Reduction and Accumulative Characteristics of Dissolved Heavy Metals in Modified Bioretention Media
Abstract
:1. Introduction
2. Materials and Methods
2.1. Media Preparation
2.2. Device Setting
2.3. Experimental Design
2.4. Analysis Methods
3. Results and Discussion
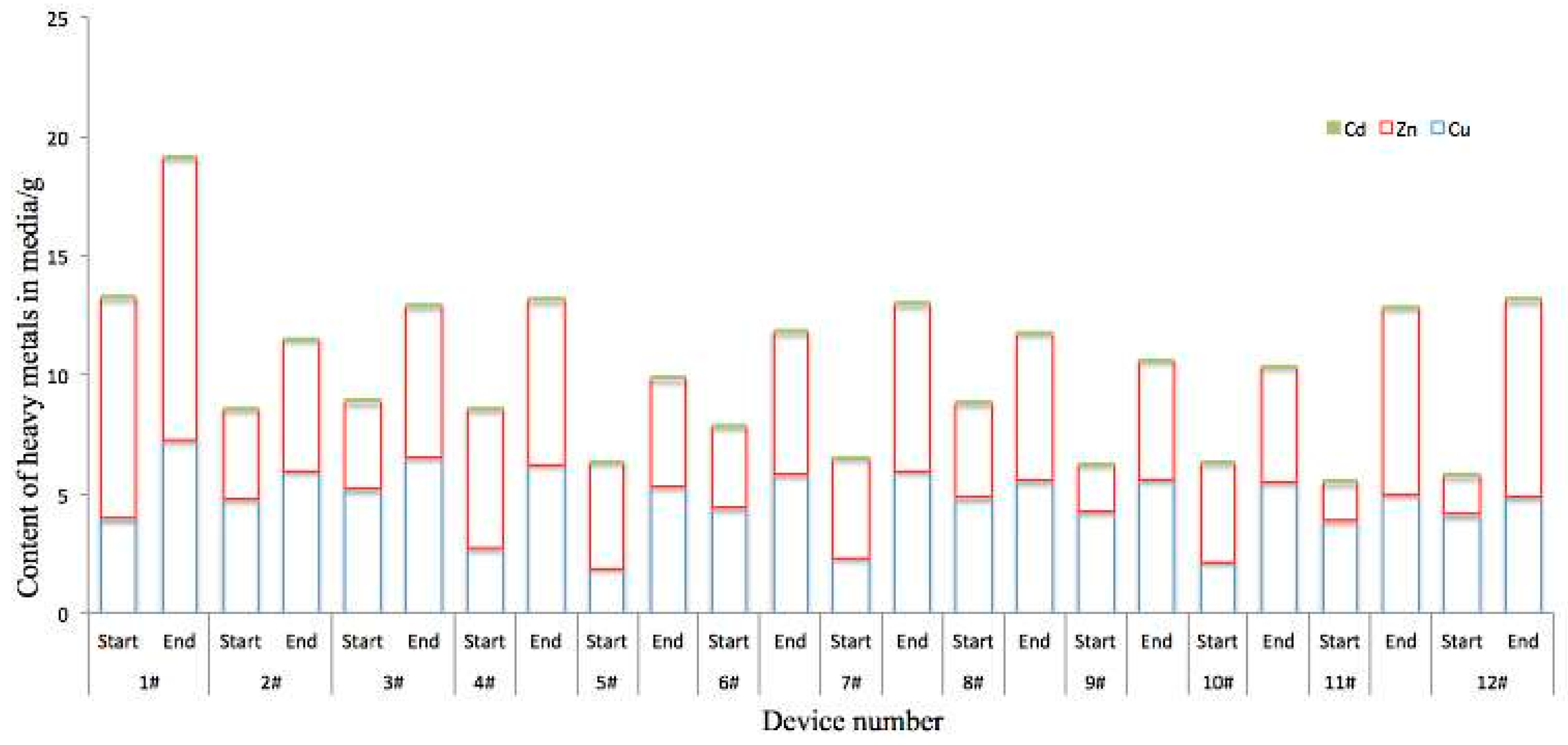
3.1. Dissolved Heavy Metal Concentration Removal Effects
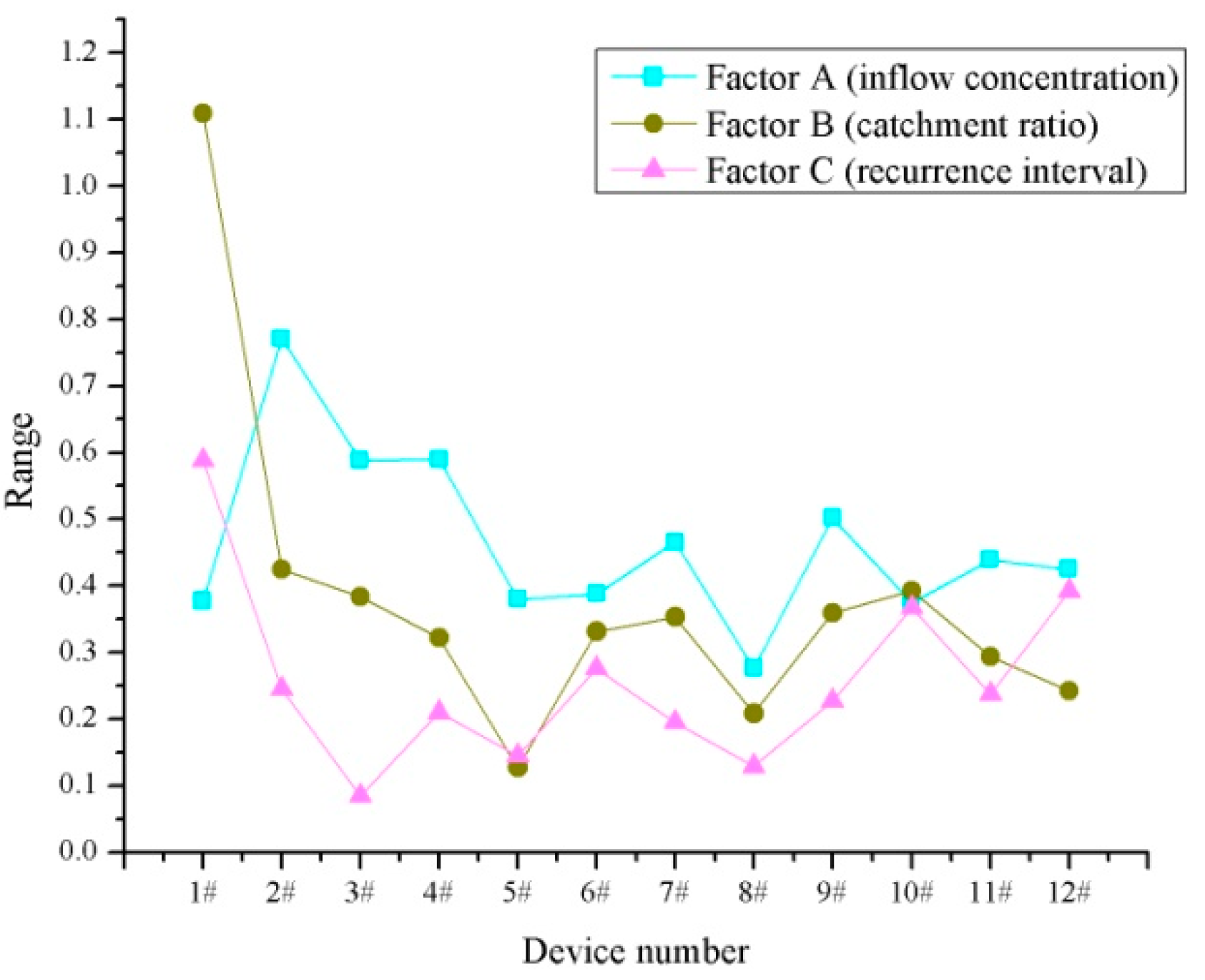
3.2. Relationship between Treatment Effect and Test Factors
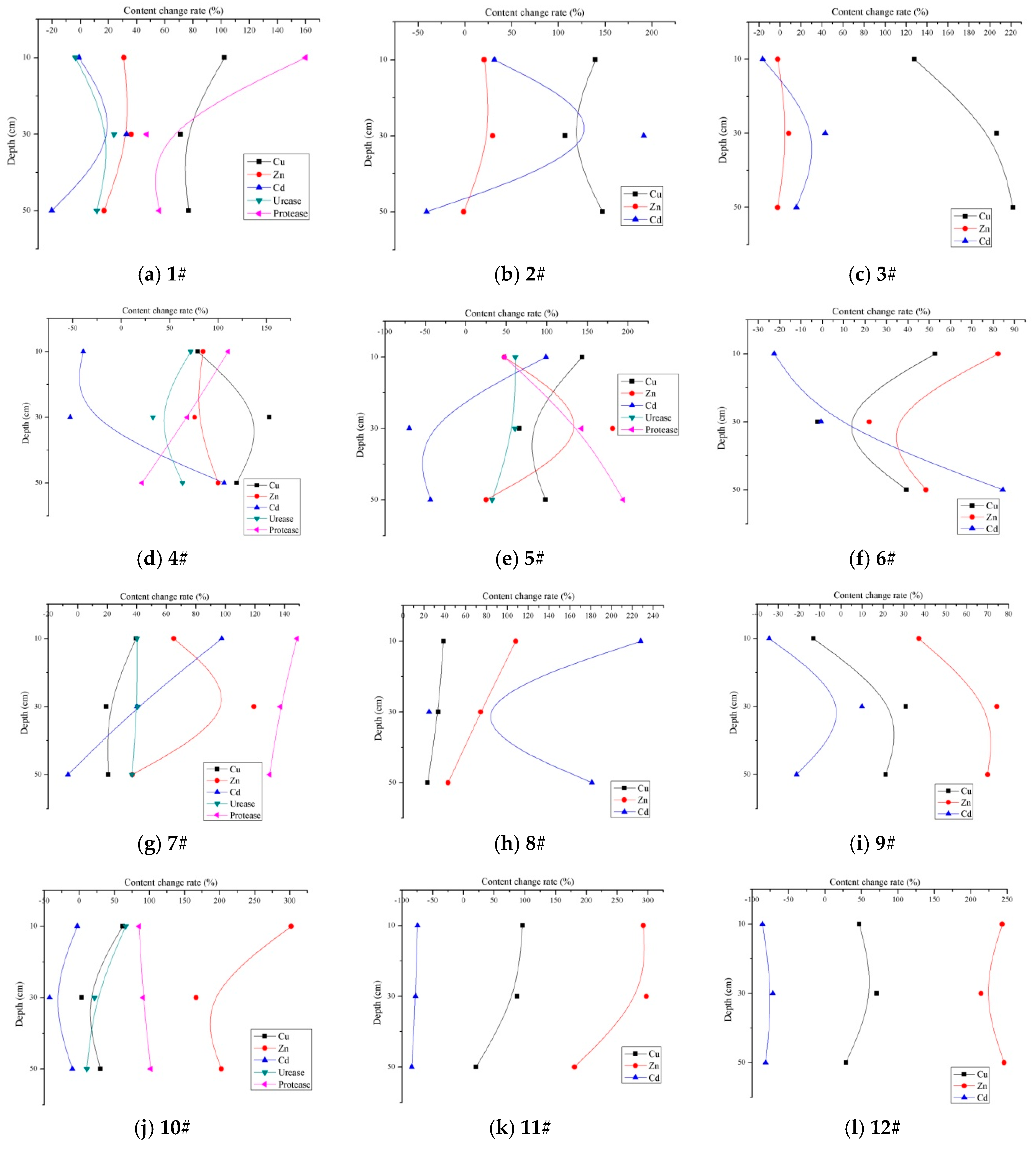
3.3. Adsorption and Accumulation of Heavy Metals and Difference of Enzyme Activity in Different Media
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hu, A.; Li, Z.; Zhang, S.; Liu, J.; Chen, J. Research Progress on urban road rain water quality. Geomat. World 2010, 46, 123–127. [Google Scholar] [CrossRef]
- Ma, Y.; Egodawatta, P.; Mcgree, J.; Liu, A.; Goonetilleke, A.; Goonetilleke, A. Human health risk assessment of heavy metals in urban stormwater. Sci. Total Environ. 2016, 557, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.M.; Fiala, M.J.; Park, D.; Wade, T.L. Review of pollutants in urban road dust and stormwater runoff: Part 1. Heavy metals released from vehicles. Int. J. Urban Sci. 2016, 20, 1–27. [Google Scholar] [CrossRef]
- Grogoglione, A.; Bombardelli, F.A.; Pitton, B.J.L.; Oki, L.R.; Haver, D.L.; Young, T.M. Role of Sediments in Insecticide Runoff from Urban Surfaces: Analysis and Modeling. Int. J. Environ. Res. Public Health 2018, 15, 1464. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Gan, J. Importance of Fine Particles in Pesticide Runoff from Concrete Surfaces and Its Prediction. Environ. Sci. Technol. 2012, 46, 6028–6034. [Google Scholar] [CrossRef] [PubMed]
- Bressy, A.; Gromaire, M.-C.; Lorgeoux, C.; Saad, M.; Leroy, F.; Chebbo, G. Towards the determination of an optimal scale for stormwater quality management: Micropollutants in a small residential catchment. Water Res. 2012, 46, 6799–6810. [Google Scholar] [CrossRef] [PubMed]
- Lefevre, G.H.; Paus, K.H.; Natarajan, P. Review of dissolved pollutants in urban storm water and their removal and fate in bioretention cells. J. Environ. Eng. 2015, 141. [Google Scholar] [CrossRef]
- Davis, A.P.; Traver, R.G.; Hunt, W.F. Improving urban stormwater quality: Applying fundamental principles. Water Res. 2010, 146, 3–10. [Google Scholar] [CrossRef]
- Clark, S.E.; Pitt, R. Targeting treatment technologies to address specific stormwater pollutants and numeric discharge limits. Water Res. 2012, 46, 6715–6730. [Google Scholar] [CrossRef] [PubMed]
- Erickson, A.J.; Gulliver, J.S.; Weiss, P.T. Enhanced sand filtration for storm water phosphorus removal. Environ. Eng. 2007, 133, 485–497. [Google Scholar] [CrossRef]
- Kayhanian, M.; Fruchtman, B.D.; Gulliver, J.S.; Montanaro, C.; Raniere, E.; Wuertz, S. Review of highway runoff characteristics: Comparative analysis and universal implications. Water Res. 2012, 46, 6609–6624. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wei, X.; Pomeroy, C.A. Review of current research and future directions of low impact development practices for storm water. Adv. Water Sci. 2011, 22, 287–293. [Google Scholar]
- Sheoran, A.S.; Sheoran, V. Heavy metal removal mechanism of acid mine drainage in wetlands: A critical review. Miner. Eng. 2006, 19, 105–116. [Google Scholar] [CrossRef]
- Ranieri, E.; Gorgoglione, A.; Montanaro, C.; Iacovelli, A.; Gikas, P. Removal capacity of BTEX and metals of constructed wetlands under the influence of hydraulic conductivity. Desalin. Water Treat. 2015, 56, 5. [Google Scholar] [CrossRef]
- De Macedo, M.B.; Rosa, A.; Do Lago, C.A.F.; Mendiondo, E.M.; de Souza, V.C.B. Learning from the operation, pathology and maintenance of a bioretention system to optimize urban drainage practices. J. Environ. Manag. 2017, 204, 454–466. [Google Scholar] [CrossRef] [PubMed]
- Ingvertsen, S.T.; Cederkvist, K.; Jensen, M.B. Assessment of existing roadside swales with engineered filter soil: II. Treatment efficiency and in situ mobilization in soil columns. J. Environ. Qual. 2012, 41, 1970–1981. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.; Davis, A.P. Evaluation and optimization of bioretention media for treatment of urban storm water runoff. J. Environ. Eng. 2005, 131, 1521–1531. [Google Scholar] [CrossRef]
- Rahmani, A.; Mousavi, H.Z.; Fazli, M. Effect of nanostructure alumina on adsorption of heavy metals. Desalination 2010, 253, 94–100. [Google Scholar] [CrossRef]
- Reddy, K.R.; Xie, T.; Dastgheibi, S. Removal of heavy metals from urban stormwater runoff using different filter materials. J. Environ. Chem. Eng. 2014, 2, 282–292. [Google Scholar] [CrossRef]
- Lim, H.S.; Lim, W.; Hu, J.Y. Comparison of filter media materials for heavy metal removal from urban stormwater runoff using biofiltration systems. J. Environ. Manag. 2015, 147, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Trenouth, W.R.; Gharabaghi, B. Soil amendments for heavy metals removal from stormwater runoff discharging to environmentally sensitive areas. J. Hydrol. 2015, 529, 1478–1487. [Google Scholar] [CrossRef]
- Bratieres, K.; Fletcher, T.D.; Deletic, A. Nutrient and sediment removal by stormwater biofilters: A large-scale design optimization study. Water Res. 2008, 42, 3930–3940. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Li, J.; Li, Y. Adsorption Characteristics of Several Bioretention-Modified Fillers for Phosphorus. Water 2018, 10, 831. [Google Scholar] [CrossRef]
- Berndtsson, J.C. Green roof performance towards management of runoff water quantity and quality: A review. Ecol. Eng. 2010, 4, 351–360. [Google Scholar] [CrossRef]
- Read, J.; Wevill, T.; Fletcher, T. Variation among plant species in pollutant removal from storm water in biofiltration systems. Water Res. 2008, 42, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J. Study on the Changes in Rainfall Patterns in Xi’an; Xi’an University of Architecture & Technology: Xi’an, China, 2015. [Google Scholar]
- Lu, J.; Cheng, Y.; Deng, Q.; Du, R.; Wang, S.; Wang, J. Derivation of rainstorm intensity Equation in Xi’an city. Chin. Water Wastewater 2010, 26, 82–84. [Google Scholar]
- Sun, X.; Davis, A.P. Heavy metal fates in laboratory bioretention systems. Chemosphere 2007, 66, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Al-Ameri, M.; Hatt, B.; Coustumer, S.L. Accumulation of heavy metals in stormwaterbioretention media: A field study of temporal and spatial variation. J. Hydrol. 2018. [Google Scholar] [CrossRef]
- Li, H.; Davis, A.P. Water quality improvement through reductions of pollutant loads using bioretention. J. Environ. Eng. 2009, 135, 567–576. [Google Scholar] [CrossRef]
- Blecken, G.T.; Marsalek, J.; Viklander, M. Laboratory study of stormwater biofiltration in low temperatures: Total and dissolved metal removals and fates. Water Air Soil Pollut. 2011, 219, 303–317. [Google Scholar] [CrossRef]
- Mullane, J.M.; Flury, M.; Iqbal, H. Intermittent rainstorms cause pulses of nitrogen, phosphorus, and copper in leach ate from compost in bioretention systems. Sci. Total Environ. 2015, 537, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Zeng, G.; Du, C. Transfer of heavy metals from compost to red soil and groundwater under simulated rainfall conditions. J. Hazard. Mater. 2010, 181, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Dietz, M.E.; Clausen, J.C. Saturation to Improve Pollutant Retention in a Rain Garden. Environ. Sci. Technol. 2006, 40, 1335–1340. [Google Scholar] [CrossRef] [PubMed]
- Muthanna, T.M.; Viklander, M.; Gjesdahl, N. Heavy Metal Removal in Cold Climate Bioretention. Water Air Soil Pollut. 2007, 183, 391–402. [Google Scholar] [CrossRef]


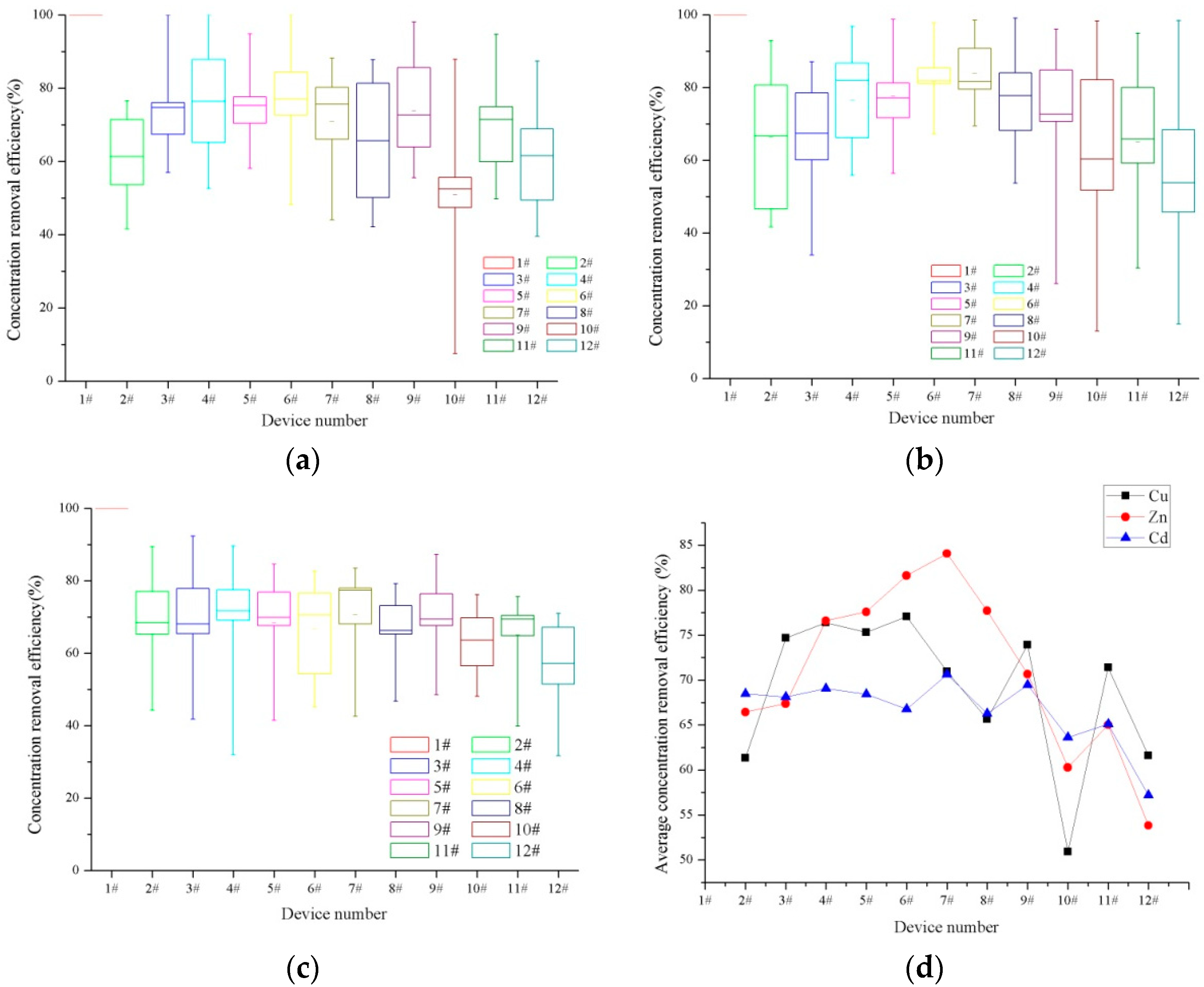
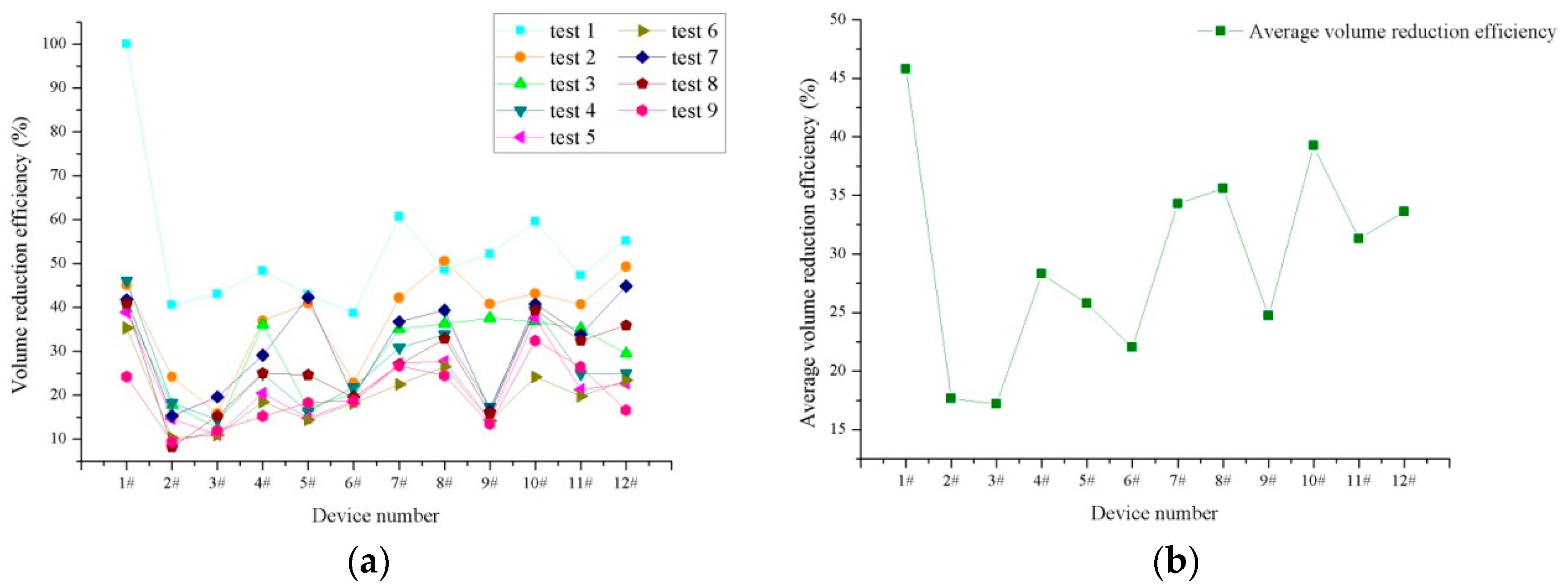
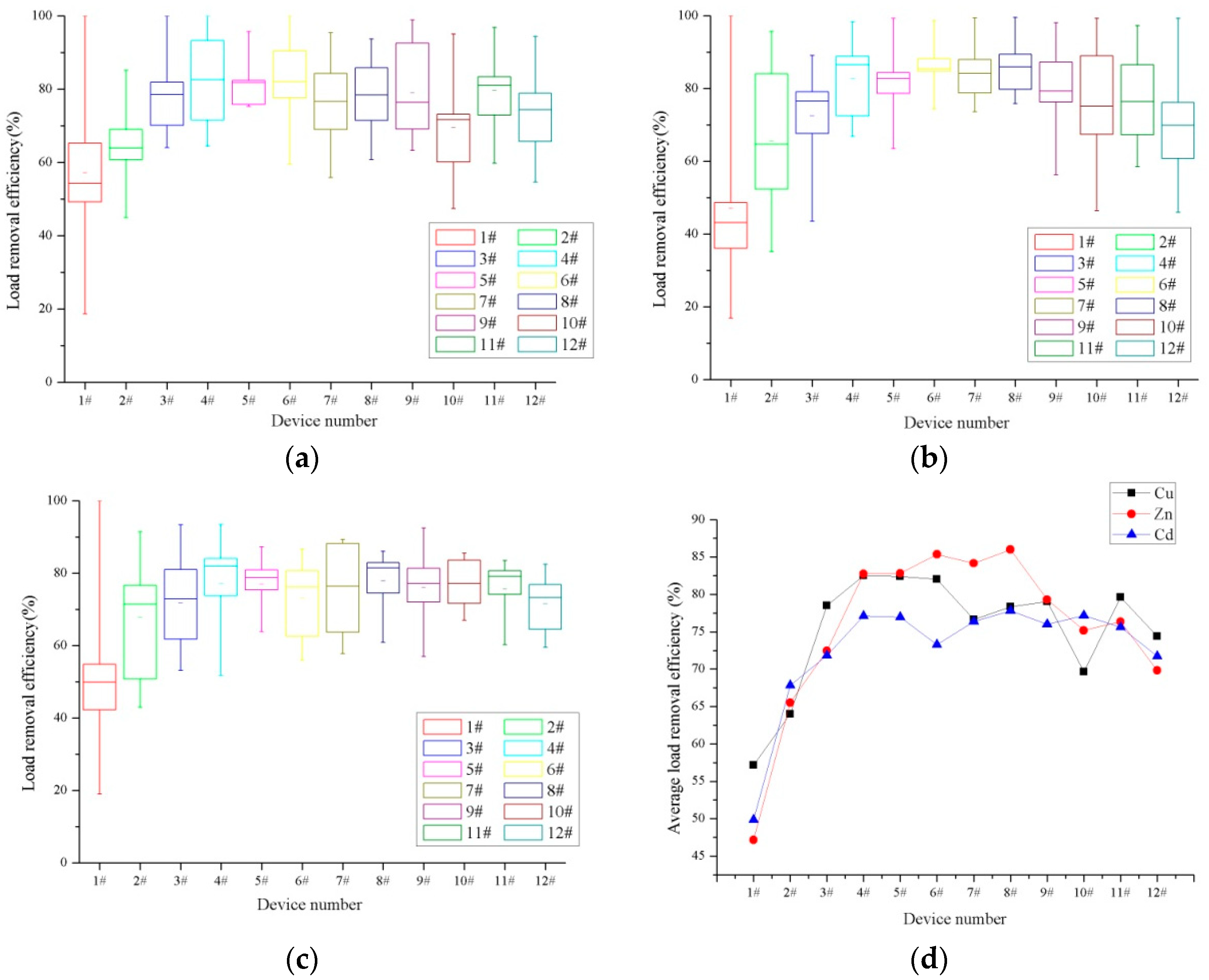
| Device Number | 1# | 2# | 3# | 4# | 5# | 6# |
| Media (by mass) | Local soil | 30%Local soil+70%Sand | Bioretention soil media (BSM) | BSM+10%WTR | BSM+10%Gz | BSM+10%Ms |
| Device Number | 7# | 8# | 9# | 10# | 11# | 12# |
| Media (by mass) | BSM+10% fly ash | BSM+5% vermiculite | BSM+5%Ts | BSM+5% coconut bran | BSM+2.5%Ms+2.5%Ts | BSM+2.5%Gz+2.5%Ts |
| Media | Organic Matter % | Specific Surface Area m2/g | Ion Exchange Capacity cmol/kg | Pore Volume cm3/g | Packed Density g/mL |
|---|---|---|---|---|---|
| Loal soil | 0.032 | 20.837 | 19.44 | 0.03 | 1.121 |
| Sand | 0.745 | 0.8037 | 11.76 | 0.0021 | 1.53 |
| BSM | 7.55 | 4.9909 | 34.45 | 0.0096 | 1.116 |
| Water treatment residue (WTR) | 10.3 | 28.433 | 9.31 | 0.0215 | 0.953 |
| Green zeolite (Gz) | 6.98 | 16.8707 | 27.50 | 0.0510 | 1.054 |
| Medicinal stone (Ms) | 4.14 | 1.2723 | 13.39 | 0.0033 | 1.297 |
| Fly ash | 2.66 | 1.3814 | 23.23 | 0.0066 | 1.008 |
| Vermiculite | 1.122 | 1.5942 | 17.38 | 0.0162 | 0.120 |
| Turfy soil (Ts) | 36.6 | 0.7400 | 14.44 | 0.0018 | 0.136 |
| Coconut bran | 4.65 | 0.8105 | 13.62 | 0.0026 | 0.092 |
| Level | Factor A (Inflow Concentration, mg·L−1) | Factor B (Discharge Ratio) | Factor C (Recurrence Interval) | ||
|---|---|---|---|---|---|
| Cu | Zn | Cd | |||
| 1 | 1 | 1.5 | 0.5 | 10:1 | 0.5a |
| 2 | 0.5 | 1 | 0.3 | 15:1 | 2a |
| 3 | 0.3 | 0.5 | 0.1 | 20:1 | 3a |
| Test | Inflow Concentration | Catchment Ratio | Recurrence Interval | Antecedent Dry Time/day | Rainfall Duration/min | Precipitation/L |
|---|---|---|---|---|---|---|
| 1 | A1 | B1 | C1 | 6 | 60 | 151.5 |
| 2 | A1 | B2 | C2 | 6 | 60 | 227.3 |
| 3 | A1 | B3 | C3 | 6 | 60 | 303.0 |
| 4 | A2 | B1 | C2 | 6 | 60 | 315.3 |
| 5 | A2 | B2 | C3 | 6 | 60 | 473.0 |
| 6 | A2 | B3 | C1 | 6 | 60 | 630.7 |
| 7 | A3 | B1 | C3 | 6 | 60 | 363.3 |
| 8 | A3 | B2 | C1 | 6 | 60 | 544.9 |
| 9 | A3 | B3 | C2 | 6 | 60 | 726.5 |
| Device Number | Range/A | Range/B | Range/C | Optimal Test Condition |
|---|---|---|---|---|
| 1# | 0.377 | 1.109 | 0.588 | A1B1C1 |
| 2# | 0.771 | 0.425 | 0.245 | A1B2C2 |
| 3# | 0.588 | 0.383 | 0.085 | A1B2C3 |
| 4# | 0.590 | 0.322 | 0.210 | A1B2C2 |
| 5# | 0.380 | 0.127 | 0.145 | A1B2C2 |
| 6# | 0.388 | 0.331 | 0.276 | A1B2C2 |
| 7# | 0.465 | 0.353 | 0.195 | A1B1C2 |
| 8# | 0.276 | 0.208 | 0.129 | A1B2C2 |
| 9# | 0.502 | 0.359 | 0.227 | A1B2C2 |
| 10# | 0.374 | 0.392 | 0.367 | A1B2C2 |
| 11# | 0.439 | 0.294 | 0.237 | A1B2C2 |
| 12# | 0.425 | 0.242 | 0.392 | A1B2C1 |
| Content Difference | Cu | Zn | Cd | Summation |
|---|---|---|---|---|
| Urease | 0.20 | 0.32 | 0.21 | 0.52 |
| Protease | −0.34 | - | −0.36 | −0.45 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Wen, M.; Li, J.; Chai, B.; Jiang, C. Reduction and Accumulative Characteristics of Dissolved Heavy Metals in Modified Bioretention Media. Water 2018, 10, 1488. https://doi.org/10.3390/w10101488
Li Y, Wen M, Li J, Chai B, Jiang C. Reduction and Accumulative Characteristics of Dissolved Heavy Metals in Modified Bioretention Media. Water. 2018; 10(10):1488. https://doi.org/10.3390/w10101488
Chicago/Turabian StyleLi, Yajiao, Meng Wen, Jiake Li, Beibei Chai, and Chunbo Jiang. 2018. "Reduction and Accumulative Characteristics of Dissolved Heavy Metals in Modified Bioretention Media" Water 10, no. 10: 1488. https://doi.org/10.3390/w10101488
APA StyleLi, Y., Wen, M., Li, J., Chai, B., & Jiang, C. (2018). Reduction and Accumulative Characteristics of Dissolved Heavy Metals in Modified Bioretention Media. Water, 10(10), 1488. https://doi.org/10.3390/w10101488




