Genetic Alterations in a Large Population of Italian Patients Affected by Neurodevelopmental Disorders
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Genetic Testing
2.3. Data Analysis
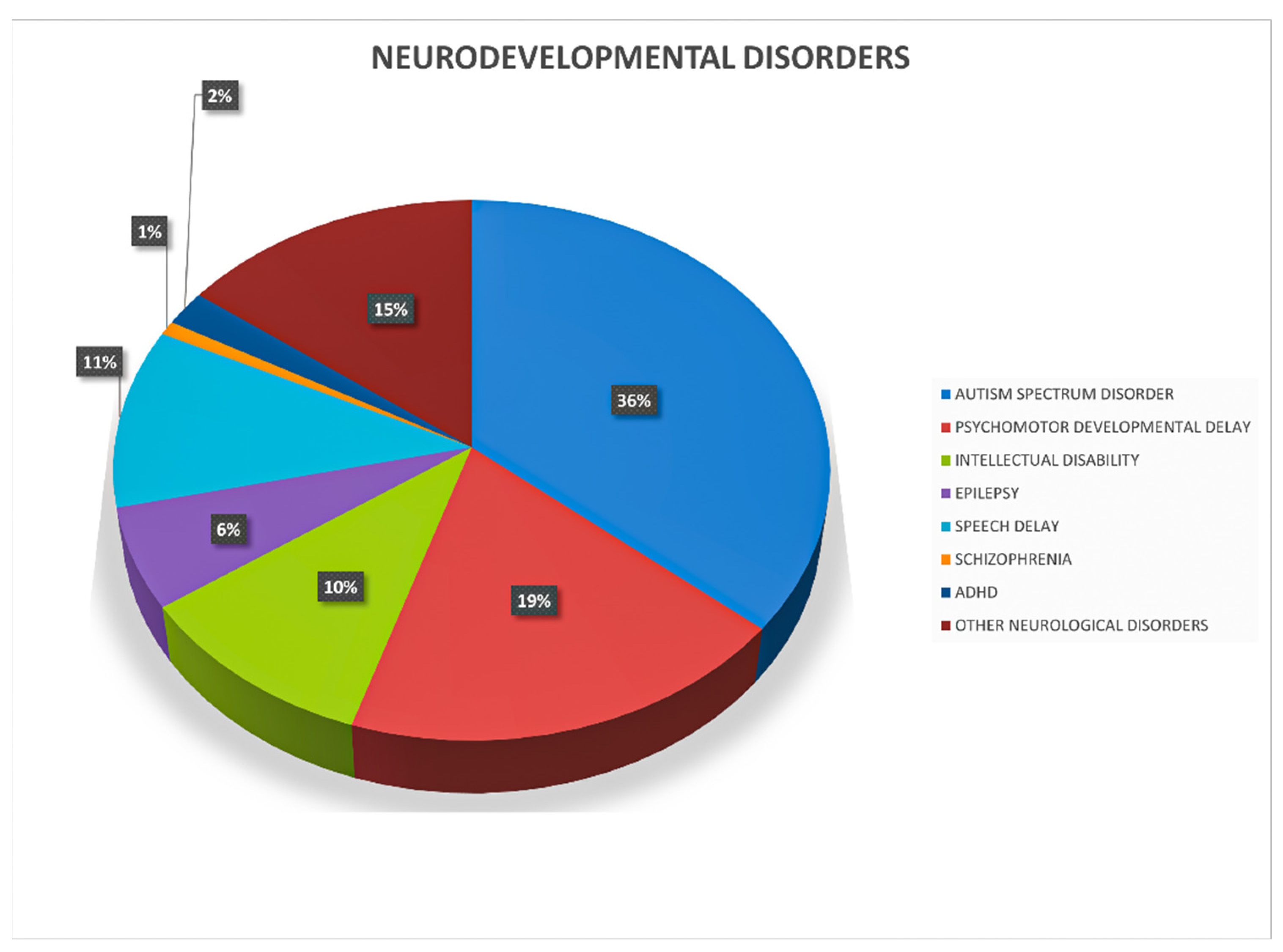
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bartnik, M.; Szczepanik, E.; Derwińska, K.; Wiśniowiecka-Kowalnik, B.; Gambin, T.; Sykulski, M.; Ziemkiewicz, K.; Keogonekdzior, M.; Gos, M.; Hoffman-Zacharska, D.; et al. Application of array comparative genomic hybridization in 102 patients with epilepsy and additional neurodevelopmental disorders. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2012, 159, 760–771. [Google Scholar] [CrossRef] [PubMed]
- Mullin, A.P.; Gokhale, A.; Moreno-De-Luca, A.; Sanyal, S.; Waddington, J.L.; Faundez, V. Neurodevelopmental disorders: Mechanisms and boundary definitions from genomes, interactomes and proteomes. Transl. Psychiatry 2013, 3, e329-6. [Google Scholar] [CrossRef] [PubMed]
- Morris-Rosendahl, D.J.; Crocq, M.A. Neurodevelopmental disorders-the history and future of a diagnostic concept. Dialogues Clin. Neurosci. 2020, 22, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Jeste Spurling, S. Neurodevelopmental behavioral and cognitive disorders. Contin. Lifelong Learn. Neurol. 2015, 21, 690. [Google Scholar] [CrossRef]
- De Felice, A.; Ricceri, L.; Venerosi, A.; Chiarotti, F.; Calamandrei, G. Multifactorial origin of neurodevelopmental disorders: Approaches to understanding complex etiologies. Toxics 2015, 3, 89–129. [Google Scholar] [CrossRef] [PubMed]
- Parenti, I.; Rabaneda, L.G.; Schoen, H.; Novarino, G. Neurodevelopmental Disorders: From Genetics to Functional Pathways. Trends Neurosci. 2020, 43, 608–621. [Google Scholar] [CrossRef] [PubMed]
- Wiśniowiecka-Kowalnik, B.; Nowakowska, B.A. Genetics and epigenetics of autism spectrum disorder—Current evidence in the field. J. Appl. Genet. 2019, 60, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Ghiani, A.; Faundez, V. Cellular and Molecular Mechanisms of Neurodevelopmental Disorders. Neurosci. Res. 2017, 95, 1093–1096. [Google Scholar] [CrossRef]
- Cheroni, C.; Caporale, N.; Testa, G. Autism spectrum disorder at the crossroad between genes and environment: Contributions, convergences, and interactions in ASD developmental pathophysiology. Mol. Autism 2020, 11, 69. [Google Scholar] [CrossRef] [PubMed]
- Fakhro, K.A. Genomics of Autism. Adv. Neurobiol. 2020, 24, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Ní Ghrálaigh, F.; Gallagher, L.; Lopez, L.M. Autism spectrum disorder genomics: The progress and potential of genomic technologies. Genomics 2020, 112, 5136–5142. [Google Scholar] [CrossRef] [PubMed]
- Lovrečić, L.; Rajar, P.; Volk, M.; Bertok, S.; Gnidovec Stražišar, B.; Osredkar, D.; Jekovec Vrhovšek, M.; Peterlin, B. Diagnostic efficacy and new variants in isolated and complex autism spectrum disorder using molecular karyotyping. J. Appl. Genet. 2018, 59, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T. Genomic aberrations associated with the pathophysiological mechanisms of neurodevelopmental disorders. Cells 2021, 10, 2317. [Google Scholar] [CrossRef] [PubMed]
- Molloy, C.J.; Quigley, C.; McNicholas, Á.; Lisanti, L.; Gallagher, L. A review of the cognitive impact of neurodevelopmental and neuropsychiatric associated copy number variants. Transl. Psychiatry 2023, 13, 116. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, B.; D’Argenio, V.; Monda, E.; Vitale, A.; Caiazza, M.; Sacchetti, L.; Pastore, L.; Limongelli, G.; Frisso, G.; Mazzaccara, C. Genetic analysis resolves differential diagnosis of a familial syndromic dilated cardiomyopathy: A new case of Alström syndrome. Mol. Genet. Genomic Med. 2020, 8, e1260. [Google Scholar] [CrossRef] [PubMed]
- Iossa, S.; Costa, V.; Corvino, V.; Auletta, G.; Barruffo, L.; Cappellani, S.; Ceglia, C.; Cennamo, G.; D’Adamo, A.P.; D’Amico, A.; et al. Phenotypic and genetic characterization of a family carrying two Xq21.1-21.3 interstitial deletions associated with syndromic hearing loss. Mol. Cytogenet. 2015, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Veneruso, I.; Di Resta, C.; Tomaiuolo, R.; D’argenio, V. Current Updates on Expanded Carrier Screening: New Insights in the Omics Era. Medicina 2022, 58, 455. [Google Scholar] [CrossRef] [PubMed]
- De Angelis, C.; Nardelli, C.; Concolino, P.; Pagliuca, M.; Setaro, M.; De Paolis, E.; De Placido, P.; Forestieri, V.; Scaglione, G.L.; Ranieri, A.; et al. Case Report: Detection of a Novel Germline PALB2 Deletion in a Young Woman With Hereditary Breast Cancer: When the Patient’s Phenotype History Doesn’t Lie. Front. Oncol. 2021, 11, 602523. [Google Scholar] [CrossRef] [PubMed]
- Arnett, A.B.; Wang, T.; Eichler, E.E.; Bernier, R.A. Reflections on the genetics-first approach to advancements in molecular genetic and neurobiological research on neurodevelopmental disorders. J. Neurodev. Disord. 2021, 13, 24. [Google Scholar] [CrossRef] [PubMed]
- Carter, M.T.; Srour, M.; Au, P.Y.B.; Buhas, D.; Dyack, S.; Eaton, A.; Inbar-Feigenberg, M.; Howley, H.; Kawamura, A.; Lewis, S.M.E.; et al. Genetic and metabolic investigations for neurodevelopmental disorders: Position statement of the Canadian College of Medical Geneticists (CCMG). J. Med. Genet. 2023, 60, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, B.; Pagani, M.; De Rosa, A.; Nunziato, M.; Migliarini, S.; Garofalo, M.; Terrile, M.; D’Argenio, V.; Galbusera, A.; Nuzzo, T.; et al. D-aspartate oxidase gene duplication induces social recognition memory deficit in mice and intellectual disabilities in humans. Transl. Psychiatry 2022, 12, 305. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, T.; Molander, F.; Taylor, M.J.; Jonsson, U.; Bölte, S. Early environmental risk factors for neurodevelopmental disorders—A systematic review of twin and sibling studies. Dev. Psychopathol. 2021, 33, 1448–1495. [Google Scholar] [CrossRef] [PubMed]
- Di Stefano, C.; Lombardo, B.; Fabbricatore, C.; Munno, C.; Caliendo, I.; Gallo, F.; Pastore, L. Oculo-facio-cardio-dental (OFCD) syndrome: The first Italian case of BCOR and co-occurring OTC gene deletion. Gene 2015, 559, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Riggs, E.; Andersen, E.; Cherry, A.; Kantarci, S.; Kearney, H.; Patel, A.; Raca, G.; Ritter, D.; South, S.; Thorland, E.; et al. Technical standards for the interpretation and reporting of constitutional copy-number variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics (ACMG) and the Clinical Genome Resource (ClinGen). Genet Med. 2020, 22, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.T.; Adam, M.P.; Aradhya, S.; Biesecker, L.G.; Brothman, A.R.; Carter, N.P.; Church, D.M.; Crolla, J.A.; Eichler, E.E.; Epstein, C.J.; et al. Consensus Statement: Chromosomal Microarray Is a First-Tier Clinical Diagnostic Test for Individuals with Developmental Disabilities or Congenital Anomalies. Am. J. Hum. Genet. 2010, 86, 749–764. [Google Scholar] [CrossRef] [PubMed]
- Heil, K.M.; Schaaf, C.P. The genetics of autism spectrum disorders—A guide for clinicians. Curr. Psychiatry Rep. 2013, 15, 334. [Google Scholar] [CrossRef] [PubMed]
- Watson, C.T.; Marques-Bonet, T.; Sharp, A.J.; Mefford, H.C. The Genetics of Microdeletion and Microduplication Syndromes: An Update. Annu. Rev. Genom. Hum. Genet. 2014, 15, 215–244. [Google Scholar] [CrossRef] [PubMed]
- Rylaarsdam, L.; Guemez-Gamboa, A. Genetic Causes and Modifiers of Autism Spectrum Disorder. Front. Cell Neurosci. 2019, 13, 385. [Google Scholar] [CrossRef] [PubMed]
- Wayhelova, M.; Smetana, J.; Vallova, V.; Hladilkova, E.; Filkova, H.; Hanakova, M.; Vilemova, M.; Nikolova, P.; Gromesova, B.; Gaillyova, R.; et al. The clinical benefit of array-based comparative genomic hybridization for detection of copy number variants in Czech children with intellectual disability and developmental delay. BMC Med. Genom. 2019, 12, 111. [Google Scholar] [CrossRef]
- Carbonell, A.U.; Cho, C.H.; Tindi, J.O.; Counts, P.A.; Bates, J.C.; Erdjument-Bromage, H.; Cvejic, S.; Iaboni, A.; Kvint, I.; Rosensaft, J.; et al. Haploinsufficiency in the ANKS1B gene encoding AIDA-1 leads to a neurodevelopmental syndrome. Nat. Commun. 2019, 10, 3529. [Google Scholar] [CrossRef]
- Jordan, B.A.; Fernholz, B.D.; Khatri, L.; Ziff, E.B. Activity-dependent AIDA-1 nuclear signaling regulates nucleolar numbers and protein synthesis in neurons. Nat. Neurosci. 2007, 10, 427–435. [Google Scholar] [CrossRef]
- Carbonell, A.U.; Freire-Cobo, C.; Deyneko, I.V.; Dobariya, S.; Erdjument-Bromage, H.; Clipperton-Allen, A.E.; Page, D.T.; Neubert, T.A.; Jordan, B.A. Comparing synaptic proteomes across five mouse models for autism reveals converging molecular similarities including deficits in oxidative phosphorylation and Rho GTPase signaling. Front. Aging Neurosci. 2023, 15, 1152562. [Google Scholar] [CrossRef] [PubMed]
- Enga, R.M.; Rice, A.C.; Weller, P.; Subler, M.A.; Lee, D.; Hall, C.P.; Windle, J.J.; Beardsley, P.M.; van den Oord, E.J.; McClay, J.L. Initial characterization of behavior and ketamine response in a mouse knockout of the post-synaptic effector gene Anks1b. Neurosci. Lett. 2017, 641, 26–32. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Uddin, M.; Tammimies, K.; Pellecchia, G.; Alipanahi, B.; Hu, P.; Wang, Z.; Pinto, D.; Lau, L.; Nalpathamkalam, T.; Marshall, C.R.; et al. Brain-expressed exons under purifying selection are enriched for de novo mutations in autism spectrum disorder. Nat. Genet. 2014, 46, 742–747. [Google Scholar] [CrossRef] [PubMed]
- Pinto, D.; Delaby, E.; Merico, D.; Barbosa, M.; Merikangas, A.; Klei, L.; Thiruvahindrapuram, B.; Xu, X.; Ziman, R.; Wang, Z.; et al. Convergence of genes and cellular pathways dysregulated in autism spectrum disorders. Am. J. Hum. Genet. 2014, 94, 677–694. [Google Scholar] [CrossRef] [PubMed]
- Cardo, L.F.; de la Fuente, D.C.; Li, M. Impaired neurogenesis and neural progenitor fate choice in a human stem cell model of SETBP1 disorder. Mol. Autism 2023, 14, 8. [Google Scholar] [CrossRef] [PubMed]
- Filges, I.; Shimojima, K.; Okamoto, N.; Röthlisberger, B.; Weber, P.; Huber, A.R.; Nishizawa, T.; Datta, A.N.; Miny, P.; Yamamoto, T. Reduced expression by SETBP1 haploinsufficiency causes developmental and expressive language delay indicating a phenotype distinct from Schinzel-Giedion syndrome. J. Med. Genet. 2011, 48, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Schinzel, A.; Binkert, F.; Lillington, D.M.; Sands, M.; Stocks, R.J.; Lindenbaum, R.H.; Matthews, H.; Sheridan, H. Interstitial deletion of the long arm of chromosome 18, del(18)(q12.2q21.1): A report of three cases of an autosomal deletion with a mild phenotype. J. Med. Genet. 1991, 28, 352–355. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bouquillon, S.; Andrieux, J.; Landais, E.; Duban-Bedu, B.; Boidein, F.; Lenne, B.; Vallée, L.; Leal, T.; Doco-Fenzy, M.; Delobel, B. A 5.3Mb deletion in chromosome 18q12.3 as the smallest region of overlap in two patients with expressive speech delay. Eur. J. Med. Genet. 2011, 54, 194–197. [Google Scholar] [CrossRef] [PubMed]
- Marseglia, G.; Scordo, M.R.; Pescucci, C.; Nannetti, G.; Biagini, E.; Scandurra, V.; Gerundino, F.; Magi, A.; Benelli, M.; Torricelli, F. 372 kb microdeletion in 18q12.3 causing SETBP1 haploinsufficiency associated with mild mental retardation and expressive speech impairment. Eur. J. Med. Genet. 2012, 55, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Jansen, N.A.; Braden, R.O.; Srivastava, S.; Otness, E.F.; Lesca, G.; Rossi, M.; Nizon, M.; Bernier, R.A.; Quelin, C.; van Haeringen, A.; et al. Clinical delineation of SETBP1 haploinsufficiency disorder. Eur. J. Hum. Genet. 2021, 29, 1198–1205. [Google Scholar] [CrossRef] [PubMed]
- Behesti, H.; Fore, T.R.; Wu, P.; Horn, Z.; Leppert, M.; Hull, C.; Hattena, M.E. ASTN2 modultes synaptic strength by trafficking and degradation of surface proteins. Proc. Natl. Acad. Sci. USA 2018, 115, E9717–E9726. [Google Scholar] [CrossRef] [PubMed]
- Glessner, J.T.; Wang, K.; Cai, G.; Korvatska, O.; Kim, C.E.; Wood, S.; Zhang, H.; Estes, A.; Brune, C.W.; Bradfield, J.P.; et al. Autism genome-wide copy number variation reveals ubiquitin and neuronal genes. Nature 2009, 459, 569–572. [Google Scholar] [CrossRef] [PubMed]
- Lionel, A.C.; Tammimies, K.; Vaags, A.K.; Rosenfeld, J.A.; Ahn, J.W.; Merico, D.; Noor, A.; Runke, C.K.; Pillalamarri, V.K.; Carter, M.T.; et al. Disruption of the ASTN2/TRIM32 locus at 9q33.1 is a risk factor in males for autism spectrum disorders, ADHD and other neurodevelopmental phenotypes. Hum. Mol. Genet. 2014, 23, 2752–2768. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Yoshida, M.; Aida, T.; Kushima, I.; Hiramatsu, Y.; Ono, M.; Yoshimi, A.; Tanaka, K.; Ozaki, N.; Noda, Y. Astrotactin 2 (ASTN2) regulates emotional and cognitive functions by affecting neuronal morphogenesis and monoaminergic systems. J. Neurochem. 2023, 165, 211–229. [Google Scholar] [CrossRef] [PubMed]
- Hartman, C.A.; Geurts, H.M.; Franke, B.; Buitelaar, J.K.; Rommelse, N.N.J. Changing ASD-ADHD symptom co-occurrence across the lifespan with adolescence as crucial time window: Illustrating the need to go beyond childhood. Neurosci. Biobehav. Rev. 2016, 71, 529–541. [Google Scholar] [CrossRef] [PubMed]
- Mikami, A.Y.; Miller, M.; Lerner, M.D. Social functioning in youth with attention-deficit/hyperactivity disorder and autism spectrum disorder: Transdiagnostic commonalities and differences. Clin. Psychol. Rev. 2019, 68, 54–70. [Google Scholar] [CrossRef] [PubMed]
- Hogart, A.; Nagarajan, R.; Patzel, K.; Yasui, D.; Lasalle, J. GABAA receptor genes are normally biallelically expressed in brain yet are subject to epigenetic dysregulation in autism-spectrum disorders. Hum. Mol. Genet. 2007, 16, 691–703. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, J.; Shuang, M.; Lu, T.; Wang, Z.; Zhang, T.; Yue, W.; Jia, M.; Ruan, Y.; Liu, J.; et al. Association study and mutation sequencing of genes on chromosome 15q11-q13 identified GABRG3 as a susceptibility gene for autism in Chinese Han population. Transl. Psychiatry 2018, 8, 152. [Google Scholar] [CrossRef] [PubMed]
- DeLorey, T.M.; Sahbaie, P.; Hashemi, E.; Homanics, G.E.; Clark, J.D. Gabrb3 gene deficient mice exhibit impaired social and exploratory behaviors, deficits in non-selective attention and hypoplasia of cerebellar vermal lobules: A potential model of autism spectrum disorder. Behav. Brain Res. 2008, 187, 207–220. [Google Scholar] [CrossRef]
- Smith, S.E.P.; Zhou, Y.-D.; Zhang, G.; Jin, Z.; Stoppel, D.C.; Anderson, M.P. Increased gene dosage of Ube3a results in autism traits and decreased glutamate synaptic transmission in mice. Sci. Transl. Med. 2011, 3, 103ra97. [Google Scholar] [CrossRef] [PubMed]
- DeLorey, T.M.; Sahbaie, P.; Hashemi, E.; Li, W.W.; Salehi, A.; Clark, D.J. Somatosensory and sensorimotor consequences associated with the heterozygous disruption of the autism candidate gene, Gabrb3. Behav. Brain Res. 2011, 216, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Menold, M.M.; Shao, Y.; Wolpert, C.M.; Donnelly, S.L.; Raiford, K.L.; Martin, E.R.; Ravan, S.A.; Abramson, R.K.; Wright, H.H.; Delong, G.R.; et al. Association Analysis of Chromosome 15 GABAA Receptor Subunit Genes in Autistic Disorder. J. Neurogenet. 2001, 15, 245–259. [Google Scholar] [CrossRef] [PubMed]
- Adak, P.; Sinha, S.; Banerjee, N. An Association Study of Gamma-Aminobutyric Acid Type A Receptor Variants and Susceptibility to Autism Spectrum Disorders. J. Autism Dev. Disord. 2021, 51, 4043–4053. [Google Scholar] [CrossRef] [PubMed]
- Coskunpinar, E.M.; Tur, S.; Cevher Binici, N.; Yazan Songür, C. Association of GABRG3, GABRB3, HTR2A gene variants with autism spectrum disorder. Gene 2023, 870, 147399. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, C.; Hümmer, S.; Masanas, M.; Gabau, E.; Guitart, M.; Jeyaprakash, A.A.; Segura, M.F.; Santamaria, A.; Ruiz, A. The Novel KIF1A Missense Variant (R169T) Strongly Reduces Microtubule Stimulated ATPase Activity and Is Associated With NESCAV Syndrome. Front. Neurosci. 2021, 15, 618098. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.; Greeny, A.; Rajendran, R.; Abdelgawad, M.A.; Ghoneim, M.M.; Raghavan, R.P.; Sudevan, S.T.; Mathew, B.; Kim, H. KIF1A-Associated Neurological Disorder: An Overview of a Rare Mutational Disease. Pharmaceuticals 2023, 16, 147. [Google Scholar] [CrossRef] [PubMed]
- Tomaselli, P.J.; Rossor, A.M.; Horga, A.; Laura, M.; Blake, J.C.; Houlden, H.; Reilly, M.M. A de novo dominant mutation in KIF1A associated with axonal neuropathy, spasticity and autism spectrum disorder. J. Peripher. Nerv. Syst. 2017, 22, 460–463. [Google Scholar] [CrossRef] [PubMed]
- Demily, C.; Lesca, G.; Poisson, A.; Till, M.; Barcia, G.; Chatron, N.; Sanlaville, D.; Munnich, A. Additive Effect of Variably Penetrant 22q11.2 Duplication and Pathogenic Mutations in Autism Spectrum Disorder: To Which Extent Does the Tree Hide the Forest? J. Autism Dev. Disord. 2018, 48, 2886–2889. [Google Scholar] [CrossRef]
- Rio, M.; Royer, G.; Gobin, S.; de Blois, M.; Ozilou, C.; Bernheim, A.; Nizon, M.; Munnich, A.; Bonnefont, J.P.; Romana, S.; et al. Monozygotic twins discordant for submicroscopic chromosomal anomalies in 2p25.3 region detected by array CGH. Clin. Genet. 2013, 84, 31–36. [Google Scholar] [CrossRef]
- Yamakawa, H.; Oyama, S.; Mitsuhashi, H.; Sasagawa, N.; Uchino, S.; Kohsaka, S.; Ishiura, S. Neuroligins 3 and 4X interact with syntrophin-γ2, and the interactions are affected by autism-related mutations. Biochem. Biophys. Res. Commun. 2007, 355, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Felix, R.A.; Chavez, V.A.; Novicio, D.M.; Morley, B.J.; Portfors, C.V. Nicotinic acetylcholine receptor subunit α7-knockout mice exhibit degraded auditory temporal processing. J. Neurophysiol. 2019, 122, 451–465. [Google Scholar] [CrossRef] [PubMed]
- Budisteanu, M.; Papuc, S.M.; Streata, I.; Cucu, M.; Pirvu, A.; Serban-Sosoi, S.; Erbescu, A.; Andrei, E.; Iliescu, C.; Ioana, D.; et al. The phenotypic spectrum of 15q13.3 region duplications: Report of 5 patients. Genes 2021, 12, 1025. [Google Scholar] [CrossRef] [PubMed]
- Gillentine, M.A.; Schaaf, C.P. The Human Clinical Phenotypes of Altered CHRNA7 Copy Number. Biochem. Pharmacol. 2015, 97, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Antony, I.; Narasimhan, M.; Shen, R.; Prakasam, R.; Kaushik, K.; Chapman, G.; Kroll, K.L. Duplication Versus Deletion through the Lens of 15q13.3: Clinical and Research Implications of Studying Copy Number Variants Associated with Neuropsychiatric Disorders in Induced Pluripotent Stem Cell-Derived Neurons. Stem. Cell Rev. Rep. 2023, 19, 639–650. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, P.; Carlström, E.; Råstam, M.; Gillberg, C.; Anckarsäter, H. The genetics of autism spectrum disorders and related neuropsychiatric disorders in childhood. Am. J. Psychiatry 2010, 167, 1357–1363. [Google Scholar] [CrossRef] [PubMed]
- Ravel, J.M.; Renaud, M.; Muller, J.; Becker, A.; Renard, É.; Remen, T.; Lefort, G.; Dexheimer, M.; Jonveaux, P.; Leheup, B.; et al. Clinical utility of periodic reinterpretation of CNVs of uncertain significance: An 8-year retrospective study. Genome Med. 2023, 15, 39. [Google Scholar] [CrossRef]
- Zamora-Moratalla, A.; Martínez de Lagrán, M.; Dierssen, M. Neurodevelopmental disorders: 2021 update. Free Neuropathol. 2021, 2, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Firdaus, Z.; Li, X. Unraveling the Genetic Landscape of Neurological Disorders: Insights into Pathogenesis, Techniques for Variant Identification, and Therapeutic Approaches. Int. J. Mol. Sci. 2024, 25, 2320. [Google Scholar] [CrossRef]
- Lombardo, B.; Ceglia, C.; Tarsitano, M.; Pierucci, I.; Salvatore, F.; Pastore, L. Identification of a deletion in the NDUFS4 gene using array-comparative genomic hybridization in a patient with suspected mitochondrial respiratory disease. Gene 2014, 535, 376–379. [Google Scholar] [CrossRef]
- Alotibi, R.S.; Sannan, N.S.; AlEissa, M.; Aldriwesh, M.G.; Al Tuwaijri, A.; Akiel, M.A.; Almutairi, M.; Alsamer, A.; Altharawi, N.; Aljawfan, G.; et al. The diagnostic yield of CGH and WES in neurodevelopmental disorders. Front. Pediatr. 2023, 11, 1133789. [Google Scholar] [CrossRef] [PubMed]
- Ranieri, A.; Veneruso, I.; La Monica, I.; Pascale, M.G.; Pastore, L.; D’argenio, V.; Lombardo, B. Combined aCGH and Exome Sequencing Analysis Improves Autism Spectrum Disorders Diagnosis: A Case Report. Medicina 2022, 58, 522. [Google Scholar] [CrossRef] [PubMed]
- Au, P.Y.B.; Eaton, A.; Dyment, D.A. Genetic Mechanisms of Neurodevelopmental Disorders, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2020; Volume 173, ISBN 9780444641502. [Google Scholar]
- Wayhelova, M.; Vallova, V.; Broz, P.; Mikulasova, A.; Smetana, J.; Dynkova Filkova, H.; Machackova, D.; Handzusova, K.; Gaillyova, R.; Kuglik, P. Exome sequencing improves the molecular diagnostics of paediatric unexplained neurodevelopmental disorders. Orphanet J. Rare Dis. 2024, 19, 41. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Love-Nichols, J.A.; Dies, K.A.; Ledbetter, D.H.; Martin, C.L.; Chung, W.K.; Firth, H.V.; Frazier, T.; Hansen, R.L.; Prock, L.; et al. Meta-analysis and multidisciplinary consensus statement: Exome sequencing is a first-tier clinical diagnostic test for individuals with neurodevelopmental disorders. Genet. Med. 2019, 21, 2413–2421. [Google Scholar] [CrossRef] [PubMed]
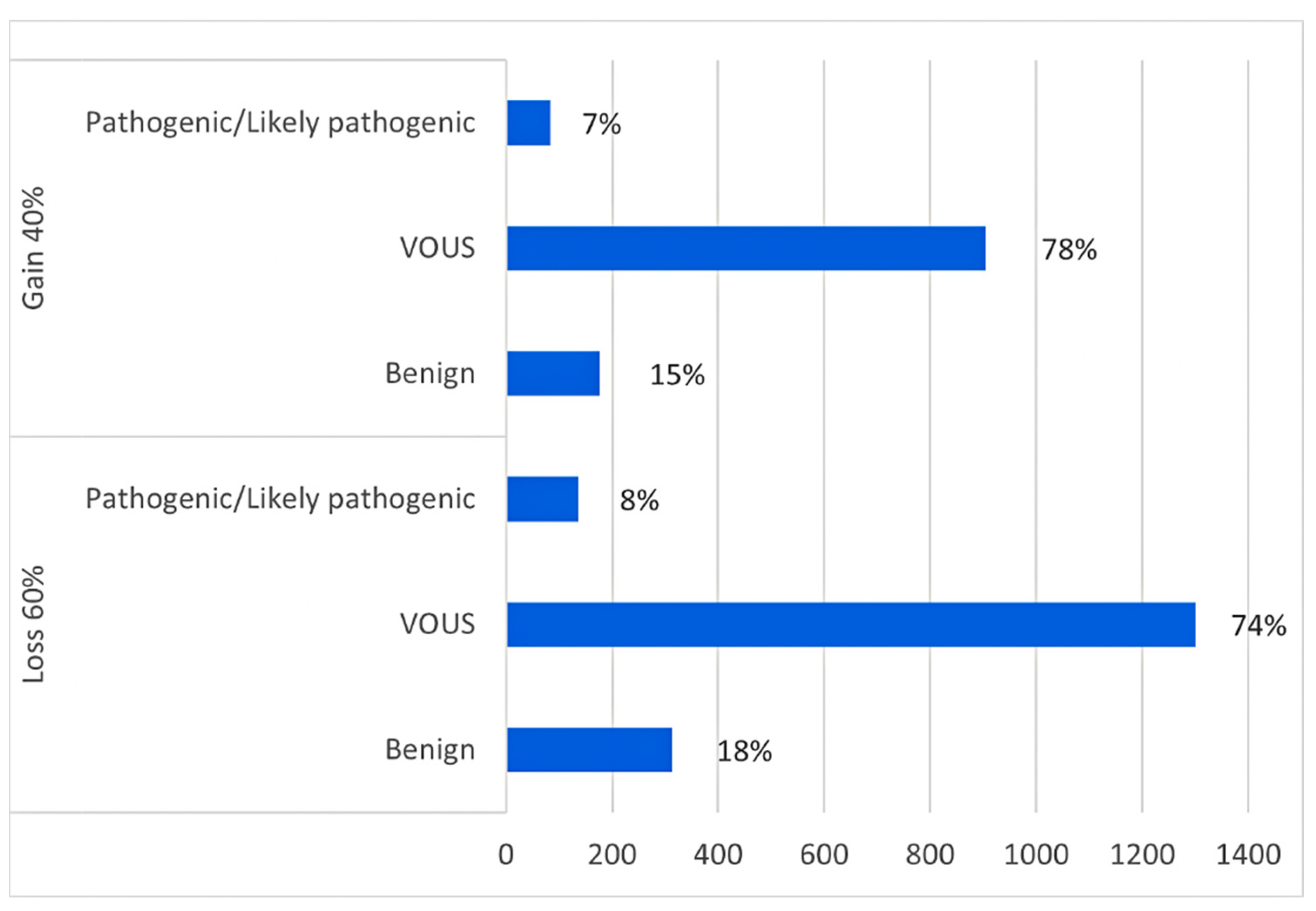
| Number of Patients Analysed | Reason for Referral | Array-CGH Results | CNV Classification | Inheritance | Related Syndrome/OMIM Number |
|---|---|---|---|---|---|
| 7 | ID, dysmorphic features, psychomotor development delay, ASD | arr 16p11.2 deletion | Pathogenic/Likely pathogenic | 1 maternal origin 2 De novo 4 ND | 16p11.2 Deletion syndrome (#611913) |
| 9 | ADHD, ID, ASD, speech delay | arr 16p11.2 duplication | Pathogenic/Likely pathogenic | 3 De novo 6 ND | 16p11.2 Duplication syndrome (#614671) |
| 22 | ID, psychomotor development delay, dysmorphic features, DiGeorge syndrome | arr 22q11.21 deletion | Pathogenic | 1 paternal origin 6 De novo 15 ND | DiGeorge syndrome (#188400) Velocardiofacial syndrome (#192430) |
| 15 | Psychomotor development delay, learning deficit, growth delay | arr 22q11.21 duplication | Pathogenic | 1 maternal origin 4 De novo 10 ND | 22q11.2 microduplication syndrome (#608363) DiGeorge syndrome (#188400) Velocardiofacial syndrome (#192430) |
| 8 | ASD, psychomotor development delay | arr 15q11.2-q13.1 duplication | Pathogenic | 2 De novo 6 ND | Duplication 15q11-q13 syndrome (#608636) Angelman syndrome (#105830) Prader–Willi syndrome (#176270) |
| 10 | Psychomotor development delay, ASD, ADHD, ID, psychomotor and growth delay, dysmorphic features, microcephaly | arr 1q21.1-q21.2 deletion | Pathogenic/Likely pathogenic | 2 maternal origin 1 paternal origin 3 De novo 4 ND | 1q21.1 Deletion syndrome (#612474) Thrombocytopenia-absent radius syndrome (#274000) |
| 11 | ASD, psychomotor development delay and epilepsy | arr 1q21.1-q21.2 duplication | Pathogenic/Likely pathogenic | 1 paternal origin 4 De novo 6 ND | 1q21.1 Duplication syndrome (#612475) |
| 6 | ASD, psychomotor development delay, Becker muscular dystrophy | arr Xp21.1 deletion | Pathogenic | 6 ND | Duchenne muscular dystrophy (#310200) Becker muscular dystrophy (#300376) |
| 5 | ADHD, speech delay, Charcot–Marie-Tooth disease type 1 | arr 17p12 duplication | Pathogenic | 1 paternal origin 1 De novo 3 ND | Charcot–Marie-Tooth disease, type 1A (#118220) |
| 4 | NDD, psychomotor development delay, speech delay | arr 17p11.2 deletion | Pathogenic | 1 paternal origin 3 ND | Smith–Magenis syndrome (#182290) |
| Number of Patients Analysed | Reason for Referral | Array-CGH Results | CNV Classification | Inheritance |
|---|---|---|---|---|
| 1 | ASD | arr 11q13.1-q13.2 duplication | Pathogenic/Likely pathogenic | De novo |
| 1 | ASD | arr 5q14.3 duplication | Pathogenic/Likely pathogenic | ND |
| 3 | ASD, psychomotor development delay | arr Xq28 deletion | Pathogenic/Likely pathogenic | 3 ND |
| 1 | ADHD | arr 13q21.32-q31.1 deletion | Pathogenic/Likely pathogenic | ND |
| 9 | ASD, psychomotor development delay, speech delay | arr 2p16.3 deletion | Pathogenic/Likely pathogenic | 1 maternal origin 1 paternal origin 2 De novo 5 ND |
| 1 | Speech delay | arr 20p12.3-p12.1 duplication | Pathogenic/Likely pathogenic | ND |
| 2 | Psychomotor development delay, growth delay, ASD | arr 15q26.3 deletion | Pathogenic/Likely pathogenic | 1 paternal origin 1 De novo |
| Number of Patients Analysed | Clinical Diagnosis | Array-CGH Results | CNV Classification | Inheritance | Gene OMIM Number and Score SFARI | Biological Function |
|---|---|---|---|---|---|---|
| 12 | ASD, psychomotor developmental delay, speech delay | arr 3p26.3-p26.2 deletion | VOUS | 1 paternal origin 2 De novo 9 ND | CNTN4 (*607280) Strong candidate | Essential for promoting dendrite growth and dendritic spine formation |
| 1 | ASD | arr 9q33.1 deletion | VOUS | De novo | ASTN2 (*612856) Strong candidate | A central role in neural-glial adhesion during neuronal migration and synaptic strength through the trafficking and degradation of surface proteins |
| 1 | NDD | arr 18q12.3 duplication | VOUS | ND | SETBP1 (*611060) High confidence | Role in controlling forebrain progenitor expansion and neurogenic differentiation |
| 6 | ASD, epilepsy, psychomotor developmental delay | arr 15q13.3 duplication | VOUS | 1 maternal origin 1 paternal origin 4 ND | CHRNA7 (*118511) Strong candidate | Involved in signal transmission at synapses |
| 1 | ASD | arr 11q13.3-q13.4 deletion | VOUS | ND | SHANK2 (*603290) High confidence | Molecular scaffolds in the postsynaptic density of excitatory synapses |
| 1 | ASD | arr 2q37.3 duplication | VOUS | Paternal origin | KIF1A (*601255) Strong syndromic candidate | Microtubule-dependent motor protein responsible for fast anterograde transport of synaptic vesicle precursors in neurons |
| 3 | ASD | arr 15q12 deletion | VOUS | 3 ND | GABRG3 (*600233) Strong candidate | Major inhibitory neurotransmitter in the mammalian brain |
| 3 | ASD | arr 3p26.3 duplication | VOUS | 1 De novo 2 ND | CNTN6 (*607220) Strong candidate | Role in the formation of axon connections in the developing nervous system |
| 1 | ASD, epilepsy | arr 16q24.3 deletion | VOUS | ND | ANKRD11 (*611192) High confidence syndromic | Regulates pyramidal neuron migration and dendritic differentiation in the developing mouse cerebral cortex |
| 3 | ASD, ID | arr 11q22.1 deletion | VOUS | 1 maternal origin 2 ND | CNTN5 (*607219) Strong candidate | Role in the formation of axon connections in the developing nervous system |
| 5 | ASD, ID, ADHD dysmorphic features | arr 12q23.1 deletion | VOUS | 1 maternal origin 1 De novo 3 ND | ANKS1B (*607815) Strong syndromic candidate | Abundant in neuronal synapses and important role in normal brain development |
| 8 | ASD, ID, psychomotor developmental delay, speech delay | arr 2p25.3 duplication | VOUS | 1 maternal origin 2 paternal origin 1 De novo 4 ND | SNTG2 (*608715) Strong candidate | A scaffolding protein that interacts with neuroligins as crucial binding factor at inhibitory synapses |
| 7 | ASD, ID, psychomotor developmental delay, speech delay, dysmorphic features | arr 22q11.2 duplication | VOUS | 1 paternal origin 6 ND | TOP3B (*603582) Strong candidate | Controls the DNA state during transcription by catalysing transient breakage and junction of a single strand of DNA |
| 3 | ASD, psychomotor developmental delay | arr 2p12 deletion | VOUS | 1 maternal origin 1 paternal origin 1 ND | CTNNA2 (*114025) Syndromic | Involved in the negative regulation of actin nucleation and in the regulation of neuron migration and the development of neuron projection |
| 12 | ASD, ID, ADHD | arr 16p13.3 deletion | VOUS | 1 paternal origin 2 De novo 9 ND | RBFOX1 (*605104) Strong candidate | Role in the regulation of the alternative splicing of large neuronal gene networks |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ranieri, A.; La Monica, I.; Di Iorio, M.R.; Lombardo, B.; Pastore, L. Genetic Alterations in a Large Population of Italian Patients Affected by Neurodevelopmental Disorders. Genes 2024, 15, 427. https://doi.org/10.3390/genes15040427
Ranieri A, La Monica I, Di Iorio MR, Lombardo B, Pastore L. Genetic Alterations in a Large Population of Italian Patients Affected by Neurodevelopmental Disorders. Genes. 2024; 15(4):427. https://doi.org/10.3390/genes15040427
Chicago/Turabian StyleRanieri, Annaluisa, Ilaria La Monica, Maria Rosaria Di Iorio, Barbara Lombardo, and Lucio Pastore. 2024. "Genetic Alterations in a Large Population of Italian Patients Affected by Neurodevelopmental Disorders" Genes 15, no. 4: 427. https://doi.org/10.3390/genes15040427
APA StyleRanieri, A., La Monica, I., Di Iorio, M. R., Lombardo, B., & Pastore, L. (2024). Genetic Alterations in a Large Population of Italian Patients Affected by Neurodevelopmental Disorders. Genes, 15(4), 427. https://doi.org/10.3390/genes15040427






