VOCs Measurements in Residential Buildings: Quantification via Thermal Desorption and Assessment of Indoor Concentrations in a Case-Study
Abstract
1. Introduction
2. Experimental Section
2.1. Vocs Sampling and Analysis by TD-GC-FID: Method Optimization
2.1.1. Solid-Adsorbent Based Sampling
2.1.2. Chemicals and Multistandard Solutions
2.1.3. Analytical Instrumentation and Analysis
2.2. Case-Study in Residential Buildings
3. Results and Discussion
3.1. TD-GC-FID Method Optimization
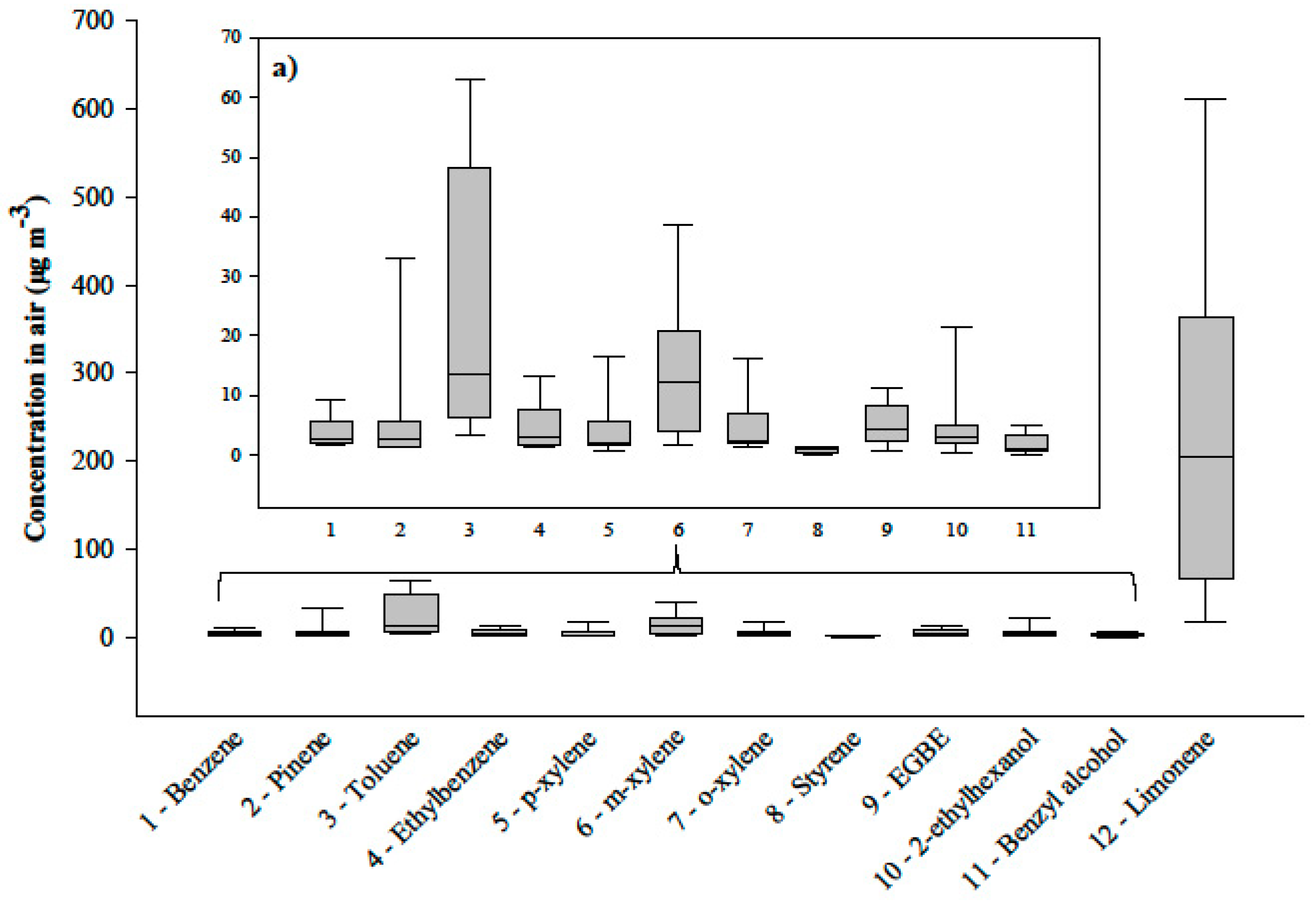
3.2. Vocs Concentrations in Residential Buildings
3.3. Comparison of Vocs Concentrations with Other Studies
3.4. Strengths and Limitations of the Study
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schweizer, C.; Edwards, R.; Bayer-Oglesby, L.; Gauderman, W.; Ilacqua, V.; Jantunen, M.; Lai, H.; Nieuwenhuijsen, M.; Kunzli, N. Indoor time-microenvironment-activity patterns in seven regions of Europe. J. Expo. Sci. Environ. Epidemiol. 2007, 17, 170–181. [Google Scholar] [CrossRef] [PubMed]
- Nørgaard, A.W.; Kofoed-Sørensen, V.; Mandin, C.; Ventura, G.; Mabilia, R.; Perreca, E.; Cattaneo, A.; Spinazzè, A.; Mihucz, V.G.; Szigeti, T.; et al. Ozone-initiated terpene reaction products in five European offices: Replacement of a floor cleaning agent. Environ. Sci. Technol. 2014, 48, 13331–13339. [Google Scholar] [CrossRef] [PubMed]
- Mandin, C.; Trantallidi, M.; Cattaneo, A.; Canha, N.; Mihucz, V.G.; Szigeti, T.; Mabilia, R.; Perreca, E.; Spinazzè, A.; Fossati, S.; et al. Assessment of indoor air quality in office buildings across Europe—The OFFICAIR study. Sci. Total Environ. 2017, 579, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.M.; Barratt, B.; Carslaw, N.; Doutsi, A.; Dunmore, R.E.; Ward, M.W.; Lewis, A.C. Unexpectedly high concentrations of monoterpenes in a study of UK homes. Environ. Sci. Process Impacts 2017, 19, 528–537. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, R.; Arey, J. Atmospheric degradation of volatile organic compounds. Chem. Rev. 2003, 103, 4605–4638. [Google Scholar] [CrossRef] [PubMed]
- Walser, M.L.; Desyaterik, Y.; Laskin, J.; Laskin, A.; Nizkorodov, S.A. High-resolution mass spectrometric analysis of secondary organic aerosol produced by ozonation of limonene. Phys. Chem. Chem. Phys. 2008, 10, 1009–1022. [Google Scholar] [CrossRef] [PubMed]
- Rohr, A.C. The health significance of gas- and particle-phase terpene oxidation products: A review. Environ. Int. J. 2013, 60, 145–162. [Google Scholar] [CrossRef]
- Wells, J.R.; Ham, J.E. A new agent for derivatizing carbonyl species used to investigate limonene ozonolysis. Atmos. Environ. 2014, 99, 519–526. [Google Scholar] [CrossRef]
- Carslaw, N. A new detailed chemical model for indoor air pollution. Atmos. Environ. 2007, 41, 1164–1179. [Google Scholar] [CrossRef]
- Stone, D.; Whalley, L.K.; Heard, D.E. Tropospheric OH and HO2 radicals: Field measurements and model comparisons. Chem. Soc. Rev. 2012, 41, 6348–6404. [Google Scholar] [CrossRef]
- Carslaw, N.; Fletcher, L.; Heard, D.; Ingham, T.; Walker, H. Significant OH production under surface cleaning and air cleaning conditions: Impact on indoor air quality. Indoor Air 2017, 27, 1091–1100. [Google Scholar] [CrossRef] [PubMed]
- Koistinen, K.; Kotzias, D.; Kephalopoulos, S.; Schlitt, C.; Carrer, P.; Jantunen, M.; Kirchner, S.; McLaughlin, J.; Mølhave, L.; Fernandes, E.O.; et al. The INDEX project: Executive summary of a European Union project on indoor air pollutants. Allergy 2008, 63, 810–819. [Google Scholar] [CrossRef] [PubMed]
- Wolkoff, P. Indoor air pollutants in office environments: Assessment of comfort, health and performance. Int. J. Hyg. Environ. Health 2013, 216, 371–394. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO), Regional Office for Europe. Guidelines for Indoor Air Quality: Selected Pollutants; WHO: Copenhagen, Denmark, 2010. [Google Scholar]
- Cacho, C.; Ventura Silva, G.; Martins, A.O.; Fernandes, E.O.; Saraga, D.E.; Dimitroulopoulou, C.; Bartzis, J.G.; Rembges, D.; Barrero-Moreno, J.; Kotzias, D. Air pollutants in office environments and emissions from electronic equipment: A review. Fresen. Environ. Bull. 2013, 22, 2488–2497. [Google Scholar]
- Wilke, O.; Jann, O.; Broedner, D. VOC- and SVOC- emissions from adhesives, floor coverings and complete floor structures. Indoor Air 2004, 14, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Missia, D.A.; Demetriou, E.; Michale, N.; Tolis, E.I.; Bartzis, J.G. Indoor exposure from building materials: A field study. Atmos. Environ. 2010, 44, 4388–4395. [Google Scholar] [CrossRef]
- Geiss, O.; Giannopoulos, G.; Tirendi, S.; Barrero-Moreno, J.; Larsen, B.R.; Kotzias, D. The AIRMEX study—VOC measurement in public buildings and schools/kindergartens in eleven European cities: Statistical analysis of the data. Atmos. Environ. 2011, 45, 3676–3684. [Google Scholar] [CrossRef]
- Hӓnninen, O.O.; Alm, S.; Katsouyanni, K.; Künzli, N.; Maroni, M.; Nieuwenhuijsen, M.J.; Saarela, K.; Srám, R.J.; Zmirou, D.; Jantunen, M.J. The EXPOLIS study: Implications for exposure research and environmental policy in Europe. J. Expo. Anal. Environ. Epidemiol. 2004, 14, 440–456. [Google Scholar] [CrossRef] [PubMed]
- Schlink, U.; Thiem, A.; Kohajda, T.; Richter, M.; Strebel, K. Quantile regression of indoor air concentrations of volatile organic compounds (VOC). Sci. Total Environ. 2010, 408, 3840–3851. [Google Scholar] [CrossRef] [PubMed]
- Weschler, C.J. Changes in indoor pollutants since the 1950s. Atmos. Environ. 2009, 43, 153–169. [Google Scholar] [CrossRef]
- Dunagan, S.C.; Dodson, R.E.; Rudel, R.A.; Brody, J.G. Toxics use reduction in the home: Lessons learned from household exposure studies. J. Clean. Prod. 2011, 19, 438–444. [Google Scholar] [CrossRef] [PubMed]
- Hӓnninen, O.O.; Alm, S.; Kaarakainen, E.; Jantunen, M. The EXPOLIS Databases, B13/2002; Publications of the National Public Health Institute: Kuopio, Finland, 2002. [Google Scholar]
- Stranger, M.; Potgieter-Vermaak, S.S.; Van Grieken, R. Comparative overview of indoor air quality in Antwerp, Belgium. Environ. Int. 2007, 33, 789–797. [Google Scholar] [CrossRef] [PubMed]
- McClenny, W.A.; Coløn, M. Measurement of volatile organic compounds by the US Environmental Protection Agency Compendium Method TO-17 Evaluation of performance criteria. J. Chromatogr. A 1998, 813, 101–111. [Google Scholar] [CrossRef]
- Peng, C.-Y.; Batterman, S. Performance evaluation of a sorbent tube sampling method using short path thermal desorption for volatile organic compunds. J. Environ. Monit. 2000, 2, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Sin, D.W.; Wong, Y.; Sham, W.; Wang, D. Development of an analytical technique and stability evaluation of 143 C3-C12 volatile organic compounds in Summa® canister by gas chromatography-mass spectrometry. Analyst 2001, 126, 310–321. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-H.; Lin, M.-N.; Feng, C.-T.; Yang, K.-L.; Lo, Y.-S.; Lo, J.-G. Measurement of toxic volatile organic compounds in indoor air of semiconductor foundries using multisorbent adsorption/thermal desorption coupled with gas chromatography-mass spectrometry. J. Chromatogr. A 2003, 996, 225–231. [Google Scholar] [CrossRef]
- Kuntasal, Ö.-O.; Karman, D.; Wang, D.; Tuncel, S.G.; Tuncel, G. Determination of volatile organic compounds in different microenvironments by multibed adsorbtion and short-path thermal desorption followed by gas chromatographic-mass spectrometry analysis. J. Chromatogr. A 2005, 1099, 43–54. [Google Scholar] [CrossRef]
- Ribes, A.; Carrera, G.; Gallego, E.; Roca, X.; José Berenguer, M.; Guardino, X. Development and validation of a method for air-quality and nuisance odors monitoring of volatile organic compounds using multi-sorbent adsorption and gas chromatography/mass spectrometry thermal desorption system. J. Chromatogr. A 2007, 1140, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Kotzias, D.; Koistinen, K.; Kephalopoulos, S. The INDEX Project: Critical Appraisal of the Setting and Implementation of Indoor Exposure Limits in the EU; European Commision, Joint Research Center. Institute for Health and Consumer Protection: Ispra, Italy, 2005. [Google Scholar]
- Howard-Reed, C.; Corsi, R.L. Mass transfer of volatile organic compounds from drinking water to indoor air: The role of residential dishwashers. Environ. Sci. Technol. 1999, 33, 2266–2272. [Google Scholar] [CrossRef]
- Howard, C.; Corsi, R.L. Volatilization of chemicals from drinking water to indoor air: The role of residential washing machines. J. Air Waste Manag. 1998, 48, 907–914. [Google Scholar] [CrossRef]
- Cocheo, C.; Boaretto, C.; Pagani, D.; Quaglio, F.; Sacco, P.; Zaratin, L.; Cottica, D. Field evaluation of thermal and chemical desorption BTEX radial diffusive sampler radiello® compared with active pumped (samplers) for ambient air measurements. J. Environ. Monit. 2009, 11, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Ongwandee, M.; Moonrinta, R.; Panyametheekul, S.; Tangbanluekal, C.; Morrison, G. Investigation of volatile organic compounds in office buildings in Bangkok, Thailand: Concentrations, sources and occupant symptoms. Build. Environ. 2011, 46, 1512–1522. [Google Scholar] [CrossRef]
- Uhde, E.; Schulz, N. Impact of room fragrance products on indoor air quality. Atmos. Environ. 2015, 106, 492–502. [Google Scholar] [CrossRef]
- Edwards, R.D.; Jurvelin, J.; Saarela, K.; Jantunen, M. VOC concentrations measured in personal samples and residential indoor, outdoor and workplace microenvironments in EXPOLIS-Helsinki, Finland. Atmos. Environ. 2001, 35, 4531–4543. [Google Scholar] [CrossRef]
- Jia, C.; Batterman, S.; Godwin, C. VOCs in industrial, urban and suburban neighborhoods, Part 1: Indoor and outdoor concentrations, variation, and risk drivers. Atmos. Environ. 2008, 42, 2083–2100. [Google Scholar] [CrossRef]
- Campagnolo, D.; Saraga, D.E.; Cattaneo, A.; Spinazzè, A.; Mandin, C.; Mabilia, R.; Perreca, E.; Sakellaris, I.; Canha, N.; Mihucz, V.G.; et al. VOCs and aldehydes sources identification in European office buildings—The OFFICAIR study. Build. Environ. 2017, 115, 18–24. [Google Scholar] [CrossRef]
- Kotzias, D.; Geiss, O.; Leva, P.; Belintani, A.; Arvanitis, A.; Kephalopoulos, S. Impact of various air exchange rates on the levels of environmental tobacco smoke (ETS) components. Fresen. Environ. Bull. 2004, 13, 1536–1549. [Google Scholar]
- HSDB, Hazardous Substances Data Bank. Specialized Information Services. National Library of Medicine. 2003. Available online: https://toxnet.nlm.nih.gov/ (accessed on 10 October 2018).
- Katsoyiannis, A.; Leva, P.; Kotzias, D. VOC and carbonyl emissions from carpets: A comparative study using four types of environmental chambers. J. Hazard. Mater. 2008, 152, 669–676. [Google Scholar] [CrossRef]
- Yu, C.; Crump, D. A review of the emission of VOCs from polymeric materials used in buildings. Build. Environ. 1998, 33, 357–374. [Google Scholar] [CrossRef]
- Uhde, E.; Salthammer, T. Impact of reaction products from building materials and furnishings on indoor air quality—A review of recent advances in indoor chemistry. Atmos. Environ. 2007, 41, 3111–3128. [Google Scholar] [CrossRef]
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; Overall Evaluation of Carcinogenity: An Updating of IARC Monographs Volumes 1 to 42. Suppl. 7; International Agency for Research on Cancer: Lyon, France, 1987; pp. 38–74.
- Rohr, A.C.; Wilkins, C.K.; Clausen, P.A.; Hammer, M.; Nielsen, G.D.; Wolkoff, P.; Spengler, J.D. Upper airway and pulmonary effects of oxidation products of (+)-α-pinene, d-limonene, and isoprene in BALB/c mice. Inhal. Toxicol. 2002, 14, 663–684. [Google Scholar] [CrossRef] [PubMed]
- Petry, T.; Vitale, D.; Joachim, F.J.; Smith, B.; Cruse, L.; Mascarenhas, R.; Schneider, S.; Singal, M. Human health risk evaluation of selected VOC, SVOC and particulate emissions from scented candles. Regul. Toxicol. Pharm. 2014, 69, 55–70. [Google Scholar] [CrossRef] [PubMed]
- ECHA. Guidance on Information Requirements and Chemical Safety Assessment. Chapter R.15: Consumer Exposure Assessment. European Chemicals Agency (Version 3.0). July 2016. Available online: https://echa.europa.eu/documents/10162/13632/information_requirements_r15_en.pdf (accessed on 10 October 2018).
- Trantallidi, M.; Dimitroulopoulou, C.; Wolkoof, P.; Kephalopoulos, S.; Carrer, P. EPHECT III: Health risk assessment of exposure to household consumer product. Sci. Total Environ. 2015, 536, 903–913. [Google Scholar] [CrossRef] [PubMed]
- Nørgaard, A.W.; Kudal, J.D.; Kofoed-Sørensen, V.; Koponen, I.K.; Wolkoff, P. Ozone-initiated VOC and particle emissions from a cleaning agent and an air freshener: Risk assessment of acute airway effects. Environ. Int. 2014, 68, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Raw, G.J.; Coward, S.K.D.; Brown, V.M.; Crump, D.R. Exposure to air pollutants in English homes. J. Expo. Anal. Environ. Epidemiol. 2004, 14, S85–S94. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, F.; Tapia, A.; Amo-Salas, M.; Notario, A.; Cabañas, B.; Martínez, E. Levels and sources of volatile organic compounds including carbonyls in indoor air of homes in Puertollano, the most industrialized city in central Iberian Peninsula. Estimation of health risk. Int. J. Hyg. Environ. Health 2015, 218, 522–534. [Google Scholar] [CrossRef] [PubMed]
- Schlink, U.; Rehwagen, M.; Damm, M.; Richter, M.; Borte, M.; Herbarth, O. Seasonal cycle of indoor-VOCs: Comparison of apartments and cities. Atmos. Environ. 2004, 38, 1181–1190. [Google Scholar] [CrossRef]
- Lai, H.K.; Kendall, M.; Ferrier, H.; Lindup, I.; Alm, S.; Hӓnninen, O.; Jantunen, M.; Mathys, P.; Colvile, R.; Ashmore, M.R.; et al. Personal exposures and microenvironment concentrations of PM2.5, VOC, NO2 and CO in Oxford, UK. Atmos. Environ. 2004, 38, 6399–6410. [Google Scholar] [CrossRef]
- Schneider, P.; Gebefügi, I.; Richter, K.; Wölke, G.; Schnelle, J.; Wichmann, H.-E.; Heinrich, J.; INGA Study Group. Indoor and outdoor BTX levels in German cities. Sci. Total Environ. 2001, 267, 41–51. [Google Scholar] [CrossRef]
- Massolo, L.; Rehwagen, M.; Porta, A.; Ronco, A.; Herbarth, O.; Mueller, A. Indoor-outdoor distribution and risk assessment of volatile organic compounds in the atmosphere of industrial and urban areas. Environ. Toxicol. 2010, 25, 339–349. [Google Scholar] [CrossRef]
| Automated Thermal Desorber (ATD) | |
|---|---|
| Carrier gas | Helium |
| Primary desorption | 280 °C, 6 min |
| Total primary desorption flow-rate | 20 mL/min |
| Inlet split flow-rate | 6.8 mL/min |
| Outlet split flow-rate | 6.5 mL/min |
| Valve temperature | 200 °C |
| Cold trap temperature | −30 °C |
| Secondary desorption | 300 °C, 6 min |
| Transfer line temperature | 200 °C |
| GC system | |
| Carrier gas | Helium (1 mL/min) |
| Auxiliary gas | Nitrogen (30 mL/min) |
| Column temperature program | 50 °C, 5 min; 2 °C/min to 100 °C; 10 °C/min to 200 °C; 200 °C, 10 min |
| Total run time | 60 min |
| FID temperature | 250 °C |
| Sample Number | Type of Site and Location | Floor | Duration of the Dishwashing Cycle (min) | Total Sampling Time (min) | Type of Product Used * |
|---|---|---|---|---|---|
| 1 | Suburban site, on a private garden. Main road over 100 m | Ground floor | 50 | 80 | Type 1 |
| 2 | Suburban site, on a private courtyard. Main road over 100 m | 1st floor | 65 | 95 | Type 2 |
| 3 | Suburban site, on a private courtyard. Main road over 100 m | 1st floor | 85 | 115 | Type 2 |
| 4 | Urban site, on a private garden. Main road over 50 m | 1st floor | 85 | 115 | Type 3 |
| 5 | Suburban site, on a private courtyard. Main road over 100 m | 1st floor | 95 | 125 | Type 4 |
| 6 | Rural site, on a private courtyard and near a private garden. Main road over 150 m | 1st floor | 45 | 75 | Type 2 |
| 7 | Rural site, on a private courtyard | 1st floor | 85 | 115 | Type 4 |
| 8 | Rural site, on a private courtyard | 1st floor | 30 | 60 | Type 5 |
| 9 | Rural site, on a private garden | Ground floor | 90 | 120 | Type 6 |
| Compound | Retention Time (min) | Mass Range for Calibration Curve (ng) | R2 | LOD (ng–µg/m3) | LOQ (ng–µg/m3) | %RSD |
|---|---|---|---|---|---|---|
| Benzene | 9.5 | 9.7–79.1 | 0.9987 | 0.21–0.018 | 0.70–0.058 | 10.4 |
| α-Pinene | 12.7 | 9.6–78.4 | 0.9916 | 0.31–0.026 | 1.02–0.085 | n.d. |
| Toluene | 14.1 | 19.6–160.0 | 0.9998 | 0.21–0.018 | 0.71–0.059 | 1.2 |
| Ethylbenzene | 19.3 | 10.2–83.3 | 0.9996 | 0.26–0.022 | 0.86–0.072 | 2.7 |
| p-xylene | 19.9 | 10.2–83.4 | 0.9994 | 0.25–0.021 | 0.84–0.070 | 3.8 |
| m-xylene | 20.5 | 10.2–82.7 | 0.9985 | 0.26–0.022 | 0.85–0.071 | 1.9 |
| o-xylene | 23.4 | 10.3–84.0 | 0.9995 | 0.25–0.021 | 0.85–0.071 | 0.8 |
| d-Limonene | 23.9 | 9.6–78.2 | 0.9996 | 0.28–0.023 | 0.93–0.078 | n.d. |
| Styrene | 28.6 | 9.8–74.1 | 0.9967 | 0.15–0.013 | 0.50–0.042 | n.d. |
| EGBE | 35.6 | 10.6–86.7 | 0.9990 | 0.25–0.021 | 0.82–0.068 | 4.5 |
| 2-Ethylhexanol | 37.9 | 10.6–86.1 | 0.9950 | 0.14–0.012 | 0.47–0.039 | 12.2 |
| Benzyl alcohol | 46.7 | 11.7–95.0 | 0.9986 | 0.22–0.018 | 0.74–0.062 | n.d. |
| Compound | n | Mean (S.D.) (µg/m3) | Median (µg/m3) | Min–Max (µg/m3) | DNELs—Short-Term (mg/m3) | DNELs—Long-Term (mg/m3) |
|---|---|---|---|---|---|---|
| Benzene | 9 | 4.0 (2.5) | 2.8 | 1.8–9.3 | / | / |
| α-Pinene | 9 | 6.2 (10.3) | 2.8 | 1.2–33.1 | / | 5.69 |
| Toluene | 9 | 23.4 (23.3) | 13.4 | 3.2–63.1 | 384 | 192 |
| Ethylbenzene | 9 | 4.8 (4.0) | 3.1 | 1.2–13.1 | / | 15 |
| p-xylene | 9 | 4.3 (5.0) | 2.0 | 0.7–16.5 | 260 | 65 |
| m-xylene | 9 | 13.8 (11.6) | 12.4 | 1.7–38.6 | 260 | 65 |
| o-xylene | 9 | 4.9 (4.8) | 2.5 | 1.4–16.3 | 260 | 65 |
| d-Limonene | 9 | 231.5 (191.3) | 204.9 | 17.0–611.3 | 66.7 | 3.6 |
| Styrene | 9 | 0.9 (0.5) | 0.9 | <LOD–1.5 | 289 | 10.2 |
| EGBE | 9 | 5.2 (3.5) | 4.3 | 0.7–11.3 | / | 98 |
| 2-Ethylhexanol | 9 | 5.1 (6.4) | 3.0 | 0.5–21.6 | / | 12.8 |
| Benzyl alcohol | 9 | 1.7 (1.7) | 0.9 | <LOD–5.0 | / | 22 |
| Reference | Location | Sampling Information | Mean [Median] (µg/m3) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (Min–Max) or (Max) (µg/m3) | ||||||||||||
| Benzene | α-Pinene | Toluene | Ethylbenzene | p-xylene | m-xylene | o-xylene | d-Limonene | Styrene | 2-Ethylhexanol | |||
| This study | Como (Italy) | 2 h sampling during the dishwasher washing cycle | 4.0 [2.8] | 6.2 [2.8] | 23.4 [13.4] | 4.8 [3.1] | 4.3 [2.0] | 13.8 [12.4] | 4.9 [2.5] | 231.5 [204.9] | 0.9 [0.9] | 5.1 [3.0] |
| (1.8–9.3) | (1.2–33.1) | (3.2–63.1) | (1.2–13.1) | (0.7–16.5) | (1.7–38.6) | (1.4–16.3) | (17.0–611.3) | (<LOD–1.5) | (0.5–21.6) | |||
| [4] | York (UK) | 5-days sampling in homes | ||||||||||
| (7–19) | (2–229) | (18–1439) | ||||||||||
| [18] | Different cities across Europe | 7-days sampling in homes | 2.8 [1.9] | 14.5 [6.1] | 11.7 [6.5] | 1.5 [1.1] | 3.8 [2.8] | 1.8 [1.2] | 29.2 [9.5] | 0.4 [0] | ||
| (0.4–32.1) | (0.2–214.1) | (1.3–160.6) | (0.2–12.8) | (0.5–28.1) | (0.2–20.5) | (0–492.9) | (0–22.1) | |||||
| [37] | Helsinki (Finland) | 48 h sampling in residential indoor microenvironments | 2.2 | 16.1 | 20.4 | 2.9 | 7.8 | 2.5 | 31.6 | 1.2 | 3.7 | |
| (14.1) | (215.6) | (247.4) | (19.0) | (62.5) | (23.9) | (494.9) | (15.2) | (34.3) | ||||
| [38] | Michigan (USA) | 4-days sampling in living room | 2.8 [1.2] | 9.0 [3.2] | 15.6 [6.8] | 2.2 [1.0] | 7.9 [3.3] | 2.4 [1.1] | 25.7 [16.6] | 0.5 [0.3] | ||
| (47.4) | (139.2) | (197.3) | (79.9) | (318.7) | (50.4) | (258.5) | (6.6) | |||||
| [51] | England | 4-weeks sampling in the bedroom of each home | 3.0 | 15.1 | 3.8 | 6.2 | ||||||
| (<0.1–93.5) | (0.3–1783.5) | (0.1–152.8) | (<0.1–308.4) | |||||||||
| [52] | Puertollano (Spain) | 2-weeks sampling in living rooms | 1.9 [1.7] | 18.5 [13.4] | 12.0 [6.3] | 3.4 [2.7] | 7.0 [4.7] | 0.8 [0.4] | 17.1 [13.4] | 2.1 [1.9] | ||
| (0.7–5.1) | (2.5–63.1) | (2.6–87.9) | (0.7–13) | (1.6–20.4) | (<LOD-3.1) | (1.8–87.2) | (0.3–6.5) | |||||
| [53] | Different cities in Germany | 4-weeks sampling in different residential rooms | 3.2 [2.3] | 23.3 [9.8] | 29.5 [18.3] | 3.6 [1.9] | 9.8 [4.6] | 2.7 [1.4] | 32.9 [16.0] | 1.4 [0.6] | ||
| (12.0) | (141.8) | (142.9) | (19.1) | (47.9) | (15.3) | (172.5) | (8.8) | |||||
| [54] | Oxford (UK) | 48 h sampling in residential indoor microenvironments | 3.6 | 16.5 | 23.7 | 2.9 | 9.0 | 19.0 | ||||
| [55] | Hamburg (Germany) | 1-week sampling in living room and bedroom | [1.5] | [20.5] | [0.7] | [2.9] | [0.8] | |||||
| Erfurt (Germany) | 1-week sampling in living room and bedroom | [2.2] | [37.3] | [1.7] | [4.2] | [1.2] | ||||||
| [56] | La Plata (Argentina) | 4-weeks sampling in homes at an urban area | 3.6 [3.2] | 15.1 [11.7] | 1.4 [1.3] | 6.5 [6.3] | 1.6 [1.4] | 0.3 [0.2] | ||||
| (12.7) | (89.0) | (2.9) | (18.8) | (4.8) | (0.7) | |||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rovelli, S.; Cattaneo, A.; Fazio, A.; Spinazzè, A.; Borghi, F.; Campagnolo, D.; Dossi, C.; Cavallo, D.M. VOCs Measurements in Residential Buildings: Quantification via Thermal Desorption and Assessment of Indoor Concentrations in a Case-Study. Atmosphere 2019, 10, 57. https://doi.org/10.3390/atmos10020057
Rovelli S, Cattaneo A, Fazio A, Spinazzè A, Borghi F, Campagnolo D, Dossi C, Cavallo DM. VOCs Measurements in Residential Buildings: Quantification via Thermal Desorption and Assessment of Indoor Concentrations in a Case-Study. Atmosphere. 2019; 10(2):57. https://doi.org/10.3390/atmos10020057
Chicago/Turabian StyleRovelli, Sabrina, Andrea Cattaneo, Arianna Fazio, Andrea Spinazzè, Francesca Borghi, Davide Campagnolo, Carlo Dossi, and Domenico M. Cavallo. 2019. "VOCs Measurements in Residential Buildings: Quantification via Thermal Desorption and Assessment of Indoor Concentrations in a Case-Study" Atmosphere 10, no. 2: 57. https://doi.org/10.3390/atmos10020057
APA StyleRovelli, S., Cattaneo, A., Fazio, A., Spinazzè, A., Borghi, F., Campagnolo, D., Dossi, C., & Cavallo, D. M. (2019). VOCs Measurements in Residential Buildings: Quantification via Thermal Desorption and Assessment of Indoor Concentrations in a Case-Study. Atmosphere, 10(2), 57. https://doi.org/10.3390/atmos10020057









