Abstract
This study aimed to characterize airborne bacteria and fungi populations present in rural nursery schools in the Upper Silesia region of Poland during winter and spring seasons through quantification and identification procedures. Bacterial and fungal concentration levels and size distributions were obtained by the use of a six-stage Andersen cascade impactor. Results showed a wide range of indoor bioaerosols levels. The maximum level of viable bacterial aerosols indoors was about 2600 CFU·m−3, two to three times higher than the outdoor level. Fungi levels were lower, from 82 to 1549 CFU·m−3, with indoor concentrations comparable to or lower than outdoor concentrations. The most prevalent bacteria found indoors were Gram-positive cocci (>65%). Using the obtained data, the nursery school exposure dose (NSED) of bioaerosols was estimated for both the children and personnel of nursery schools. The highest dose for younger children was estimated to range: 327–706 CFU·kg−1 for bacterial aerosols and 31–225 CFU·kg−1 for fungal aerosols. These results suggest an elevated risk of adverse health effects on younger children. These findings may contribute to the promotion and implementation of preventative public health programs and the formulation of recommendations aimed at providing healthier school environments.
1. Introduction
The amount of time that people spend in indoor environments intensifies concern regarding the potential health hazards of social infrastructures. According to studies conducted over the last 20 years by the U.S. Environmental Protection Agency, indoor air can be 70–100 times more polluted than outdoor air [1,2]. Most indoor air pollution occurs at concentrations that are not considered to be harmful, yet prolonged exposure may adversely affect human health.
Among indoor environments, nursery schools are one of the most critical in any given society, since children are particularly at risk of lung damage and infection caused by poor air quality [3]. Children are more vulnerable than adults to contamination because their activities are more diverse, they breathe more air than adults do relative to their body size [4], and their immune systems and bodies are less mature [5]. Moreover, organisms in growth phases are more susceptible to damage than already-developed organisms [6].
Bioaerosols are a large category of airborne particles, comprising bacteria, fungi, viruses, pollen, and fragments of these or their metabolic products (e.g., endotoxins, mycotoxins). Bioaerosols are, to a great extent, of natural origin (e.g., rotting leaves, mold growth in damp areas) and are therefore ubiquitous in natural environments [7]. It has been reported that approximately 24% of total atmospheric particles and 5%–10% of total suspended particulates are bioaerosols [8].
Most of these studies have focused on the total concentration of bioaerosols and regard to the changes of airborne microbial community with season or location. This information is indispensable for the assessment of population exposure, as well as for the identification of biological aerosol emission sources.
Bioaerosols vary considerably in size, from approximately 0.02 to 100 µm. The size distribution of bioaerosols depends on the type of microorganism species, age of the spore and nutrient medium, humidity, differences in aggregation rates of the spores, and the type of particles with which the spores are associated, such as mist or dust. It should be noted that bioaerosol particles can occur in air as single cells or aggregates of cells, as well as in fragments. They are often transported attached to other particles, such as skin flakes, soil, dust, saliva, or water droplets [9,10].
Although bacteria are ubiquitous in every habitat on Earth—growing in soil, water, and organic matter, including in the live bodies of plants and animals—their presence in indoor environments is mostly related to human and animal occupancy [11,12]. Some reviewed studies have reported that the predominant bacteria species are Micrococcus spp., Staphylococcus spp. and Bacillus spp., while Penicillium spp., Aspergillus spp., and Cladosporium spp. are the most abundant mold genera in indoor air [13,14,15,16]. Fungi consist of long, microscopic strands of hyphae and require specific growth conditions, such as the availability of an appropriate substrate, optimum temperature, and humidity [15]. Worldwide, some studies have been carried out on airborne bacteria and fungi in different regions and settings [8,16,17]. A significant part of these investigations concerns the various types of generally non-compliant indoor environments: schools [3,18,19,20], residential settings [9,13,16,21], hospitals [22,23], less is known about children’s daycare centers [24,25,26,27]. In reviewing these studies, we found relatively few examples of research focusing on younger children [14,28,29,30]. Researchers often encounter problems in gaining access to institutions like nursery schools, as well as in installing the necessary measuring equipment in such a way as to avoid disturbances during the measurement process and to limit children’s curiosity regarding the measuring devices [31].
The aim of this study is to investigate the concentration levels of culturable bacterial and fungal aerosols, their size distribution, and seasonal (winter-spring) variations in two rural nursery schools—in older and younger children’s classrooms, respectively. The aim of this study was to investigate: (a) the concentration levels of culturable airborne bacteria and fungi during two seasons characterized by the lowest and highest concentrations of these bioaerosols (winter and spring); (b) the size distributions with particular attention on respirable fraction; (c) the difference between bacterial community structure in older and younger children’s classrooms. To our knowledge, this is the first study to present bioaerosol data regarding a rural area of Poland. There are currently no formally regulated standards for bioaerosol levels in Poland. This study can thus increase awareness and provide references for better understanding of IAQ in the rural areas of developing countries.
2. Materials
The study was carried out in two nursery schools situated about 10 km north of the city of Gliwice (Figure 1), which is located in the west district of the industrial region of Upper Silesia, Poland. The major activities influencing the ambient air quality of these two sites are agricultural activities (during non-heating periods) and burning of biomass and fossil fuels for domestic needs (during heating periods). There were two sampling campaigns: from the 18 February–6 March 2014, designated as the winter campaign, and 8–21 of May 2014, called the spring campaign.
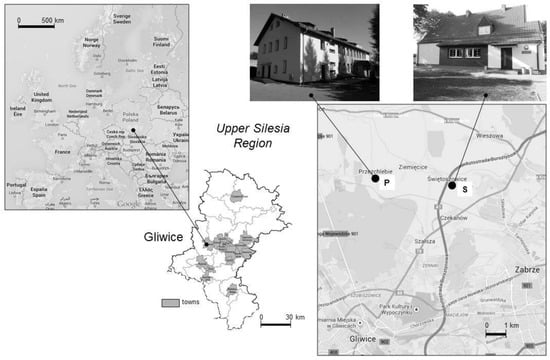
Figure 1.
Location of the investigated nursery schools (Map data: 2015© Google, ORION-ME).
The first building (50°22′6.35″ N, 18°41′2.87″ E), labelled “P” for the Przezchlebie village, represents a typical rural location, while the second building (50°21′50.99″ N, 18°43′56.73″ E), labelled “S” for the Świętoszowice village, is located 50 m from the A1 highway (a section that has been open since 2011). This building is separated from the highway by highway screens. Both nursery schools are public and managed entirely by the municipality authorities and the Ministry of Education using public funds. They are both detached buildings (Figure 1) that underwent thermal efficiency improvement processes, completed in the summer of 2013. During the thermal insulation process, natural ventilation using the buildings’ air duct systems was left unchanged. Consequently, the IAQ is primarily ensured by means of stack ventilation and airing through open and unsealed windows. Both nursery schools are located in detached buildings with two floors. The (P) building was constructed in the 1970s, while the (S) building in the 1930s before the Second World War. The classrooms in (P) building have wooden parquet with one-third covered with carpet. In (S) building, the classrooms are covered in halves with laminated HDF panels and carpet. The classrooms in both buildings were subject to the same cleaning schedule, some daily cleaning in corridors and common spaces were made during children’s occupancy in the classrooms and the deep cleaning was made daily after the occupancy period. The buildings both are equipped with a central heating system and have kitchens that use gas stoves. The kitchens are located on the ground (P) or first (S) floor. Children attending the nursery schools are divided into three groups. Depending on the season, each group contained 9–25 children, with one or two nursery staff members. The measurements were conducted in both the classrooms of older (five- to six-year-old, O) and younger (three- to four-year-old, Y) children. Table 1 summarizes the specification of the two sampling sites, including basic IAQ parameters and the median occupancy of each classroom during the winter and spring seasons.

Table 1.
Summary of the occupancy and IAQ parameter characteristics for each studied classroom, as well as for the outdoor air.
3. Methods
A six-stage Andersen cascade impactor was used to sample airborne bacteria and fungi. The aerodynamic diameter ranges for each stage were: stage 1 (>7 µm), stage 2 (4.7–7 µm), stage 3 (3.3–4.7 µm), stage 4 (2.1–3.3 µm), stage 5 (1.1–2.1 µm), and stage 6 (0.65–1.1 µm). Air was sampled at a constant flow rate of 28.3 L·min−1 for 10 min using a calibrated pump and impacted directly onto agar plates (bacterial aerosols) or malt extract agar plates (fungal aerosols). During the sampling periods, indoor and outdoor temperature, relative humidity (RH), and CO2 concentrations were also monitored using automatic portable monitors (model 77535, Az Instruments International Ltd., Hong Kong, China). Each monitor, equipped with a nondispersive infrared sensor, was connected to a PC with RS232 software installed. The precision of measurements ranged between 0 and 10,000 ppm CO2: ±100 ppm CO2 or ±3% at a concentration below 100 ppm. The monitors displayed and recorded measurements in real time, allowing logged data to be downloaded for analysis. The selected sampling interval was 60 s.
Bioaerosols sampling was carried out at a height of approximately 1 m from the ground, in the children’s breathing zone, in the center of each classroom. These measurements were performed after three series of tests for bacterial and fungal aerosols in the outdoor and indoor area. During each season, three independent samples were collected. Each day, three samples were collected between 10:00 and 13:00. The hours of sample collection were selected during five hours of compulsory preschool education (from 8:00 to 13:00). Each sample included six impaction stages with Petri dishes, resulting in a total of 432 Petri dishes (without blanks) containing biological material analysed during the study.
The bioaerosol samples were impacted onto different culture plates of 90 mm diameter. In order to determine the total number of heterotrophic bacteria, Tryptic Soy Agar (TSA) was used with cycloheximide added to inhibit fungal growth in the agar medium. Malt Extract Agar (MEA 2%) was applied for fungi, with chloramphenicol added to inhibit bacterial growth. Bacterial cultures were incubated at 36 ± 1 °C for 48 h, while fungal plates were incubated at 26 °C for five days. Following the incubation of material, colonies were enumerated and airborne concentrations of bacterial and fungal aerosols, in colony-forming units per cubic metre (CFU·m−3), were calculated for each impactor stage. The next step involved the identification of the collected bacteria, which took place in two stages. The first stage involved an analysis of morphological and microscopic colonies of grown cells stained with Gram. In the second stage, analytical profile index (API) biochemical tests (bioMerieux, Marcy-l’Etoile, France) were carried out, which allowed for the differentiation of the bacterial strains on the basis of their metabolic properties. Bacterial identification was based on morphology, Gram staining and endospore formation. The bacteria identification results are presented in supplementary material (Tables S1 and S2). 17 bacterial species from 8 genera were identified. The bacterial species from Staphylococcus (four species) and Micrococcus/Kocuria (two species) genera were predominant indoors. In the outdoor dominant genus was Bacillus (six species).
A quality control procedure was practised throughout the analyses to avoid any interference and minimize the risk of error, following the previous studies [32,33].
All statistical calculations, including univariate and multivariate analyses (comprising Pearson correlation coefficient, nonparametric Mann–Whitney U and Wilcoxon matched pairs tests) were performed using the statistical package Statistica 10 (StatSoft). The Mann–Whitney U test was used to analyze whether the total concentrations of bioaerosols differed between seasons and locations (either outdoors or inside the classrooms). Meanwhile, the Wilcoxon test was performed in order to compare the size distributions of bioaerosols between different seasons in the indoor air (for both the older and younger children’s classrooms). Throughout the study, a p value of <0.05 was considered to indicate statistical significance.
4. Results and Discussion
4.1. Total Concentration of Bacterial and Fungal Aerosols
Table 2 presents the average concentrations of bacterial and fungal aerosols collected in the indoor and outdoor air of rural nursery schools during the analyzed winter and spring seasons. Three samples were collected for each bioaerosol during both seasons, both inside and outside each location (outdoor—OUT; older children classrooms—O; younger children classrooms—Y). Generally, bacteria levels (258–2588 CFU·m−3) were higher than fungi levels (82–1549 CFU·m−3), except in the case of outdoor samples collected in spring. Additionally, during spring season a negative control sample was collected. The concentration of bacterial aerosol collected at 6:30 without children’s presence was in the range from 390 to 440 CFU·m−3, five times lower compared to results obtained during children’s activity. In the same way, the concentration of fungal aerosol was in the range from 330 to 520 CFU·m−3, obtained values were similar to the samples collected when the nursery schools were open. The results present that bacterial instead of fungal concentration depends on occupant’s activity. The small influence of human activity on fungal levels also confirmed Shin et al. [27]. The highest total concentration of bacterial (2588 CFU·m−3) and fungal (1549 CFU·m−3) aerosols was found during spring, respectively inside and outside the S building.

Table 2.
Average concentrations of total bacterial and fungal colony-forming units per cubic metre of air in two nursery schools (P, S) in winter and spring season; (OUT)—outdoor; (Y)—younger children classroom; (O)—older children classroom.
During the present study, the total concentrations of bacterial aerosols obtained in indoor air during the spring season were of a comparable level to those of the previous study conducted in urban nursery schools, which found levels of between 2545 and 2890 CFU·m−3. However, the concentrations recorded inside the urban classrooms during the winter season were in some cases almost five times higher [34] than the results obtained in the rural nursery schools. Other studies [35] concerning IAQ in two nursery schools in Bydgoszcz, conducted between April and February 2014, reported higher average concentrations of bacteria in the indoor air (3697 CFU·m−3, range: 1520–7780 CFU·m−3) and lower average concentrations in the outdoor air (137 CFU·m−3, range: 100–180 CFU·m−3). Research performed in Ankara, Turkey, underlined that, among indoor urban environments, the highest concentrations of total bacteria aerosols were observed in kindergartens, at 649 and 1462 CFU·m−3 in the winter and summer seasons, respectively [8]. Similar studies carried out in three schools in Portugal showed that the concentrations of bacteria in the schools’ indoor air was between 500 and 1600 CFU·m−3 [36]. Yang et al. [37] studied South Korean schools between July and December, and reported the average concentration of bacterial aerosol to be 1300 CFU·m−3, while the maximum concentration reached 4700 CFU·m−3. Concentrations of airborne bacteria found in homes in Upper Silesian were on average 1021 CFU·m−3, while the same measure was 300 CFU m−3 in offices [38]. In the sports hall of an elementary school in this same area, the concentration of airborne bacteria was found to be very high, reaching almost 5500 CFU·m−3 during the first class of the day and rapidly increasing with lesson hours, finally reaching a level higher than 12,000 CFU·m−3 [39]. The average concentration of fungal aerosol in the indoor air was 368 CFU·m−3 (range: 78–788 CFU·m−3), while in the outdoor air it was 623 CFU·m−3 (range: 65–1792 CFU·m−3). In Portuguese primary schools, a similar concentration range indoors of 16–1686 CFU·m−3, and a lower concentration range outdoors of 53–590 CFU·m−3 (mean values of 332 ± 274 CFU·m−3 and 224 ± 127 CFU·m−3, respectively) were reported [40]. Similar levels (36–2494 CFU·m−3) were reported by Pastuszka et al. [38] for non-moldy homes in multi-storey buildings during the summer in Poland, with a geometric mean concentration of 225 CFU·m−3. Similar overall concentrations of indoor fungi (235 CFU·m−3) were found by Hunter et al. in the UK houses [9]. Higher levels of indoor fungal concentrations were reported by Zuraimi et al. [41], finding 1424 CFU·m−3 during dry and 2930 CFU·m−3 during rainy weather in childcare centers in Singapore. However, in kindergartens located in Ankara, Turkey, lower indoor levels of fungal aerosols of 27–53 CFU·m−3 have been reported [15].
The variations in bacterial and fungal aerosol concentrations were found to be similar to those of the above-mentioned studies. However, our results cannot be directly compared to those environments that differ from preschools. Moreover, in Poland, the legislation governing microbiological standards for air pollution have not been developed and implemented. Since appropriate standards and guidelines do not yet exist, expertise must be sought and research must be conducted to determine contaminant concentrations and exposures that are acceptable. Górny et al. [42], in a wide review of propositions for non-industrial workplaces and public buildings, propose a total concentration of bacteria of 7000 CFU·m−3 as the upper limit for regulatory levels of bacterial aerosols. In other research [16] based on correlation analysis between health complaints and fungi concentrations, the authors suggest that a concentration of fungi in indoor air above 2000 CFU·m−3 be considered a serious risk factor for the health of the occupants. It can be seen that the concentration levels of airborne bioaerosols obtained in our study are below these proposed standards. However, research by the Occupational Health and Safety Research Institute Robert Sauvé (IRSST) indicates that, in the case of total airborne bacteria concentrations above 1000 CFU·m−3, possible microbial contamination justifies further investigation of the situation and an action requirement [43]. In the case of fungal species, the need for remedial action is highlighted for concentrations exceeding 500 CFU·m−3 as such levels may be indicative of building-related bioaerosol sources, poor ventilation rates, or overcrowding [36].
According to reported recommendations, our research on bioaerosols collected in rural nursery schools should focus mainly on the spring season. However, seasonal (winter-spring) variations between both nursery schools are also interesting. For fungal aerosols, the difference between the (P) and (S) sites differs significantly between the seasons (p < 0.01) while, for bacterial aerosols, the significant difference between concentration levels was observed during the winter season. In spring, the relation between both nursery schools is not statistically significant (Figure 2).
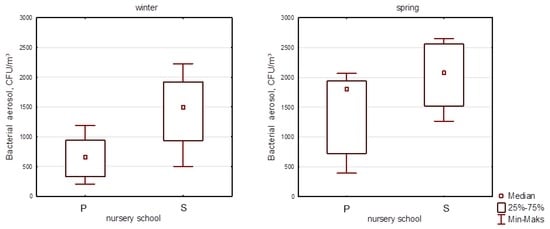
Figure 2.
Comparison of bacterial aerosol levels between rural sites (P) and (S) during winter and spring seasons.
The non-significant difference between the (P) and (S) sites during the spring season may point to the influence of a common parameter on the bacterial aerosol levels. For example, increases in humidity and temperature are both associated with significantly higher culturable bacteria and fungi concentrations in indoor air [13]. In our research, environmental parameters such as temperature, RH, and CO2 concentrations in both indoor and outdoor air during the sampling periods were measured to estimate the influence of these factors on bioaerosol levels. The most significant physical factors influencing viability of microorganisms are temperature and relative humidity (RH). Higher temperature and RH could promote the growth of bacteria and fungi [44,45]. This is confirmed by the higher concentration levels of bioaerosols obtained in the (S) building. The summary of environmental parameters for both spring and winter seasons is provided in Table 1. Significant correlations between RH and bioaerosol levels were found. For bacterial aerosol levels and fungal aerosol levels, the correlation coefficients (R) with indoor RH were, respectively, 0.94 and 0.77. As can be seen from the table, regardless of the season, the RH measured in the indoor air ranged from 25% to 47%, approximately, and especially during winter RH fell below the recommended comfort range of 30%–60% for indoor air [46]. Although low RH (<60%) is desirable for the prevention of mold formation, humidity levels that are too low (<30%) can cause sensations of dryness, irritation, and other health complaints, such as drying mucous membranes and increased risk of catching colds [8]. The other environmental parameters—temperature and CO2 concentrations—were not significantly correlated with bacterial or fungal aerosol levels (R < 0.5).
4.2. Indoor-Outdoor and Winter-Spring Relationships
During bioaerosol sampling for any indoor environment, outdoor bioaerosol sampling should be performed for comparison of indoor and outdoor source strengths. Comparison of indoor and outdoor bioaerosol levels can be a useful tool to indicate whether a proliferation site exists indoors [8]. The indoor-to-outdoor (I/O) ratio indicates the source of bioaerosols. If this ratio is >1.0, there is a difference between outdoor and indoor bioaerosol sources and the source exists in the indoor environment. The calculated average I/O ratios of bacterial and fungal aerosol levels according to sampling site and groups of children for both winter and spring seasons, representing parallel indoor and outdoor samples collected for each sampling day, are given in Table 2. As can be seen from Table 2, the results indicate between two and four times higher indoor concentrations of bacterial aerosols than outdoor samples. The average I/O ratio calculated for all indoor and outdoor bacteria concentrations for the winter season was 2.84, while the average I/O ratio for the spring season was 2.50. Since the I/O ratio was >1 in both of these sampling site groups, it can be concluded that the major sources of these bioaerosols are likely internal, such as building occupants (in this case children and their activities) as well as building materials that host microbiological growth (especially carpets).
When we classified all indoor and outdoor bacteria levels according to three different categories (outdoor, indoor older children and indoor younger children), the Mann–Whitney U test results showed that there was a significant difference between indoor and outdoor bacterial aerosols levels (p < 0.01), while the difference between levels of bacterial aerosols collected indoors in older versus younger children’s classrooms were statistically non-significant (p > 0.05). On the other hand, the indoor bacteria levels were moderately correlated with the corresponding outdoor levels (R > 0.87 for winter and >0.72 for spring), demonstrating that, in addition to children’s activities, outdoor infiltration could contribute to direct indoor transportation.
The I/O ratio for fungal aerosol levels varied between seasons, being <1 (0.43–0.70) in spring and >1 (1.10–1.55) in winter. Since the mean I/O ratio for fungi levels in the spring season is below 1.0 and, in the winter season, the concentrations in indoor environments are relatively low (<250 CFU·m−3), it can be concluded that there is no significant mold source in these indoor environments.
Analogously to bacteria levels, we classified all indoor and outdoor fungi levels according to three different categories (outdoor, indoor older children, and indoor younger children). The Mann–Whitney U test results here showed that there was a non-significant difference between fungal aerosols levels collected indoors (for both older and younger children’s classrooms) and outdoors (p > 0.05). In addition, the indoor fungi levels were highly correlated with the corresponding outdoor levels (R > 0.96 for winter and >0.95 for spring). This result could demonstrate the significant role of outdoor infiltration on indoor fungi levels. Nevertheless, the I/O ratio <1.0 during spring suggests stronger deposition on solid surfaces, or decomposition in the indoor air, rather than differences in the filtering of the ventilation air when crossing the building threshold.
It is generally accepted that microbiological concentration in the healthy buildings is similar to the corresponding outdoor values, which means that the I/O concentration ratio is close to 1. Comparing the obtained average values of I/O ratios with the research of Stryjakowska-Sekulska et al. and the reference therein [47], the indoor fungi levels in the tested rooms can be estimated to be relatively safe, in contrast to their poor air quality according to bacterial aerosol levels (I/O ≤ 1.5: good indoor air conditions; I/O = 1.5–2.0: regular; I/O > 2: poor).
Since meteorological conditions have substantial effects on both bioaerosol taxa and levels [8], investigating the seasonal variations of air quality in terms of bioaerosol content should be helpful to evaluate the measurement results accurately. Therefore, winter-to-spring (W/S) ratios were calculated for each bacterial and fungal concentration pair obtained from the data, measured in each sampling site for both winter and spring seasons. The average W/S ratio value was computed as 0.56 for pairs of bacteria levels and 0.20 for pairs of fungi levels. Since the average W/S ratio was below 1.00, we can conclude that winter bacteria levels were lower than spring levels for these study environments. According to Mann–Whitney U test results, a non-significant relationship was found between sample types (i.e., indoor and outdoor bacteria levels) measured in both seasons (p > 0.05). Additionally, for fungi levels, two statistically different pairs were recorded: W/S outdoor levels and W/S indoor levels (in both older and younger children’s classrooms). For these two data pairs, the outdoor fungi levels significantly differed between seasons (p < 0.05), while no significant association was found between winter fungi levels and spring fungi levels for both indoor environments (p > 0.05). A comparison with other studies regarding winter-to-spring [48] and winter-to-summer [8] ratios revealed that ratios lower than 1.0 were found in most studies, mainly due to meteorological conditions.
4.3. Size Distribution of Bacterial and Fungal Aerosol
The size distribution of airborne bacteria inside the studied nursery school (S) is presented in the supplementary material (Figure S1). It can be seen that the shape of the obtained size distribution is almost the same for all sites. The distributions are unimodal, with the peak located in the size range between 2.1 and 3.3 µm. Only the peak in Figure S1a is shifted into the 3.3–4.7 µm range. Similar results were obtained for homes in Katowice [49], which confirms that the studied bacterial aerosol indoors was dominated by fresh bacteria emitted from human organisms. It can be noted that the obtained size distributions of bacterial aerosol in the nursery school (S) do not significantly depend on the season (winter or spring), meaning there is a low exchange of air between the outdoor and indoor areas (poor ventilation). It is a different situation for the second nursery school (P), where, in the spring, a bimodal size distribution is recorded (Figure S3). The second peak, in the range 3.3–4.7 µm, indicates the influence of the migration indoors of outdoor bioaerosols containing a relatively higher amount of coarse bioaerosol particles in spring than in winter [50]. This result suggests that the ventilation rate is significantly higher in school (P) than in school (S).
The size distributions of fungal aerosols in both nursery schools in the spring are unimodal, with the peak for the particles having an aerodynamic diameter of between 3.3 and 4.7 µm, i.e., shifted towards coarser particles as compared to airborne bacteria (Figures S2 and S4). The same result was obtained for healthy homes in summer in Upper Silesia, Poland, while in winter the peak typically appeared in the 2.1–3.3 µm range [38]. It should be noted that all size distributions of fungal aerosols found in homes were unimodal [38], yet the results obtained in the studied nursery schools were much more complicated. In some cases, the size distribution was the same as in homes, but in other cases it was not, sometimes even showing a bimodal pattern (see Figure S4b,d). These differences reflect the significantly different dynamics of air movement inside the classrooms, strongly dependent on the physical activity of the children. For example, the resuspension of settled fungi-contained dust particles certainly plays a very important role in changing the primary (background) size distribution. However, additional studies are required to explain this process in detail.
It is common knowledge that the potential health risk caused by exposure to airborne bacteria and fungi is related to the concentrations of respirable airborne bacteria and fungi. The respirable fraction of bacteria and fungi is defined as the sum of the third stage (3.3–4.7 µm), fourth stage (2.1–3.3 µm), fifth stage (1.1–2.1 µm), and sixth stage (0.65–1.1 µm), with respect to the total concentration of bacteria or fungi [17]. The ratios of the respirable fraction of airborne bacteria and fungi concentrations were calculated in the 63%–80% range (Figure 3).
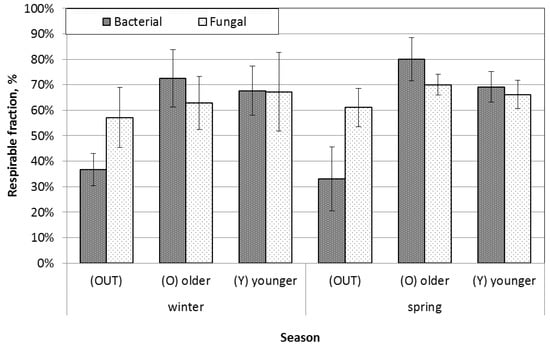
Figure 3.
Respirable fraction of bacterial and fungal aerosols.
The respirable fraction ratio of indoor airborne bacteria was higher than that of outdoor airborne bacteria. Based on the findings of Pastuszka et al. [38] that indoor contamination with airborne bacteria can be suspected if the indoor respirable/total ratio is higher than the outdoor ratio, the level of airborne bacteria contamination in the investigated nursery schools cannot be concluded to be safe. Considering the reports, however, that the ratio of respirable/total airborne bacteria inside the public buildings ranged between 30% and 60% [17,38], the range reaching 80% revealed through this study is likely to be a serious level. On the other hand, the range between 57% and 70% for airborne fungi was of much the same level as that of the outdoor concentration, which was lower than other reports that showed a range of 70%–80% [17,38]. Thus, we can conclude that the indoor air of the nursery school buildings investigated in this study was not contaminated with airborne fungi at a high level.
4.4. Characterization of Bacterial Aerosols
Four groups of viable bacteria were identified: Gram-positive cocci, non-sporing Gram-positive rods, sporing Gram-positive rods (family: Bacillaceae), and Gram-negative rods (Figure 4). Airborne Gram-positive bacteria were the most abundant, accounting for approximately 90% of the measured population. Gram-negative bacteria were present in less than 10% of outdoor samples in our study, while indoors the contribution of Gram-positive bacteria was 5% higher. Similar proportions were found for child day centers in Turkey [14].
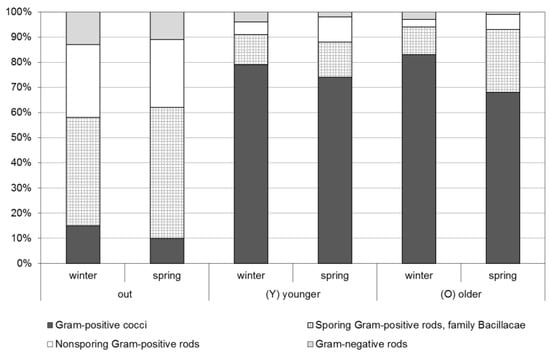
Figure 4.
The average percentage of main groups of bacteria isolated from the outdoor and indoor air during winter and spring seasons.
Gram-positive cocci were dominant in the indoor air, while Gram-positive rods were more dominant in the outdoor air (Figure 4). The results of bacterial species identification for aerosols in the outdoor environment revealed that the largest percentage relative to the total bacterial flora were Gram-positive bacilli-forming endospores; which accounted for 43% of the total during winter and 52% in the spring. Aerobic and facultative anaerobic Gram-positive bacilli are commonly found in soil and water habitats, and in much of the normal skin and mucous membrane flora of humans and various animals. The virulence of Gram-positive bacilli is highly variable. Many have the potential to be opportunistic pathogens, capable of producing disease only in persons with compromised host resistance [14], characteristic especially of small children. The second most frequently isolated group of bacteria was Gram-positive non-sporing rods, at 29% and 27% in winter and spring, respectively.
In indoor air, Gram-positive cocci were dominant during both seasons, which coheres with other research [14,18,32,38]. Gram-positive bacteria, particularly the cocci, are microorganisms that are widespread in nature and can be isolated either from the environment or as commensal inhabitants of the skin, mucous membranes, and other body sites in humans and animals [3,12,14].
Children’s activity is normally high and, thus, Gram-positive cocci can be transmitted to the air from their bodies and respiratory tracts. With regard to bacterial groups, some bacteria isolated from all the sampling sites were identified. The results of qualitative analysis obtained in the rural nursery schools are comparable to those obtained in studies conducted in urban nursery schools in Gliwice, as well as in schools in Lublin, Poland [18,32]. The dominant bacteria genera/species isolated from outdoor and indoor air are presented in the supplementary material (Tables S1 and S2). In outdoor air, the most isolated were Gram-positive rods forming endospores (34%–56%). In indoor air samples, the most abundant were Gram-positive cocci (66%–94%): Micrococcus spp. and Staphylococcus spp. Micrococcus is generally thought to be a harmless bacterium, but Staphylococci are indicators of the severity of air pollution, and their presence may indicate the further presence of pathogenic bacteria [35]. Similar results were also obtained by Kim et al. [51] in the indoor environment of elementary schools in Ulsan, South Korea; they found that 84% of identified bacteria were Gram-positive, and Micrococcus spp. was the most abundant group with 61% of tested isolates. The study carried out by Stryjakowska-Sekulska et al. [47] in various rooms of university buildings in Poznań, Poland, also showed that the predominant airborne bacteria were Gram-positive cocci.
Generally, in the air of the studied nursery school, exposure to bacterial aerosol does not constitute an immediate risk of any acute health effects; however, the long-term inhalation of such high doses of airborne bacteria can cause some adverse health effects, especially among vulnerable persons. Such persons may have greater susceptibility to diseases of the upper respiratory tract and allergic symptoms such as headaches, watery eyes, itchy skin, coughing, etc. [52]. Although the level of the microbial pollution inside the studied kindergarten should be classified as safe, some action to improve indoor air quality is needed. It can be expected that if some sick children are present in the kindergarten the level of pathogenic bacteria will rapidly elevate in this building, especially in the rooms with younger children. Therefore, an increase of the air exchange rate is strongly recommended. Certainly, such a recommendation can be addressed to a huge number of the preschools in Poland.
4.5. Nursery School Exposure Dose (NSED)
Interest in exposure to bioaerosols has increased over the last few decades because they are associated with a wide range of health effects with major public health impacts, including infection by diseases, acute toxic effects, allergies, and cancer. However, with regard only to infectious diseases, no clear correlation has been found between concentrations of culturable microorganisms in the air and infection [53]. One reason for this could be that infection should be correlated with the dose rather than the concentration. Although dose-response relationships still have not been established for most biological agents, the bioaerosols expert network concluded recently that the measurement of bioaerosols should be performed according to a protocol representative of exposure patterns and duration, which means—in fact—the dose [7,54]. This is not an entirely new idea. For example, in 1994 Alekseev et al. [55] reported their study on the dynamics of P. pseudomallei infection and its antigens in white rats after aerogenic infection with a dose equal to 941 CFU. Pastuszka [21] estimated that children living in healthy homes in Upper Silesia, Poland, inhaled daily doses of 1780 and 560 CFU of airborne bacteria and fungi, respectively. Therefore, estimating the dose of culturable bacteria and fungi inhaled by those in the nursery schools of the present study might well be important for possible future exposure analysis. We think, however, that assessment of the risk of infection for children and staff depends not only on the number of inhaled culturable bacteria but also on the individual’s body mass. According to this hypothesis, we calculated the doses of culturable bacteria and fungi per mean body weight [32].
This NSED has been calculated on the basis of the EPA’s Child-Specific Exposure Factors Handbook [56] and other publications [57,58,59]. Total concentration levels were used in the calculations of the absorbed dose of airborne bacteria, although particles of an aerodynamic diameter >10 µm usually cannot be inhaled. Based on the measured concentrations of bacterial aerosols—as well as additional data, including children and staff activity patterns and general characteristics, received from questionnaires—the doses inhaled by adults (in this case nursery staff), older children, and younger children were estimated. The calculations were based on the following equation:
where, NSED: nursery school exposure dose for indoor environment, CFU·kg −1; C: bacterial aerosol concentration, CFU·m−3; IEF: indoor exposure fraction—hours spent over a day in nursery school, concerning diverse activity patterns hour (in sum on average: children 7 h, adults 8 h); IR: inhalation rate coefficient, characteristic for selected activity levels. It was assumed that the weighted average of IR was 0.72 m3·h−1 for children and 0.76 m3·h−1 for staff [60] BW: mean body weight, kg. The measured mean body weight was equal to 23.5 kg and 15.8 kg for older (O) and younger (Y) children, respectively, while for the staff it was 59 kg.
The detailed values of the Inhalation Rate (IR) and Indoor Exposure Fraction (IEF) obtained in our study, dependent on the children and staff’s physical activity levels, are contained in Table 3, while in Table 4 the calculated results of the inhaled doses of culturable airborne bacteria are shown. Generally, it can be concluded that the higher dose in the case of children as compared to staff is attributable to the former’s greater inhalation of air relative to body weight.

Table 3.
Dependence of the Inhalation Rate (IR) and Indoor Exposure Fraction (IEF) on children and staff’s physical activity levels.

Table 4.
Calculated exposure dose (NSED) of bacterial and fungal aerosols inhaled by younger (Y) and older (O) children, as well as by nursery school staff.
It can be seen that both children and staff inhaled higher doses of bacterial and fungal aerosols in spring. However, the most important point to note is that younger children inhaled significantly higher NSED than their older counterparts. A similar study [32] conducted in an urban kindergarten in Gliwice, Upper Silesia, showed higher values of NSED of bacterial aerosol inhaled by children (545.8–929.9 CFU·kg−1) and staff (272.6–331.9 CFU·kg−1). Although the bacterial dose inhaled by the staff in the studied kindergarten in spring is comparable to the dose absorbed by adults inside apartments in Katowice, Upper Silesia, Poland (175.4 CFU·kg−1) [34], their NSED is three times lower than the dose inhaled by younger children. This could be one of the reasons why children, especially those who are starting their preschool education, are frequently sick—that adverse health effects strongly depend on the absorbed dose of air pollutants. Additionally, infectious diseases spread faster among children than among adults. In this age group, there is no proper awareness of the importance of hygiene in the prevention of infection and, in terms of preschool, children are often only in the process of learning good hygiene. Inadequate hand-washing, direct contact during games, and being in constant motion aid the spread of bacterial infections, which increases the risk of exposing educators to harmful microorganisms.
5. Conclusions
To our knowledge, this is the first study in Poland that investigated the levels and size distribution of airborne bacteria and fungi in indoor air in rural nursery schools. The data showed that bacteria and fungi levels varied between seasons, with spring concentrations being higher than those in winter. The bacteria concentrations varied in the range from 258 to 2588 CFU·m−3, while fungi levels varied from 82 to 1549 CFU·m−3. The indoor bacteria levels were between two and three times higher than the outdoor levels, while the I/O ratio for fungi was >1 in winter and <1 in spring.
The size distribution of bacterial and fungal aerosols inside the classrooms indicates that smaller particles are more prevalent in indoor air. The highest number of culturable bacteria was isolated for particles <4.7 µm, which is respirable and thus has the potential to be deposited either in the tracheal, bronchial, or alveolar regions of the lungs. The prevalence of smaller size ranges indicated higher concentrations of individual bacterial cells, which could be due to higher occupancy and poor ventilation in nursery schools.
The level of NSED for children is up to three times higher than that for staff. The highest doses of bacterial and fungal aerosols were inhaled by younger children (Y), who absorbed between 327.4 and 705.7 culturable bacteria counts per kg of body weight, and between 31.1 and 224.6 fungi counts per kg of body weight. The higher NSED of younger children is connected to their higher inhalation rate relative to their body weight.
The dominant airborne bacteria species detected in outdoor air differed from those in indoor air. Outdoor air was dominated by Bacillus cereus and Bacillus subtilis, while indoor air was dominated by Micrococcus spp. and Staphylococcus chromogens.
Further, knowledge regarding airborne bioaerosols allows us to better understand the prevalence and ecology of indoor airborne aerosols, which may be useful in the management and prevention of both long- and short-term health problems for children and teachers. In addition, the data gathered demonstrated that indoor humidity levels were correlated with indoor bacteria and fungi levels. The results obtained may serve as a reference for future assessments and provide a useful contribution to: (1) policy reviews; (2) implementation of preventative public health programs; and (3) formulation of recommendations aimed at providing healthier preschool environments.
Supplementary Materials
The following are available online at http://www.mdpi.com/2073-4433/7/11/142/s1, Figure S1: Comparison of size distribution of bacterial aerosols in classrooms of younger and older children during winter and spring seasons in nursery school (S). (a) Classroom of younger children-spring, (b) Classroom of younger children-winter, (c) Classroom of older children-spring, (d) Classroom of older children-winter, Figure S2: Comparison of size distribution of fungal aerosols in classrooms of younger and older children during winter and spring seasons in nursery school (S). (a) Classroom of younger children-spring, (b) Classroom of younger children-winter, (c) Classroom of older children-spring, (d) Classroom of older children-winter, Figure S3: Comparison of size distribution of bacterial aerosols in classrooms of younger and older children during winter and spring seasons in nursery school (P). (a) Classroom of younger children-spring, (b) Classroom of younger children-winter, (c) Classroom of older children-spring, (d) Classroom of older children-winter, Figure S4: Comparison of size distribution of fungal aerosols in classrooms of younger and older children during winter and spring seasons in nursery school (P). (a) Classroom of younger children-spring, (b) Classroom of younger children-winter, (c) Classroom of older children-spring, (d) Classroom of older children-winter, Table S1: Viable bacterial genera identified outdoors and indoors nursery school (S) during winter and spring seasons, Table S2: Viable bacterial genera identified outdoors and indoors nursery school (P) during winter and spring seasons.
Acknowledgments
This work was supported by Teacher Benefit Grant KIC InnoEnergy Master School. The results used in this paper have received funding from the Polish-Norwegian Research Program operated by the National Center for Research and Development under the Norwegian Financial Mechanism 2009–2014 in the frame of Project Contract No Pol Nor/210247/20/2013. The authors would like to thank the principals and staff of the nursery schools that participated in the study for their support.
Author Contributions
Ewa Brągoszewska performed the experiments and drafted the manuscript. Anna Mainka designed the study and prepared the final manuscript. Jozef S. Pastuszka helped with the data analysis.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kotzias, D. Indoor air and human exposure assessment—Needs and approaches. Exp. Toxicol. Pathol. 2005, 57, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Yassin, M.F.; Almouqatea, S. Assessment of airborne bacteria and fungi in an indoor and outdoor environment. Int. J. Environ. Sci. Technol. 2010, 7, 535–544. [Google Scholar] [CrossRef]
- D’Arcy, N.; Canales, M.; Spratt, D.A. Healthy schools: Standardisation of culturing methods for seeking airborne pathogens in bioaerosols emitted from human sources. Aerobiologia (Bologna) 2012, 28, 413–422. [Google Scholar] [CrossRef]
- Demirel, G.; Özden, Ö.; Döǧeroǧlu, T.; Gaga, E.O. Personal exposure of primary school children to BTEX, NO2 and ozone in Eskişehir, Turkey: Relationship with indoor/outdoor concentrations and risk assessment. Sci. Total Environ. 2014, 473–474, 537–548. [Google Scholar] [CrossRef] [PubMed]
- Yoon, C.; Lee, K.; Park, D. Indoor air quality differences between urban and rural preschools in Korea. Environ. Sci. Pollut. Res. 2011, 18, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Patelarou, E.; Tzanakis, N.; Kelly, F.J. Exposure to Indoor Pollutants and Wheeze and Asthma Development during Early Childhood. Int. J. Environ. Res. Public Health 2015, 12, 3993–4017. [Google Scholar] [CrossRef] [PubMed]
- Walser, S.M.; Gerstner, D.G.; Brenner, B.; Bünger, J.; Eikmann, T.; Janssen, B.; Kolb, S.; Kolk, A.; Nowak, D.; Raulf, M.; et al. Evaluation of exposure–response relationships for health effects of microbial bioaerosols—A systematic review. Int. J. Hyg. Environ. Health 2015, 218, 577–589. [Google Scholar] [CrossRef] [PubMed]
- Mentese, S.; Rad, A.Y.; Arisoy, M.; Gullu, G. Seasonal and Spatial variations of bioaerosols in indoor urban environments, Ankara, Turkey. Indoor Built Environ. 2012, 21, 797–810. [Google Scholar] [CrossRef]
- Nasir, Z.A.; Colbeck, I. Assessment of bacterial and fungal aerosol in different residential settings. Water Air Soil Pollut. 2010, 211, 367–377. [Google Scholar] [CrossRef]
- Fabian, M.P.; Miller, S.L.; Reponen, T.; Hernandez, M.T. Ambient bioaerosol indices for indoor air quality assessments of flood reclamation. J. Aerosol Sci. 2005, 36, 763–783. [Google Scholar] [CrossRef]
- Nazaroff, W.W. Indoor bioaerosol dynamics. Indoor Air 2015, 26, 61–78. [Google Scholar] [CrossRef] [PubMed]
- Hospodsky, D.; Qian, J.; Nazaroff, W.W.; Yamamoto, N.; Bibby, K.; Rismani-Yazdi, H.; Peccia, J. Human occupancy as a source of indoor airborne bacteria. PLoS ONE 2012, 7, e34867. [Google Scholar] [CrossRef] [PubMed]
- Moon, K.W.; Huh, E.H.; Jeong, H.C. Seasonal evaluation of bioaerosols from indoor air of residential apartments within the metropolitan area in South Korea. Environ. Monit. Assess. 2014, 186, 2111–2120. [Google Scholar] [CrossRef] [PubMed]
- Aydogdu, H.; Asan, A.; Tatman Otkun, M. Indoor and outdoor airborne bacteria in child day-care centers in Edirne City (Turkey), seasonal distribution and influence of meteorological factors. Environ. Monit. Assess. 2010, 164, 53–66. [Google Scholar] [CrossRef] [PubMed]
- Menteşe, S.; Arisoy, M.; Rad, A.Y.; Güllü, G. Bacteria and fungi levels in various indoor and outdoor environments in Ankara, Turkey. Clean Soil Air Water 2009, 37, 487–493. [Google Scholar] [CrossRef]
- Górny, R.L.; Dutkiewicz, J. Bacterial and fungal aerosols in indoor environment in central and eastern European countries. Ann. Agric. Environ. Med. 2002, 9, 17–23. [Google Scholar] [PubMed]
- Kim, K.Y.; Kim, C.N. Airborne microbiological characteristics in public buildings of Korea. Build. Environ. 2007, 42, 2188–2196. [Google Scholar] [CrossRef]
- Dumała, S.M.; Dudzińska, M.R. Microbiological indoor air quality in Polish schools. Annu. Set Environ. Prot. 2013, 15, 231–244. [Google Scholar]
- Canha, N.; Almeida, S.M.; do Carmo Freitas, M.; Wolterbeek, H.T. Assessment of bioaerosols in urban and rural primary schools using passive and active sampling methodologies. Arch. Environ. Prot. 2015, 41, 11–22. [Google Scholar] [CrossRef]
- Hospodsky, D.; Yamamoto, N.; Nazaroff, W.W.; Miller, D.; Gorthala, S.; Peccia, J. Characterizing airborne fungal and bacterial concentrations and emission rates in six occupied children’s classrooms. Indoor Air 2015, 25, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Pastuszka, J.S. Exposure of the General Population Living in Upper Silesia Industrial Zone to the Particulate, Fibrous and Biological (Bacteria and Fungi) Aerosols; Wroclaw Technical University: Wroclaw, Poland, 2001. (In Polish) [Google Scholar]
- Pastuszka, J.S.; Marchwińska-Wyrwał, E.; Wlazło, A. Bacterial aerosol in Silesian hospitals: Preliminary results. Pol. J. Environ. Stud. 2005, 14, 883–890. [Google Scholar]
- Nasir, Z.A.; Mula, V.; Stokoe, J.; Colbeck, I.; Loeffler, M. Evaluation of total concentration and size distribution of bacterial and fungal aerosol in healthcare built environments. Indoor Built Environ. 2015, 24, 269–279. [Google Scholar] [CrossRef]
- Prussin, A.J.; Vikram, A.; Bibby, K.J.; Marr, L.C. Seasonal dynamics of the airborne bacterial community and selected viruses in a children’s daycare center. PLoS ONE 2016, 11, e0151004. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.; Tin, S.; Kelley, S.T. Culture-independent analysis of bacterial diversity in a child-care facility. BMC Microbiol. 2007, 7, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Andersson, A.M.; Weiss, N.; Rainey, F.; Salkinoja-Salonen, M.S. Dust-borne bacteria in animal sheds, schools and children’s day care centers. J. Appl. Microbiol. 1999, 86, 622–634. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.-K.; Kim, J.; Ha, S.; Oh, H.-S.; Chun, J.; Sohn, J.; Yi, H. Metagenomic insights into the bioaerosols in the indoor and outdoor environments of childcare facilities. PLoS ONE 2015, 10, e0126960. [Google Scholar] [CrossRef] [PubMed]
- Cyprowski, M.; Buczyńska, A.; Szadkowska-Stańczyk, I. Indoor allergens in settled dust from kindergartens in city of Łódź, Poland. Int. J. Occup. Med. Environ. Health 2013, 26, 890–899. [Google Scholar] [CrossRef] [PubMed]
- Górny, R.; Gołofit-Szymczak, M.; Stobnicka, A. Biological agents in kindergartens [in Polish Szkodliwe czynniki biologiczne w przedszkolach]. Bezpieczeństwo Pracy Nauka i Praktyka 2014, 2, 16–20. [Google Scholar]
- Deng, W.; Chai, Y.; Lin, H.; So, W.W.M.; Ho, K.W.K.; Tsui, A.K.Y.; Wong, R.K.S. Distribution of bacteria in inhalable particles and its implications for health risks in kindergarten children in Hong Kong. Atmos. Environ. 2016, 128, 268–275. [Google Scholar] [CrossRef]
- Mainka, A.; Brągoszewska, E.; Kozielska, B.; Pastuszka, J.S.; Zajusz-Zubek, E. Indoor air quality in urban nursery schools in Gliwice, Poland: Analysis of the case study. Atmos. Pollut. Res. 2015, 6, 1098–1104. [Google Scholar] [CrossRef]
- Brągoszewska, E.; Mainka, A.; Pastuszka, J.S. Bacterial aerosols in an urban nursery school in Gliwice, Poland: A case study. Aerobiologia (Bologna) 2016, 32, 469–480. [Google Scholar] [CrossRef]
- Mainka, A.; Zajusz-Zubek, E. Indoor Air quality in urban and rural preschools in Upper Silesia, Poland: Particulate Matter and carbon dioxide. Int. J. Environ. Res. Public Health 2015, 12, 7697–7711. [Google Scholar] [CrossRef] [PubMed]
- Brągoszewska, E. Bacterial Aerosol Occuring in the Atmospheric Air in Gliwice and Its Share of the Total Human Exposure to the Bacteria Absorbed by Inhalation. Ph.D. Thesis, Silesian University of Technology, Gliwice, Poland, 2014. [Google Scholar]
- Kubera, Ł.; Studzińska, J.; Dokładna, W.; Małecka-Adamowicz, M.; Donderski, W. Microbiological air quality in some kindergartens and antibiotic resistance of bactaria of the Staphylococcus spp. genus. Med. Pr. 2015, 66, 49–56. [Google Scholar] [PubMed]
- Pegas, P.N.; Evtyugina, M.G.; Alves, C.A.; Nunes, T.; Cerqueira, M.; Franchi, M.; Pio, C.; Almeida, S.M.; Freitas, M.C. Outdoor/indoor air quality in primary schools in Lisbon: A preeliminary study. Quim. Nova 2010, 33, 1145–1149. [Google Scholar] [CrossRef]
- Yang, W.; Sohn, J.; Kim, J.; Son, B.; Park, J. Indoor air quality investigation according to age of the school buildings in Korea. J. Environ. Manag. 2009, 90, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Pastuszka, J.S.; Kyaw Tha Paw, U.; Lis, D.O.; Wlazło, A.; Ulfig, K. Bacterial and fungal aerosol in indoor environment in Upper Silesia, Poland. Atmos. Environ. 2000, 34, 3833–3842. [Google Scholar] [CrossRef]
- Pastuszka, J.S.; Wlazło, A.; Łudzeń-Izbińska, B.; Pastuszka, K. Bacterial and fungal aerosol in the school sport hall. Ochrona Powietrza i Problemy Odpadów 2004, 38, 62–66. (In Polish) [Google Scholar]
- Madureira, J.; Pereira, C.; Paciência, I.; Teixeira, J.P.; de Oliveira Fernandes, E. Identification and levels of airborne fungi in Portuguese primary schools. J. Toxicol. Environ. Health A 2014, 77, 816–826. [Google Scholar] [CrossRef] [PubMed]
- Zuraimi, M.S.; Fang, L.; Tan, T.K.; Chew, F.T.; Tham, K.W. Airborne fungi in low and high allergic prevalence child care centers. Atmos. Environ. 2009, 43, 2391–2400. [Google Scholar] [CrossRef]
- Górny, R.; Cyprowski, M.; Ławniczek-Wałczyk, A.; Gołofit-Szymczak, M.; Zapór, L. Biohazards in the indoor environment-a role for threshold limit values in exposure assessment. In Management of Indoor Air Quality; Dudzińska, M.R., Ed.; Taylor & Francis Group CRC Press: London, UK, 2011; pp. 1–20. [Google Scholar]
- Goyer, N.; Lavoie, J.; Lazure, L.; Marchand, G. Bioaerosols in the Workplace: Evaluations, Control and Prevention Guide; IRSST, Occupational Health and Safety Research Institute Robert Sauvé: Montréal, QC, Canada, 2001. [Google Scholar]
- Zhong, X.; Qi, J.; Li, H.; Dong, L.; Gao, D. Seasonal distribution of microbial activity in bioaerosols in the outdoor environment of the Qingdao coastal region. Atmos. Environ. 2016, 140, 506–513. [Google Scholar] [CrossRef]
- Wu, Y.H.; Chan, C.C.; Chew, G.L.; Shih, P.W.; Lee, C.T.; Chao, H.J. Meteorological factors and ambient bacterial levels in a subtropical urban environment. Int. J. Biometeorol. 2012, 56, 1001–1009. [Google Scholar] [CrossRef] [PubMed]
- ASHRAE. Standard 62-1999 Ventilation for Acceptable Indoor Air Quality; American Society of Heating, Refrigerating and Air-Conditioning Engineers, Inc.: Atlanta, GA, USA, 1999. [Google Scholar]
- Stryjakowska-Sekulska, M.; Piotraszewska-Pajak, A.; Szyszka, A.; Nowicki, M.; Filipiak, M. Microbiological quality of indoor air in university rooms. Pol. J. Environ. Stud. 2007, 16, 623–632. [Google Scholar]
- Wang, Y.F.; Wang, C.H.; Hsu, K.L. Size and seasonal distributions of airborne bioaerosols in commuting trains. Atmos. Environ. 2010, 44, 4331–4338. [Google Scholar] [CrossRef]
- Brągoszewska, E.; Świercz, M.; Pastuszka, J.S. Charakterystyka aerozolu bakteryjnego występującego w powietrzu pomieszczeń mieszkalnych oraz w powietrzu atmosferycznym wybranej dzielnicy Katowic. In Inżynieria i Ochrona Powietrza; Kuropka, J., Musialik-Piotrowska, A., Eds.; Oficyna Wydawnicza Politechniki Wrocławskiej: Wrocław, Poland, 2014; pp. 89–97. [Google Scholar]
- Brągoszewska, E.; Kowal, A.; Pastuszka, J.S. Bacterial aerosol occurring in atmospheric air in Gliwice, Upper Silesia, Poland. Archit. Civ. Eng. Environ. 2013, 4, 61–66. [Google Scholar]
- Kim, N.Y.; Kim, Y.R.; Kim, M.K.; Cho, D.W.; Kim, J. Isolation and characterization of airborne bacteria and fungi in indoor environment of elementary schools. Korean J. Microbiol. 2007, 43, 193–200. [Google Scholar]
- Daisey, J.M.; Angell, W.J.; Apte, M.G. Indoor air quality, ventilation and health symptoms in schools: An analysis of existing information. Indoor Air 2003, 13, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Henningson, E.W.; Lundquist, M.; Larsson, E.; Sandström, G.; Forsman, M. A comparative study of different methods to determine the total number and the survival ratio of bacteria in aerobiological samples. J. Aerosol Sci. 1997, 28, 459–469. [Google Scholar] [CrossRef]
- Douwes, J.; Thorne, P.; Pearce, N.; Heederik, D. Bioaerosol Health effects and exposure assessment: progress and prospects. Ann. Occup. Hyg. 2003, 47, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Alekseev, V.V.; Tikhonov, N.G.; Savchenko, S.T.; Kurilov, V.Y.; Vjazmina, T.N.; Yakovlev, A.T.; Denisov, I.I.; Popov, S.F. Experimental bacterial aerosols: Some physical and biological (pathogenic) properties. J. Aerosol Sci. 1994, 25, 1413–1423. [Google Scholar] [CrossRef]
- US Environmental Protection Agency. Child-Specific Exposure Factors Handbook; Environmental Protection Agency: Washington, DC, USA, 2002.
- Johnson-Restrepo, B.; Kannan, K. An assessment of sources and pathways of human exposure to polybrominated diphenyl ethers in the United States. Chemosphere 2009, 76, 542–548. [Google Scholar] [CrossRef] [PubMed]
- Ott, W. Exposure Analysis; Taylor & Francis Group CRC Press: London, UK, 2006. [Google Scholar]
- US Environmental Protection Agency. Risk Assessment: “Supplemental Guidance for Dermal Risk Assessment”, Part E of Risk Assessment Guidance for Superfund, Human Health Evaluation Manual; US Environmental Protection Agency: Washington, DC, USA, 2004.
- US Environmental Protection Agency. Exposure Factors Handbook; US Environmental Protection Agency: Washington, DC, USA, 2011.
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).