Long-Range Transport Influence on Key Chemical Components of PM2.5 in the Seoul Metropolitan Area, South Korea, during the Years 2012–2016
Abstract
1. Introduction
2. Data and Methods
2.1. Surface Observation Data
2.2. Air-Quality Modeling
2.3. Model Performance Evaluation
2.4. Estimation of Chinese Emission Impacts
3. Results and Discussion
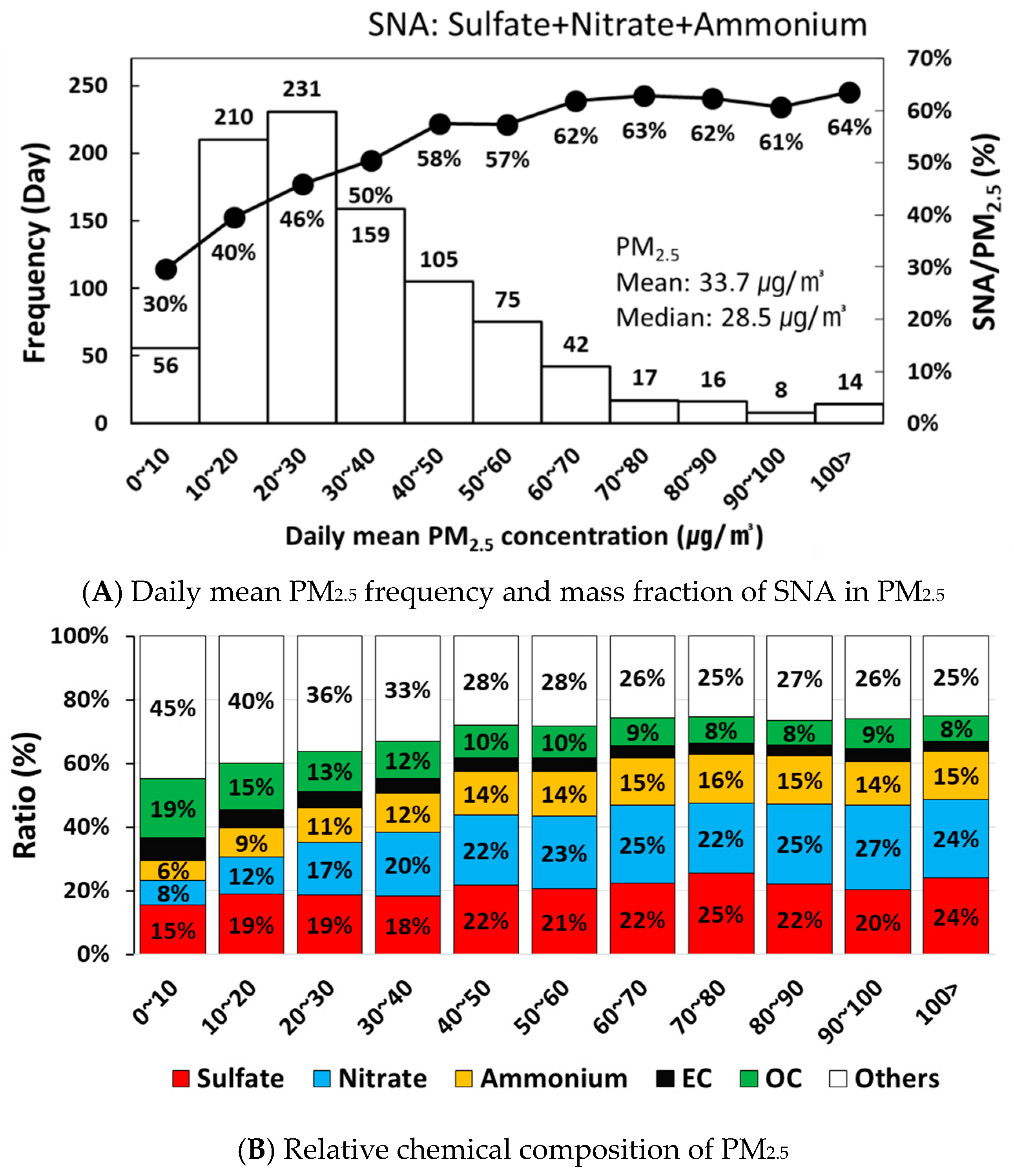
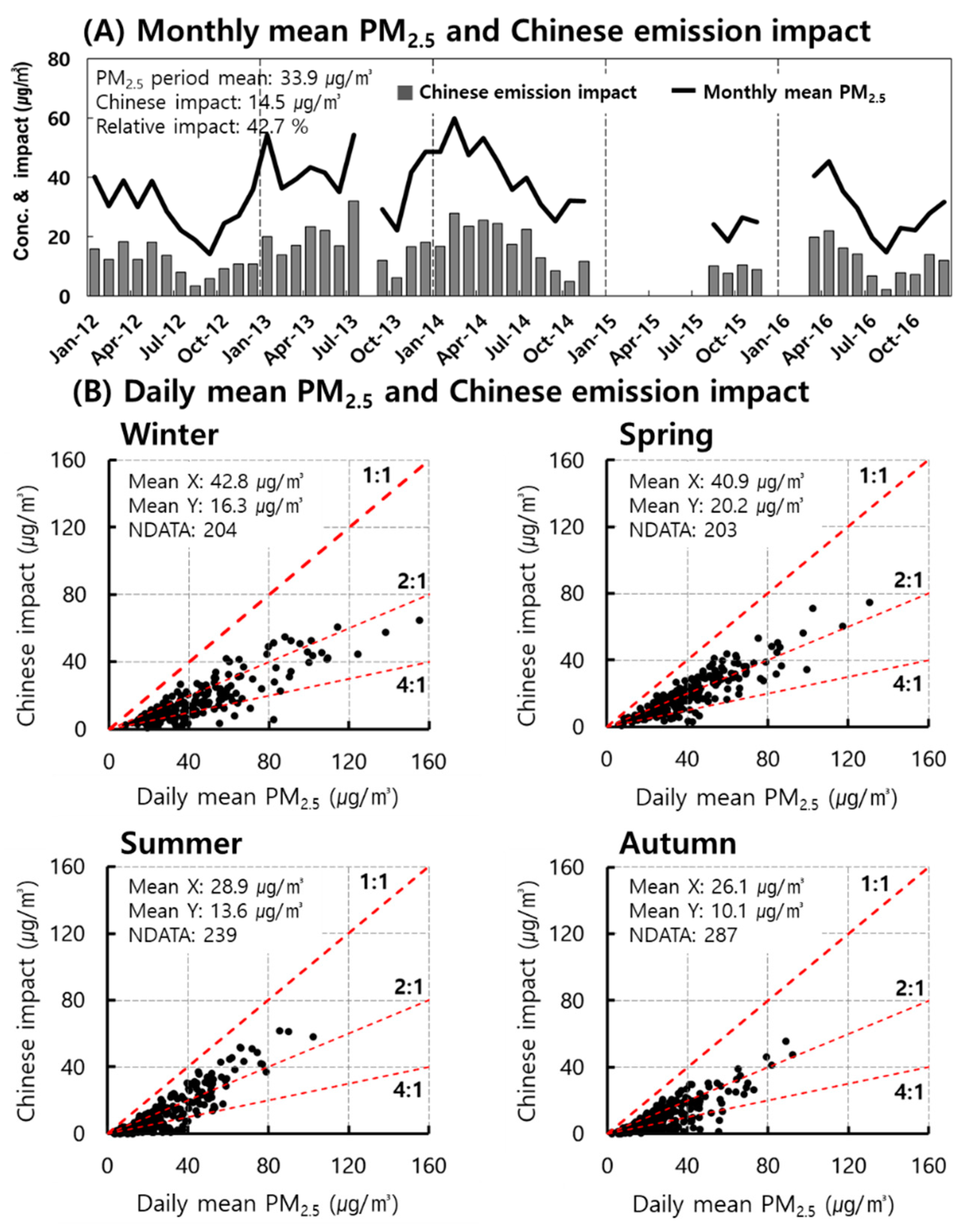
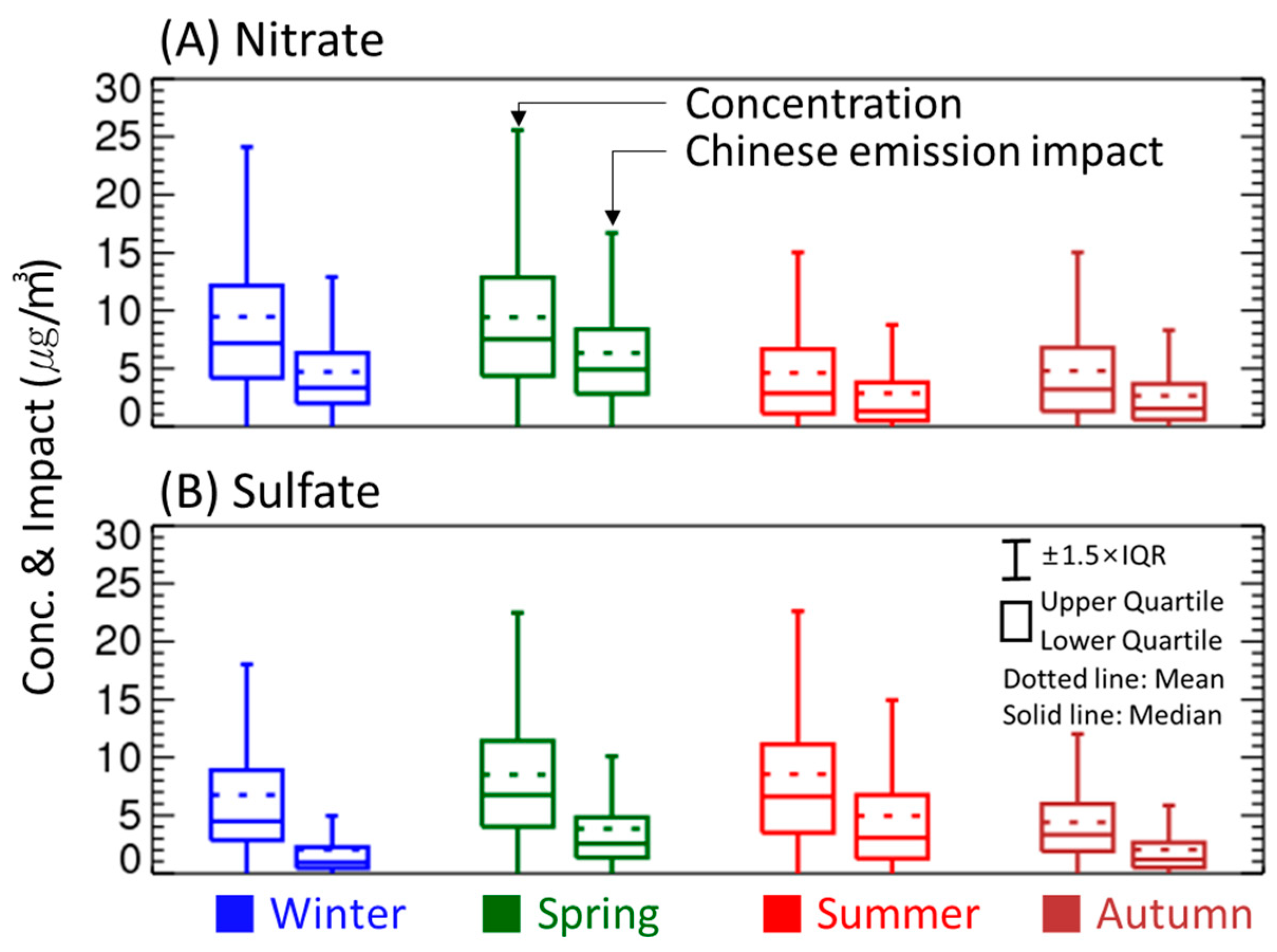
3.1. Concentration and Chemical Composition of PM2.5
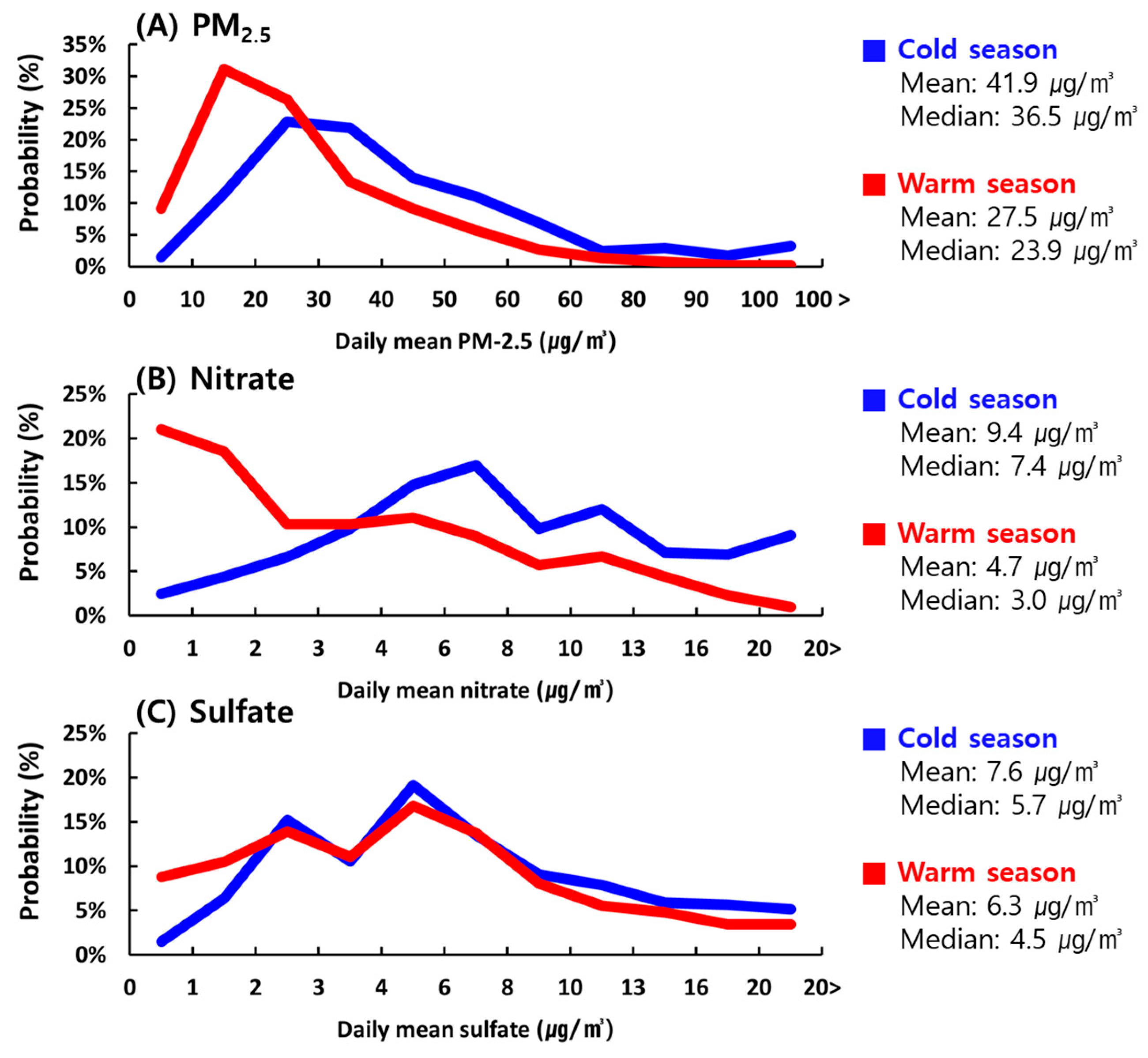
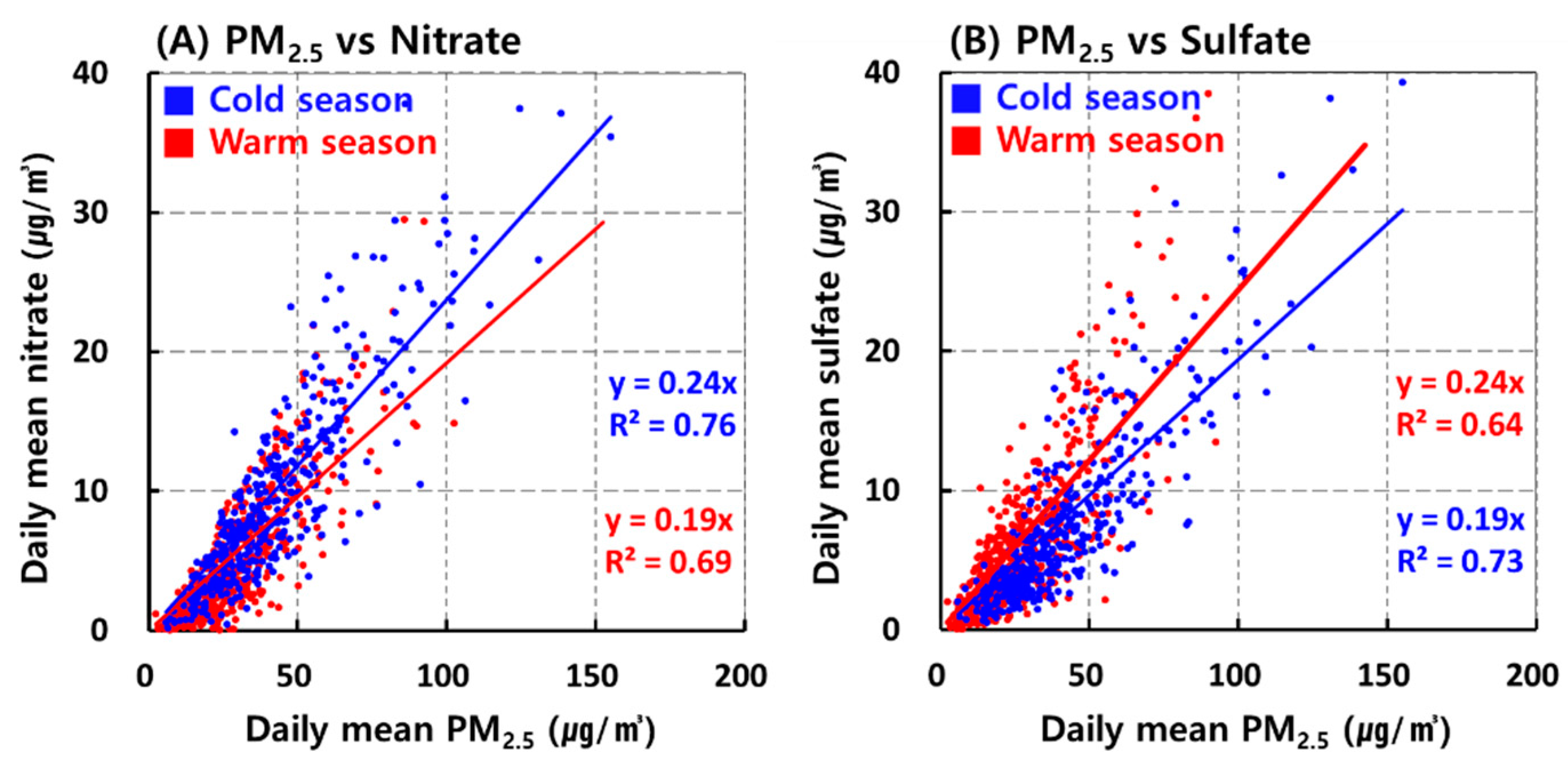
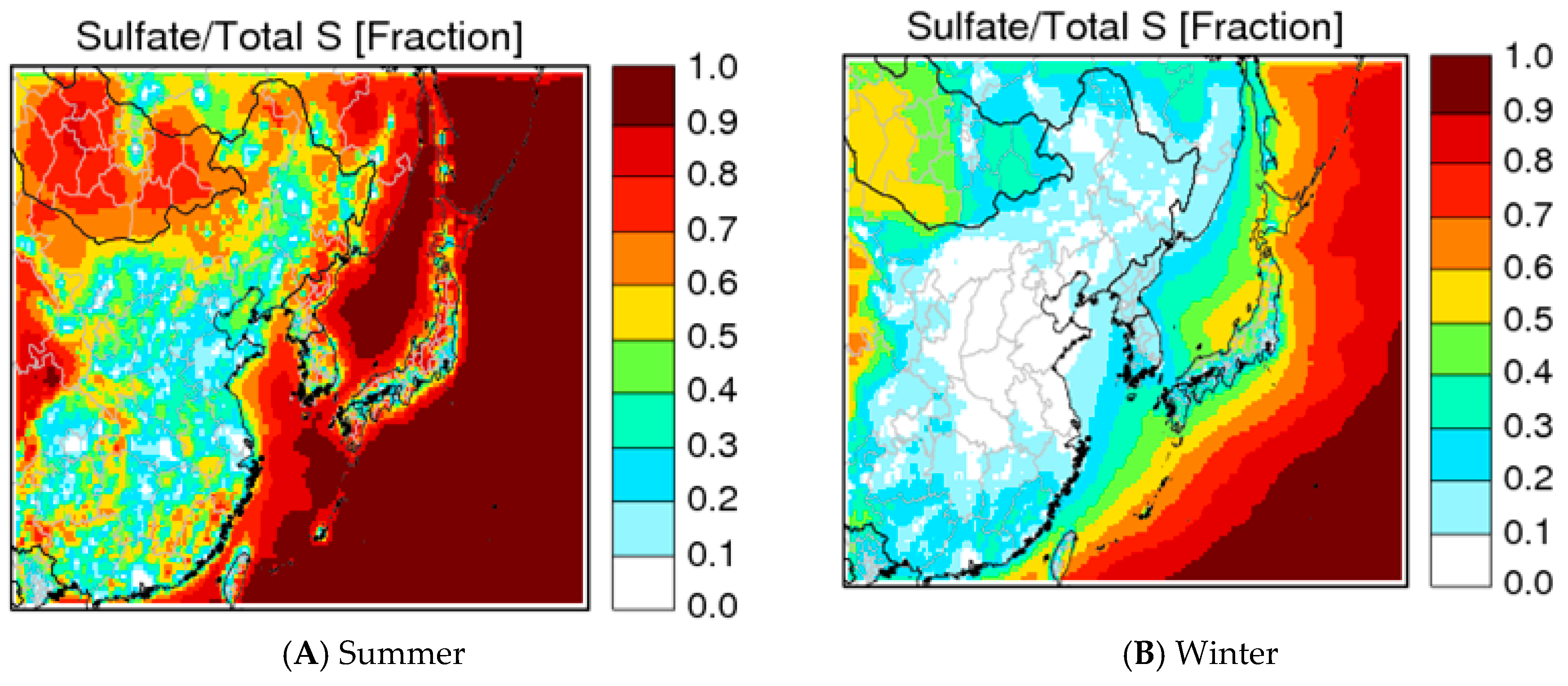
3.2. Seasonal Changes
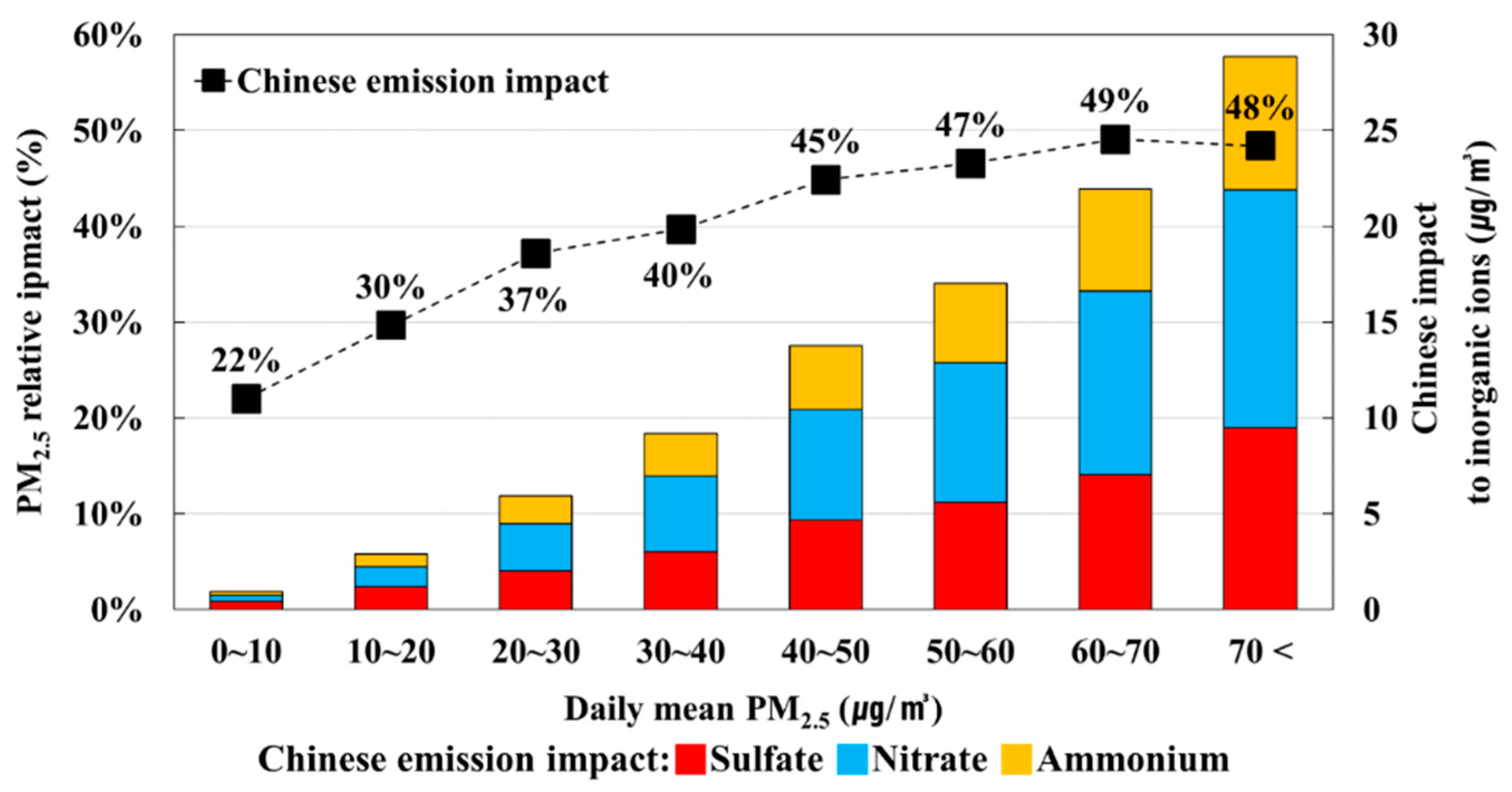
3.3. Seasonal Chinese Emission Impacts on PM2.5
4. Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Snider, G.; Weagle, C.L.; Murdymootoo, K.K.; Ring, A.; Ritchie, Y.; Stone, E.; Walsh, A.; Akoshile, C.; Anh, N.X.; Balasubramanian, R.; et al. Variation in global chemical composition of PM 2.5:Emerging results from SPARTAN. Atmos. Chem. Phys. 2016, 16, 9629–9653. [Google Scholar]
- Cheng, Z.; Luo, L.; Wang, S.; Wang, Y.; Sharma, S.; Shimadera, H.; Wang, X.; Bressi, M.; de Miranda, R.M.; Jiang, J.; et al. Status and characteristics of ambient PM2.5 pollution in global megacities. Environ. Int. 2016, 89–90, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Choi, S.; Li, C.; Fioletov, V.E.; McLinden, C.A.; Joiner, J.; Krotkov, N.A.; Bian, H.; Janssens-Maenhout, G.; Darmenov, A.S. A new global anthropogenic SO2 emission inventory for the last decade: A mosaic of satellite-derived and bottom-up emissions. Atmos. Chem. Phys. 2018, 18, 16571–16586. [Google Scholar] [CrossRef]
- Park, E.H.; Heo, J.; Hirakura, S.; Hashizume, M.; Deng, F.; Kim, H.; Yi, S.-M. Characteristics of PM2.5 and its chemical constituents in Beijing, Seoul, and Nagasaki. Air Qual. Atmos. Health 2018, 11, 1167–1178. [Google Scholar]
- Moon, K.-J.; Park, S.-M.; Park, J.-S.; Song, I.-H.; Jang, S.-K.; Kim, J.-C.; Lee, S.-J. Chemical Characteristics and Source Apportionment ofPM 2.5 in Seoul Metropolitan Area in 2010. J. Korean Soc. Atmos. Environ. 2011, 27, 711–722, (In Korean with English Abstract). [Google Scholar]
- Zhang, X.Y.; Wang, Y.Q.; Niu, T.; Zhang, X.C.; Gong, S.L.; Zhang, Y.M.; Sun, J.Y. Atmospheric aerosol compositions in China: Spatial/temporal variability, chemical signature, regional haze distribution and comparisons with global aerosols. Atmos. Chem. Phys. 2012, 12, 779–799. [Google Scholar] [CrossRef]
- Li, H.; Ma, Y.; Duan, F.; He, K.; Zhu, L.; Huang, T.; Kimoto, T.; Ma, X.; Ma, T.; Xu, L.; et al. Typical winter haze pollution in Zibo, an industrial city in China: Characteristics, secondary formation, and regional contribution. Environ. Pollut. 2017, 229, 339–349. [Google Scholar] [CrossRef]
- Ma, Q.; Wu, Y.; Zhang, D.; Wang, X.; Xia, Y.; Liu, X.; Tian, P.; Han, Z.; Xia, X.; Wang, Y.; et al. Roles of regional transport and heterogeneous reactions in the PM2.5 increase during winter haze episodes in Beijing. Sci. Total Environ. 2017, 599–600, 246–253. [Google Scholar] [CrossRef]
- Saxena, M.; Sharma, A.; Sen, A.; Saxena, P.; Mandal, T.K.; Sharma, S.K.; Sharma, C. Water soluble inorganic species of PM10 and PM2.5 at an urban site of Delhi, India: Seasonal variability and sources. Atmos. Res. 2017, 184, 112–125. [Google Scholar]
- Dunker, A.M.; Yarwood, G.; Ortmann, J.P.; Wilson, G.M. Comparison of source apportionment and source sensitivity of ozone in a three-dimensional air quality model. Environ. Sci. Technol. 2002, 36, 2953–2964. [Google Scholar]
- Ghim, Y.S.; Kim, J.Y.; Chang, Y.-S. Concentration variations in particulate matter in Seoul associated with Asian dust and smog episodes. Aerosol Air Qual. Res. 2017, 17, 3128–3140. [Google Scholar] [CrossRef]
- Huang, X.H.; Bian, Q.; Ng, W.M.; Louie, P.K.; Yu, J.Z. Characterization of PM2. 5 major components and source investigation in suburban Hong Kong: A one year monitoring study. Aerosol Air Qual. Res. 2014, 14, 237–250. [Google Scholar] [CrossRef]
- Liu, H.-Y.; Dunea, D.; Iordache, S.; Pohoata, A. A Review of Airborne Particulate Matter Effects on Young Children’s Respiratory Symptoms and Diseases. Atmosphere 2018, 9, 150. [Google Scholar] [CrossRef]
- Di, Q.; Wang, Y.; Zanobetti, A.; Wang, Y.; Koutrakis, P.; Choirat, C.; Dominici, F.; Schwartz, J.D. Air Pollution and Mortality in the Medicare Population. N. Engl. J. Med. 2017, 376, 2513–2522. [Google Scholar] [CrossRef]
- Zhang, Q.; Jiang, X.; Tong, D.; Davis, S.J.; Zhao, H.; Geng, G.; Feng, T.; Zheng, B.; Lu, Z.; Streets, D.G.; et al. Transboundary health impacts of transported global air pollution and international trade. Nature 2017, 543, 705–709. [Google Scholar] [CrossRef]
- Yu, G.H.; Park, S.S.; Ghim, Y.S.; Shin, H.J.; Lim, C.S.; Ban, S.J.; Yu, J.A.; Kang, H.J.; Seo, Y.K.; Kang, K.S.; et al. Difference in Chemical Composition of PM2.5 and Investigation of its Causing Factors between 2013 and 2015 in Air Pollution Intensive Monitoring Stations. J. Korean Soc. Atmos. Environ. 2018, 34, 16–37, (In Korean with English Abstract). [Google Scholar] [CrossRef]
- Shin, H.J.; Park, S.-M.; Park, J.S.; Song, I.H.; Hong, Y.D. Chemical characteristics of high PM episodes occurring in Spring 2014, Seoul, Korea. Adv. Meteorol. 2016, 2016, 2424875. [Google Scholar] [CrossRef]
- Wang, L.; Hao, J.; He, K.; Wang, S.; Li, J.; Zhang, Q.; Streets, D.G.; Fu, J.S.; Jang, C.J.; Takekawa, H. A modeling study of coarse particulate matter pollution in Beijing: Regional source contributions and control implications for the 2008 Summer Olympics. J. Air Waste Manag. Assoc. 2008, 58, 1057–1069. [Google Scholar] [CrossRef]
- Zheng, B.; Tong, D.; Li, M.; Liu, F.; Hong, C.; Geng, G.; Li, H.; Li, X.; Peng, L.; Qi, J.; et al. Trends in China’s anthropogenic emissions since 2010 as the consequence of clean air actions. Atmos. Chem. Phys. 2018, 18, 14095–14111. [Google Scholar] [CrossRef]
- Zhang, Q.; Zheng, Y.; Tong, D.; Shao, M.; Wang, S.; Zhang, Y.; Xu, X.; Wang, J.; He, H.; Liu, W. Drivers of improved PM2. 5 air quality in China from 2013 to 2017. Proc. Natl. Acad. Sci. USA 2019, 116, 24463–24469. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, F.; Pilot, E.; Yu, J.; Nie, C.; Holdaway, J.; Yang, L.; Li, Y.; Wang, W.; Vardoulakis, S.; et al. Taking Action on Air Pollution Control in the Beijing-Tianjin-Hebei (BTH) Region: Progress, Challenges and Opportunities. IJERPH 2018, 15, 306. [Google Scholar] [CrossRef] [PubMed]
- KORUS-AQ. Introduction to the KORUS-AQ Rapid Synthesis Report. 2017. Available online: https://espo.nasa.gov/sites/default/files/documents/KORUS-AQ-ENG.pdf (accessed on 28 December 2019).
- Oh, H.-R.; Ho, C.-H.; Kim, J.; Chen, D.; Lee, S.; Choi, Y.-S.; Chang, L.-S.; Song, C.-K. Long-range transport of air pollutants originating in China: A possible major cause of multi-day high-PM10 episodes during cold season in Seoul, Korea. Atmos. Environ. 2015, 109, 23–30. [Google Scholar] [CrossRef]
- Kaneyasu, N.; Yamamoto, S.; Sato, K.; Takami, A.; Hayashi, M.; Hara, K.; Kawamoto, K.; Okuda, T.; Hatakeyama, S. Impact of long-range transport of aerosols on the PM2.5 composition at a major metropolitan area in the northern Kyushu area of Japan. Atmos. Environ. 2014, 97, 416–425. [Google Scholar] [CrossRef]
- Heo, J.-B.; Hopke, P.K.; Yi, S.-M. Source apportionment of PM 2.5 in Seoul, Korea. Atmos. Chem. Phys. 2009, 9, 4957–4971. [Google Scholar] [CrossRef]
- Chen, L.; Gao, Y.; Zhang, M.; Fu, J.S.; Zhu, J.; Liao, H.; Li, J.; Huang, K.; Ge, B.; Wang, X. MICS-Asia III: Multi-model comparison and evaluation of aerosol over East Asia. Atmos. Chem. Phys. 2019, 19, 11911–11937. [Google Scholar] [CrossRef]
- Ying, Q.; Wu, L.; Zhang, H. Local and inter-regional contributions to PM2.5 nitrate and sulfate in China. Atmos. Environ. 2014, 94, 582–592. [Google Scholar] [CrossRef]
- Kim, H.-S.; Huh, J.-B.; Hopke, P.K.; Holsen, T.M.; Yi, S.-M. Characteristics of the major chemical constituents of PM2.5 and smog events in Seoul, Korea in 2003 and 2004. Atmos. Environ. 2007, 41, 6762–6770. [Google Scholar] [CrossRef]
- Park, S.-S.; Cho, S.-Y.; Jung, C.-H.; Lee, K.-H. Characteristics of water-soluble inorganic species in PM10 and PM2.5 at two coastal sites during spring in Korea. Atmos. Pollut. Res. 2016, 7, 370–383. [Google Scholar] [CrossRef]
- Koo, Y.-S.; Yun, H.-Y.; Choi, D.-R.; Han, J.-S.; Lee, J.-B.; Lim, Y.-J. An analysis of chemical and meteorological characteristics of haze events in the Seoul metropolitan area during January 12–18, 2013. Atmos. Environ. 2018, 178, 87–100. [Google Scholar] [CrossRef]
- Kang, C.-M.; Lee, H.S.; Kang, B.-W.; Lee, S.-K.; Sunwoo, Y. Chemical characteristics of acidic gas pollutants and PM2.5 species during hazy episodes in Seoul, South Korea. Atmos. Environ. 2004, 38, 4749–4760. [Google Scholar] [CrossRef]
- Han, S.H.; Kim, Y.P. Long-term Trends of the Concentrations of Mass and Chemical Composition in PM 2.5 over Seoul. J. Korean Soc. Atmos. Environ. 2015, 31, 143–156, (In Korean with English Abstract). [Google Scholar] [CrossRef]
- Kim, H.C.; Kwon, S.; Kim, B.-U.; Kim, S. Review of Shandong Peninsular Emissions Change and South Korean Air Quality. J. Korean Soc. Atmos. Environ. 2018, 34, 356–365, (In Korean with English Abstract). [Google Scholar] [CrossRef]
- Han, Y.-J.; Kim, T.-S.; Kim, H. Ionic constituents and source analysis of PM2.5 in three Korean cities. Atmos. Environ. 2008, 42, 4735–4746. [Google Scholar] [CrossRef]
- Han, Y.-J.; Kim, S.-R.; Jung, J.-H. Long-term measurements of atmospheric PM2.5 and its chemical composition in rural Korea. J. Atmos. Chem. 2011, 68, 281–298. [Google Scholar] [CrossRef]
- Choi, J.; Park, R.J.; Lee, H.-M.; Lee, S.; Jo, D.S.; Jeong, J.I.; Henze, D.K.; Woo, J.-H.; Ban, S.-J.; Lee, M.-D.; et al. Impacts of local vs. trans-boundary emissions from different sectors on PM2.5 exposure in South Korea during the KORUS-AQ campaign. Atmos. Environ. 2019, 203, 196–205. [Google Scholar] [CrossRef]
- U.S. EPA. Modeling Procedures for Demonstrating Compliance with PM2.5 NAAQS; U.S. EPA: Washington, DC, USA, 2010.
- Aikawa, M.; Ohara, T.; Hiraki, T.; Oishi, O.; Tsuji, A.; Yamagami, M.; Murano, K.; Mukai, H. Significant geographic gradients in particulate sulfate over Japan determined from multiple-site measurements and a chemical transport model: Impacts of transboundary pollution from the Asian continent. Atmos. Environ. 2010, 44, 381–391. [Google Scholar] [CrossRef]
- Cohan, D.S.; Boylan, J.W.; Marmur, A.; Khan, M.N. An Integrated Framework for Multipollutant Air Quality Management and Its Application in Georgia. Environ. Manag. 2007, 40, 545–554. [Google Scholar] [CrossRef]
- Skamarock, W.C.; Klemp, J.B.; Dudhia, J.; Gill, D.O.; Barker, D.M.; Wang, W.; Powers, J.G. A Description of the Advanced Research WRF Version 2; National Center for Atmospheric Research: Boulder, CO, USA, 2007; p. 100. [Google Scholar]
- Benjey, W.G. Implementation of the SMOKE Emission Data Processor and SMOKE Tool Input Data Processor in Models-3. Presented at the Emission Inventory Conference, Denver, CO, USA, 1–4 May 2001; p. 15. [Google Scholar]
- Byun, D.; Schere, K.L. Review of the governing equations, computational algorithms, and other components of the Models-3 Community Multiscale Air Quality (CMAQ) modeling system. Appl. Mech. Rev. 2006, 59, 51–77. [Google Scholar] [CrossRef]
- Kim, B.-U.; Bae, C.; Kim, H.C.; Kim, E.; Kim, S. Spatially and chemically resolved source apportionment analysis: Case study of high particulate matter event. Atmos. Environ. 2017, 162, 55–70. [Google Scholar] [CrossRef]
- Koo, Y.-S.; Kim, S.-T.; Yun, H.-Y.; Han, J.-S.; Lee, J.-Y.; Kim, K.-H.; Jeon, E.-C. The simulation of aerosol transport over East Asia region. Atmos. Res. 2008, 90, 264–271. [Google Scholar] [CrossRef]
- Yim, S.H.L.; Gu, Y.; Shapiro, M.A.; Stephens, B. Air quality and acid deposition impacts of local emissions and transboundary air pollution in Japan and South Korea. Atmos. Chem. Phys. 2019, 19, 13309–13323. [Google Scholar] [CrossRef]
- Kim, H.C.; Kim, E.; Bae, C.; Cho, J.H.; Kim, B.-U.; Kim, S. Regional contributions to particulate matter concentration in the Seoul metropolitan area, South Korea: Seasonal variation and sensitivity to meteorology and emissions inventory. Atmos. Chem. Phys. 2017, 17, 10315–10332. [Google Scholar] [CrossRef]
- Lee, H.-M.; Park, R.J.; Henze, D.K.; Lee, S.; Shim, C.; Shin, H.-J.; Moon, K.-J.; Woo, J.-H. PM2. 5 source attribution for Seoul in May from 2009 to 2013 using GEOS-Chem and its adjoint model. Environ. Pollut. 2017, 221, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Ho, C.-H.; Choi, Y.-S. High-PM10 concentration episodes in Seoul, Korea: Background sources and related meteorological conditions. Atmos. Environ. 2011, 45, 7240–7247. [Google Scholar] [CrossRef]
- Chung, A.; Chang, D.P.; Kleeman, M.J.; Perry, K.D.; Cahill, T.A.; Dutcher, D.; McDougall, E.M.; Stroud, K. Comparison of real-time instruments used to monitor airborne particulate matter. J. Air Waste Manag. Assoc. 2001, 51, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-S.; Jung, S.-A.; Gong, B.-J.; Cho, S.-Y.; Lee, S.-J. Characteristics of PM2.5 Haze Episodes Revealed by Highly Time-Resolved Measurements at an Air Pollution Monitoring Supersite in Korea. Aerosol Air Qual. Res. 2013, 13, 957–976. [Google Scholar] [CrossRef]
- Park, S.-S.; Kim, S.-J.; Gong, B.-J.; Lee, K.-H.; Cho, S.-Y.; Kim, J.-C.; Lee, S.-J. Investigation on a Haze Episode of Fine Particulate Matter using Semi-continuous Chemical Composition Data. J. Korean Soc. Atmos. Environ. 2013, 29, 642–655, (In Korean with English Abstract). [Google Scholar] [CrossRef][Green Version]
- Guenther, A.; Karl, T.; Harley, P.; Wiedinmyer, C.; Palmer, P.I.; Geron, C. Estimates of global terrestrial isoprene emissions using MEGAN (Model of Emissions of Gases and Aerosols from Nature). Atmos. Chem. Phys. 2006, 6, 3181–3210. [Google Scholar] [CrossRef]
- Li, M.; Zhang, Q.; Kurokawa, J.; Woo, J.-H.; He, K.; Lu, Z.; Ohara, T.; Song, Y.; Streets, D.G.; Carmichael, G.R.; et al. MIX: A mosaic Asian anthropogenic emission inventory under the international collaboration framework of the MICS-Asia and HTAP. Atmos. Chem. Phys. 2017, 17, 935–963. [Google Scholar] [CrossRef]
- Itahashi, S.; Uno, I.; Kim, S. Source contributions of sulfate aerosol over East Asia estimated by CMAQ-DDM. Environ. Sci. Technol. 2012, 46, 6733–6741. [Google Scholar] [CrossRef]
- Lee, D.-G.; Lee, Y.-M.; Jang, K.-W.; Yoo, C.; Kang, K.-H.; Lee, J.-H.; Jung, S.-W.; Park, J.-M.; Lee, S.-B.; Han, J.-S.; et al. Korean National Emissions Inventory System and 2007 Air Pollutant Emissions. Asian J. Atmos. Environ. 2011, 5, 278–291. [Google Scholar] [CrossRef]
- Kim, S.; Moon1), N.; Byun, D.W. Korea Emissions Inventory Processing Using the US EPA’s SMOKE System. Asian J. Atmos. Environ. 2008, 2, 34–46. [Google Scholar] [CrossRef]
- Carter, W.P. Implementation of the SAPRC-99 Chemical Mechanism into the Models-3 Framework; Report to the United States Environmental Protection Agency; United States Environmental Protection Agency: Washington, DC, USA, 2000; p. 29.
- Carlton, A.G.; Bhave, P.V.; Napelenok, S.L.; Edney, E.O.; Sarwar, G.; Pinder, R.W.; Pouliot, G.A.; Houyoux, M. Model representation of secondary organic aerosol in CMAQv4. 7. Environ. Sci. Technol. 2010, 44, 8553–8560. [Google Scholar] [CrossRef] [PubMed]
- Emery, C.; Liu, Z.; Russell, A.G.; Odman, M.T.; Yarwood, G.; Kumar, N. Recommendations on statistics and benchmarks to assess photochemical model performance. J. Air Waste Manag. Assoc. 2017, 67, 582–598. [Google Scholar] [CrossRef]
- Emery, C.; Tai, E.; Yarwood, G. Enhanced Meteorological Modeling and Performance Evaluation for Two Texas Ozone Episodes; Prepared for the Texas natural resource conservation commission, by Environ International Corporation; Texas Natural Resource Conservation Commission: Austin, TX, USA, 2001. [Google Scholar]
- Bartnicki, J. Computing Source-Receptor Matrices with the Emep Eulerian Acid Deposition Model; Norwegian Meteorological Institute: Oslo, Norway, 1999; p. 37. [Google Scholar]
- Li, X.; Zhang, Q.; Zhang, Y.; Zheng, B.; Wang, K.; Chen, Y.; Wallington, T.J.; Han, W.; Shen, W.; Zhang, X. Source contributions of urban PM2. 5 in the Beijing–Tianjin–Hebei region: Changes between 2006 and 2013 and relative impacts of emissions and meteorology. Atmos. Environ. 2015, 123, 229–239. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, W.; Cheng, M.; Liu, S.; Xu, J.; He, Y.; Meng, F. The contribution of residential coal combustion to PM 2.5 pollution over China’s Beijing-Tianjin-Hebei region in winter. Atmos. Environ. 2017, 159, 147–161. [Google Scholar] [CrossRef]
- Tonnesen, G.S.; Dennis, R.L. Analysis of radical propagation efficiency to assess ozone sensitivity to hydrocarbons and NOx: 1. Local indicators of instantaneous odd oxygen production sensitivity. J. Geophys. Res. Atmos. 2000, 105, 9213–9225. [Google Scholar] [CrossRef]
- Marmur, A.; Unal, A.; Mulholland, J.A.; Russell, A.G. Optimization-based source apportionment of PM2.5 incorporating gas-to-particle ratios. Environ. Sci. Technol. 2005, 39, 3245–3254. [Google Scholar] [CrossRef]
- EPA, U. Guidance on the Use of Models and Other Analyses for Demonstrating Attainment of Air Quality Goals for Ozone, PM2. 5, and Regional Haze; US Environmental Protection Agency, Office of Air Quality Planning and Standards: Washington, DC, USA, 2007.
- Bae, C.; Kim, E.; Kim, B.-U.; Kim, H.C.; Woo, J.-H.; Moon, K.-J.; Shin, H.-J.; Song, I.H.; Kim, S. Impact of Emission Inventory Choices on PM10 Forecast Accuracy and Contributions in the Seoul Metropolitan Area. J. Korean Soc. Atmos. Environ. 2017, 33, 497–514, (In Korean with English Abstract). [Google Scholar] [CrossRef]
- Park, S.-M.; Song, I.-H.; Park, J.S.; Oh, J.; Moon, K.J.; Shin, H.J.; Ahn, J.Y.; Lee, M.-D.; Kim, J.; Lee, G. Variation of PM2.5 Chemical Compositions and their Contributions to Light Extinction in Seoul. Aerosol Air Qual. Res. 2018, 18, 2220–2229. [Google Scholar] [CrossRef]
- Kim, H.; Zhang, Q.; Heo, J. Influence of intense secondary aerosol formation and long-range transport on aerosol chemistry and properties in the Seoul Metropolitan Area during spring time: Results from KORUS-AQ. Atmos. Chem. Phys. 2018, 18, 7149–7168. [Google Scholar] [CrossRef]
- Khoder, M.I. Atmospheric conversion of sulfur dioxide to particulate sulfate and nitrogen dioxide to particulate nitrate and gaseous nitric acid in an urban area. Chemosphere 2002, 49, 675–684. [Google Scholar] [CrossRef]
- Eatough, D.J.; Caka, F.M.; Farber, R.J. The conversion of SO2 to sulfate in the atmosphere. Isr. J. Chem. 1994, 34, 301–314. [Google Scholar] [CrossRef]
- Matsumoto, K.; Tanaka, H. Formation and dissociation of atmospheric particulate nitrate and chloride: An approach based on phase equilibrium. Atmos. Environ. 1996, 30, 639–648. [Google Scholar] [CrossRef]
- Wang, P.; Cao, J.; Shen, Z.; Han, Y.; Lee, S.; Huang, Y.; Zhu, C.; Wang, Q.; Xu, H.; Huang, R. Spatial and seasonal variations of PM 2.5 mass and species during 2010 in Xi’an, China. Sci. Total Environ. 2015, 508, 477–487. [Google Scholar] [CrossRef] [PubMed]
- He, K.; Yang, F.; Ma, Y.; Zhang, Q.; Yao, X.; Chan, C.K.; Cadle, S.; Chan, T.; Mulawa, P. The characteristics of PM2.5 in Beijing, China. Atmos. Environ. 2001, 35, 4959–4970. [Google Scholar] [CrossRef]
- Zhang, F.; Cheng, H.; Wang, Z.; Lv, X.; Zhu, Z.; Zhang, G.; Wang, X. Fine particles (PM2.5) at a CAWNET background site in Central China: Chemical compositions, seasonal variations and regional pollution events. Atmos. Environ. 2014, 86, 193–202. [Google Scholar] [CrossRef]
- Zhang, T.; Cao, J.J.; Tie, X.X.; Shen, Z.X.; Liu, S.X.; Ding, H.; Han, Y.M.; Wang, G.H.; Ho, K.F.; Qiang, J.; et al. Water-soluble ions in atmospheric aerosols measured in Xi’an, China: Seasonal variations and sources. Atmos. Res. 2011, 102, 110–119. [Google Scholar] [CrossRef]
- Kundu, S.; Kawamura, K.; Kobayashi, M.; Tachibana, E.; Lee, M.; Fu, P.Q.; Jung, J. A sub-decadal trend in diacids in atmospheric aerosols in eastern Asia. Atmos. Chem. Phys. 2016, 16, 585–596. [Google Scholar] [CrossRef]
- Pani, S.K.; Lee, C.-T.; Chou, C.C.-K.; Shimada, K.; Hatakeyama, S.; Takami, A.; Wang, S.-H.; Lin, N.-H. Chemical characterization of wintertime aerosols over islands and mountains in East Asia: Impacts of the continental Asian outflow. Aerosol Air Qual. Res. 2017, 17, 3006–3036. [Google Scholar] [CrossRef]
- Seinfeld, J.H.; Pandis, S.N. Atmospheric Chemistry and Physics: From Air Pollution to Climate Change; John Wiley & Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
- Pandis, S.N.; Seinfeld, J.H. Sensitivity analysis of a chemical mechanism for aqueous-phase atmospheric chemistry. J. Geophys. Res. Atmos. 1989, 94, 1105–1126. [Google Scholar] [CrossRef]
- Singh, H.B.; Salas, L.J.; Viezee, W. Global distribution of peroxyacetyl nitrate. Nature 1986, 321, 588. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.; An, J.; He, Y.; Zheng, J. An overview of emissions of SO2 and NOx and the long-range transport of oxidized sulfur and nitrogen pollutants in East Asia. J. Environ. Sci. 2016, 44, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-J.; Jo, H.-Y.; Kim, S.-W.; Park, M.-S.; Kim, C.-H. Impacts of atmospheric vertical structures on transboundary aerosol transport from China to South Korea. Sci. Rep. 2019, 9, 13040. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Liu, J.; Zhang, J.; Gui, H.; Du, P.; Yu, T.; Wang, J.; Lu, Y.; Liu, W.; Cheng, Y. Identification of long-range transport pathways and potential sources of PM2. 5 and PM10 in Beijing from 2014 to 2015. J. Environ. Sci. 2017, 56, 214–229. [Google Scholar] [CrossRef]
- Itahashi, S.; Yumimoto, K.; Uno, I.; Hayami, H.; Fujita, S.; Pan, Y.; Wang, Y. A 15-year record (2001–2015) of the ratio of nitrate to non-sea-salt sulfate in precipitation over East Asia. Atmos. Chem. Phys. 2018, 18, 2835–2852. [Google Scholar] [CrossRef]
- Wang, X.; Chen, W.; Chen, D.; Wu, Z.; Fan, Q. Long-term trends of fine particulate matter and chemical composition in the Pearl River Delta Economic Zone (PRDEZ), China. Front. Environ. Sci. Eng. 2016, 10, 53–62. [Google Scholar] [CrossRef]
- Woo, J.-H.; Bu, C.; Kim, J.; Ghim, Y.S.; Kim, Y. Analysis of regional and inter-annual changes of air pollutants emissions in China. J. Korean Soc. Atmos. Environ. 2018, 34, 87–100, (In Korean with English Abstract). [Google Scholar] [CrossRef]
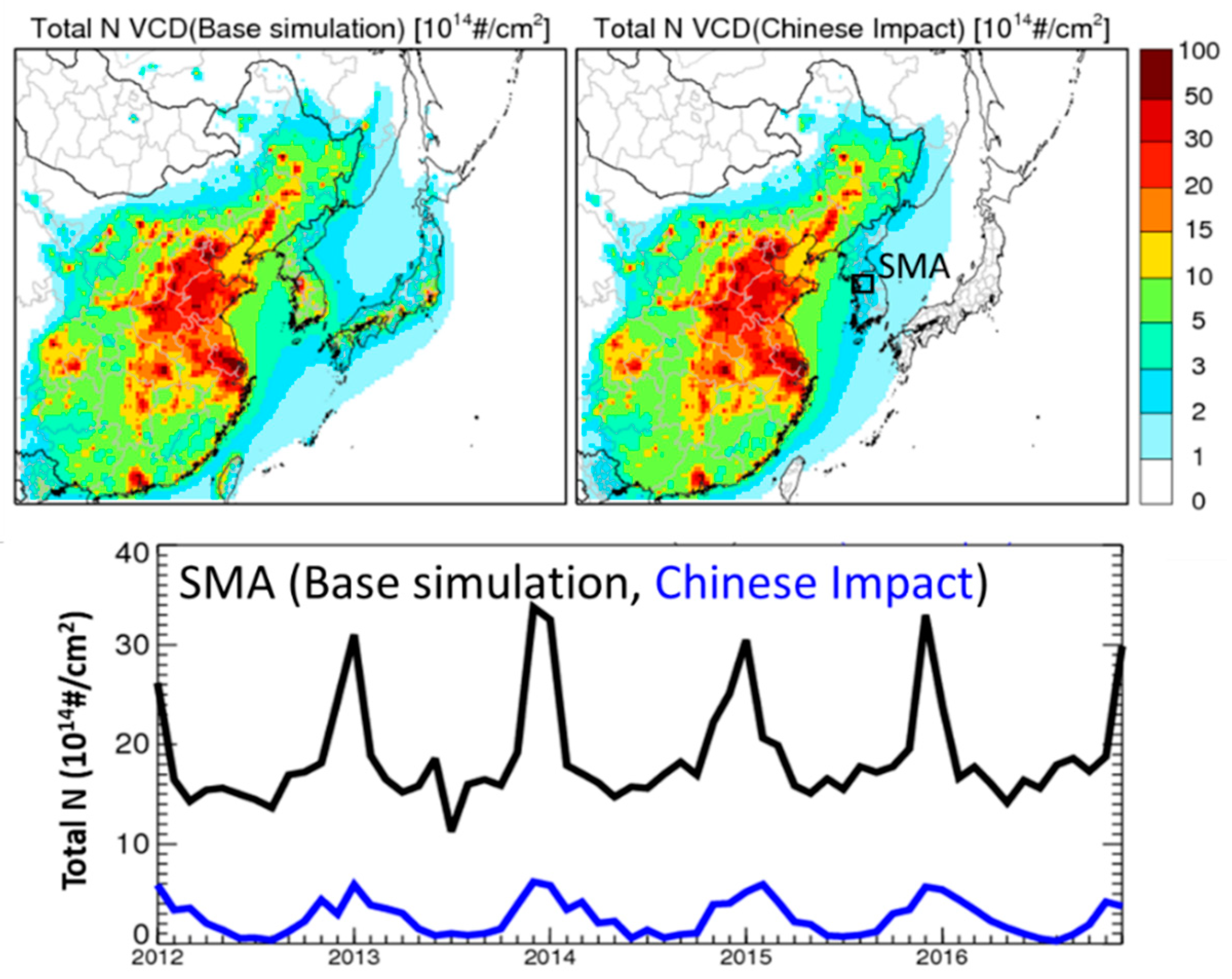
| Jan | Feb | Mar | Apr | May | Jun | Jul | Aug | Sep | Oct | Nov | Dec | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| # of daily samples (Daily Mean) | 69 | 60 | 71 | 67 | 65 | 80 | 81 | 78 | 83 | 107 | 97 | 75 |
| WRF | Description |
|---|---|
| Version | 3.4.1 |
| Initial field | FNL |
| Planetary boundary layer scheme | YSU |
| Microphysics | WSM6 |
| Land surface model scheme | NOAH |
| Short wave radiation | NASA Goddard |
| CMAQ | Description |
|---|---|
| Version | 4.7.1 |
| Chemical Mechanism | SAPRC99 |
| Chemical Solver | EBI |
| Aerosol Module | AERO5 |
| Boundary Condition | Default profile |
| Advection Scheme | YAMO |
| Horizontal Diffusion | Multiscale |
| Vertical Diffusion | Eddy |
| Chinese Emission Impact | Sulfate | Nitrate | |
|---|---|---|---|
| (a) Base case | Concentration (µg/m3) | 2.3 | 5.1 |
| Relative (%) | 46.2 | 57.8 | |
| (b) CCF case | Concentration (µg/m3) | 3.2 | 3.9 |
| Relative (%) | 48.7 | 58.4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bae, C.; Kim, B.-U.; Kim, H.C.; Yoo, C.; Kim, S. Long-Range Transport Influence on Key Chemical Components of PM2.5 in the Seoul Metropolitan Area, South Korea, during the Years 2012–2016. Atmosphere 2020, 11, 48. https://doi.org/10.3390/atmos11010048
Bae C, Kim B-U, Kim HC, Yoo C, Kim S. Long-Range Transport Influence on Key Chemical Components of PM2.5 in the Seoul Metropolitan Area, South Korea, during the Years 2012–2016. Atmosphere. 2020; 11(1):48. https://doi.org/10.3390/atmos11010048
Chicago/Turabian StyleBae, Changhan, Byeong-Uk Kim, Hyun Cheol Kim, Chul Yoo, and Soontae Kim. 2020. "Long-Range Transport Influence on Key Chemical Components of PM2.5 in the Seoul Metropolitan Area, South Korea, during the Years 2012–2016" Atmosphere 11, no. 1: 48. https://doi.org/10.3390/atmos11010048
APA StyleBae, C., Kim, B.-U., Kim, H. C., Yoo, C., & Kim, S. (2020). Long-Range Transport Influence on Key Chemical Components of PM2.5 in the Seoul Metropolitan Area, South Korea, during the Years 2012–2016. Atmosphere, 11(1), 48. https://doi.org/10.3390/atmos11010048






