Abstract
Spherical acoustic resonant cavities have been increasingly reported in photoacoustic spectroscopy due to their small volume and enhanced effective gas absorption path length. For further reducing the acoustic cavity volume and exploiting broadband LED as a light source, this paper reports a low-cost, LED-based photoacoustic gas-sensing system using a hemispherical acoustic resonant (HAR) cavity with a radius of 15 mm and a volume of 7.07 mL. The placement of both the excitation light source and transducer, as important elements in photoacoustic spectroscopy, was systematically optimized for improving the generation efficient of photoacoustic signal. The frequency response of the HAR cavity was thoroughly characterized for exploring an optimal operation frequency of the light source. Through positional and frequency optimization, the developed low-cost, LED-based photoacoustic spectrophone realized highly sensitive measurements of hydrocarbon gases with measurement sensitivities of 111.6 ppm (3σ) for isobutane, 140.1 ppm (3σ) for propane, and 866.4 ppm (3σ) for ethylene at an integration time of 1 s. These results demonstrate the strong potential of low-cost, LED-HAR-based PA-sensing systems in the field of gas sensing for widespread deployment in distributed sensor networks and atmospheric monitoring platforms.
1. Introduction
Highly flammable and explosive hydrocarbons gases, such as isobutane, propane, and ethylene, serve as essential fuels and critical raw materials across the petrochemical, chemical manufacturing, and pharmaceutical industries [1,2,3,4]. Isobutane is widely used as an efficient refrigerant, offering notable advantages over traditional Freon-based refrigerants [1]. Propane functions both as a common industrial fuel and as an anesthetic in surgery [2]. Ethylene represents a fundamental feedstock in the synthesis of various industrial products, including synthetic fibers, plastics, and synthetic rubber [3]. Consequently, continuous and reliable monitoring of these hydrocarbon gas concentrations, particularly within chemical plant environments, is crucial for ensuring operational safety, process control, and emission management.
Photoacoustic (PA) spectroscopy, as a common optical absorption spectroscopy, is a zero-background approach with advantages of simple structure, low price, and high sensitivity [5,6,7]. PA spectroscopy is based on the detection of acoustic signals generated from the light absorption of photonic energy by the target species [8,9]. An acoustic resonator is a critical component for amplifying the generated sound signal in the PA gas-sensing system. Common resonator geometries include cylindrical resonance chambers [9,10,11], differential Holmhertz resonators [12], and spherical resonators [13]. Cylindrical acoustic resonators are widely used, as they are closely matched to the axisymmetric beam with the acoustic field, enabling high photoacoustic signal gain [14]. Differential Helmholtz resonators employ two identical resonators for eliminating background noise, making them particularly suitable for high-sensitivity trace gas detection [15]. More recently, spherical resonators have attracted growing interest, owing to their ability to provide long effective gas absorption path lengths and high-quality factor [16,17,18]. Li et al. developed a spherical cell made of high-reflectance polytetrafluoroethylene for increasing optical path, achieving a CO2 measurement sensitivity of 0.35 ppm with an integration time of 865 s [16]. Wei et al. demonstrated simultaneous multi-gas detection of water vapor, carbon dioxide, and methane by exploiting multiple resonant modes of a spherical cavity [17].
While most previous studies utilizing spherical resonators have employed lasers as excitation sources, light-emitting diodes (LEDs) remain relatively unexplored for PA applications, mainly due to their poor spatial coherence and beam quality. However, LED offers compelling benefits, such as low cost, long lifetime, and broad emission bands. A notable contribution by Scholz et al. demonstrated CO2 sensing using an LED-powered PA system with a half-ellipsoid resonator operating at a modulation frequency of 300 Hz [18]. To further advance resonator miniaturization and fully leverage the resonant characteristics of spherical structures, this work introduces a hemispherical acoustic resonator excited by a broadband LED source.
This paper presents a PA spectrophone based on a light-emitting diode (LED) and a hemispherical acoustic resonant (HAR) cavity for the detection of hydrocarbon gases. To maximize the photoacoustic signal generation efficiency, the positions of both the optical source and the acoustic transducer within the HAR cavity were systematically optimized, as their placement critically influences the excitation and detection of the acoustic signal. The frequency response of the resonator was thoroughly characterized to identify the optimal modulation frequency. Time-series measurements of isobutane, propane, and ethylene were conducted to demonstrate the performance and validation of the LED-based photoacoustic sensor.
2. Measurement Principle
In photoacoustic spectroscopy, the absorption of intensity-modulated optical radiation by the target species produces a periodic thermal wave through non-radiative relaxation processes. This thermal wave propagates into the surrounding medium, inducing local pressure oscillations. When the modulation frequency coincides with an eigenfrequency of the acoustic resonator, the pressure fluctuations excite a corresponding resonant mode, resulting in a standing acoustic wave. The amplitude of this wave is directly proportional to the absorber concentration. The geometry and dimensions of the resonator define the spatial distribution of acoustic modes and the fundamental to higher-order resonance frequencies. In a standard spherical resonant cavity, the amplitude Aj(ω) of the resonant mode is expressed as follows [19,20]:
where ω is the angular frequency of modulation; ωj is the angular frequency of the j = (n, l, m) eigenmode component; n, l, and m are the order of the radial, theta, and ϕ angle direction acoustic modes. Qj is the quality factor, and Vres is the resonator volume. The integral ʃpj(r)g(r)dV, which is taken over the cell volume Vres, describes the spatial overlap between the propagating laser beam and the pressure distribution of the jth acoustic eigenmode of the resonator. c is the speed of sound; η is the ratio of the specific heat at constant pressure to the specific heat at constant volume, N is the number concentration of the absorber, σ is the absorption cross-section, L is the PA resonator optical length, and P is the light power.
The placement of the acoustic transducer is a critical parameter influencing the signal amplitude in a PA-sensing system, as it must be located within the antinode of the targeted acoustic resonant mode. In standard spherical resonators, the first radial mode is often preferentially excited due to its low viscous damping losses [17]. However, positioning the transducer at the center of the sphere, where this mode exhibits its pressure antinode, can perturb the acoustic field distribution [19]. Alternative mounting strategies have been proposed to minimize such interference, including integrating the transducer at the inner surface of the cavity or coupling it via a resonant tube attached to the sphere’s exterior [16,20].
In the present study employing a HAR cell, the transducer location was experimentally evaluated between two candidate positions: the center of the hemispherical base and the center of the arc top. Furthermore, a broadband LED served as the excitation source, and its optimal placement was also empirically determined to maximize the overlap between the excitation volume and the acoustic mode shape, thereby enhancing the generated photoacoustic signal.
3. Experimental Setup
Figure 1a shows the schematic diagram of the designed HAR cavity with a diameter of 30 mm and a volume of 7.07 mL, which is much lower than the commonly used H-typed cylindrical acoustic cavity with two buffer chambers [21]. The light source location is experimentally determined after assessing different positions marked as LED-A, LED-B, and LED-C in Figure 1. The transducer is installed at the arc top of the HAR cell after optimization. The inlet and outlet of the resonator are located at the bottom. An LED (Hamamatsu Photonics K.K., Hamamatsu, Japan, L15893-0330M) emitting at 3.4 μm is employed as an excitation source with a full width at half maxima of 400 nm and a light power of 1.9 mW. The divergence angle is 100°, fully enabling the cavity volume of the excitation light radiation. Hydrocarbon gases exhibit strong infrared absorption around 3.4 μm, primarily due to C–H bond stretching vibrations [22]. The 3.4 μm broadband LED light source can effectively interact with these hydrocarbon gases to generate an effective infrared PA signal.
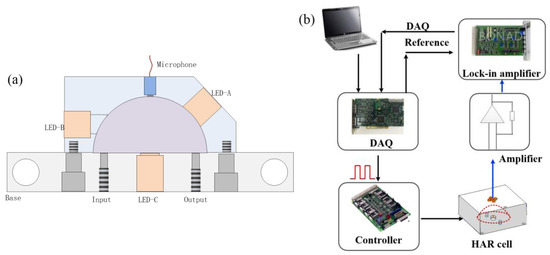
Figure 1.
(a) Schematic drawing of HAR cavity; (b) schematic diagram of LED-based PA spectrophone for measurement of hydrocarbon gases.
The LED-based PA spectrophone in Figure 1b is developed for characterizing the HAR cell. The PA spectrophone mainly includes a light source module, an HAR cell module, and a data-processing module. The instrument’s operation process is as follows:
(1) Light source module: The LED light source is controlled by a light source controller and is modulated by a square wave generated by a data acquisition (DAQ) card. The modulated LED radiation is incited in the HAR cell to excite a photoacoustic signal, which is detected via an electret microphone.
(2) Data-processing module: The electret microphone signal is amplified by a low-noise amplifier and is connected to a lock-in amplifier (SR830, Stanford Research Systems, Sunnyvale, CA, USA) that demodulates photoacoustic signal at the refer modulation frequency; then, an NI data acquisition card (NI-6251, National Instruments, Austin, TX, USA) is used to collect and save the PA signal to a computer through a labview software program (Labview 2014, National Instruments, Austin, TX, USA).
4. Results
4.1. Frequency Response with Different Locations of LED and Microphone
Operating the LED at its optimal modulation frequency is crucial for efficiently exciting the PA signal. In PA spectroscopy, the signal is generated when absorbed photons are converted into thermal energy, leading to localized heating and subsequent sound wave emission via thermoelastic expansion. For effective signal generation, the light must fully interact with the target gas molecules inside the cavity. While laser beams are typically directed through spherical resonators, the positioning of LEDs in such cavities has received limited attention. Similarly, the microphone should be located at the antinode of the resonant acoustic mode to maximize signal detection. Previous studies have suggested mounting the microphone at either the center or the surface of hemispherical cavities [16,20].
Given the significant influence of both source and detector placement on the PA signal amplitude, this study evaluates five distinct configurations to optimize the instrument performance, as illustrated in Figure 2. In Figure 2a, the microphone is fixed at the center of the hemispherical arc top, while the LED is positioned at three different locations: on the inclined plane (LED-A), on the side (LED-B), and at the bottom (LED-C) of the HAR cavity. Figure 2b shows a second arrangement where the microphone is placed at the bottom center, and the LED is tested at two positions (LED-A and LED-B). To determine the optimal modulation frequency, the frequency response of the HAR cell filled with isobutane and nitrogen was characterized using a 3.4 μm LED.
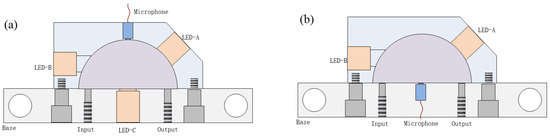
Figure 2.
The microphone installed at the center of the (a) arc top or (b) bottom for evaluating different locations of the LED: LED-A, LED-B, and LED-C.
Figure 3 presents the frequency response of the HAR cavity, characterized by measuring the PA signal from 5000 ppm isobutane and nitrogen under LED modulation frequencies ranging from 100 Hz to 20 kHz. Nitrogen, which exhibits negligible absorption around 3400 nm, was used to measure the background PA signal. Figure 3a displays the response obtained with the microphone positioned at the center of the arc top and the LED placed at three distinct locations: LED-A (inclined plane), LED-B (side), and LED-C (bottom). A significant PA signal difference between isobutane and nitrogen is observed at approximately 250 Hz, indicating a prominent low-frequency resonant mode of the HAR cavity. This low-frequency behavior is further corroborated in Figure 3b, which shows results for the microphone located at the bottom center with the LED at positions LED-A and LED-B. All five configurations demonstrate effective PA signal generation near 250 Hz, consistent with previous reports, such as that by Scholz et al. [18], who identified a low-frequency mode around 300 Hz in an LED-driven elliptical resonator—though their study did not explore frequencies beyond 2500 Hz.

Figure 3.
(a) Frequency response of HAR with a microphone installed at the center of the (a) arc top or (b) bottom with isobutane and nitrogen filling in the HAR cell.
In addition to the low-frequency response, a strong high-frequency resonance is observed near 11.9 kHz when the LED is located at positions LED-A or LED-B, as evidenced by a clear enhancement in the isobutane signal relative to the nitrogen background (Figure 3a). In contrast, the amplitude measured with LED-C at this frequency is comparable to the background level, indicating significantly lower PA generation efficiency at this position. This high-frequency resonance is attributed to an angular acoustic mode of the HAR cavity, as confirmed in our previous work [23], which identified eigenmodes at approximately 7114 Hz, 12,000 Hz, and 15,775 Hz. Among these, the mode near 12,000 Hz is most effectively excited under the current experimental conditions, likely due to stronger overlap with the acoustic mode shape and the spatial distribution of the LED beam, particularly from LED-A and LED-C, which provide more uniform illumination of the cavity compared to LED-B. The weaker excitation of the other modes may result from limited available optical power and spatial mismatch with the acoustic field. To further evaluate the performance of each sensor configuration, PA signals were recorded for various isobutane concentrations under each geometric and excitation condition.
4.2. Instrument Performance
To evaluate the detection sensitivity, the PA signal amplitude was recorded as a function of the isobutane concentration for each of the five sensor configurations described above. Figure 4 displays the measured PA signals for different concentrations under each configuration, using the specific modulation frequencies optimized for the LED source. The corresponding analytical performance metrics are summarized in Table 1, including the linear relationship between the PA signal and concentration, the signal amplitude for 5000 ppm isobutane and nitrogen, the signal-to-noise ratio (SNR), and the calculated detection sensitivity.


Figure 4.
Time series measurements of isobutane under different schemes: (a) microphone installed at the center of arc top with LED-A location and modulation frequency of 350 Hz, (b) microphone installed at the center of arc top with LED-A location and modulation frequency of 19,000 Hz, (c) microphone installed at the center of arc top with LED-B location and modulation frequency of 200 Hz, (d) microphone installed at the center of arc top with LED-C location and modulation frequency of 250 Hz, (e) microphone installed at the center of arc top with LED-C location and modulation frequency of 11,850 Hz, (f) microphone installed at the center of bottom with LED-A location and modulation frequency of 250 Hz, and (g) microphone installed at the center of bottom with LED-B location and modulation frequency of 250 Hz.

Table 1.
Linear relationship of PA signal vs. isobutane concentration under different configurations of LED and microphone position, PA signal of 5000 ppm isobutane and nitrogen, signal-to-noise ratio, and detection sensitivity.
The SNR was determined by dividing the effective PA (EPA) signal of 5000 ppm isobutane (obtained by subtracting the nitrogen background signal from the total measured signal) by the standard deviation (1σ) of the nitrogen PA signal. As indicated in Table 1, the configuration with the microphone at the top position and the LED at location LED-A, modulated at 11.9 kHz, yielded the highest sensitivity, achieving a detection limit of 35 ppm for isobutane. This configuration was identified as optimal for highly sensitive measurement.
4.3. Allan Deviation
An Allan variance analysis is carried out to evaluate the system stability. A time series of 1200 data points is recorded in 1200 s with N2 flushing in the HAR cell. The resulting Allan deviation curve, plotted in Figure 5b, indicates that the system’s stability is predominantly limited by white noise, as depicted by the red reference line [24]. The Allan variance curve in Figure 5 indicates an optimum sensing performance of the PA sensor: a minimum Allan deviation (8 ppmv) is obtained with an optimum averaging (integration) time of 254 s. The detection limit is given as three times the noise standard deviation (STD). The standard deviations of the recorded 1200 data are calculated to be 39 ppm, which leads to a detection limit of 117 ppm (3σ), with an averaging time of 1 s.
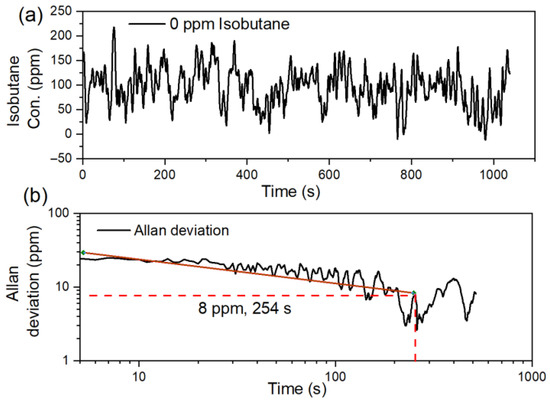
Figure 5.
(a) Time series measurements of 0 ppm isobutane; (b) Allan deviation as a function of averaging (integration) time; red line represents the white noise regime.
4.4. Measurement of Isobutane, Propane, and Ethylene
Hydrocarbon gases show broadband light absorption around 3.4 μm, enabling the detection of multiple hydrocarbon gases using the developed LED-based PA spectrophone. In this experiment, isobutane, propane, and ethylene are individually detected to evaluate their capability of quantifying hydrocarbon gases. The reference concentrations of isobutane (5000 ppm), propane (5000 ppm), and ethylene (15,000 ppm) are diluted with pure nitrogen to obtain different concentrations using two mass flow controllers. Figure 6a shows the time series measurement of isobutane, propane, and ethylene with a resolution time of 1 s. The recorded PA signals well follow the variation in isobutane, propane, and ethylene concentrations. Figure 6b plots the PA signals vs. different concentrations of isobutane, propane, and ethylene. Good linear relationships of PA signal vs. isobutane, propane, and ethylene concentration are found: y = (0.085 ± 0.003)x + 1058 for isobutane, y = (0.076 ± 0.003)x + 1050 for propane, and y = (0.0100 ± 0.0005)x + 1063 for ethylene, with regression coefficient R = 0.99, which indicates the retrieved concentrations are highly accurate.

Figure 6.
(a) Time-series measurements of isobutane, propane, and ethylene; (b) linear relationship of PA signals on isobutane, propane, and ethylene concentrations.
The measurement sensitivities of isobutane, propane, and ethylene are recognized by the PA signal amplitude and the STD of the background PA (BP) signal. The signal-to-noise ratios of PA signals excited by 1250 ppm isobutane, 1250 ppm propane, and 5000 ppm ethylene are deduced as 33.6, 26.8, and 17.3 by using the corresponding effective PA signal amplitude (after removing the BP signal) and the STD of the BP signal. Based on the SNR of the PA signals, the measurement sensitivity values of isobutane, propane, and ethylene are deduced as 111.6 ppm (3σ), 140.1 ppm (3σ), and 866.4 ppm (3σ) at an integration time of 1 s.
Table 2 summarizes the current research status of spherical acoustic resonance cavity-based PA systems using LED and laser light sources. It reveals that the sensitivity of LED-based photoacoustic spectroscopy still lags behind that of laser-driven PA instruments, but the principal appeal of LED-based PA sensors lies in their potential for significant cost reduction and miniaturization. This advantage enables them as a promising technology with broad applications in environmental monitoring, industrial safety, and medical diagnostics.

Table 2.
Summary of the current research status of spherical acoustic resonance cavity-based PA system using LED and laser light source.
5. Conclusions
This study successfully demonstrates a practical and cost-optimized PA spectrophone for hydrocarbon gas detection by integrating a broadband mid-infrared LED with a miniaturized hemispherical acoustic resonator (HAR). This design significantly reduces system cost and size compared to conventional laser-based PA configurations. The LED-based PA spectrophone effectively leverages the advantages of LED, namely low cost, long lifetime, and broad spectral emission. The prototype achieves detection sensitivities of 111.6 ppm (3σ) for isobutane, 140.1 ppm (3σ) for propane, and 866.4 ppm (3σ) for ethylene at the one-second integration, with stability confirmed by the Allan deviation analysis, yielding an 8 ppmv limit at 254 s. The optimized structure (the microphone at the arc top and the LED at position LED-A, modulated at 11.9 kHz) ensures efficient acoustic excitation in a compact cavity volume of only 7.07 mL.
The LED-HAR system is a highly promising candidate for low-cost gas sensing in scenarios such as leak detection in chemical parks, safety monitoring in refineries, and air quality oversight in confined spaces. For future work, efforts should focus on hybrid LED arrays or time-multiplexed multi-wavelength schemes to extend their capability of simultaneous detection of multiple gases in mixtures and, ultimately, in field conditions. With these developments, this technology holds strong potential for widespread deployment in distributed sensor networks and atmospheric monitoring platforms.
Author Contributions
Conceptualization, G.W.; methodology, G.W.; software, L.H., F.L. and L.W.; investigation, C.F. and X.H.; writing—original draft preparation, G.W. and L.H.; writing—review and editing, G.W.; supervision, S.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Zhejiang Province Leading Earth Goose Program (2025C02235), National Natural Science Foundation of China (62305290), Ningbo Science and Technology Project (2023Z179), National Key R&D Program (2022YFC3601002), Zhejiang Provincial Natural Science Foundation of China (LQ22F050014).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Butrymowicz, D.; Smierciew, K.; Karwacki, J.; Gagan, J. Experimental investigations of low-temperature driven ejection refrigeration cycle operating with isobutane. Int. J. Refrig. 2014, 39, 196–209. [Google Scholar] [CrossRef]
- Speight, J.G. Handbook of Petrochemical Processes, 1st ed.; CRC Press: Boca Raton, FL, USA, 2019; pp. 209–266. [Google Scholar]
- Weissermel, K.; Arpe, H.J. Industrial Organic Chemistry, 4th ed.; Wiley-VCH: Hoboken, NJ, USA, 2003; pp. 209–266. [Google Scholar]
- Mei, H.; Wang, G.; Xu, Y.; He, H.; Yao, J.; He, S. Simultaneous measurement of methane, propane and isobutane using a compact mid-infrared photoacoustic spectrophone. Photoacostics 2024, 39, 100635. [Google Scholar] [CrossRef]
- Miklós, A.; Hess, P. Application of acoustic resonators in photoacoustic trace gas analysis and metrology. Rev. Sci. Instrum. 2001, 72, 1937–1955. [Google Scholar] [CrossRef]
- Li, J.; Chen, W.; Yu, B. Recent Progress on Infrared Photoacoustic Spectroscopy Techniques. Appl. Spectrosc. Rev. 2011, 46, 440. [Google Scholar] [CrossRef]
- Palzer, S. Photoacoustic-Based gas sensing: A review. Sensors 2020, 20, 2745. [Google Scholar] [CrossRef]
- Sampaolo, A.; Patimisco, P.; Giglio, M.; Zifarelli, A.; Wu, H.; Dong, L.; Spagnolo, V. Quartz-enhanced photoacoustic spectroscopy for multi-gas detection: A review. Anal. Chim. Acta 2022, 1202, 338894. [Google Scholar] [CrossRef]
- Wang, G.; Meng, L.; Shen, F.; Fei, C.; Hong, X.; He, S. Integrated photoacoustic spectroscopy and scattering nephelometer for simultaneous measurement of aerosol light absorption and scattering properties. Sens. Actuators B Chem. 2025, 444, 138468. [Google Scholar] [CrossRef]
- Bozóki, Z.; Pogány, A.; Szabó, G. Photoacoustic Instruments for Practical Applications: Present, Potentials, and Future Challenges. Appl. Spectrosc. Rev. 2011, 46, 1–37. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, T.; Jiang, Y.; He, S. Compact photoacoustic spectrophone for simultaneously monitoring the concentrations of dichloromethane and trichloromethane with a single acoustic resonator. Opt. Express 2022, 30, 7053. [Google Scholar] [CrossRef] [PubMed]
- Rouxel, J.; Coutard, J.; Gidon, S.; Lartigue, O.; Nicoletti, S.; Parvitte, B.; Vallon, R.; Zéninari, V.; Glière, A. Miniaturized differential Helmholtz resonators for photoacoustic trace gas detection. Sens. Actuators B Chem. 2016, 236, 1104–1110. [Google Scholar] [CrossRef]
- Li, Z.; Si, G.; Ning, Z.; Liu, J.; Fang, Y.; Si, B.; Cheng, Z.; Yang, C. Highly Sensitive Sphere-Tube Coupled Photoacoustic Cell Suitable for Detection of a Variety of Trace Gases: NO2 as an Example. Sensors 2022, 22, 281. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Zhou, S.; He, T.; Li, J. Modeling of a cylindrical resonant photoacoustic cell for high sensitive gas detection. Microw. Opt. Technol. Lett. 2021, 63, 2528. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, L.; Liu, Y.; Zhang, X.; Huan, H.; Yin, X.; Xi, T.; Shao, X. Advances in differential photoacoustic spectroscopy for trace gas detection. Microw. Opt. Technol. Lett. 2023, 65, 1506–1515. [Google Scholar] [CrossRef]
- Li, Z.; Si, G.; Ning, Z.; Liu, J.; Fang, Y.; Cheng, Z.; Si, B.; Yang, C. Research on Long Optical Path and Resonant Carbon Dioxide Gas Photoacoustic Sensor. Spectrosc. Spect. Anal. 2023, 43, 43–49. [Google Scholar]
- Wei, Y.; Huang, Q.; Li, J. Dual-gas detection based on high-performance spherical photoacoustic cells. Sens. Actuators A Phys. 2023, 360, 114542. [Google Scholar] [CrossRef]
- Scholz, L.; Perez, A.; Bierer, B.; Eaksen, P.; Wöllenstein, J.; Palzer, S. Miniature Low-Cost Carbon Dioxide Sensor for Mobile Devices. IEEE Sens. J. 2017, 17, 2889–2895. [Google Scholar] [CrossRef]
- Huang, Q.; Wei, Y.; Li, J. Simultaneous detection of multiple gases using multi-resonance photoacoustic spectroscopy. Sens. Actuators B Chem. 2022, 369, 132234. [Google Scholar] [CrossRef]
- Jiao, Y.; Fan, H.; Gong, Z.; Yang, K.; Shen, F.; Chen, K.; Mei, L.; Peng, W.; Yu, Q. Trace CH4 Gas Detection Based on an Integrated Spherical Photoacoustic Cell. Appl. Sci. 2021, 11, 4997. [Google Scholar] [CrossRef]
- Yuan, W.; Yao, J.; Gong, P.; Jiao, M.; Lei, C.; Jiang, Y.; Xie, L. A Compact NO2 Gas Sensor with an Optimized Photoacoustic Cell and 450 nm-LD. Atmosphere 2023, 14, 704. [Google Scholar] [CrossRef]
- Silverstein, R.M.; Webster, F.X.; Kiemle, D.J.; Bryce, D.L. Spectrometric Identification of Organic Compounds, 8th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2014; pp. 71–125. [Google Scholar]
- Wand, G.; Ding, Z.; Gao, X. Research on Gas Sensing System Using Photoacoustic Spectroscopy Based on a Hemispherical Acoustic Resonant Cavity. Spectrosc. Spect. Anal. 2025, 45, 608. [Google Scholar]
- Wang, G.; Meng, M.; Gou, Q.; Hanoune, B.; Crumeyrolle, S.; Fagniez, T.; Coeur, C.; Akiki, R.; Chen, W. Novel Broadband Cavity-Enhanced Absorption Spectrometer for Simultaneous Measurements of NO2 and Particulate Matter. Anal. Chem. 2023, 95, 3640–3647. [Google Scholar] [CrossRef] [PubMed]
- Wittstock, V.; Scholz, L.; Bierera, B.; Perez, A.O.; Wöllenstein, J.; Palzera, S. Design of a LED-based sensor for monitoring the lower explosion limitof methane. Sens. Actuators B Chem. 2017, 247, 930–939. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).