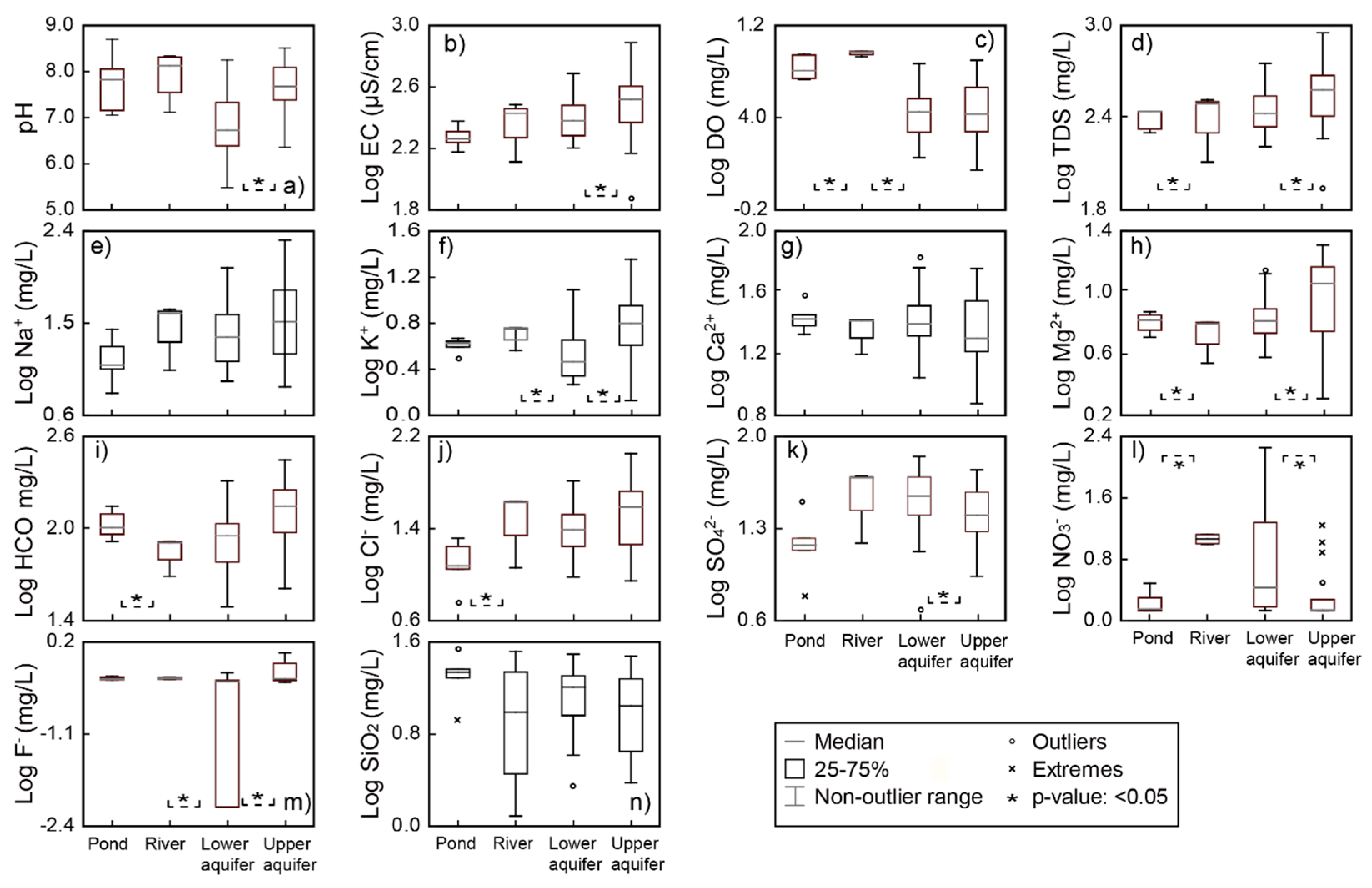
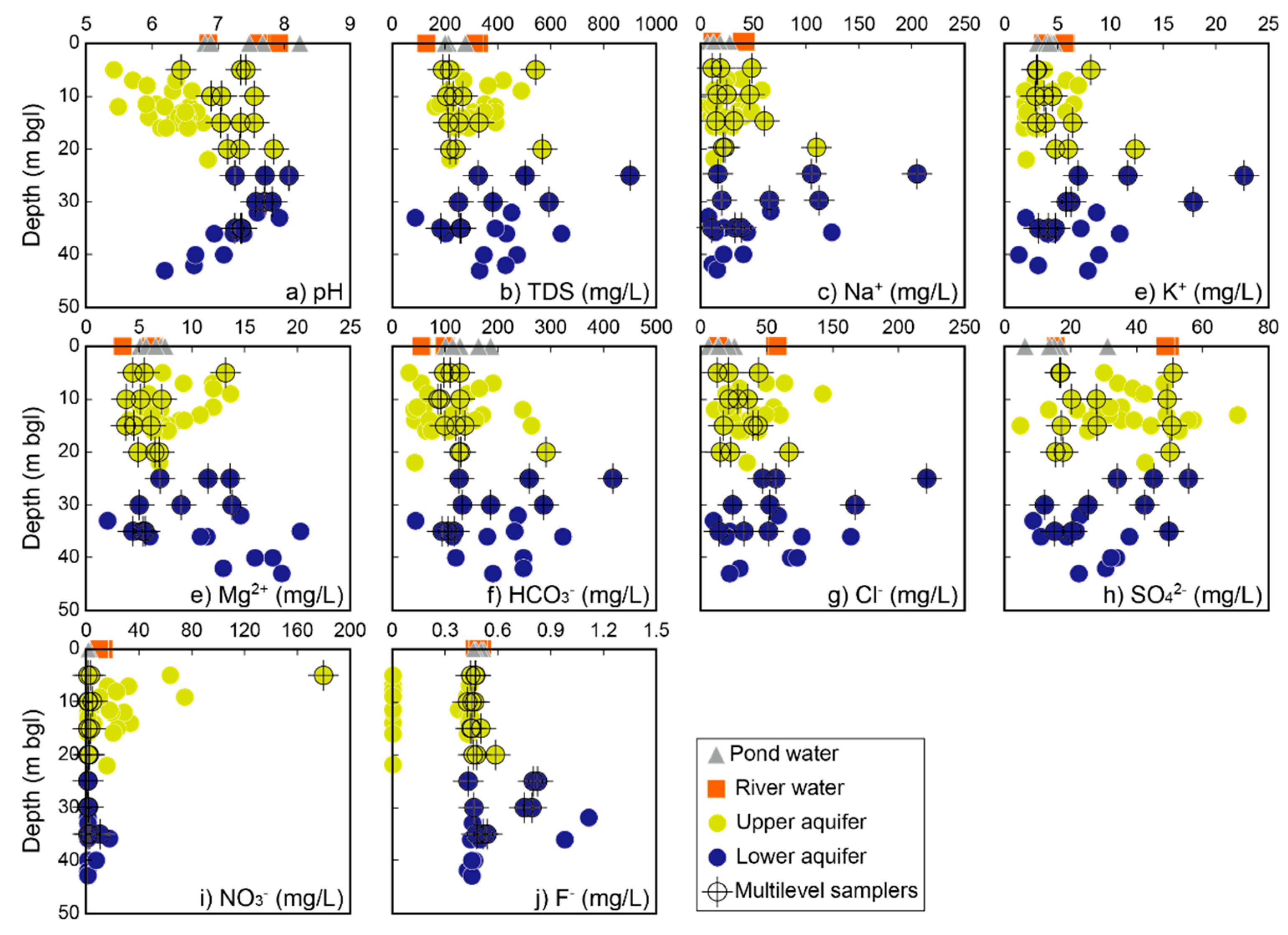
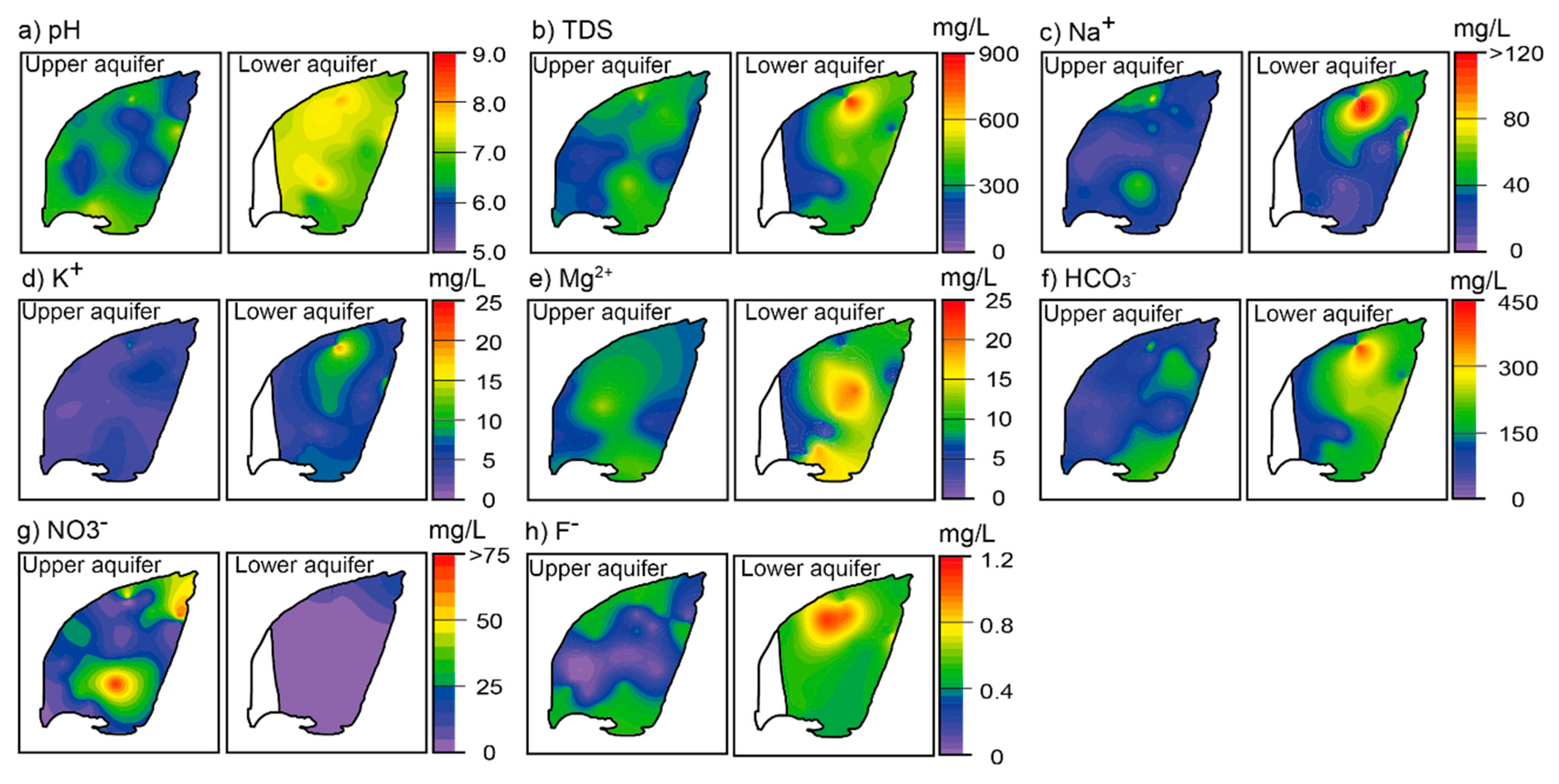
4.2. General Hydrochemistry
4.3. Vertical Change of Hydrochemistry
4.4. Evaluation of Potential Flow Paths
4.4.1. Flow Path 1: Hyporheic Flow
4.4.2. Flow Path 2: Regional Groundwater Flow
4.5. Potential Evaluation of the Use of River Bank Filtration
5. Summary and Conclusions
- The island that was studied was recently formed by point-bar sedimentation and subsequent erosion near the confluence of the Miryang River to the Nakdong River. The alluvial sediments are about 40 m thick and consist of fine- to medium-grained sand (upper aquifer; depth to about <25 m), silty sand with clay intercalations, and sandy gravel (lower aquifer; at depths between approximately 25 and 40 m) in descending order. Due to the presence of the intermediate layer as an aquitard that extends below the bottom of the river, the upper unconfined aquifer and lower confined aquifer are not hydrologically interconnected. Measurements of potentiometric head levels in the two aquifers support the different hydrogeologic conditions of the two aquifers in the riverine island.
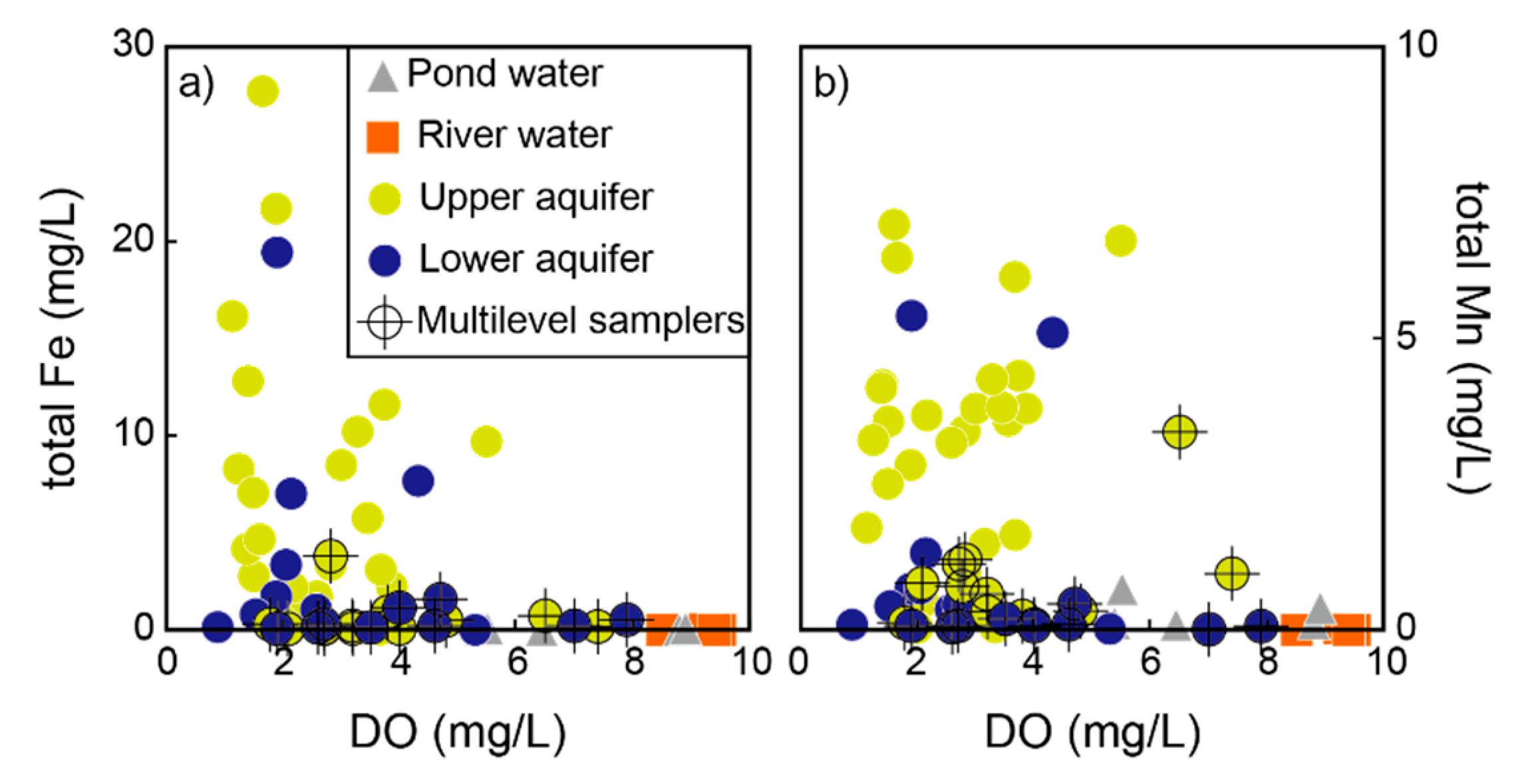
- Dissolved Mn is originated from agricultural activities on the surface, and likely, recharges from the direct infiltration. The separation of the two alluvial aquifers are also indicated by hydrochemical characteristics. Groundwater chemistry on the small island is highly variable, from Ca−Cl(−NO3) type to Na−HCO3 type. The upper aquifer groundwater is highly contaminated by nitrate and dissolved Fe in rainwater, and hyporheic flow of river water. The zone of horizontal hyporheic flow is recognized by the zone of low TDS and Mg2+ concentrations in the upper aquifer. On the other hand, the lower aquifer groundwater is enriched in TDS, Na+, K+, Mg2+, HCO3−, and F−, likely due to the water–rock interaction during regional groundwater flow. The quality of the lower aquifer is also influenced by remnant seawater under freshening.
- The results of this study indicate that the lower aquifer is not directly connected to the river channel. Therefore, sustainable large-scale bank filtration is not promising at the study site. This study implies that careful examination of groundwater chemistry can be very helpful to evaluate the potential of the use of RBF.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Doussan, C.; Ledoux, E.; Detay, M. River-groundwater exchanges, bank filtration, and groundwater quality: Ammonium behavior. J. Environ. Qual. 1998, 27, 1418–1427. [Google Scholar] [CrossRef]
- International Riverbank Filtration Conference. Proceedings of the International Riverbank Filtration Conference: 2–4 November 2000. Available online: https://d-nb.info/962327611/04 (accessed on 15 September 2018).
- Tufenkji, N.; Ryan, J.N.; Elimelech, M. The promise of bank filtration. Environ. Sci. Technol. 2002, 36, 422A–428A. [Google Scholar] [CrossRef] [PubMed]
- Ray, C.; Grischek, T.; Schubert, J.; Wang, Z.; Speth, T.F. A perspective of riverbank filtration. J. Am. Water Works Assoc. 2002, 94, 149–160. [Google Scholar] [CrossRef]
- Ray, C.; Melin, G.; Linsky, R.B. Riverbank Filtration: Improving Source Water Quality; Water Science and Technology Library: New York, NY, USA, 2002. [Google Scholar]
- Stuyfzand, P.J.; Juhasz-Holterman, M.H.A.; De Lange, W.J. Riverbed filtration in the Netherlands: Well fields, clogging and geochemical reactions. In Riverbank Filtration Hydrology—Impacts on System Capacity and Water Quality; Hubbs, S.A., Ed.; Springer: Dordrecht, The Netherlands, 2013; pp. 119–153. [Google Scholar]
- Von Gunten, H.R.; Kull, T.P. Infiltration of inorganic compounds from the Glatt river, Switzerland, into a groundwater aquifer. Water Air Soil Pollut. 1986, 29, 333–346. [Google Scholar] [CrossRef]
- Kühn, W.; Müller, U. Riverbank filtration. J. Am. Water Works Assoc. 2000, 92, 60–69. [Google Scholar] [CrossRef]
- Bouwer, H. Artificial recharge of groundwater: Hydrogeology and engineering. Hydrogeol. J. 2002, 10, 121–142. [Google Scholar] [CrossRef]
- Dash, R.R.; Mehrotra, I.; Kumar, P.; Grischek, T. Lake bank filtration at Naintal, India: Water quality evaluation. Hydrogeol. J. 2008, 16, 1089–1099. [Google Scholar] [CrossRef]
- Dash, R.R.; Bhanu Prakash, E.V.P.; Kumar, P.; Mehrotra, I.; Sandhu, C.; Grischek, T. Riverbank filtration in Haridwar, India: Removal or turbidity, organics, and bacteria. Hydrogeol. J. 2010, 18, 973–983. [Google Scholar] [CrossRef]
- Sudhakaran, S.; Lattemann, S.; Amy, G.L. Appropriate drinking water treatment processes for organic micropollutants removal based on experimental and model studies-a multi-criteria analysis study. Sci. Total Environ. 2013, 442, 478–488. [Google Scholar] [CrossRef] [PubMed]
- Gianni, G.; Richon, J.; Perrochet, P.; Vogel, A.; Brunner, P. Rapid identification of transience in streambed conductance by inversion of floodwave responses. Water Resour. Res. 2016, 52, 2647–2658. [Google Scholar] [CrossRef]
- Grischek, T.; Schoenheinz, D.; Worch, E.; Hiscock, K.M. Bank filtration in Europe-an overview of aquifer conditions and hydraulic controls. In Management of Aquifer Recharge for Sustainability; Dillon, P., Ed.; Balkema: Rotterdam, The Netherlands, 2002; pp. 485–488. [Google Scholar]
- Grischek, T.; Schoenheinz, D.; Ray, C. Siting and design issues for riverbank filtration schemes. In Riverbank Filtration; Ray, C., Melin, G., Linksy, R.B., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002; pp. 291–302. [Google Scholar]
- Sandhu, C.; Grischek, T.; Kumar, P. Potential for riverbank filtration in India. Clean Technol. Environ. Policy 2011, 13, 295–316. [Google Scholar] [CrossRef]
- Ojha, C.S.P. Simulating turbidity removal at a river bank filtration site in India using SCS-CN approach. J. Hydrol. Eng. 2012, 17, 1240–1244. [Google Scholar] [CrossRef]
- Singh, A.K.; Shah, G.; Sharma, V. Revival of defunct radial collector wells for urban water supply using river bank filtration technique in India. J. Indian Water Works Assoc. 2012, 49, 24–39. [Google Scholar]
- Chang, L.C.; Ho, C.C.; Yeh, M.S.; Yang, C.C. An integrating approach for conjunctive-use planning of surface and subsurface water system. Water Resour. Manag. 2011, 25, 59–78. [Google Scholar] [CrossRef]
- Wang, L.; Ye, X.; Du, X. Suitability evaluation of river bank filtration along the second Songhua River, China. Water 2016, 8, 176. [Google Scholar] [CrossRef]
- Korea Water Corporation. Unpublished Internal Report on Pilot Survey of Hydraulic Property of Fluvial Deposits for Water Resource Utilization; Korea Water Corporation: Daejeon, Korea, 1995; p. 132. [Google Scholar]
- Hamm, S.-Y.; Cheong, J.-Y.; Ryu, S.M.; Kim, M.J.; Kim, H.S. Hydrogeological characteristics of bank storage area in Daesan-Myeon, Changwon City, Korea. J. Geol. Soc. Korea 2002, 38, 595–610. [Google Scholar]
- Hamm, S.-Y.; Cheong, J.-Y.; Kim, H.S.; Hahn, J.S.; Cha, Y.H. Groundwater flow modeling in a riverbank filtration area, Deasan-Myeon, Changwon City. Econ. Environ. Geol. 2005, 38, 67–78. (In Korean) [Google Scholar]
- Cheong, J.-Y.; Hamm, S.-Y.; Kim, H.-S.; Ko, E.-J.; Yang, K.; Lee, J.-H. Estimating hydraulic conductivity using grain-size analyses, aquifer tests, and numerical modeling in a riverside alluvial system in South Korea. Hydrogeol. J. 2008, 16, 1129–1143. [Google Scholar] [CrossRef]
- Seo, J.A.; Kim, Y.C.; Kim, J.S.; Kim, Y.J. Site prioritization for artificial recharge in Korea using GIS mapping. J. Soil Groundwater Environ. 2011, 16, 66–78. (In Korean) [Google Scholar] [CrossRef]
- Lee, S.-H.; Hamm, S.-Y.; Ha, K.; Kim, Y.C.; Koh, D.-C.; Yoon, H.; Kim, S.-W. Hydrogeologic and paleo-geographic characteristics of riverside alluvium at an artificial recharge site in Korea. Water 2018, 10, 835. [Google Scholar] [CrossRef]
- Hiscock, K.M.; Grischek, T. Attenuation of groundwater pollution by bank filtration. J. Hydrol. 2002, 266, 139–144. [Google Scholar] [CrossRef]
- Diem, S.; Cirpka, O.A.; Schirmer, M. Modeling the dynamics of oxygen consumption upon river bank filtration by a stochastic-convective approach. J. Hydrol. 2013, 505, 352–363. [Google Scholar] [CrossRef]
- Huntscha, S.; Rodriguez Velosa, D.M; Schroth, M.H; Hollender, J. Degradation of polar organic micropollutants during riverbank filtration: Complementary results from spatiotemporal sampling and push-pull tests. Environ. Sci. Technol. 2013, 47, 11512–11521. [Google Scholar] [CrossRef] [PubMed]
- Kvitsand, H.M.L.; Myrmel, M.; Fiksdal, L.; Østerhus, S.W. Evaluation of bank filtration as a pretreatment method for the provision of hygienically safe drinking water in Norway: Results from monitoring at two full-scale sites. Hydrogeol. J. 2017, 25, 1257–1269. [Google Scholar] [CrossRef]
- Sontheimer, H. Experience with river bank filtration along the Rhine River. J. Am. Water Works Assoc. 1980, 72, 386–390. [Google Scholar] [CrossRef]
- Bourg, A.C.M.; Darmendrail, D.; Ricour, J. Geochemical filtration of riverbank and migration of heavy metals between the Deule River and the Ansereuilles alluvion-chalk aquifer (Nord, France). Geoderma 1989, 4, 229–244. [Google Scholar] [CrossRef]
- Bertin, C.; Bourg, A.C.M. Radon-222 and chloride as natural tracers of the infiltration of river water into an alluvial aquifer in which there is significant river/groundwater mixing. Environ. Sci. Technol. 1994, 28, 794–798. [Google Scholar] [CrossRef] [PubMed]
- Squillace, P.J. Observed and simulated movement of bank storage water. Ground Water 1996, 34, 121–134. [Google Scholar] [CrossRef]
- Triska, F.J.; Duff, J.H.; Avanzino, R.J. The role of water exchange between a stream channel and its hyporheic zone in nitrogen cycling at the terrestrial-aquatic interface. Hydrobiologia 1993, 251, 167–184. [Google Scholar] [CrossRef]
- Hoehn, E. Hydrogeological issues of riverbank filtration—A review. In Riverbank Filtration: Understanding Contaminant Biogeochemistry and Pathogen Removal; Ray, C., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002; pp. 17–41. [Google Scholar]
- Conant Jr., B.; Cherry, J.A.; Gillham, R.W. A PCE groundwater plume discharging to a river: Influence of the streambed and near-river zone on contaminant distributions. J. Contam. Hydrol. 2004, 73, 249–279. [Google Scholar] [CrossRef] [PubMed]
- Bencala, K.E.; Gooseff, M.N.; Kimball, B.A. Rethinking hyporheic flow and transient storage to advance understanding of stream-catchment connections. Water Resour. Res. 2011, 47, W00H03. [Google Scholar] [CrossRef]
- Boano, F.; Harvey, J.W.; Marion, A.; Packman, A.I.; Revelli, R.; Ridolfi, L.; Worman, A. Hyporheic flow and transport processes: Mechanisms, models, and biogeochemical implications. Rev. Geophys. 2015, 52, 603–679. [Google Scholar] [CrossRef]
- Hunt, H.; Schubert, J.; Ray, C. Conceptual design of riverbank filtration systems. In Riverbank Filtration: Improving Source-Water Quality; Ray, C., Melin, G., Linksy, R.B., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2003; pp. 19–27. [Google Scholar]
- Gollnitz, W.D.; Whitteberry, B.L.; Vogt, J.A. Riverbank filtration: Induced filtration and groundwater quality. J. Am. Water Works Assoc. 2004, 96, 98–110. [Google Scholar] [CrossRef]
- Massmann, G.; Nogeitzig, A.; Taute, T.; Pekdeger, A. Seasonal and spatial distribution of redox zones during lake bank filtration in Berlin, Germany. Environ. Geol. 2008, 54, 53–65. [Google Scholar] [CrossRef]
- Hubbs, S.A. Laboratory-simulated RBF particle removal processes. J. Am. Water Works Assoc. 2010, 102, 57–66. [Google Scholar] [CrossRef]
- Su, X.; Lu, S.; Gao, R.; Su, D.; Yuan, W.; Dai, Z.; Papavasilopoulos, E.N. Groundwater flow path determination during riverbank filtration affected by groundwater exploitation: A case study of Liao River, Northeast China. Hydrol. Sci. J. 2017, 62, 2331–2347. [Google Scholar] [CrossRef]
- Min, J.H.; Yun, S.T.; Kim, K.; Kim, H.S.; Kim, D.J. Geologic controls on the chemical behavior of nitrate in riverside alluvial aquifers, Korea. Hydrol. Proc. 2003, 17, 1197–1211. [Google Scholar] [CrossRef]
- Choi, B.Y.; Yun, S.T.; Mayer, B.; Chae, G.T.; Kim, K.H.; Kim, K.; Koh, Y.K. Identification of groundwater recharge sources and processes in a heterogeneous alluvial aquifer: Results from multi-level monitoring of hydrochemistry and environmental isotopes in a riverside agricultural area in Korea. Hydrol. Proc. 2010, 24, 317–330. [Google Scholar] [CrossRef]
- Daewoo Construction Co. Survey Report for Bank Filtration Project at Gimhae, Unpublished Report. 2006. (In Korean)
- Paik, S.; Cheong, D.; Shin, S.; Kim, J.C.; Park, Y.-H.; Lim, H.S. A paleoenvironmental study of Holocene delta sediments in Nakdong River Estuary. J. Geol. Soc. Korea 2016, 52, 15–30. [Google Scholar] [CrossRef]
- Kelly, W.R. Heterogeneities in ground-water geochemistry in a sand aquifer beneath an irrigated field. J. Hydrol. 1997, 198, 154–176. [Google Scholar] [CrossRef]
- Owens, L.B.; van Keuren, R.W.; Edward, W.M. Budgets of non-nitrogen nutrients in a high fertility pasture system. Agric. Ecosyst. Environ. 1998, 70, 7–18. [Google Scholar] [CrossRef]
- Böhlke, J.K. Groundwater recharge and agricultural contamination. Hydrogeol. J. 2002, 10, 153–179. [Google Scholar] [CrossRef]
- Chae, G.T.; Kim, K.; Yun, S.T.; Kim, K.; Kim, S.; Choi, B.; Kim, H.; Rhee, C.W. Hydrogeochemistry of alluvial groundwaters in an agricultural area: An implication for groundwater contamination susceptibility. Chemosphere 2004, 55, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Yun, S.T.; Choi, B.Y.; Chae, G.T.; Joo, Y.; Kim, K.; Kim, H.S. Hydrochemical and multivariate statistical interpretations of spatial controls of nitrate concentrations in a shallow alluvial aquifer around oxbow lakes (Osong area, central Korea). J. Contam. Hydrol. 2009, 107, 114–127. [Google Scholar] [CrossRef]
- Wright, R.F.; Lotse, E.; Semb, A. Reversibility of acidification shown by whole-catchment experiments. Nature 1988, 334, 670–675. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Aber, J.D.; Howarth, R.W.; Likens, G.E.; Matson, P.A.; Schindler, D.W.; Schlesinger, W.H.; Tilman, D.G. Human alteration of the global nitrogen cycle: Sources and consequences. Ecol. Appl. 1997, 7, 737–750. [Google Scholar] [CrossRef]
- Chae, G.T.; Yun, S.T.; Kwon, M.J.; Kim, Y.S.; Mayer, B. Batch dissolution of granite and biotite in water: Implication for fluorine geochemistry in groundwater. Geochem. J. 2006, 40, 95–102. [Google Scholar] [CrossRef]
- Pierso n-Wickmann, A.C.; Aquilina, L.; Weyer, C.; Molénat, J.; Lischeid, G. Acidification processes and soil leaching influenced by agricultural practices revealed by strontium isotopic ratios. Geochim. Cosmochim. Acta 2009, 73, 4688–4704. [Google Scholar] [CrossRef]
- Kim, K.; Jeong, G.Y. Factors influencing natural occurrence of fluoride-rich groundwaters: A case study in the southeastern part of the Korean Peninsula. Chemosphere 2005, 58, 1399–1408. [Google Scholar] [CrossRef] [PubMed]
- Chae, G.T.; Yun, S.T.; Mayer, B.; Kim, K.H.; Kim, S.Y.; Kwon, J.S.; Kim, K.; Koh, Y.K. Fluorine geochemistry in bedrock groundwater of South Korea. Sci. Total Environ. 2007, 385, 272–283. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.Y.; Yun, S.T.; Kim, K.H.; Kim, J.W.; Kim, H.M.; Koh, Y.K. Hydrogeochemical interpretation of South Korean groundwater monitoring data using Self-Organizing Maps. J. Geochem. Explor. 2014, 137, 73–84. [Google Scholar] [CrossRef]
- Lee, S.; Currell, M.; Cendón, D.I. Marine water from mid-Holocene sea level highstand trapped in a coastal aquifer: Evidence from groundwater isotopes, and environmental significance. Sci. Total Environ. 2016, 544, 995–1007. [Google Scholar] [CrossRef] [PubMed]
- Thomas, N.E.; Kan, K.T.; Bray, D.I.; MacQuarrie, K.T.B. Temporal changes in manganese concentrations in water from the Fredericton Aquifer, New Brunswick. Ground Water 1994, 32, 650–656. [Google Scholar] [CrossRef]
- Brown, C.J.; Schoonen, M.A.A.; Candela, J.L. Geochemical modeling of iron, sulfur, oxygen and carbon in a coastal plain aquifer. J. Hydrol. 2000, 237, 147–168. [Google Scholar] [CrossRef]
- Liaghati, T.; Preda, M.; Cox, M. Heavy metal distribution and controlling factors within coastal plain sediments, Bells Creek catchment, southeast Queensland, Australia. Environ. Int. 2004, 29, 935–948. [Google Scholar] [CrossRef]
- Bencala, K.E. Hyporheic zone hydrological processes. Hydrol. Proc. 2000, 14, 2797–2798. [Google Scholar] [CrossRef]
| Title | pH | EC (μS/cm) | DO | Na+ | K+ | Ca2+ | Mg2+ | Cl− | NO3− | SO42− | HCO3− | F− | Fe | Mn | SiO2 | TDS | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Unit: mg/L | |||||||||||||||||
| Pond water (n = 5) | Min | 6.8 | 150 | 5.4 | 6.6 | 3.1 | 21.3 | 5.1 | 5.9 | 1.3 | 6.2 | 101 | 0.5 | 0.0 | 0.1 | 9.0 | 200 |
| Median | 7.5 | 183 | 6.4 | 12.4 | 4.2 | 26.7 | 6.6 | 13.7 | 1.4 | 15.0 | 128 | 0.5 | 0.0 | 0.1 | 21.8 | 277 | |
| Max | 8.2 | 239 | 8.9 | 27.5 | 4.7 | 38.2 | 7.5 | 25.6 | 3.1 | 31.3 | 186 | 0.5 | 0.1 | 0.7 | 34.2 | 277 | |
| STD | 0.6 | 33.6 | 1.7 | 8.1 | 0.6 | 6.4 | 1.0 | 7.7 | 0.7 | 9.1 | 35.3 | 0.0 | 0.0 | 0.3 | 9.0 | 39.1 | |
| River water (n = 4) | Min | 6.9 | 130 | 8.5 | 11.0 | 3.7 | 15.7 | 3.5 | 13.1 | 10.0 | 15.5 | 54.9 | 0.5 | 0.0 | 0.0 | 1.2 | 129 |
| Median | 7.7 | 269 | 9.3 | 39.5 | 5.6 | 26.1 | 6.3 | 57.3 | 11.8 | 48.7 | 98.4 | 0.5 | 0.0 | 0.0 | 10.6 | 310 | |
| Max | 7.9 | 305 | 9.5 | 43.2 | 5.8 | 26.6 | 6.4 | 58.3 | 13.3 | 50.1 | 101 | 0.5 | 0.0 | 0.0 | 33.0 | 328 | |
| STD | 0.5 | 77.5 | 0.4 | 14.9 | 1.0 | 5.3 | 1.4 | 22.3 | 1.8 | 16.8 | 22.2 | 0.0 | 0.0 | 0.0 | 13.9 | 93.7 | |
| Upper (5 to 22 m deep) aquifer groundwater (n = 38) | Min | 5.4 | 159 | 1.1 | 8.6 | 1.8 | 11.1 | 3.8 | 10.7 | 1.4 | 4.8 | 32.0 | 0.0 | 0.0 | 0.0 | 2.2 | 163 |
| Median | 6.5 | 240 | 2.8 | 23.2 | 2.9 | 24.9 | 6.5 | 30.9 | 2.7 | 35.3 | 111 | 0.4 | 2.0 | 2.7 | 16.2 | 268 | |
| Max | 7.8 | 488 | 7.4 | 110 | 12.4 | 67.3 | 13.7 | 92.6 | 180 | 70.7 | 291 | 0.6 | 27.7 | 7.0 | 31.2 | 568 | |
| STD | 0.6 | 89.2 | 1.4 | 19.7 | 2.2 | 11.6 | 2.6 | 17.4 | 32.1 | 15.2 | 59.6 | 0.2 | 6.4 | 2.0 | 7.5 | 96.4 | |
| Lower (25 to 42 m deep) aquifer groundwater (n = 19) | Min | 6.2 | 69.8 | 0.9 | 7.6 | 1.3 | 7.5 | 2.0 | 9.8 | 1.4 | 8.7 | 44.2 | 0.4 | 0.0 | 0.0 | 2.4 | 87.4 |
| Median | 7.3 | 331 | 2.7 | 32.8 | 6.3 | 20.0 | 11.4 | 51.5 | 1.4 | 25.3 | 186 | 0.5 | 0.5 | 0.2 | 11.1 | 381 | |
| Max | 8.1 | 776 | 7.9 | 205 | 22.7 | 57.0 | 20.3 | 171 | 17.6 | 55.7 | 418 | 1.1 | 19.4 | 5.4 | 30.0 | 901 | |
| STD | 0.5 | 160 | 1.9 | 51.9 | 5.4 | 14.0 | 5.3 | 41.6 | 4.3 | 13.5 | 92.9 | 0.2 | 4.7 | 1.6 | 8.1 | 186 | |
| Aquifer | pH | EC (μS/cm) | DO | Na+ | K+ | Ca2+ | Mg2+ | Cl− | NO3− | SO42− | HCO3− | F− | Fe | Mn | SiO2 | TDS | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mg/L | |||||||||||||||||
| Upper (5 to 22 m deep) aquifer groundwater (n = 12) | Min | 6.4 | 164 | 1.8 | 11.1 | 2.9 | 11.1 | 3.8 | 12.7 | 1.4 | 15.5 | 87.0 | 0.4 | 0.0 | 0.1 | 5.9 | 191 |
| Median | 7.3 | 198 | 3.2 | 24.5 | 4.2 | 19.7 | 5.3 | 25.3 | 2.0 | 24.1 | 124 | 0.5 | 0.2 | 0.7 | 9.2 | 236 | |
| Max | 7.8 | 462 | 7.4 | 110 | 12.4 | 67.3 | 13.2 | 66.9 | 180 | 51.1 | 291 | 0.6 | 3.8 | 3.4 | 19.6 | 568 | |
| STD | 0.4 | 105 | 1.7 | 28.0 | 2.8 | 14.5 | 2.5 | 15.8 | 51.3 | 15.4 | 53.9 | 0.0 | 1.1 | 0.9 | 4.1 | 129 | |
| Lower (25 to 42 m deep) aquifer groundwater (n = 9) | Min | 7.3 | 147 | 1.9 | 10.7 | 3.2 | 9.3 | 4.4 | 14.0 | 1.4 | 12.1 | 94.6 | 0.4 | 0.1 | 0.0 | 2.4 | 184 |
| Median | 7.6 | 299 | 4.0 | 38.7 | 6.3 | 16.6 | 7.0 | 51.5 | 1.4 | 34.1 | 133 | 0.5 | 0.2 | 0.1 | 10.6 | 325 | |
| Max | 8.1 | 776 | 7.9 | 205 | 22.7 | 35.1 | 13.8 | 171 | 10.5 | 55.7 | 418 | 0.8 | 1.6 | 0.4 | 21.3 | 901 | |
| STD | 0.3 | 188 | 2.0 | 63.7 | 6.8 | 7.9 | 3.8 | 50.0 | 3.0 | 15.8 | 109 | 0.2 | 0.5 | 0.1 | 6.9 | 227 | |
| p-Value * | 0.049 | 0.169 | 0.602 | 0.310 | 0.069 | 0.310 | 0.111 | 0.049 | 0.049 | 0.862 | 0.193 | 0.023 | 0.508 | 0.000 | 0.917 | 0.129 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moniruzzaman, M.; Lee, J.-H.; Jung, K.M.; Kwon, J.S.; Kim, K.-H.; Yun, S.-T. Lithologic Control of the Hydrochemistry of a Point-Bar Alluvial Aquifer at the Low Reach of the Nakdong River, South Korea: Implications for the Evaluation of Riverbank Filtration Potential. Water 2018, 10, 1763. https://doi.org/10.3390/w10121763
Moniruzzaman M, Lee J-H, Jung KM, Kwon JS, Kim K-H, Yun S-T. Lithologic Control of the Hydrochemistry of a Point-Bar Alluvial Aquifer at the Low Reach of the Nakdong River, South Korea: Implications for the Evaluation of Riverbank Filtration Potential. Water. 2018; 10(12):1763. https://doi.org/10.3390/w10121763
Chicago/Turabian StyleMoniruzzaman, Md, Jeong-Ho Lee, Kyung Moon Jung, Jang Soon Kwon, Kyoung-Ho Kim, and Seong-Taek Yun. 2018. "Lithologic Control of the Hydrochemistry of a Point-Bar Alluvial Aquifer at the Low Reach of the Nakdong River, South Korea: Implications for the Evaluation of Riverbank Filtration Potential" Water 10, no. 12: 1763. https://doi.org/10.3390/w10121763
APA StyleMoniruzzaman, M., Lee, J.-H., Jung, K. M., Kwon, J. S., Kim, K.-H., & Yun, S.-T. (2018). Lithologic Control of the Hydrochemistry of a Point-Bar Alluvial Aquifer at the Low Reach of the Nakdong River, South Korea: Implications for the Evaluation of Riverbank Filtration Potential. Water, 10(12), 1763. https://doi.org/10.3390/w10121763






