Internal Structure of Asian Dust Particles over the Western North Pacific: Analyses Using Focused Ion Beam and Transmission Electron Microscopy
Abstract
1. Introduction
2. Dust Aerosol Sampling and Laboratory Analysis
2.1. Shipboard Sample Collection and Peripheral Aerosol Measurement
2.2. Preparation of Particle Cross-Sections with FIB and Detailed Analysis Using TEM-EDS-SAED
3. Results
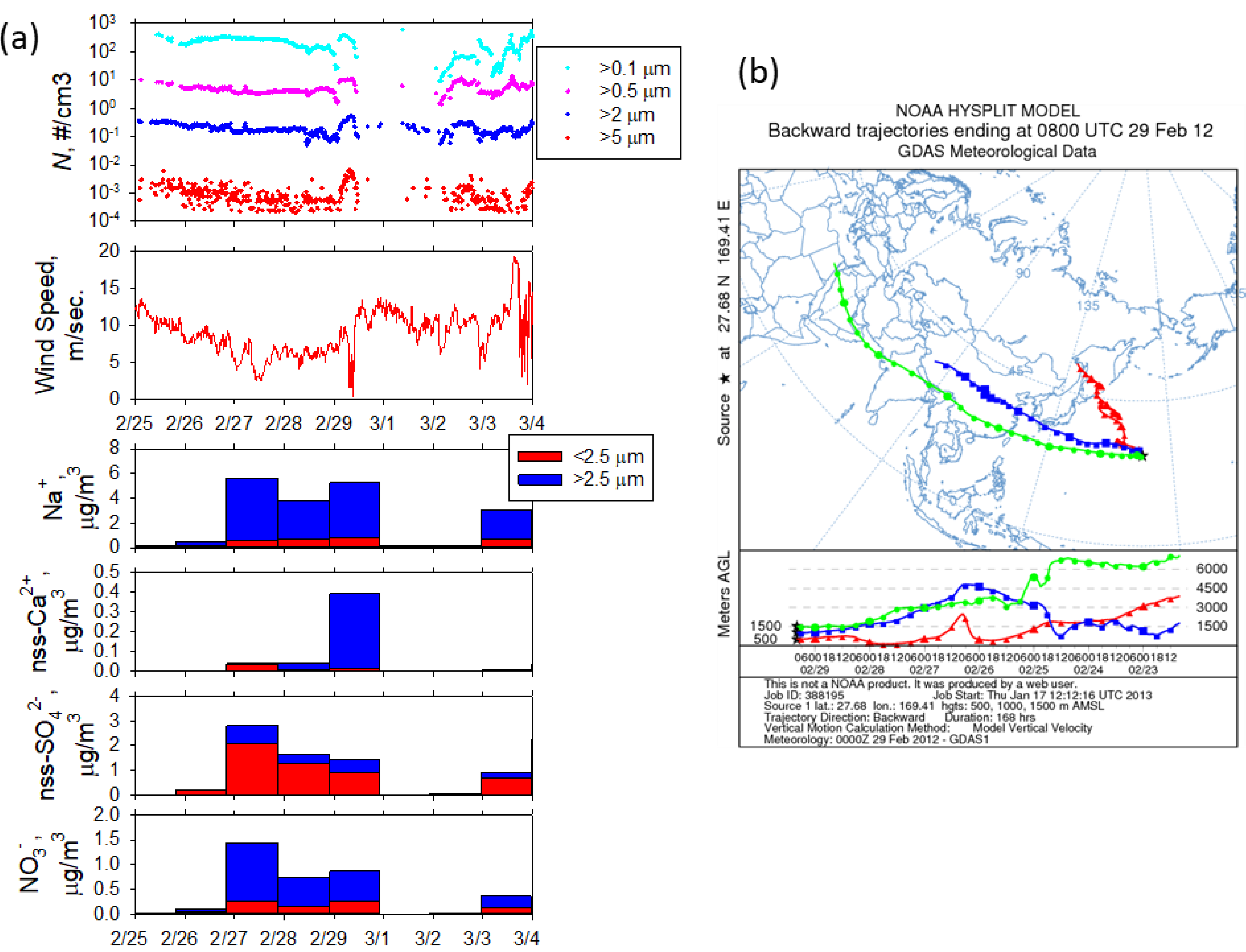
3.1. Sampling Condition of Long-Range Transported Dust Particles
3.2. Dust Particles Collected at the Western North Pacific
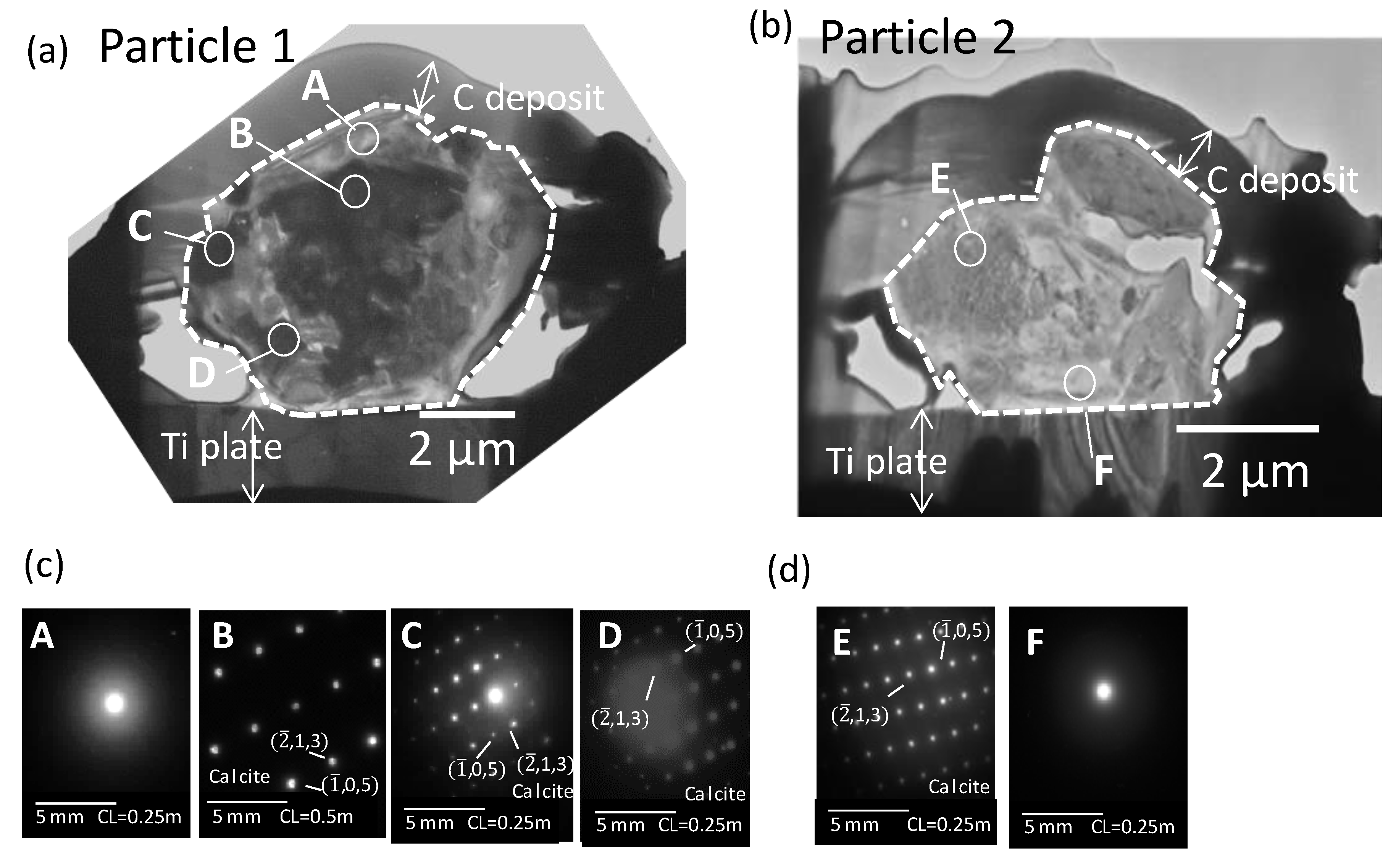
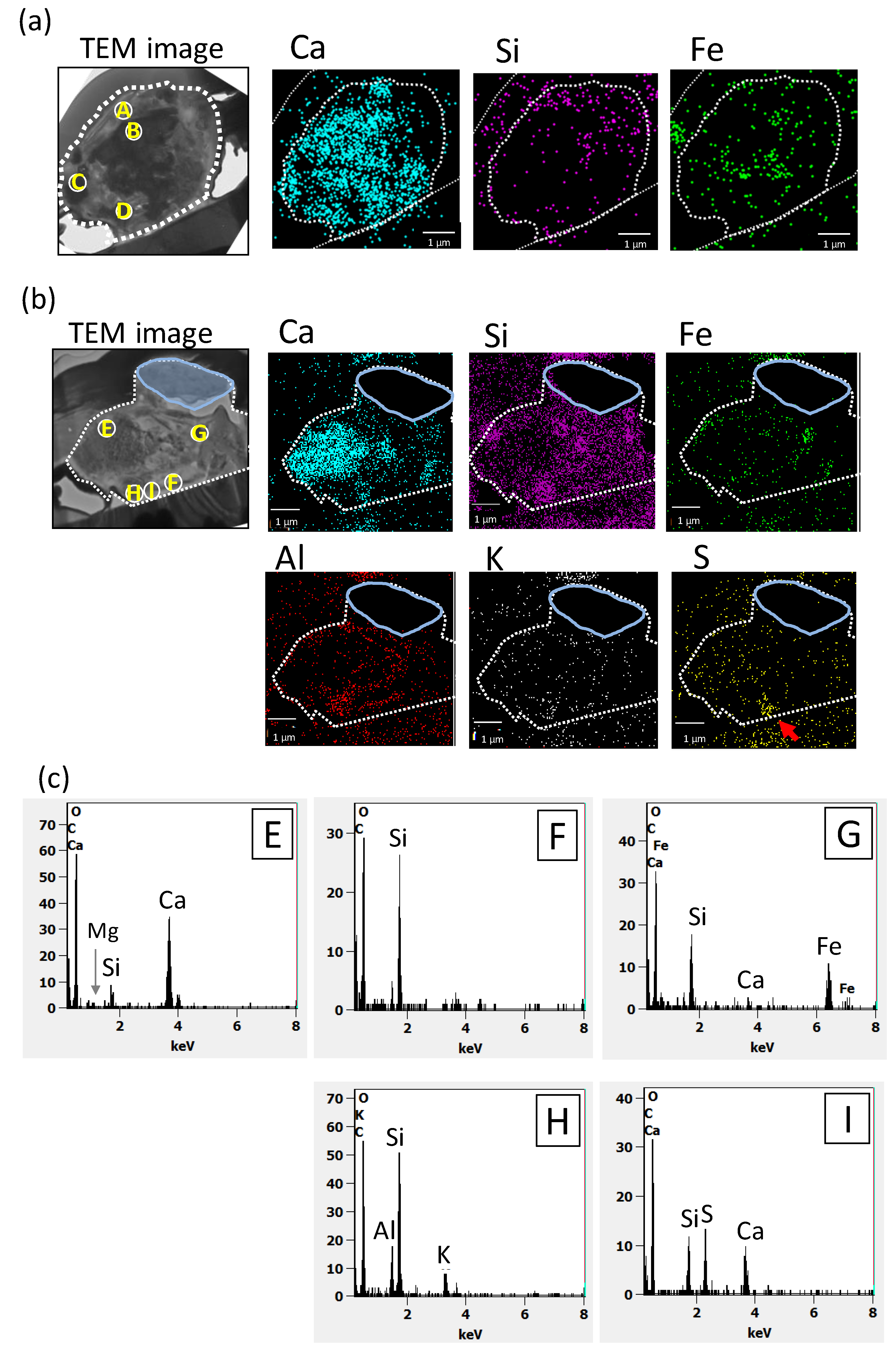
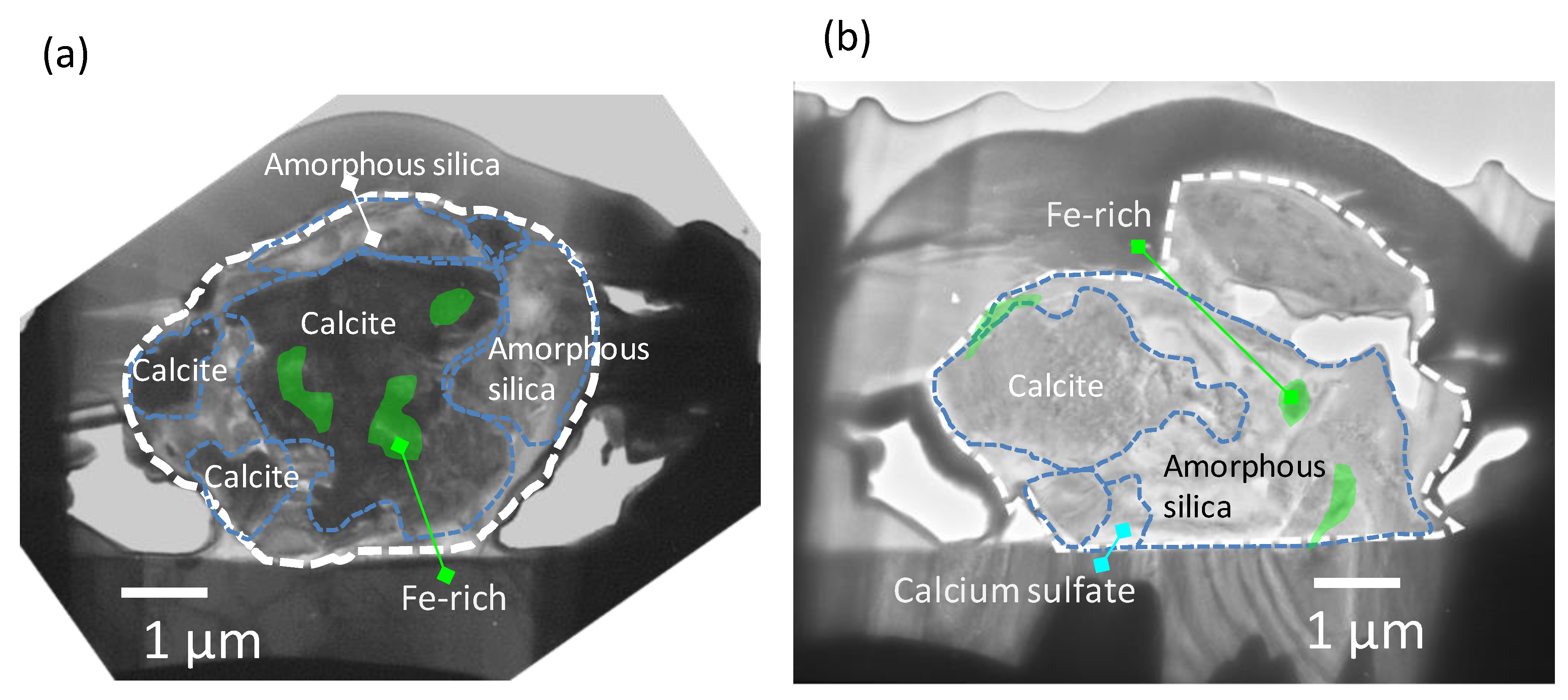
3.3. Detailed TEM Analysis of Dust Particle Cross-Sections
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Uematsu, M.; Duce, R.A.; Prospero, J.M.; Chen, L.; Merrill, J.T.; McDonald, R.L. Transport of mineral aerosol from Asia over the North Pacific Ocean. J. Geophys. Res. 1893, 88, 5343–5352. [Google Scholar] [CrossRef]
- Iwamoto, Y.; Yumimoto, K.; Toratani, M.; Tsuda, A.; Miura, K.; Uno, I.; Uematsu, M. Biogeochemical implications of increased mineral particle concentrations in surface waters of the northwestern North Pacific during an Asian dust event. Geophys. Res. Lett. 2011, 38, L01604. [Google Scholar] [CrossRef]
- Harrison, P.J.; Boyd, P.W.; Varela, D.E.; Takeda, S.; Shiomoto, A.; Odate, T. Comparison of factors controlling phytoplankton productivity in the NE and NW subarctic Pacific gyres. Prog. Oceanogr. 1999, 43, 205–234. [Google Scholar] [CrossRef]
- Ingall, E.D.; Feng, Y.; Longo, A.F.; Lai, B.; Shelley, R.U.; Landing, W.M.; Morton, P.L.; Nenes, A.; Mihalopoulos, N.; Violaki, K.; et al. Enhanced Iron Solubility at Low pH in Global Aerosols. Atmosphere 2018, 9, 201. [Google Scholar] [CrossRef]
- Baker, A.R.; Jickells, T.D. Mineral particle size as a control on aerosol iron solubility. Geophys. Res. Lett. 2006, 33. [Google Scholar] [CrossRef]
- Sullivan, R.C.; Guazzotti, S.A.; Sodeman, D.A.; Prather, K.A. Direct observations of the atmospheric processing of Asian mineral dust. Atmos. Chem. Phys. 2007, 7, 1213–1236. [Google Scholar] [CrossRef]
- Journet, E.; Desboeufs, K.V.; Caquineau, S.; Colin, J.L. Mineralogy as a critical factor of dust iron solubility. Geophys. Res. Lett. 2008, 35. [Google Scholar] [CrossRef]
- Kumar, A.; Sarin, M. Aerosol iron solubility in a semi-arid region: Temporal trend and impact of anthropogenic sources. Tellus Ser. B Chem. Phys. Meteorol. 2009, 62, 125–132. [Google Scholar] [CrossRef]
- Li, J.; Osada, K. Water-insoluble particles in spring snow at Mt. Tateyama, Japan: Characteristics of the shape factors and size distribution in relation with their origin and transportation. J. Meteorol. Sci. Jpn. 2007, 85, 137–149. [Google Scholar] [CrossRef]
- Li, J.; Osada, K. Preferential settling of elongated mineral dust particles in the atmosphere. Geophys. Res. Lett. 2007, 34. [Google Scholar] [CrossRef]
- Jeong, G.Y.; Nousiainen, T. TEM analysis of the internal structures and mineralogy of Asian dust particles and the implications for optical modeling. Atmos. Chem. Phys. 2014, 14, 7233–7254. [Google Scholar] [CrossRef]
- Jeong, G.Y.; Park, M.Y.; Kandler, K.; Nousiainen, T.; Kemppinen, O. Mineralogical properties and internal structures of individual fine particles of Saharan dust. Atmos. Chem. Phys. 2016, 16, 12397–12410. [Google Scholar] [CrossRef]
- Jeong, G.Y.; Kim, J.Y.; Seo, J.; Kim, G.M.; Jin, H.C.; Chun, Y. Long-range transport of giant particles in Asian dust identified by physical, mineralogical, and meteorological analysis. Atmos. Chem. Phys. 2014, 14, 505–521. [Google Scholar] [CrossRef]
- Matsuki, A.; Iwasaka, Y.; Shi, G.; Zhang, D.; Trochkine, D.; Yamada, M.; Kim, Y.-S.; Chen, B.; Nagatani, T.; Miyazawa, T.; et al. Morphological and chemical modification of mineral dust: Observational insight into the heterogeneous uptake of acidic gases. Geophys. Res. Lett. 2005, 32, L22806. [Google Scholar] [CrossRef]
- Okada, K.; Qin, Y.; Kai, K. Elemental composition and mixing properties of atmospheric mineral particles collected in Hohhot, China. Atmos. Res. 2005, 73, 45–67. [Google Scholar] [CrossRef]
- Zhang, D.; Iwasaka, Y. Nitrate and sulfate in individual Asian dust-storm particles in Beijing, China in spring of 1995 and 1996. Atmos. Environ. 1999, 33, 3213–3223. [Google Scholar] [CrossRef]
- Okada, K.; Naruse, H.; Tanaka, T.; Nemoto, O.; Iwasaka, Y.; Wu, P.-M.; Ono, A.; Duce, R.A.; Uematsu, M.; Merrill, J.T.; et al. X-ray spectrometry of individual Asian dust-storm particles over the Japanese islands and the North Pacific Ocean. Atmos. Environ. 1990, 24A, 1369–1378. [Google Scholar] [CrossRef]
- Conny, J.M.; Willis, R.D.; Ortiz-Montalvo, D.L. Analysis and optical modeling of individual heterogeneous Asian Dust Particles collected at Mauna Loa Observatory. J. Geophys. Res. Atmos. 2019, 124, 270–2723. [Google Scholar] [CrossRef]
- Reid, E.A.; Reid, J.S.; Meier, M.M.; Dunlap, M.R.; Cliff, S.S.; Broumas, A.; Perry, K.; Maring, H. Characterization of African dust transported to Puerto Rico by individual particle and size segregated bulk analysis. J. Geophys. Res. 2003, 108, 8591. [Google Scholar] [CrossRef]
- Li, W.; Xu, L.; Liu, X.; Zhang, J.; Lin, Y.; Yao, X.; Gao, H.; Zhang, D.; Chen, J.; Wang, W.; et al. Air pollution-aerosol interactions produce more bioavailable iron for ocean ecosystems. Sci. Adv. 2017, 3, e1601749. [Google Scholar] [CrossRef]
- Kaegi, R.; Gasser, P. Application of the focused ion beam technique in aerosol science: Detailed investigation of selected, airborne particles. J. Microsc. 2006, 224, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Grassian, V.H.; Saraf, L.V.; Laskin, A. Chemical imaging analysis of environmental particles using the focused ion beam/scanning electron microscopy technique: Microanalysis insights into atmospheric chemistry of fly ash. Analyst 2013, 138, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Conny, J.M. Internal composition of atmospheric dust particles from focused ion-beam scanning electron microscopy. Environ. Sci. Technol. 2013, 47, 8575–8581. [Google Scholar] [CrossRef]
- Miki, Y.; Ueda, S.; Miura, K.; Furutani, H.; Uematsu, M. Atmospheric Fe-containing particles over the North Pacific Ocean: The mixing states with water soluble materials. Earozoru Kenkyo 2014, 29, 104–111. [Google Scholar]
- Cui, Y.; Suzuki, S.; Omori, Y.; Wong, S.-K.; Ijichi, M.; Kaneko, R.; Kameyama, S.; Tanimoto, H.; Hamasaki, K. Abundance and distribution of dimethylsulfoniopropionate degradation genes and the corresponding bacterial community structure at dimethyl sulfide hot spots in the tropical and subtropical Pacific Ocean. Appl. Environ. Microbiol. 2015, 81, 4184–4194. [Google Scholar] [CrossRef]
- Kamogawa, M.; Ohhora, O.; Ueda, S.; Miura, K.; Yajima, K.; Hashamoto, S.; Nakamura, M.; Kakinami, Y.; Furutani, H.; Uematsu, M. Simultaneous observations of atmospheric electric field, aerosols, and c1ouds on the R/V Hakuho Maru over the Pacific Ocean. J. Atmos. Electr. 2014, 34, 12–26. [Google Scholar]
- Omori, Y.; Tanimoto, H.; Inomata, S.; Ikeda, K.; Iwata, T.; Kameyama, S.; Uematsu, M.; Gamo, T.; Ogawa, H.; Furuya, K. Sea-to-air flux of dimethyl sulfide in the South and North Pacific Ocean as measured by proton transfer reaction-mass spectrometry coupled with the gradient flux technique. J. Geophys. Res. Atmos. 2017, 122, 7216–7231. [Google Scholar] [CrossRef]
- Ueda, S.; Miura, K.; Kawata, R.; Furutani, H.; Uematsu, M.; Omori, Y.; Tanimoto, H. Number–size distribution of aerosol particles and new particle formation events in tropical and subtropical Pacific Ocean. Atmos. Environ. 2016, 142, 324–339. [Google Scholar] [CrossRef]
- Jung, J.; Furutani, H.; Uematsu, M.; Park, J. Distribution of atmospheric non-sea-salt sulfate and methanesulfonic acid over the Pacific Ocean between 48°N and 55°S during summer. Atmos. Environ. 2014, 99, 374–384. [Google Scholar] [CrossRef]
- Rolph, G.; Stein, A.; Stunder, B. Real-time Environmental Applications and Display sYstem: READY. Environ. Model. Softw. 2017, 95, 210–228. [Google Scholar] [CrossRef]
- Stein, A.F.; Draxler, R.R.; Rolph, G.D.; Stunder, B.J.B.; Cohen, M.D.; Ngan, F. NOAA’s HYSPLIT atmospheric transport and dispersion modeling system. Bull. Am. Meteorol. Soc. 2015, 96, 2059–2077. [Google Scholar] [CrossRef]
- Kogure, T. Geometry of electron diffraction and its calculation. Kenbikyo 2015, 50, 6–10. [Google Scholar] [CrossRef]
- Kawakam, N.; Osada, K.; Nishita, C.; Yabuki, M.; Kobayashi, H.; Hara, K.; Shiobara, M. Factors controlling sea salt modification and dry deposition of nonsea-salt components to the ocean. J. Geophys. Res. 2008, 113, D14216. [Google Scholar] [CrossRef]
- Chessin, H.; Hamilton, W.C.; Post, B. Position and thermal parameters of oxygen atoms in calcite. Acta Cryst. 1965, 18, 689–693. [Google Scholar] [CrossRef]

| Paper | Sampling Site | Date | Site Type | Method | Analyzed Particle Size | Co-Existence of Ca and S |
|---|---|---|---|---|---|---|
| Okada et al. [14] | Hohhot (China) | April, 1991 | urban, near source | TEM-EDX | 0.2–5 μm | Most Ca-rich particles contained S |
| Matsuki et al. [15] | Beijing (China) | October, 2004 | suburban, tethered balloon | TEM-EDX | >1 μm | 62%–78% Ca-rich and carbonate particles contained S |
| Zhang and Iwasaka [16] | Beiing (China) | Spring of 1995–1996 | urban | TEM-EDX | >1 μm | 14.6% dust contained S |
| Jeong and Nousianinen [11] | Seoul (Korea) | March, 2009 | urban | FIB+TEM (slicing, SAED, EDS) | ca. 30 μm | - |
| Jeong et al. [13] | Deokjeok Island (Korea) | March and April, 2012 | coast, mid-distant from dust source | FIB+TEM (slicing, SAED, EDS) | <60 μm | - |
| Okada et al. [17] | Nagasaki and Nagoya (Japan) | April and May, 1986 | urban, mid-distant from dust source | TEM-EDX | 2–4 μm | <0.2 (Nagasaki) and 0.78 (Nagoya) mean weight ratio of S/Ca |
| This study | Western North Pacific | February, 2012 | remote, HNLC area | FIB+TEM (slicing, SAED, EDS) | ca. 6 μm | Large Ca-rich domains not containing S |
| Okada et al. [17] | Eastern North Pacific | April and May, 1986 | remote, north of Hawaii | TEM-EDX | 2–4 μm | 0.38–1.08 mean weight ratio of S/Ca |
| Conny et al. [18] | Hawaii (USA) | March, 2011 | remote | FIB-SEM (elemental mapping, tomography) | 0.4–10 μm | S was detected in all Ca-rich particles |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ueda, S.; Miki, Y.; Kato, H.; Miura, K.; Nakayama, H.; Furutani, H.; Uematsu, M. Internal Structure of Asian Dust Particles over the Western North Pacific: Analyses Using Focused Ion Beam and Transmission Electron Microscopy. Atmosphere 2020, 11, 78. https://doi.org/10.3390/atmos11010078
Ueda S, Miki Y, Kato H, Miura K, Nakayama H, Furutani H, Uematsu M. Internal Structure of Asian Dust Particles over the Western North Pacific: Analyses Using Focused Ion Beam and Transmission Electron Microscopy. Atmosphere. 2020; 11(1):78. https://doi.org/10.3390/atmos11010078
Chicago/Turabian StyleUeda, Sayako, Yusuke Miki, Hiroki Kato, Kazuhiko Miura, Hiroyasu Nakayama, Hiroshi Furutani, and Mitsuo Uematsu. 2020. "Internal Structure of Asian Dust Particles over the Western North Pacific: Analyses Using Focused Ion Beam and Transmission Electron Microscopy" Atmosphere 11, no. 1: 78. https://doi.org/10.3390/atmos11010078
APA StyleUeda, S., Miki, Y., Kato, H., Miura, K., Nakayama, H., Furutani, H., & Uematsu, M. (2020). Internal Structure of Asian Dust Particles over the Western North Pacific: Analyses Using Focused Ion Beam and Transmission Electron Microscopy. Atmosphere, 11(1), 78. https://doi.org/10.3390/atmos11010078





