Airborne Aerosols and Human Health: Leapfrogging from Mass Concentration to Oxidative Potential
Abstract
1. Introduction
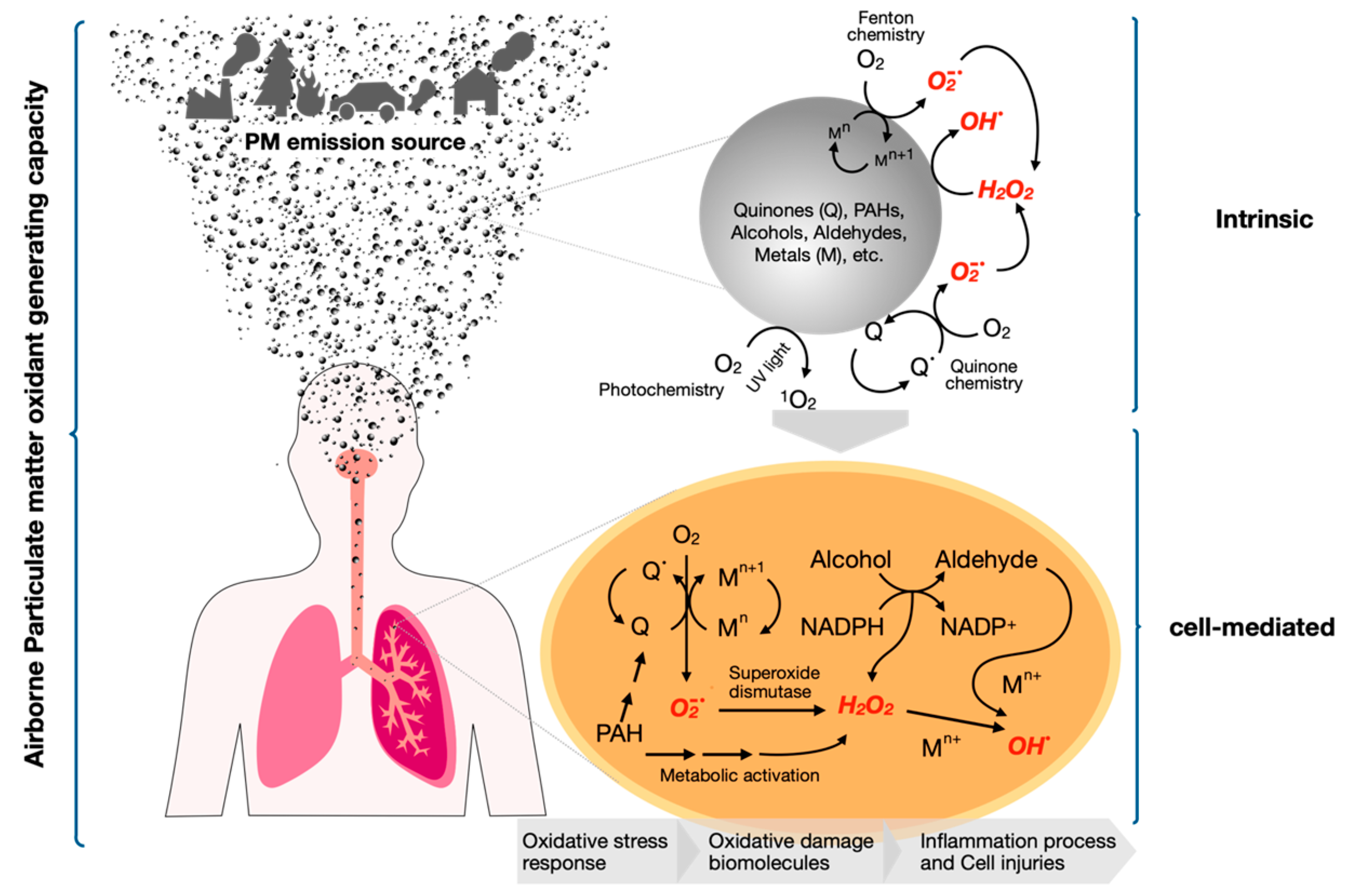
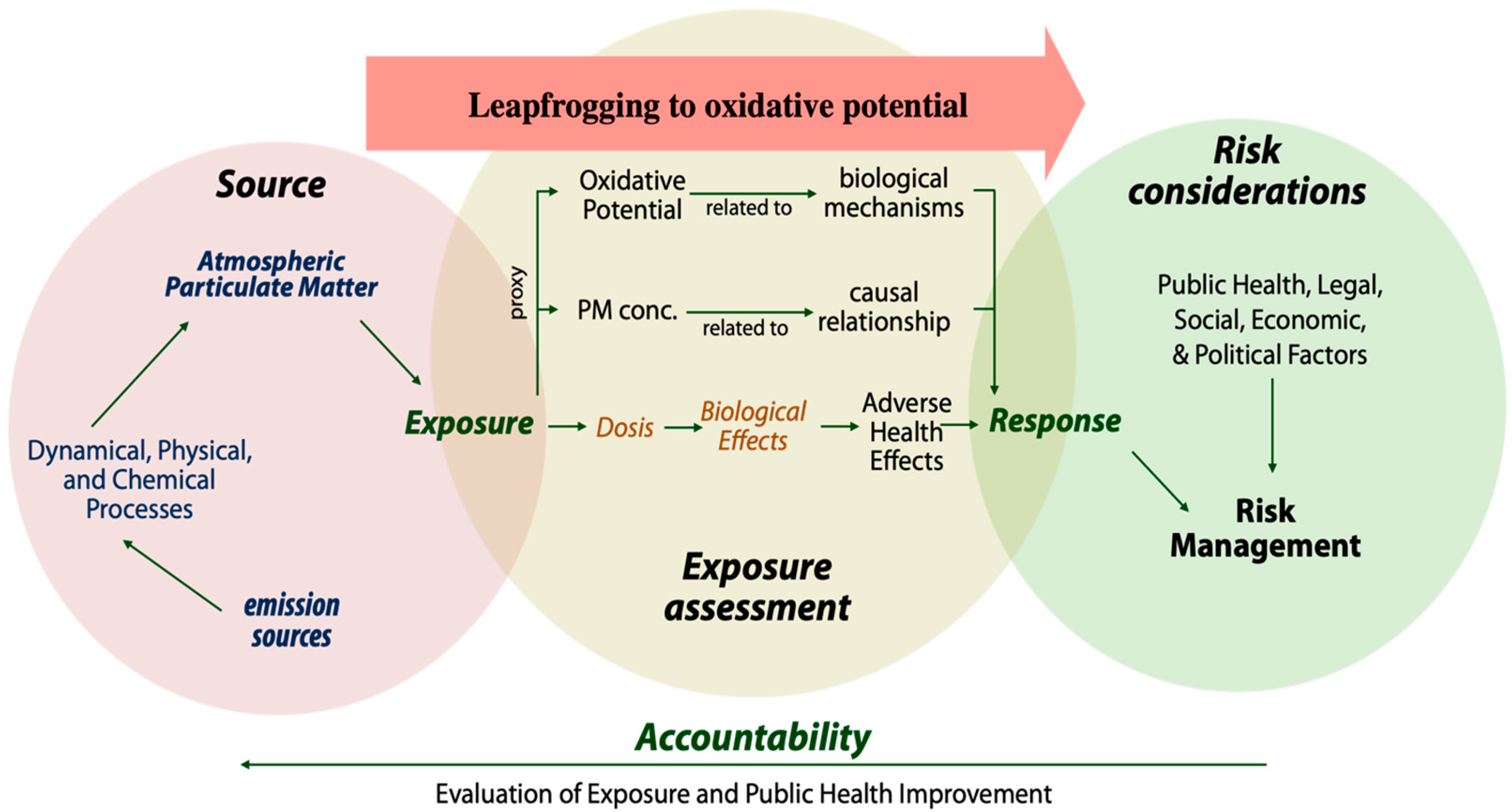
2. Leapfrogging to Oxidative Potential
3. Oxidative Potential Measurement Assays
4. OP Environmental Monitoring
5. OP and Chemical Composition of PM
6. Oxidative Potential and Size Segregated PM
7. Oxidative Potential and PM Source
8. Final Remarks
Author Contributions
Funding
Conflicts of Interest
References
- UN, United Nations, Department of Economic and Social Affairs. World Urbanization Prospects, The 2018 Revision 2018. 2018. Available online: https://population.un.org/wup/Publications/Files/WUP2018-Report.pdf (accessed on 29 May 2020).
- Anderson, J.O.; Thundiyil, J.G.; Stolbach, A. Clearing the Air: A Review of the Effects of Particulate Matter Air Pollution on Human Health. J. Med. Toxicol. 2012, 8, 166–175. [Google Scholar] [CrossRef]
- Kim, K.-H.; Kabir, E.; Kabir, S. A Review on the Human Health Impact of Airborne Particulate Matter. Environ. Int. 2015, 74, 136–143. [Google Scholar] [CrossRef]
- Landrigan, P.J.; Fuller, R.; Acosta, N.J.R.; Adeyi, O.; Arnold, R.; Basu, N.; Baldé, A.B.; Bertollini, R.; Bose-O’Reilly, S.; Boufford, J.I.; et al. The Lancet Commission on Pollution and Health. Lancet 2018, 391, 462–512. [Google Scholar] [CrossRef]
- Dockery, D.W. Health Effects of Particulate Air Pollution. Ann. Epidemiol. 2009, 19, 257–263. [Google Scholar] [CrossRef]
- IARC. International Agency for Research on Cancer Press Release N° 221 IARC: Outdoor Air Pollution a Leading Environmental Cause of Cancer Deaths. 2013. Available online: https://www.iarc.fr/wp-content/uploads/2018/07/pr221_E.pdf (accessed on 29 May 2020).
- Yang, B.Y.; Qian, Z.M.; Li, S.; Chen, G.; Bloom, M.S.; Elliott, M.; Syberg, K.W.; Heinrich, J.; Markevych, I.; Wang, S.Q.; et al. Ambient Air Pollution in Relation to Diabetes and Glucose-Homoeostasis Markers in China: A Cross-Sectional Study with Findings from the 33 Communities Chinese Health Study. Lancet Planet. Health 2018, 2, e64–e73. [Google Scholar] [CrossRef]
- Li, Z.; Wen, Q.; Zhang, R. Sources, Health Effects and Control Strategies of Indoor Fine Particulate Matter (PM2.5): A Review. Sci. Total Environ. 2017, 586, 610–622. [Google Scholar] [CrossRef]
- Li, M.H.; Fan, L.C.; Mao, B.; Yang, J.W.; Choi, A.M.K.; Cao, W.J.; Xu, J.F. Short-Term Exposure to Ambient Fine Particulate Matter Increases Hospitalizations and Mortality in COPD: A Systematic Review and Meta-Analysis. Chest 2016, 149, 447–458. [Google Scholar] [CrossRef]
- Fu, P.; Guo, X.; Cheung, F.M.H.; Yung, K.K.L. The Association between PM 2.5 Exposure and Neurological Disorders: A Systematic Review and Meta-Analysis. Sci. Total Environ. 2019, 10, 1240–1248. [Google Scholar] [CrossRef]
- Zhang, H.; Li, S.; Chen, G.; Abdulai, T.; Liu, X.; Wang, Y.; Liang, H.; Hou, J.; Huo, W.; Mao, Z.; et al. Ambient Air Pollutants Aggravate Association of Snoring with Prevalent Hypertension: Results from the Henan Rural Cohort. Chemosphere 2020, 256, 127108. [Google Scholar] [CrossRef]
- Ostro, B. The Association of Air Pollution and Mortality Examining the Case for Inference. Arch. Environ. Health 1993, 48, 336–342. [Google Scholar] [CrossRef]
- Shiraiwa, M.; Ueda, K.; Pozzer, A.; Lammel, G.; Kampf, C.J.; Fushimi, A.; Enami, S.; Arangio, A.M.; Fröhlich-Nowoisky, J.; Fujitani, Y.; et al. Aerosol Health Effects from Molecular to Global Scales. Environ. Sci. Technol. 2017, 51, 13545–13567. [Google Scholar] [CrossRef]
- Chen, R.; Hu, B.; Liu, Y.; Xu, J.; Yang, G.; Xu, D.; Chen, C. Beyond PM2.5: The Role of Ultrafine Particles on Adverse Health Effects of Air Pollution. Biochim. Biophys. 2016, 1860, 2844–2855. [Google Scholar] [CrossRef]
- Incecik, S.; Gertler, A.; Kassomenos, P. Aerosols and Air Quality. Sci. Total Environ. 2014, 488, 488–489. [Google Scholar] [CrossRef]
- Mcdonald, K. Air Pollution in the Urban Atmosphere: Sources and Consequences. In Metropolitan Sustainability. Understanding and Improving the Urban Environment; Woodhead Publishing: Amsterdam, The Netherlands, 2012; pp. 231–259. [Google Scholar]
- Xing, Y.F.; Xu, Y.H.; Shi, M.H.; Lian, Y.X. The Impact of PM2.5 on the Human Respiratory System. J. Thorac. Dis. 2016, 8, E69–E74. [Google Scholar]
- Di, Q.; Dai, L.; Wang, Y.; Zanobetti, A.; Choirat, C.; Schwartz, J.D.; Dominici, F. Association of Short-Term Exposure to Air Pollution with Mortality in Older Adults. JAMA J. Am. Med. Assoc. 2017, 318, 2446–2456. [Google Scholar] [CrossRef]
- Malley, C.S.; Kuylenstierna, J.C.I.; Vallack, H.W.; Henze, D.K.; Blencowe, H.; Ashmore, M.R. Preterm Birth Associated with Maternal Fine Particulate Matter Exposure: A Global, Regional and National Assessment. Environ. Int. 2017, 101, 173–182. [Google Scholar] [CrossRef]
- Mortamais, M.; Pujol, J.; Martínez-Vilavella, G.; Fenoll, R.; Reynes, C.; Sabatier, R.; Rivas, I.; Forns, J.; Vilor-Tejedor, N.; Alemany, S.; et al. Effects of Prenatal Exposure to Particulate Matter Air Pollution on Corpus Callosum and Behavioral Problems in Children. Environ. Res. 2019, 178, 108734. [Google Scholar] [CrossRef]
- Deng, Q.; Deng, L.; Miao, Y.; Guo, X.; Li, Y. Particle Deposition in the Human Lung: Health Implications of Particulate Matter from Different Sources. Environ. Res. 2019, 169, 237–245. [Google Scholar] [CrossRef]
- USEPA. Regulatory Impact Analyses for the Particulate Matter and Ozone National Ambient Air Quality Standards and Proposed Regional Haze Rule; United States Environmental Protection Agency, Office of Air Quality Planning and Standards, Research Triangle Park: Washington, DC, USA, 1996; Appendix A-2; pp. A-7–A-8.
- Shi, L.; Zanobetti, A.; Kloog, I.; Coull, B.A.; Koutrakis, P.; Melly, S.J.; Schwartz, J.D. Low-Concentration PM2.5 and Mortality: Estimating Acute and Chronic Effects in a Population-Based Study. Environ. Health Perspect. 2016, 124, 46–52. [Google Scholar] [CrossRef]
- Saliba, N. A Comparative Review of PM Levels, Sources, and Their Likely Fates in the Eastern Mediterranean Region. In Urban Airborne Particulate Matter: Origin, Chemistry, Fate, and Health Impacts; Zereini, F., Wiseman, C.L.S., Eds.; Springer: Heidelberg, Germany; New York, NY, USA, 2010; pp. 3–17. [Google Scholar]
- Arhami, M.; Hosseini, V.; Zare Shahne, M.; Bigdeli, M.; Lai, A.; Schauer, J.J. Seasonal Trends, Chemical Speciation and Source Apportionment of Fine PM in Tehran. Atmos. Environ. 2017, 153, 70–82. [Google Scholar] [CrossRef]
- Snider, G.; Weagle, C.L.; Murdymootoo, K.K.; Ring, A.; Ritchie, Y.; Stone, E.; Walsh, A.; Akoshile, C.; Anh, N.X.; Balasubramanian, R.; et al. Variation in Global Chemical Composition of PM2.5: Emerging Results from SPARTAN. Atmos. Chem. Phys. 2016, 16, 9629–9653. [Google Scholar] [CrossRef]
- Amato, F.; Alastuey, A.; Karanasiou, A.; Lucarelli, F.; Nava, S.; Calzolai, G.; Severi, M.; Becagli, S.; Gianelle, V.L.; Colombi, C.; et al. AIRUSE-LIFE+: A Harmonized PM Speciation and Source Apportionment in Five Southern European Cities. Atmos. Chem. Phys. 2016, 16, 3289–3309. [Google Scholar] [CrossRef]
- Tao, J.; Zhang, L.; Cao, J.; Zhang, R. A Review of Current Knowledge Concerning PM2.5 Chemical Composition, Aerosol Optical Properties and Their Relationships across China. Atmos. Chem. Phys. 2017, 17, 9485–9518. [Google Scholar] [CrossRef]
- Wei, K.; Zou, Z.; Zheng, Y.; Li, J.; Shen, F.; Wu, C.Y.; Wu, Y.; Hu, M.; Yao, M. Ambient Bioaerosol Particle Dynamics Observed during Haze and Sunny Days in Beijing. Sci. Total Environ. 2016, 550, 751–759. [Google Scholar] [CrossRef]
- Kelly, F.J.; Fussell, J.C. Size, Source and Chemical Composition as Determinants of Toxicity Attributable to Ambient Particulate Matter. Atmos. Environ. 2012, 60, 504–526. [Google Scholar] [CrossRef]
- Kim, K.H.; Jahan, S.A.; Kabir, E.; Brown, R.J. Review of Airborne Polycyclic Aromatic Hydrocarbons (PAHs) and Their Human Health Effects. Environ. Int. 2013, 60, 71–80. [Google Scholar] [CrossRef]
- Adams, K.; Greenbaum, D.S.; Shaikh, R.; van Erp, A.M.; Russell, A.G. Particulate Matter Components, Sources, and Health: Systematic Approaches to Testing Effects. J. Air Waste Manag. Assoc. 2015, 65, 544–558. [Google Scholar] [CrossRef]
- Levy, J.I.; Diez, D.; Dou, Y.; Barr, C.D.; Dominici, F. A Meta-Analysis and Multisite Time-Series Analysis of the Differential Toxicity of Major Fine Particulate Matter Constituents. Am. J. Epidemiol. 2012, 175, 1091–1099. [Google Scholar] [CrossRef]
- Bell, M.L. Assessment of the Health Impacts of Particulate Matter Characteristics. Res. Rep. Health Eff. Inst. 2012, 161, 5–38. [Google Scholar]
- Bernard, A. Cadmium & Its Adverse Effects on Human Health. Indian J. Med. Res. 2008, 128, 557–564. [Google Scholar]
- White, A.J.; O’Brien, K.M.; Niehoff, N.M.; Carroll, R.; Sandler, D.P. Metallic Air Pollutants and Breast Cancer Risk in a Nationwide Cohort Study. Epidemiology 2019, 30, 20–28. [Google Scholar] [CrossRef]
- Bouwmeester, H.; Lynch, I.; Marvin, H.J.P.; Dawson, K.A.; Berges, M.; Braguer, D.; Byrne, H.J.; Casey, A.; Chambers, G.; Clift, M.J.D.; et al. Minimal Analytical Characterization of Engineered Nanomaterials Needed for Hazard Assessment in Biological Matrices. Nanotoxicology 2011, 5, 1–11. [Google Scholar] [CrossRef]
- Wilson, W. The exposure paradox in particulate matter community time-series epidemiology: Can ambient concentrations of PM be used as a surrogate for personal exposure to PM? Epidemiology 2001, 12, S67. [Google Scholar]
- Hellack, B.; Nickel, C.; Albrecht, C.; Kuhlbusch, T.A.J.; Boland, S.; Baeza-Squiban, A.; Wohlleben, W.; Schins, R.P.F. Analytical Methods to Assess the Oxidative Potential of Nanoparticles: A Review. Environ. Sci. Nano 2017, 4, 1920–1934. [Google Scholar] [CrossRef]
- Kelly, F.J.; Fuller, G.W.; Walton, H.A.; Fussell, J.C. Monitoring Air Pollution: Use of Early Warning Systems for Public Health. Respirology 2012, 17, 7–19. [Google Scholar] [CrossRef]
- Ricci, P.F.; Cirillo, M.C. Uncertainty in Health Risk Analysis. J. Hazard. Mater. 1985, 10, 433–447. [Google Scholar] [CrossRef]
- WHO. Review of Evidence on Health Aspects of Air Pollution—REVIHAAP; First Results; World Health Organization, Regional Office for Europe: Copenhagen, Denmark, 2013; Available online: https://bit.ly/2KOhOMx (accessed on 29 May 2020).
- Miranda, A.I.; Valente, J.; Costa, A.M.; Lopes, M.; Borrego, C. Current Environmental Issues and Challenges. Air Pollution and Health Effects; Springer: Dordrecht, The Netherlands, 2014; pp. 1–13. [Google Scholar]
- Rohr, A.C.; Wyzga, R.E. Attributing Health Effects to Individual Particulate Matter Constituents. Atmos. Environ. 2012, 62, 130–152. [Google Scholar] [CrossRef]
- Yang, W.; Omaye, S.T. Air Pollutants, Oxidative Stress and Human Health. Mutat. Res. Genet. Toxicol. Environ. 2009, 674, 45–54. [Google Scholar] [CrossRef]
- Øvrevik, J.; Refsnes, M.; Låg, M.; Holme, J.; Schwarze, P. Activation of Proinflammatory Responses in Cells of the Airway Mucosa by Particulate Matter: Oxidant- and Non-Oxidant-Mediated Triggering Mechanisms. Biomolecules 2015, 5, 1399–1440. [Google Scholar] [CrossRef]
- Darbre, P.D. Overview of Air Pollution and Endocrine Disorders. Int. J. Gen. Med. 2018, 11, 191–207. [Google Scholar] [CrossRef]
- Delfino, R.J.; Staimer, N.; Tjoa, T.; Gillen, D.L.; Schauer, J.J.; Shafer, M.M. Airway Inflammation and Oxidative Potential of Air Pollutant Particles in a Pediatric Asthma Panel. J. Expos. Sci. Environ. Epidemiol. 2013, 23, 466–473. [Google Scholar] [CrossRef]
- Hoek, G.; Krishnan, R.M.; Beelen, R.; Peters, A.; Ostro, B.; Brunekreef, B.; Kaufman, J.D. Long-Term Air Pollution Exposure and Cardio-Respiratory Mortality: A Review. Environ. Health 2013, 12, 12–43. [Google Scholar] [CrossRef]
- Li, Q.; Wyatt, A.; Kamens, R.M. Oxidant Generation and Toxicity Enhancement of Aged-Diesel Exhaust. Atmos. Environ. 2009, 43, 1037–1042. [Google Scholar] [CrossRef]
- Lin, P.; Yu, J.Z. Generation of Reactive Oxygen Species Mediated by Humic-like Substances in Atmospheric Aerosols. Environ. Sci. Technol. 2011, 45, 10362–10368. [Google Scholar] [CrossRef]
- Valavanidis, A.; Vlachogianni, T.; Fiotakis, K.; Loridas, S. Pulmonary Oxidative Stress, Inflammation and Cancer: Respirable Particulate Matter, Fibrous Dusts and Ozone as Major Causes of Lung Carcinogenesis through Reactive Oxygen Species Mechanisms. Int. J. Environ. Res. Public Health 2013, 10, 3886–3907. [Google Scholar] [CrossRef]
- Clift, M.J.D.; Rothen-Rutishauser, B. Studying the Oxidative Stress Paradigm In Vitro: A Theoretical and Practical Perspective. Methods Mol. Biol. 2013, 1028, 115–133. [Google Scholar]
- Charrier, J.G.; Anastasio, C. Impacts of Antioxidants on Hydroxyl Radical Production from Individual and Mixed Transition Metals in a Surrogate Lung Fluid. Atmos. Environ. 2011, 45, 7555–7562. [Google Scholar] [CrossRef]
- Jiang, H.; Ahmed, C.M.S.; Canchola, A.; Chen, J.Y.; Lin, Y.-H. Use of Dithiothreitol Assay to Evaluate the Oxidative Potential of Atmospheric Aerosols. Atmosphere 2019, 10, 571. [Google Scholar] [CrossRef]
- Frezzini, M.A.; Castellani, F.; De Francesco, N.; Ristorini, M.; Canepari, S. Application of DPPH Assay for Assessment of Particulate Matter Reducing Properties. Atmosphere 2019, 10, 816. [Google Scholar] [CrossRef]
- Tuet, W.Y.; Fok, S.; Verma, V.; Tagle Rodriguez, M.S.; Grosberg, A.; Champion, J.A.; Ng, N.L. Dose-Dependent Intracellular Reactive Oxygen and Nitrogen Species (ROS/RNS) Production from Particulate Matter Exposure: Comparison to Oxidative Potential and Chemical Composition. Atmos. Environ. 2016, 144, 335–344. [Google Scholar] [CrossRef]
- Arnhold, J. Chapter 2—Role of Reactive Species in Destructions. In Cell and Tissue Destruction: Mechanisms, Protection, Disorders; Academic Press: Cambridge, MA, USA, 2020; pp. 23–54. [Google Scholar] [CrossRef]
- Di Meo, S.; Reed, T.T.; Venditti, P.; Victor, V.M. Role of ROS and RNS Sources in Physiological and Pathological Conditions. Oxid. Med. Cell. Longev. 2016, 2016, 44. [Google Scholar] [CrossRef] [PubMed]
- Chio, C.P.; Chen, S.C.; Chiang, K.C.; Chou, W.C.; Liao, C.M. Oxidative Stress Risk Analysis for Exposure to Diesel Exhaust Particle-Induced Reactive Oxygen Species. Sci. Total Environ. 2007, 387, 113–127. [Google Scholar] [CrossRef] [PubMed]
- Venkatachari, P.; Hopke, P.K.; Brune, W.H.; Ren, X.; Lesher, R.; Mao, J.; Mitchel, M. Characterization of Wintertime Reactive Oxygen Species Concentrations in Flushing, New York. Aerosol Sci. Technol. 2007, 41, 97–111. [Google Scholar] [CrossRef]
- Zhang, Y.; Schauer, J.J.; Shafer, M.M.; Hannigan, M.P.; Dutton, S.J. Source Apportionment of in Vitro Reactive Oxygen Species Bioassay Activity from Atmospheric Particulate Matter. Environ. Sci. Technol. 2008, 42, 7502–7509. [Google Scholar] [CrossRef]
- Øvrevik, J. Oxidative Potential versus Biological Effects: A Review on the Relevance of Cell-Free/Abiotic Assays as Predictors of Toxicity from Airborne Particulate Matter. Int. J. Mol. Sci. 2019, 20, 4772. [Google Scholar] [CrossRef]
- Cho, A.K.; Sioutas, C.; Miguel, A.H.; Kumagai, Y.; Schmitz, D.A.; Singh, M.; Eiguren-Fernandez, A.; Froines, J.R. Redox Activity of Airborne Particulate Matter at Different Sites in the Los Angeles Basin. Environ. Res. 2005, 99, 40–47. [Google Scholar] [CrossRef]
- Charrier, J.G.; Anastasio, C. On Dithiothreitol (DTT) as a Measure of Oxidative Potential for Ambient Particles: Evidence for the Importance of Soluble Transition Metals. Atmos. Chem. Phys. 2012, 12, 11317–11350. [Google Scholar] [CrossRef]
- Molina, C.; Andrade, C.; Manzano, C.A.; Richard Toro, A.; Verma, V.; Leiva-Guzmán, M.A. Dithiothreitol-Based Oxidative Potential for Airborne Particulate Matter: An Estimation of the Associated Uncertainty. Environ. Sci. Pollut. Res. 2020, 27, 29672–29680. [Google Scholar] [CrossRef]
- Pietrogrande, M.C.; Bertoli, I.; Manarini, F.; Russo, M. Ascorbate Assay as a Measure of Oxidative Potential for Ambient Particles: Evidence for the Importance of Cell-Free Surrogate Lung Fluid Composition. Atmos. Environ. 2019, 211, 103–112. [Google Scholar] [CrossRef]
- Kurmi, O.P.; Dunster, C.; Ayres, J.G.; Kelly, F.J. Oxidative Potential of Smoke from Burning Wood and Mixed Biomass Fuels. Free Radic. Res. 2013, 47, 829–835. [Google Scholar] [CrossRef]
- Godri, K.J.; Harrison, R.M.; Evans, T.; Baker, T.; Dunster, C.; Mudway, I.S.; Kelly, F.J. Increased Oxidative Burden Associated with Traffic Component of Ambient Particulate Matter at Roadside and Urban Background Schools Sites in London. PLoS ONE 2011, 6, e21961. [Google Scholar] [CrossRef] [PubMed]
- Jovanovic, M.V.; Savic, J.Z.; Salimi, F.; Stevanovic, S.; Bottle, S.; Ristovski, Z.D. Measurements of Oxidative Potential of Particulate Matter at Belgrade Tunnel; Comparison of BPEAnit, DTT and DCFH Assays. Int. J. Environ. Res. Public Health 2019, 16, 4906. [Google Scholar] [CrossRef] [PubMed]
- Taghvaee, S.; Sowlat, M.H.; Diapouli, E.; Manousakas, M.I.; Vasilatou, V.; Eleftheriadis, K.; Sioutas, C. Source Apportionment of the Oxidative Potential of Fine Ambient Particulate Matter (PM2.5) in Athens, Greece. Sci. Total Environ. 2019, 653, 1407–1416. [Google Scholar] [CrossRef] [PubMed]
- Janssen, N.A.H.; Strak, M.; Yang, A.; Hellack, B.; Kelly, F.J.; Kuhlbusch, T.A.J.; Harrison, R.M.; Brunekreef, B.; Cassee, F.R.; Steenhof, M.; et al. Associations between Three Specific A-Cellular Measures of the Oxidative Potential of Particulate Matter and Markers of Acute Airway and Nasal Inflammation in Healthy Volunteers. Occup. Environ. Med. 2015, 72, 49–56. [Google Scholar] [CrossRef]
- Hogervorst, J.G.F.; De Kok, T.M.C.M.; Briedé, J.J.; Wesseling, G.; Kleinjans, J.C.S.; Van Schayck, C.P. Relationship between Radical Generation by Urban Ambient Particulate Matter and Pulmonary Function of School Children. J. Toxicol. Environ. Health Part A 2006, 69, 245–262. [Google Scholar] [CrossRef]
- Bates, J.T.; Fang, T.; Verma, V.; Zeng, L.; Weber, R.J.; Tolbert, P.E.; Abrams, J.Y.; Sarnat, S.E.; Klein, M.; Mulholland, J.A.; et al. Review of Acellular Assays of Ambient Particulate Matter Oxidative Potential: Methods and Relationships with Composition, Sources, and Health Effects. Environ. Sci. Technol. 2019, 53, 4003–4019. [Google Scholar] [CrossRef]
- Hedayat, F.; Stevanovic, S.; Miljevic, B.; Bottle, S.; Ristovski, Z.D. Review-Evaluating the Molecular Assays for Measuring the Oxidative Potential of Particulate Matter. Chem. Ind. Chem. Eng. Q. 2015, 21, 201–210. [Google Scholar] [CrossRef]
- Piacentini, D.; Falasca, G.; Canepari, S.; Massimi, L. Potential of PM-Selected Components to Induce Oxidative Stress and Root System Alteration in a Plant Model Organism. Environ. Int. 2019, 132, 105094. [Google Scholar] [CrossRef]
- Simonetti, G.; Conte, E.; Perrino, C.; Canepari, S. Oxidative Potential of Size-Segregated PM in an Urban and an Industrial Area of Italy. Atmos. Environ. 2018, 187, 292–300. [Google Scholar] [CrossRef]
- Manigrasso, M.; Simonetti, G.; Astolfi, M.L.; Perrino, C.; Canepari, S.; Protano, C.; Antonucci, A.; Avino, P.; Vitali, M. Oxidative Potential Associated with Urban Aerosol Deposited into the Respiratory System and Relevant Elemental and Ionic Fraction Contributions. Atmosphere 2020, 11, 6. [Google Scholar] [CrossRef]
- Gao, D.; Godri Pollitt, K.J.; Mulholland, J.A.; Russell, A.G.; Weber, R.J. Characterization and Comparison of PM2.5 Oxidative Potential Assessed by Two Acellular Assays. Atmos. Chem. Phys. 2020, 20, 5197–5210. [Google Scholar] [CrossRef]
- Abrams, J.Y.; Weber, R.J.; Klein, M.; Samat, S.E.; Chang, H.H.; Strickland, M.J.; Verma, V.; Fang, T.; Bates, J.T.; Mulholland, J.A.; et al. Associations between Ambient Fine Particulate Oxidative Potential and Cardiorespiratory Emergency Department Visits. Environ. Health Perspect. 2017, 125, 10. [Google Scholar]
- Yang, A.; Janssen, N.A.H.; Brunekreef, B.; Cassee, F.R.; Hoek, G.; Gehring, U. Children’s Respiratory Health and Oxidative Potential of PM2.5: The PIAMA Birth Cohort Study. Occup. Environ. Med. 2016, 73, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Weichenthal, S.; Crouse, D.L.; Pinault, L.; Godri-Pollitt, K.; Lavigne, E.; Evans, G.; van Donkelaar, A.; Martin, R.V.; Burnett, R.T. Oxidative Burden of Fine Particulate Air Pollution and Risk of Cause-Specific Mortality in the Canadian Census Health and Environment Cohort (CanCHEC). Environ. Res. 2016, 146, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Fang, T.; Verma, V.; Bates, J.T.; Abrams, J.; Klein, M.; Strickland, J.M.; Sarnat, E.S.; Chang, H.H.; Mulholland, A.J.; Tolbert, E.P.; et al. Oxidative Potential of Ambient Water-Soluble PM2.5 in the Southeastern United States: Contrasts in Sources and Health Associations between Ascorbic Acid (AA) and Dithiothreitol (DTT) Assays. Atmos. Chem. Phys. 2016, 16, 3865–3879. [Google Scholar] [CrossRef]
- Fu, H.; Liu, X.; Li, W.; Zu, Y.; Zhou, F.; Shou, Q.; Ding, Z. PM2.5 Exposure Induces Inflammatory Response in Macrophages via the TLR4/COX-2/NF-ΚB Pathway. Inflammation 2020, 6, 1–11. [Google Scholar]
- Figliuzzi, M.; Tironi, M.; Longaretti, L.; Mancini, A.; Teoldi, F.; Sangalli, F.; Remuzzi, A. Copper-Dependent Biological Effects of Particulate Matter Produced by Brake Systems on Lung Alveolar Cells. Arch. Toxicol. 2020, 94, 2965–2979. [Google Scholar] [CrossRef]
- MohseniBandpi, A.; Eslami, A.; Shahsavani, A.; Khodagholi, F.; Alinejad, A. Physicochemical Characterization of Ambient PM2.5 in Tehran Air and Its Potential Cytotoxicity in Human Lung Epithelial Cells (A549). Sci. Total Environ. 2017, 593–594, 182–190. [Google Scholar] [CrossRef]
- Yang, L.; Liu, G.; Lin, Z.; Wang, Y.; He, H.; Liu, T.; Kamp, D.W. Pro-Inflammatory Response and Oxidative Stress Induced by Specific Components in Ambient Particulate Matter in Human Bronchial Epithelial Cells. Environ. Toxicol. 2016, 31, 923–936. [Google Scholar] [CrossRef]
- Vaughan, A.; Stevanovic, S.; Banks, A.P.W.; Zare, A.; Rahman, M.M.; Bowman, R.V.; Fong, K.M.; Ristovski, Z.D.; Yang, I.A. The Cytotoxic, Inflammatory and Oxidative Potential of Coconut Oil-Substituted Diesel Emissions on Bronchial Epithelial Cells at an Air-Liquid Interface. Environ. Sci. Pollut. Res. 2019, 26, 27783–27791. [Google Scholar] [CrossRef]
- Ndong Ba, A.; Cazier, F.; Verdin, A.; Garcon, G.; Cabral, M.; Courcot, L.; Diouf, A.; Courcot, D.; Gualtieri, M.; Fall, M. Physico-Chemical Characterization and in Vitro Inflammatory and Oxidative Potency of Atmospheric Particles Collected in Dakar City’s (Senegal). Environ. Pollut. 2019, 245, 568–581. [Google Scholar] [CrossRef]
- Xu, Z.; Wu, H.; Zhang, H.; Bai, J.; Zhang, Z. Interleukins 6/8 and Cyclooxygenase-2 Release and Expressions Are Regulated by Oxidative Stress-JAK2/STAT3 Signaling Pathway in Human Bronchial Epithelial Cells Exposed to Particulate Matter ≤ 2.5 Mm. J. Appl. Toxicol. 2020, 40, 1210–1218. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yang, M.; Meng, T.; Niu, Y.; Dai, Y.; Zhang, L.; Zheng, X.; Jalava, P.; Dong, G.; Gao, W.; et al. Oxidative Stress Induced by Ultrafine Carbon Black Particles Can Elicit Apoptosis in Vivo and Vitro. Sci. Total Environ. 2020, 709, 135802. [Google Scholar] [CrossRef] [PubMed]
- Mantecca, P.; Sancini, G.; Moschini, E.; Farina, F.; Gualtieri, M.; Rohr, A.; Miserocchi, G.; Palestini, P.; Camatini, M. Lung Toxicity Induced by Intratracheal Instillation of Size-Fractionated Tire Particles. Toxicol. Lett. 2009, 189, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Jang, M.; Sabo-Attwood, T.; Robinson, S.E. Oxidative Potential of Secondary Organic Aerosols Produced from Photooxidation of Different Hydrocarbons Using Outdoor Chamber under Ambient Sunlight. Atmos. Environ. 2016, 131, 382–389. [Google Scholar] [CrossRef]
- Xu, C.; Zhang, M.; Chen, W.; Jiang, L.; Chen, C.; Qin, J. Assessment of Air Pollutant PM2.5 Pulmonary Exposure Using a 3D Lung-on-Chip Model. ACS Biomater. Sci. Eng. 2020, 6, 3081–3090. [Google Scholar] [CrossRef]
- Yang, J.W.; Shen, Y.C.; Lin, K.C.; Cheng, S.J.; Chen, S.L.; Chen, C.Y.; Kumar, P.V.; Lin, S.F.; Lu, H.E.; Chen, G.Y. Organ-on-a-Chip: Opportunities for Assessing the Toxicity of Particulate Matter. Front. Bioeng. Biotechnol. 2020, 8, 519. [Google Scholar] [CrossRef]
- Mauriac, H.; Pannetier, C.; Velvé Casquillas, G. Organs on Chip Review—Elveflow. 2017. Available online: https://www.elveflow.com/microfluidic-reviews/organs-on-chip-3d-cell-culture/organs-chip-review/ (accessed on 13 August 2020).
- Li, Y.; Hu, C.; Wang, P.; Liu, Y.; Wang, L.; Pi, Q.; Gong, Z.; Yang, X.; Mak, M.; Wu, Y. Indoor Nanoscale Particulate Matter-Induced Coagulation Abnormality Based on a Human 3D Microvascular Model on a Microfluidic Chip. J. Nanobiotechnol. 2019, 17, 20. [Google Scholar] [CrossRef]
- Møller, P.; Loft, S. Oxidative Damage to DNA and Lipids as Biomarkers of Exposure to Air Pollution. Environ. Health Perspect. 2010, 118, 1126–1136. [Google Scholar] [CrossRef]
- Zhang, X.; Staimer, N.; Gillen, D.L.; Tjoa, T.; Schauer, J.J.; Shafer, M.M.; Hasheminassab, S.; Pakbin, P.; Vaziri, N.D.; Sioutas, C.; et al. Associations of Oxidative Stress and Inflammatory Biomarkers with Chemically-Characterized Air Pollutant Exposures in an Elderly Cohort. Environ. Res. 2016, 150, 306–319. [Google Scholar] [CrossRef]
- Strak, M.; Janssen, N.A.H.; Godri, K.J.; Gosens, I.; Mudway, I.S.; Cassee, F.R.; Lebret, E.; Kelly, F.J.; Harrison, R.M.; Brunekreef, B.; et al. Respiratory Health Effects of Airborne Particulate Matter: The Role of Particle Size, Composition, and Oxidative Potential-the RAPTES Project. Environ. Health Perspect. 2012, 120, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Borm, P.J.A.; Kelly, F.; Künzli, N.; Schins, R.P.F.; Donaldson, K. Oxidant Generation by Particulate Matter: From Biologically Effective Dose to a Promising, Novel Metric. Occup. Environ. Med. 2007, 64, 73–74. [Google Scholar] [CrossRef] [PubMed]
- Verma, V.; Pakbin, P.; Cheung, K.L.; Cho, A.K.; Schauer, J.J.; Shafer, M.M.; Kleinman, M.T.; Sioutas, C. Physicochemical and Oxidative Characteristics of Semi-Volatile Components of Quasi-Ultrafine Particles in an Urban Atmosphere. Atmos. Environ. 2011, 45, 1025–1033. [Google Scholar] [CrossRef]
- Daher, N.; Ning, Z.; Cho, A.K.; Shafer, M.; Schauer, J.J.; Sioutas, C. Comparison of the Chemical and Oxidative Characteristics of Particulate Matter (PM) Collected by Different Methods: Filters, Impactors, and BioSamplers. Aerosol Sci. Technol. 2011, 45, 1294–1304. [Google Scholar] [CrossRef]
- Wang, D.; Pakbin, P.; Shafer, M.M.; Antkiewicz, D.; Schauer, J.J.; Sioutas, C. Macrophage Reactive Oxygen Species Activity of Water-Soluble and Water-Insoluble Fractions of Ambient Coarse, PM2.5 and Ultrafine Particulate Matter (PM) in Los Angeles. Atmos. Environ. 2013, 77, 301–310. [Google Scholar] [CrossRef]
- Guo, H.B.; Li, M.; Lyu, Y.; Cheng, T.T.; Xv, J.J.; Li, X. Size-Resolved Particle Oxidative Potential in the Office, Laboratory, and Home: Evidence for the Importance of Water-Soluble Transition Metals. Environ. Pollut. 2019, 246, 704–709. [Google Scholar] [CrossRef]
- Calas, A.; Uzu, G.; Kelly, F.J.; Houdier, S.; Martins, J.M.F.; Thomas, F.; Molton, F.; Charron, A.; Dunster, C.; Oliete, A.; et al. Comparison between Five Acellular Oxidative Potential Measurement Assays Performed with Detailed Chemistry on PM10 Samples from the City of Chamonix (France). Atmos. Chem. Phys. 2018, 18, 7863–7875. [Google Scholar] [CrossRef]
- Liu, W.J.; Xu, Y.S.; Liu, W.X.; Liu, Q.Y.; Yu, S.Y.; Liu, Y.; Wang, X.; Tao, S. Oxidative Potential of Ambient PM2.5in the Coastal Cities of the Bohai Sea, Northern China: Seasonal Variation and Source Apportionment. Environ. Pollut. 2018, 236, 514–528. [Google Scholar] [CrossRef]
- Gao, D.; Mulholland, J.A.; Russell, A.G.; Weber, R.J. Characterization of Water-Insoluble Oxidative Potential of PM2.5 Using the Dithiothreitol Assay. Atmos. Environ. 2020, 224, 117327. [Google Scholar] [CrossRef]
- Yu, S.; Liu, W.; Xu, Y.; Yi, K.; Zhou, M.; Tao, S.; Liu, W. Characteristics and Oxidative Potential of Atmospheric PM2.5 in Beijing: Source Apportionment and Seasonal Variation. Sci. Total Environ. 2019, 650, 277–287. [Google Scholar] [CrossRef]
- Shao, L.; Hu, Y.; Shen, R.; Schäfer, K.; Wang, J.; Wang, J.; Schnelle-Kreis, J.; Zimmermann, R.; BéruBé, K.; Suppan, P. Seasonal Variation of Particle-Induced Oxidative Potential of Airborne Particulate Matter in Beijing. Sci. Total Environ. 2017, 579, 1152–1160. [Google Scholar] [CrossRef] [PubMed]
- Perrone, M.R.; Bertoli, I.; Romano, S.; Russo, M.; Rispoli, G.; Pietrogrande, M.C. PM2.5 and PM10 Oxidative Potential at a Central Mediterranean Site: Contrasts between Dithiothreitol- and Ascorbic Acid-Measured Values in Relation with Particle Size and Chemical Composition. Atmos. Environ. 2019, 210, 143–155. [Google Scholar] [CrossRef]
- Paraskevopoulou, D.; Bougiatioti, A.; Stavroulas, I.; Fang, T.; Lianou, M.; Liakakou, E.; Gerasopoulos, E.; Weber, R.; Nenes, A.; Mihalopoulos, N. Yearlong Variability of Oxidative Potential of Particulate Matter in an Urban Mediterranean Environment. Atmos. Environ. 2019, 206, 183–196. [Google Scholar] [CrossRef]
- Chen, Q.; Wang, M.; Wang, Y.; Zhang, L.; Li, Y.; Han, Y. Oxidative Potential of Water-Soluble Matter Associated with Chromophoric Substances in PM 2.5 over Xi’an, China. Environ. Sci. Technol. 2019, 53, 8574–8584. [Google Scholar] [CrossRef]
- Wang, J.; Lin, X.; Lu, L.; Wu, Y.; Zhang, H.; Lv, Q.; Liu, W.; Zhang, Y.; Zhuang, S. Temporal Variation of Oxidative Potential of Water Soluble Components of Ambient PM2.5 Measured by Dithiothreitol (DTT) Assay. Sci. Total Environ. 2019, 649, 969–978. [Google Scholar] [CrossRef]
- Janssen, N.A.H.; Yang, A.; Strak, M.; Steenhof, M.; Hellack, B.; Gerlofs-Nijland, M.E.; Kuhlbusch, T.; Kelly, F.; Harrison, R.; Brunekreef, B.; et al. Oxidative Potential of Particulate Matter Collected at Sites with Different Source Characteristics. Sci. Total Environ. 2014, 472, 572–581. [Google Scholar] [CrossRef]
- Boogaard, H.; Janssen, N.A.H.; Fischer, P.H.; Kos, G.P.A.; Weijers, E.P.; Cassee, F.R.; van der Zee, S.C.; de Hartog, J.J.; Brunekreef, B.; Hoek, G. Contrasts in Oxidative Potential and Other Particulate Matter Characteristics Collected near Major Streets and Background Locations. Environ. Health Perspect. 2012, 120, 185–191. [Google Scholar] [CrossRef]
- Ntziachristos, L.; Froines, J.R.; Cho, A.K.; Sioutas, C. Relationship between Redox Activity and Chemical Speciation of Size-Fractionated Particulate Matter. Part. Fibre Toxicol. 2007, 4, 5. [Google Scholar] [CrossRef]
- Venkatachari, P.; Hopke, P.K. Development and Laboratory Testing of an Automated Monitor for the Measurement of Atmospheric Particle-Bound Reactive Oxygen Species (ROS). Aerosol Sci. Technol. 2008, 42, 629–635. [Google Scholar] [CrossRef]
- Wragg, F.P.H.; Fuller, S.J.; Freshwater, R.; Green, D.C.; Kelly, F.J.; Kalberer, M. An Automated Online Instrument to Quantify Aerosol-Bound Reactive Oxygen Species (ROS) for Ambient Measurement and Health-Relevant Aerosol Studies. Atmos. Meas. Tech. 2016, 9, 4891–4900. [Google Scholar] [CrossRef]
- Berg, K.E.; Clark, K.M.; Li, X.; Carter, E.M.; Volckens, J.; Henry, C.S. High-Throughput, Semi-Automated Dithiothreitol (DTT) Assays for Oxidative Potential of Fine Particulate Matter. Atmos. Environ. 2020, 222, 117132. [Google Scholar] [CrossRef]
- Fang, T.; Verma, V.; Guo, H.; King, L.E.; Edgerton, E.S.; Weber, R.J. A Semi-Automated System for Quantifying the Oxidative Potential of Ambient Particles in Aqueous Extracts Using the Dithiothreitol (DTT) Assay: Results from the Southeastern Center for Air Pollution and Epidemiology (SCAPE). Atmos. Meas. Tech. 2015, 8, 471–482. [Google Scholar] [CrossRef]
- Eiguren-Fernandez, A.; Kreisberg, N.; Hering, S. An Online Monitor of the Oxidative Capacity of Aerosols (o-MOCA). Atmos. Meas. Tech. 2017, 10, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Puthussery, J.V.; Zhang, C.; Verma, V. Development and Field Testing of an Online Instrument for Measuring the Real-Time Oxidative Potential of Ambient Particulate Matter Based on Dithiothreitol Assay. Atmos. Meas. Tech. 2018, 11, 5767–5780. [Google Scholar] [CrossRef]
- Son, Y.; Mishin, V.; Welsh, W.; Lu, S.-E.; Laskin, J.D.; Kipen, H.; Meng, Q. A Novel High-Throughput Approach to Measure Hydroxyl Radicals Induced by Airborne Particulate Matter. Int. J. Environ. Res. Public Health 2015, 12, 13678–13695. [Google Scholar] [CrossRef]
- Yu, H.; Puthussery, J.V.; Verma, V. A Semi-Automated Multi-Endpoint Reactive Oxygen Species Activity Analyzer (SAMERA) for Measuring the Oxidative Potential of Ambient PM2.5 Aqueous Extracts. Aerosol Sci. Technol. 2019, 54, 304–320. [Google Scholar] [CrossRef]
- Quinn, C.; Miller-Lionberg, D.D.; Klunder, K.J.; Kwon, J.; Noth, E.M.; Mehaffy, J.; Leith, D.; Magzamen, S.; Hammond, S.K.; Henry, C.S.; et al. Personal Exposure to PM2.5 Black Carbon and Aerosol Oxidative Potential Using an Automated Microenvironmental Aerosol Sampler (AMAS). Environ. Sci. Technol. 2018, 52, 11267–11275. [Google Scholar] [CrossRef]
- Brehmer, C.; Lai, A.; Clark, S.; Shan, M.; Ni, K.; Ezzati, M.; Yang, X.; Baumgartner, J.; Schauer, J.J.; Carter, E. The Oxidative Potential of Personal and Household PM2.5 in a Rural Setting in Southwestern China. Environ. Sci. Technol. 2019, 53, 2788–2798. [Google Scholar] [CrossRef]
- Roper, C.; Perez, A.; Barrett, D.; Hystad, P.; Massey Simonich, S.L.; Tanguay, R.L. Workflow for Comparison of Chemical and Biological Metrics of Filter Collected PM2.5. Atmos. Environ. 2020, 226, 117379. [Google Scholar] [CrossRef]
- Brehmer, C.; Norris, C.; Barkjohn, K.K.; Bergin, M.H.; Zhang, J.; Cui, X.; Teng, Y.; Zhang, Y.; Black, M.; Li, Z.; et al. The Impact of Household Air Cleaners on the Oxidative Potential of PM2.5 and the Role of Metals and Sources Associated with Indoor and Outdoor Exposure. Environ. Res. 2020, 181, 108919. [Google Scholar] [CrossRef]
- Zhan, Y.; Johnson, K.; Norris, C.; Shafer, M.M.; Bergin, M.H.; Zhang, Y.; Zhang, J.; Schauer, J.J. The Influence of Air Cleaners on Indoor Particulate Matter Components and Oxidative Potential in Residential Households in Beijing. Sci. Total Environ. 2018, 626, 507–518. [Google Scholar] [CrossRef] [PubMed]
- Charrier, J.G.; Richards-Henderson, N.K.; Bein, K.J.; McFall, A.S.; Wexler, A.S.; Anastasio, C. Oxidant Production from Source-Oriented Particulate Matter—Part 1: Oxidative Potential Using the Dithiothreitol (DTT) Assay. Atmos. Chem. Phys. 2015, 15, 2327–2340. [Google Scholar] [CrossRef]
- Pirhadi, M.; Mousavi, A.; Taghvaee, S.; Shafer, M.M.; Sioutas, C. Semi-Volatile Components of PM2.5 in an Urban Environment: Volatility Profiles and Associated Oxidative Potential. Atmos. Environ. 2020, 223, 117197. [Google Scholar] [CrossRef] [PubMed]
- Verma, V.; Rico-Martinez, R.; Kotra, N.; King, L.; Liu, J.; Snell, T.W.; Weber, R.J. Contribution of Water-Soluble and Insoluble Components and Their Hydrophobic/Hydrophilic Subfractions to the Reactive Oxygen Species-Generating Potential of Fine Ambient Aerosols. Environ. Sci. Technol. 2012, 46, 11384–11392. [Google Scholar] [CrossRef]
- CARB. Determination of Reactive Oxygen Species Activity in PM and Enhanced Exposure Assessment for the NIH, NIEHS Study Entitled: Ultrafine Particulate Matter and Cardiorespiratory Health; California Air Resources Board and the California Environmental Protection Agency: Street Sacramento, CA, USA, 2009. Available online: http://www.arb.ca.gov/research/apr/past/03-329.pdf (accessed on 29 May 2020).
- Verma, V.; Fang, T.; Guo, H.; King, L.; Bates, J.T.; Peltier, R.E.; Edgerton, E.; Russell, A.G.; Weber, R.J. Reactive Oxygen Species Associated with Water-Soluble PM2.5 in the Southeastern United States: Spatiotemporal Trends and Source Apportionment. Atmos. Chem. Phys. 2014, 14, 12915–12930. [Google Scholar] [CrossRef]
- Saffari, A.; Daher, N.; Shafer, M.M.; Schauer, J.J.; Sioutas, C. Global Perspective on the Oxidative Potential of Airborne Particulate Matter: A Synthesis of Research Findings. Environ. Sci. Technol. 2014, 48, 7576–7583. [Google Scholar] [CrossRef]
- Mirowsky, J.E.; Jin, L.; Thurston, G.; Lighthall, D.; Tyner, T.; Horton, L.; Galdanes, K.; Chillrud, S.; Ross, J.; Pinkerton, K.E.; et al. In Vitro and in Vivo Toxicity of Urban and Rural Particulate Matter from California. Atmos. Environ. 2015, 103, 256–262. [Google Scholar] [CrossRef]
- Jomova, K.; Valko, M. Advances in Metal-Induced Oxidative Stress and Human Disease. Toxicology 2011, 283, 65–87. [Google Scholar] [CrossRef]
- Verma, V.; Ning, Z.; Cho, A.K.; Schauer, J.J.; Shafer, M.M.; Sioutas, C. Redox Activity of Urban Quasi-Ultrafine Particles from Primary and Secondary Sources. Atmos. Environ. 2009, 43, 6360–6368. [Google Scholar] [CrossRef]
- Frank, L.D.; Iroz-Elardo, N.; MacLeod, K.E.; Hong, A. Pathways from Built Environment to Health: A Conceptual Framework Linking Behavior and Exposure-Based Impacts. J. Transp. Health 2019, 12, 319–335. [Google Scholar] [CrossRef]
- Künzli, N.; Mudway, I.S.; Götschi, T.; Shi, T.; Kelly, F.J.; Cook, S.; Burney, P.; Forsberg, B.; Gauderman, J.W.; Hazenkamp, M.E.; et al. Comparison of Oxidative Properties, Light Absorbance, and Total and Elemental Mass Concentration of Ambient PM2.5 Collected at 20 European Sites. Environ. Health Perspect. 2006, 114, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Risom, L.; Dybdahl, M.; Bornholdt, J.; Vogel, U.; Wallin, H.; Møller, P.; Loft, S. Oxidative DNA Damage and Defense Gene Expression in the Mouse Lung after Short-Term Exposure to Diesel Exhaust Particles by Inhalation. Carcinogenesis 2003, 24, 1847–1852. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, M.; Autrup, H.; Møller, P.; Hertel, O.; Jensen, S.S.; Vinzents, P.; Knudsen, L.E.; Loft, S. Linking Exposure to Environmental Pollutants with Biological Effects. Mutat. Res. Rev. Mutat. 2003, 544, 255–271. [Google Scholar] [CrossRef] [PubMed]
- Squadrito, G.L.; Cueto, R.; Dellinger, B.; Pryor, W.A. Quinoid Redox Cycling as a Mechanism for Sustained Free Radical Generation by Inhaled Airborne Particulate Matter. Free Radic. Biol. Med. 2001, 31, 1132–1138. [Google Scholar] [CrossRef]
- Penning, T.M.; Burczynski, M.E.; Hung, C.F.; McCoull, K.D.; Palackal, N.T.; Tsuruda, L.S. Dihydrodiol Dehydrogenases and Polycyclic Aromatic Hydrocarbon Activation: Generation of Reactive and Redox Active o-Quinones. Chem. Res. Toxicol. 1999, 12, 1–18. [Google Scholar] [CrossRef]
- McWhinney, R.D.; Badali, K.; Liggio, J.; Li, S.M.; Abbatt, J.P.D. Filterable Redox Cycling Activity: A Comparison between Diesel Exhaust Particles and Secondary Organic Aerosol Constituents. Environ. Sci. Technol. 2013, 47, 3362–3369. [Google Scholar] [CrossRef]
- Cheng, W.Y.; Currier, J.; Bromberg, P.A.; Silbajoris, R.; Simmons, S.O.; Samet, J.M. Linking Oxidative Events to Inflammatory and Adaptive Gene Expression Induced by Exposure to an Organic Particulate Matter Component. Environ. Health Perspect. 2012, 120, 267–274. [Google Scholar] [CrossRef]
- Taguchi, K.; Fujii, S.; Yamano, S.; Cho, A.K.; Kamisuki, S.; Nakai, Y.; Sugawara, F.; Froines, J.R.; Kumagai, Y. An Approach to Evaluate Two-Electron Reduction of 9,10-Phenanthraquinone and Redox Activity of the Hydroquinone Associated with Oxidative Stress. Free Radic. Biol. Med. 2007, 43, 789–799. [Google Scholar] [CrossRef]
- Saffari, A.; Daher, N.; Shafer, M.M.; Schauer, J.J.; Sioutas, C. Seasonal and Spatial Variation in Dithiothreitol (DTT) Activity of Quasi-Ultrafine Particles in the Los Angeles Basin and Its Association with Chemical Species. J. Environ. Sci. Health Part A Toxic Hazard. Substain. Environ. Eng. 2014, 49, 441–451. [Google Scholar] [CrossRef]
- Happo, M.S.; Hirvonen, M.R.; Halinen, A.I.; Jalava, P.I.; Pennanen, A.S.; Sillanpaa, M.; Hillamo, R.; Salonen, R.O. Chemical Compositions Responsible for Inflammation and Tissue Damage in the Mouse Lung by Coarse and Fine Particulate Samples from Contrasting Air Pollution in Europe. Inhal. Toxicol. 2008, 20, 1215–1231. [Google Scholar] [CrossRef]
- Kramer, A.J.; Rattanavaraha, W.; Zhang, Z.; Gold, A.; Surratt, J.D.; Lin, Y.-H. Assessing the Oxidative Potential of Isoprene-Derived Epoxides and Secondary Organic Aerosol. Atmos. Environ. 2016, 130, 211–218. [Google Scholar] [CrossRef]
- Arashiro, M.; Lin, Y.-H.; Zhang, Z.; Sexton, K.G.; Gold, A.; Jaspers, I.; Fry, R.C.; Surratt, J.D. Effect of Secondary Organic Aerosol from Isoprene-Derived Hydroxyhydroperoxides on the Expression of Oxidative Stress Response Genes in Human Bronchial Epithelial Cells. Environ. Sci. Process. Impacts 2018, 20, 332–339. [Google Scholar] [CrossRef] [PubMed]
- McWhinney, R.D.; Zhou, S.; Abbatt, J.P.D. Naphthalene SOA: Redox Activity and Naphthoquinone Gas–Particle Partitioning. Atmos. Chem. Phys. 2013, 13, 9731–9744. [Google Scholar] [CrossRef]
- Tuet, W.Y.; Chen, Y.; Xu, L.; Fok, S.; Gao, D.; Weber, R.J.; Ng, N.L. Chemical Oxidative Potential of Secondary Organic Aerosol (SOA) Generated from the Photooxidation of Biogenic and Anthropogenic Volatile Organic Compounds. Atmos. Chem. Phys. 2017, 17, 839–853. [Google Scholar] [CrossRef]
- Tong, H.; Lakey, P.S.J.; Arangio, A.M.; Socorro, J.; Shen, F.; Lucas, K.; Brune, W.H.; Pöschl, U.; Shiraiwa, M. Reactive Oxygen Species Formed by Secondary Organic Aerosols in Water and Surrogate Lung Fluid. Environ. Sci. Technol. 2018, 52, 11642–11651. [Google Scholar] [CrossRef]
- Fang, T.; Guo, H.; Zeng, L.; Verma, V.; Nenes, A.; Weber, R.J. Highly Acidic Ambient Particles, Soluble Metals, and Oxidative Potential: A Link between Sulfate and Aerosol Toxicity. Environ. Sci. Technol. 2017, 51, 2611–2620. [Google Scholar] [CrossRef]
- Gao, D.; Ripley, S.; Weichenthal, S.; Godri Pollitt, K.J. Ambient Particulate Matter Oxidative Potential: Chemical Determinants, Associated Health Effects, and Strategies for Risk Management. Free Radic. Biol. Med. 2020, 151, 7–25. [Google Scholar] [CrossRef]
- Samake, A.; Uzu, G.; Martins, J.M.F.; Calas, A.; Vince, E.; Parat, S.; Jaffrezo, J.L. The Unexpected Role of Bioaerosols in the Oxidative Potential of PM. Sci. Rep. 2017, 7, 10978. [Google Scholar] [CrossRef]
- Yu, H.; Wei, J.; Cheng, Y.; Subedi, K.; Verma, V. Synergistic and Antagonistic Interactions among the Particulate Matter Components in Generating Reactive Oxygen Species Based on the Dithiothreitol Assay. Environ. Sci. Technol. 2018, 52, 2261–2270. [Google Scholar] [CrossRef]
- Lin, M.; Yu, J.Z. Effect of Metal-Organic Interactions on the Oxidative Potential of Mixtures of Atmospheric Humic-like Substances and Copper/Manganese as Investigated by the Dithiothreitol Assay. Sci. Total Environ. 2019, 697, 134012. [Google Scholar] [CrossRef]
- Guo, H.; Fu, H.; Jin, L.; Huang, S.; Li, X. Quantification of Synergistic, Additive and Antagonistic Effects of Aerosol Components on Total Oxidative Potential. Chemosphere 2020, 252, 126573. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Sioutas, C.; Cho, A.; Schmitz, D.; Misra, C.; Sempf, J.; Wang, M.; Oberley, T.; Froines, J.; Nel, A. Ultrafine Particulate Pollutants Induce Oxidative Stress and Mitochondrial Damage. Environ. Health Perspect. 2003, 111, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Ohlwein, S.; Kappeler, R.; Kutlar Joss, M.; Künzli, N.; Hoffmann, B. Health Effects of Ultrafine Particles: A Systematic Literature Review Update of Epidemiological Evidence. Int. J. Public Health 2019, 64, 547–559. [Google Scholar] [CrossRef] [PubMed]
- Lyu, Y.; Guo, H.; Cheng, T.; Li, X. Particle Size Distributions of Oxidative Potential of Lung-Deposited Particles: Assessing Contributions from Quinones and Water-Soluble Metals. Environ. Sci. Technol. 2018, 52, 6592–6600. [Google Scholar] [CrossRef] [PubMed]
- Saffari, A.; Daher, N.; Shafer, M.; Schauer, J.; Sioutas, C. Toxic Potential of Ambient Particulate Matter in Various Urban Areas of the World and Its Relation to Chemical Composition and Emission Sources. Eur. Respir. J. 2014, 44, 4132. [Google Scholar]
- Cheung, K.L.; Polidori, A.; Ntziachristos, L.; Tzamkiozis, T.; Samaras, Z.; Cassee, F.R.; Gerlofs, M.; Sioutas, C. Chemical Characteristics and Oxidative Potential of Particulate Matter Emissions from Gasoline, Diesel, and Biodiesel Cars. Environ. Sci. Technol. 2009, 43, 6334–6340. [Google Scholar] [CrossRef]
- Sauvain, J.-J.; Deslarzes, S.; Riediker, M. Nanoparticle Reactivity toward Dithiothreitol. Nanotoxicology 2008, 2, 121–129. [Google Scholar] [CrossRef]
- Breznan, D.; Nazemof, N.; Kunc, F.; Hill, M.; Vladisavljevic, D.; Gomes, J.; Johnston, L.J.; Vincent, R.; Kumarathasan, P. Acellular Oxidative Potential Assay for Screening of Amorphous Silica Nanoparticles. Analyst 2020, 145, 4867–4879. [Google Scholar] [CrossRef]
- Liu, Q.; Shahpoury, P.; Liggio, J.; Harner, T.; Li, K.; Lee, P.; Li, S.-M. Understanding the Key Role of Atmospheric Processing in Determining the Oxidative Potential of Airborne Engineered Nanoparticles. Environ. Sci. Technol. Lett. 2020, 7, 7–13. [Google Scholar] [CrossRef]
- Cesari, D.; Merico, E.; Grasso, F.M.; Decesari, S.; Belosi, F.; Manarini, F.; de Nuntiis, P.; Rinaldi, M.; Volpi, F.; Gambaro, A.; et al. Source Apportionment of PM2.5 and of Its Oxidative Potential in an Industrial Suburban Site in South Italy. Atmosphere 2019, 10, 758. [Google Scholar] [CrossRef]
- Yang, A.; Wang, M.; Eeftens, M.; Beelen, R.; Dons, E.; Leseman, D.L.A.C.; Brunekreef, B.; Cassee, F.R.; Janssen, N.A.H.; Hoek, G. Spatial Variation and Land Use Regression Modeling of the Oxidative Potential of Fine Particles. Environ. Health Perspect. 2015, 123, 1187–1192. [Google Scholar] [CrossRef] [PubMed]
- Jedynska, A.; Hoek, G.; Wang, M.; Yang, A.; Eeftens, M.; Cyrys, J.; Keuken, M.; Ampe, C.; Beelen, R.; Cesaroni, G.; et al. Spatial Variations and Development of Land Use Regression Models of Oxidative Potential in Ten European Study Areas. Atmos. Environ. 2017, 150, 24–32. [Google Scholar] [CrossRef]
- Gulliver, J.; Morley, D.; Dunster, C.; McCrea, A.; van Nunen, E.; Tsai, M.-Y.; Probst-Hensch, N.; Eeftens, M.; Imboden, M.; Ducret-Stich, R.; et al. Land Use Regression Models for the Oxidative Potential of Fine Particles (PM 2.5) in Five European Areas. Environ. Res. 2018, 160, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Weichenthal, S.; Shekarrizfard, M.; Traub, A.; Kulka, R.; Al-Rijleh, K.; Anowar, S.; Evans, G.; Hatzopoulou, M. Within-City Spatial Variations in Multiple Measures of PM2.5 Oxidative Potential in Toronto, Canada. Environ. Sci. Technol. 2019, 53, 2799–2810. [Google Scholar] [CrossRef] [PubMed]
- Yang, A.; Hoek, G.; Montagne, D.; Leseman, D.L.A.C.; Hellack, B.; Kuhlbusch, T.A.J.; Cassee, F.R.; Brunekreef, B.; Janssen, N.A.H. Agreement of Central Site Measurements and Land Use Regression Modeled Oxidative Potential of PM2.5 with Personal Exposure. Environ. Res. 2015, 140, 397–404. [Google Scholar] [CrossRef]
- Hellack, B.; Sugiri, D.; Schins, R.P.F.; Schikowski, T.; Krämer, U.; Kuhlbusch, T.A.J.; Hoffmann, B. Land Use Regression Modeling of Oxidative Potential of Fine Particles, NO2, PM2.5 Mass and Association to Type Two Diabetes Mellitus. Atmos. Environ. 2017, 171, 181–190. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molina, C.; Toro A., R.; Manzano, C.A.; Canepari, S.; Massimi, L.; Leiva-Guzmán, M.A. Airborne Aerosols and Human Health: Leapfrogging from Mass Concentration to Oxidative Potential. Atmosphere 2020, 11, 917. https://doi.org/10.3390/atmos11090917
Molina C, Toro A. R, Manzano CA, Canepari S, Massimi L, Leiva-Guzmán MA. Airborne Aerosols and Human Health: Leapfrogging from Mass Concentration to Oxidative Potential. Atmosphere. 2020; 11(9):917. https://doi.org/10.3390/atmos11090917
Chicago/Turabian StyleMolina, Carolina, Richard Toro A., Carlos A. Manzano, Silvia Canepari, Lorenzo Massimi, and Manuel. A. Leiva-Guzmán. 2020. "Airborne Aerosols and Human Health: Leapfrogging from Mass Concentration to Oxidative Potential" Atmosphere 11, no. 9: 917. https://doi.org/10.3390/atmos11090917
APA StyleMolina, C., Toro A., R., Manzano, C. A., Canepari, S., Massimi, L., & Leiva-Guzmán, M. A. (2020). Airborne Aerosols and Human Health: Leapfrogging from Mass Concentration to Oxidative Potential. Atmosphere, 11(9), 917. https://doi.org/10.3390/atmos11090917