A Preliminary Spatial Analysis of the Association of Asthma and Traffic-Related Air Pollution in the Metropolitan Area of Calgary, Canada
Abstract
1. Introduction
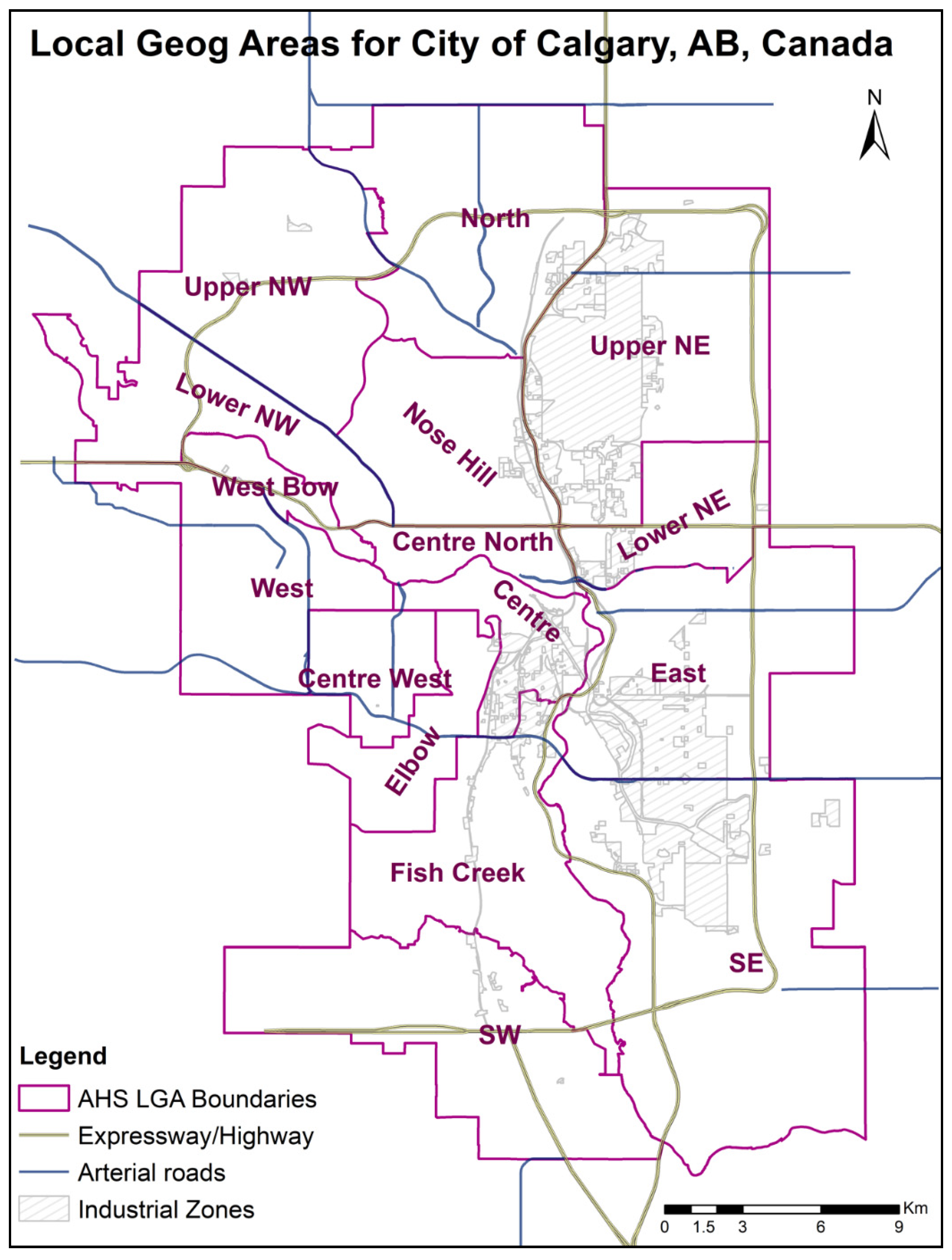
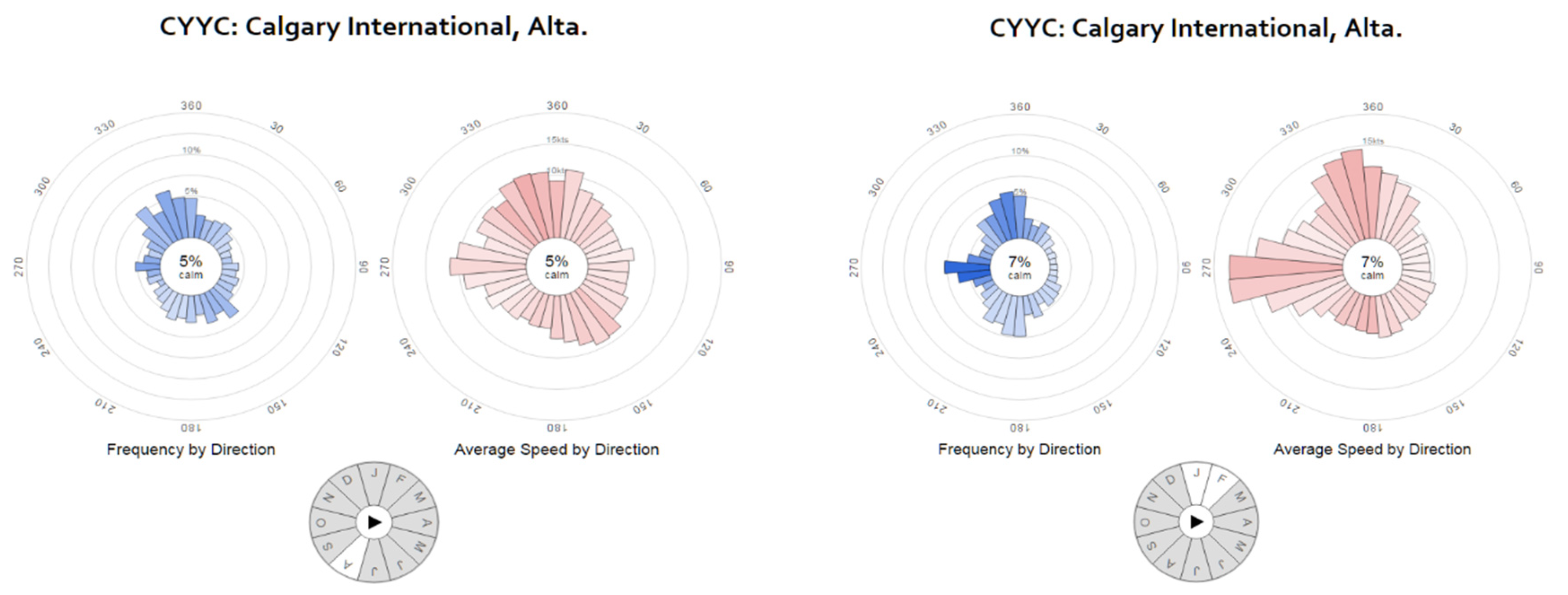
2. Study Region, Data, and Methods
3. Results
4. Discussion
4.1. Pollutants and Seasonality
4.2. Temporal Trend
4.3. Spatial Analysis
4.4. Limitations, Novelty, and Future Work
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Holgate, S.T.; Wenzel, S.; Postma, D.S.; Weiss, S.T.; Renz, H.; Sly, P.D. Asthma. Nat. Rev. Dis. Primers. 2014, 1, 15025. [Google Scholar] [CrossRef]
- Understanding Asthma. Available online: https://asthma.ca/get-help/understanding-asthma/ (accessed on 2 September 2020).
- Asthma. Available online: https://www.canada.ca/en/public-health/services/diseases/asthma.html (accessed on 2 September 2020).
- Asthma. Available online: https://www.albertahealthservices.ca/info/Page15627.aspx (accessed on 11 August 2020).
- Bosonea, A.M.; Sharpe, H.; Wang, T.; Bakal, J.; Befus, D.; Svenson, L.; Vliagoftis, H. Developments in asthma incidence and prevalence in Alberta between 1995 and 2015. Allergy Asthma Clin. Immunol. 2020. under review. [Google Scholar]
- Asthma—Age-Sex Specific Incidence Rate. Available online: http://www.ahw.gov.ab.ca/IHDA_Retrieval/redirectToURL.do?cat=6&subCat=1049 (accessed on 31 August 2020).
- Tétreault, L.F.; Doucet, M.; Gamache, P.; Fournier, M.; Brand, A.; Kosatsky, T.; Smargiassi, A. Childhood exposure to ambient air pollutants and the onset of asthma: An administrative cohort study in Québec. Environ. Health Perspect. 2016, 124, 1276–1282. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.Y.; Ding, H.; Jiang, L.N.; Chen, S.W.; Zheng, J.P.; Qiu, M.; Zhou, Y.X.; Chen, Q.; Guan, W.J. Association between air pollutants and asthma emergency room visits and hospital admissions in time series studies: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0138146. [Google Scholar] [CrossRef] [PubMed]
- Bouazza, N.; Foissac, F.; Urien, S.; Guedj, R.; Carbajal, R.; Tréluyer, J.M.; Chappuy, H. Fine particulate pollution and asthma exacerbations. Arch. Dis. Child. 2018, 103, 828–831. [Google Scholar] [CrossRef]
- Rosychuk, R.J.; Ospina, M.; Zhang, J.; Leigh, R.; Cave, A.; Rowe, B.H. Sex differences in outcomes after discharge from Alberta emergency departments for asthma: A large population-based study. J. Asthma 2018, 55, 817–825. [Google Scholar] [CrossRef] [PubMed]
- Requia, W.J.; Adams, M.D.; Koutrakis, P. Association of PM2.5 with diabetes, asthma, and high blood pressure incidence in Canada: A spatiotemporal analysis of the impacts of the energy generation and fuel sales. Sci. Total Environ. 2017, 584–585, 1077–1083. [Google Scholar] [CrossRef]
- Villamizar, L.A. Outdoor Air Pollution and Children’s Asthma in the Census Metropolitan Area of Edmonton, Alberta: The Influence of Place of Residence and Socioeconomic Position. Ph.D. Thesis, University of Alberta, Edmonton, AB, Canada, 2016. [Google Scholar]
- Kozyrskyj, A.L.; Kendall, G.E.; Jacoby, P.; Sly, P.D.; Zubrick, S.R. Association between socioeconomic status and the development of asthma: Analyses of income trajectories. Am. J. Public Health 2010, 100, 540–546. [Google Scholar] [CrossRef]
- Almqvist, C.; Pershagen, G.; Wickman, M. Low socioeconomic status as a risk factor for asthma, rhinitis and sensitization at 4 years in a birth cohort. Clin. Exp. Allergy 2005, 35, 612–618. [Google Scholar] [CrossRef]
- Bacon, S.L.; Bouchard, A.; Loucks, E.B.; Lavoie, K.L. Individual-level socioeconomic status is associated with worse asthma morbidity in patients with asthma. Respir. Res. 2009, 10, 125. [Google Scholar] [CrossRef]
- Cakmak, S.; Dales, R.E.; Judek, S. Respiratory health effects of air pollution gases: Modification by education and income. Arch. Environ. Occup. Health 2006, 61, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Castner, J.; Guo, L.; Yin, Y. Ambient air pollution and emergency department visits for asthma in Erie County, New York 2007–2012. Int. Arch. Occup. Environ. Health 2018, 91, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Orellano, P.; Quaranta, N.; Reynoso, J.; Balbi, B.; Vasquez, J. Effect of outdoor air pollution on asthma exacerbations in children and adults: Systematic review and multilevel meta-analysis. PLoS ONE 2017, 12, e0174050. [Google Scholar] [CrossRef] [PubMed]
- Khreis, H.; Kelly, C.; Tate, J.; Parslow, R.; Lucas, K.; Nieuwenhuijsen, M. Exposure to traffic-related air pollution and risk of development of childhood asthma: A systematic review and meta-analysis. Environ. Int. 2017, 100, 1–31. [Google Scholar] [CrossRef]
- Khreis, H.; Nieuwenhuijsen, M.J. Traffic-related air pollution and childhood asthma: Recent advances and remaining gaps in the exposure assessment methods. Int. J. Environ. Res. Public Health 2017, 14, 312. [Google Scholar] [CrossRef]
- Gauderman, W.J.; Vora, H.; McConnell, R.; Berhane, K.; Gilliland, F.; Thomas, D.; Lurmann, F.; Avol, E.; Kunzli, N.; Jerrett, M.; et al. Effect of exposure to traffic on lung development from 10 to 18 years of age: A cohort study. Lancet 2007, 369, 571–577. [Google Scholar] [CrossRef]
- Rosenlund, M.; Forastiere, F.; Porta, D.; De Sario, M.; Badaloni, C.; Perucci, C.A. Traffic-related air pollution in relation to respiratory symptoms, allergic sensitisation and lung function in schoolchildren. Thorax 2009, 64, 573–580. [Google Scholar] [CrossRef]
- Dell, S.D.; Jerrett, M.; Beckerman, B.; Brook, J.R.; Foty, R.G.; Gilbert, N.L.; Marshall, L.; Miller, J.D.; To, T.; Walter, S.D.; et al. Presence of other allergic disease modifies the effect of early childhood traffic-related air pollution exposure on asthma prevalence. Environ. Int. 2014, 65, 83–92. [Google Scholar] [CrossRef]
- Hesam, M.; Shakerkhatibi, M.; Samadi, M.T.; Poorolajal, J.; Rahmani, A.; Rafieemehr, H. Long-term exposure to outdoor VOCs and lung function in urban adults: A cross-sectional study in Tabriz an industrialized city in the northwest of Iran. Hum. Ecol. Risk Assess. 2020, 26, 1512–1528. [Google Scholar] [CrossRef]
- Clark, N.A.; Demers, P.A.; Karr, C.J.; Koehoorn, M.; Lencar, C.; Tamburic, L.; Brauer, M. Effect of early life exposure to air pollution on development of childhood asthma. Environ. Health Perspect. 2010, 118, 284–290. [Google Scholar] [CrossRef]
- Inhalable Particulate Matter and Health (PM2.5 and PM10). Available online: https://ww2.arb.ca.gov/resources/inhalable-particulate-matter-and-health (accessed on 1 September 2020).
- Dehghani, M.; Fazlzadeh, M.; Sorooshian, A.; Tabatabaee, H.R.; Miri, M.; Baghani, A.N.; Delikhoon, M.; Mahvi, A.H.; Rashidi, M. Characteristics and health effects of BTEX in a hot spot for urban pollution. Ecotoxic Environ. Saf. 2018, 155, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Volatile Organic Compounds. Available online: https://www.lung.org/clean-air/at-home/indoor-air-pollutants/volatile-organic-compounds (accessed on 3 September 2020).
- Black Carbon Research. Available online: https://www.epa.gov/air-research/black-carbon-research (accessed on 3 September 2020).
- Particulate Matter Literature Review. Available online: http://www.burncohowesound.com/wp-content/uploads/2016/08/9.1_E_APP%20PM%20Literature%20Review.pdf (accessed on 2 September 2020).
- Nurvatov, U.B.; Tagiyeva, N.; Semple, S.; Devereux, G.; Sheikh, A. Volatile organic compounds and risk of asthma and allergy: A systematic review. Eur. Respir. Rev. 2015, 24, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Dissemination Block. Available online: https://www150.statcan.gc.ca/n1/pub/92-195-x/2011001/geo/db-id/db-id-eng.htm (accessed on 23 September 2020).
- Bertazzon, S.; Couloigner, I.; Underwood, F.E. Spatial land use regression of nitrogen dioxide over a 5-year interval in Calgary, Canada. Int. J. Geogr. Inf. Sci. 2019, 33, 1335–1354. [Google Scholar] [CrossRef]
- Population and Dwelling Count Highlight Tables, 2016 Census. Available online: https://www12.statcan.gc.ca/census-recensement/2016/dp-pd/hlt-fst/pd-pl/Table.cfm?Lang=Eng&T=301&S=3&O=D (accessed on 13 June 2020).
- City of Calgary Statistics Profile. Available online: http://www.municipalaffairs.alberta.ca/cfml/MunicipalProfiles/index.cfm?fuseaction=BasicReport&MunicipalityType=CITY&stakeholder=46&profileType=STAT (accessed on 13 June 2020).
- Calgary Facts. Available online: https://www.lifeincalgary.ca/moving/calgary-facts (accessed on 13 June 2020).
- Where is Calgary? Available online: https://calgaryeconomicdevelopment.com/why-calgary/where-is-calgary/where-is-calgary/ (accessed on 13 June 2020).
- Average Annual Wind Speed at Canadian Cities. Available online: https://www.currentresults.com/Weather/Canada/Cities/wind-annual-average.php (accessed on 13 June 2020).
- Bertazzon, S.; Shahid, R. Schools, air pollution, and active transportation: An exploratory spatial analysis of Calgary, Canada. Int. J. Environ. Res. Public Health 2017, 14, 834. [Google Scholar] [CrossRef]
- Chapter 5 Airport Climatology Alberta. Available online: https://www.navcanada.ca/EN/media/Publications/Local%20Area%20Weather%20Manuals/LAWM-Prairies-5-EN.pdf (accessed on 3 September 2020).
- Official Standard Geographic Areas. Available online: https://open.alberta.ca/dataset/a14b50c9-94b2-4024-8ee5-c13fb70abb4a/resource/70fd0f2c-5a7c-45a3-bdaa-e1b4f4c5d9a4/download/Official-Standard-Geographic-Area-Document.pdf (accessed on 28 July 2020).
- Bertazzon, S.; Johnson, M.; Eccles, K.; Kaplan, G.G. Accounting for spatial effects in land use regression for urban air pollution modelling. Spat. Spatio-Temporal Epidemiol. 2015, 14–15, 9–21. [Google Scholar] [CrossRef]
- Bertazzon, S.; Underwood, F.; Johnson, M.; Zhang, J. Land use regression of particulate matter in Calgary, Canada. In Proceedings of the Ninth International Conference on Geographic Information Science, Montreal, QC, Canada, 27–30 September 2016. [Google Scholar] [CrossRef]
- Bertazzon, S.; Couloigner, I. Exploratory spatial analysis of air pollution over a large metropolitan area. Riv. Geogr. Ital. 2018, 125, 525–548. [Google Scholar]
- Liu, X.; Bertazzon, S. Fine scale spatiotemporal modelling of urban air pollution. In Geographic Information Science, Proceedings of the Ninth International Conference on Geographic Information Science. GIScience 2016, Montreal, QC, Canada, 27–30 September 2016; Miller, J., O’Sullivan, D., Wiegand, N., Eds.; Springer: Berlin, Germany, 2016; Volume 9927, pp. 210–224. [Google Scholar]
- Getis, A.; Ord, J.K. The analysis of spatial association by use of distance statistics. Geogr. Anal. 1992, 24, 189–206. [Google Scholar] [CrossRef]
- Getis, A. A history of the concept of spatial autocorrelation: A geographer’s perspective. Geogr. Anal. 2008, 40, 297–309. [Google Scholar] [CrossRef]
- Burt, J.; Barber, G.; Rigby, D.L. Elementary Statistics for Geographers, 3rd ed.; Guilford Press: New York, NY, USA, 2009. [Google Scholar]
- Anselin, L. Spatial Econometrics: Methods and Models; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1998. [Google Scholar]
- Wind History for CYYC: Calgary International, Alta. Available online: http://windhistory.com/station.html?CYYC (accessed on 26 September 2020).
- Rumchev, K.; Spickett, J.; Bulsara, M.; Phillips, M.; Stick, S. Association of domestic exposure to volatile organic compounds with asthma in young children. Thorax 2004, 59, 746–751. [Google Scholar] [CrossRef]
- Hulin, M.; Caillaud, D.; Annesi-Maesano, I. Indoor air pollution and childhood asthma: Variations between urban and rural areas. Indoor Air 2010, 20, 502–514. [Google Scholar] [CrossRef]
- Roberts-Semple, D.; Song, F.; Gao, Y. Seasonal characteristics of ambient nitrogen oxides and ground-level ozone in metropolitan northeastern New Jersey. Atmos. Pollut. Res. 2012, 3, 247–257. [Google Scholar] [CrossRef]
- Byers, N.; Ritchey, M.; Vaidyanathan, A.; Brandt, A.J.; Yip, F. Short-term effects of ambient air pollutants on asthma-related emergency department visits in Indianapolis, Indiana, 2007–2011. J. Asthma 2016, 53, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Erbas, B.; Jazayeri, M.; Lambert, K.A.; Katelaris, C.H.; Prendergast, L.A.; Tham, R.; Parrodi, M.J.; Davies, J.; Newbigin, E.; Abramson, M.J.; et al. Outdoor pollen is a trigger of child and adolescent asthma emergency department presentations: A systematic review and meta-analysis. Allergy 2018, 73, 1632–1641. [Google Scholar] [CrossRef] [PubMed]
- Côté, A.; Turmel, J.; Boulet, L.P. Exercise and Asthma. Semin Respir Crit. Care Med. 2018, 39, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, M.; Bertazzon, S.; Couloigner, I. Modeling wildfire smoke pollution by integrating land use regression and remote sensing data: Regional multi-temporal estimates for public health and exposure models. Atmosphere 2018, 9, 335. [Google Scholar] [CrossRef]
- Mirzaei, M.; Bertazzon, S.; Couloigner, I.; Farjad, B.; Ngom, R. Estimation of local daily PM2.5 concentration during wildfire episodes: Integrating MODIS AOD with multivariate linear mixed effect (LME) models. Air Qual. Atmos. Health 2020, 13, 173–185. [Google Scholar]
- Tischer, C.; Chen, C.M.; Heinrich, J. Association between domestic mould and mould components, and asthma and allergy in children: A systematic review. Eur. Respiry J. 2011, 38, 812–824. [Google Scholar] [CrossRef]
- Tischer, C.G.; Hohmann, C.; Thiering, E.; Herbarth, O.; Müller, A.; Henderson, J.; Granell, R.; Fantini, M.P.; Luciano, L.; Bergström, A.; et al. Meta-analysis of mould and dampness exposure on asthma and allergy in eight European birth cohorts: An ENRIECO initiative. Allergy 2011, 66, 1570–1579. [Google Scholar] [CrossRef]
- Jeedrychowski, W.; Maugeri, U.; Zembala, M.; Perzanowski, M.S.; Hajto, B.; Flak, E.; Mróz, E.; Jacek, R.; Sowa, A.; Perera, F.P. Risk of wheezing associated with house-dust mite allergens and indoor air quality among three-year-old children. Kraków inner city study. Int. J. Occup. Med. Environ. Health 2007, 20, 117–126. [Google Scholar] [CrossRef]
- Eccles, K.M.; Checkley, S.; Sjogren, D.; Barkema, H.W.; Bertazzon, S. Lessons learned from the 2013 Calgary flood: Assessing risk of drinking water well contamination. Appl. Geogr. 2017, 80, 78–85. [Google Scholar] [CrossRef]
- Gamble, J.M.; Eurich, D.T.; Johnson, J.A. A comparison of drug coverage in Alberta before and after the introduction of the national common drug review process. Healthc. Policy 2010, 6, e117–e144. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pieters, W.R.; Wilson, K.K.; Smith, H.C.; Tamminga, J.J.; Sondhi, S. Salmeterol/fluticasone propionate versus fluticasone propionate plus montelukast: A cost-effective comparison for asthma. Treat Respir. Med. 2005, 4, 129–138. [Google Scholar] [CrossRef] [PubMed]
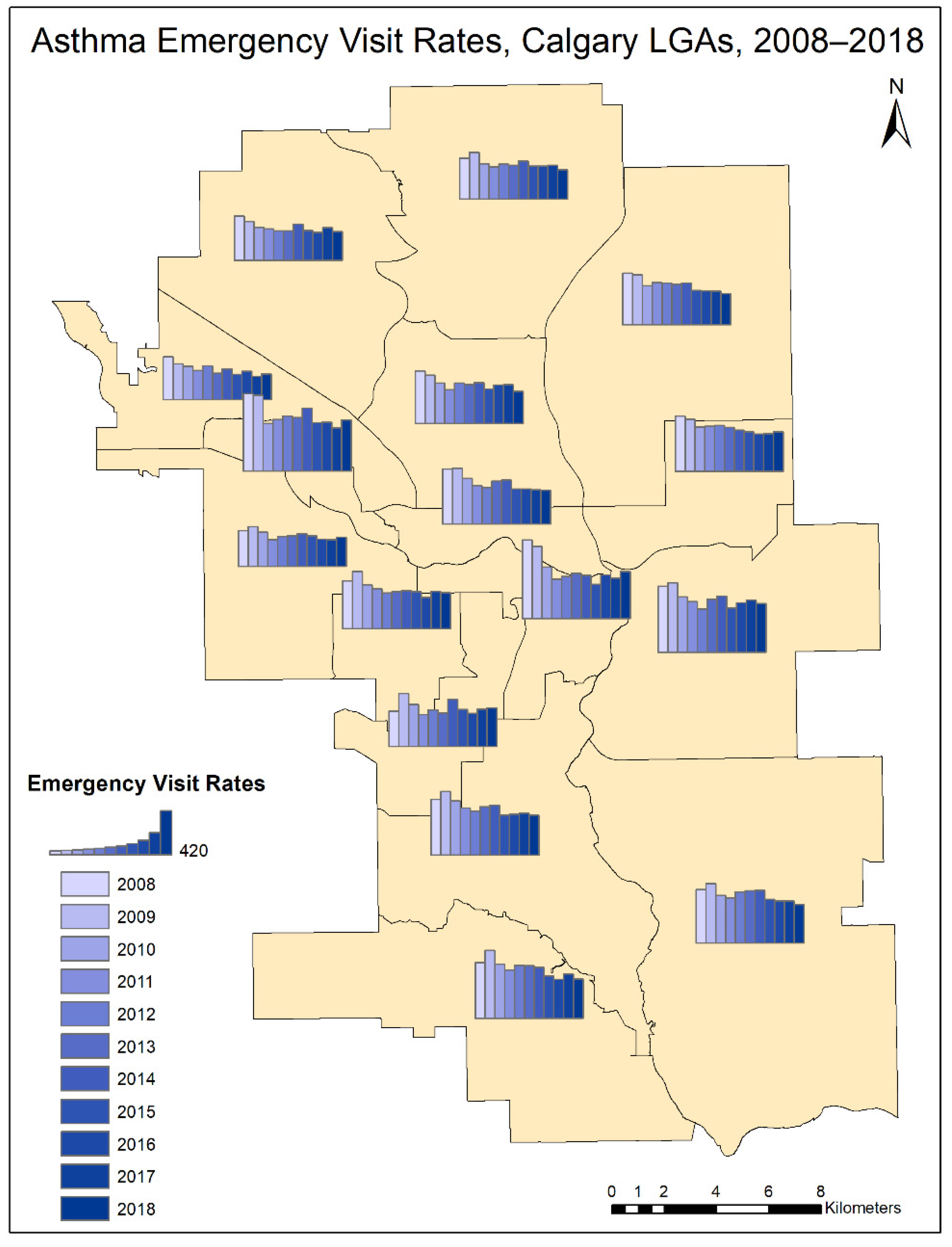
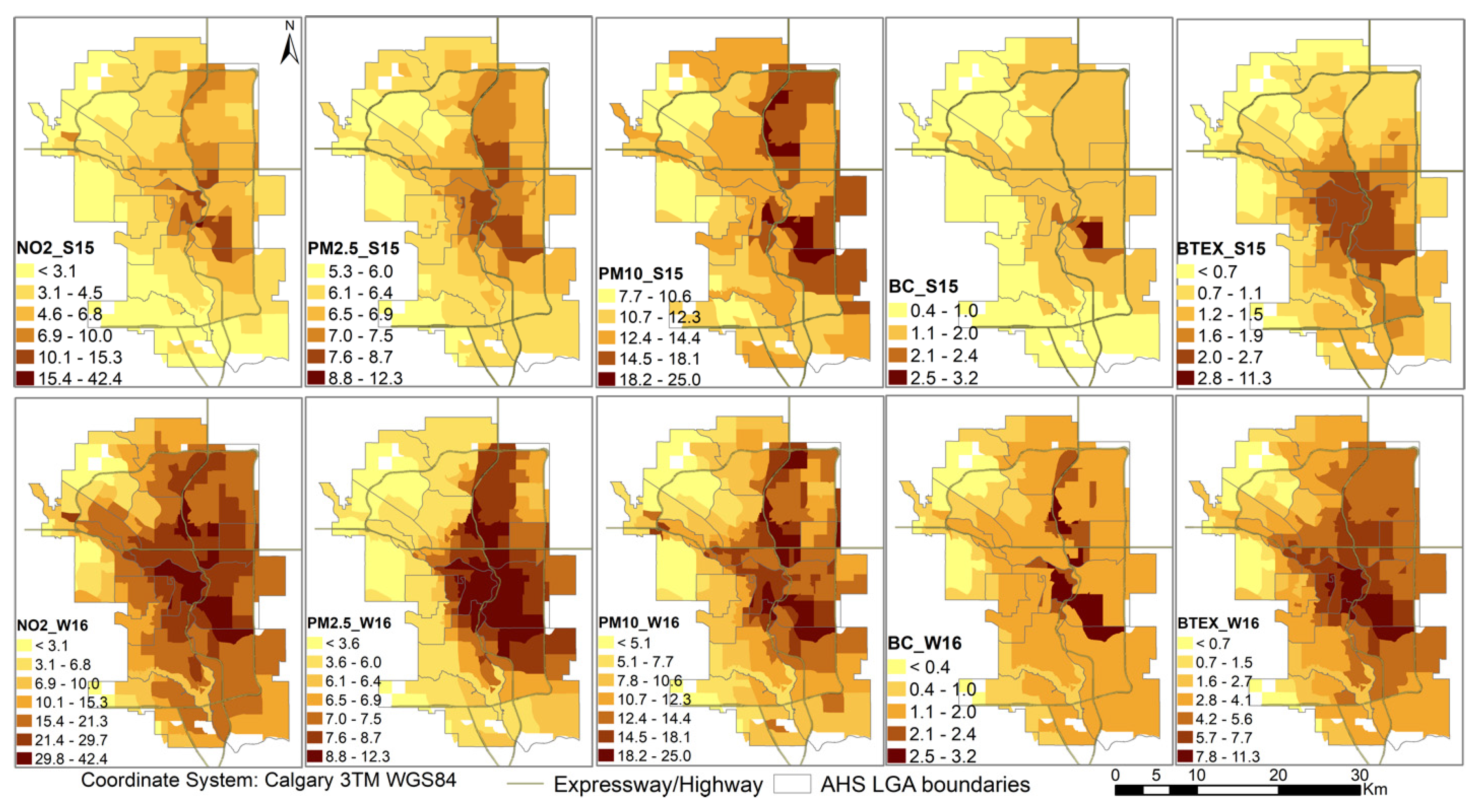
| Year | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Min. | 375.22 | 381.30 | 352.66 | 283.96 | 314.41 | 280.88 | 327.33 | 269.88 | 286.47 | 246.93 | 274.96 |
| Mean | 566.23 | 591.22 | 467.70 | 422.65 | 435.30 | 441.52 | 471.50 | 392.82 | 393.20 | 399.67 | 393.29 |
| Max. | 839.00 | 810.94 | 593.65 | 549.35 | 587.23 | 571.44 | 669.66 | 516.43 | 531.83 | 560.67 | 547.85 |
| Std. Dev. | 135.40 | 130.20 | 81.84 | 85.78 | 83.10 | 97.36 | 93.10 | 64.47 | 74.21 | 75.63 | 80.99 |
| S-W | 0.92 | 0.97 | 0.94 | 0.94 | 0.96 | 0.94 | 0.97 | 0.98 | 0.96 | 0.97 | 0.94 |
| (p-value) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) |
| Year | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Moran I | 0.50 | 0.64 | 0.60 | 0.67 | 0.67 | 0.70 | 0.71 | 0.67 | 0.64 | 0.68 | 0.63 |
| z(I) | 1.90 | 2.38 | 2.28 | 2.46 | 2.46 | 2.57 | 2.60 | 2.48 | 2.42 | 2.51 | 2.36 |
| p-value | (*) | (**) | (**) | (***) | (***) | (***) | (***) | (***) | (**) | (***) | (**) |
| Pollutants | NO2_S | NO2_W | PM2.5_S | PM2.5_W | PM10_S | PM10_W | BC_S | BC_W | BTEX_S | BTEX_W |
|---|---|---|---|---|---|---|---|---|---|---|
| AEVR_08 | 0.32 | 0.22 | 0.31 | 0.30 | 0.37 | 0.11 | 0.35 | 0.25 | 0.35 | 0.29 |
| (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | |
| AEVR_09 | 0.19 | 0.24 | 0.22 | 0.32 | 0.29 | 0.14 | 0.24 | 0.27 | 0.56 | 0.40 |
| (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (**) | (ns) | |
| AEVR_10 | 0.21 | 0.32 | 0.24 | 0.43 | 0.34 | 0.21 | 0.28 | 0.37 | 0.55 | 0.49 |
| (ns) | (ns) | (ns) | (*) | (ns) | (ns) | (ns) | (ns) | (**) | (**) | |
| AEVR_11 | 0.14 | 0.23 | 0.16 | 0.38 | 0.39 | 0.22 | 0.24 | 0.34 | 0.24 | 0.25 |
| (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | |
| AEVR_12 | 0.23 | 0.17 | 0.27 | 0.30 | 0.50 | 0.15 | 0.32 | 0.29 | 0.37 | 0.17 |
| (ns) | (ns) | (ns) | (ns) | (**) | (ns) | (ns) | (ns) | (ns) | (ns) | |
| AEVR_13 | 0.26 | 0.19 | 0.26 | 0.33 | 0.46 | 0.16 | 0.32 | 0.28 | 0.36 | 0.25 |
| (ns) | (ns) | (ns) | (ns) | (*) | (ns) | (ns) | (ns) | (ns) | (ns) | |
| AEVR_14 | 0.21 | 0.19 | 0.22 | 0.33 | 0.38 | 0.14 | 0.24 | 0.27 | 0.48 | 0.32 |
| (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (*) | (ns) | |
| AEVR_15 | 0.05 | 0.19 | 0.11 | 0.32 | 0.29 | 0.14 | 0.14 | 0.27 | 0.42 | 0.30 |
| (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | |
| AEVR_16 | 0.43 | 0.26 | 0.41 | 0.37 | 0.56 | 0.21 | 0.44 | 0.31 | 0.44 | 0.35 |
| (*) | 0.33 | 0.12 | 0.16 | (**) | 0.43 | (*) | 0.24 | (*) | 0.18 | |
| AEVR_17 | 0.11 | 0.16 | 0.14 | 0.28 | 0.33 | 0.09 | 0.17 | 0.21 | 0.39 | 0.32 |
| (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | (ns) | |
| AEVR_18 | 0.26 | 0.33 | 0.30 | 0.43 | 0.37 | 0.24 | 0.31 | 0.38 | 0.55 | 0.44 |
| (ns) | (ns) | (ns) | (*) | (ns) | (ns) | (ns) | (ns) | (**) | (*) |
| Pollutants | NO2_S | NO2_W | PM2.5_S | PM2.5_W | PM10_S | PM10_W | BC_S | BC_W | BTEX_S | BTEX_W |
|---|---|---|---|---|---|---|---|---|---|---|
| Low Income | 0.68 | 0.67 | 0.66 | 0.57 | 0.47 | 0.56 | 0.62 | 0.56 | 0.40 | 0.52 |
| (**) | (**) | (*) | (**) | (*) | (**) | (**) | (**) | (ns) | (**) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertazzon, S.; Calder-Bellamy, C.; Shahid, R.; Couloigner, I.; Wong, R. A Preliminary Spatial Analysis of the Association of Asthma and Traffic-Related Air Pollution in the Metropolitan Area of Calgary, Canada. Atmosphere 2020, 11, 1066. https://doi.org/10.3390/atmos11101066
Bertazzon S, Calder-Bellamy C, Shahid R, Couloigner I, Wong R. A Preliminary Spatial Analysis of the Association of Asthma and Traffic-Related Air Pollution in the Metropolitan Area of Calgary, Canada. Atmosphere. 2020; 11(10):1066. https://doi.org/10.3390/atmos11101066
Chicago/Turabian StyleBertazzon, Stefania, Caitlin Calder-Bellamy, Rizwan Shahid, Isabelle Couloigner, and Richard Wong. 2020. "A Preliminary Spatial Analysis of the Association of Asthma and Traffic-Related Air Pollution in the Metropolitan Area of Calgary, Canada" Atmosphere 11, no. 10: 1066. https://doi.org/10.3390/atmos11101066
APA StyleBertazzon, S., Calder-Bellamy, C., Shahid, R., Couloigner, I., & Wong, R. (2020). A Preliminary Spatial Analysis of the Association of Asthma and Traffic-Related Air Pollution in the Metropolitan Area of Calgary, Canada. Atmosphere, 11(10), 1066. https://doi.org/10.3390/atmos11101066





