Genomic Insights into Date Palm Origins
Abstract
Highlights
- A large amount of genomic data has been generated for the date palm in the past few years.
- This review records these data and re-evaluates questions about date palm domestication origins and diffusion in light of genomic data
- Remaining questions are highlighted, and the future perspective of the field discussed.
1. Introduction
2. High-Throughput Sequencing of Date Palms
2.1. Nuclear Reference Genome
2.2. Organellar Genomes
2.3. Resequencing Projects
2.4. Transcriptomic Data and Analyses
2.5. Ancient DNA of Date Palm
3. The Date Palm and the Genus Phoenix
3.1. Number of Species in the Genus Phoenix
3.2. Phylogenetic Relationships in The Genus Phoenix
4. The Wild Ancestor of the Date Palm
4.1. Identifying Wild Phoenix dactylifera Population
4.2. Consequences of Domestication
5. The Geography of the Domestication
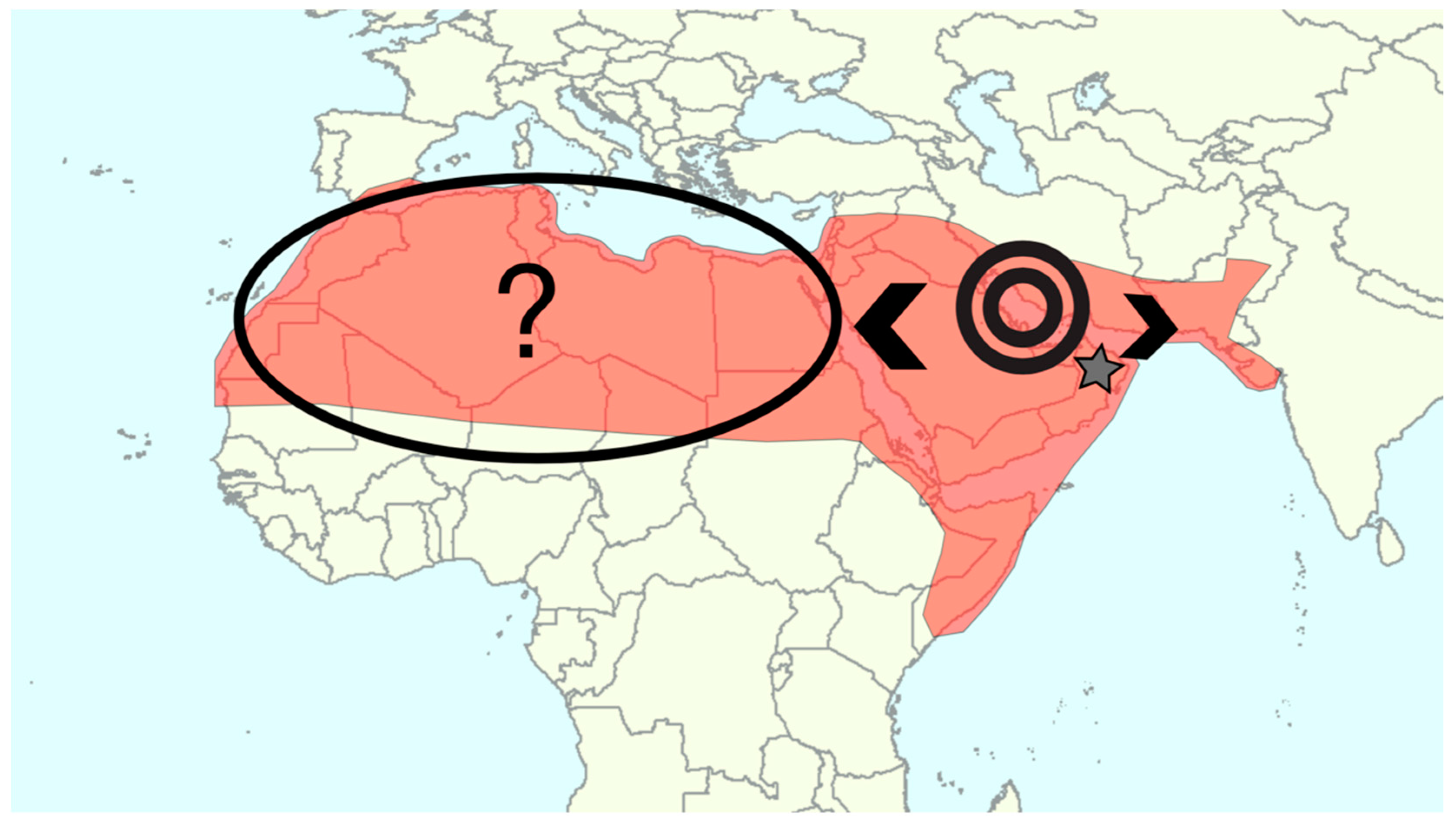
5.1. What is the Original Date Palm Distribution?
5.2. Domestication Center(s)
5.3. Diffusion and Interspecific Hybridization
6. Domestication and Diversification Genes
7. Conclusions and Prospects
Funding
Acknowledgments
Conflicts of Interest
References
- Cubry, P.; Tranchant-Dubreuil, C.; Thuillet, A.-C.; Monat, C.; Ndjiondjop, M.-N.; Labadie, K.; Cruaud, C.; Engelen, S.; Scarcelli, N.; Rhoné, B.; et al. The rise and fall of african rice cultivation revealed by analysis of 246 new genomes. Curr. Biol. 2018, 28, 2274–2282.e6. [Google Scholar] [CrossRef] [PubMed]
- Stitzer, M.C.; Ross-Ibarra, J. Maize domestication and gene interaction. New Phytol. 2018, 220, 395–408. [Google Scholar] [CrossRef] [PubMed]
- Swinnen, G.; Goossens, A.; Pauwels, L. Lessons from domestication: Targeting cis-regulatory elements for crop improvement. Trends Plant Sci. 2016, 21, 506–515. [Google Scholar] [CrossRef] [PubMed]
- Sang, T.; Ge, S. Understanding rice domestication and implications for cultivar improvement. Curr. Opin. Plant Biol. 2013, 16, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Barrow, S. A monograph of Phoenix L. (Palmae: Coryphoideae). Kew Bull. 1998, 53, 513–575. [Google Scholar] [CrossRef]
- Henderson, A. Phoenix. In Palms of Southern Asia; Princeton University Press: Princeton, NJ, USA, 2009; pp. 140–142. ISBN 0691134499. [Google Scholar]
- FAO FAOSTAT. Available online: http://faostat.fao.org/default.aspx (accessed on 1 September 2018).
- Tengberg, M. Beginnings and early history of date palm garden cultivation in the Middle East. J. Arid Environ. 2012, 86, 139–147. [Google Scholar] [CrossRef]
- Chao, C.T.; Krueger, R.R. The date palm (Phoenix dactylifera L.): Overview of biology, uses and cultivation. HortScience 2007, 42, 1077–1082. [Google Scholar]
- Food and Agricultural Organization of the United Nations. Date Palm Cultivation; FAO: Rome, Italy, 2002; ISBN 92-5-104863-0. [Google Scholar]
- Bodian, A.; El Houmaizi, M.A.; Ndoye Ndir, K. Genetic diversity analysis of date palm (Phoenix dactylifera L.) cultivars from Figuig oasis (Morocco) using SSR markers. Int. J. Sci. Adv. Technol. 2012, 2, 96–104. [Google Scholar]
- Battesti, V.; Gros-Balthazard, M.; Ogéron, C.; Ivorra, S.; Terral, J.F.; Newton, C. Date palm agrobiodiversity (Phoenix dactylifera L.) in Siwa Oasis, Egypt: Combining ethnography, morphometry, and genetics. Hum. Ecol. 2018, 46, 529–546. [Google Scholar] [CrossRef]
- Pintaud, J.-C.; Ludena, B.; Zehdi, S.; Gros-Balthazard, M.; Ivorra, S.; Terral, J.-F.; Newton, C.; Tengberg, M.; Santoni, S.; Boughedoura, N. Biogeography of the date palm (Phoenix dactylifera L., Arecaceae): Insights on the origin and on the structure of modern diversity. ISHS Acta Hortic. 2013, 994. [Google Scholar] [CrossRef]
- Shendure, J.; Balasubramanian, S.; Church, G.M.; Gilbert, W.; Rogers, J.; Schloss, J.A.; Waterston, R.H. DNA sequencing at 40: Past, present and future. Nature 2017, 550, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Bleidorn, C. Third generation sequencing: technology and its potential impact on evolutionary biodiversity research. Syst. Biodivers. 2016, 14, 1–8. [Google Scholar] [CrossRef]
- Goff, S.A.; Ricke, D.; Lan, T.-H.; Presting, G.; Wang, R.; Dunn, M.; Glazebrook, J.; Sessions, A.; Oeller, P.; Varma, H.; et al. A draft sequence of the rice genome (Oryza sativa L. ssp. japonica). Science 2002, 296, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Hu, S.; Wang, J.; Wong, G.K.-S.; Li, S.; Liu, B.; Deng, Y.; Dai, L.; Zhou, Y.; Zhang, X.; et al. A draft sequence of the rice genome (Oryza sativa L. ssp. indica). Science 2002, 296, 79–92. [Google Scholar] [CrossRef] [PubMed]
- International Wheat Genome Sequencing Consortium (IWGSC); IWGSC RefSeq principal investigators; Appels, R.; Eversole, K.; Feuillet, C.; Keller, B.; Rogers, J.; Stein, N.; IWGSC whole-genome assembly principal investigators; Pozniak, C.J.; et al. Shifting the limits in wheat research and breeding using a fully annotated reference genome. Science 2018, 361. [Google Scholar] [CrossRef]
- Al-Dous, E.K.; George, B.; Al-Mahmoud, M.E.; Al-Jaber, M.Y.; Wang, H.; Salameh, Y.M.; Al-Azwani, E.K.; Chaluvadi, S.; Pontaroli, A.C.; DeBarry, J.; et al. De novo genome sequencing and comparative genomics of date palm (Phoenix dactylifera). Nat. Biotechnol. 2011, 29, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Al-Mssallem, I.S.; Hu, S.; Zhang, X.; Lin, Q.; Liu, W.; Tan, J.; Yu, X.; Liu, J.; Pan, L.; Zhang, T.; et al. Genome sequence of the date palm Phoenix dactylifera L. Nat. Commun. 2013, 4, 2274. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Zhang, X.W.; Liu, G.M.; Yin, Y.X.; Chen, K.F.; Yun, Q.Z.; Zhao, D.J.; Al-Mssallem, I.S.; Yu, J. The complete chloroplast genome sequence of date palm (Phoenix dactylifera L.). PLoS ONE 2010, 5, e12762. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Ishtiaq, A.K.; Berthold, H.; Azim, M.K. The chloroplast genome sequence of date palm (Phoenix dactylifera L. cv. ’Aseel’). Plant Mol. Biol. Rep. 2012, 30, 666–678. [Google Scholar] [CrossRef]
- Khan, A.L.; Asaf, S.; Lee, I.; Al-harrasi, A.; Al-rawahi, A. First chloroplast genomics study of Phoenix dactylifera (var. Naghal and Khanezi): A comparative analysis. PLoS ONE 2018, 31, e0200104. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Wu, H.; Zhang, T.; Yang, M.; Yin, Y.; Pan, L.; Yu, X.; Zhang, X.; Hu, S.; Al-Mssallem, I.S.; et al. A complete sequence and transcriptomic analyses of date palm (Phoenix dactylifera L.) mitochondrial genome. PLoS ONE 2012, 7, e37164. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Zhang, X.; Fang, Y.; Pan, L.; Sun, G.; Xin, C.; Ba Abdullah, M.M.; Yu, X.; Hu, S.; Al-Mssallem, I.S.; et al. High-throughput sequencing-based gene profiling on multi-staged fruit development of date palm (Phoenix dactylifera, L.). Plant Mol. Biol. 2012, 78, 617–626. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Xia, W.; Yang, Y.; Mason, A.S.; Lei, X.; Ma, Z. Characterization and evolution of conserved MicroRNA through duplication events in date palm (Phoenix dactylifera). PLoS ONE 2013, 8, e71435. [Google Scholar] [CrossRef] [PubMed]
- Mathew, L.S.; Seidel, M.A.; George, B.; Mathew, S.; Spannagl, M.; Haberer, G.; Torres, M.F.; Al-Dous, E.K.; Al-Azwani, E.K.; Diboun, I.; et al. A genome-wide survey of date palm cultivars supports two major subpopulations in Phoenix dactylifera. G3 (Bethesda) 2015, 5, 1429–1438. [Google Scholar] [CrossRef] [PubMed]
- Radwan, O.; Arro, J.; Keller, C.; Korban, S.S. RNA-seq transcriptome analysis in date palm suggests multi-dimensional responses to salinity stress. Trop. Plant Biol. 2015, 8, 74–86. [Google Scholar] [CrossRef]
- Yaish, M.W.; Patankar, H.V.; Assaha, D.V.M.; Zheng, Y.; Al-Yahyai, R.; Sunkar, R. Genome-wide expression profiling in leaves and roots of date palm (Phoenix dactylifera L.) exposed to salinity. BMC Genom. 2017, 18, 246. [Google Scholar] [CrossRef] [PubMed]
- Xin, C.; Liu, W.; Lin, Q.; Zhang, X.; Cui, P.; Li, F.; Zhang, G.; Pan, L.; Al-Amer, A.; Mei, H.; et al. Profiling microRNA expression during multi-staged date palm (Phoenix dactylifera L.) fruit development. Genomics 2015, 105, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Yaish, M.W.; Sunkar, R.; Zheng, Y.; Ji, B.; Al-Yahyai, R.; Farooq, S.A. A genome-wide identification of the miRNAome in response to salinity stress in date palm (Phoenix dactylifera L.). Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Hazzouri, K.M.; Flowers, J.M.; Visser, H.J.; Khierallah, H.S.M.; Rosas, U.; Pham, G.M.; Meyer, R.S.; Johansen, C.K.; Fresquez, Z.A.; Masmoudi, K.; et al. Whole genome resequencing of date palms yields insights into diversification of a fruit tree crop. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Gros-Balthazard, M.; Galimberti, M.; Kousathanas, A.; Newton, C.; Ivorra, S.; Paradis, L.; Vigouroux, Y.; Carter, R.; Tengberg, M.; Battesti, V.; et al. The discovery of wild date palms in Oman reveals a complex domestication history involving centers in the Middle East and Africa. Curr. Biol. 2017, 27, 2211–2218. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.F.; Mathew, L.S.; Ahmed, I.; Al-Azwani, I.K.; Krueger, R.; Rivera-Nuñez, D.; Mohamoud, Y.A.; Clark, A.G.; Suhre, K.; Malek, J.A. Genus-wide sequencing supports a two-locus model for sex-determination in Phoenix. Nat. Commun. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Zhang, C.; Liu, W.; Lin, Q.; Wei, T.; Aljohi, H.A.; Chen, W.-H.; Hu, S. DRDB: An online date palm genomic resource database. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Beal, J.M. Cytological studies in the genus Phoenix. Bot. Gaz. 1937, 99, 400–407. [Google Scholar] [CrossRef]
- Chin, C.-S.; Peluso, P.; Sedlazeck, F.J.; Nattestad, M.; Concepcion, G.T.; Clum, A.; Dunn, C.; O’Malley, R.; Figueroa-Balderas, R.; Morales-Cruz, A.; et al. Phased diploid genome assembly with single-molecule real-time sequencing. Nat. Methods 2016, 13, 1050–1054. [Google Scholar] [CrossRef] [PubMed]
- Mathew, L.S.; Spannagl, M.; Al-Malki, A.; George, B.; Torres, M.F.; Al-Dous, E.K.; Al-Azwani, E.K.; Hussein, E.; Mathew, S.; Mayer, K.F.; et al. A first genetic map of date palm (Phoenix dactylifera) reveals long-range genome structure conservation in the palms. BMC Genom. 2014, 15, 285. [Google Scholar] [CrossRef] [PubMed]
- Sattar, M.N.; Iqbal, Z.; Tahir, M.N.; Shahid, M.S.; Khurshid, M.; Al-Khateeb, A.A.; Al-Khateeb, S.A. CRISPR/Cas9: A practical approach in date palm genome editing. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Sabir, J.S.M.; Arasappan, D.; Bahieldin, A.; Abo-Aba, S.; Bafeel, S.; Zari, T.A.; Edris, S.; Shokry, A.M.; Gadalla, N.O.; Ramadan, A.M.; et al. Whole mitochondrial and plastid genome SNP analysis of nine date palm cultivars reveals plastid heteroplasmy and close phylogenetic relationships among cultivars. PLoS ONE 2014, 9, e94158. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Pan, L.; Yin, Y.; Liu, W.; Huang, D.; Zhang, T.; Wang, L.; Xin, C.; Lin, Q.; Sun, G.; et al. Large-scale collection and annotation of gene models for date palm (Phoenix dactylifera, L.). Plant Mol. Biol. 2012, 79, 521–536. [Google Scholar] [CrossRef] [PubMed]
- Bourgis, F.; Kilaru, A.; Cao, X.; Ngando-Ebongue, G.-F.; Drira, N.; Ohlrogge, J.B.; Arondel, V. Comparative transcriptome and metabolite analysis of oil palm and date palm mesocarp that differ dramatically in carbon partitioning. Proc. Natl. Acad. Sci. USA 2011, 108, 12527–12532. [Google Scholar] [CrossRef] [PubMed]
- Sallon, S.; Solowey, E.; Cohen, Y.; Korchinsky, R.; Egli, M.; Woodhatch, I.; Simchoni, O.; Kislev, M. Germination, genetics, and growth of an ancient date seed. Science 2008, 320. [Google Scholar] [CrossRef] [PubMed]
- Dransfield, J.; Uhl, N.W.; Asmussen, C.B.; Baker, W.J.; Harley, M.M.; Lewis, C.E. Genera Palmarum: The Evolution and Classification of Palms; Kew Publishing: Kew, UK, 2008; p. 663. ISBN 1842461826. [Google Scholar]
- Govaerts, R.; Dransfield, J. World Checklist of Palms; Royal Botanic Gardens, Kew: Kew, UK, 2005; p. 235. ISBN 1842460846. [Google Scholar]
- Newton, C.; Gros-Balthazard, M.; Ivorra, S.; Paradis, L.; Pintaud, J.-C.; Terral, J.-F. Phoenix dactylifera and P. sylvestris in Northwestern India: A glimpse into their complex relationships. Palms 2013, 57, 37–50. [Google Scholar]
- Palmweb—Palms of the World Online. Available online: http://www.palmweb.org/ (accessed on 1 September 2018).
- Henderson, S.A.; Billotte, N.; Pintaud, J.C. Genetic isolation of Cape Verde Island Phoenix atlantica (Arecaceae) revealed by microsatellite markers. Conserv. Genet. 2006, 7, 213–223. [Google Scholar] [CrossRef]
- Rivera, D.; de Castro, C.O.; Carreño, E.; Inocencio, C.; Alcaraz, F.; Rios, S.; Palazon, J.A.; Vazquez, L.; Laguna, E. Morphological systematics of date-palm diversity (Phoenix, Arecaceae) in Western Europe and some preliminary molecular results. In 5th International Symposium on the Taxonomy of Cultivated Plants; [CDROM]; Groendijk-Wilders, N., Alexander, C., VandenBerg, R.G., Hetterscheid, W.L.A., Eds.; ISHS Acta Horticulturae: Wageningen, The Netherlands, 2007; Volume 1. [Google Scholar]
- Pintaud, J.C.; Zehdi, S.; Couvreur, T.L.P.; Barrow, S.; Henderson, S.; Aberlenc-Bertossi, F.; Tregear, J.; Billotte, N. Species delimitation in the genus Phoenix (Arecaceae) based on SSR markers, with emphasis on the identity of the Date Palm (Phoenix dactylifera L.). In Diversity Phylogeny, and Evolution in the Monocotyledons; Seberg, O., Petersen, G., Barford, A., Davis, J., Eds.; Aarhus University Press: Aarhus, Denmark, 2010; pp. 267–286. ISBN 8779343988. [Google Scholar]
- Munier, P. Le Palmier-Dattier; Techniques Agricoles et Productions Tropicales Series 24; Maisonneuve & Larose: Paris, France, 1973. [Google Scholar]
- González-Pérez, M.A.; Caujapé-Castells, J.; Sosa, P.A. Molecular evidence of hybridisation between the endemic Phoenix canariensis and the widespread P. dactylifera with random amplified polymorphic DNA (RAPD) markers. Plant Syst. Evol. 2004, 247, 165–175. [Google Scholar] [CrossRef]
- Gonzáles-Perez, M.A.; Sosa, P.A. Hybridisation and introgression between the endemic Phoenix canariensis and the introduced P. dactylifera in the Canary Islands. Open For. Sci. J. 2009, 2, 78–85. [Google Scholar] [CrossRef]
- Gros-Balthazard, M. Hybridization in the genus Phoenix: A review. Emirates J. Food Agric. 2013, 25, 831–842. [Google Scholar] [CrossRef]
- Ballardini, M.; Mercuri, A.; Littardi, C.; Abbas, S.; Couderc, M.; Ludeña, B.; Pintaud, J.-C. The chloroplast DNA locus psbZ-trnfM as a potential barcode marker in Phoenix L. (Arecaceae). Zookeys 2013, 365, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Zohary, D.; Spiegel-Roy, P. Beginnings of fruit growing in Old World. Science 1975, 187, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Zohary, D.; Hopf, M.; Weiss, E. Fruit trees and nuts. In Domestication of Plants in the Old World, 4th ed.; Oxford University Press: Oxford, UK, 2012; pp. 114–152. ISBN 0199688176. [Google Scholar]
- Tengberg, M. Research into the origins of date palm domestication. In The Date Palm: From Traditional Resource to Green Wealth; Emirates Center for Strategic Studies and Research: Abu Dhabi, UAE, 2003; pp. 51–64. ISBN 9948005511. [Google Scholar]
- Terral, J.F.; Newton, C.; Ivorra, S.; Gros-Balthazard, M.; Tito de Morais, C.; Picq, S.; Tengberg, M.; Pintaud, J.C. Insights into the historical biogeography of the date palm (Phoenix dactylifera L.) using geometric morphometry of modern and ancient seeds. J. Biogeogr. 2012, 39, 929–941. [Google Scholar] [CrossRef]
- Terral, J.F.; Alonso, N.; Capdevila, R.B.I.; Chatti, N.; Fabre, L.; Fiorentino, G.; Marinval, P.; Jorda, G.P.; Pradat, B.; Rovira, N.; et al. Historical biogeography of olive domestication (Olea europaea L.) as revealed by geometrical morphometry applied to biological and archaeological material. J. Biogeogr. 2003, 31. [Google Scholar] [CrossRef]
- Terral, J.F.; Tabard, E.; Bouby, L.; Ivorra, S.; Pastor, T.; Figueiral, I.; Picq, S.; Chevance, J.B.; Jung, C.; Fabre, L.; et al. Evolution and history of grapevine (Vitis vinifera) under domestication: New morphometric perspectives to understand seed domestication syndrome and reveal origins of ancient European cultivars. Ann. Bot. 2010, 105, 443–455. [Google Scholar] [CrossRef] [PubMed]
- Gros-Balthazard, M.; Newton, C.; Ivorra, S.; Pierre, M.-H.; Pintaud, J.-C.; Terral, J.-F. The domestication syndrome in Phoenix dactylifera seeds: Toward the identification of wild date palm populations. PLoS ONE 2016, 11, e0152394. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.J.; Gross, B.L. From forest to field: perennial fruit crop domestication. Am. J. Bot. 2011, 98, 1389–1414. [Google Scholar] [CrossRef] [PubMed]
- Solecki, R.S.; Leroi-gourhan, A. Palaeoclimatology and archaeology in the near east. Ann. N. Y. Acad. Sci. 1961, 95, 729–739. [Google Scholar] [CrossRef]
- Liphschitz, N.; Nadel, D. Epipalaeolithic (19,000 B.P.) charred wood remains from Ohalo II. J. Isr. Prehist. Soc. 1997, 27, 5–18. [Google Scholar]
- Zehdi-Azouzi, S.; Cherif, E.; Moussouni, S.; Gros-Balthazard, M.; Abbas Naqvi, S.; Ludeña, B.; Castillo, K.; Chabrillange, N.; Bouguedoura, N.; Bennaceur, M.; et al. Genetic structure of the date palm (Phoenix dactylifera) in the Old World reveals a strong differentiation between eastern and western populations. Ann. Bot. 2015, 116, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Cherif, E.; Zehdi-Azouzi, S.; Crabos, A.; Castillo, K.; Chabrillange, N.; Pintaud, J.-C.; Amel, S.-H.; Glémin, S.; Aberlenc-Bertossi, F. Evolution of sex chromosomes prior to speciation in the dioecious Phoenix species. J. Evol. Biol. 2016, 1513–1522. [Google Scholar] [CrossRef] [PubMed]
- Cherif, E.; Zehdi, S.; Castillo, K.; Chabrillange, N.; Abdoulkader, S.; Pintaud, J.-C.; Santoni, S.; Salhi-Hannachi, A.; Glémin, S.; Aberlenc-Bertossi, F. Male-specific DNA markers provide genetic evidence of an XY chromosome system, a recombination arrest and allow the tracing of paternal lineages in date palm. New Phytol. 2013, 197, 409–415. [Google Scholar] [CrossRef] [PubMed]
| Genome | Cultivar | GenBank Reference Number | Size | Number of Scaffolds | N50 | Reference |
|---|---|---|---|---|---|---|
| Nuclear | Khalas | GCA_000181215.2 | ~381 Mb | 57,277 | 30,480 | Al-Dous et al., 2011 [19] |
| Nuclear | Khalas | GCA_000413155.1 | ~558 Mb | 82,354 | 329,900 | Al-Mssallem et al., 2013 [20] |
| Chloroplastic | Khalas | NC_013991.2 | 158,462 bp | / | / | Yang et al., 2010 [21] |
| Chloroplastic | Aseel | FJ212316.3 | 158,458 bp | / | / | Khan et al., 2012 [22] |
| Chloroplastic | Khanezi | / | 158,211 bp | / | / | Khan et al., 2018 [23] |
| Chloroplastic | Naghal | / | 158,210 bp | / | / | Khan et al., 2018 [23] |
| Mitochondrial | Khalas | NC_016740 | 715,001 bp | / | / | Fang et al., 2012 [24] |
| Mitochondrial | unknown | MG257490.1 | / | / | / | |
| Mitochondrial | Khanezi | MH176159.1 | / | / | / |
| Reference | # Accession (# New) | Summary of Major Findings |
|---|---|---|
| Al-Dous et al., 2011 [19] | 9 (9) cultivated date palms | First genome assembly of the date palm genome (cultivar Khalas). The paper further focuses on sex determination, providing the evidence that the date palm employs an XY system of gender inheritance. |
| Al-Mssallem et al., 2013 [20] | 11 (4) cultivated date palms | Improved genome assembly of the date palm genome (cultivar Khalas) and study of genetic diversity among a few cultivars. Functional genes involved in stress resistance and sugar metabolism were brought to light. |
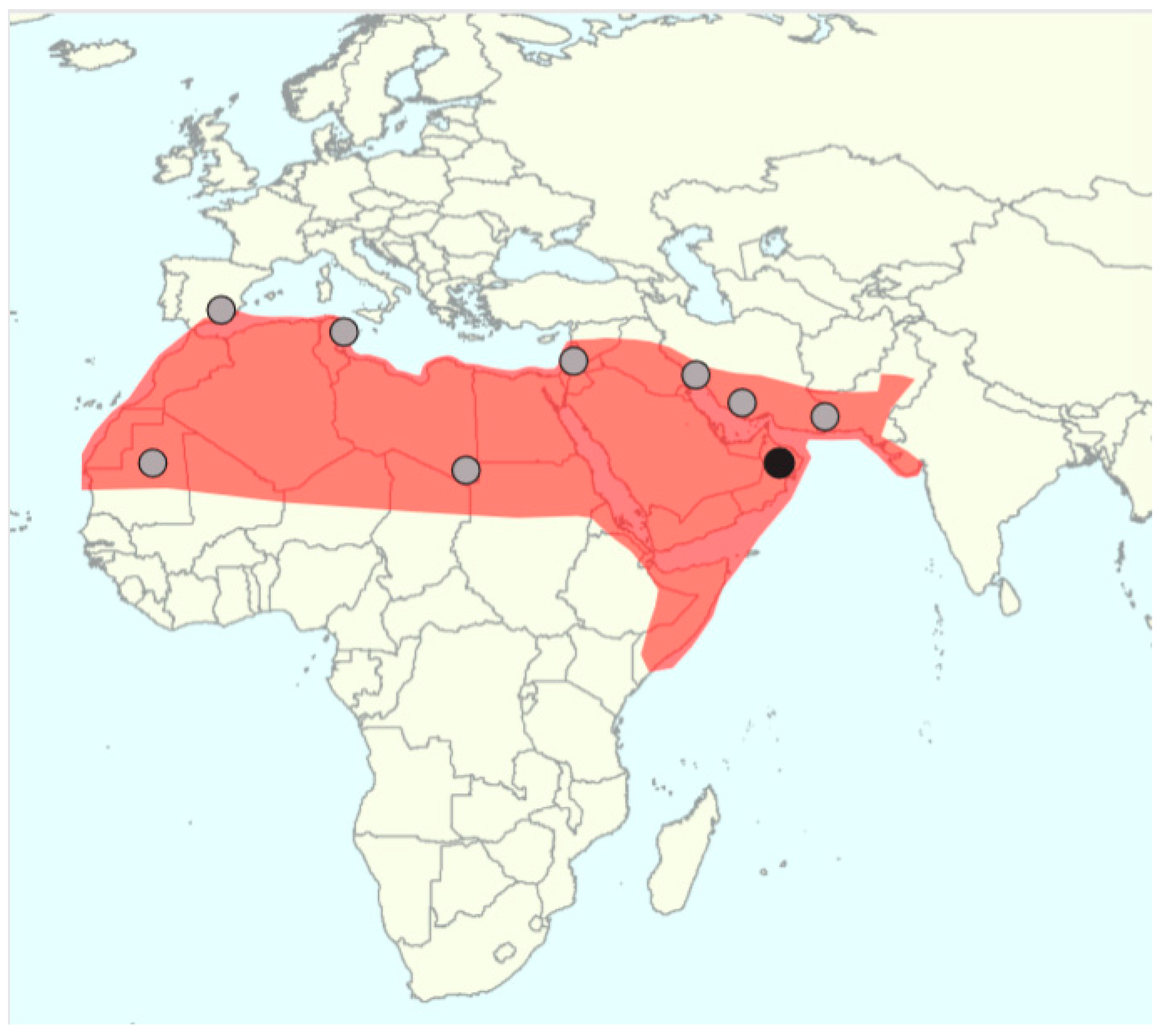
| Hazzouri et al., 2015 [32] | 62 (62) cultivated date palms | Resequencing study of 62 cultivars from North Africa and the Middle East providing evidence for a large differentiation between these two gene pools. A larger diversity in North African date palms is noted, challenging the classic scenario stating that they derive from Middle Eastern cultivars. The orthologue of the oil palm Virescens gene was linked to color polymorphism (red/yellow) in dates. |
| Gros-Balthazard et al., 2017 [33] | 16 (2) date palms 3 (3) wild date palms 1 (1) Phoenix sylvestris 1 (1) Phoenix atlantica | Candidate wild date palms growing in Oman were hypothesized based on seed morphometric features and diversity analyses (microsatellite data). Further whole-genome analyses, including structure, diversity, and modeling, demonstrated that they are ancestral, leading to the first report of wild date palms. African date palms were shown to mostly derive from Middle Eastern cultivars although an unknown source of variability was noted. |
| Torres et al., 2018 [34] | 15 female and 13 male individuals representing all 14 species (no male Phoenix pusilla could be identified for sequencing) | Whole-genome sequencing of males and females from all Phoenix species allowed the identification of male-specific sequences. The sex-determination region was further sequenced using long read technologies and annotated. Four genes were identified and their analysis supported a two-mutation model for the evolution of dioecy in Phoenix. |
| Species | Current Status | Distribution |
|---|---|---|
| Phoenix acaulis | Recognized species | Southern Asia |
| Phoenix andamanensis | Recognized species | Andaman and Nicobar Islands |
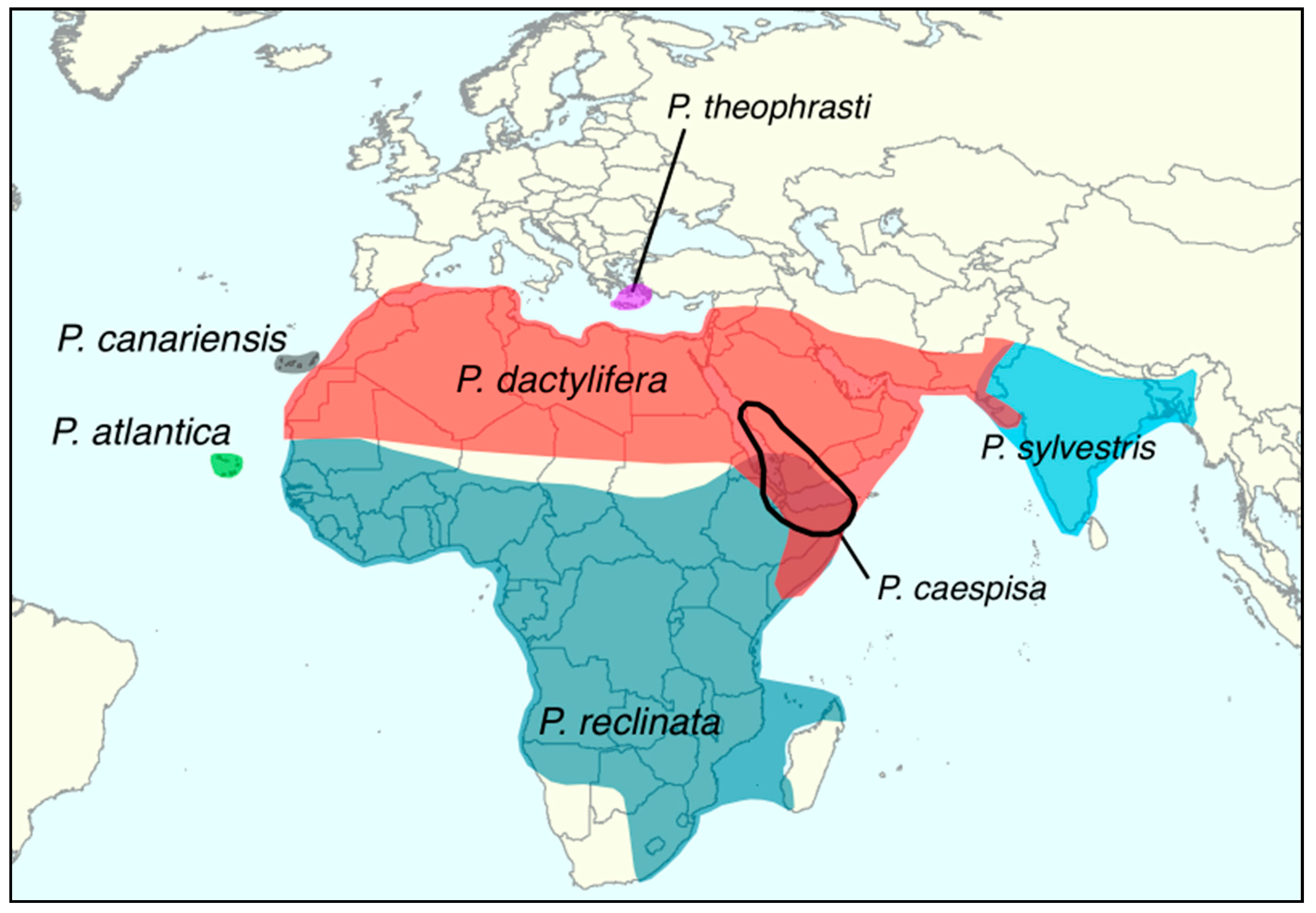
| Phoenix atlantica [48] | Recognized species but status warrant verification following a genomic study that failed to differentiate it from African date palms [33] | Cape Verde (see Figure 1) |
| Phoenix caespitosa | Recognized species | Southern Arabia and Horn of Africa (see Figure 1) |
| Phoenix canariensis | Recognized species | Canary Islands (see Figure 1) |
| Phoenix chevalierii [49] | Unrecognized species | Southern Spain |
| Phoenix dactylifera | Recognized species | Southern Spain, North Africa, the Middle East, Pakistan, and Northwestern India (see Figure 1) Recently introduced in many locations including California and China |
| Phoenix iberica [49] | Unrecognized species | Southern Spain |
| Phoenix loureiroi | Recognized species | Southern Asia |
| Phoenix paludosa | Recognized species | Southern Asia |
| Phoenix pusilla | Recognized species | Indian subcontinent |
| Phoenix reclinata | Recognized species | Sub-Saharan Africa (see Figure 1) |
| Phoenix roebelenii | Recognized species | Southeast Asia |
| Phoenix rupicola | Recognized species | Foothills of the Himalayas |
| Phoenix sylvestris | Recognized species | Indian subcontinent (see Figure 1) |
| Phoenix theophrasti | Recognized species | Crete and coastal Turkey (see Figure 1) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gros-Balthazard, M.; Hazzouri, K.M.; Flowers, J.M. Genomic Insights into Date Palm Origins. Genes 2018, 9, 502. https://doi.org/10.3390/genes9100502
Gros-Balthazard M, Hazzouri KM, Flowers JM. Genomic Insights into Date Palm Origins. Genes. 2018; 9(10):502. https://doi.org/10.3390/genes9100502
Chicago/Turabian StyleGros-Balthazard, Muriel, Khaled Michel Hazzouri, and Jonathan Mark Flowers. 2018. "Genomic Insights into Date Palm Origins" Genes 9, no. 10: 502. https://doi.org/10.3390/genes9100502
APA StyleGros-Balthazard, M., Hazzouri, K. M., & Flowers, J. M. (2018). Genomic Insights into Date Palm Origins. Genes, 9(10), 502. https://doi.org/10.3390/genes9100502





