Fundamentals of Ornamental Plants in Removing Benzene in Indoor Air
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Cultured Condition
2.2. Fumigation Chamber
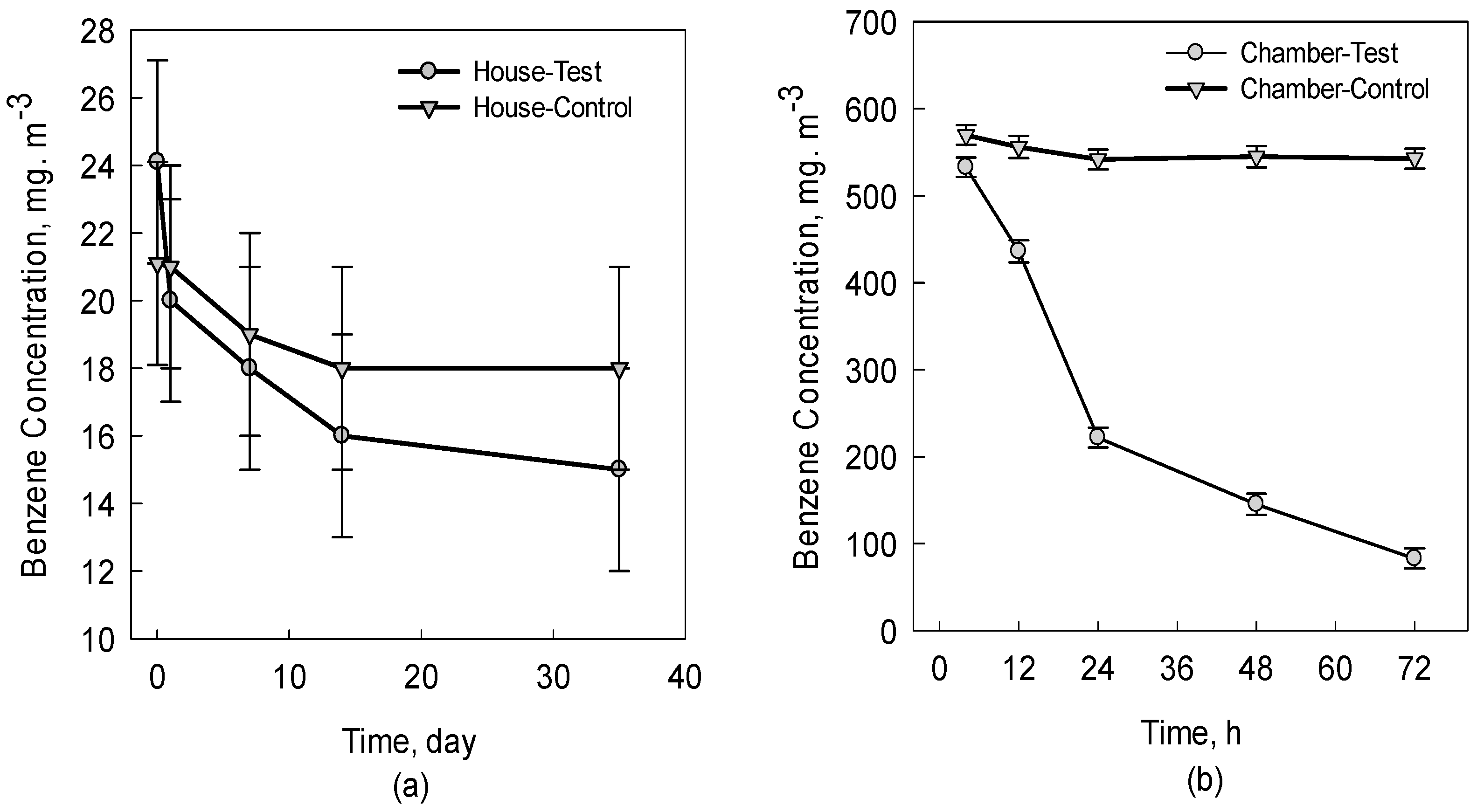
2.3. Removal of Benzene in House
2.4. Transpiration Rate, POD and Chlorophyll Concentration Analysis
2.5. Benzene Removal Efficiency
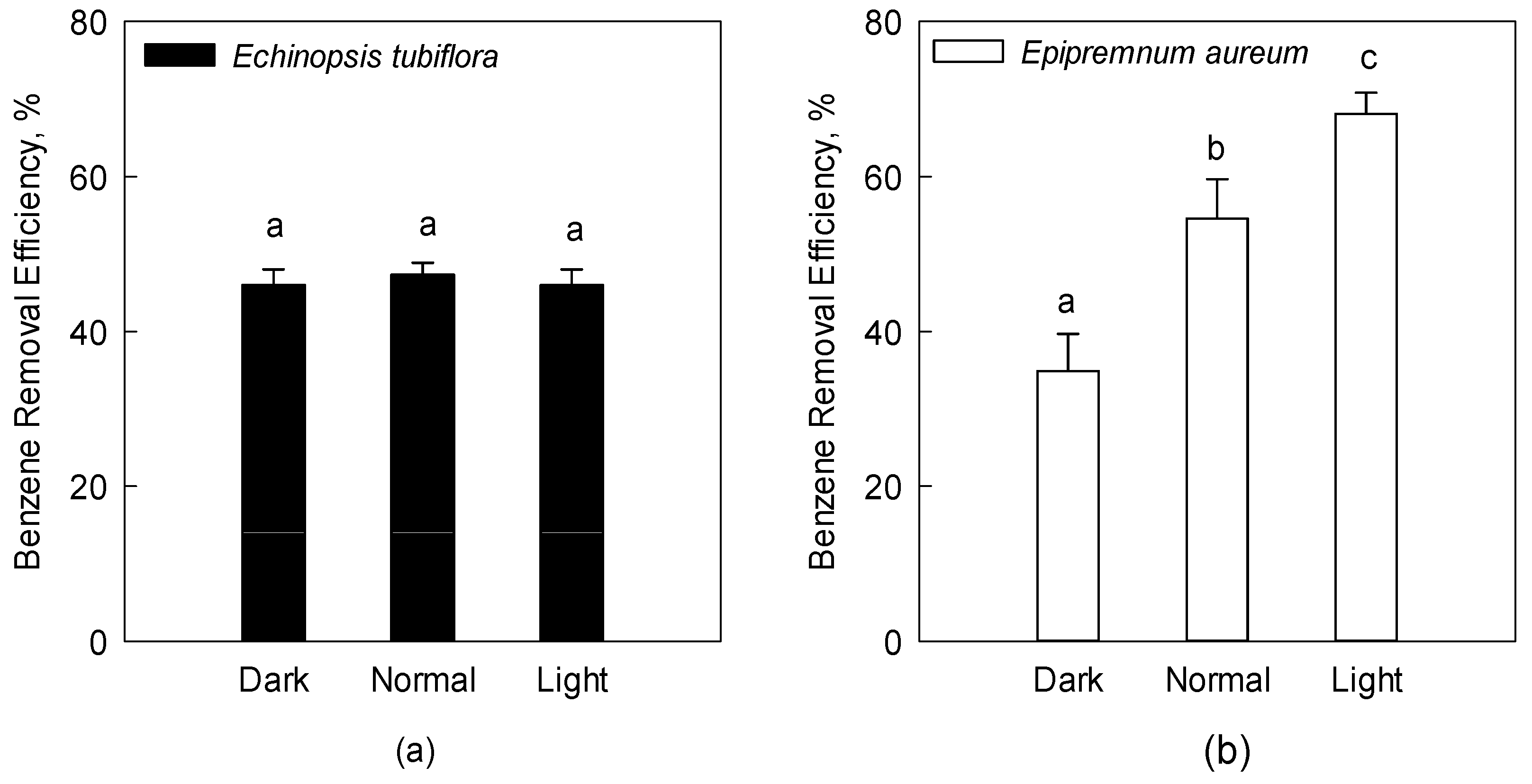
2.6. Effect of Light Conditions
2.7. Effect of Different Benzene Concentrations
2.8. Gas Analysis
2.9. Statistical Analysis
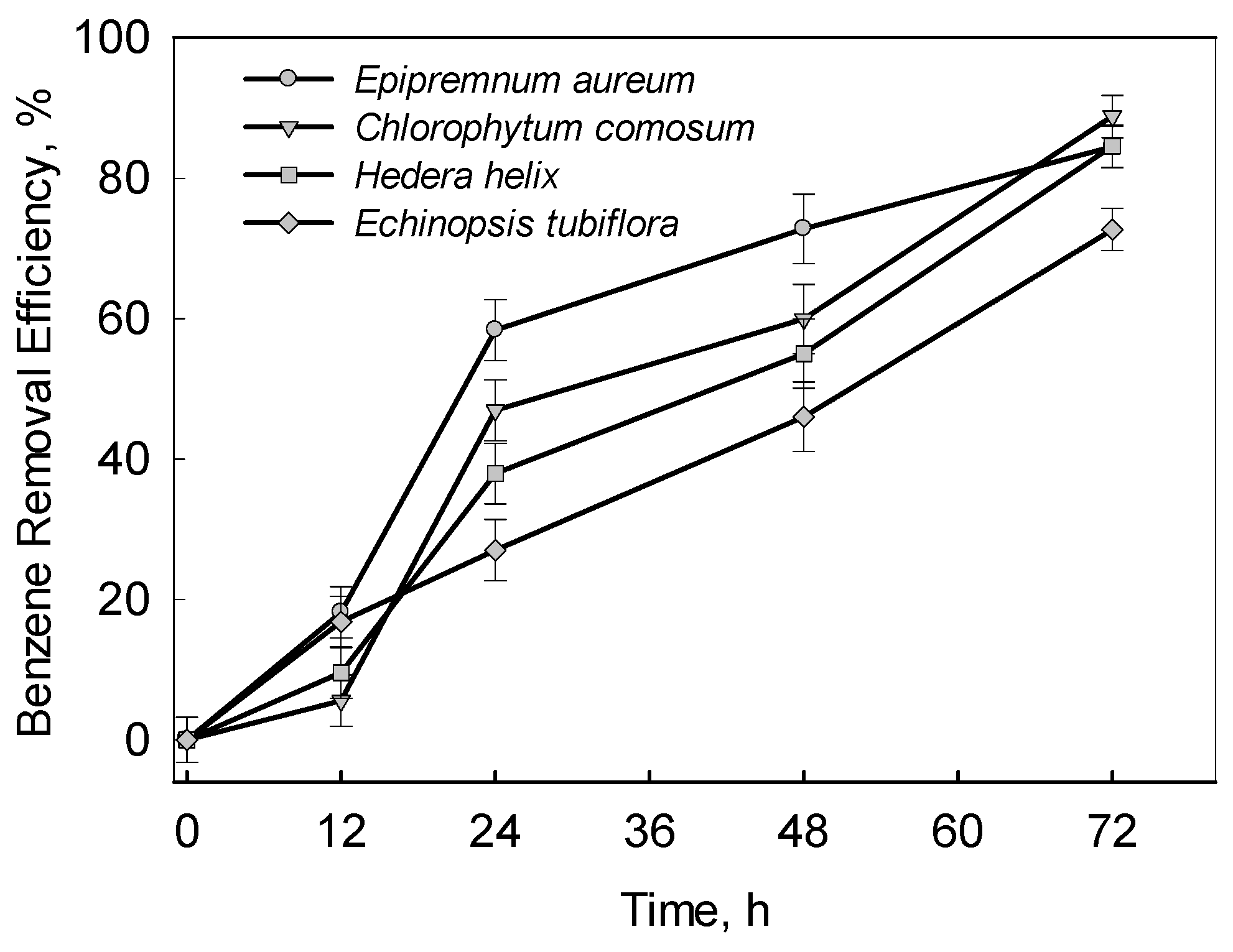
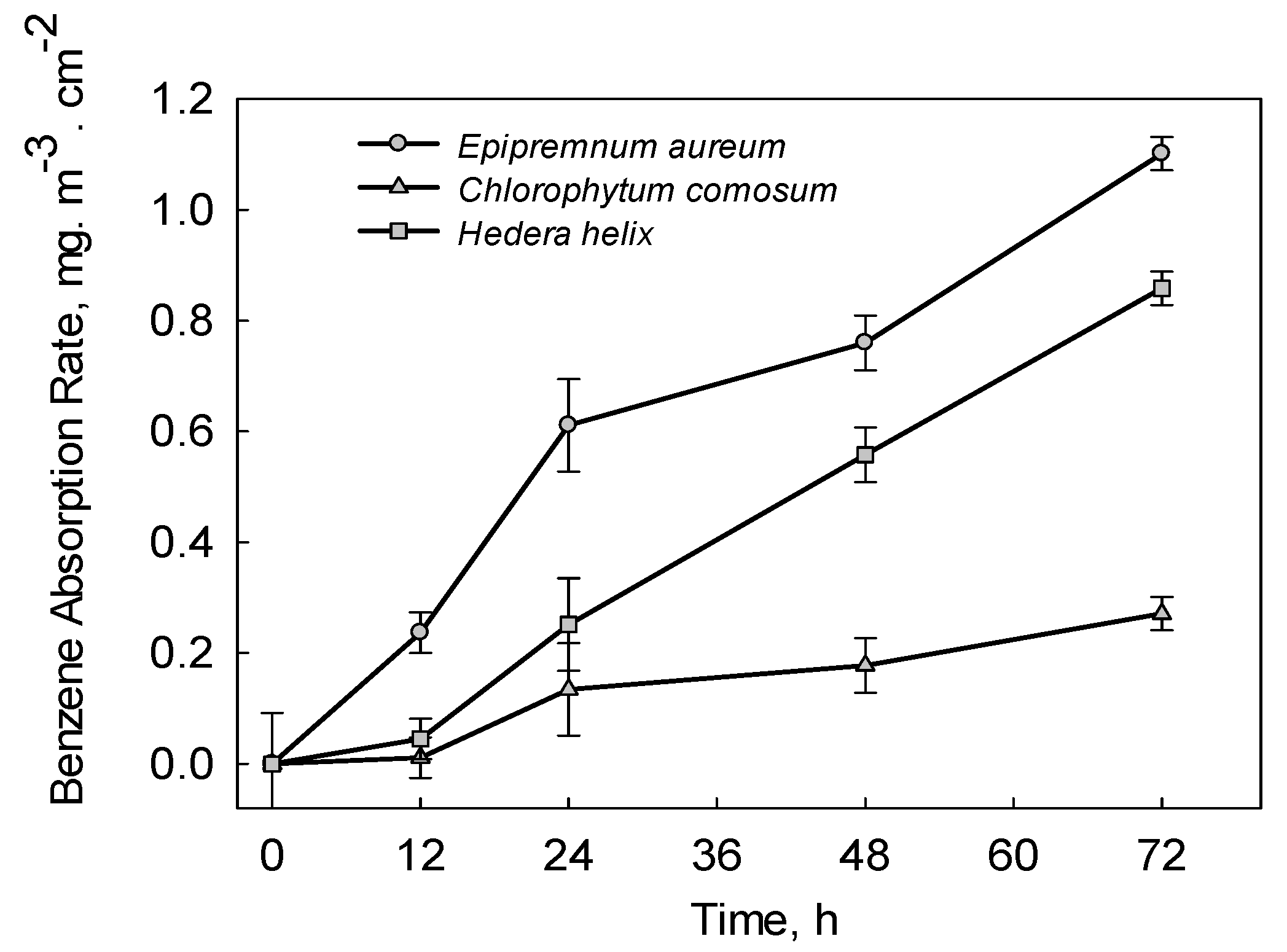
3. Results and Discussions
3.1. Benzene Removal Efficiency of Experimental Plants
3.2. Regulating Factors of Benzene Removal Efficiency
3.2.1. Chlorophyll Concentration, POD and Transpiration Rate
3.2.2. Effect of Light Conditions
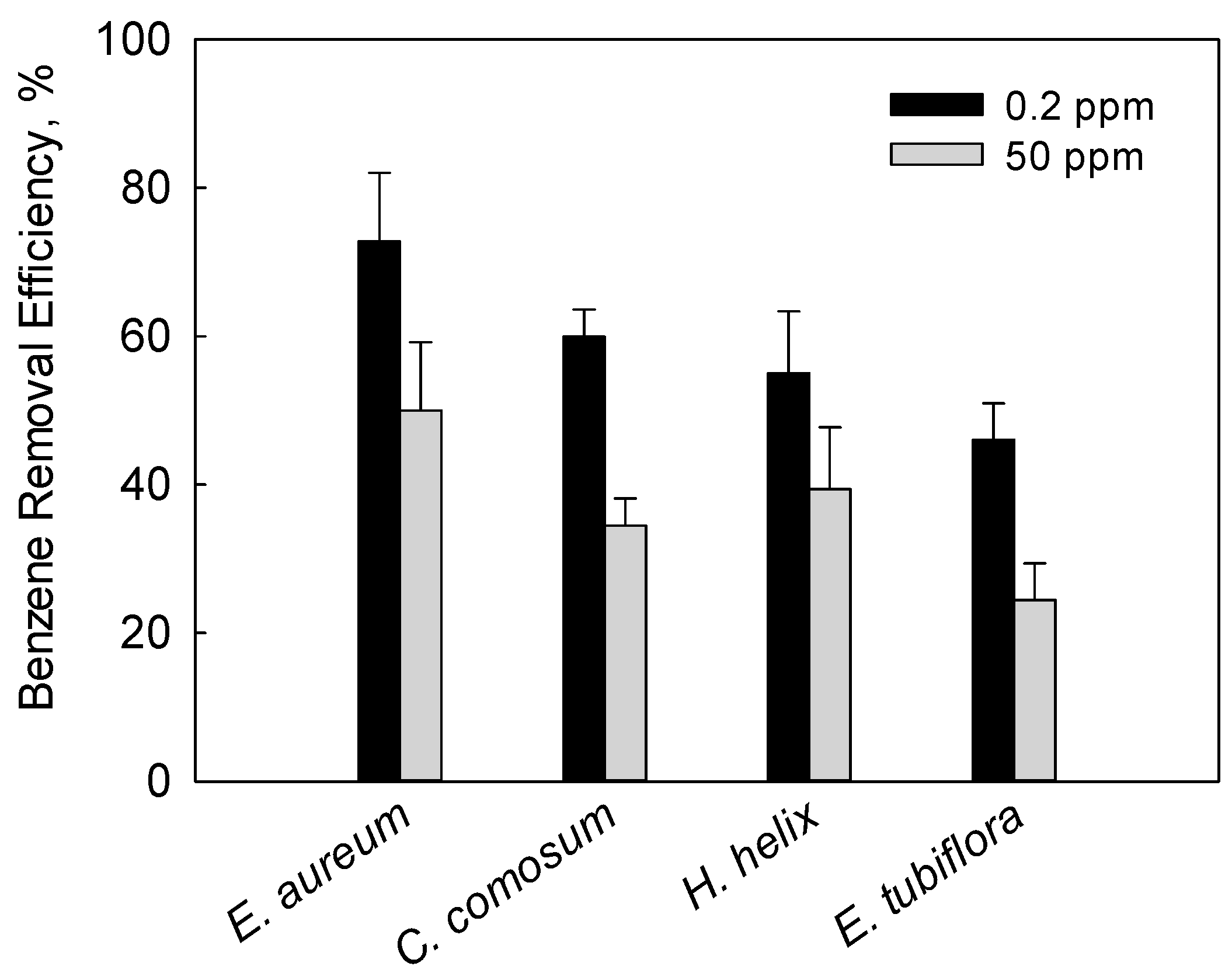
3.3.3. Effect of Benzene Concentration
4. Conclusion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rinsky, R.A. Benzene and leukemia an epidemiological risk assessment. Environ. Health Perspect. 1989, 82, 189–191. [Google Scholar] [CrossRef]
- Schnatter, A.R.; Rosamilia, K.; Wojcik, N.C. Review of the literature on benzene exposure and leukemia subtypes. Chem. Biol. Interact. 2005, 153, 9–21. [Google Scholar] [CrossRef]
- Klepeis, N.E.; Nelson, W.C.; Ott, W.R.; Robinson, J.P.; Tsang, A.M.; Switzer, P.; Behar, J.V. The national human activity pattern survey (nhaps): A resource for assessing exposure to environmental pollutants. J. Expo. Anal. Environ. Epidemiol. 2001, 11, 231–252. [Google Scholar] [CrossRef]
- Yu, B.F.; Hu, Z.B.; Liu, M.; Yang, H.L.; Kong, Q.X.; Liu, Y.H. Review of research on air-conditioning systems and indoor air quality control for human health. Int. J. Refrig. 2009, 32, 3–20. [Google Scholar] [CrossRef]
- Guieysse, B.; Hort, C.; Platel, V.; Munoz, R.; Ondarts, M.; Revah, S. Biological treatment of indoor air for VOC removal: Potential and challenges. Biotechnol. Adv. 2008, 26, 398–410. [Google Scholar] [CrossRef] [PubMed]
- Wolverton, B.C.; McDonald, R.C.; Watkins, E. Foliage plants for removing indoor air pollutants from energy-efficient homes. Econ. Bot. 1984, 38, 224–228. [Google Scholar] [CrossRef]
- Godish, T.; Guindon, C. An assessment of botanical air purification as a formaldehyde mitigation measure under dynamic laboratory chamber conditions. Environ. Pollut. 1989, 62, 13–20. [Google Scholar] [CrossRef]
- Dela Cruz, M.; Christensen, J.H.; Thomsen, J.D.; Muller, R. Can ornamental potted plants remove volatile organic compounds from indoor air? A review. Environ. Sci. Pollut. Res. Int. 2014, 21, 13909–13928. [Google Scholar] [CrossRef] [PubMed]
- Abbass, O.A.; Sailor, D.J.; Gall, E.T. Effectiveness of indoor plants for passive removal of indoor ozone. Build. Environ. 2017, 119, 62–70. [Google Scholar] [CrossRef]
- Dela Cruz, M.; Muller, R.; Svensmark, B.; Pedersen, J.S.; Christensen, J.H. Assessment of volatile organic compound removal by indoor plants—A novel experimental setup. Environ. Sci. Pollut. Res. Int. 2014, 21, 7838–7846. [Google Scholar] [CrossRef]
- Orwell, R.L.; Wood, R.L.; Tarran, J.; Torpy, F.; Burchett, M.D. Removal of benzene by the indoor plant/substrate microcosm and implications for air quality. Water Air Soil Pollut. 2004, 82, 189–191. [Google Scholar] [CrossRef]
- Yoo, M.H.; Kwon, Y.J.; Son, K.; Kays, S.J. Efficacy of indoor plants for the removal of single and mixed volatile organic pollutants and physiological effects of the volatiles on the plants. J. Am. Soc. Hortic. Sci. 2006, 131, 452–458. [Google Scholar] [CrossRef]
- Liu, Y.-J.; Mu, Y.-J.; Zhu, Y.-G.; Ding, H.; Crystal Arens, N. Which ornamental plant species effectively remove benzene from indoor air? Atmos. Environ. 2007, 41, 650–654. [Google Scholar] [CrossRef]
- Yang, D.S.; Pennisi, S.V.; Son, K.C.; Kays, S.J. Screening indoor plants for volatile organic pollutant removal efficiency. HortScience 2009, 44, 1377–1381. [Google Scholar] [CrossRef]
- Chun, S.C.; Yoo, M.H.; Moon, Y.S.; Shin, M.H.; Son, K.C.; Chung, I.M.; Kays, S. Effect of bacterial population from rhizosphere of various foliage plants on removal of indoor volatile organic compounds. Korean J. Hortic. Sci. Technol. 2010, 28, 476–483. [Google Scholar]
- Sriprapat, W.; Thiravetyan, P. Phytoremediation of BTEX from indoor air by Zamioculcas zamiifolia. Water Air Soil Pollut. 2013, 224, 1482. [Google Scholar] [CrossRef]
- Wood, R.A.; Burchett, M.D.; Alquezar, R.; Orwell, R.L.; Tarran, J.; Torpy, F. The potted-plant microcosm substantially reduces indoor air voc pollution: I. Office field-study. Water Air Soil Pollut. 2006, 175, 163–180. [Google Scholar] [CrossRef]
- Treesubsuntorn, C.; Thiravetyan, P. Removal of benzene from indoor air by Dracaena sanderiana: Effect of wax and stomata. Atmos. Environ. 2012, 57, 317–321. [Google Scholar] [CrossRef]
- Irga, P.J.; Torpy, F.R.; Burchett, M.D. Can hydroculture be used to enhance the performance of indoor plants for the removal of air pollutants? Atmos. Environ. 2013, 77, 267–271. [Google Scholar] [CrossRef]
- Weyens, N.; Thijs, S.; Popek, R.; Witters, N.; Przybysz, A.; Espenshade, J.; Gawronska, H.; Vangronsveld, J.; Gawronski, S.W. The role of plant-microbe interactions and their exploitation for phytoremediation of air pollutants. Int. J. Mol. Sci. 2015, 16, 25576–25604. [Google Scholar] [CrossRef]
- Nelson, M.; Wolverton, B.C. Plants+soil/wetland microbes: Food crop systems that also clean air and water. Adv. Space Res. 2011, 47, 582–590. [Google Scholar] [CrossRef]
- Giese, M.; Bauerdoranth, U.; Langebartels, C.; Sander-mann, H. Detoxification of formaldehyde by the spider plant (Chlorophytum comosum L.) and by soybean (Glycine max L.) cell-suspension cultures. J. Plant Physiol. 1994, 104, 1301–1309. [Google Scholar] [CrossRef]
- Biddington, N.; Thomas, T.H. Influence of different cytokinins on the transpiration and senescence of excised oat leaves. Physiol. Plant. 1987, 42, 369–374. [Google Scholar] [CrossRef]
- Lim, Y.W.; Kim, H.H.; Yang, J.Y.; Kim, K.J.; Lee, J.Y.; Shin, D.C. Improvement of indoor air quality by houseplants in new-built apartment buildings. Hortc. J. 2009, 78, 456–462. [Google Scholar] [CrossRef]
- Kvesitadze, E.; Sadunishvili, T.; Kvesitadze, G. Mechanisms of organic contaminants uptake and degradation in plants. Int. J. Biol. Biomol. Agric. Food Biotechnol. Eng. 2009, 3, 361–371. [Google Scholar]
- Sriprapat, W.; Boraphech, P.; Thiravetyan, P. Factors affecting xylene-contaminated air removal by the ornamental plant Zamioculcas zamiifolia. Environ. Sci. Pollut. Res. Int. 2014, 21, 2603–2610. [Google Scholar] [CrossRef]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence—A practical guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef]
- Sadunishvili, T.; Kvesitadze, E.; Betsiashvili, M.; Kuprava, N.; Zaalishvili, G.; Kvesitadze, G. Influence of hydrocarbons on plant cell ultrastructure and main metabolic enzymes. World Acad. Sci. Eng. Technol. 2009, 3, 271–276. [Google Scholar]
- Takahama, U.; Oniki, T. A peroxidase/phenolics/ascorbate system can scavenge hydrogen peroxide in plant cells. J. Plant Physiol. 1997, 101, 845–852. [Google Scholar] [CrossRef]
- Jin, C.J.; Zhou, X.J.; Zhao, H.T.; Liu, X.M.; Feng, K. Comparison of removal of formaldehyde capacity between Hedera helix and Melissa officinalis. Asian J. Chem. 2013, 25, 3823–3826. [Google Scholar] [CrossRef]
- Collins, C.; Fryer, M.; Grosso, A. Plant uptake of non-ionic organic chemicals. Environ. Sci. Technol. 2006, 40, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S. Stomatal opening mechanism of CAM plants. J. Plant Biol. 2010, 53, 19–23. [Google Scholar] [CrossRef]
| Plant Species | Mean Concentration ± S.D. (mg·L−1) | ||||
|---|---|---|---|---|---|
| 0 h | 12 h | 24 h | 48 h | 72 h | |
| Epipremnum Aureum | 38.63 ± 0.05b | 37.79 ± 0.06b | 32.93 ± 0.05a | 22.14 ± 0.05b | 20.71 ± 0.07b |
| Chlorophytum Comosum | 28.06 ± 0.07c | 25.91 ± 0.06c | 20.32 ± 0.04c | 14.82 ± 0.04c | 10.82 ± 0.05c |
| Hedera Helix | 44.84 ± 0.08a | 38.16 ± 0.05a | 27.96 ± 0.03b | 24.37 ± 0.07a | 22.57 ± 0.05a |
| Plant Species | Mean Concentration ± S.D.(U·(g·min)−1) | ||||
|---|---|---|---|---|---|
| 0 h | 12 h | 24 h | 48 h | 72 h | |
| Epipremnum Aureum | 20.06 ± 0.16a | 23.04 ± 0.21a | 27.80 ± 0.30a | 29.82 ± 0.04b | 36.44 ± 0.06a |
| Chlorophytum Comosum | 16.39 ± 0.23c | 17.90 ± 0.20c | 19.75 ± 0.20b | 20.31 ± 0.10c | 21.20 ± 0.07b |
| Hedera Helix | 18.44 ± 0.14b | 22.47 ± 0.15a | 27.37 ± 0.21a | 30.55 ± 0.20a | 32.50 ± 0.06a |
| Plant Species | Mean Concentration ± S.D. (10-6 g·m−2) | ||||
|---|---|---|---|---|---|
| 0 h | 12 h | 24 h | 48 h | 72 h | |
| Epipremnum Aureum | 12.61 ± 0.21a | 10.14 ± 0.21a | 3.65 ± 0.11a | 3.26 ± 0.07a | 2.26 ± 0.06a |
| Chlorophytum Comosum | 0.39 ± 0.12b | 0.38 ± 0.11b | 0.26 ± 0.12b | 0.25 ± 0.14b | 0.18 ± 0.09b |
| Hedera Helix | 0.24 ± 0.13b | 0.22 ± 0.09b | 0.18 ± 0.11b | 0.04 ± 0.02c | 0.03 ± 0.02c |
| Plant Species | Chlorophyll Concentration | Transpiration Rate | POD Activity |
|---|---|---|---|
| Epipremnum Aureum | 0.901 * | 0.711 * | 0.469 |
| Chlorophytum Comosum | 0.751 * | 0.871 * | 0.420 |
| Hedera Helix | 0.875 * | 0.710 * | 0.481 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gong, Y.; Zhou, T.; Wang, P.; Lin, Y.; Zheng, R.; Zhao, Y.; Xu, B. Fundamentals of Ornamental Plants in Removing Benzene in Indoor Air. Atmosphere 2019, 10, 221. https://doi.org/10.3390/atmos10040221
Gong Y, Zhou T, Wang P, Lin Y, Zheng R, Zhao Y, Xu B. Fundamentals of Ornamental Plants in Removing Benzene in Indoor Air. Atmosphere. 2019; 10(4):221. https://doi.org/10.3390/atmos10040221
Chicago/Turabian StyleGong, Yu, Tao Zhou, Peiran Wang, Yinuo Lin, Ruomeng Zheng, Youcai Zhao, and Bin Xu. 2019. "Fundamentals of Ornamental Plants in Removing Benzene in Indoor Air" Atmosphere 10, no. 4: 221. https://doi.org/10.3390/atmos10040221
APA StyleGong, Y., Zhou, T., Wang, P., Lin, Y., Zheng, R., Zhao, Y., & Xu, B. (2019). Fundamentals of Ornamental Plants in Removing Benzene in Indoor Air. Atmosphere, 10(4), 221. https://doi.org/10.3390/atmos10040221






