Angiogenic microRNAs in Systemic Sclerosis: Insights into Microvascular Dysfunction and Therapeutic Implications
Highlights
- Identification of a novel set of angiogenic microRNAs that are significantly dysregulated in patients with systemic sclerosis, offering potential biomarkers for disease progression and vascular dysfunction.
- Demonstration of a clear correlation between altered miRNA profiles and key clinical features of systemic sclerosis (e.g., digital ulcers and pulmonary hypertension), supporting a mechanistic link between angiogenic-miRNA dysregulation and microvascular injury.
- Integration of machine-learning-driven pathway analysis reveals previously unrecognized gene networks regulated by these miRNAs—including VEGF, Notch, and TGF-β signaling—which may serve as therapeutic targets in systemic sclerosis.
- Proposal of a translational framework: from profiling miRNA signatures through functional validation to envisaging patient-stratified therapeutic strategies, thereby paving the way for personalized vascular care in systemic sclerosis.
Abstract
1. Introduction
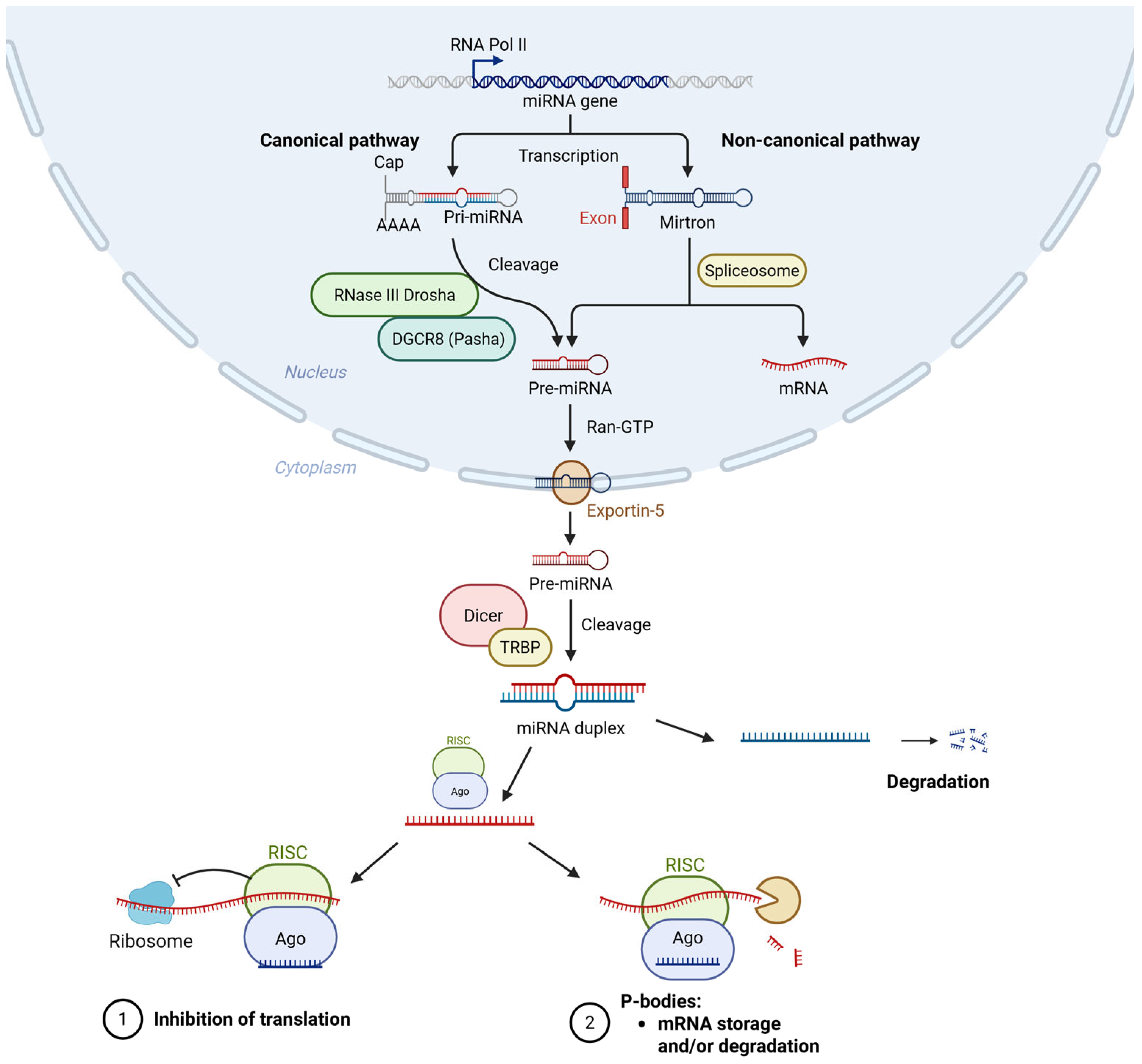
2. microRNAs Biogenesis and Function
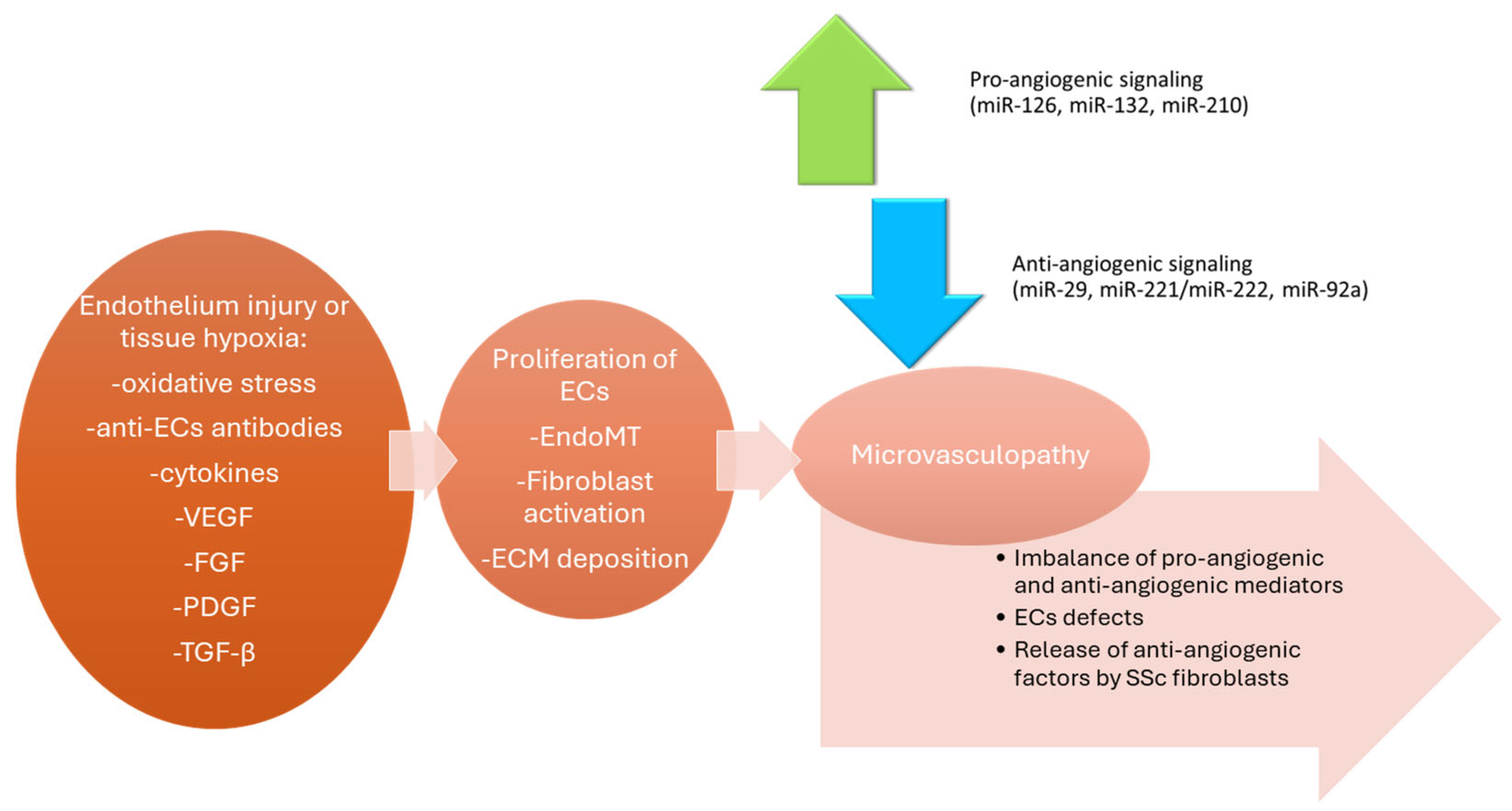
3. Endothelial Dysfunction and Microvascular Damage in SSc
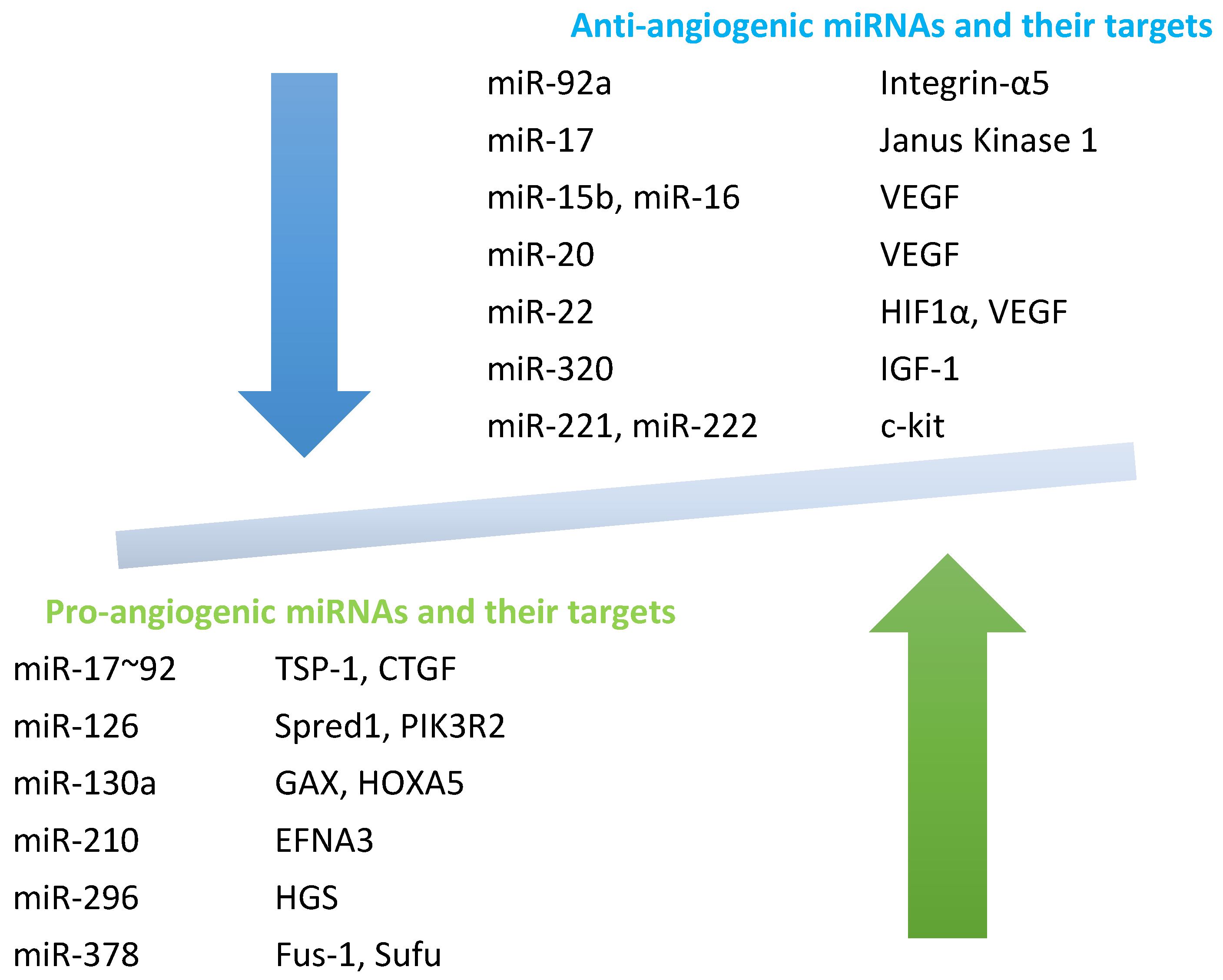
4. Angiogenic and Anti-Angiogenic miRNAs in SSc
4.1. Pro-Angiogenic miRNAs
4.1.1. miR-126
4.1.2. miR-130a
4.1.3. miR-210
4.1.4. miR-296
4.1.5. miR-148b
4.1.6. miR-155
4.1.7. let-7f and miR-27b
4.1.8. miR-152
4.1.9. miR-193b
4.1.10. miR-20a
4.1.11. miR-146a
4.1.12. miR-125a
4.2. Anti-Angiogenic miRNAs
4.2.1. miR-221/222
4.2.2. miR-92a
4.2.3. miR-15b, miR-16, miR-20a/b
4.2.4. miR-22
4.2.5. miR-200c
4.2.6. miR-34a
4.2.7. miR-214
4.2.8. miR-217
4.2.9. miR-328
4.3. miRNAs Regulating EndoMT and EC Apoptosis
5. miRNAs as Diagnostic and Prognostic Biomarkers Results
6. Therapeutic Potential of Targeting miRNAs
6.1. Experimental miRNA-Based Therapies in SSc
6.2. Challenges in Delivery and Specificity
6.3. miRNA–lncRNA–circRNA Interactions
6.4. Clinical Translation: Current Status
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AECAs | Anti-endothelial cell antibodies |
| Ago2 | Argonaut 2 |
| AT1R | Angiotensin II type 1 receptor |
| CD133 | Prominin-1 |
| ceRNAs | Competing endogenous RNAs |
| CEUS | Contrast-enhanced ultrasound |
| circRNAs | Circular RNAs |
| c-Kit (CD117) | Stem cell factor receptor |
| COL1A1 | Collagen type 1, alpha 1 |
| CTGF | Connective tissue growth factor |
| CUL2 | Cullin-2 |
| DNMT1 | DNA methyltransferase 1 |
| ECM | Extracellular Matrix |
| ECs | Endothelial cells |
| EFNA3 | Ephrin-A3 |
| EndoMT | Endothelial-to-mesenchymal transition |
| eNOS | Endothelial nitric oxide synthase |
| EPCs | Endothelial progenitor cells |
| ET-1 | Endothelin-1 |
| FBs | Fibroblasts |
| FGF | Fibroblast growth factor |
| Fli1 | Friend leukemia integration 1 transcription factor |
| FOXO | Forkhead family of transcription factors |
| Fra2 | Fos-related antigen 2 |
| FUS-1 | Nuclear fusion protein |
| GAX | Growth arrest-specific homeobox gene |
| HGS | Hepatocyte growth factor-regulated tyrosine kinase substrate |
| HIF1α | Hypoxia-inducible factor 1alfa |
| HOX-A5 | Homeobox protein HOX-A5 |
| HUVECs | Human umbilical vein endothelial cells |
| ICAM-1 | Intercellular adhesion molecule 1 |
| IGF-1 | Insulin-like growth factor 1 |
| IL-1β | Interleukin 1beta |
| ITG-α5 | Integrin alfa5 |
| KLF-4, 5 | Kruppel-like factor 4, 5 |
| LNA | Locked nucleic acid |
| lncRNAs | Long non-coding RNAs |
| MAP3K7 | Mitogen-activated protein kinase 7 |
| MiRNAs | MicroRNAs |
| MKK3 | Mitogen-activated protein kinase 3 |
| MMP1 | Matrix metalloproteinase 1 |
| MVECs | Microvascular endothelial cells |
| NFκβ | Nuclear factor kappa beta |
| NO | Nitric oxide |
| NOX4 | NADPH oxidase 4 |
| NPM1 | Nucleophosmin 1 |
| NPTX1 | Neuronal pentraxin 1 |
| NVC | Nailfold videocapillaroscopy |
| OCTA | Optical coherence tomography angiography |
| p53 | Protein p53 |
| PAH | Pulmonary hypertension |
| PAK1 | p21-Activated kinase 1 |
| PDGF | Platelet derived growth factor |
| PDGFRB | Platelet derived growth factor receptor beta |
| PFKFB3 | 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3 |
| PHD1,2 | Prolyl hydroxylase |
| PIK3R2 | Phosphoinositide-3-kinase regulatory subunit 2 |
| PLAU | Plasminogen activator, urokinase |
| Pre-miRNAs | Precursor miRNAs |
| Pri-miRNAs | Primary miRNAs |
| PTEN | Phosphatase and tensin homolog |
| RAS/ERK pathway | Rat Sarcoma/Extracellular Signal-Regulated Kinase |
| RISC | RNA-induced silencing complex |
| ROBO1 | Roundabout guidance receptor 1 |
| ROS | Reactive oxygen species |
| SEMA6A | Semaphoring 6A |
| siRNAs | Silent interfering RNAs |
| SIRT1 | Sirtuin 1 |
| SMAD3/7 | Mothers against decapentaplegic homologue 3/7 |
| SPRED | Sprouty-related, EVH1 do-main-containing protein 1 |
| SSc | Systemic sclerosis |
| STAT5a | Signal transducer and activator of transcription 5a |
| SUFU | Suppressor of fused homolog |
| TAK1 | Transforming growth factor beta-activated kinase 1 |
| TGF-β1 | Transforming growth factor beta-1 |
| TIMP-1 | Tissue inhibitor of metalloproteinases |
| TNF-α | Tumor necrosis factor alfa |
| TSP-1 | Thrombospondin 1 |
| TSR | Methyl-accepting chemotaxis protein |
| UTR | Untranslated region |
| VASH1 | Vasohibin 1 |
| VCAM-1 | Vascular cell adhesion protein 1 |
| VE-cadherin | Vascular endothelia cadherin |
| VEGF | Vascular endothelial growth factor |
| VEGFR-2 | Vascular endothelial growth factor receptor 2 |
| VSMCs | Vascular smooth muscle cells |
| XPO5 | Exportin 5 |
| ZEB1 | Zinc finger E-box-binding homeobox 1 |
| α-SMA | alfa-smooth muscle actin |
| β-TRC | beta-transducin repeat-containing gene |
References
- Trojanowska, M. Cellular and Molecular Aspects of Vascular Dysfunction in Systemic Sclerosis. Nat. Rev. Rheumatol. 2010, 6, 453. [Google Scholar] [CrossRef] [PubMed]
- Fleming, J.N.; Schwartz, S.M. The Pathology of Scleroderma Vascular Disease. Rheum. Dis. Clin. N. Am. 2008, 34, 41–55. [Google Scholar] [CrossRef] [PubMed]
- Varga, J.; Abraham, D. Systemic Sclerosis: A Prototypic Multisystem Fibrotic Disorder. J. Clin. Investig. 2007, 117, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Shumnalieva, R.; Monov, S.; Velikova, T. MicroRNAs in Systemic Sclerosis: Involvement in Disease Pathogenesis and Potential Use as Diagnostic Biomarkers and Therapeutic Targets. Biomedicines 2025, 13, 1216. [Google Scholar] [CrossRef]
- Jimenez, S.A.; Mendoza, F.A.; Piera-Velazquez, S. A Review of Recent Studies on the Pathogenesis of Systemic Sclerosis: Focus on Fibrosis Pathways. Front. Immunol. 2025, 16, 1551911. [Google Scholar] [CrossRef]
- Flower, V.A.; Barratt, S.L.; Ward, S.; Pauling, J.D. The Role of Vascular Endothelial Growth Factor in Systemic Sclerosis. Curr. Rheumatol. Rev. 2018, 15, 99–109. [Google Scholar] [CrossRef]
- Maurer, B.; Distler, A.; Suliman, Y.A.; Gay, R.E.; Michel, B.A.; Gay, S.; Distler, J.H.W.; Distler, O. Vascular Endothelial Growth Factor Aggravates Fibrosis and Vasculopathy in Experimental Models of Systemic Sclerosis. Ann. Rheum. Dis. 2014, 73, 1880–1887. [Google Scholar] [CrossRef]
- Campitiello, R.; Soldano, S.; Gotelli, E.; Hysa, E.; Montagna, P.; Casabella, A.; Paolino, S.; Pizzorni, C.; Sulli, A.; Smith, V.; et al. The Intervention of Macrophages in Progressive Fibrosis Characterizing Systemic Sclerosis: A Systematic Review. Autoimmun. Rev. 2024, 23, 103637. [Google Scholar] [CrossRef]
- Dudley, A.C.; Griffioen, A.W. Pathological Angiogenesis: Mechanisms and Therapeutic Strategies. Angiogenesis 2023, 26, 313–347. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Wei, J.; Varga, J. Understanding Fibrosis in Systemic Sclerosis: Shifting Paradigms, Emerging Opportunities. Nat. Rev. Rheumatol. 2012, 8, 42–54. [Google Scholar] [CrossRef]
- Al-Dhaher, F.F.; Pope, J.E.; Ouimet, J.M. Determinants of Morbidity and Mortality of Systemic Sclerosis in Canada. Semin. Arthritis Rheum. 2010, 39, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Simeon-Aznar, C.P.; Fonollosa-Pla, V.; Tolosa-Vilella, C.; Espinosa-Garriga, G.; Campillo-Grau, M.; Ramos-Casals, M.; Garcia-Hernandez, F.J.; Castillo-Palma, M.J.; Sanchez-Roman, J.; Callejas-Rubio, J.L.; et al. Registry of the Spanish Network for Systemic Sclerosis: Survival, Prognostic Factors, and Causes of Death. Medicine 2015, 94, e1728. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lunardi, C.; Bason, C.; Navone, R.; Millo, E.; Damonte, G.; Corrocher, R.; Puccetti, A. Systemic Sclerosis Immunoglobulin G Autoantibodies Bind the Human Cytomegalovirus Late Protein UL94 and Induce Apoptosis in Human Endothelial Cells. Nat. Med. 2000, 6, 1183–1186. [Google Scholar] [CrossRef]
- Lunardi, C.; Dolcino, M.; Peterlana, D.; Bason, C.; Navone, R.; Tamassia, N.; Beri, R.; Corrocher, R.; Puccetti, A. Antibodies against Human Cytomegalovirus in the Pathogenesis of Systemic Sclerosis: A Gene Array Approach. PLoS Med. 2005, 3, e2. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, Y.; Wang, Q.; Xiao, R.; Lu, Q. Systemic Sclerosis: Genetics and Epigenetics. J. Autoimmun. 2013, 41, 161–167. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, Y.; Shu, Y.; Lu, Q.; Xiao, R. Epigenetic Mechanisms: An Emerging Role in Pathogenesis and Its Therapeutic Potential in Systemic Sclerosis. Int. J. Biochem. Cell Biol. 2015, 67, 92–100. [Google Scholar] [CrossRef]
- Dolcino, M.; Pelosi, A.; Fiore, P.F.; Patuzzo, G.; Tinazzi, E.; Lunardi, C.; Puccetti, A. Gene Profiling in Patients with Systemic Sclerosis Reveals the Presence of Oncogenic Gene Signatures. Front. Immunol. 2018, 9, 449. [Google Scholar] [CrossRef]
- Altorok, N.; Almeshal, N.; Wang, Y.; Kahaleh, B. Epigenetics, the Holy Grail in the Pathogenesis of Systemic Sclerosis. Rheumatology 2015, 54, 1759–1770. [Google Scholar] [CrossRef]
- Makino, T.; Jinnin, M. Genetic and Epigenetic Abnormalities in Systemic Sclerosis. J. Dermatol. 2016, 43, 10–18. [Google Scholar] [CrossRef]
- Fioretto, B.S.; Rosa, I.; Matucci-Cerinic, M.; Romano, E.; Manetti, M. Current Trends in Vascular Biomarkers for Systemic Sclerosis: A Narrative Review. Int. J. Mol. Sci. 2023, 24, 4097. [Google Scholar] [CrossRef]
- Matucci-Cerinic, M.; Manetti, M.; Bruni, C.; Chora, I.; Bellando-Randone, S.; Lepri, G.; De Paulis, A.; Guiducci, S. The “Myth” of Loss of Angiogenesis in Systemic Sclerosis: A Pivotal Early Pathogenetic Process or Just a Late Unavoidable Event? Arthritis Res. Ther. 2017, 19, 162. [Google Scholar] [CrossRef] [PubMed]
- Romano, E.; Rosa, I.; Fioretto, B.S.; Manetti, M. Recent Insights into Cellular and Molecular Mechanisms of Defective Angiogenesis in Systemic Sclerosis. Biomedicines 2024, 12, 1331. [Google Scholar] [CrossRef] [PubMed]
- Broen, J.C.A.; Radstake, T.R.D.J.; Rossato, M. The Role of Genetics and Epigenetics in the Pathogenesis of Systemic Sclerosis. Nat. Rev. Rheumatol. 2014, 10, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Luo, H.; Zuo, X. MicroRNAs: Their Involvement in Fibrosis Pathogenesis and Use as Diagnostic Biomarkers in Scleroderma. Exp. Mol. Med. 2013, 45, e41. [Google Scholar] [CrossRef]
- Bartel, D.P. Metazoan MicroRNAs. Cell 2018, 173, 20–51. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Genomics, Biogenesis, Mechanism, and Function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Singhvi, G.; Manchanda, P.; Krishna Rapalli, V.; Kumar Dubey, S.; Gupta, G.; Dua, K. MicroRNAs as Biological Regulators in Skin Disorders. Biomed. Pharmacother. 2018, 108, 996–1004. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Target Recognition and Regulatory Functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef]
- Agarwal, V.; Bell, G.W.; Nam, J.-W.; Bartel, D.P. Predicting Effective MicroRNA Target Sites in Mammalian MRNAs. elife 2015, 4, e05005. [Google Scholar] [CrossRef]
- Duttagupta, R.; Jiang, R.; Gollub, J.; Getts, R.C.; Jones, K.W. Impact of Cellular MiRNAs on Circulating MiRNA Biomarker Signatures. PLoS ONE 2011, 6, e20769. [Google Scholar] [CrossRef]
- Deng, X.; Su, Y.; Wu, H.; Wu, R.; Zhang, P.; Dai, Y.; Chan, T.M.; Zhao, M.; Lu, Q. The Role of MicroRNAs in Autoimmune Diseases with Skin Involvement. Scand. J. Immunol. 2015, 81, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Ruan, K. MicroRNA Detection by Microarray. Anal. Bioanal. Chem. 2009, 394, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, C.C.; Cheng, H.H.; Tewari, M. MicroRNA Profiling: Approaches and Considerations. Nat. Rev. Genet. 2012, 13, 358–369. [Google Scholar] [CrossRef] [PubMed]
- Reid, G.; Kirschner, M.B.; van Zandwijk, N. Circulating MicroRNAs: Association with Disease and Potential Use as Biomarkers. Crit. Rev. Oncol. Hematol. 2011, 80, 193–208. [Google Scholar] [CrossRef]
- Andreasen, D.; Fog, J.U.; Biggs, W.; Salomon, J.; Dahslveen, I.K.; Baker, A.; Mouritzen, P. Improved MicroRNA Quantification in Total RNA from Clinical Samples. Methods 2010, 50, S6–S9. [Google Scholar] [CrossRef]
- O’Connell, R.M.; Rao, D.S.; Baltimore, D. MicroRNA Regulation of Inflammatory Responses. Annu. Rev. Immunol. 2012, 30, 295–312. [Google Scholar] [CrossRef]
- Quiat, D.; Olson, E.N. MicroRNAs in Cardiovascular Disease: From Pathogenesis to Prevention and Treatment. J. Clin. Investig. 2013, 123, 11–18. [Google Scholar] [CrossRef]
- Turchinovich, A.; Weiz, L.; Langheinz, A.; Burwinkel, B. Characterization of Extracellular Circulating MicroRNA. Nucleic Acids Res. 2011, 39, 7223–7233. [Google Scholar] [CrossRef]
- Murakami, Y.; Tanahashi, T. Analysis of Circulating MicroRNA by Microarray in Liver Disease. Methods Mol. Biol. 2013, 1024, 173–182. [Google Scholar] [CrossRef]
- Ma, B.; Wang, S.; Wu, W.; Shan, P.; Chen, Y.; Meng, J.; Xing, L.; Yun, J.; Hao, L.; Wang, X.; et al. Mechanisms of CircRNA/LncRNA-MiRNA Interactions and Applications in Disease and Drug Research. Biomed. Pharmacother. 2023, 162, 114672. [Google Scholar] [CrossRef]
- Denzler, R.; Agarwal, V.; Stefano, J.; Bartel, D.P.; Stoffel, M. Assessing the CeRNA Hypothesis with Quantitative Measurements of MiRNA and Target Abundance. Mol. Cell 2014, 54, 766–776. [Google Scholar] [CrossRef]
- Gao, Y.; Takenaka, K.; Xu, S.-M.; Cheng, Y.; Janitz, M. Recent Advances in Investigation of CircRNA/LncRNA-MiRNA-MRNA Networks Through RNA Sequencing Data Analysis. Brief. Funct. Genom. 2025, 24, elaf005. [Google Scholar] [CrossRef]
- Wu, F.; Yang, Z.; Li, G. Role of Specific MicroRNAs for Endothelial Function and Angiogenesis. Biochem. Biophys. Res. Commun. 2009, 386, 549–553. [Google Scholar] [CrossRef] [PubMed]
- Zaccone, V.; Falsetti, L.; Contegiacomo, S.; Cataldi, S.; Benfaremo, D.; Moroncini, G. Systemic Sclerosis: A Key Model of Endothelial Dysfunction. Biomedicines 2025, 13, 1771. [Google Scholar] [CrossRef] [PubMed]
- Kazerounian, S.; Lawler, J. Integration of Pro- and Anti-Angiogenic Signals by Endothelial Cells. J. Cell Commun. Signal. 2018, 12, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Asano, Y.; Ihn, H.; Yamane, K.; Jinnin, M.; Tamaki, K. Increased Expression of Integrin Avβ5 Induces the Myofibroblastic Differentiation of Dermal Fibroblasts. Am. J. Pathol. 2006, 168, 499–510. [Google Scholar] [CrossRef] [PubMed]
- Lawler, P.R.; Lawler, J. Molecular Basis for the Regulation of Angiogenesis by Thrombospondin-1 and -2. Cold Spring Harb. Perspect. Med. 2012, 2, a006627. [Google Scholar] [CrossRef]
- Altorok, N.; Wang, Y.; Kahaleh, B. Endothelial Dysfunction in Systemic Sclerosis. Curr. Opin. Rheumatol. 2014, 26, 615–620. [Google Scholar] [CrossRef]
- Bonella, F.; Patuzzo, G.; Lunardi, C. Biomarker Discovery in Systemic Sclerosis: State of the Art. Curr. Biomark. Find. 2015, 5, 47–68. [Google Scholar] [CrossRef]
- Kuwana, M.; Okazaki, Y.; Yasuoka, H.; Kawakami, Y.; Ikeda, Y. Defective Vasculogenesis in Systemic Sclerosis. Lancet 2004, 364, 603–610. [Google Scholar] [CrossRef]
- Whitfield, M.L.; Finlay, D.R.; Murray, J.I.; Troyanskaya, O.G.; Chi, J.-T.; Pergamenschikov, A.; McCalmont, T.H.; Brown, P.O.; Botstein, D.; Connolly, M.K. Systemic and Cell Type-Specific Gene Expression Patterns in Scleroderma Skin. Proc. Natl. Acad. Sci. USA 2003, 100, 12319–12324. [Google Scholar] [CrossRef]
- Matucci-Cerinic, M.; Kahaleh, B.; Wigley, F.M. Review: Evidence That Systemic Sclerosis Is a Vascular Disease. Arthritis Rheum. 2013, 65, 1953–1962. [Google Scholar] [CrossRef] [PubMed]
- Manetti, M.; Romano, E.; Rosa, I.; Guiducci, S.; Bellando-Randone, S.; De Paulis, A.; Ibba-Manneschi, L.; Matucci-Cerinic, M. Endothelial-to-Mesenchymal Transition Contributes to Endothelial Dysfunction and Dermal Fibrosis in Systemic Sclerosis. Ann. Rheum. Dis. 2017, 76, 924–934. [Google Scholar] [CrossRef] [PubMed]
- LeRoy, E.C.; Black, C.; Fleischmajer, R.; Jablonska, S.; Krieg, T.; Medsger, T.A.J.; Rowell, N.; Wollheim, F. Scleroderma (Systemic Sclerosis): Classification, Subsets and Pathogenesis. J. Rheumatol. 1988, 15, 202–205. [Google Scholar] [PubMed]
- Rabquer, B.J.; Koch, A.E. Angiogenesis and Vasculopathy in Systemic Sclerosis: Evolving Concepts. Curr. Rheumatol. Rep. 2012, 14, 56–63. [Google Scholar] [CrossRef]
- Tsou, P.-S.; Rabquer, B.J.; Ohara, R.A.; Stinson, W.A.; Campbell, P.L.; Amin, M.A.; Balogh, B.; Zakhem, G.; Renauer, P.A.; Lozier, A.; et al. Scleroderma Dermal Microvascular Endothelial Cells Exhibit Defective Response to Pro-Angiogenic Chemokines. Rheumatology 2016, 55, 745–754. [Google Scholar] [CrossRef]
- Folkman, J. Angiogenesis. Annu. Rev. Med. 2006, 57, 1–18. [Google Scholar] [CrossRef]
- Manetti, M.; Guiducci, S.; Ibba-Manneschi, L.; Matucci-Cerinic, M. Mechanisms in the Loss of Capillaries in Systemic Sclerosis: Angiogenesis versus Vasculogenesis. J. Cell. Mol. Med. 2010, 14, 1241–1254. [Google Scholar] [CrossRef]
- Liakouli, V.; Cipriani, P.; Marrelli, A.; Alvaro, S.; Ruscitti, P.; Giacomelli, R. Angiogenic Cytokines and Growth Factors in Systemic Sclerosis. Autoimmun. Rev. 2011, 10, 590–594. [Google Scholar] [CrossRef]
- Distler, O.; Del Rosso, A.; Giacomelli, R.; Cipriani, P.; Conforti, M.L.; Guiducci, S.; Gay, R.E.; Michel, B.A.; Bruhlmann, P.; Muller-Ladner, U.; et al. Angiogenic and Angiostatic Factors in Systemic Sclerosis: Increased Levels of Vascular Endothelial Growth Factor Are a Feature of the Earliest Disease Stages and Are Associated with the Absence of Fingertip Ulcers. Arthritis Res. 2002, 4, R11. [Google Scholar] [CrossRef]
- Faller, D. V Endothelial Cell Responses to Hypoxic Stress. Clin. Exp. Pharmacol. Physiol. 1999, 26, 74–84. [Google Scholar] [CrossRef]
- Toyama, T.; Asano, Y.; Miyagawa, T.; Nakamura, K.; Hirabayashi, M.; Yamashita, T.; Saigusa, R.; Miura, S.; Ichimura, Y.; Takahashi, T.; et al. The Impact of Transcription Factor Fli1 Deficiency on the Regulation of Angiogenesis. Exp. Dermatol. 2017, 26, 912–918. [Google Scholar] [CrossRef]
- Noda, S.; Asano, Y.; Nishimura, S.; Taniguchi, T.; Fujiu, K.; Manabe, I.; Nakamura, K.; Yamashita, T.; Saigusa, R.; Akamata, K.; et al. Simultaneous Downregulation of KLF5 and Fli1 Is a Key Feature Underlying Systemic Sclerosis. Nat. Commun. 2014, 5, 5797. [Google Scholar] [CrossRef]
- Corallo, C.; Franci, B.; Lucani, B.; Montella, A.; Chirico, C.; Gonnelli, S.; Nuti, R.; Giordano, N. From Microvasculature to Fibroblasts: Contribution of Anti-Endothelial Cell Antibodies in Systemic Sclerosis. Int. J. Immunopathol. Pharmacol. 2015, 28, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Ingegnoli, F.; Gualtierotti, R.; Lubatti, C.; Bertolazzi, C.; Gutierrez, M.; Boracchi, P.; Fornili, M.; De Angelis, R. Nailfold Capillary Patterns in Healthy Subjects: A Real Issue in Capillaroscopy. Microvasc. Res. 2013, 90, 90–95. [Google Scholar] [CrossRef]
- Avouac, J.; Vallucci, M.; Smith, V.; Senet, P.; Ruiz, B.; Sulli, A.; Pizzorni, C.; Frances, C.; Chiocchia, G.; Cutolo, M.; et al. Correlations between Angiogenic Factors and Capillaroscopic Patterns in Systemic Sclerosis. Arthritis Res. Ther. 2013, 15, R55. [Google Scholar] [CrossRef]
- Smith, V.; Riccieri, V.; Pizzorni, C.; Decuman, S.; Deschepper, E.; Bonroy, C.; Sulli, A.; Piette, Y.; De Keyser, F.; Cutolo, M. Nailfold Capillaroscopy for Prediction of Novel Future Severe Organ Involvement in Systemic Sclerosis. J. Rheumatol. 2013, 40, 2023–2028. [Google Scholar] [CrossRef]
- Cutolo, M.; Cerinic, M.M. Nailfold Videocapillaroscopy for the Early Diagnosis of Systemic Sclerosis in Raynaud’s Phenomenon. Future Rheumatol. 2006, 1, 41–51. [Google Scholar] [CrossRef]
- Anghel, D.; Prioteasă, O.-G.; Nicolau, I.-N.; Bucurică, S.; Belinski, D.-O.; Popescu, G.-G.; Ghinescu, M.C.; Bobircă, A.; Groșeanu, M.-L.; Bojincă, V.-C. The Role of Nailfold Videocapillaroscopy in the Diagnosis and Monitoring of Interstitial Lung Disease Associated with Rheumatic Autoimmune Diseases. Diagnostics 2025, 15, 362. [Google Scholar] [CrossRef]
- Abou Ali, A.N.; Fittipaldi, A.; Rocha-Neves, J.; Ruaro, B.; Benedetto, F.; Al Ghadban, Z.; Simon, G.; Lepidi, S.; D’Oria, M. Clinical Applications of Contrast-Enhanced Ultrasound in Vascular Surgery: State-of-the-Art Narrative and Pictorial Review. JVS-Vasc. Insights 2025, 3, 100254. [Google Scholar] [CrossRef]
- Schinkel, A.F.L.; Kaspar, M.; Staub, D. Contrast-Enhanced Ultrasound: Clinical Applications in Patients with Atherosclerosis. Int. J. Cardiovasc. Imaging 2016, 32, 35–48. [Google Scholar] [CrossRef]
- Spaide, R.F.; Fujimoto, J.G.; Waheed, N.K.; Sadda, S.R.; Staurenghi, G. Optical Coherence Tomography Angiography. Prog. Retin. Eye Res. 2018, 64, 1–55. [Google Scholar] [CrossRef]
- Wang, S.; Aurora, A.B.; Johnson, B.A.; Qi, X.; McAnally, J.; Hill, J.A.; Richardson, J.A.; Bassel-Duby, R.; Olson, E.N. The Endothelial-Specific MicroRNA MiR-126 Governs Vascular Integrity and Angiogenesis. Dev. Cell 2008, 15, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Ha, M.; Kim, V.N. Regulation of MicroRNA Biogenesis. Nat. Rev. Mol. Cell Biol. 2014, 15, 509–524. [Google Scholar] [CrossRef] [PubMed]
- Nalbant, E.; Akkaya-Ulum, Y.Z. Exploring Regulatory Mechanisms on MiRNAs and Their Implications in Inflammation-Related Diseases. Clin. Exp. Med. 2024, 24, 142. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Romero, I.A.; Guerra-Calderas, L.; Salgado-Albarrán, M.; Maldonado-Huerta, T.; Soto-Reyes, E. The Regulatory Roles of Non-Coding RNAs in Angiogenesis and Neovascularization from an Epigenetic Perspective. Front. Oncol. 2019, 9, 1091. [Google Scholar] [CrossRef]
- Fahs, F.; Bi, X.; Yu, F.S.; Zhou, L.; Mi, Q.S. New Insights into MicroRNAs in Skin Wound Healing. IUBMB Life 2015, 67, 889–896. [Google Scholar] [CrossRef]
- Li, Y.; Huang, J.; Guo, M.; Zuo, X. MicroRNAs Regulating Signaling Pathways: Potential Biomarkers in Systemic Sclerosis. Genom. Proteom. Bioinform. 2015, 13, 234–241. [Google Scholar] [CrossRef]
- Suarez, Y.; Fernandez-Hernando, C.; Yu, J.; Gerber, S.A.; Harrison, K.D.; Pober, J.S.; Iruela-Arispe, M.L.; Merkenschlager, M.; Sessa, W.C. Dicer-Dependent Endothelial MicroRNAs Are Necessary for Postnatal Angiogenesis. Proc. Natl. Acad. Sci. USA 2008, 105, 14082–14087. [Google Scholar] [CrossRef]
- Kuehbacher, A.; Urbich, C.; Zeiher, A.M.; Dimmeler, S. Role of Dicer and Drosha for Endothelial MicroRNA Expression and Angiogenesis. Circ. Res. 2007, 101, 59–68. [Google Scholar] [CrossRef]
- Fish, J.E.; Santoro, M.M.; Morton, S.U.; Yu, S.; Yeh, R.-F.; Wythe, J.D.; Ivey, K.N.; Bruneau, B.G.; Stainier, D.Y.R.; Srivastava, D. MiR-126 Regulates Angiogenic Signaling and Vascular Integrity. Dev. Cell 2008, 15, 272–284. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Gorski, D.H. Regulation of Angiogenesis Through a MicroRNA (MiR-130a) That down-Regulates Antiangiogenic Homeobox Genes GAX and HOXA5. Blood 2008, 111, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Fasanaro, P.; D’Alessandra, Y.; Di Stefano, V.; Melchionna, R.; Romani, S.; Pompilio, G.; Capogrossi, M.C.; Martelli, F. MicroRNA-210 Modulates Endothelial Cell Response to Hypoxia and Inhibits the Receptor Tyrosine Kinase Ligand Ephrin-A3. J. Biol. Chem. 2008, 283, 15878–15883. [Google Scholar] [CrossRef]
- Wurdinger, T.; Tannous, B.A.; Saydam, O.; Skog, J.; Grau, S.; Soutschek, J.; Weissleder, R.; Breakefield, X.O.; Krichevsky, A.M. MiR-296 Regulates Growth Factor Receptor Overexpression in Angiogenic Endothelial Cells. Cancer Cell 2008, 14, 382–393. [Google Scholar] [CrossRef]
- Lee, D.Y.; Deng, Z.; Wang, C.-H.; Yang, B.B. MicroRNA-378 Promotes Cell Survival, Tumor Growth, and Angiogenesis by Targeting SuFu and Fus-1 Expression. Proc. Natl. Acad. Sci. USA 2007, 104, 20350–20355. [Google Scholar] [CrossRef]
- Bonauer, A.; Carmona, G.; Iwasaki, M.; Mione, M.; Koyanagi, M.; Fischer, A.; Burchfield, J.; Fox, H.; Doebele, C.; Ohtani, K.; et al. MicroRNA-92a Controls Angiogenesis and Functional Recovery of Ischemic Tissues in Mice. Science 2009, 324, 1710–1713. [Google Scholar] [CrossRef]
- Doebele, C.; Bonauer, A.; Fischer, A.; Scholz, A.; Reiss, Y.; Urbich, C.; Hofmann, W.-K.; Zeiher, A.M.; Dimmeler, S. Members of the MicroRNA-17-92 Cluster Exhibit a Cell-Intrinsic Antiangiogenic Function in Endothelial Cells. Blood 2010, 115, 4944–4950. [Google Scholar] [CrossRef]
- Hua, Z.; Lv, Q.; Ye, W.; Wong, C.-K.A.; Cai, G.; Gu, D.; Ji, Y.; Zhao, C.; Wang, J.; Yang, B.B.; et al. MiRNA-Directed Regulation of VEGF and Other Angiogenic Factors under Hypoxia. PLoS ONE 2006, 1, e116. [Google Scholar] [CrossRef]
- Wang, X.H.; Qian, R.Z.; Zhang, W.; Chen, S.F.; Jin, H.M.; Hu, R.M. MicroRNA-320 Expression in Myocardial Microvascular Endothelial Cells and Its Relationship with Insulin-like Growth Factor-1 in Type 2 Diabetic Rats. Clin. Exp. Pharmacol. Physiol. 2009, 36, 181–188. [Google Scholar] [CrossRef]
- Poliseno, L.; Tuccoli, A.; Mariani, L.; Evangelista, M.; Citti, L.; Woods, K.; Mercatanti, A.; Hammond, S.; Rainaldi, G. MicroRNAs Modulate the Angiogenic Properties of HUVECs. Blood 2006, 108, 3068–3071. [Google Scholar] [CrossRef]
- Landskroner-Eiger, S.; Moneke, I.; Sessa, W.C. MiRNAs as Modulators of Angiogenesis. Cold Spring Harb. Perspect. Med. 2013, 3, a006643. [Google Scholar] [CrossRef] [PubMed]
- Suárez, Y.; Sessa, W.C. MicroRNAs as Novel Regulators of Angiogenesis. Circ. Res. 2009, 104, 442–454. [Google Scholar] [CrossRef] [PubMed]
- Staszel, T.; Zapala, B.; Polus, A.; Sadakierska-Chudy, A.; Kiec-Wilk, B.; Stepien, E.; Wybranska, I.; Chojnacka, M.; Dembinska-Kiec, A. Role of MicroRNAs in Endothelial Cell Pathophysiology. Pol. Arch. Med. Wewn. 2011, 121, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, M.; Zheng, M.; Hayashi, M.; Lee, J.-D.; Yoshino, O.; Lin, S.; Han, J. Impaired MicroRNA Processing Causes Corpus Luteum Insufficiency and Infertility in Mice. J. Clin. Investig. 2008, 118, 1944–1954. [Google Scholar] [CrossRef]
- Xie, M.; Dart, D.A.; Guo, T.; Xing, X.-F.; Cheng, X.-J.; Du, H.; Jiang, W.G.; Wen, X.-Z.; Ji, J.-F. MicroRNA-1 Acts as a Tumor Suppressor MicroRNA by Inhibiting Angiogenesis-Related Growth Factors in Human Gastric Cancer. Gastric Cancer 2018, 21, 41–54. [Google Scholar] [CrossRef]
- Fang, Y.; Shi, C.; Manduchi, E.; Civelek, M.; Davies, P.F. MicroRNA-10a Regulation of Proinflammatory Phenotype in Athero-Susceptible Endothelium in Vivo and in Vitro. Proc. Natl. Acad. Sci. USA 2010, 107, 13450–13455. [Google Scholar] [CrossRef]
- Nikolic, I.; Plate, K.-H.; Schmidt, M.H. EGFL7 Meets MiRNA-126: An Angiogenesis Alliance. J. Angiogenes Res. 2010, 2, 9. [Google Scholar] [CrossRef]
- Harris, T.A.; Yamakuchi, M.; Ferlito, M.; Mendell, J.T.; Lowenstein, C.J. MicroRNA-126 Regulates Endothelial Expression of Vascular Cell Adhesion Molecule 1. Proc. Natl. Acad. Sci. USA 2008, 105, 1516–1521. [Google Scholar] [CrossRef]
- Zomorrod, M.S.; Kouhkan, F.; Soleimani, M.; Aliyan, A.; Tasharrofi, N. Overexpression of MiR-133 Decrease Primary Endothelial Cells Proliferation and Migration via FGFR1 Targeting. Exp. Cell Res. 2018, 369, 11–16. [Google Scholar] [CrossRef]
- Yu, B.T.; Yu, N.; Wang, Y.; Zhang, H.; Wan, K.; Sun, X.; Zhang, C.S. Role of MiR-133a in Regulating TGF-Β1 Signaling Pathway in Myocardial Fibrosis after Acute Myocardial Infarction in Rats. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 8588–8597. [Google Scholar]
- Vasa-Nicotera, M.; Chen, H.; Tucci, P.; Yang, A.L.; Saintigny, G.; Menghini, R.; Mahe, C.; Agostini, M.; Knight, R.A.; Melino, G.; et al. MiR-146a Is Modulated in Human Endothelial Cell with Aging. Atherosclerosis 2011, 217, 326–330. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, R.M.; Taganov, K.D.; Boldin, M.P.; Cheng, G.; Baltimore, D. MicroRNA-155 Is Induced During the Macrophage Inflammatory Response. Proc. Natl. Acad. Sci. USA 2007, 104, 1604–1609. [Google Scholar] [CrossRef] [PubMed]
- Christmann, R.B.; Wooten, A.; Sampaio-Barros, P.; Borges, C.L.; Carvalho, C.R.R.; Kairalla, R.A.; Feghali-Bostwick, C.; Ziemek, J.; Mei, Y.; Goummih, S.; et al. MiR-155 in the Progression of Lung Fibrosis in Systemic Sclerosis. Arthritis Res. Ther. 2016, 18, 155. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, Z.; Xiao-qin; Hu, H.; Tian, S.; Lu, Z.; Zhang, T.; Bai, Y. Down-Regulation of MicroRNA-155 Attenuates Retinal Neovascularization via the PI3K/Akt Pathway. Mol. Vis. 2015, 21, 1173–1184. [Google Scholar]
- Meng, Y.-C.; Ding, Z.-Y.; Wang, H.-Q.; Ning, L.-P.; Wang, C. Effect of MicroRNA-155 on Angiogenesis after Cerebral Infarction of Rats Through AT1R/VEGFR2 Pathway. Asian Pac. J. Trop. Med. 2015, 8, 829–835. [Google Scholar] [CrossRef]
- Shan, S.W.; Lee, D.Y.; Deng, Z.; Shatseva, T.; Jeyapalan, Z.; Du, W.W.; Zhang, Y.; Xuan, J.W.; Yee, S.-P.; Siragam, V.; et al. MicroRNA MiR-17 Retards Tissue Growth and Represses Fibronectin Expression. Nat. Cell Biol. 2009, 11, 1031–1038. [Google Scholar] [CrossRef]
- Yin, R.; Wang, R.; Guo, L.; Zhang, W.; Lu, Y. MiR-17-3p Inhibits Angiogenesis by Downregulating Flk-1 in the Cell Growth Signal Pathway. J. Vasc. Res. 2013, 50, 157–166. [Google Scholar] [CrossRef]
- Fontana, L.; Pelosi, E.; Greco, P.; Racanicchi, S.; Testa, U.; Liuzzi, F.; Croce, C.M.; Brunetti, E.; Grignani, F.; Peschle, C. MicroRNAs 17-5p-20a-106a Control Monocytopoiesis Through AML1 Targeting and M-CSF Receptor Upregulation. Nat. Cell Biol. 2007, 9, 775–787. [Google Scholar] [CrossRef]
- Kuhnert, F.; Kuo, C.J. MiR-17-92 Angiogenesis Micromanagement. Blood 2010, 115, 4631–4633. [Google Scholar] [CrossRef]
- Guan, J.-T.; Li, X.-X.; Peng, D.-W.; Zhang, W.-M.; Qu, J.; Lu, F.; D’Amato, R.J.; Chi, Z.-L. MicroRNA-18a-5p Administration Suppresses Retinal Neovascularization by Targeting FGF1 and HIF1A. Front. Pharmacol. 2020, 11, 276. [Google Scholar] [CrossRef]
- Qin, X.; Wang, X.; Wang, Y.; Tang, Z.; Cui, Q.; Xi, J.; Li, Y.-S.J.; Chien, S.; Wang, N. MicroRNA-19a Mediates the Suppressive Effect of Laminar Flow on Cyclin D1 Expression in Human Umbilical Vein Endothelial Cells. Proc. Natl. Acad. Sci. USA 2010, 107, 3240–3244. [Google Scholar] [CrossRef] [PubMed]
- Correia, A.C.P.; Moonen, J.-R.A.J.; Brinker, M.G.L.; Krenning, G. FGF2 Inhibits Endothelial–Mesenchymal Transition Through MicroRNA-20a-Mediated Repression of Canonical TGF-β Signaling. J. Cell Sci. 2016, 129, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Luo, H.; Li, Y.; Zhou, Y.; Jiang, Y.; Chai, J.; Xiao, X.; You, Y.; Zuo, X. MicroRNA-21 in Scleroderma Fibrosis and Its Function in TGF-β- Regulated Fibrosis-Related Genes Expression. J. Clin. Immunol. 2013, 33, 1100–1109. [Google Scholar] [CrossRef] [PubMed]
- Lou, Y.-L.; Guo, F.; Liu, F.; Gao, F.-L.; Zhang, P.-Q.; Niu, X.; Guo, S.-C.; Yin, J.-H.; Wang, Y.; Deng, Z.-F. MiR-210 Activates Notch Signaling Pathway in Angiogenesis Induced by Cerebral Ischemia. Mol. Cell. Biochem. 2012, 370, 45–51. [Google Scholar] [CrossRef]
- Menghini, R.; Casagrande, V.; Cardellini, M.; Martelli, E.; Terrinoni, A.; Amati, F.; Vasa-Nicotera, M.; Ippoliti, A.; Novelli, G.; Melino, G.; et al. MicroRNA 217 Modulates Endothelial Cell Senescence via Silent Information Regulator 1. Circulation 2009, 120, 1524–1532. [Google Scholar] [CrossRef]
- Zhang, X.; Dong, J.; He, Y.; Zhao, M.; Liu, Z.; Wang, N.; Jiang, M.; Zhang, Z.; Liu, G.; Liu, H.; et al. MiR-218 Inhibited Tumor Angiogenesis by Targeting ROBO1 in Gastric Cancer. Gene 2017, 615, 42–49. [Google Scholar] [CrossRef]
- Yamakuchi, M.; Yagi, S.; Ito, T.; Lowenstein, C.J. MicroRNA-22 Regulates Hypoxia Signaling in Colon Cancer Cells. PLoS ONE 2011, 6, e20291. [Google Scholar] [CrossRef]
- Celic, T.; Metzinger-Le Meuth, V.; Six, I.; Massy, Z.A.; Metzinger, L. The Mir-221/222 Cluster Is a Key Player in Vascular Biology via the Fine-Tuning of Endothelial Cell Physiology. Curr. Vasc. Pharmacol. 2017, 15, 40–46. [Google Scholar] [CrossRef]
- Liu, X.; Cheng, Y.; Zhang, S.; Lin, Y.; Yang, J.; Zhang, C. A Necessary Role of MiR-221 and MiR-222 in Vascular Smooth Muscle Cell Proliferation and Neointimal Hyperplasia. Circ. Res. 2009, 104, 476–487. [Google Scholar] [CrossRef]
- Dentelli, P.; Rosso, A.; Orso, F.; Olgasi, C.; Taverna, D.; Brizzi, M.F. MicroRNA-222 Controls Neovascularization by Regulating Signal Transducer and Activator of Transcription 5A Expression. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 1562–1568. [Google Scholar] [CrossRef]
- Hsu, Y.-L.; Hung, J.-Y.; Chang, W.-A.; Lin, Y.-S.; Pan, Y.-C.; Tsai, P.-H.; Wu, C.-Y.; Kuo, P.-L. Hypoxic Lung Cancer-Secreted Exosomal MiR-23a Increased Angiogenesis and Vascular Permeability by Targeting Prolyl Hydroxylase and Tight Junction Protein ZO-1. Oncogene 2017, 36, 4929–4942. [Google Scholar] [CrossRef]
- Urbich, C.; Kaluza, D.; Frömel, T.; Knau, A.; Bennewitz, K.; Boon, R.A.; Bonauer, A.; Doebele, C.; Boeckel, J.-N.; Hergenreider, E.; et al. MicroRNA-27a/b Controls Endothelial Cell Repulsion and Angiogenesis by Targeting Semaphorin 6A. Blood 2012, 119, 1607–1616. [Google Scholar] [CrossRef]
- Zhong, L.; Simard, M.J.; Huot, J. Endothelial MicroRNAs Regulating the NF-ΚB Pathway and Cell Adhesion Molecules During Inflammation. FASEB J. 2018, 32, 4070–4084. [Google Scholar] [CrossRef]
- Ito, T.; Yagi, S.; Yamakuchi, M. MicroRNA-34a Regulation of Endothelial Senescence. Biochem. Biophys. Res. Commun. 2010, 398, 735–740. [Google Scholar] [CrossRef] [PubMed]
- Yamakuchi, M.; Ferlito, M.; Lowenstein, C.J. MiR-34a Repression of SIRT1 Regulates Apoptosis. Proc. Natl. Acad. Sci. USA 2008, 105, 13421–13426. [Google Scholar] [CrossRef] [PubMed]
- Tabuchi, T.; Satoh, M.; Itoh, T.; Nakamura, M. MicroRNA-34a Regulates the Longevity-Associated Protein SIRT1 in Coronary Artery Disease: Effect of Statins on SIRT1 and MicroRNA-34a Expression. Clin. Sci. 2012, 123, 161–171. [Google Scholar] [CrossRef]
- Fan, W.; Fang, R.; Wu, X.; Liu, J.; Feng, M.; Dai, G.; Chen, G.; Wu, G. Shear-Sensitive MicroRNA-34a Modulates Flow-Dependent Regulation of Endothelial Inflammation. J. Cell Sci. 2015, 128, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Xu, W.W.; Han, L.; Chan, K.T.; Tsao, S.W.; Lee, N.P.Y.; Law, S.; Xu, L.Y.; Li, E.M.; Chan, K.W.; et al. MicroRNA-377 Suppresses Initiation and Progression of Esophageal Cancer by Inhibiting CD133 and VEGF. Oncogene 2017, 36, 3986–4000. [Google Scholar] [CrossRef]
- Ghosh, G.; Subramanian, I.V.; Adhikari, N.; Zhang, X.; Joshi, H.P.; Basi, D.; Chandrashekhar, Y.S.; Hall, J.L.; Roy, S.; Zeng, Y.; et al. Hypoxia-Induced MicroRNA-424 Expression in Human Endothelial Cells Regulates HIF-α Isoforms and Promotes Angiogenesis. J. Clin. Investig. 2010, 120, 4141–4154. [Google Scholar] [CrossRef]
- Zhang, T.; Jing, L.; Li, H.; Ding, L.; Ai, D.; Lyu, J.; Zhong, L. MicroRNA-4530 Promotes Angiogenesis by Targeting VASH1 in Breast Carcinoma Cells. Oncol. Lett. 2017, 14, 111–118. [Google Scholar] [CrossRef]
- Sing, T.; Jinnin, M.; Yamane, K.; Honda, N.; Makino, K.; Kajihara, I.; Makino, T.; Sakai, K.; Masuguchi, S.; Fukushima, S.; et al. MicroRNA-92a Expression in the Sera and Dermal Fibroblasts Increases in Patients with Scleroderma. Rheumatology 2012, 51, 1550–1556. [Google Scholar] [CrossRef]
- Daniel, J.-M.; Penzkofer, D.; Teske, R.; Dutzmann, J.; Koch, A.; Bielenberg, W.; Bonauer, A.; Boon, R.A.; Fischer, A.; Bauersachs, J.; et al. Inhibition of MiR-92a Improves Re-Endothelialization and Prevents Neointima Formation Following Vascular Injury. Cardiovasc. Res. 2014, 103, 564–572. [Google Scholar] [CrossRef]
- Fang, Y.; Davies, P.F. Site-Specific MicroRNA-92a Regulation of Kruppel-like Factors 4 and 2 in Atherosusceptible Endothelium. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Hong, Q.; Wang, Y.; Hou, K.; Wang, L.; Zhang, Y.; Fu, B.; Zhou, Y.; Zheng, W.; Chen, X.; et al. High Concentrations of Uric Acid Inhibit Angiogenesis via Regulation of the Krüppel-like Factor 2-Vascular Endothelial Growth Factor-A Axis by MiR-92a. Circ. J. 2015, 79, 2487–2498. [Google Scholar] [CrossRef] [PubMed]
- Wade, S.M.; Ohnesorge, N.; McLoughlin, H.; Biniecka, M.; Carter, S.P.; Trenkman, M.; Cunningham, C.C.; McGarry, T.; Canavan, M.; Kennedy, B.N.; et al. Dysregulated MiR-125a Promotes Angiogenesis through Enhanced Glycolysis. EBioMedicine 2019, 47, 402–413. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Nazari-Jahantigh, M.; Neth, P.; Weber, C.; Schober, A. MicroRNA-126, -145, and -155: A Therapeutic Triad in Atherosclerosis? Arterioscler. Thromb. Vasc. Biol. 2013, 33, 449–454. [Google Scholar] [CrossRef]
- Guenther, S.P.W.; Schrepfer, S. MiR-126: A Potential New Key Player in Hypoxia and Reperfusion? Ann. Transl. Med. 2016, 4, 377. [Google Scholar] [CrossRef]
- van Solingen, C.; Bijkerk, R.; de Boer, H.C.; Rabelink, T.J.; van Zonneveld, A.J. The Role of MicroRNA-126 in Vascular Homeostasis. Curr. Vasc. Pharmacol. 2015, 13, 341–351. [Google Scholar] [CrossRef]
- Iwamoto, N.; Vettori, S.; Maurer, B.; Brock, M.; Pachera, E.; Jüngel, A.; Calcagni, M.; Gay, R.E.; Whitfield, M.L.; Distler, J.H.W.; et al. Downregulation of MiR-193b in Systemic Sclerosis Regulates the Proliferative Vasculopathy by Urokinase-Type Plasminogen Activator Expression. Ann. Rheum. Dis. 2015, 75, 303–310. [Google Scholar] [CrossRef]
- Yang, C.; Tahiri, H.; Cai, C.; Gu, M.; Gagnon, C.; Hardy, P. MicroRNA-181a Inhibits Ocular Neovascularization by Interfering with Vascular Endothelial Growth Factor Expression. Cardiovasc. Ther. 2018, 36, e12329. [Google Scholar] [CrossRef]
- Li, Y.; Kuscu, C.; Banach, A.; Zhang, Q.; Pulkoski-Gross, A.; Kim, D.; Liu, J.; Roth, E.; Li, E.; Shroyer, K.R.; et al. MiR-181a-5p Inhibits Cancer Cell Migration and Angiogenesis via Downregulation of Matrix Metalloproteinase-14. Cancer Res. 2015, 75, 2674–2685. [Google Scholar] [CrossRef]
- Sun, W.; Wang, X.; Li, J.; You, C.; Lu, P.; Feng, H.; Kong, Y.; Zhang, H.; Liu, Y.; Jiao, R.; et al. MicroRNA-181a Promotes Angiogenesis in Colorectal Cancer by Targeting SRCIN1 to Promote the SRC/VEGF Signaling Pathway. Cell Death Dis. 2018, 9, 438. [Google Scholar] [CrossRef]
- Magenta, A.; Cencioni, C.; Fasanaro, P.; Zaccagnini, G.; Greco, S.; Sarra-Ferraris, G.; Antonini, A.; Martelli, F.; Capogrossi, M.C. MiR-200c Is Upregulated by Oxidative Stress and Induces Endothelial Cell Apoptosis and Senescence via ZEB1 Inhibition. Cell Death Differ. 2011, 18, 1628–1639. [Google Scholar] [CrossRef]
- Huang, J.; Wang, Y.; Guo, Y.; Sun, S. Down-Regulated MicroRNA-152 Induces Aberrant DNA Methylation in Hepatitis B Virus-Related Hepatocellular Carcinoma by Targeting DNA Methyltransferase 1. Hepatology 2010, 52, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Suto, A.; Ikeda, K.; Sanayama, Y.; Nakagomi, D.; Iwamoto, T.; Suzuki, K.; Kambe, N.; Matsue, H.; Matsumura, R.; et al. Alteration of Circulating MiRNAs in SSc: MiR-30b Regulates the Expression of PDGF Receptor Beta. Rheumatology 2013, 52, 1963–1972. [Google Scholar] [CrossRef] [PubMed]
- Howe, G.A.; Kazda, K.; Addison, C.L. MicroRNA-30b Controls Endothelial Cell Capillary Morphogenesis Through Regulation of Transforming Growth Factor Beta 2. PLoS ONE 2017, 12, e0185619. [Google Scholar] [CrossRef] [PubMed]
- Urbich, C.; Kuehbacher, A.; Dimmeler, S. Role of MicroRNAs in Vascular Diseases, Inflammation, and Angiogenesis. Cardiovasc. Res. 2008, 79, 581–588. [Google Scholar] [CrossRef]
- Woźniak, O.; Mierzejewski, B.; Brzoska, E. MicroRNA-126: A Key Regulator of Angiogenesis, Inflammation, and Tumorigenesis—Exploring Its Multifaceted Functions in Vascular Health and Cancer. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2025, 1871, 167984. [Google Scholar] [CrossRef]
- Zhou, Q.; Anderson, C.; Hanus, J.; Zhao, F.; Ma, J.; Yoshimura, A.; Wang, S. Strand and Cell Type-Specific Function of MicroRNA-126 in Angiogenesis. Mol. Ther. 2016, 24, 1823–1835. [Google Scholar] [CrossRef]
- Wang, J.-M.; Xu, W.-D.; Yuan, Z.-C.; Wu, Q.; Zhou, J.; Huang, A.-F. Serum Levels and Gene Polymorphisms of Angiopoietin 2 in Systemic Lupus Erythematosus Patients. Sci. Rep. 2021, 11, 10. [Google Scholar] [CrossRef]
- Enteshari-Moghadam, A.; Fouladi, N.; Pordel, S.; Jeddi, F.; Asghariazar, V.; Eterafi, M.; Safarzadeh, E. Evaluation of the MiRNA-126 and VCAM-1 in Scleroderma Patients and Its Association with Clinical Characteristics. Am. J. Med. Sci. 2025, 369, 339–345. [Google Scholar] [CrossRef]
- Wajda, A.; Walczyk, M.; Dudek, E.; Stypińska, B.; Lewandowska, A.; Romanowska-Próchnicka, K.; Chojnowski, M.; Olesińska, M.; Paradowska-Gorycka, A. Serum MicroRNAs in Systemic Sclerosis, Associations with Digital Vasculopathy and Lung Involvement. Int. J. Mol. Sci. 2022, 23, 10731. [Google Scholar] [CrossRef]
- Liakouli, V.; Cipriani, P.; Di Benedetto, P.; Panzera, N.; Ruscitti, P.; Pantano, I.; Berardicurti, O.; Carubbi, F.; Esteves, F.; Mavria, G.; et al. Epidermal Growth Factor Like-Domain 7 and MiR-126 Are Abnormally Expressed in Diffuse Systemic Sclerosis Fibroblasts. Sci. Rep. 2019, 9, 4589. [Google Scholar] [CrossRef]
- Guo, B.; Gu, J.; Zhuang, T.; Zhang, J.; Fan, C.; Li, Y.; Zhao, M.; Chen, R.; Wang, R.; Kong, Y.; et al. MicroRNA-126: From Biology to Therapeutics. Biomed. Pharmacother. 2025, 185, 117953. [Google Scholar] [CrossRef] [PubMed]
- Miscianinov, V.; Martello, A.; Rose, L.; Parish, E.; Cathcart, B.; Mitic, T.; Gray, G.A.; Meloni, M.; Al Haj Zen, A.; Caporali, A. MicroRNA-148b Targets the TGF-Beta Pathway to Regulate Angiogenesis and Endothelial-to-Mesenchymal Transition During Skin Wound Healing. Mol. Ther. 2018, 26, 1996–2007. [Google Scholar] [CrossRef] [PubMed]
- Zhu, N.; Zhang, D.; Chen, S.; Liu, X.; Lin, L.; Huang, X.; Guo, Z.; Liu, J.; Wang, Y.; Yuan, W.; et al. Endothelial Enriched MicroRNAs Regulate Angiotensin II-Induced Endothelial Inflammation and Migration. Atherosclerosis. 2011, 215, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Ceolotto, G.; Papparella, I.; Bortoluzzi, A.; Strapazzon, G.; Ragazzo, F.; Bratti, P.; Fabricio, A.S.C.; Squarcina, E.; Gion, M.; Palatini, P.; et al. Interplay Between MiR-155, AT1R A1166C Polymorphism, and AT1R Expression in Young Untreated Hypertensives. Am. J. Hypertens. 2011, 24, 241–246. [Google Scholar] [CrossRef]
- Tkachuk, V.A.; Plekhanova, O.S.; Parfyonova, Y. V Regulation of Arterial Remodeling and Angiogenesis by Urokinase-Type Plasminogen Activator. Can. J. Physiol. Pharmacol. 2009, 87, 231–251. [Google Scholar] [CrossRef]
- Manetti, M.; Rosa, I.; Milia, A.F.; Guiducci, S.; Carmeliet, P.; Ibba-Manneschi, L.; Matucci-Cerinic, M. Inactivation of Urokinase-Type Plasminogen Activator Receptor (UPAR) Gene Induces Dermal and Pulmonary Fibrosis and Peripheral Microvasculopathy in Mice: A New Model of Experimental Scleroderma? Ann. Rheum. Dis. 2014, 73, 1700–1709. [Google Scholar] [CrossRef]
- van Mil, A.; Grundmann, S.; Goumans, M.-J.; Lei, Z.; Oerlemans, M.I.; Jaksani, S.; Doevendans, P.A.; Sluijter, J.P.G. MicroRNA-214 Inhibits Angiogenesis by Targeting Quaking and Reducing Angiogenic Growth Factor Release. Cardiovasc. Res. 2012, 93, 655–665. [Google Scholar] [CrossRef]
- Suarez, Y.; Fernandez-Hernando, C.; Pober, J.S.; Sessa, W.C. Dicer Dependent MicroRNAs Regulate Gene Expression and Functions in Human Endothelial Cells. Circ. Res. 2007, 100, 1164–1173. [Google Scholar] [CrossRef] [PubMed]
- Chistiakov, D.A.; Sobenin, I.A.; Orekhov, A.N.; Bobryshev, Y. V Human MiR-221/222 in Physiological and Atherosclerotic Vascular Remodeling. BioMed Res. Int. 2015, 2015, 354517. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, A.; Yanagisawa, K.; Tanaka, M.; Cao, K.; Matsuyama, Y.; Goto, H.; Takahashi, T. Identification of Hypoxia-Inducible Factor-1 Alpha as a Novel Target for MiR-17-92 MicroRNA Cluster. Cancer Res. 2008, 68, 5540–5545. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-H.; Lee, D.Y.; Deng, Z.; Jeyapalan, Z.; Lee, S.-C.; Kahai, S.; Lu, W.-Y.; Zhang, Y.; Yang, B.B. MicroRNA MiR-328 Regulates Zonation Morphogenesis by Targeting CD44 Expression. PLoS ONE 2008, 3, e2420. [Google Scholar] [CrossRef]
- Raitoharju, E.; Lyytikäinen, L.-P.; Levula, M.; Oksala, N.; Mennander, A.; Tarkka, M.; Klopp, N.; Illig, T.; Kähönen, M.; Karhunen, P.J.; et al. MiR-21, MiR-210, MiR-34a, and MiR-146a/b Are up-Regulated in Human Atherosclerotic Plaques in the Tampere Vascular Study. Atherosclerosis 2011, 219, 211–217. [Google Scholar] [CrossRef]
- Ge, X.; Huang, S.; Gao, H.; Han, Z.; Chen, F.; Zhang, S.; Wang, Z.; Kang, C.; Jiang, R.; Yue, S.; et al. MiR-21-5p Alleviates Leakage of Injured Brain Microvascular Endothelial Barrier in Vitro Through Suppressing Inflammation and Apoptosis. Brain Res. 2016, 1650, 31–40. [Google Scholar] [CrossRef]
- Condrat, C.E.; Thompson, D.C.; Barbu, M.G.; Bugnar, O.L.; Boboc, A.; Cretoiu, D.; Suciu, N.; Cretoiu, S.M.; Voinea, S.C. MiRNAs as Biomarkers in Disease: Latest Findings Regarding Their Role in Diagnosis and Prognosis. Cells 2020, 9, 276. [Google Scholar] [CrossRef]
- Chen, X.; Ba, Y.; Ma, L.; Cai, X.; Yin, Y.; Wang, K.; Guo, J.; Zhang, Y.; Chen, J.; Guo, X.; et al. Characterization of MicroRNAs in Serum: A Novel Class of Biomarkers for Diagnosis of Cancer and Other Diseases. Cell Res. 2008, 18, 997–1006. [Google Scholar] [CrossRef]
- Kumar, M.A.; Baba, S.K.; Sadida, H.Q.; Marzooqi, S.A.; Jerobin, J.; Altemani, F.H.; Algehainy, N.; Alanazi, M.A.; Abou-Samra, A.-B.; Kumar, R.; et al. Extracellular Vesicles as Tools and Targets in Therapy for Diseases. Signal Transduct. Target. Ther. 2024, 9, 27. [Google Scholar] [CrossRef]
- Lei, X.; Ring, S.; Jin, S.; Singh, S.; Mahnke, K. Extracellular Vesicles and Their Role in Skin Inflammatory Diseases: From Pathogenesis to Therapy. Int. J. Mol. Sci. 2025, 26, 3827. [Google Scholar] [CrossRef]
- Kalluri, R.; LeBleu, V.S. The Biology, Function, and Biomedical Applications of Exosomes. Science 2020, 367, eaau6977. [Google Scholar] [CrossRef]
- Makino, K.; Jinnin, M.; Kajihara, I.; Honda, N.; Sakai, K.; Masuguchi, S.; Fukushima, S.; Inoue, Y.; Ihn, H. Circulating MiR-142-3p Levels in Patients with Systemic Sclerosis. Clin. Exp. Dermatol. 2012, 37, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Chistiakov, D.A.; Orekhov, A.N.; Bobryshev, Y.V. The Role of MiR-126 in Embryonic Angiogenesis, Adult Vascular Homeostasis, and Vascular Repair and Its Alterations in Atherosclerotic Disease. J. Mol. Cell. Cardiol. 2016, 97, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Hernández-López, J.R.; Flores-García, M.; García-Flores, E.; Cazarín-Santos, B.G.; Peña-Duque, M.A.; Sánchez-Muñoz, F.; Ballinas-Verdugo, M.A.; Delgadillo-Rodríguez, H.; Martínez-Ríos, M.A.; Angles-Cano, E.; et al. Circulating Microvesicles Enriched in MiR–126–5p and MiR–223–3p: Potential Biomarkers in Acute Coronary Syndrome. Biomedicines 2025, 13, 510. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; He, X.; Liu, M.; Yun, L.; Cong, B. Diagnostic Value of Cardiac MiR-126-5p, MiR-134-5p, and MiR-499a-5p in Coronary Artery Disease-Induced Sudden Cardiac Death. Front. Cardiovasc. Med. 2022, 9, 944317. [Google Scholar] [CrossRef]
- Szabo, I.; Muntean, L.; Crisan, T.; Rednic, V.; Sirbe, C.; Rednic, S. Novel Concepts in Systemic Sclerosis Pathogenesis: Role for MiRNAs. Biomedicines 2021, 9, 1471. [Google Scholar] [CrossRef]
- Chirshev, E.; Oberg, K.C.; Ioffe, Y.J.; Unternaehrer, J.J. Let-7 as Biomarker, Prognostic Indicator, and Therapy for Precision Medicine in Cancer. Clin. Transl. Med. 2019, 8, e24. [Google Scholar] [CrossRef]
- Mahesh, G.; Biswas, R. MicroRNA-155: A Master Regulator of Inflammation. J. Interferon Cytokine Res. 2019, 39, 321–330. [Google Scholar] [CrossRef]
- Zapata-Martínez, L.; Águila, S.; de los Reyes-García, A.M.; Carrillo-Tornel, S.; Lozano, M.L.; González-Conejero, R.; Martínez, C. Inflammatory MicroRNAs in Cardiovascular Pathology: Another Brick in the Wall. Front. Immunol. 2023, 14, 1196104. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, M.; Qin, G.; Weintraub, N.L.; Tang, Y. MiR-92a Regulates Viability and Angiogenesis of Endothelial Cells under Oxidative Stress. Biochem. Biophys. Res. Commun. 2014, 446, 952–958. [Google Scholar] [CrossRef]
- Chang, Y.-J.; Li, Y.-S.; Wu, C.-C.; Wang, K.-C.; Huang, T.-C.; Chen, Z.; Chien, S. Extracellular MicroRNA-92a Mediates Endothelial Cell–Macrophage Communication. Arterioscler. Thromb. Vasc. Biol. 2019, 39, 2492–2504. [Google Scholar] [CrossRef]
- Wronska, A. The Role of MicroRNA in the Development, Diagnosis, and Treatment of Cardiovascular Disease: Recent Developments. J. Pharmacol. Exp. Ther. 2023, 384, 123–132. [Google Scholar] [CrossRef]
- Al-Gburi, S.; Moinzadeh, P.; Krieg, T. Pathophysiology in Systemic Sclerosis: Current Insights and Future Perspectives. Sclerosis 2025, 3, 17. [Google Scholar] [CrossRef]
- Santos, C.S.; Galdo, F. Del New Horizons in Systemic Sclerosis Treatment: Advances and Emerging Therapies in 2025. RMD Open 2025, 11, e005776. [Google Scholar] [CrossRef] [PubMed]
- Izumiya, Y.; Jinnn, M.; Kimura, Y.; Wang, Z.; Onoue, Y.; Hanatani, S.; Araki, S.; Ihn, H.; Ogawa, H. Expression of Let-7 Family MicroRNAs in Skin Correlates Negatively with Severity of Pulmonary Hypertension in Patients with Systemic Scleroderma. IJC Heart Vasc. 2015, 8, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wu, H.; Zhao, M.; Lu, Q. Meta-Analysis of Differentially Expressed MicroRNAs in Systemic Sclerosis. Int. J. Rheum. Dis. 2020, 23, 1297–1304. [Google Scholar] [CrossRef] [PubMed]
- Martino, M.T.D.; Tagliaferri, P.; Tassone, P. MicroRNA in Cancer Therapy: Breakthroughs and Challenges in Early Clinical Applications. J. Exp. Clin. Cancer Res. 2025, 44, 126. [Google Scholar] [CrossRef]
- Seyhan, A.A. Trials and Tribulations of MicroRNA Therapeutics. Int. J. Mol. Sci. 2024, 25, 1469. [Google Scholar] [CrossRef]
- Fu, Y.; Chen, J.; Huang, Z. Recent Progress in MicroRNA-Based Delivery Systems for the Treatment of Human Disease. ExRNA 2019, 1, 24. [Google Scholar] [CrossRef]
- Makino, K.; Jinnin, M.; Hirano, A.; Yamane, K.; Eto, M.; Kusano, T.; Honda, N.; Kajihara, I.; Makino, T.; Sakai, K.; et al. The Downregulation of MicroRNA Let-7a Contributes to the Excessive Expression of Type I Collagen in Systemic and Localized Scleroderma. J. Immunol. 2013, 190, 3905–3915. [Google Scholar] [CrossRef]
- Li, Q.; Yao, Y.; Shi, S.; Zhou, M.; Zhou, Y.; Wang, M.; Chiu, J.-J.; Huang, Z.; Zhang, W.; Liu, M.; et al. Inhibition of MiR-21 Alleviated Cardiac Perivascular Fibrosis via Repressing EndMT in T1DM. J. Cell. Mol. Med. 2020, 24, 910–920. [Google Scholar] [CrossRef]
- Man, S.; Sanchez Duffhues, G.; ten Dijke, P.; Baker, D. The Therapeutic Potential of Targeting the Endothelial-to-Mesenchymal Transition. Angiogenesis 2019, 22, 3–13. [Google Scholar] [CrossRef]
- Khalaji, A.; Mehrtabar, S.; Jabraeilipour, A.; Doustar, N.; Youshanlouei, H.R.; Tahavvori, A.; Fattahi, P.; Alavi, S.M.A.; Taha, S.R.; Fazlollahpour-Naghibi, A.; et al. Inhibitory Effect of MicroRNA-21 on Pathways and Mechanisms Involved in Cardiac Fibrosis Development. Ther. Adv. Cardiovasc. Dis. 2024, 18, 17539447241253134. [Google Scholar] [CrossRef]
- Pagoni, M.; Cava, C.; Sideris, D.C.; Avgeris, M.; Zoumpourlis, V.; Michalopoulos, I.; Drakoulis, N. MiRNA-Based Technologies in Cancer Therapy. J. Pers. Med. 2023, 13, 1586. [Google Scholar] [CrossRef]
- Mc Cormack, B.A.; González-Cantó, E.; Agababyan, C.; Espinoza-Sánchez, N.A.; Tomás-Pérez, S.; Llueca, A.; Marí-Alexandre, J.; Götte, M.; Gilabert-Estellés, J. MiRNAs in the Era of Personalized Medicine: From Biomarkers to Therapeutics. Int. J. Mol. Sci. 2021, 22, 8154. [Google Scholar] [CrossRef]
- Diener, C.; Keller, A.; Meese, E. Emerging Concepts of MiRNA Therapeutics: From Cells to Clinic. Trends Genet. 2022, 38, 613–626. [Google Scholar] [CrossRef] [PubMed]
- Castañón-Cortés, L.G.; Bravo-Vázquez, L.A.; Santoyo-Valencia, G.; Medina-Feria, S.; Sahare, P.; Duttaroy, A.K.; Paul, S. Current Advances in the Development of MicroRNA-Integrated Tissue Engineering Strategies: A Cornerstone of Regenerative Medicine. Front. Bioeng. Biotechnol. 2024, 12, 1484151. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.-H.; Xu, X.-W.; Fu, X.-Y.; Zhou, L.-D.; Liu, S.-P.; Tan, D.-M. Long Non-Coding RNA MALAT1 Promotes Angiogenesis and Immunosuppressive Properties of HCC Cells by Sponging MiR-140. Am. J. Physiol.-Cell Physiol. 2019, 318, C649–C663. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Wang, S. Angio-LncRs: LncRNAs That Regulate Angiogenesis and Vascular Disease. Theranostics 2018, 8, 3654–3675. [Google Scholar] [CrossRef]
- Ran, L.; Pan, W.; Feng, J.; Tang, L. Long Non-Coding RNA MALAT1: A Crucial Factor in Fibrotic Diseases. Mol. Ther. Nucleic Acids 2025, 36, 102630. [Google Scholar] [CrossRef]
- Chen, X.; Yang, T.; Wang, W.; Xi, W.; Zhang, T.; Li, Q.; Yang, A.; Wang, T. Circular RNAs in Immune Responses and Immune Diseases. Theranostics 2019, 9, 588–607. [Google Scholar] [CrossRef]
- Zhao, M.; Lin, M.; Zhang, Z.; Ye, L. Research Progress of Circular RNA FOXO3 in Diseases (Review). Glob. Med. Genet. 2025, 12, 100003. [Google Scholar] [CrossRef]
- Gallant-Behm, C.L.; Piper, J.; Dickinson, B.A.; Dalby, C.M.; Pestano, L.A.; Jackson, A.L. A Synthetic MicroRNA-92a Inhibitor (MRG-110) Accelerates Angiogenesis and Wound Healing in Diabetic and Nondiabetic Wounds. Wound Repair Regen. 2018, 26, 311–323. [Google Scholar] [CrossRef]
- Abplanalp, W.T.; Fischer, A.; John, D.; Zeiher, A.M.; Gosgnach, W.; Darville, H.; Montgomery, R.; Pestano, L.; Allée, G.; Paty, I.; et al. Efficiency and Target Derepression of Anti-MiR-92a: Results of a First in Human Study. Nucleic Acid. Ther. 2020, 30, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Gallant-Behm, C.L.; Piper, J.; Lynch, J.M.; Seto, A.G.; Hong, S.J.; Mustoe, T.A.; Maari, C.; Pestano, L.A.; Dalby, C.M.; Jackson, A.L.; et al. A MicroRNA-29 Mimic (Remlarsen) Represses Extracellular Matrix Expression and Fibroplasia in the Skin. J. Investig. Dermatol. 2019, 139, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Anastasiadou, E.; Seto, A.G.; Beatty, X.; Hermreck, M.; Gilles, M.-E.; Stroopinsky, D.; Pinter-Brown, L.C.; Pestano, L.; Marchese, C.; Avigan, D.; et al. Cobomarsen, an Oligonucleotide Inhibitor of MiR-155, Slows DLBCL Tumor Cell Growth In Vitro and In Vivo. Clin. Cancer Res. 2021, 27, 1139–1149. [Google Scholar] [CrossRef] [PubMed]
- Seto, A.G.; Beatty, X.; Lynch, J.M.; Hermreck, M.; Tetzlaff, M.; Duvic, M.; Jackson, A.L. Cobomarsen, an Oligonucleotide Inhibitor of MiR-155, Co-Ordinately Regulates Multiple Survival Pathways to Reduce Cellular Proliferation and Survival in Cutaneous T-Cell Lymphoma. Br. J. Haematol. 2018, 183, 428–444. [Google Scholar] [CrossRef]
| miRNAs | Level | Targets | Effects | Ref |
|---|---|---|---|---|
| Angiogenesis | ||||
| let-7f | ↓ | TIMP-1 | Migration and proliferation of ECs, sprout formation | [94] |
| miR-1 | ↓ | VEGF-A | Tube formation and proliferation of ECs | [95] |
| miR-10a | ↑ | MAP3K7, TAK1, βTRC | Modulation of proinflammatory EC phenotypes in atherosusceptible regions in vivo | [96] |
| miR-126 | ↑ | VCAM-1 SPRED, PIK3R2, VEGFR-2, p85-β | Vascular integrity, cell adhesion and migration Proliferation of ECs Angiogenesis in vivo | [81,97,98] |
| miR-130a | ↑ | GAX, HOX-A5 | Inhibition of ECs migration and proliferation | [82] |
| miR-133a/b | ↑ | TGF-β1 | ECs’ proliferation and branch formation | [99,100] |
| miR-146a | ↑ | VEGF, PAK-1 | Formation of new blood vessels | [101] |
| miR-155 | ↑ | AT1R, VEGFR-2 | Migration and proliferation of ECs Angiogenesis in the region of ischemia | [102,103,104,105] |
| miR-17 | ↓ | ICAM-1, Janus Kinase 1 | EC’s adhesion and migration | [106,107] |
| miR-17-5p | ↑ | TSP-1/CTGF, TIMP1 | Migration and growth of ECs | [108] |
| miR-17~92 | ↑ | TSP-1/CTGF | Migration and growth of ECs | [87,109] |
| miR-18a | ↑ | TSR/VEGFR-2 | Migration and growth of ECs | [110] |
| miR-19a | ↑ | TSR/VEGFR-2 | Migration and growth of ECs | [111] |
| miR-20a | ↓ ↑ | VEGF MKK3 | Migration and growth of ECs ECs’ migration and angiogenesis | [112] |
| miR-21 | ↓ | PTEN, SMAD7 | ECs’ migration and proliferation | [113] |
| miR-210 | ↑ | Ephrin-A3 NPTX1 | ECs’ tube formation, proliferation, and migration ECs-mediated angiogenesis | [83,114] |
| miR-217 | ↑ | FOXO, eNOS, SIRT1 | Vessel formation, maturation | [115] |
| miR-218 | ↓ | ROBO-1 | Neovascularization Dysregulated endothelial migration Impaired retinal vasculature | [116] |
| miR-22 | ↑ | HIF1α, VEGF | Inhibition of VEGF secretion | [117] |
| miR-221 | ↓ | c-Kit, eNOS | Migration and proliferation of ECs Vessel permeability Tube formation, migration, and impaired wound healing | [118,119] |
| miR-222 | ↓ | c-Kit, eNOS STAT5a | Migration and proliferation of ECs Vessel permeability Inflammation-mediated vascular remodeling Tube formation, migration, and impaired wound healing | [119,120] |
| miR-23a | ↑ | PHD1,2 | Vascular permeability and cellular migration | [121] |
| miR-27b | ↑ | SEMA6A | Sprout formation | [122] |
| miR-296 | ↑ | HGS | Tube formation and migration in vitro, angiogenesis in vivo | [84] |
| miR-31 | ↓ | E-selectin | Immune cell infiltration at sites of inflammation | [123] |
| miR-34a | ↑ | SIRT1, p53 | Angiogenesis blockade in ECs | [124,125,126,127] |
| miR-320 | ↓ | IGF-1 | Angiogenesis in diabetic ECs | [89] |
| miR-377 | ↓ | CD133, VEGF | Angiogenesis | [128] |
| miR-378 | ↑ | FUS-1, SUFU | Angiogenesis | [85] |
| miR-424 | ↑ | CUL-2, HIF-1α | Cell proliferation, chemotaxis, angiogenesis, vascular remodeling | [129] |
| miR-4530 | ↑ | VASH-1 | Angiogenesis | [130] |
| miR-92a | ↓ | ITG-α5 KLF-4 KLF-5 | Angiogenesis and vessel formation Proliferation Cell adhesion and cell interactions | [86,131,132,133,134] |
| Vascular inflammation | ||||
| miR-125a | ↓ | PFKFB3 | ECs metabolism | [135] |
| miR-126 | ↑ | SPRED-1 VCAM-1 ITG-α5 | Inflammatory response Vascular integrity and homeostasis Angiogenesis | [73,81,98,136,137,138] |
| miR-193b | ↓ | PLAU | uPA signaling in MVECs | [139] |
| Cellular senescence | ||||
| miR-146a | ↑ | NOX4, KLF-4 | Cell growth | [101] |
| miR-181a | ↑ | NOX4 | Cell growth | [140,141,142] |
| miR-200c | ↑ | ZEB1 | Cell growth | [143] |
| miR-217 | ↑ | SIRT1, FOXO | Stress resistance | [115] |
| miR-34a | ↓ | SIRT1, p53 | Stress resistance | [124,125] |
| miR-152 | ↓ | DNMT1 | Hypermethylation in MVECs | [144] |
| miR-30b | ↓ | PDGFRB | PDGF signaling | [145,146] |
| miRNA | Direction of Change | Validated Targets or Pathways | Functional Role | Associated Vascular Complication | Diagnostic or Prognostic Potential |
|---|---|---|---|---|---|
| miR-126 | ↓ | SPRED1, PIK3R2 (VEGF signaling) | Pro-angiogenic (lost in SSc) | Capillary loss, impaired angiogenesis | Circulating biomarker of SSc vasculopathy |
| miR-130a | ↓ | HOXA5, GAX | Pro-angiogenic (downregulated) | Defective neovascularization | Potential marker of angiogenesis defects |
| miR-210 | ↑ | EFNA3, PHDs | Hypoxia-induced angiogenesis | Hypoxia-driven angiogenesis | Associated with hypoxia severity; candidate circulating biomarker |
| miR-132 | ↑ | RAS/ERK pathway regulators | Pro-angiogenic, endothelial proliferation | Enhanced vascular repair | Exploratory biomarker (needs validation) |
| miR-92a | ↑ | ITGα5, KLF4/5 | Anti-angiogenic | Inhibited angiogenesis, vascular rarefaction | Linked to digital ulcers; candidate biomarker |
| miR-155 | ↑ | AT1R, VCAM-1 | Pro-inflammatory, vascular injury | Inflammation-driven vasculopathy, PAH | Associated with PAH and inflammation severity |
| miR-29 | ↓ | Collagens, ECM genes | Anti-fibrotic (loss promotes fibrosis) | Fibrosis, vascular remodeling | Circulating biomarker of fibrosis severity |
| miR-221 | ↑ | c-Kit, endothelial proliferation | Anti-angiogenic, impairs endothelial repair | Endothelial dysfunction, impaired repair | Preliminary evidence in SSc; exploratory biomarker |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rusek, M. Angiogenic microRNAs in Systemic Sclerosis: Insights into Microvascular Dysfunction and Therapeutic Implications. Genes 2025, 16, 1057. https://doi.org/10.3390/genes16091057
Rusek M. Angiogenic microRNAs in Systemic Sclerosis: Insights into Microvascular Dysfunction and Therapeutic Implications. Genes. 2025; 16(9):1057. https://doi.org/10.3390/genes16091057
Chicago/Turabian StyleRusek, Marta. 2025. "Angiogenic microRNAs in Systemic Sclerosis: Insights into Microvascular Dysfunction and Therapeutic Implications" Genes 16, no. 9: 1057. https://doi.org/10.3390/genes16091057
APA StyleRusek, M. (2025). Angiogenic microRNAs in Systemic Sclerosis: Insights into Microvascular Dysfunction and Therapeutic Implications. Genes, 16(9), 1057. https://doi.org/10.3390/genes16091057






