Aerosol from Biomass Combustion in Northern Europe: Influence of Meteorological Conditions and Air Mass History
Abstract
1. Introduction
2. Methods
2.1. Measurement Site
2.2. Alkali Aerosol Mass Spectrometer Measurements
2.3. Supporting Measurements
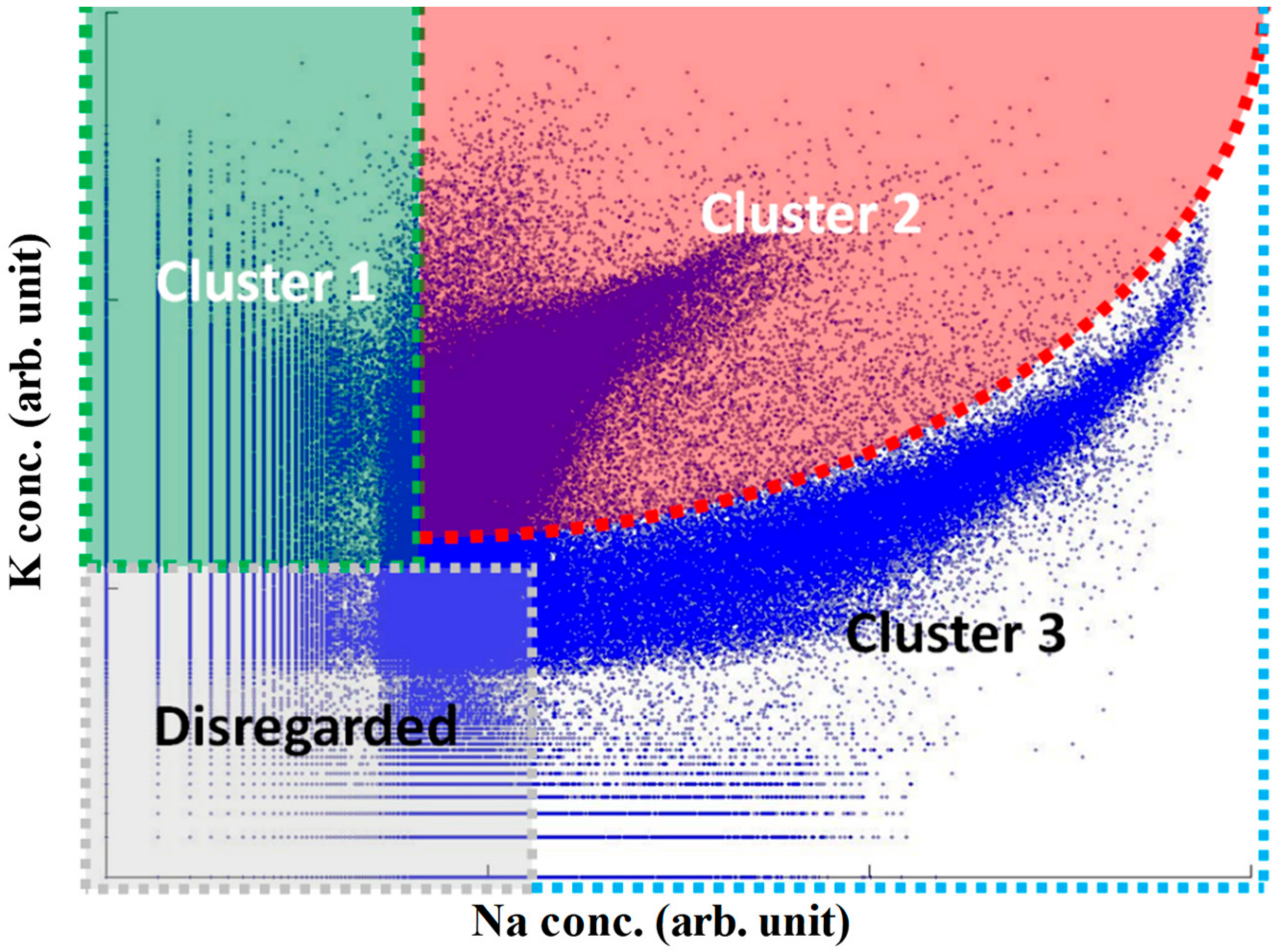
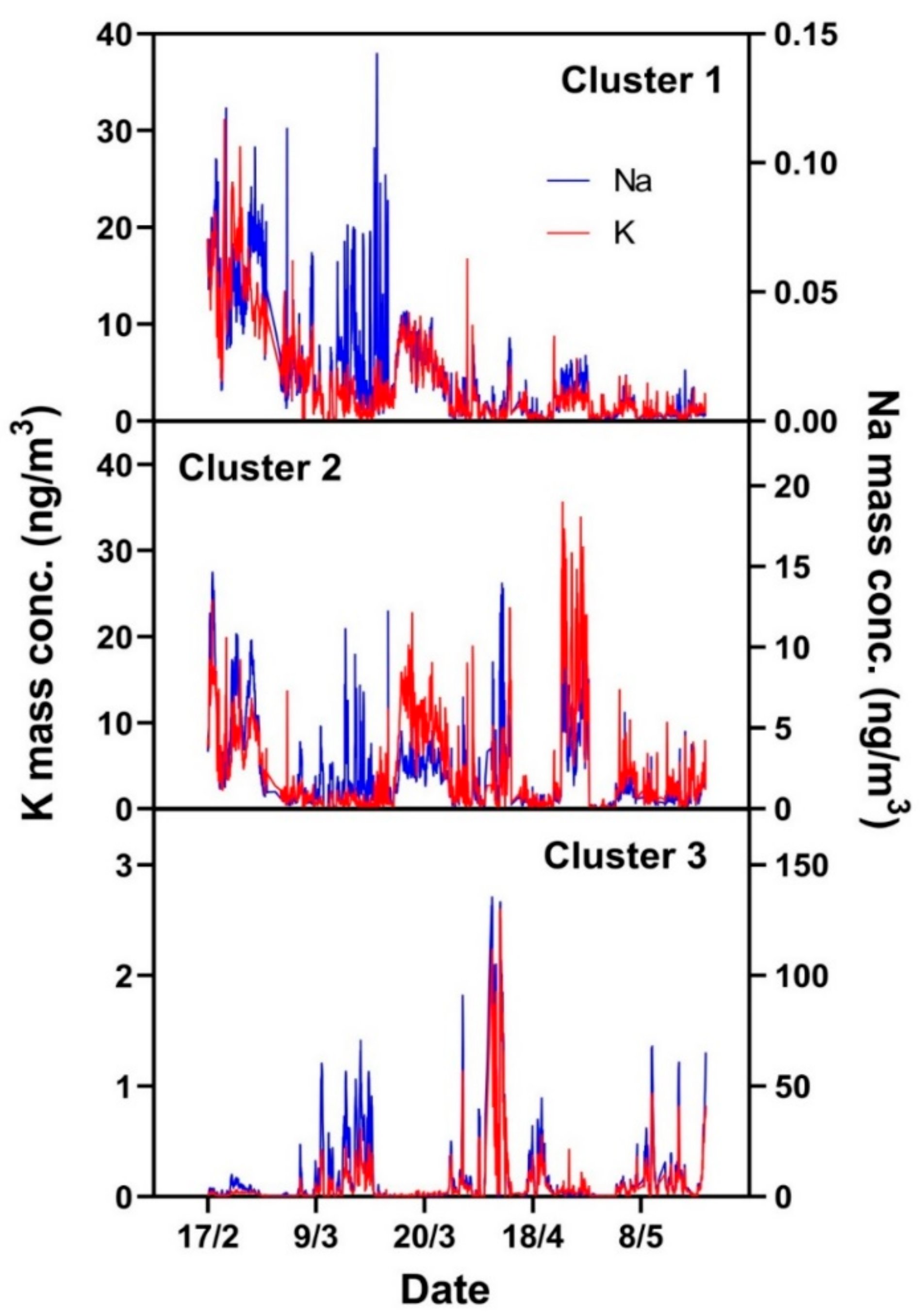
2.4. Cluster Analysis of Mass Spectra
2.5. Air Mass Back-Trajectories
2.6. Chemical Transport Modelling
3. Results and Discussion
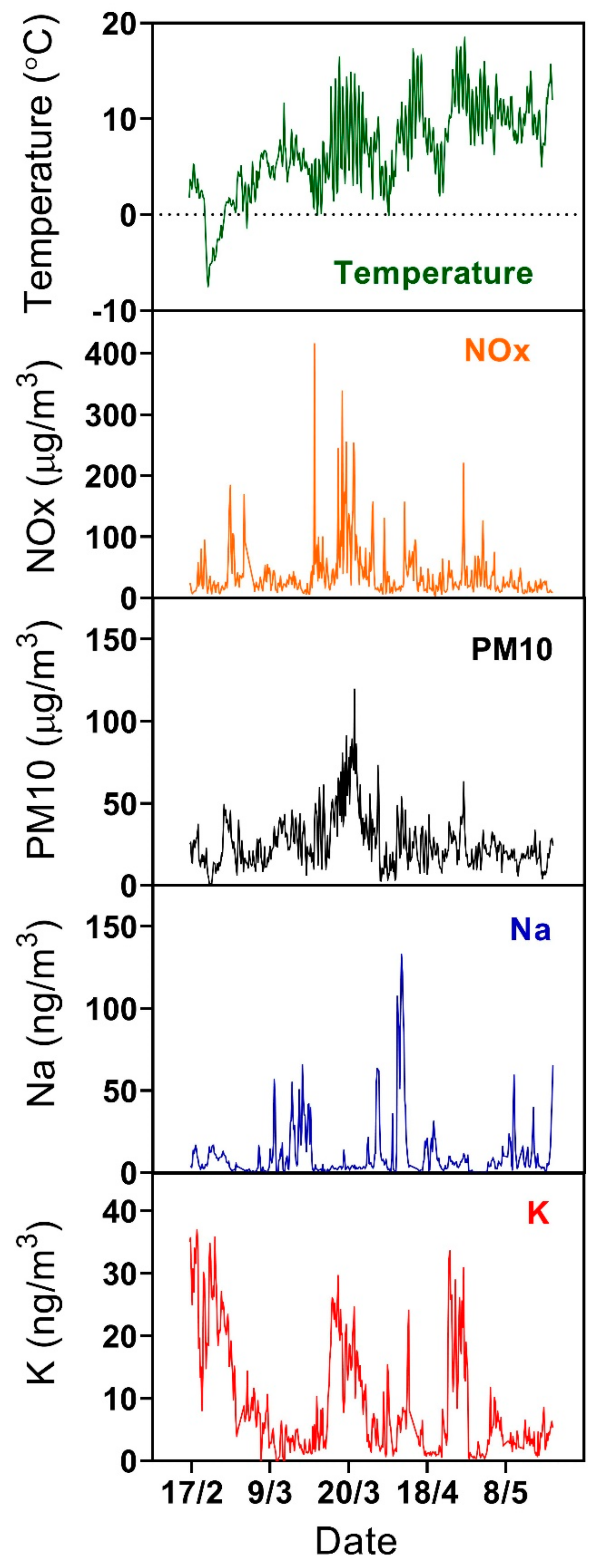
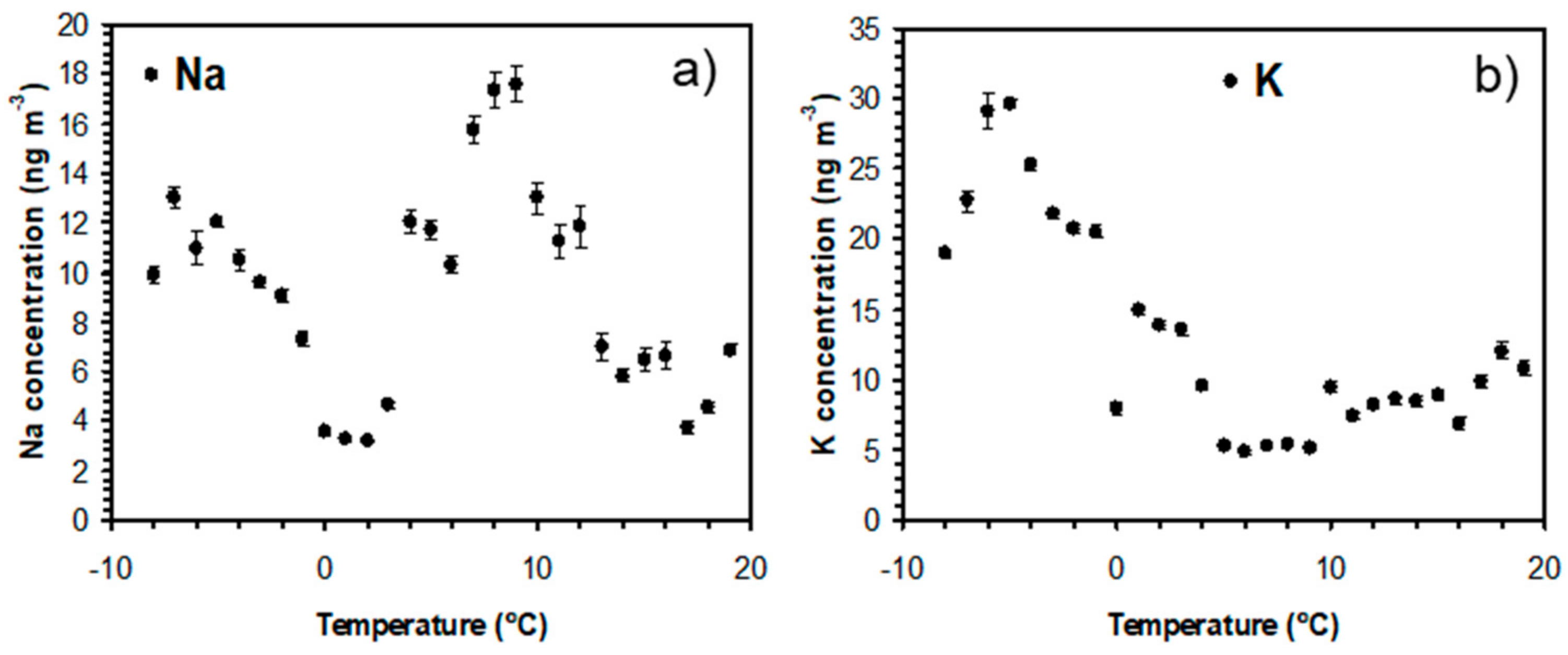
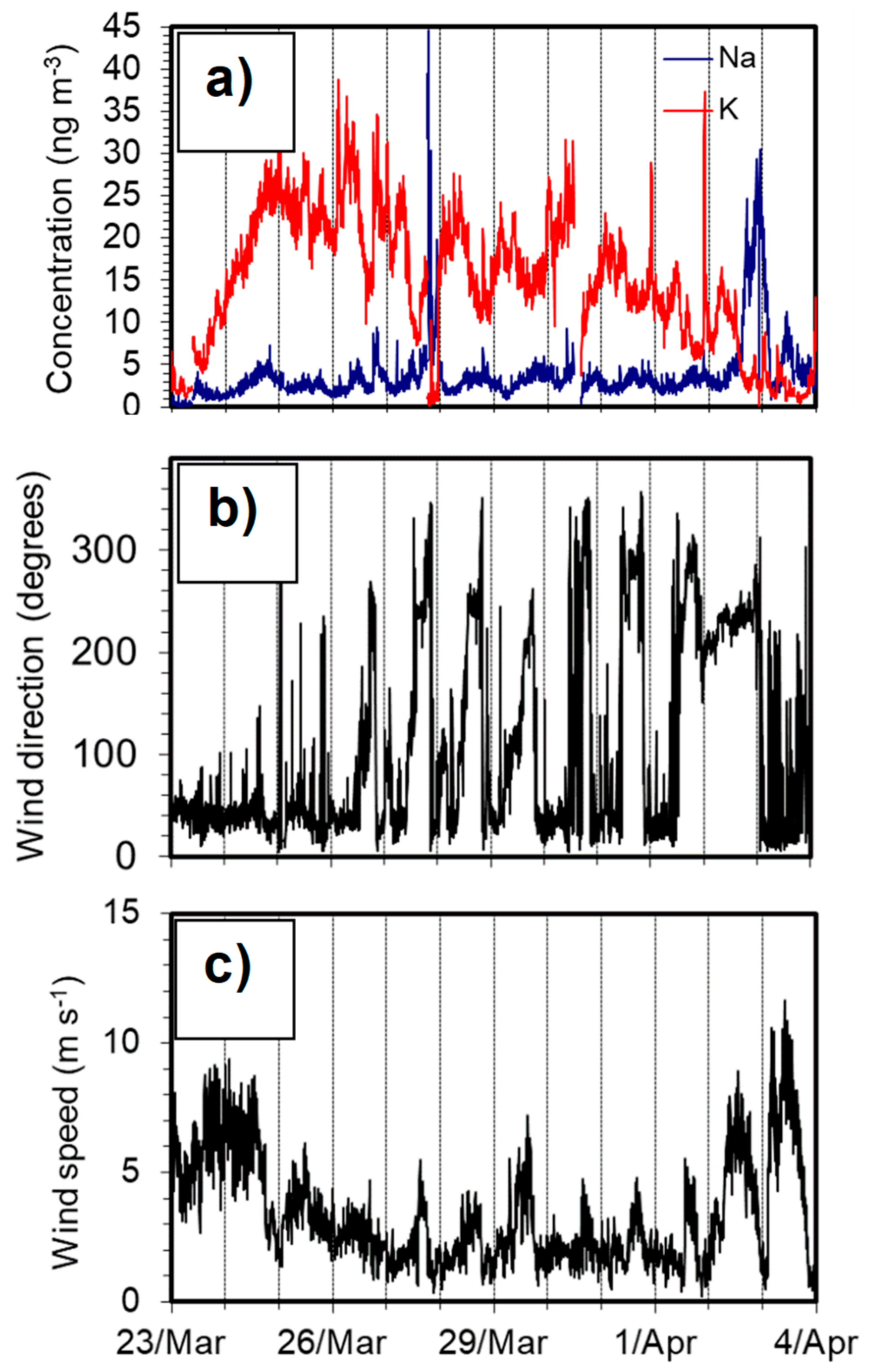
3.1. Overview of Measured K and Na Concentrations
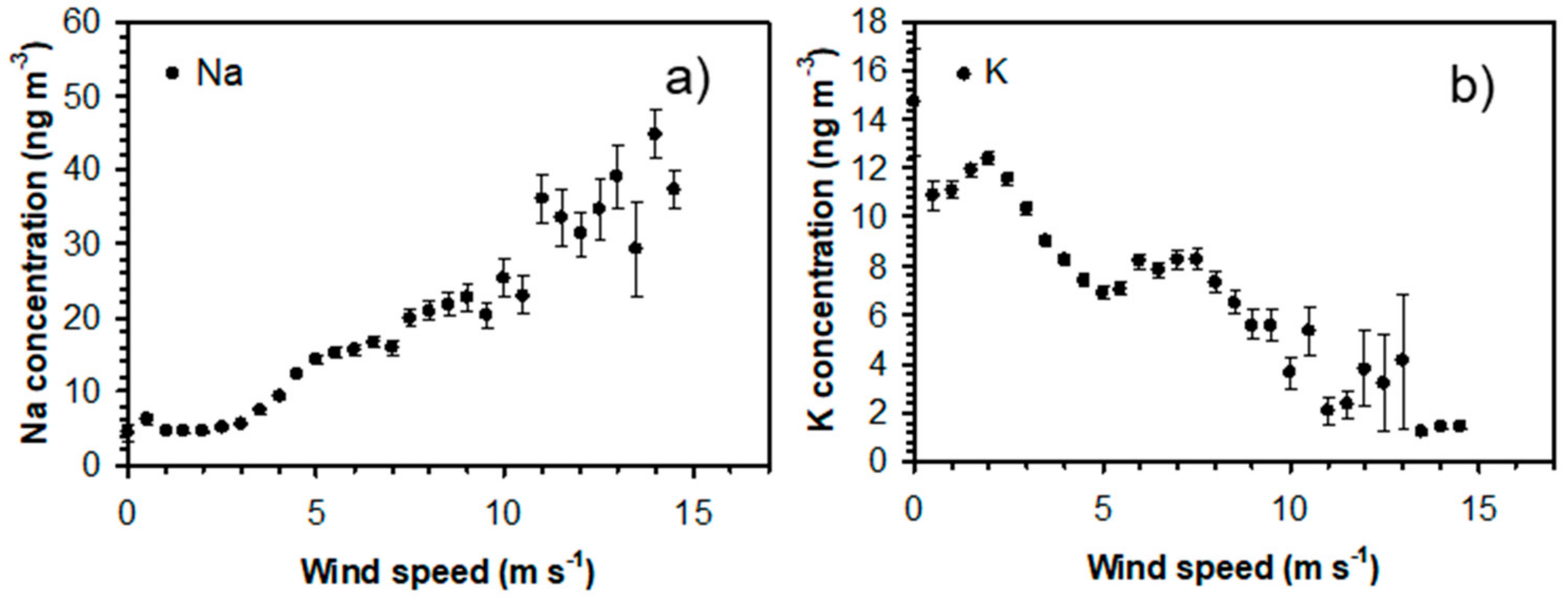
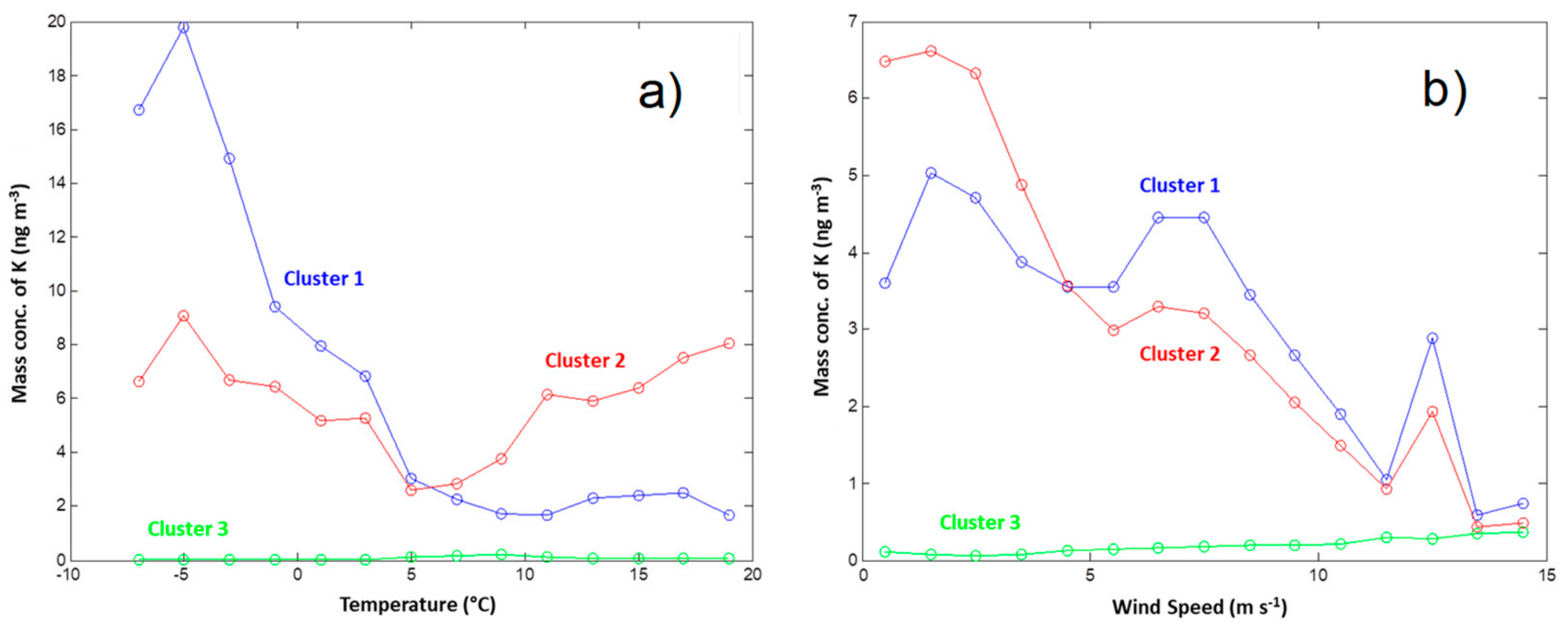
3.2. Influence of Local Meteorological Conditions
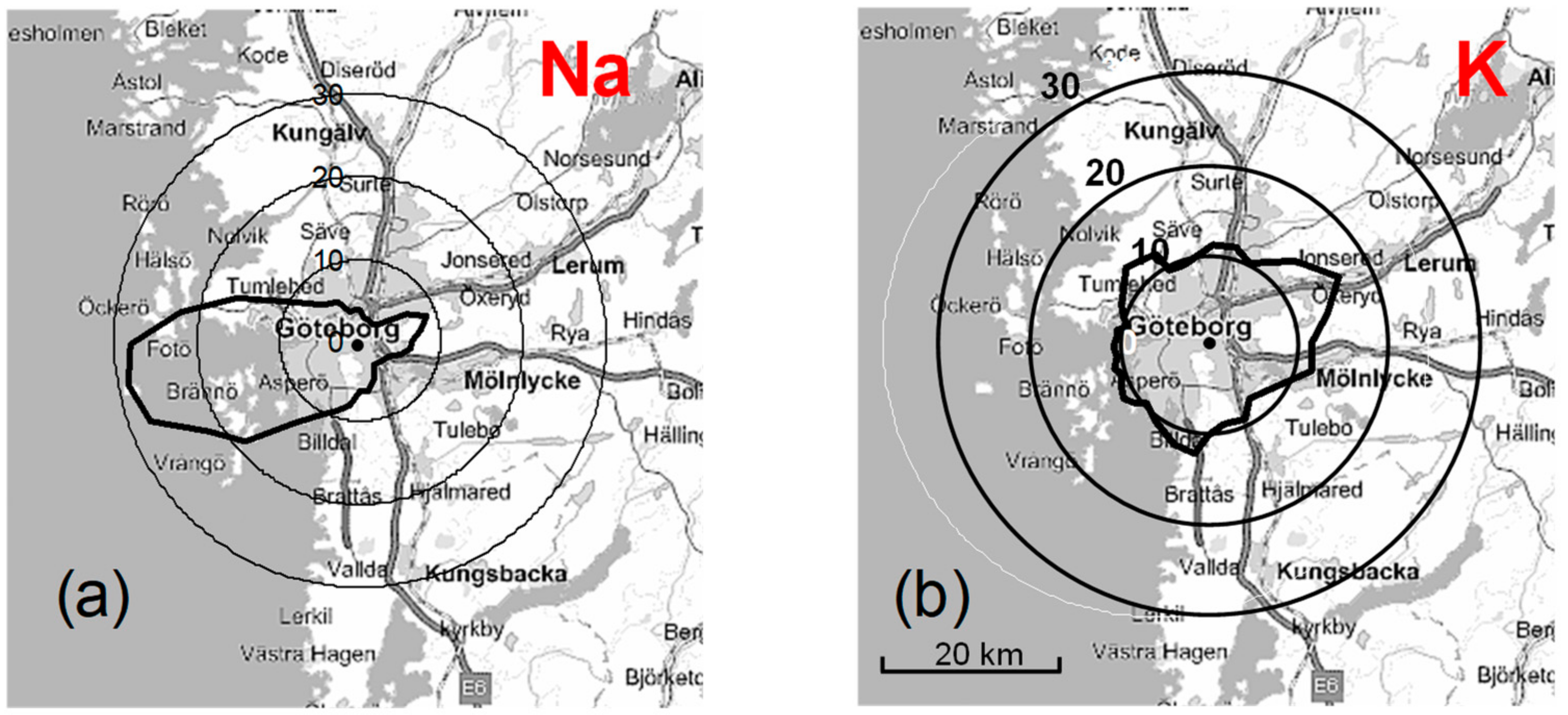
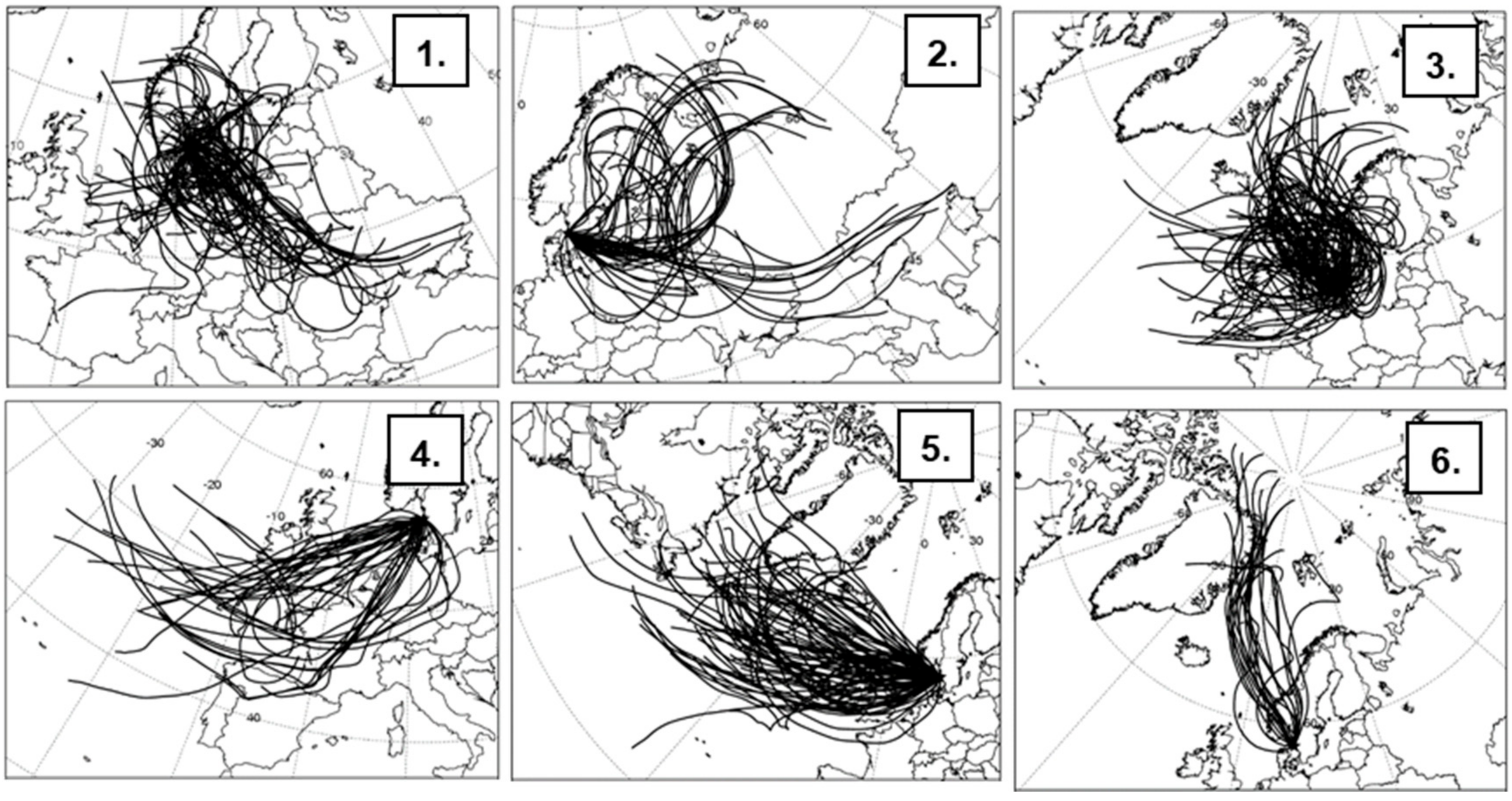
3.3. Influence of Air Mass History
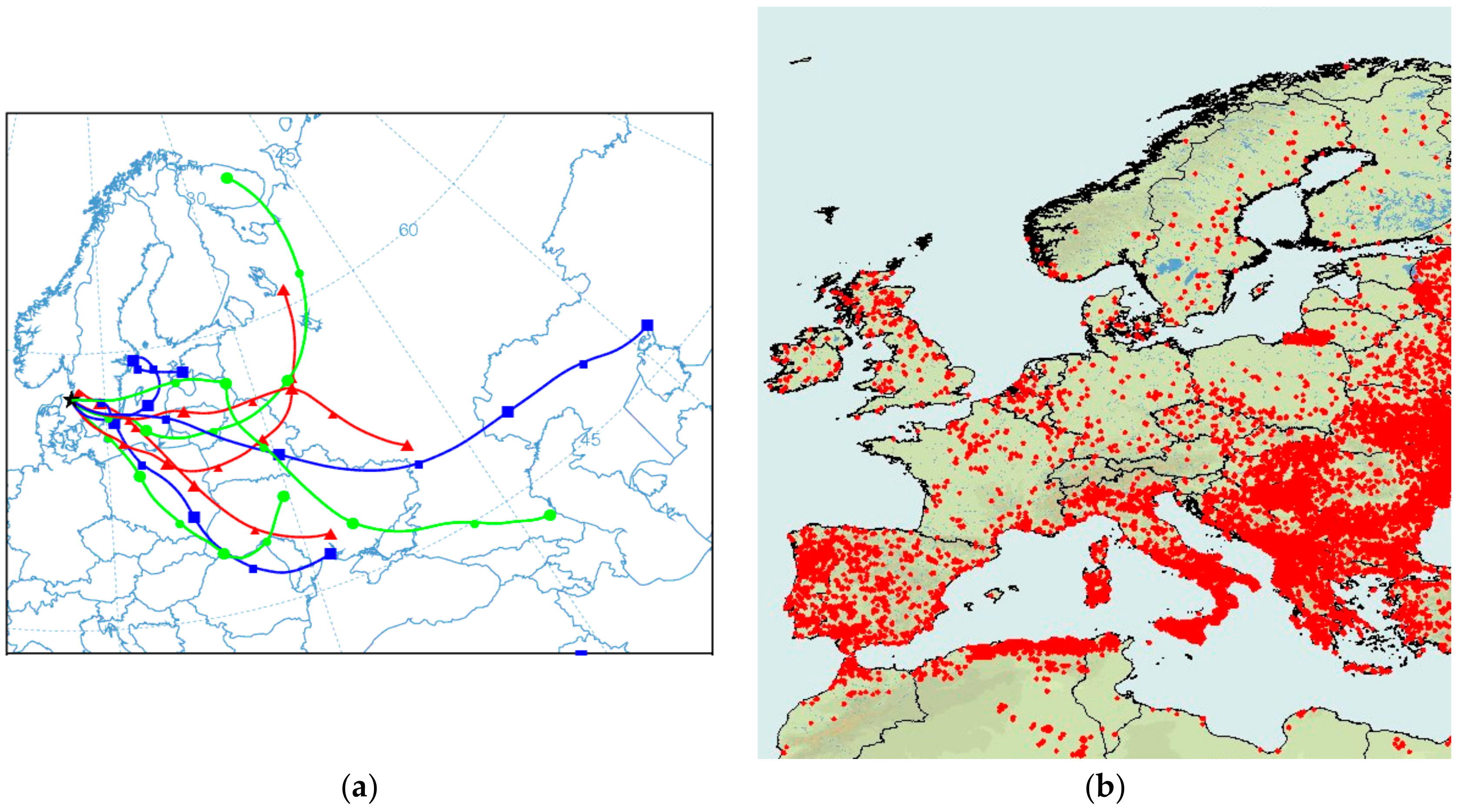
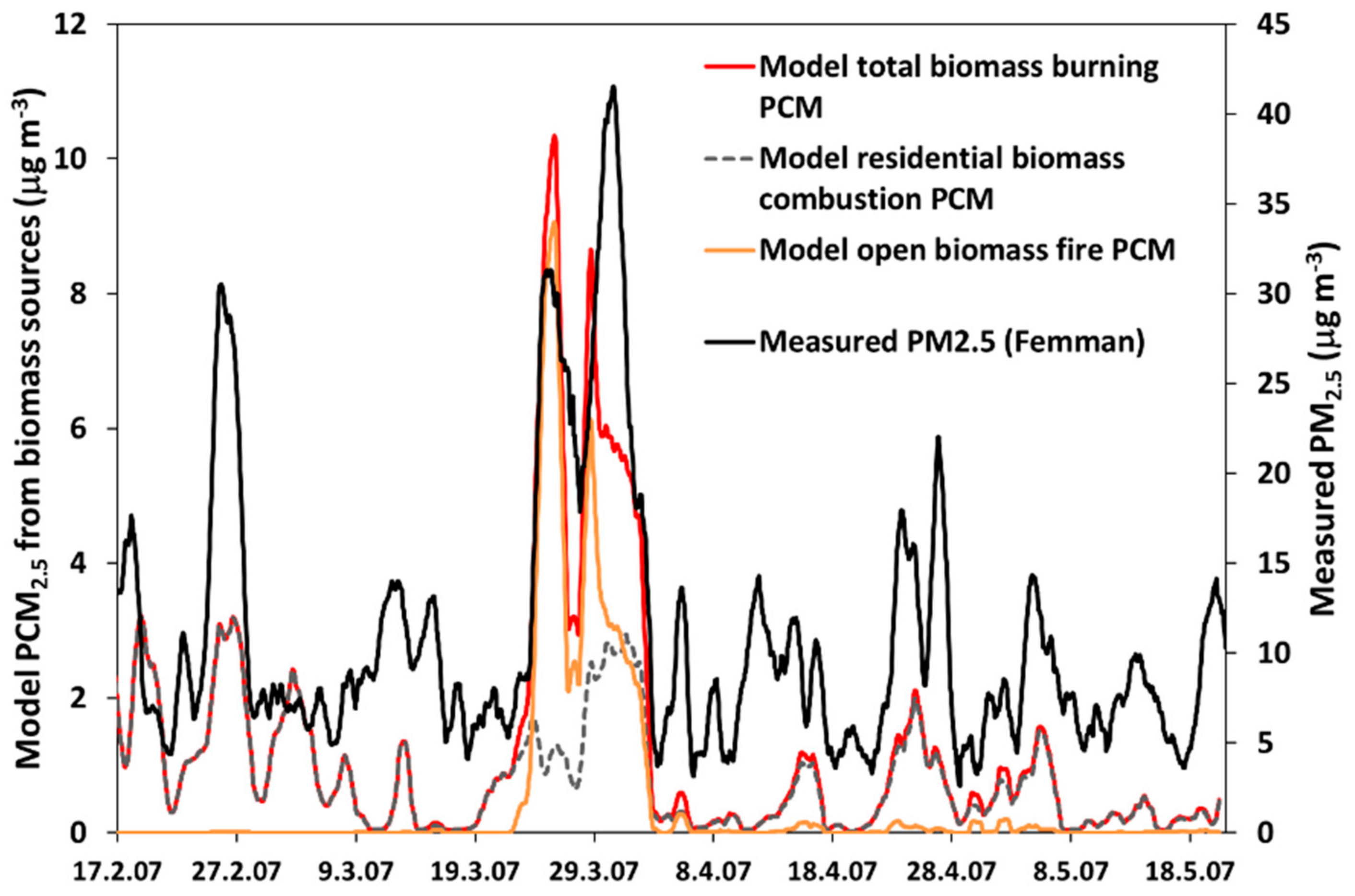
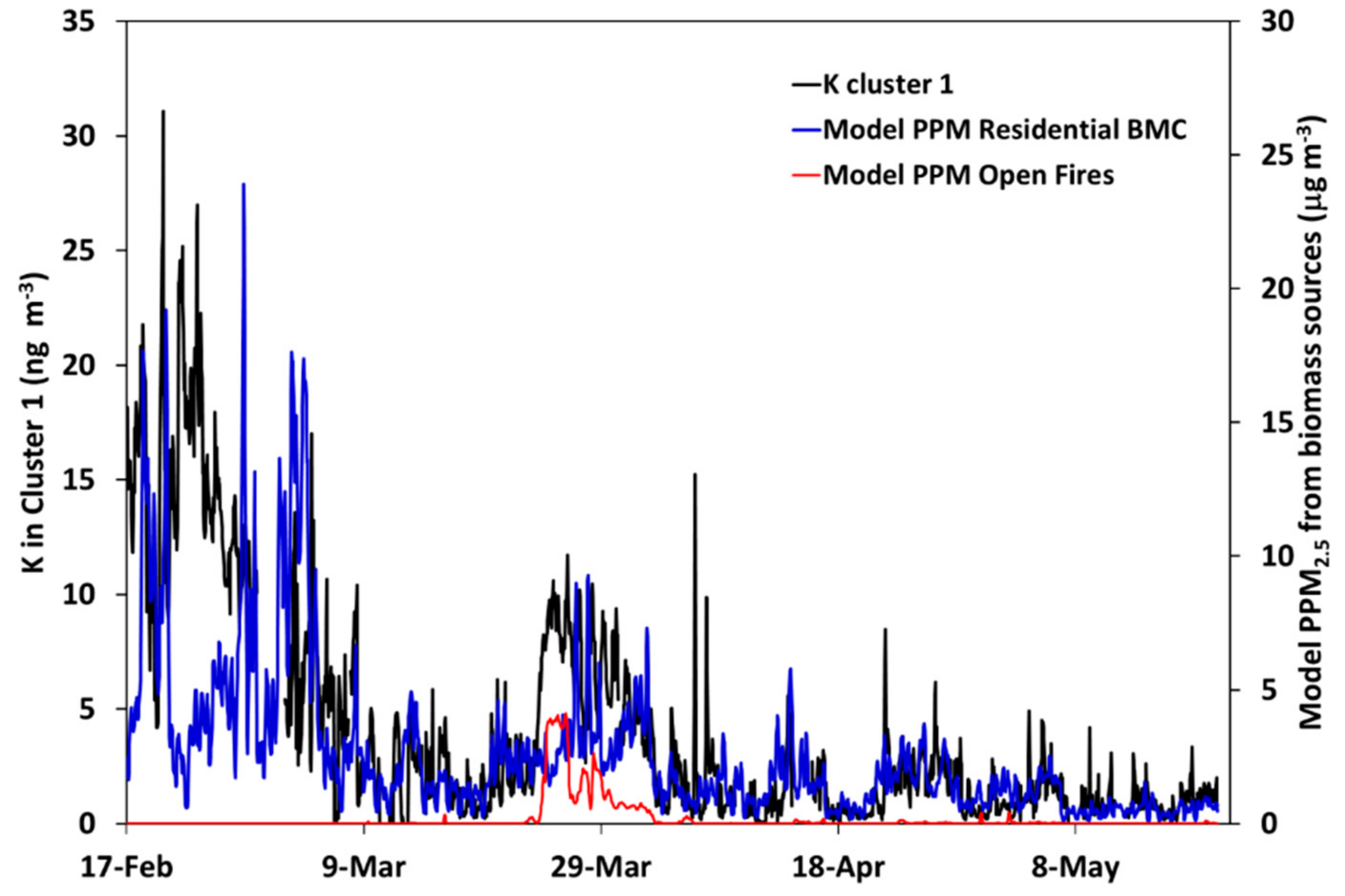
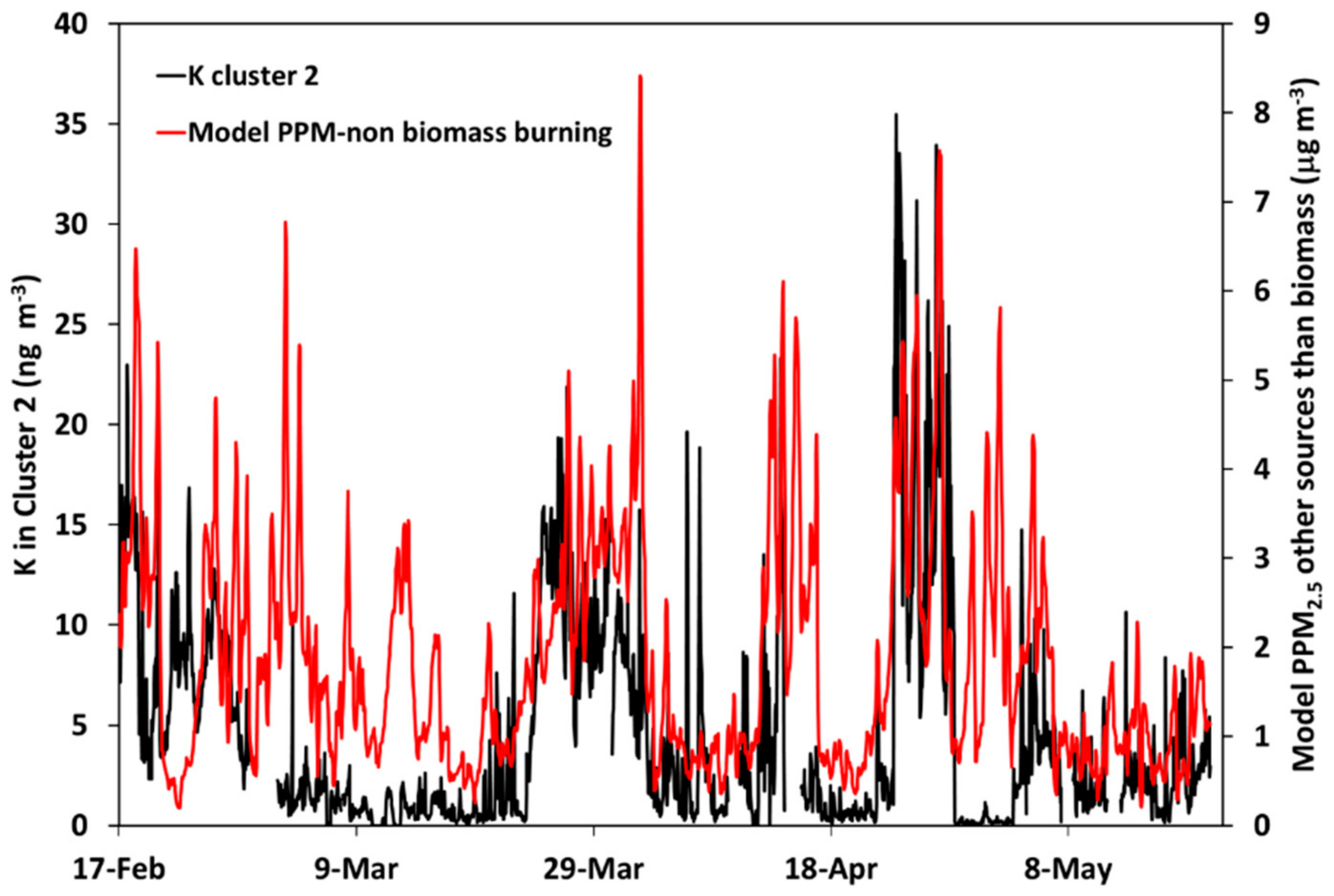
3.4. Comparison between Alkali-AMS and Chemical Transport Modeling Results
3.5. Comparison with Earlier Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, J.; Li, C.; Ristovski, Z.; Milic, A.; Gu, Y.; Islam, M.S.; Wang, S.; Hao, J.; Zhang, H.; He, C.; et al. A review of biomass burning: Emissions and impacts on air quality, health and climate in China. Sci. Total Environ. 2017, 579, 1000–1034. [Google Scholar] [CrossRef] [PubMed]
- Kelly, F.J.; Fussell, J.C. Size, source and chemical composition as determinants of toxicity attributable to ambient particulate matter. Atmos. Environ. 2012, 60, 504–526. [Google Scholar] [CrossRef]
- Lee, A.; Kinney, P.; Chillrud, S.; Jack, D. A Systematic Review of Innate Immunomodulatory Effects of Household Air Pollution Secondary to the Burning of Biomass Fuels. Ann. Glob. Health 2015, 81, 368–374. [Google Scholar] [CrossRef]
- Deering-Rice, C.E.; Nguyen, N.; Lu, Z.; Cox, J.E.; Shapiro, D.; Romero, E.G.; Mitchell, V.K.; Burrell, K.L.; Veranth, J.M.; Reilly, C.A. Activation of TRPV3 by Wood Smoke Particles and Roles in Pneumotoxicity. Chem. Res. Toxicol. 2018, 31, 291–301. [Google Scholar] [CrossRef]
- Scott, A.F.; Reilly, C.A. Wood and Biomass Smoke: Addressing Human Health Risks and Exposures. Chem. Res. Toxicol. 2019, 32, 219–221. [Google Scholar] [CrossRef]
- Viana, M.; Kuhlbusch, T.A.J.; Querola, X.; Alastueya, A.; Harrison, R.M.; Hopke, P.K.; Winiwarter, W.; Vallius, M.; Szidat, S.; Prévôt, A.S.H.; et al. Source apportionment of particulate matter in Europe: A review of methods and results. Aerosol Sci. 2008, 39, 827–849. [Google Scholar] [CrossRef]
- Andersen, Z.J.; Wåhlin, P.; Raaschou-Nielsen, O.; Scheike, T.; Loft, S. Ambient particle source apportionment and daily hospital admissions among children and elderly in Copenhagen. J. Expo. Sci. Environ. Epidemiol. 2007, 17, 625–636. [Google Scholar] [CrossRef]
- Denier van der Gon, H.A.C.; Bergström, R.; Fountoukis, C.; Johansson, C.; Pandis, S.N.; Simpson, D.; Visschedijk, A.J.H. Particulate emissions from residential wood combustion in Europe–revised estimates and an evaluation. Atmos. Chem. Physics 2015, 15, 6503–6519. [Google Scholar] [CrossRef]
- Glasius, M.; Hansen, A.; Claeys, M.; Henzing, J.; Jedynska, A.; Kasper-Giebl, A.; Kistler, M.; Kristensen, K.; Martinsson, J.; Maenhaut, W. Composition and sources of carbonaceous aerosols in Northern Europe during winter. Atmos. Environ. 2018, 173, 127–141. [Google Scholar] [CrossRef]
- Szidat, S.; Jenk, T.M.; Synal, H.-A.; Kalberer, M.; Wacker, L.; Hajdas, I.; Kasper-Giebl, A.; Baltensperger, U. Contributions of fossil fuel, biomass burning, and biogenic emissions to carbonaceous aerosols in Zürich as traced by 14C. J. Geophys. Res. 2006, 111, D07206. [Google Scholar] [CrossRef]
- Szidat, S.; Prévôt, A.S.H.; Sandradewi, J.; Alfarra, M.R.; Synal, H-A.; Wacker, L.; Baltensperger, U. Dominant impact of residential wood burning on particulate matter in Alpine valleys during winter. Geophys. Res. Lett. 2007, 34, L05820. [Google Scholar] [CrossRef]
- Szidat, S.; Ruff, M.; Wacker, L.; Synal, H.A.; Hallquist, M.; Shannigrahi, A.S.; Yttri, K.E.; Dye, C.; Simpson, D. Fossil and non-fossil sources of organic carbon (OC) and elemental carbon (EC) in Göteborg, Sweden. Atmos. Chem. Phys. 2009, 9, 1521–1535. [Google Scholar] [CrossRef]
- Simoneit, B.R.T. Biomass Burning—A Review of Organic Tracers for Smoke from Incomplete Combustion. Appl. Geochem. 2002, 17, 129–162. [Google Scholar] [CrossRef]
- Elsasser, M.; Crippa, M.; Orasche, J.; DeCarlo, P.F.; Oster, M.; Pitz, M.; Cyrys, J.; Gustafson, T.L.; Pettersson, J.B.C.; Schnelle-Kreis, J.; et al. Organic molecular markers and signature from wood combustion particles in winter ambient aerosols: aerosol mass spectrometer (AMS) and high time-resolved GC-MS measurements in Augsburg, Germany. Atmos. Chem. Phys. 2012, 12, 6113–6128. [Google Scholar] [CrossRef]
- Tao, J.; Zhang, L.M.; Zhang, R.J.; Wu, Y.F.; Zhang, Z.S.; Zhang, X.L.; Tang, Y.X.; Cao, J.J.; Zhang, Y.H. Uncertainty Assessment of Source Attribution of PM2.5 and its Water-soluble Organic Carbon Content using Different Biomass Burning Tracers in Positive Matrix Factorization Analysis–A Case Study in Beijing, China. Sci. Total Environ. 2016, 543, 326–335. [Google Scholar] [CrossRef]
- Fourtziou, L.; Liakakou, E.; Stavroulas, I.; Theodosi, C.; Zarmpas, P.; Psiloglou, B.; Sciare, J.; Maggos, T.; Bairachtari, K.; Bougiatioti, A. Multi-tracer Approach to Characterize Domestic Wood Burning in Athens (Greece) during Wintertime. Atmos. Environ. 2017, 148, 89–101. [Google Scholar] [CrossRef]
- Nielsen, I.E.; Eriksson, A.C.; Lindgren, R.; Martinsson, J.; Nystrom, R.; Nordin, E.Z.; Sadiktsis, I.; Boman, C.; Nojgaard, J.K.; Pagels, J. Time-resolved Analysis of Particle Emissions from Residential Biomass Combustion Emissions of Refractory Black Carbon, PAHs and Organic Tracers. Atmos. Environ. 2017, 165, 179–190. [Google Scholar] [CrossRef]
- Achad, M.; Caumo, S.; Vasconcellos, P.D.; Bajano, H.; Gomez, D.; Smichowski, P. Chemical Markers of Biomass Burning: Determination of Levoglucosan, and Potassium in Size-classified Atmospheric Aerosols Collected in Buenos Aires, Argentina by Different Analytical Techniques. Microchem. J. 2018, 139, 181–187. [Google Scholar] [CrossRef]
- Rad, F.M.; Spinicci, S.; Silvergren, S.; Nilsson, U.; Westerholm, R. Validation of a HILIC/ESI-MS/MS Method for the Wood Burning Marker Levoglucosan and its Isomers in Airborne Particulate Matter. Chemosphere 2018, 211, 617–623. [Google Scholar]
- Elsasser, M.; Busch, C.; Orasche, J.; Schön, C.; Hartmann, H.; Schnelle-Kreis, J.; Zimmermann, R. Dynamic Changes of the Aerosol Composition and Concentration during Different Burning Phases of Wood Combustion. Energy Fuels 2013, 27, 4959–4968. [Google Scholar] [CrossRef]
- Pagels, J.; Dutcher, D.D.; Stolzenburg, M.R.; McMurry, P.H.; Galli, M.E.; Gross, D.S. Fine-particle emissions from solid biofuel combustion studied with single-particle mass spectrometry: Identification of markers for organics, soot, and ash components. J. Geophys. Res. Atmos. 2013, 118, 859–870. [Google Scholar] [CrossRef]
- Echalar, F.; Gaudichet, A.; Cachier, H.; Artaxo, P. Aerosol Emissions by Tropical Forest and Savanna Biomass Burning: Characteristic Trace-Elements and Fluxes. Geophys. Res. Lett. 1995, 22, 3039–3042. [Google Scholar] [CrossRef]
- Lee, T.; Sullivan, A.P.; Mack, L.; Jimenez, J.L.; Kreidenweis, S.M.; Onasch, T.B.; Worsnop, D.R.; Malm, W.; Wold, C.E.; Hao, W.M. Chemical Smoke Marker Emissions During Flaming and Smoldering Phases of Laboratory Open Burning of Wildland Fuels. Aerosol Sci. Tech. 2010, 44, I–V. [Google Scholar] [CrossRef]
- Suess, D.T.; Prather, K.A. Mass Spectrometry of Aerosols. Chem. Rev. 1999, 99, 3007. [Google Scholar] [CrossRef]
- Johnston, M.V. Sampling and Analysis of Individual Particles by Aerosol Mass Spectrometry. J. Mass. Spectrom. 2000, 35, 585–595. [Google Scholar] [CrossRef]
- Noble, C.A.; Prather, K.A. Real-time single particle mass spectrometry: A historical review of a quarter century of the chemical analysis of aerosols. Mass Spectrom. Rev. 2000, 19, 248–274. [Google Scholar] [CrossRef]
- Coe, H.; Allan, J.D.; Alfarra, M.R.; Bower, K.N.; Flynn, M.J.; McFiggans, G.B.; Topping, D.O.; Williams, P.I.; O’Dowd, C.D.; Dall’Osto, M. Chemical and physical characteristics of aerosol particles at a remote coastal location, Mace Head, Ireland, during NAMBLEX. Atmos. Chem. Phys. 2006, 6, 3289–3301. [Google Scholar] [CrossRef]
- Canagaratna, M.R.; Jayne, J.T.; Jimenez, J.L.; Allan, J.D.; Alfarra, M.R.; Zhang, Q.; Onasch, T.B.; Drewnick, F.; Coe, H.; Middlebrook, A. Chemical and microphysical characterization of ambient aerosols with the aerodyne aerosol mass spectrometer. Mass Spectrom. Rev. 2007, 26, 185–222. [Google Scholar] [CrossRef]
- Svane, M.; Hagström, M.; Pettersson, J.B.C. Chemical Analysis of Individual Alkali-Containing Aerosol Particles: Design and Performance of a Surface Ionization Particle Beam Mass Spectrometer. Aerosol Sci. Technol. 2004, 38, 655–663. [Google Scholar] [CrossRef]
- Svane, M.; Gustafsson, T.L.; Kovacevik, B.; Noda, J.; Andersson, P.U.; Nilsson, E.D.; Pettersson, J.B.C. On-line chemical analysis of individual alkali-containing aerosol particles by surface ionization combined with time-of-flight mass spectrometry. Aerosol Sci. Technol. 2009, 43, 453–661. [Google Scholar] [CrossRef]
- Svane, M.; Janhäll, S.; Hagström, M.; Hallquist, M.; Pettersson, J.B.C. On-line Alkali Analysis of Individual Aerosol Particles in Urban Air. Atmos. Environ. 2005, 39, 6919–6930. [Google Scholar] [CrossRef]
- Svane, M.; Hagström, M.; Pettersson, J.B.C. Online Measurements of Individual Alkali-Containing Particles Formed in Biomass and Coal Combustion: Demonstration of an Instrument Based on Surface Ionization Technique. Energy Fuels. 2005, 19, 411–417. [Google Scholar] [CrossRef]
- Svane, M.; Hagström, M.; Davidsson, K.O.; Boman, J.; Pettersson, J.B.C. Cesium as a Tracer for Alkali Processes in a Large-scale Combustion Facility. Energy Fuels 2006, 20, 979–985. [Google Scholar] [CrossRef]
- Hu, Q.H.; Xie, Z.Q.; Wang, X.M.; Kang, H.; Zhang, P. Levoglucosan indicates high levels of biomass burning aerosols over oceans from the Arctic to Antarctic. Sci. Rep. 2013, 3, 3119. [Google Scholar] [CrossRef] [PubMed]
- Pratap, V.; Chen, Y.; Yao, G.; Nakao, S. Temperature effects on multiphase reactions of organic molecular markers: A modeling study. Atmos. Environ. 2018, 179, 40–48. [Google Scholar] [CrossRef]
- Simpson, D.; Benedictow, A.; Berge, H.; Bergström, R.; Emberson, L.D.; Fagerli, H.; Flechard, C.R.; Hayman, G.D.; Gauss, M.; Jonson, J.E. The EMEP MSC-W chemical transport model–Technical description. Atmos. Chem. Phys. 2012, 12, 7825–7865. [Google Scholar] [CrossRef]
- Liu, P.; Ziemann, P.J.; Kittelson, D.B.; McMurry, P.H. Generating Particle Beams of Controlled Dimensions and Divergence: I. Theory of Particle Motion in Aerodynamic Lenses and Nozzle Expansions. Aerosol Sci. Technol. 1995, 22, 292–313. [Google Scholar] [CrossRef]
- Liu, P.; Ziemann, P.J.; Kittelson, D.B.; McMurry, P.H. Generating Particle Beams of Controlled Dimensions and Divergence: Ii. Experimental Evaluation of Particle Motion in Aerodynamic Lenses and Nozzle Expansions. Aerosol Sci. Technol. 1995, 22, 314–324. [Google Scholar] [CrossRef]
- Jayne, J.T.; Leard, D.C.; Zhang, X.; Davidovits, P.; Smith, K.A.; Kolb, C.A.; Worsnop, D.R. Development of an Aerosol Mass Spectrometer for Size and Composition Analysis of Submicron Particles. Aerosol Sci. Technol. 2000, 33, 49–70. [Google Scholar] [CrossRef]
- Jimenez, J.L.; Jayne, J.T.; Shi, Q.; Kolb, C.A.; Worsnop, D.R.; Yourshaw, I.; Seinfeld, J.H.; Flagan, R.C.; Zhang, X.; Smith, K.A. Ambient Aerosol Sampling Using the Aerodyne Aerosol Mass Spectrometer. J. Geophys. Res. 2003, 108, 8425. [Google Scholar] [CrossRef]
- Ionov, N.I. Progress in Surface Science. S G Davison, Pergamon Press: Oxford, UK, 1972; Volume 1. [Google Scholar]
- Zandberg, E.Y. Surface-Ionization Detection of Particles. Tech. Phys. 1995, 65, 1–38. [Google Scholar]
- The Environmental Management office of the City of Gothenburg (miljoforvaltningen@miljo.goteborg.se). Air quality monitoring data from the Femman site in Gothenburg (and most other Swedish air quality monitoring stations). Available online: https://shair.smhi.se/portal/concentrations-in-air (accessed on 1 December 2019).
- Ester, M.; Kriegel, H.; Sander, J.; Xu, X. A density-based algorithm for discovering clusters in large spatial databases with noise. In Proceedings of the Second International Conference on Knowledge Discovery and Data Mining, Portland, OR, USA, 2–4 August 1996; pp. 226–231. [Google Scholar]
- Daszykowski, M.; Walczak, B.; Massart, D.L. Looking for natural patterns in data. Part 1. Density-based approach. Chemom. Intell. Lab. Syst. 2001, 56, 83–92. [Google Scholar] [CrossRef]
- Draxler, R.R.; Hess, G.D. Description of the HYSPLIT4 Modeling System, NOAA Technical Memorandum ERL ARL-224; NOAA: Silver Spring, MD, USA, 2004. [Google Scholar]
- Bergström, R.; Denier van der Gon, H.A.C.; Prévôt, A.S.H.; Yttri, K.E.; Simpson, D. Modelling of organic aerosols over Europe (2002–2007) using a volatility basis set (VBS) framework: Application of different assumptions regarding the formation of secondary organic aerosol. Atmos. Chem. Phys. 2012, 12, 8499–8527. [Google Scholar] [CrossRef]
- Simpson, D.; Bergström, R.; Tsyro, S.; Wind, P. Updates to the EMEP/MSC-W Model, 2018–2019, Transboundary Particulate Matter, Photo-Oxidants, Acidifying and Eutrophying Components. EMEP Status Report 1/2019; The Norwegian Meteorological Institute: Oslo, Norway, 2018; pp. 145–155. Available online: www.emep.int (accessed on 2 December 2019).
- Aas, W.; Tsyro, S.; Bieber, E.; Bergström, R.; Ceburnis, D.; Ellermann, T.; Fagerli, H.; Frölich, M.; Gehrig, R.; Makkonen, U. Lessons learnt from the first EMEP intensive measurement periods. Atmos. Chem. Phys. 2012, 12, 8073–8094. [Google Scholar] [CrossRef]
- Genberg, J.; Denier van der Gon, H.A.C.; Simpson, D.; Swietlicki, E.; Areskoug, H.; Beddows, D.; Ceburnis, D.; Fiebig, M.; Hansson, H.C.; Harrison, R.M.; et al. Light-absorbing carbon in Europe–measurement and modelling, with a focus on residential wood combustion emissions. Atmos. Chem. Phys. 2013, 13, 8719–8738. [Google Scholar] [CrossRef]
- Jonson, J.E.; Simpson, D.; Fagerli, H.; Solberg, S. Can we explain the trends in European ozone levels? Atmos. Chem. Phys. 2006, 6, 51–66. [Google Scholar] [CrossRef]
- Yttri, K.E.; Simpson, D.; Bergström, R.; Kiss, G.; Szidat, S.; Ceburnis, D.; Eckhardt, S.; Hueglin, C.; Nøjgaard, J.K.; Perrino, C. The EMEP Intensive Measurement Period campaign, 2008–2009: Characterizing carbonaceous aerosol at nine rural sites in Europe. Atmos. Chem. Phys. 2019, 19, 4211–4233. [Google Scholar] [CrossRef]
- Bergström, R.; Hallquist, M.; Simpson, D.; Wildt, J.; Mentel, T.F. Biotic stress: a significant contributor to organic aerosol in Europe? Atmos. Chem. Phys. 2014, 14, 13643–13660. [Google Scholar] [CrossRef]
- Mareckova, K.; Wankmueller, R.; Anderl, M.; Poupa, S.; Wieser, M. Inventory Review 2009. Emission Data Reported Under the LRTAP Convention and NEC Directive. Stage 1 and 2 Review. Status of Gridded Data and LPS Data; EMEP/CEIP Technical Report 1/2009; Umweltbundesamt GmbH: Vienna, Austria, 2009. [Google Scholar]
- Wiedinmyer, C.; Akagi, S.K.; Yokelson, R.J.; Emmons, L.K.; Al-Saadi, J.A.; Orlando, J.J.; Soja, A.J. The Fire Inventory from NCAR (FINN): A high resolution global model to estimate the emissions from open burning. Geosci. Model Dev. 2011, 4, 625–641. [Google Scholar] [CrossRef]
- Janhäll, S.; Andersson, P.U.; Olofson, K.F.G.; Pettersson, J.B.C.; Hallquist, M. Evolution of the Urban Aerosol during Winter Temperature Inversion Episodes. Atmos. Environ. 2006, 28, 5355–5366. [Google Scholar] [CrossRef]
- Olofson, K.F.G.; Andersson, P.U.; Hallquist, M.; Ljungström, E.; Tang, L.; Chen, D.; Pettersson, J.B.C. Urban aerosol evolution and particle formation during wintertime temperature inversions. Atmos. Environ. 2008, 43, 340–346. [Google Scholar] [CrossRef]
- Roy, D.P.; Borak, J.S.; Devadiga, S.; Wolfe, R.E.; Zheng, M.; Descloitres, J. The MODIS Land product quality assessment approach. Remote Sens. Environ. 2002, 83, 62–76. [Google Scholar] [CrossRef]
- Ooki, A.; Uematsu, M.; Miura, K.; Nakae, S. Sources of sodium in atmospheric fine particles. Atmos. Environ. 2002, 36, 4367–4374. [Google Scholar] [CrossRef]
- Mamane, Y. Estimate of Municipal Refuse Incinerator Contribution to Philadelphia Aerosol. 1. Source Analysis. Atmos. Environ. 1988, 22, 2411–2418. [Google Scholar] [CrossRef]
- Stohl, A.; Berg, T.; Burkhart, J.F.; Fjaeraa, A.M.; Forster, C.; Herber, A.; Hov, O.; Lunder, C.; McMillan, W.W.; Oltmans, S. Arctic smoke—Record high air pollution levels in the European Arctic due to agricultural fires in Eastern Europe in spring 2006. Atmos. Chem. Phys. 2007, 7, 511–534. [Google Scholar] [CrossRef]
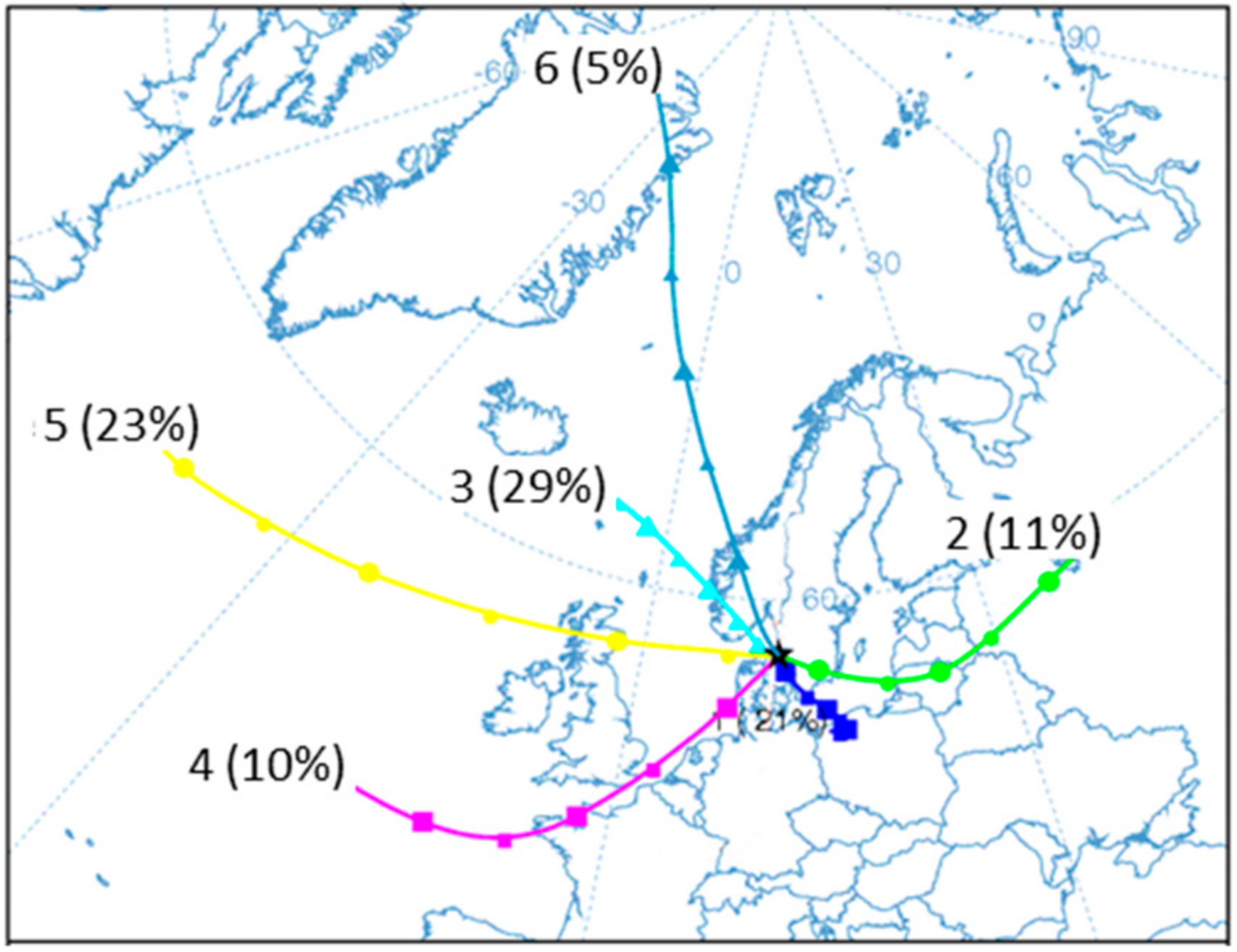
| Origin Group (Initial) | Samples | % | Na | K | Na/K | PM10 | O3 | NO | NOx | T | WS |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. Central Eur. (C) | 3238 | 19 | 3.9 | 14 | 0.27 | 34.8 (± 4.3) | 46.2 | 11.5 | 51.4 | 7.0 | 3.5 |
| 2. Eastern Eur. (E) | 2171 | 13 | 6.2 | 21 | 0.29 | 34.1 (± 5.1) | 53.8 | 16.6 | 57.6 | 2.9 | 4.7 |
| 3. Northern Eur.(N) | 5027 | 30 | 6.0 | 5.6 | 1.1 | 20.1 (± 2.0) | 57.6 | 7.8 | 36.0 | 8.3 | 3.5 |
| 4. Western Eur. (W) | 1393 | 8 | 4.9 | 11 | 0.45 | 25.3 (± 4.7) | 49.3 | 4.6 | 32.0 | 8.0 | 3.6 |
| 5. Atlantic (A) | 4188 | 25 | 33 | 3.6 | 9.2 | 27.2 (± 2.9) | 66.6 | 2.7 | 23.3 | 7.2 | 5.4 |
| 6. Polar (P) | 847 | 5 | 8.6 | 2.7 | 3.2 | 20.1 (± 4.8) | 57.1 | 9.1 | 36.9 | 4.9 | 4.7 |
| Correlation Coefficient (r) | ||||
|---|---|---|---|---|
| Model Tracer Compound/Source Category | Cluster 1 Number | Cluster 1 K | Cluster 2 Number | Cluster 2 K |
| Total biomass burning (residential + fires) | 0.76 | 0.64 | 0.24 | 0.31 |
| Residential biomass combustion (SNAP-2) | 0.71 | 0.62 | 0.15 | 0.23 |
| Open biomass burning (fires) | 0.38 | 0.21 | 0.44 | 0.41 |
| Total other anthropogenic PPM sources a | 0.41 | 0.26 | 0.63 | 0.61 |
| Non-industrial/residential combustion fossil fuels (coal, oil, gas) (SNAP-2) | 0.51 | 0.54 | 0.27 | 0.37 |
| Manufacturing industry (SNAP-3) | 0.53 | 0.30 | 0.61 | 0.58 |
| Road transport (SNAP-7) | 0.39 | 0.19 | 0.57 | 0.52 |
| International shipping | -0.04 | -0.08 | 0.33 | 0.30 |
| Other mobile sources/machinery | 0.28 | 0.12 | 0.53 | 0.50 |
| Agriculture (SNAP-10) | 0.33 | 0.12 | 0.65 | 0.59 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noda, J.; Bergström, R.; Kong, X.; Gustafsson, T.L.; Kovacevik, B.; Svane, M.; Pettersson, J.B.C. Aerosol from Biomass Combustion in Northern Europe: Influence of Meteorological Conditions and Air Mass History. Atmosphere 2019, 10, 789. https://doi.org/10.3390/atmos10120789
Noda J, Bergström R, Kong X, Gustafsson TL, Kovacevik B, Svane M, Pettersson JBC. Aerosol from Biomass Combustion in Northern Europe: Influence of Meteorological Conditions and Air Mass History. Atmosphere. 2019; 10(12):789. https://doi.org/10.3390/atmos10120789
Chicago/Turabian StyleNoda, Jun, Robert Bergström, Xiangrui Kong, Torbjörn L. Gustafsson, Borka Kovacevik, Maria Svane, and Jan B. C. Pettersson. 2019. "Aerosol from Biomass Combustion in Northern Europe: Influence of Meteorological Conditions and Air Mass History" Atmosphere 10, no. 12: 789. https://doi.org/10.3390/atmos10120789
APA StyleNoda, J., Bergström, R., Kong, X., Gustafsson, T. L., Kovacevik, B., Svane, M., & Pettersson, J. B. C. (2019). Aerosol from Biomass Combustion in Northern Europe: Influence of Meteorological Conditions and Air Mass History. Atmosphere, 10(12), 789. https://doi.org/10.3390/atmos10120789






