Size-Segregated Chemical Compositions of HULISs in Ambient Aerosols Collected during the Winter Season in Songdo, South Korea
Abstract
1. Introduction
2. Experiments
2.1. Aerosol Sampling
2.2. Preparation of HULISs from Size-Segregated Aerosols
2.3. FT–ICR MS Analysis and Data Processing for Elemental Composition Assignments
2.4. Air Mass Backward Trajectory Analysis
3. Results and Discussion
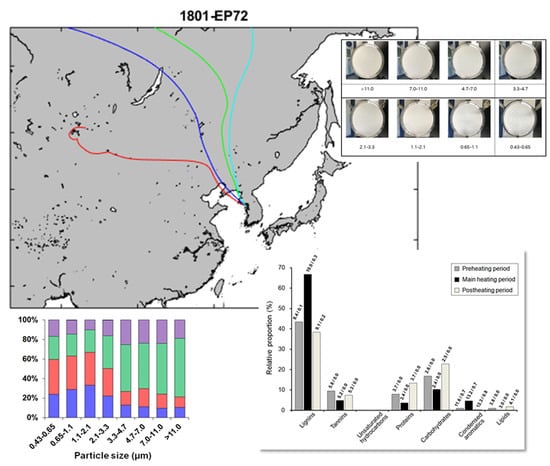
3.1. Size-Segregated Sampling of Ambient Aerosols during the Winter Season in Songdo, South Korea
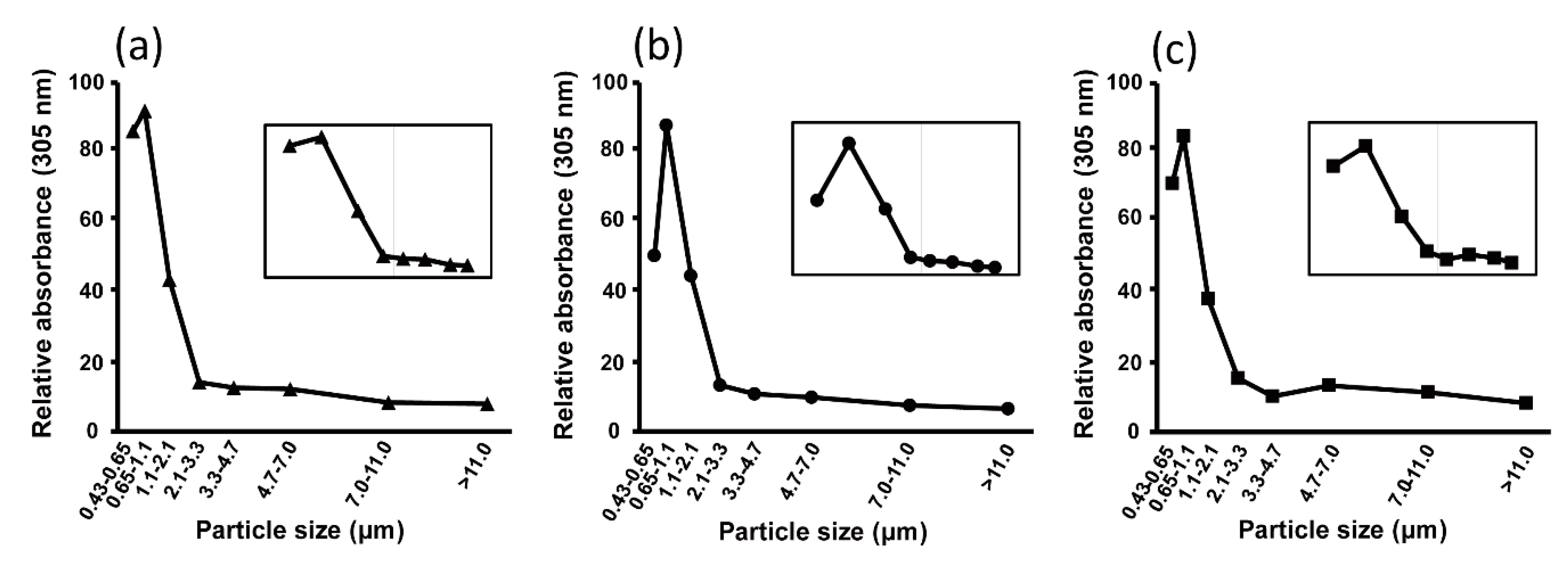
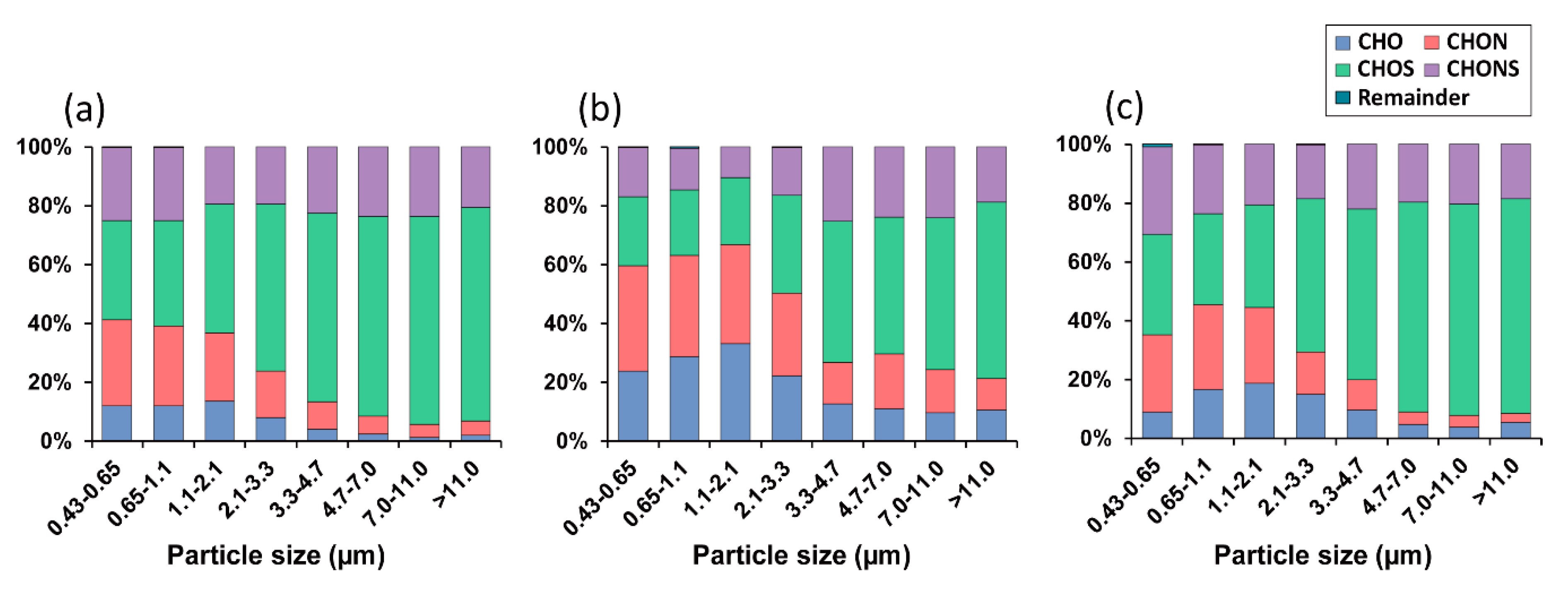
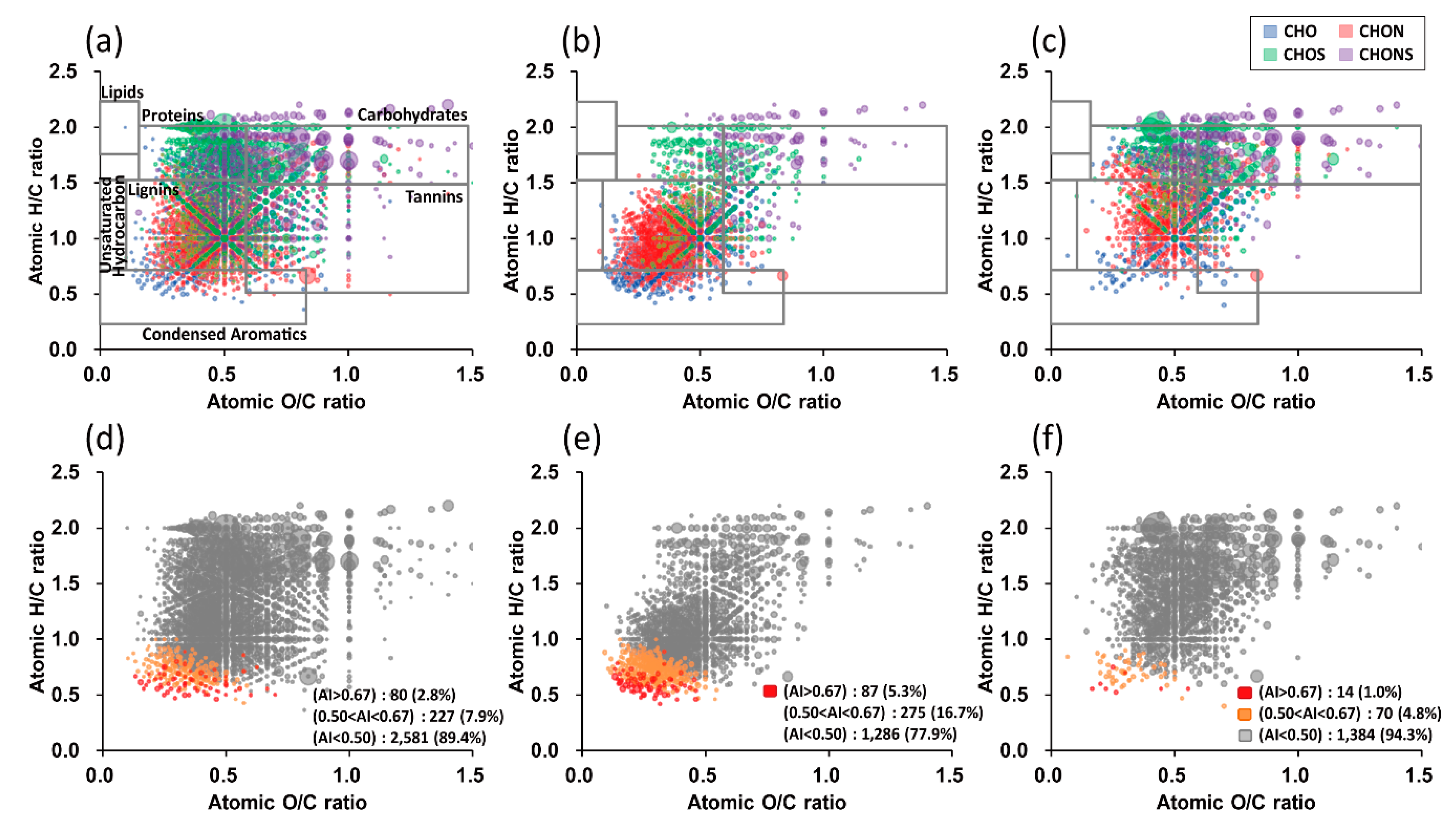
3.2. UHR FT–ICR MS Profiles of HULISs in Size-Segregated Aerosols
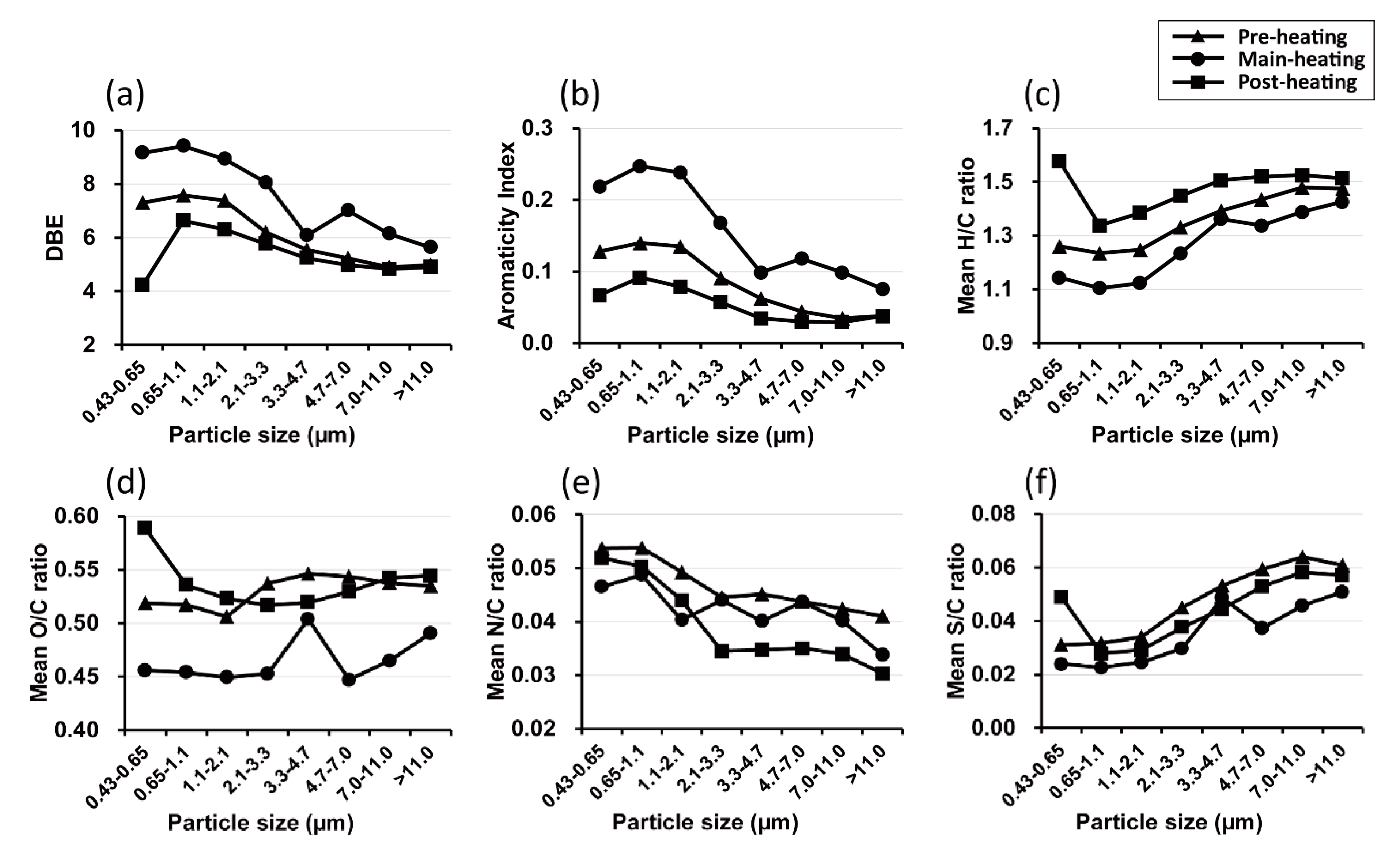
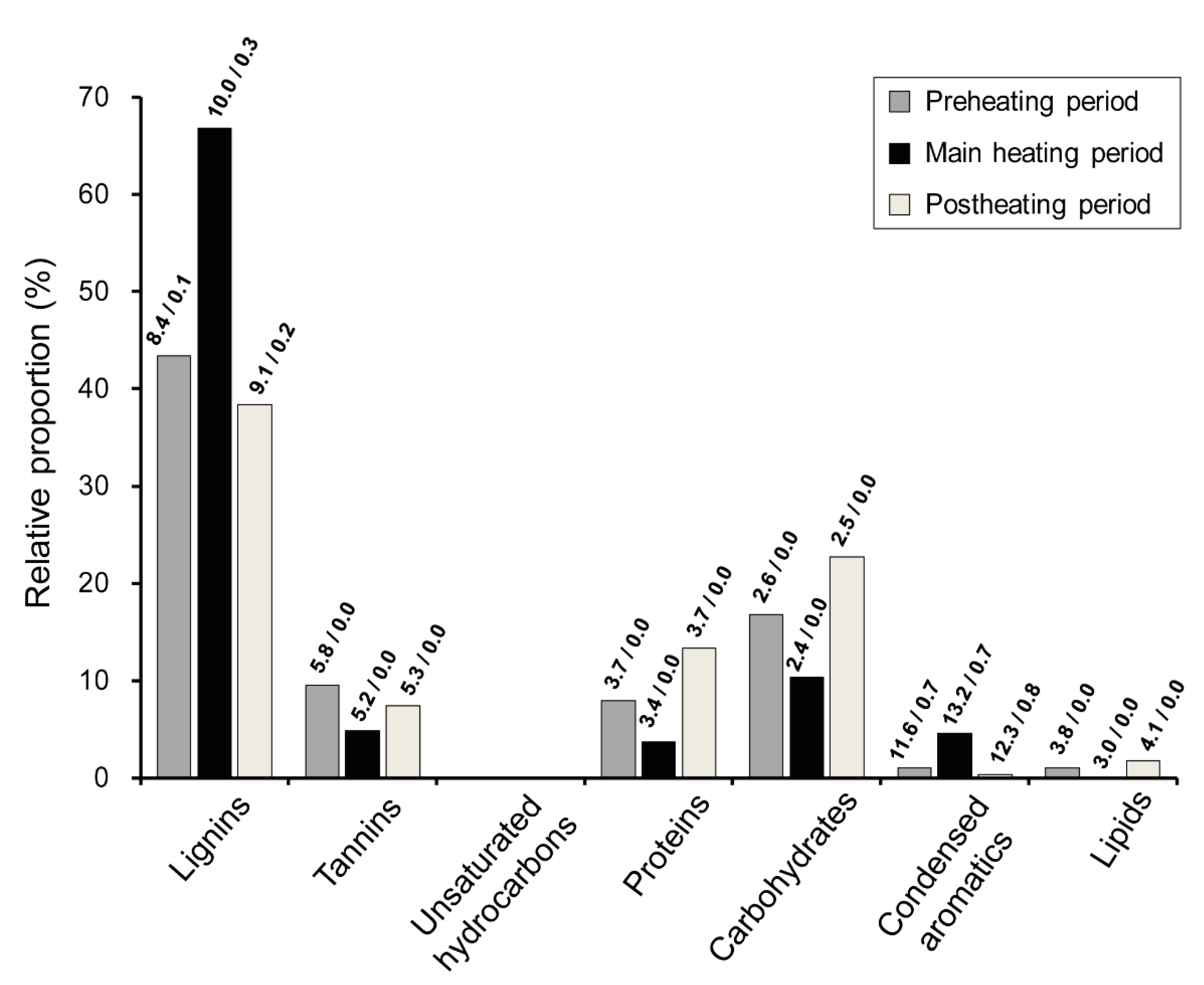
3.3. Seasonal Characteristics of Ambient Size-Resolved HULIS Samples
3.4. Implications of Size-Dependent Chemical Compositions and Their Source Origins
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ohara, T.; Akimoto, H.; Kurokawa, J.; Horii, N.; Yamaji, K.; Yan, X.; Hayasaka, T. An Asian emission inventory of anthropogenic emission sources for the period 1980–2020. Atmos Chem Phys 2007, 7, 4419–4444. [Google Scholar] [CrossRef]
- Monks, P.S.; Granier, C.; Fuzzi, S.; Stohl, A.; Williams, M.L.; Akimoto, H.; Amann, M.; Baklanov, A.; Baltensperger, U.; Bey, I.; et al. Atmospheric composition change—Global and regional air quality. Atmos. Environ. 2009, 43, 5268–5350. [Google Scholar] [CrossRef]
- Kroll, J.H.; Seinfeld, J.H. Chemistry of secondary organic aerosol: Formation and evolution of low-volatility organics in the atmosphere. Atmos. Environ. 2008, 42, 3593–3624. [Google Scholar] [CrossRef]
- Poschl, U. Atmospheric aerosols: Composition, transformation, climate and health effects. Angew. Chem. Int. Ed. Engl. 2005, 44, 7520–7540. [Google Scholar] [CrossRef] [PubMed]
- Hallquist, M.; Wenger, J.C.; Baltensperger, U.; Rudich, Y.; Simpson, D.; Claeys, M.; Dommen, J.; Donahue, N.M.; George, C.; Goldstein, A.H.; et al. The formation, properties and impact of secondary organic aerosol: Current and emerging issues. Atmos. Chem. Phys. 2009, 9, 5155–5236. [Google Scholar] [CrossRef]
- Ervens, B.; Turpin, B.J.; Weber, R.J. Secondary organic aerosol formation in cloud droplets and aqueous particles (aqSOA): A review of laboratory, field and model studies. Atmos. Chem. Phys. 2011, 11, 11069–11102. [Google Scholar] [CrossRef]
- Bernstein, J.A.; Alexis, N.; Barnes, C.; Bernstein, I.L.; Bernstein, J.A.; Nel, A.; Peden, D.; Diaz-Sanchez, D.; Tarlo, S.M.; Williams, P.B. Health effects of air pollution. J. Allergy Clin. Immunol. 2004, 114, 1116–1123. [Google Scholar] [CrossRef]
- Yu, H. Environmental carcinogenic polycyclic aromatic hydrocarbons: Photochemistry and phototoxicity. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2002, 20, 149–183. [Google Scholar] [CrossRef]
- Chowdhury, P.H.; He, Q.; Male, T.L.; Brune, W.H.; Rudich, Y.; Pardo, M. Exposure of lung epithelial cells to photochemically aged secondary organic aerosol shows increased toxic effects. Environ. Sci. Technol. Lett. 2018, 5, 424–430. [Google Scholar] [CrossRef]
- Graber, E.R.; Rudich, Y. Atmospheric HULIS: How humic-like are they? A comprehensive and critical review. Atmos. Chem. Phys. 2006, 6, 729–753. [Google Scholar] [CrossRef]
- 1Han, H.; Kim, G. Significant seasonal change in optical properties by atmospheric humic-like substances (HULIS) in water-soluble organic carbon aerosols. Atmos. Chem. Phys. Discuss. 2017, 7. [Google Scholar] [CrossRef]
- Chen, Q.; Ikemori, F.; Higo, H.; Asakawa, D.; Mochida, M. Chemical Structural Characteristics of HULIS and Other Fractionated Organic Matter in Urban Aerosols: Results from Mass Spectral and FT-IR Analysis. Environ. Sci. Technol. 2016, 50, 1721–1730. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.; Rincon, A.G.; Kalberer, M.; Yu, J.Z. Elemental composition of HULIS in the Pearl River Delta Region, China: Results inferred from positive and negative electrospray high resolution mass spectrometric data. Environ. Sci. Technol. 2012, 46, 7454–7462. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Xiang, P.; Zhou, X.; Duan, J.; Ma, Y.; He, K.; Cheng, Y.; Yu, J.; Querol, X. Chemical characterization of humic-like substances (HULIS) in PM2.5 in Lanzhou, China. Sci. Total. Environ. 2016, 573, 1481–1490. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Qiao, T.; Li, Y.; Tang, X.; Xiu, G.; Yu, J.Z. Temporal variations and source apportionment of Hulis-C in PM2.5 in urban Shanghai. Sci. Total Environ. 2016, 571, 18–26. [Google Scholar] [CrossRef]
- Wu, G.; Wan, X.; Gao, S.; Fu, P.; Yin, Y.; Li, G.; Zhang, G.; Kang, S.; Ram, K.; Cong, Z. Humic-Like Substances (HULIS) in Aerosols of Central Tibetan Plateau (Nam Co, 4730 m asl): Abundance, Light Absorption Properties, and Sources. Environ. Sci. Technol. 2018, 52, 7203–7211. [Google Scholar] [CrossRef]
- Liu, J.; Mo, Y.; Ding, P.; Li, J.; Shen, C.; Zhang, G. Dual carbon isotopes ((14)C and (13)C) and optical properties of WSOC and HULIS-C during winter in Guangzhou, China. Sci. Total Environ. 2018, 633, 1571–1578. [Google Scholar] [CrossRef]
- Ghosh, S.; Biswas, J.; Guttikunda, S.; Roychowdhury, S.; Nayak, M. An investigation of potential regional and local source regions affecting fine particulate matter concentrations in Delhi, India. J. Air Waste Manag. Assoc. 2015, 65, 218–231. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, M.; Bai, X.; Tan, H.; Li, S.; Liu, J.; Zhang, R.; Wolters, M.A.; Qin, X.; Zhang, M.; et al. Large-scale transport of PM2.5 in the lower troposphere during winter cold surges in China. Sci. Rep. 2017, 7, 13238. [Google Scholar] [CrossRef]
- Havers, N.; Burba, P.; Lambert, J.; Klockow, D. Spectroscopic characterization of humic-like substances in airborne particulate matter. J. Atmos. Chem. 1998, 29, 45–54. [Google Scholar] [CrossRef]
- Varga, B.; Kiss, G.; Ganszky, I.; Gelencser, A.; Krivacsy, Z. Isolation of water-soluble organic matter from atmospheric aerosol. Talanta 2001, 55, 561–572. [Google Scholar] [CrossRef]
- Lin, P.; Yu, J.Z.; Engling, G.; Kalberer, M. Organosulfates in humic-like substance fraction isolated from aerosols at seven locations in East Asia: A study by ultra-high-resolution mass spectrometry. Environ. Sci. Technol. 2012, 46, 13118–13127. [Google Scholar] [CrossRef]
- Choi, J.H.; Kim, Y.G.; Lee, Y.K.; Pack, S.P.; Jung, J.Y.; Jang, K.S. Chemical characterization of dissolved organic matter in moist acidic tussock tundra soil using ultra-high resolution 15T FT-ICR mass spectrometry. Biotechnol. Bioprocess. Eng. 2017, 22, 637–646. [Google Scholar] [CrossRef]
- Choi, J.H.; Ryu, J.; Jeon, S.; Seo, J.; Yang, Y.H.; Pack, S.P.; Choung, S.; Jang, K.S. In-depth compositional analysis of water-soluble and -insoluble organic substances in fine (PM2.5) airborne particles using ultra-high-resolution 15T FT-ICR MS and GCxGC-TOFMS. Environ. Pollut. 2017, 225, 329–337. [Google Scholar] [CrossRef]
- Mazzoleni, L.R.; Ehrmann, B.M.; Shen, X.; Marshall, A.G.; Collett, J.L., Jr. Water-soluble atmospheric organic matter in fog: Exact masses and chemical formula identification by ultrahigh-resolution fourier transform ion cyclotron resonance mass spectrometry. Environ. Sci. Technol. 2010, 44, 3690–3697. [Google Scholar] [CrossRef]
- Kim, S.; Kramer, R.W.; Hatcher, P.G. Graphical method for analysis of ultrahigh-resolution broadband mass spectra of natural organic matter, the van Krevelen diagram. Anal. Chem. 2003, 75, 5336–5344. [Google Scholar] [CrossRef]
- Hockaday, W.C.; Grannas, A.M.; Kim, S.; Hatcher, P.G. Direct molecular evidence for the degradation and mobility of black carbon in soils from ultrahigh-resolution mass spectral analysis of dissolved organic matter from a fire-impacted forest soil. Org. Geochem. 2006, 37, 501–510. [Google Scholar] [CrossRef]
- Koch, B.P.; Dittmar, T. From mass to structure: An aromaticity index for high-resolution mass data of natural organic matter. Rapid Commun. Mass Spectrom. 2006, 20, 926–932. [Google Scholar] [CrossRef]
- Stein, A.F.; Draxler, R.R.; Rolph, G.D.; Stunder, B.J.B.; Cohen, M.D.; Ngan, F. NOAA’s HYSPLIT atmospheric transport and dispersion modeling system. Bull. Amer. Meteor. Soc. 2015, 96, 2059–2077. [Google Scholar] [CrossRef]
- Carr, N.; Davis, C.E.; Blackbird, S.; Daniels, L.R.; Preece, C.; Woodward, M.; Mahaffey, C. Seasonal and spatial variability in the optical characteristics of DOM in a temperate shelf sea. Prog. Oceanogr. 2018, 2. [Google Scholar] [CrossRef]
- Kowalczuk, P.; Tilstone, G.H.; Zablocka, M.; Röttgers, R.; Thomas, R. Composition of dissolved organic matter along an Atlantic Meridional Transect from fluorescence spectroscopy and Parallel Factor Analysis. Mar. Chem. 2013, 157, 170–184. [Google Scholar] [CrossRef]
- Yue, D.; Hu, M.; Wu, Z.; Wang, Z.; Guo, S.; Wehner, B.; Nowak, A.; Achtert, P.; Wiedensohler, A.; Jung, J.; et al. Characteristics of aerosol size distributions and new particle formation in the summer in Beijing. J. Geophys. Res. 2009, 114, D00G12. [Google Scholar] [CrossRef]
- Ueda, S.; Miura, K.; Kawata, R.; Furutani, H.; Uematsu, M.; Omori, Y. Number-size distribution of aerosol particles and new particle formation events in tropical and subtropical Pacific Oceans. Atmos. Environ. 2016, 142, 324–339. [Google Scholar] [CrossRef]
- Al-Kindi, S.S.; Pope, F.D.; Beddows, D.C.; Bloss, W.J.; Harrison, R.M. Size-dependent chemical ageing of oleic acid aerosol under dry and humidified conditions. Atmos. Chem. Phys. 2016, 16, 15561–15579. [Google Scholar] [CrossRef]
- Smith, J.N.; Dunn, M.J.; VanReken, T.M.; Iida, K.; Stolzenburg, M.R.; McMurry, P.H.; Huey, L.G. Chemical composition of atmospheric nanoparticles formed from nucleation in Tecamac, Mexico: Evidence for an important role for organic species in nanoparticle growth. Geophys. Res. Lett. 2008, 35, 2–6. [Google Scholar] [CrossRef]
- Seinfeld, J.H.; Erdakos, G.B.; Asher, W.E.; Pankow, J.F. Modeling the formation of secondary organic aerosol (SOA). 2. The predicted effects of relative humidity on aerosol formation in the alpha-pinene-, beta-pinene-, sabinene-, delta 3-carene-, and cyclohexene-ozone systems. Environ. Sci. Technol. 2001, 35, 1806–1817. [Google Scholar] [CrossRef]
- Vasconcelos, L.A.P.; Macias, E.S.; White, W.H. Aerosol composition as a function of haze and humidity levels in the Southwestern U.S. Atmos. Environ. 2003, 28, 3679–3691. [Google Scholar] [CrossRef]
- Tong, H.; Kourtchev, I.; Pant, P.; Keyte, I.J.; O’Connor, I.P.; Wenger, J.C.; Pope, F.D.; Harrison, R.M.; Kalberer, M. Molecular composition of organic aerosols at urban background and road tunnel sites using ultra-high resolution mass spectrometry. Faraday Discuss. 2016, 189, 51–68. [Google Scholar] [CrossRef]
- Shakya, K.M.; Louchouarn, P.; Griffin, R.J. Lignin-derived phenols in Houston aerosols: Implications for natural background sources. Environ. Sci. Technol. 2011, 45, 8268–8275. [Google Scholar] [CrossRef]
- Ravindra, K.; Sokhi, R.; van Grieken, R. Atmospheric polycyclic aromatic hydrocarbons: Source attribution, emission factors and regulation. Atmos. Environ. 2008, 42, 2895–2921. [Google Scholar] [CrossRef]
- Chang, K.F.; Fang, G.C.; Chen, J.C.; Wu, Y.S. Atmospheric polycyclic aromatic hydrocarbons (PAHs) in Asia: A review from 1999 to 2004. Environ. Pollut. 2006, 142, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Liang, Y.; Xu, C.; Zhang, J.; Hu, M.; Shi, Q. Polycyclic aromatic hydrocarbons (PAHs) in ambient aerosols from Beijing: Characterization of low volatile PAHs by positive-ion atmospheric pressure photoionization (APPI) coupled with Fourier transform ion cyclotron resonance. Environ. Sci. Technol. 2014, 48, 4716–4723. [Google Scholar] [CrossRef] [PubMed]
- Petters, M.D.; Prenni, A.J.; Kreidenweis, S.M.; DeMott, P.J.; Matsunaga, A.; Lim, Y.B.; Ziemann, P.J. Chemical aging and the hydrophobic-to-hydrophilic conversion of carbonaceous aerosol. Geophys. Res. Lett. 2006, 33, L24806. [Google Scholar] [CrossRef]
- Denjean, C.; Formenti, P.; Picquet-Varrault, B.; Camredon, M.; Pangui, E.; Zapf, P.; Katrib, Y.; Giorio, C.; Tapparo, A.; Temime-Roussel, B.; et al. Aging of secondary organic aerosol generated from the ozonolysis of α-pinene: Effects of ozone, light and temperature. Atmos. Chem. Phys. 2015, 15, 883–897. [Google Scholar] [CrossRef]
- Carslaw, D.C. Evidence of an increasing NO2/NOX emissions ratio from road traffic emissions. Atmos. Environ. 2005, 39, 4793–4802. [Google Scholar] [CrossRef]
- Yli-Tuomi, T.; Aarnio, P.; Pirjola, L.; Mäkelä, T.; Hillamo, R.; Jantunen, M. Emissions of fine particles, NOx, andCO from on-road vehicles in Finland. Atmos. Environ. 2005, 39, 6696–6706. [Google Scholar] [CrossRef]
- Cho, K.D.; Kim, J.S. The Status of the Atmospheric Environment and Air Quality Management Measures of the Port. of Incheon; The Incheon Institute: Incheon, Korea, 2009. [Google Scholar]
- Blair, S.L.; MacMillan, A.C.; Drozd, G.T.; Goldstein, A.H.; Chu, R.K.; Pasa-Tolic, L.; Shaw, J.B.; Tolic, N.; Lin, P.; Laskin, J.; et al. Molecular Characterization of Organosulfur Compounds in Biodiesel and Diesel Fuel Secondary Organic Aerosol. Environ. Sci. Technol. 2017, 51, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Surratt, J.D.; Lin, Y.H.; Bapat, J.; Kamens, R.M. Effect of relative humidity on SOA formation from isoprene/NO photooxidation: Enhancement of 2-methylglyceric acid and its corresponding oligoesters under dry conditions. Atmos. Chem. Phys. 2011, 11, 6411–6424. [Google Scholar] [CrossRef]
- Emanuelsson, E.U.; Watne, A.K.; Lutz, A.; Ljungstrom, E.; Hallquist, M. Influence of humidity, temperature, and radicals on the formation and thermal properties of secondary organic aerosol (SOA) from ozonolysis of beta-pinene. J. Phys. Chem. A 2013, 117, 10346–10358. [Google Scholar] [CrossRef]
- Lepri, L.; Bubba, M.D.; Masi, F.; Udisti, R.; Cini, R. Particle size distribution of organic compounds in aqueous aerosols collected from above sewage aeration tanks. Aerosol Sci. Technol. 2000, 32, 404–420. [Google Scholar] [CrossRef]
- Bostrom, C.E.; Gerde, P.; Hanberg, A.; Jernstrom, B.; Johansson, C.; Kyrklund, T.; Rannug, A.; Tornqvist, M.; Victorin, K.; Westerholm, R. Cancer risk assessment, indicators, and guidelines for polycyclic aromatic hydrocarbons in the ambient air. Environ. Health Perspect. 2002, 110, 451–488. [Google Scholar]
- Hauser, R.; Calafat, A.M. Phthalates and human health. Occup. Environ. Med. 2005, 6, 806–818. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, S.; Masai, E.; Kamimura, N.; Takahashi, K.; Anderson, R.C.; Faisal, P.A. Phthalates impact human health: Epidemiological evidences and plausible mechanism of action. J. Hazard. Mater. 2017, 340, 360–383. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Wang, M.; Chen, Q.; Zhu, C.; Jie, J.; Li, X.; Dong, X.; Miao, Z.; Shen, M.; Bu, Q. Spatial, seasonal and particle size dependent variations of PAH contamination in indoor dust and the corresponding human health risk. Sci. Total Environ. 2019, 653, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Ksionzek, K.B.; Lechtenfeld, O.J.; McCallister, S.L.; Schmitt-Kopplin, P.; Geuer, J.K.; Geibert, W.; Koch, B.P. Dissolved organic sulfur in the ocean: Biogeochemistry of a petagram inventory. Science 2016, 354, 456–459. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, K.-S.; Choi, A.Y.; Choi, M.; Kang, H.; Kim, T.-W.; Park, K.-T. Size-Segregated Chemical Compositions of HULISs in Ambient Aerosols Collected during the Winter Season in Songdo, South Korea. Atmosphere 2019, 10, 226. https://doi.org/10.3390/atmos10040226
Jang K-S, Choi AY, Choi M, Kang H, Kim T-W, Park K-T. Size-Segregated Chemical Compositions of HULISs in Ambient Aerosols Collected during the Winter Season in Songdo, South Korea. Atmosphere. 2019; 10(4):226. https://doi.org/10.3390/atmos10040226
Chicago/Turabian StyleJang, Kyoung-Soon, A Young Choi, Mira Choi, Hyunju Kang, Tae-Wook Kim, and Ki-Tae Park. 2019. "Size-Segregated Chemical Compositions of HULISs in Ambient Aerosols Collected during the Winter Season in Songdo, South Korea" Atmosphere 10, no. 4: 226. https://doi.org/10.3390/atmos10040226
APA StyleJang, K.-S., Choi, A. Y., Choi, M., Kang, H., Kim, T.-W., & Park, K.-T. (2019). Size-Segregated Chemical Compositions of HULISs in Ambient Aerosols Collected during the Winter Season in Songdo, South Korea. Atmosphere, 10(4), 226. https://doi.org/10.3390/atmos10040226