Trace Organic Removal during River Bank Filtration for Two Types of Sediment
Abstract
1. Introduction
2. Materials and Methods
2.1. Core Sampling
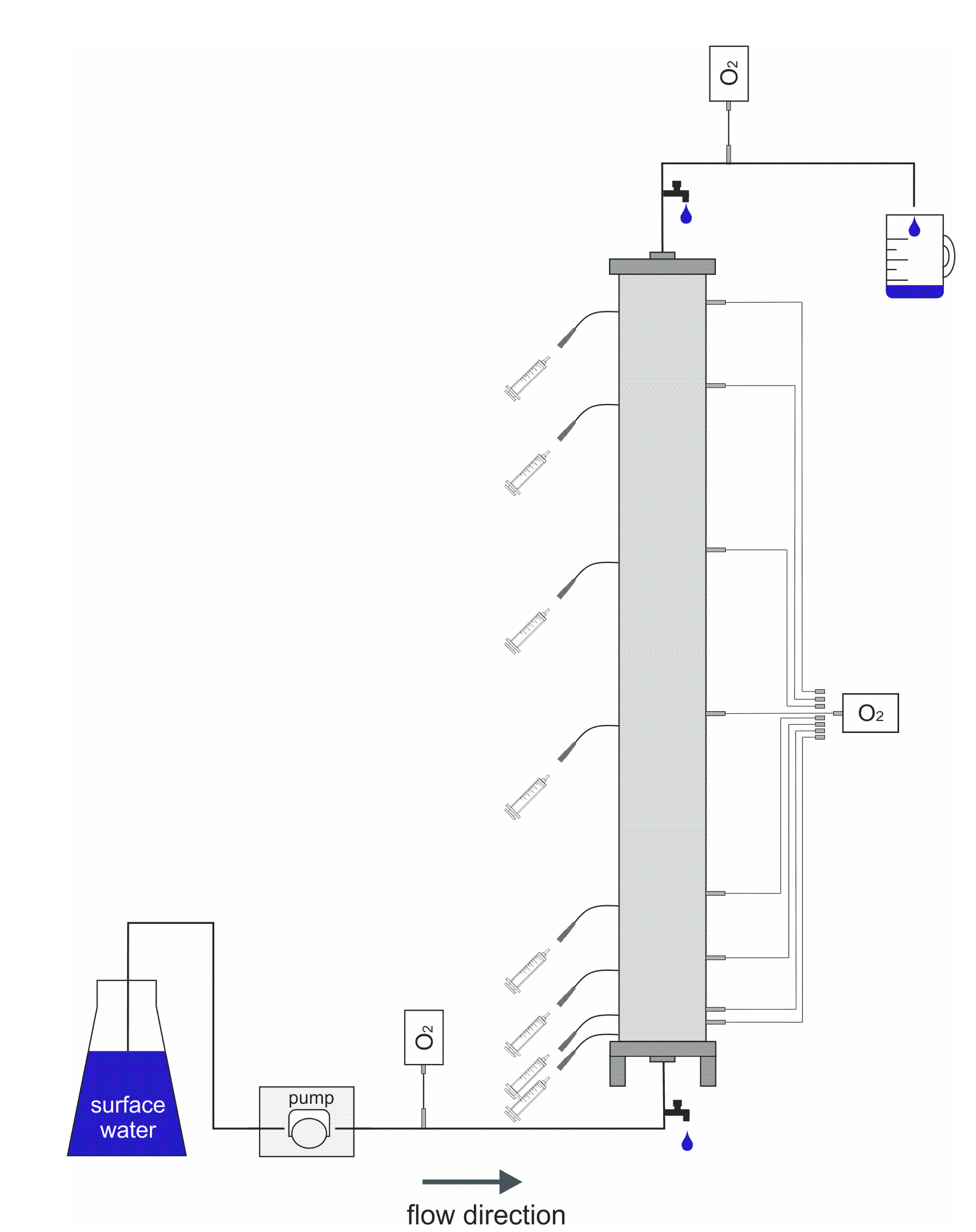
2.2. Experimental Setup
2.3. Sampling and Sample Analysis
2.4. Data Evaluation
3. Results and Discussion
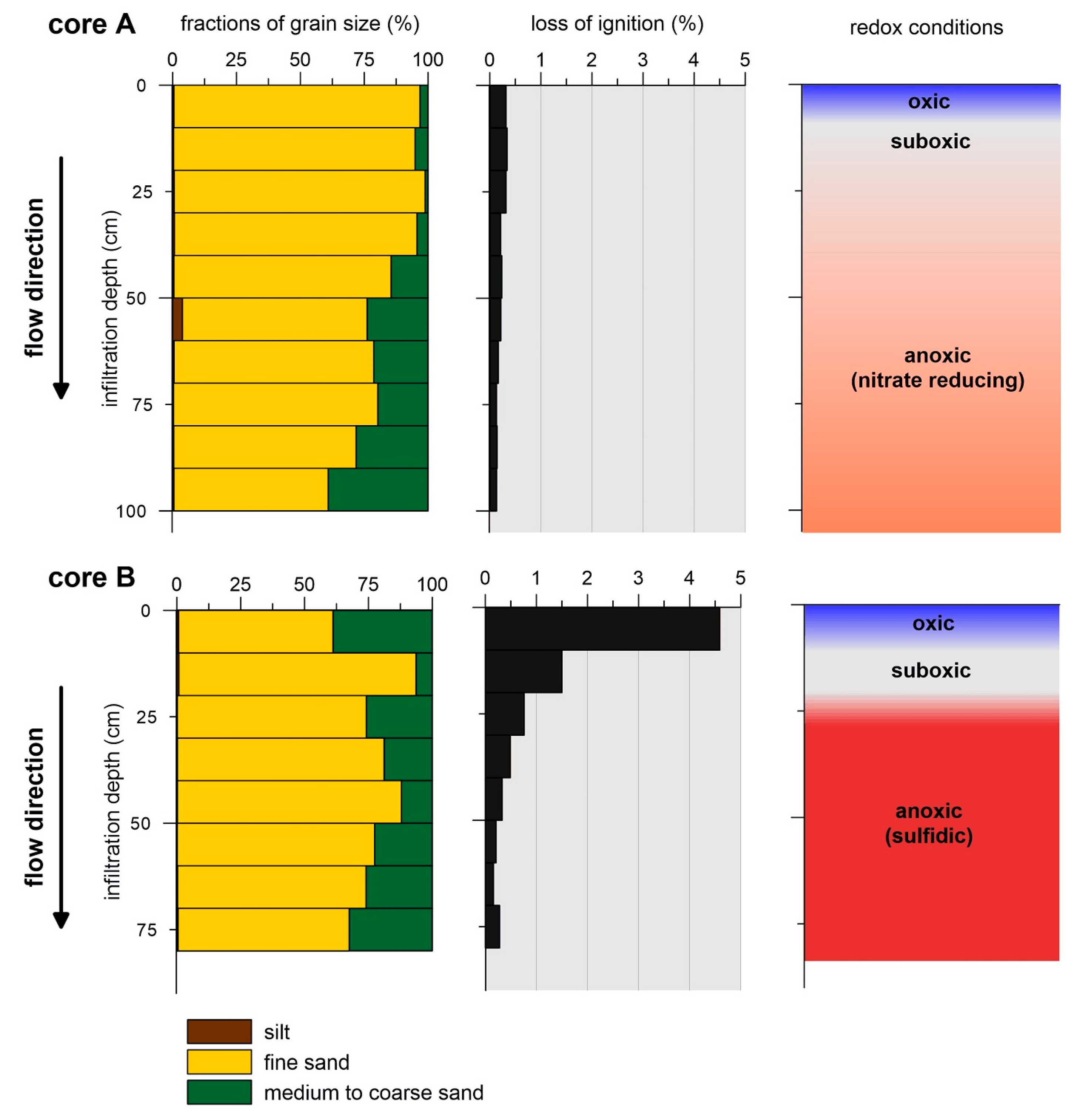
3.1. Sediment Properties and Hydrochemical Conditions
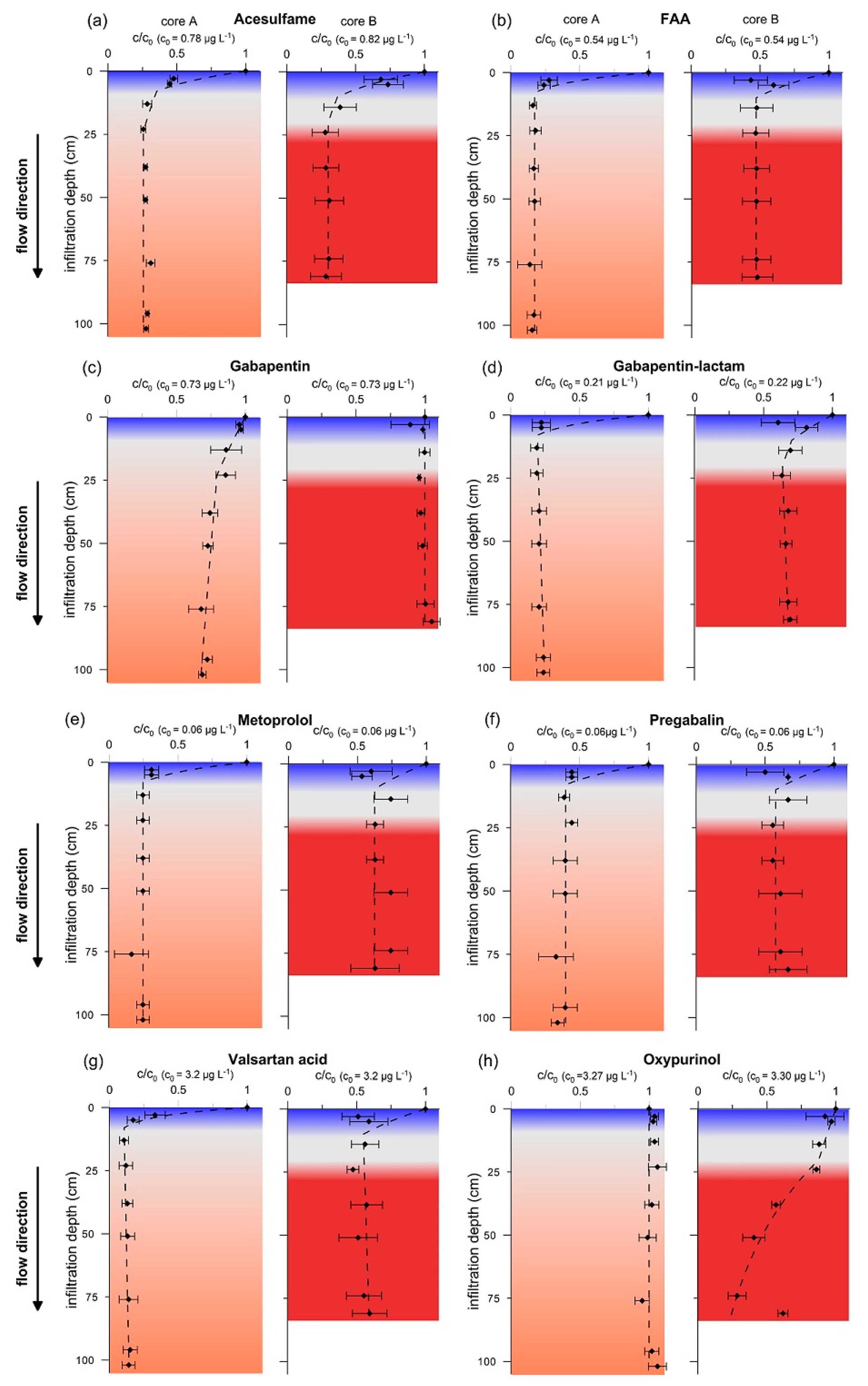
3.2. Fate of Organic Micropollutants
3.2.1. Persistent Compounds
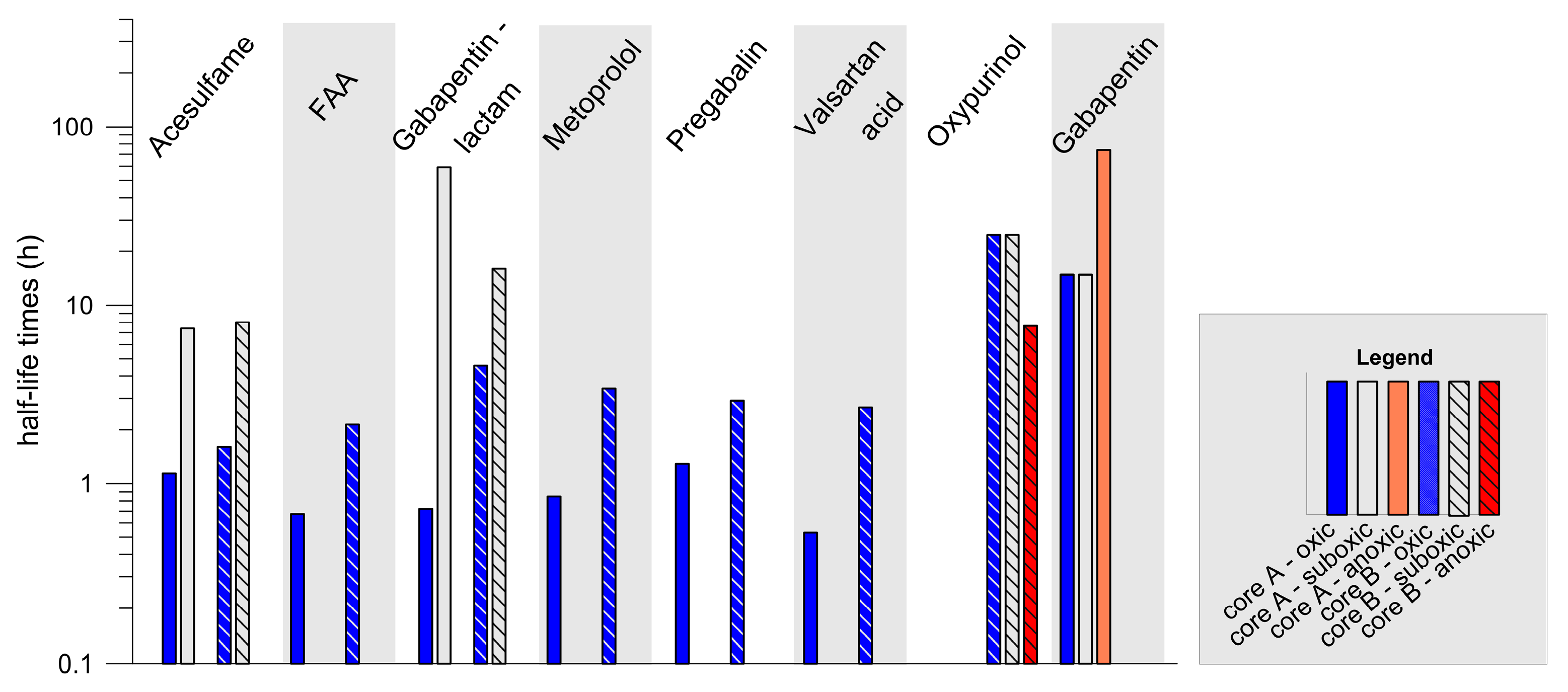
3.2.2. Reactive Compounds
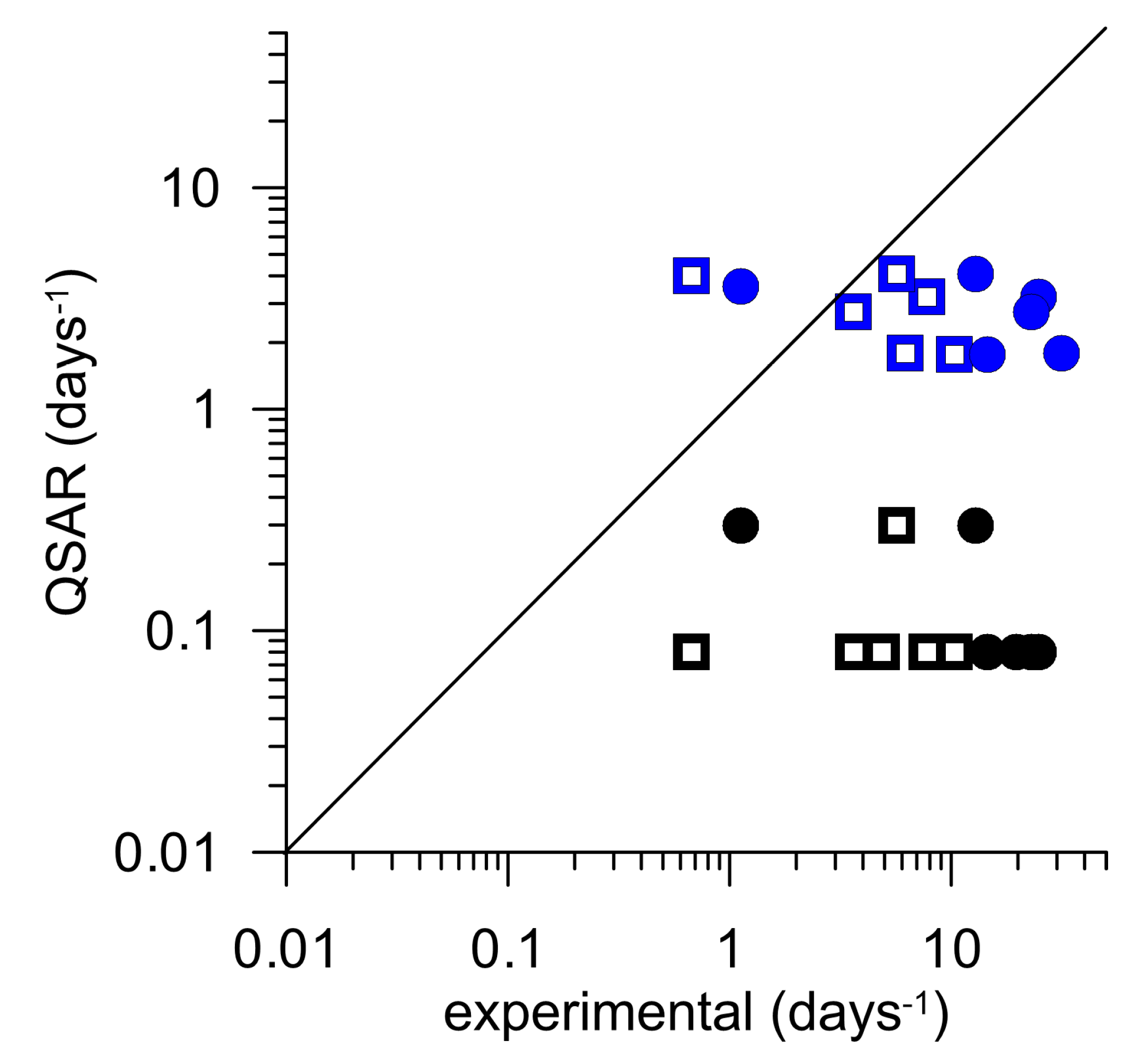
3.3. Comparison with Recent QSAR Approaches
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dillon, P. Future management of aquifer recharge. Hydrogeol. J. 2005, 13, 313–316. [Google Scholar] [CrossRef]
- Hiscock, K.M.; Grischek, T. Attenuation of groundwater pollution by bank filtration. J. Hydrol. 2002, 266, 139–144. [Google Scholar] [CrossRef]
- Dillon, P.; Stuyfzand, P.; Grischek, T.; Lluria, M.; Pyne, R.D.G.; Jain, R.C.; Bear, J.; Schwarz, J.; Wang, W.; Fernandez, E.; et al. Sixty years of global progress in managed aquifer recharge. Hydrogeol. J. 2018. [Google Scholar] [CrossRef]
- Luo, Y.; Guo, W.; Ngo, H.H.; Nghiem, L.D.; Hai, F.I.; Zhang, J.; Liang, S.; Wang, X.C. A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment. Sci. Total Environ. 2014, 473–474, 619–641. [Google Scholar] [CrossRef] [PubMed]
- Vieno, N.; Tuhkanen, T.; Kronberg, L. Elimination of pharmaceuticals in sewage treatment plants in Finland. Water Res. 2007, 41, 1001–1012. [Google Scholar] [CrossRef] [PubMed]
- Bartelt-Hunt, S.L.; Snow, D.D.; Damon, T.; Shockley, J.; Hoagland, K. The occurrence of illicit and therapeutic pharmaceuticals in wastewater effluent and surface waters in Nebraska. Environ. Pollut. 2009, 157, 786–791. [Google Scholar] [CrossRef] [PubMed]
- Kasprzyk-Hordern, B.; Dinsdale, R.M.; Guwy, A.J. The removal of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs during wastewater treatment and its impact on the quality of receiving waters. Water Res. 2009, 43, 363–380. [Google Scholar] [CrossRef] [PubMed]
- Gros, M.; Petrovic, M.; Ginebreda, A.; Barcelo, D. Removal of pharmaceuticals during wastewater treatment and environmental risk assessment using hazard indexes. Environ. Int. 2010, 36, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenbach, R.P.; Escher, B.I.; Fenner, K.; Hofstetter, T.B.; Johnson, C.A.; von Gunten, U.; Wehrli, B. The challenge of micropollutants in aquatic systems. Science 2006, 313, 1072–1077. [Google Scholar] [CrossRef] [PubMed]
- Kovacevic, S.; Radisic, M.; Lausevic, M.; Dimkic, M. Occurrence and behavior of selected pharmaceuticals during riverbank filtration in The Republic of Serbia. Environ. Sci. Pollut. Res. 2017, 24, 2075–2088. [Google Scholar] [CrossRef]
- Henzler, A.F.; Greskowiak, J.; Massmann, G. Modeling the fate of organic micropollutants during river bank filtration (Berlin, Germany). J. Contam. Hydrol. 2014, 156, 78–92. [Google Scholar] [CrossRef] [PubMed]
- Hamann, E.; Stuyfzand, P.J.; Greskowiak, J.; Timmer, H.; Massmann, G. The fate of organic micropollutants during long-term/long-distance river bank filtration. Sci. Total Environ. 2016, 545, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Maeng, S.K.; Ameda, E.; Sharma, S.K.; Grutzmacher, G.; Amy, G.L. Organic micropollutant removal from wastewater effluent-impacted drinking water sources during bank filtration and artificial recharge. Water Res. 2010, 44, 4003–4014. [Google Scholar] [CrossRef] [PubMed]
- Burke, V.; Greskowiak, J.; Asmuss, T.; Bremermann, R.; Taute, T.; Massmann, G. Temperature dependent redox zonation and attenuation of wastewater-derived organic micropollutants in the hyporheic zone. Sci. Total Environ. 2014, 482, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Trinh, T.; van den Akker, B.; Coleman, H.M.; Stuetz, R.M.; Drewes, J.E.; Le-Clech, P.; Khan, S.J. Seasonal variations in fate and removal of trace organic chemical contaminants while operating a full-scale membrane bioreactor. Sci. Total Environ. 2016, 550, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Gulde, R.; Helbling, D.E.; Scheidegger, A.; Fenner, K. pH-Dependent Biotransformation of Ionizable Organic Micropollutants in Activated Sludge. Environ. Sci. Technol. 2014, 48, 13760–13768. [Google Scholar] [CrossRef] [PubMed]
- Greskowiak, J.; Prommer, H.; Massmann, G.; Nutzmann, G. Modeling seasonal redox dynamics and the corresponding fate of the pharmaceutical residue phenazone during artificial recharge of groundwater. Environ. Sci. Technol. 2006, 40, 6615–6621. [Google Scholar] [CrossRef]
- Bertelkamp, C.; Verliefde, A.R.D.; Schoutteten, K.; Vanhaecke, L.; Bussche, J.; Singhal, N.; van der Hoek, J.P. The effect of redox conditions and adaptation time on organic micropollutant removal during river bank filtration: A laboratory-scale column study. Sci. Total Environ. 2016, 544, 309–318. [Google Scholar] [CrossRef]
- Filter, J.; Jekel, M.; Ruhl, A.S. Impacts of Accumulated Particulate Organic Matter on Oxygen Consumption and Organic Micro-Pollutant Elimination in Bank Filtration and Soil Aquifer Treatment. Water 2017, 9, 12. [Google Scholar] [CrossRef]
- Li, D.; Alidina, M.; Drewes, J.E. Role of primary substrate composition on microbial community structure and function and trace organic chemical attenuation in managed aquifer recharge systems. Appl. Microbiol. Biotechnol. 2014, 98, 5747–5756. [Google Scholar] [CrossRef]
- Alidina, M.; Li, D.; Ouf, M.; Drewes, J.E. Role of primary substrate composition and concentration on attenuation of trace organic chemicals in managed aquifer recharge systems. J. Environ. Manag. 2014, 144, 58–66. [Google Scholar] [CrossRef]
- Massmann, G.; Greskowiak, J.; Duennbier, U.; Zuehlke, S.; Knappe, A.; Pekdeger, A. The impact of variable temperatures on the redox conditions and the behaviour of pharmaceutical residues during artificial recharge. J. Hydrol. 2006, 328, 141–156. [Google Scholar] [CrossRef]
- Greskowiak, J.; Hamann, E.; Burke, V.; Massmann, G. The uncertainty of biodegradation rate constants of emerging organic compounds in soil and groundwater—A compilation of literature values for 82 substances. Water Res. 2017, 126, 122–133. [Google Scholar] [CrossRef]
- Whelton, P.K.; He, J.; Appel, L.J.; Cutler, J.A.; Havas, S.; Kotchen, T.A.; Roccella, E.J.; Stout, R.; Vallbona, C.; Winston, M.C.; et al. Primary prevention of hypertension: Clinical and public health advisory from The National High Blood Pressure Education Program. JAMA 2002, 288, 1882–1888. [Google Scholar] [CrossRef]
- Murray, C.J.L.; Lopez, A.D. Evidence-based health policy–Lessons from the global burden of disease study. Science 1996, 274, 740–743. [Google Scholar] [CrossRef]
- Stafford, R.S.; Monti, V.; Furberg, C.D.; Ma, J. Long-term and short-term changes in antihypertensive prescribing by office-based physicians in the United States. Hypertension 2006, 48, 213–218. [Google Scholar] [CrossRef]
- Campbell, N.R.C.; McAlister, F.A.; Brant, R.; Levine, M.; Drouin, D.; Feldman, R.; Herman, R.; Zarnke, K.; Canadian Hypertension Eval, P. Temporal trends in anti hypertensive drug prescriptions in Canada before and after introduction of the Canadian Hypertension Education Program. J. Hypertens. 2003, 21, 1591–1597. [Google Scholar] [CrossRef]
- Gurke, R.; Rossmann, J.; Schubert, S.; Sandmann, T.; Rossler, M.; Oertel, R.; Fauler, J. Development of a SPE-HPLC-MS/MS method for the determination of most prescribed pharmaceuticals and related metabolites in urban sewage samples. J. Chromatogr. B 2015, 990, 23–30. [Google Scholar] [CrossRef]
- Bayer, A.; Asner, R.; Schussler, W.; Kopf, W.; Weiss, K.; Sengl, M.; Letzel, M. Behavior of sartans (antihypertensive drugs) in wastewater treatment plants, their occurrence and risk for the aquatic environment. Environ. Sci Pollut. Res. 2014, 21, 10830–10839. [Google Scholar] [CrossRef]
- Letzel, T.; Bayer, A.; Schulz, W.; Heermann, A.; Lucke, T.; Greco, G.; Grosse, S.; Schüssler, W.; Sengl, M.; Letzel, M. LC-MS screening techniques for wastewater analysis and analytical data handling strategies: Sartans and their transformation products as an example. Chemosphere 2015, 137, 198–206. [Google Scholar] [CrossRef]
- Nödler, K.; Hillebrand, O.; Idzik, K.; Strathmann, M.; Schiperski, F.; Zirlewagen, J.; Licha, T. Occurrence and fate of the angiotensin II receptor antagonist transformation product valsartan acid in the water cycle—A comparative study with selected beta-blockers and the persistent anthropogenic wastewater indicators carbamazepine and acesulfame. Water Res. 2013, 47, 6650–6659. [Google Scholar] [CrossRef]
- Hermes, N.; Jewell, K.S.; Wick, A.; Ternes, T.A. Quantification of more than 150 micropollutants including transformation products in aqueous samples by liquid chromatography-tandem mass spectrometry using scheduled multiple reaction monitoring. J. Chromatogr. A 2018, 1531, 64–73. [Google Scholar] [CrossRef]
- Sperlich, A.; Harder, M.; Zietzschmann, F.; Gnirss, R. Fate of trace organic compounds in Granular Activated Carbon (GAC) adsorbers for drinking water treatment. Water 2017, 9. [Google Scholar] [CrossRef]
- Massmann, G.; Duennbier, U.; Heberer, T.; Taute, T. Behaviour and redox sensitivity of pharmaceutical residues during bank filtration—Investigation of residues of phenazone-type analgesics. Chemosphere 2008, 71, 1476–1485. [Google Scholar] [CrossRef]
- Bichler, A.; Muellegger, C.; Brünjes, R.; Hofmann, T. Quantification of river water infiltration in shallow aquifers using acesulfame and anthropogenic gadolinium. Hydrol. Process. 2015. [Google Scholar] [CrossRef]
- Storck, F.R.; Skark, C.; Remmler, F.; Brauch, H.J. Environmental fate and behavior of acesulfame in laboratory experiments. Water Sci. Technol. 2016, 74, 2832–2842. [Google Scholar] [CrossRef]
- Funke, J.; Prasse, C.; Eversloh, C.L.; Ternes, T.A. Oxypurinol—A novel marker for wastewater contamination of the aquatic environment. Water Res. 2015, 74, 257–265. [Google Scholar] [CrossRef]
- Heberer, T.; Mechlinski, A.; Fanck, B.; Knappe, A.; Massmann, G.; Pekdeger, A.; Fritz, B. Field studies on the fate and transport of pharmaceutical residues in bank filtration. Ground Water Monit. Remediat. 2004, 24, 70–77. [Google Scholar] [CrossRef]
- Massmann, G.; Sueltenfuss, J.; Duennbier, U.; Knappe, A.; Taute, T.; Pekdeger, A. Investigation of groundwater a residence times during bank filtration in Berlin: Multi-tracer approach. Hydrol. Process. 2008, 22, 788–801. [Google Scholar] [CrossRef]
- Schimmelpfennig, S.; Kirillin, G.; Engelhardt, C.; Nutzmann, G.; Dunnbier, U. Seeking a compromise between pharmaceutical pollution and phosphorus load: Management strategies for Lake Tegel, Berlin. Water Res. 2012, 46, 4153–4163. [Google Scholar] [CrossRef]
- Wurl, O. Practical Guidelines for the Analysis of Seawater; CRC Press: Boca Raton, FL, USA; Taylor & Francis Group: Abingdon-on-Thames, UK, 2009. [Google Scholar]
- Regnery, J.; Wing, A.D.; Alidina, M.; Drewes, J.E. Biotransformation of trace organic chemicals during groundwater recharge: How useful are first-order rate constants? J. Contam. Hydrol. 2015, 179, 65–75. [Google Scholar] [CrossRef]
- Fenz, R.; Blaschke, A.P.; Clara, M.; Kroiss, H.; Mascher, D.; Zessner, M. Monitoring of carbamazepine concentrations in wastewater and groundwater to quantify sewer leakage. Water Sci. Technol. 2005, 52, 205–213. [Google Scholar] [CrossRef]
- Scheurer, M.; Storck, F.R.; Graf, C.; Brauch, H.J.; Ruck, W.; Lev, O.; Lange, F.T. Correlation of six anthropogenic markers in wastewater, surface water, bank filtrate, and soil aquifer treatment. J. Environ. Monit. 2011, 13, 966–973. [Google Scholar] [CrossRef]
- Foolad, M.; Ong, S.L.; Hu, J. Transport of sewage molecular markers through saturated soil column and effect of easily biodegradable primary substrate on their removal. Chemosphere 2015, 138, 553–559. [Google Scholar] [CrossRef]
- Calisto, V.; Esteves, V.I. Adsorption of the antiepileptic carbamazepine onto agricultural soils. J. Environ. Monit. 2012, 14, 1597–1603. [Google Scholar] [CrossRef]
- Yu, L.; Fink, G.; Wintgens, T.; Melina, T.; Ternes, T.A. Sorption behavior of potential organic wastewater indicators with soils. Water Res. 2009, 43, 951–960. [Google Scholar] [CrossRef]
- Kaiser, E.; Prasse, C.; Wagner, M.; Broder, K.; Ternes, T.A. Transformation of oxcarbazepine and human metabolites of carbamazepine and oxcarbazepine in wastewater treatment and sand filters. Environ. Sci. Technol. 2014, 48, 10208–10216. [Google Scholar] [CrossRef]
- Brezina, E.; Prasse, C.; Meyer, J.; Mückter, H.; Ternes, T.A. Investigation and risk evaluation of the occurrence of carbamazepine, oxcarbazepine, their human metabolites and transformation products in the urban water cycle. Environ. Pollut. 2017, 225, 261–269. [Google Scholar] [CrossRef]
- Hellauer, K.; Mergel, D.; Ruhl, A.S.; Filter, J.; Hubner, U.; Jekel, M.; Drewes, J.E. Advancing sequential managed aquifer recharge technology (SMART) using different intermediate oxidation processes. Water 2017, 9, 14. [Google Scholar] [CrossRef]
- Khan, U.; Nicell, J. Human health relevance of pharmaceutically active compounds in drinking water. AAPS J. 2015, 17, 558–585. [Google Scholar] [CrossRef]
- Hass, U.; Duennbier, U.; Massmann, G. Occurrence and distribution of psychoactive compounds and their metabolites in the urban water cycle of Berlin (Germany). Water Res. 2012, 46, 6013–6022. [Google Scholar] [CrossRef]
- Lin, K.D.; Bondarenko, S.; Gan, J. Sorption and persistence of wastewater-borne psychoactive and antilipidemic drugs in soils. J. Soils Sediments 2011, 11, 1363–1372. [Google Scholar] [CrossRef]
- Nham, H.T.T.; Greskowiak, J.; Hamann, E.; Meffe, R.; Hass, U.; Massmann, G. Long-term transport behavior of psychoactive compounds in sewage-affected groundwater. Grundwasser 2016, 21, 321–332. [Google Scholar] [CrossRef]
- Buerge, I.J.; Buser, H.R.; Kahle, M.; Muller, M.D.; Poiger, T. Ubiquitous occurrence of the artificial sweetener acesulfame in the aquatic environment: An ideal chemical marker of domestic wastewater in groundwater. Environ. Sci. Technol. 2009, 43, 4381–4385. [Google Scholar] [CrossRef]
- Scheurer, M.; Brauch, H.-J.; Lange, F.T. Analysis and occurrence of seven artificial sweeteners in German waste water and surface water and in soil aquifer treatment (SAT). Anal. Bioanal. Chem. 2009, 394, 1585–1594. [Google Scholar] [CrossRef]
- Li, S.L.; Ren, Y.H.; Fu, Y.Y.; Gao, X.S.; Jiang, C.; Wu, G.; Ren, H.Q.; Geng, J.J. Fate of artificial sweeteners through wastewater treatment plants and water treatment processes. PLoS ONE 2018, 13, e16. [Google Scholar] [CrossRef]
- Li, Z.; Sobek, A.; Radke, M. fate of pharmaceuticals and their transformation products in four small European rivers receiving treated wastewater. Environ. Sci. Technol. 2016, 50, 5614–5621. [Google Scholar] [CrossRef]
- Müller, C.E.; Gerecke, A.C.; Alder, A.C.; Scheringer, M.; Hungerbühler, K. Identification of perfluoroalkyl acid sources in Swiss surface waters with the help of the artificial sweetener acesulfame. Environ. Pollut. 2011, 159, 1419–1426. [Google Scholar] [CrossRef]
- Yang, Y.Y.; Zhao, J.L.; Liu, Y.S.; Liu, W.R.; Zhang, Q.Q.; Yao, L.; Hu, L.X.; Zhang, J.N.; Jiang, Y.X.; Ying, G.G. Pharmaceuticals and personal care products (PPCPs) and artificial sweeteners (ASs) in surface and ground waters and their application as indication of wastewater contamination. Sci. Total Environ. 2018, 616, 816–823. [Google Scholar] [CrossRef]
- Spoelstra, J.; Senger, N.D.; Schiff, S.L. Artificial sweeteners reveal septic system effluent in rural groundwater. J. Environ. Qual. 2017, 46, 1434–1443. [Google Scholar] [CrossRef]
- Robertson, W.D.; Van Stempvoort, D.R.; Solomon, D.K.; Homewood, J.; Brown, S.J.; Spoelstra, J.; Schiff, S.L. Persistence of artificial sweeteners in a 15-year-old septic system plume. J. Hydrol. 2013, 477, 43–54. [Google Scholar] [CrossRef]
- Castronovo, S.; Wick, A.; Scheurer, M.; Nodler, K.; Schulz, M.; Ternes, T.A. Biodegradation of the artificial sweetener acesulfame in biological wastewater treatment and sandfilters. Water Res. 2017, 110, 342–353. [Google Scholar] [CrossRef]
- Kahl, S.; Kleinsteuber, S.; Nivala, J.; van Afferden, M.; Reemtsma, T. Emerging biodegradation of the previously persistent Artificial Sweetener Acesulfame in biological wastewater treatment. Environ. Sci. Technol. 2018, 52, 2717–2725. [Google Scholar] [CrossRef]
- Burke, V.; Richter, D.; Hass, U.; Duennbier, U.; Greskowiak, J.; Massmann, G. Redox-dependent removal of 27 organic trace pollutants: Compilation of results from tank aeration experiments. Environ. Earth Sci. 2014, 71, 3685–3695. [Google Scholar] [CrossRef]
- Burke, V.; Greskowiak, J.; Grunenbaum, N.; Massmann, G. Redox and temperature dependent attenuation of twenty organic micropollutants—A systematic column study. Water Environ. Res. 2017, 89, 155–167. [Google Scholar] [CrossRef]
- de Wilt, A.; He, Y.; Sutton, N.; Langenhoff, A.; Rijnaarts, H. Sorption and biodegradation of six pharmaceutically active compounds under four different redox conditions. Chemosphere 2018, 193, 811–819. [Google Scholar] [CrossRef]
- Henning, N.; Kunkel, U.; Wick, A.; Ternes, T.A. Biotransformation of gabapentin in surface water matrices under different redox conditions and the occurrence of one major TP in the aquatic environment. Water Res. 2018, 137, 290–300. [Google Scholar] [CrossRef]
- Altmann, J.; Rehfeld, D.; Trader, K.; Sperlich, A.; Jekel, M. Combination of granular activated carbon adsorption and deep-bed filtration as a single advanced wastewater treatment step for organic micropollutant and phosphorus removal. Water Res. 2016, 92, 131–139. [Google Scholar] [CrossRef]
- Altmann, J.; Sperlich, A.; Jekel, M. Integrating organic micropollutant removal into tertiary filtration: Combining PAC adsorption with advanced phosphorus removal. Water Res. 2015, 84, 58–65. [Google Scholar] [CrossRef]
- Hellauer, K.; Karakurt, S.; Sperlich, A.; Burke, V.; Massmann, G.; Hubner, U.; Drewes, J.E. Establishing sequential managed aquifer recharge technology (SMART) for enhanced removal of trace organic chemicals: Experiences from field studies in Berlin, Germany. J. Hydrol. 2018, 563, 1161–1168. [Google Scholar] [CrossRef]
- Schaffer, M.; Kroger, K.F.; Nodler, K.; Ayora, C.; Carrera, J.; Hernandez, M.; Licha, T. Influence of a compost layer on the attenuation of 28 selected organic micropollutants under realistic soil aquifer treatment conditions: Insights from a large scale column experiment. Water Res. 2015, 74, 110–121. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Estimation Programs Interface Suite™ for Microsoft® Windows, v 4.1; United States Environmental Protection Agency: Washington, DC, USA, 2012.
- Bertelkamp, C.; Verliefde, A.; Reynisson, J.; Singhal, N.; Cabo, A.; De Jonge, M.; van der Hoek, J.P. A predictive multi-linear regression model for organic micropollutants, based on a laboratory-scale column study simulating the river bank filtration process. J. Hazard. Mater. 2016, 304, 502–511. [Google Scholar] [CrossRef]
- Santschi, P.; Hohener, P.; Benoit, G.; Buchholtztenbrink, M. Chemical processes at the sediment water interface. Mar. Chem 1990, 30, 269–315. [Google Scholar] [CrossRef]
- Heberer, T.; Massmann, G.; Fanck, B.; Taute, T.; Dunnbier, U. Behaviour and redox sensitivity of antimicrobial residues during bank filtration. Chemosphere 2008, 73, 451–460. [Google Scholar] [CrossRef]
- Jüttner, F. Efficacy of bank filtration for the removal of fragrance compounds and aromatic hydrocarbons. Water Sci. Technol. 1999, 40, 123–128. [Google Scholar] [CrossRef]
| Redox Environment | Criteria | |
|---|---|---|
| oxic | DO > 1 mg L−1 | |
| suboxic | 0 < DO < 1 mg L−1 | |
| anoxic | nitrate reducing | NO3− > 0 mg L−1 and Mn2+ < 0.05 mg L−1 and Fe2+ < 0.05 mg L−1 |
| mangnese/iron reducing | Mn2+ > 0.05 mg L−1 or/and Fe2+ > 0.05 mg L−1 | |
| sulfidic | S2− > 0 mg L−1 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burke, V.; Schneider, L.; Greskowiak, J.; Zerball-van Baar, P.; Sperlich, A.; Dünnbier, U.; Massmann, G. Trace Organic Removal during River Bank Filtration for Two Types of Sediment. Water 2018, 10, 1736. https://doi.org/10.3390/w10121736
Burke V, Schneider L, Greskowiak J, Zerball-van Baar P, Sperlich A, Dünnbier U, Massmann G. Trace Organic Removal during River Bank Filtration for Two Types of Sediment. Water. 2018; 10(12):1736. https://doi.org/10.3390/w10121736
Chicago/Turabian StyleBurke, Victoria, Laura Schneider, Janek Greskowiak, Patricia Zerball-van Baar, Alexander Sperlich, Uwe Dünnbier, and Gudrun Massmann. 2018. "Trace Organic Removal during River Bank Filtration for Two Types of Sediment" Water 10, no. 12: 1736. https://doi.org/10.3390/w10121736
APA StyleBurke, V., Schneider, L., Greskowiak, J., Zerball-van Baar, P., Sperlich, A., Dünnbier, U., & Massmann, G. (2018). Trace Organic Removal during River Bank Filtration for Two Types of Sediment. Water, 10(12), 1736. https://doi.org/10.3390/w10121736






