Cilia and Cancer: From Molecular Genetics to Therapeutic Strategies
Abstract
1. Introduction
2. Role of Primary Cilia in Oncogenic Programs
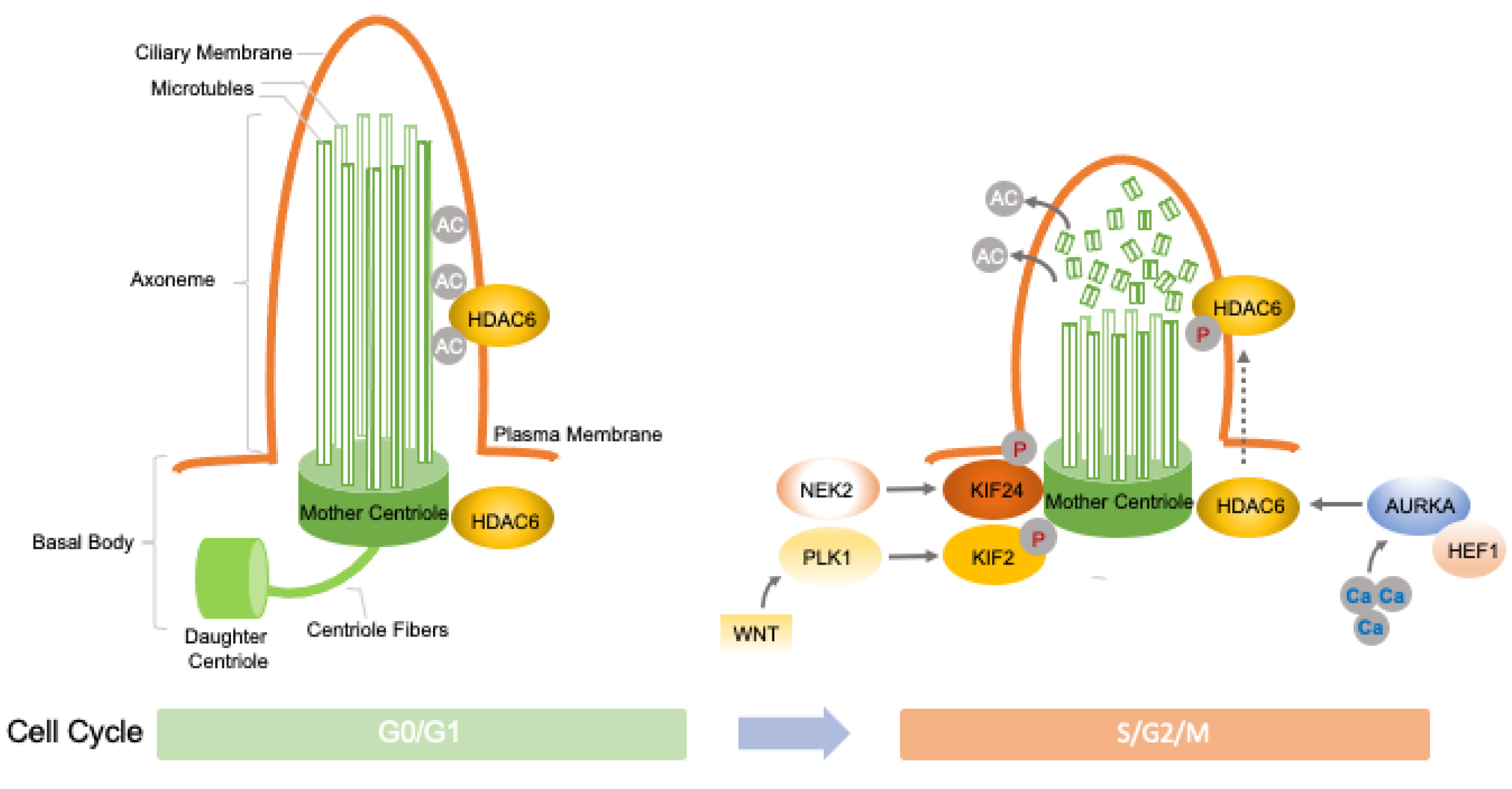
2.1. The Functional Link between Cilia and Cell-Cycle-Related Oncogenic Programs
2.2. The Functional Link between Cilia and Cancer-Related Signaling Networks
2.2.1. Hedgehog Pathway
2.2.2. Notch Pathway
2.2.3. Wnt Pathway
2.2.4. Receptor Tyrosine Kinases and Other Membrane-Associated Kinases
2.2.5. Hippo Pathway
2.2.6. DNA Damage/Repair Pathway
2.2.7. Autophagy Network
2.2.8. The Polycystin Signaling
3. Primary Cilia Defects in Cancer: Implication for Molecular Oncology
3.1. Brain Cancers
3.2. Skin Cancers
3.3. Gastrointestinal Cancers
3.4. Genito-Urinary and Endocrine Cancers
3.5. Sarcomas
4. Molecular Oncology of Primary Cilia: Clinical Implications
4.1. Brain Tumors
4.2. Skin Cancers
4.3. Gastrointestinal Cancers
4.4. Genito-Urinary and Endocrine Cancers
4.5. Implication of PC in Cancer Therapeutics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| MCs | motile cilia |
| PCs | primary cilia |
| BB | basal body |
| PKD | polycystic kidney disease |
| NPHP | nephronophthisis |
| BBS | Bardet–Biedl |
| OFD1 | oral–facial–digital type 1 syndrome |
| MTOC | microtubule-organizing center |
| PLK1 | polo-like kinase 1 |
| AURKA | Aurora A kinase |
| CDK1 | cyclin-dependent kinase |
| NEK2 | never-in-mitosis-A-related kinase |
| HEF1 | human enhancer of filamentation 1 |
| CaM | calmodulin |
| Dvl | dishevelled |
| Dvl2 | disheveled segment polarity protein 2 |
| IFT | intraflagellar transport |
| KIF24 | kinesin family member 24 |
| HDAC6 | deacetylase histone deacetylase 6 |
| HH | Hedgehog |
| PTCH1 | patched 1 |
| GPR161 | G-protein-coupled receptor |
| CCND1 | cyclin D1 |
| SMO | Smoothened |
| NICD | Notch intracellular domain |
| RTKs | receptor tyrosine kinases |
| CEP164 | centrosomal protein 164 |
| IFT20 | intraflagellar transport protein 20 |
| PCM1 | pericentriolar material 1 |
| MVA | mevalonate |
| PTC | thyroid cancer |
| FTC | follicular thyroid cancer |
| PDTC | poorly differentiated thyroid cancer |
| ATC | anaplastic thyroid cancer |
| MTC | medullary thyroid cancer |
| OS | overall survival |
References
- Reiter, J.F.; Leroux, M.R. Genes and Molecular Pathways Underpinning Ciliopathies. Nat. Rev. Mol. Cell Biol. 2017, 18, 533–547. [Google Scholar] [CrossRef] [PubMed]
- Kiesel, P.; Alvarez Viar, G.; Tsoy, N.; Maraspini, R.; Gorilak, P.; Varga, V.; Honigmann, A.; Pigino, G. The Molecular Structure of Mammalian Primary Cilia Revealed by Cryo-Electron Tomography. Nat. Struct. Mol. Biol. 2020, 27, 1115–1124. [Google Scholar] [CrossRef] [PubMed]
- Vertii, A.; Hung, H.-F.; Hehnly, H.; Doxsey, S. Human Basal Body Basics. Cilia 2016, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Takeda, S.; Narita, K. Structure and Function of Vertebrate Cilia, towards a New Taxonomy. Differentiation 2012, 83, S4–S11. [Google Scholar] [CrossRef]
- Klena, N.; Pigino, G. Structural Biology of Cilia and Intraflagellar Transport. Annu. Rev. Cell Dev. Biol. 2022, 38, 103–123. [Google Scholar] [CrossRef]
- Yoshiba, S.; Shiratori, H.; Kuo, I.Y.; Kawasumi, A.; Shinohara, K.; Nonaka, S.; Asai, Y.; Sasaki, G.; Belo, J.A.; Sasaki, H.; et al. Cilia at the Node of Mouse Embryos Sense Fluid Flow for Left-Right Determination via Pkd2. Science 2012, 338, 226–231. [Google Scholar] [CrossRef]
- Falk, N.; Lösl, M.; Schröder, N.; Gießl, A. Specialized Cilia in Mammalian Sensory Systems. Cells 2015, 4, 500–519. [Google Scholar] [CrossRef]
- Shah, A.S.; Ben-Shahar, Y.; Moninger, T.O.; Kline, J.N.; Welsh, M.J. Motile Cilia of Human Airway Epithelia Are Chemosensory. Science 2009, 325, 1131–1134. [Google Scholar] [CrossRef]
- Roth, Y.; Kimhi, Y.; Edery, H.; Aharonson, E.; Priel, Z. Ciliary Motility in Brain Ventricular System and Trachea of Hamster. Brain Res. 1985, 330, 291–297. [Google Scholar] [CrossRef]
- Aprea, I.; Nöthe-Menchen, T.; Dougherty, G.W.; Raidt, J.; Loges, N.T.; Kaiser, T.; Wallmeier, J.; Olbrich, H.; Strünker, T.; Kliesch, S.; et al. Motility of Efferent Duct Cilia Aids Passage of Sperm Cells through the Male Reproductive System. Mol. Hum. Reprod. 2021, 27, gaab009. [Google Scholar] [CrossRef]
- Brody, S.L. Sensory Functions of Motile Cilia and Implication for Bronchiectasis. Front. Biosci. 2012, S4, 320. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Singla, V.; Reiter, J.F. The Primary Cilium as the Cell’s Antenna: Signaling at a Sensory Organelle. Science 2006, 313, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Eguether, T.; Hahne, M. Mixed Signals from the Cell’s Antennae: Primary Cilia in Cancer. EMBO Rep. 2018, 19, e46589. [Google Scholar] [CrossRef] [PubMed]
- Christensen, S.T.; Pedersen, L.B.; Schneider, L.; Satir, P. Sensory Cilia and Integration of Signal Transduction in Human Health and Disease. Traffic 2007, 8, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Strissel, K.J.; Lishko, P.V.; Trieu, L.H.; Kennedy, M.J.; Hurley, J.B.; Arshavsky, V.Y. Recoverin Undergoes Light-Dependent Intracellular Translocation in Rod Photoreceptors. J. Biol. Chem. 2005, 280, 29250–29255. [Google Scholar] [CrossRef]
- Buck, L.; Axel, R. A Novel Multigene Family May Encode Odorant Receptors: A Molecular Basis for Odor Recognition. Cell 1991, 65, 175–187. [Google Scholar] [CrossRef]
- Leibovici, M.; Verpy, E.; Goodyear, R.J.; Zwaenepoel, I.; Blanchard, S.; Lainé, S.; Richardson, G.P.; Petit, C. Initial Characterization of Kinocilin, a Protein of the Hair Cell Kinocilium. Hear. Res. 2005, 203, 144–153. [Google Scholar] [CrossRef]
- Spasic, M.; Jacobs, C.R. Primary Cilia: Cell and Molecular Mechanosensors Directing Whole Tissue Function. Semin. Cell Dev. Biol. 2017, 71, 42–52. [Google Scholar] [CrossRef]
- Mohieldin, A.M.; Pala, R.; Beuttler, R.; Moresco, J.J.; Yates, J.R.; Nauli, S.M. Ciliary Extracellular Vesicles Are Distinct from the Cytosolic Extracellular Vesicles. J. Extracell. Vesicles 2021, 10, e12086. [Google Scholar] [CrossRef]
- Dubreuil, V.; Marzesco, A.-M.; Corbeil, D.; Huttner, W.B.; Wilsch-Bräuninger, M. Midbody and Primary Cilium of Neural Progenitors Release Extracellular Membrane Particles Enriched in the Stem Cell Marker Prominin-1. J. Cell Biol. 2007, 176, 483–495. [Google Scholar] [CrossRef]
- Vinay, L.; Belleannée, C. EV Duty Vehicles: Features and Functions of Ciliary Extracellular Vesicles. Front. Genet. 2022, 13, 916233. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Sirinakis, G.; Allgeyer, E.S.; Schroeder, L.K.; Duim, W.C.; Kromann, E.B.; Phan, T.; Rivera-Molina, F.E.; Myers, J.R.; Irnov, I.; et al. Ultra-High Resolution 3D Imaging of Whole Cells. Cell 2016, 166, 1028–1040. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Ning, J.; Hernandez-Lara, C.I.; Belzile, O.; Wang, Q.; Dutcher, S.K.; Liu, Y.; Snell, W.J. Uni-Directional Ciliary Membrane Protein Trafficking by a Cytoplasmic Retrograde IFT Motor and Ciliary Ectosome Shedding. Elife 2015, 4. [Google Scholar] [CrossRef]
- Nager, A.R.; Goldstein, J.S.; Herranz-Pérez, V.; Portran, D.; Ye, F.; Garcia-Verdugo, J.M.; Nachury, M.V. An Actin Network Dispatches Ciliary GPCRs into Extracellular Vesicles to Modulate Signaling. Cell 2017, 168, 252–263.e14. [Google Scholar] [CrossRef]
- Wang, W.; Jack, B.M.; Wang, H.H.; Kavanaugh, M.A.; Maser, R.L.; Tran, P.V. Intraflagellar Transport Proteins as Regulators of Primary Cilia Length. Front. Cell Dev. Biol. 2021, 9, 661350. [Google Scholar] [CrossRef] [PubMed]
- Long, H.; Zhang, F.; Xu, N.; Liu, G.; Diener, D.R.; Rosenbaum, J.L.; Huang, K. Comparative Analysis of Ciliary Membranes and Ectosomes. Curr. Biol. 2016, 26, 3327–3335. [Google Scholar] [CrossRef] [PubMed]
- Hoang-Minh, L.B.; Dutra-Clarke, M.; Breunig, J.J.; Sarkisian, M.R. Glioma Cell Proliferation Is Enhanced in the Presence of Tumor-Derived Cilia Vesicles. Cilia 2018, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Rilla, K. Diverse Plasma Membrane Protrusions Act as Platforms for Extracellular Vesicle Shedding. J. Extracell. Vesicles 2021, 10, e12148. [Google Scholar] [CrossRef]
- Gherman, A.; Davis, E.E.; Katsanis, N. The Ciliary Proteome Database: An Integrated Community Resource for the Genetic and Functional Dissection of Cilia. Nat. Genet. 2006, 38, 961–962. [Google Scholar] [CrossRef]
- Avidor-Reiss, T.; Maer, A.M.; Koundakjian, E.; Polyanovsky, A.; Keil, T.; Subramaniam, S.; Zuker, C.S. Decoding Cilia Function. Cell 2004, 117, 527–539. [Google Scholar] [CrossRef]
- Andersen, J.S.; Wilkinson, C.J.; Mayor, T.; Mortensen, P.; Nigg, E.A.; Mann, M. Proteomic Characterization of the Human Centrosome by Protein Correlation Profiling. Nature 2003, 426, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Otto, E.A.; Hurd, T.W.; Airik, R.; Chaki, M.; Zhou, W.; Stoetzel, C.; Patil, S.B.; Levy, S.; Ghosh, A.K.; Murga-Zamalloa, C.A.; et al. Candidate Exome Capture Identifies Mutation of SDCCAG8 as the Cause of a Retinal-Renal Ciliopathy. Nat. Genet. 2010, 42, 840–850. [Google Scholar] [CrossRef]
- Otto, E.A.; Ramaswami, G.; Janssen, S.; Chaki, M.; Allen, S.J.; Zhou, W.; Airik, R.; Hurd, T.W.; Ghosh, A.K.; Wolf, M.T.; et al. Mutation Analysis of 18 Nephronophthisis Associated Ciliopathy Disease Genes Using a DNA Pooling and next Generation Sequencing Strategy. J. Med. Genet. 2011, 48, 105–116. [Google Scholar] [CrossRef] [PubMed]
- van Dam, T.J.P.; Kennedy, J.; van der Lee, R.; de Vrieze, E.; Wunderlich, K.A.; Rix, S.; Dougherty, G.W.; Lambacher, N.J.; Li, C.; Jensen, V.L.; et al. CiliaCarta: An Integrated and Validated Compendium of Ciliary Genes. PLoS ONE 2019, 14, e0216705. [Google Scholar] [CrossRef]
- Lee, J.E.; Gleeson, J.G. A Systems-Biology Approach to Understanding the Ciliopathy Disorders. Genome Med. 2011, 3, 59. [Google Scholar] [CrossRef] [PubMed]
- Baker, K.; Beales, P.L. Making Sense of Cilia in Disease: The Human Ciliopathies. Am. J. Med. Genet. C Semin. Med. Genet. 2009, 151C, 281–295. [Google Scholar] [CrossRef]
- Han, Y.-G.; Alvarez-Buylla, A. Role of Primary Cilia in Brain Development and Cancer. Curr. Opin. Neurobiol. 2010, 20, 58–67. [Google Scholar] [CrossRef]
- Failler, M.; Giro-Perafita, A.; Owa, M.; Srivastava, S.; Yun, C.; Kahler, D.J.; Unutmaz, D.; Esteva, F.J.; Sánchez, I.; Dynlacht, B.D. Whole-Genome Screen Identifies Diverse Pathways That Negatively Regulate Ciliogenesis. Mol. Biol. Cell 2021, 32, 169–185. [Google Scholar] [CrossRef]
- Tilley, A.E.; Walters, M.S.; Shaykhiev, R.; Crystal, R.G. Cilia Dysfunction in Lung Disease. Annu. Rev. Physiol. 2015, 77, 379–406. [Google Scholar] [CrossRef]
- Peixoto, E.; Richard, S.; Pant, K.; Biswas, A.; Gradilone, S.A. The Primary Cilium: Its Role as a Tumor Suppressor Organelle. Biochem. Pharmacol. 2020, 175, 113906. [Google Scholar] [CrossRef]
- Mansini, A.P.; Peixoto, E.; Thelen, K.M.; Gaspari, C.; Jin, S.; Gradilone, S.A. The Cholangiocyte Primary Cilium in Health and Disease. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2018, 1864, 1245–1253. [Google Scholar] [CrossRef] [PubMed]
- Izawa, I.; Goto, H.; Kasahara, K.; Inagaki, M. Current Topics of Functional Links between Primary Cilia and Cell Cycle. Cilia 2015, 4, 12. [Google Scholar] [CrossRef] [PubMed]
- Plotnikova, O.V.; Pugacheva, E.N.; Golemis, E.A. Primary Cilia and the Cell Cycle. Methods Cell Biol. 2009, 94, 137–160. [Google Scholar] [CrossRef]
- Kasahara, K.; Inagaki, M. Primary Ciliary Signaling: Links with the Cell Cycle. Trends Cell Biol. 2021, 31, 954–964. [Google Scholar] [CrossRef] [PubMed]
- Nigg, E.A.; Stearns, T. The Centrosome Cycle: Centriole Biogenesis, Duplication and Inherent Asymmetries. Nat. Cell Biol. 2011, 13, 1154–1160. [Google Scholar] [CrossRef]
- Ghossoub, R.; Molla-Herman, A.; Bastin, P.; Benmerah, A. The Ciliary Pocket: A Once-Forgotten Membrane Domain at the Base of Cilia. Biol. Cell 2011, 103, 131–144. [Google Scholar] [CrossRef]
- Sorokin, S.P. Reconstructions of Centriole Formation and Ciliogenesis in Mammalian Lungs. J. Cell Sci. 1968, 3, 207–230. [Google Scholar] [CrossRef]
- Lee, K.H.; Johmura, Y.; Yu, L.-R.; Park, J.-E.; Gao, Y.; Bang, J.K.; Zhou, M.; Veenstra, T.D.; Yeon Kim, B.; Lee, K.S. Identification of a Novel Wnt5a-CK1ε-Dvl2-Plk1-Mediated Primary Cilia Disassembly Pathway. EMBO J. 2012, 31, 3104–3117. [Google Scholar] [CrossRef]
- Kishi, K.; van Vugt, M.A.T.M.; Okamoto, K.; Hayashi, Y.; Yaffe, M.B. Functional Dynamics of Polo-Like Kinase 1 at the Centrosome. Mol. Cell. Biol. 2009, 29, 3134–3150. [Google Scholar] [CrossRef]
- García-Álvarez, B.; de Cárcer, G.; Ibañez, S.; Bragado-Nilsson, E.; Montoya, G. Molecular and Structural Basis of Polo-like Kinase 1 Substrate Recognition: Implications in Centrosomal Localization. Proc. Natl. Acad. Sci. USA 2007, 104, 3107–3112. [Google Scholar] [CrossRef]
- Wang, G.; Chen, Q.; Zhang, X.; Zhang, B.; Zhuo, X.; Liu, J.; Jiang, Q.; Zhang, C. PCM1 Recruits Plk1 to Pericentriolar Matrix to Promote Primary Cilia Disassembly before Mitotic Entry. J. Cell Sci. 2013, 126, 1355–1365. [Google Scholar] [CrossRef] [PubMed]
- Gutteridge, R.E.A.; Ndiaye, M.A.; Liu, X.; Ahmad, N. Plk1 Inhibitors in Cancer Therapy: From Laboratory to Clinics. Mol. Cancer Ther. 2016, 15, 1427–1435. [Google Scholar] [CrossRef] [PubMed]
- Pugacheva, E.N.; Jablonski, S.A.; Hartman, T.R.; Henske, E.P.; Golemis, E.A. HEF1-Dependent Aurora A Activation Induces Disassembly of the Primary Cilium. Cell 2007, 129, 1351–1363. [Google Scholar] [CrossRef] [PubMed]
- Plotnikova, O.V.; Nikonova, A.S.; Loskutov, Y.V.; Kozyulina, P.Y.; Pugacheva, E.N.; Golemis, E.A. Calmodulin Activation of Aurora-A Kinase (AURKA) Is Required during Ciliary Disassembly and in Mitosis. Mol. Biol. Cell 2012, 23, 2658–2670. [Google Scholar] [CrossRef]
- Dere, R.; Perkins, A.L.; Bawa-Khalfe, T.; Jonasch, D.; Walker, C.L. β-Catenin Links von Hippel-Lindau to Aurora Kinase A and Loss of Primary Cilia in Renal Cell Carcinoma. J. Am. Soc. Nephrol. 2015, 26, 553–564. [Google Scholar] [CrossRef]
- Hasanov, E.; Chen, G.; Chowdhury, P.; Weldon, J.; Ding, Z.; Jonasch, E.; Sen, S.; Walker, C.L.; Dere, R. Ubiquitination and Regulation of AURKA Identifies a Hypoxia-Independent E3 Ligase Activity of VHL. Oncogene 2017, 36, 3450–3463. [Google Scholar] [CrossRef]
- Gradilone, S.A.; Radtke, B.N.; Bogert, P.S.; Huang, B.Q.; Gajdos, G.B.; LaRusso, N.F. HDAC6 Inhibition Restores Ciliary Expression and Decreases Tumor Growth. Cancer Res. 2013, 73, 2259–2270. [Google Scholar] [CrossRef]
- Li, Y.; Bavarva, J.H.; Wang, Z.; Guo, J.; Qian, C.; Thibodeau, S.N.; Golemis, E.A.; Liu, W. HEF1, a Novel Target of Wnt Signaling, Promotes Colonic Cell Migration and Cancer Progression. Oncogene 2011, 30, 2633–2643. [Google Scholar] [CrossRef]
- Kim, S.; Lee, K.; Choi, J.-H.; Ringstad, N.; Dynlacht, B.D. Nek2 Activation of Kif24 Ensures Cilium Disassembly during the Cell Cycle. Nat. Commun. 2015, 6, 8087. [Google Scholar] [CrossRef]
- Kokuryo, T.; Yokoyama, Y.; Yamaguchi, J.; Tsunoda, N.; Ebata, T.; Nagino, M. NEK2 Is an Effective Target for Cancer Therapy with Potential to Induce Regression of Multiple Human Malignancies. Anticancer. Res. 2019, 39, 2251–2258. [Google Scholar] [CrossRef]
- Sung, C.-H.; Leroux, M.R. The Roles of Evolutionarily Conserved Functional Modules in Cilia-Related Trafficking. Nat. Cell Biol. 2013, 15, 1387–1397. [Google Scholar] [CrossRef] [PubMed]
- Lechtreck, K.F. IFT–Cargo Interactions and Protein Transport in Cilia. Trends Biochem. Sci. 2015, 40, 765–778. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Deng, X.; Yuan, C.; Xin, H.; Liu, G.; Zhu, Y.; Jiang, X.; Wang, C. IFT80 Improves Invasion Ability in Gastric Cancer Cell Line via Ift80/P75NGFR/MMP9 Signaling. Int. J. Mol. Sci. 2018, 19, 3616. [Google Scholar] [CrossRef] [PubMed]
- Yauch, R.L.; Gould, S.E.; Scales, S.J.; Tang, T.; Tian, H.; Ahn, C.P.; Marshall, D.; Fu, L.; Januario, T.; Kallop, D.; et al. A Paracrine Requirement for Hedgehog Signalling in Cancer. Nature 2008, 455, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Feigin, M.E.; Xue, B.; Hammell, M.C.; Muthuswamy, S.K. G-Protein–Coupled Receptor GPR161 Is Overexpressed in Breast Cancer and Is a Promoter of Cell Proliferation and Invasion. Proc. Natl. Acad. Sci. USA 2014, 111, 4191–4196. [Google Scholar] [CrossRef]
- Begemann, M.; Waszak, S.M.; Robinson, G.W.; Jäger, N.; Sharma, T.; Knopp, C.; Kraft, F.; Moser, O.; Mynarek, M.; Guerrini-Rousseau, L.; et al. Germline GPR161 Mutations Predispose to Pediatric Medulloblastoma. J. Clin. Oncol. 2020, 38, 43–50. [Google Scholar] [CrossRef]
- Shimada, I.S.; Hwang, S.-H.; Somatilaka, B.N.; Wang, X.; Skowron, P.; Kim, J.; Kim, M.; Shelton, J.M.; Rajaram, V.; Xuan, Z.; et al. Basal Suppression of the Sonic Hedgehog Pathway by the G-Protein-Coupled Receptor Gpr161 Restricts Medulloblastoma Pathogenesis. Cell Rep. 2018, 22, 1169–1184. [Google Scholar] [CrossRef]
- Han, Y.-G.; Kim, H.J.; Dlugosz, A.A.; Ellison, D.W.; Gilbertson, R.J.; Alvarez-Buylla, A. Dual and Opposing Roles of Primary Cilia in Medulloblastoma Development. Nat. Med. 2009, 15, 1062–1065. [Google Scholar] [CrossRef]
- Wong, S.Y.; Seol, A.D.; So, P.-L.; Ermilov, A.N.; Bichakjian, C.K.; Epstein, E.H.; Dlugosz, A.A.; Reiter, J.F. Primary Cilia Can Both Mediate and Suppress Hedgehog Pathway–Dependent Tumorigenesis. Nat. Med. 2009, 15, 1055–1061. [Google Scholar] [CrossRef]
- Smith, M.J.; Beetz, C.; Williams, S.G.; Bhaskar, S.S.; O’Sullivan, J.; Anderson, B.; Daly, S.B.; Urquhart, J.E.; Bholah, Z.; Oudit, D.; et al. Germline Mutations in SUFU Cause Gorlin Syndrome–Associated Childhood Medulloblastoma and Redefine the Risk Associated With PTCH1 Mutations. J. Clin. Oncol. 2014, 32, 4155–4161. [Google Scholar] [CrossRef]
- Zhou, B.; Lin, W.; Long, Y.; Yang, Y.; Zhang, H.; Wu, K.; Chu, Q. Notch Signaling Pathway: Architecture, Disease, and Therapeutics. Signal Transduct. Target. Ther. 2022, 7, 95. [Google Scholar] [CrossRef] [PubMed]
- Aster, J.C.; Pear, W.S.; Blacklow, S.C. The Varied Roles of Notch in Cancer. Annu. Rev. Pathol. Mech. Dis. 2017, 12, 245–275. [Google Scholar] [CrossRef] [PubMed]
- Ezratty, E.J.; Stokes, N.; Chai, S.; Shah, A.S.; Williams, S.E.; Fuchs, E. A Role for the Primary Cilium in Notch Signaling and Epidermal Differentiation during Skin Development. Cell 2011, 145, 1129–1141. [Google Scholar] [CrossRef] [PubMed]
- Zhan, T.; Rindtorff, N.; Boutros, M. Wnt Signaling in Cancer. Oncogene 2017, 36, 1461–1473. [Google Scholar] [CrossRef] [PubMed]
- VanHook, A.M. Wnt Signaling and Cilia Intertwined. Sci. Signal. 2012, 5, ec22. [Google Scholar] [CrossRef]
- Wallingford, J.B.; Mitchell, B. Strange as It May Seem: The Many Links between Wnt Signaling, Planar Cell Polarity, and Cilia: Figure 1. Genes Dev. 2011, 25, 201–213. [Google Scholar] [CrossRef]
- Simons, M.; Gloy, J.; Ganner, A.; Bullerkotte, A.; Bashkurov, M.; Krönig, C.; Schermer, B.; Benzing, T.; Cabello, O.A.; Jenny, A.; et al. Inversin, the Gene Product Mutated in Nephronophthisis Type II, Functions as a Molecular Switch between Wnt Signaling Pathways. Nat. Genet. 2005, 37, 537–543. [Google Scholar] [CrossRef]
- Corbit, K.C.; Shyer, A.E.; Dowdle, W.E.; Gaulden, J.; Singla, V.; Reiter, J.F. Kif3a Constrains β-Catenin-Dependent Wnt Signalling through Dual Ciliary and Non-Ciliary Mechanisms. Nat. Cell Biol. 2008, 10, 70–76. [Google Scholar] [CrossRef]
- Christensen, S.T.; Morthorst, S.K.; Mogensen, J.B.; Pedersen, L.B. Primary Cilia and Coordination of Receptor Tyrosine Kinase (RTK) and Transforming Growth Factor β (TGF-β) Signaling. Cold Spring Harb. Perspect. Biol. 2017, 9, a028167. [Google Scholar] [CrossRef]
- Egeberg, D.L.; Lethan, M.; Manguso, R.; Schneider, L.; Awan, A.; Jørgensen, T.S.; Byskov, A.G.; Pedersen, L.B.; Christensen, S.T. Primary Cilia and Aberrant Cell Signaling in Epithelial Ovarian Cancer. Cilia 2012, 1, 15. [Google Scholar] [CrossRef]
- Corless, C.L.; Barnett, C.M.; Heinrich, M.C. Gastrointestinal Stromal Tumours: Origin and Molecular Oncology. Nat. Rev. Cancer 2011, 11, 865–878. [Google Scholar] [CrossRef] [PubMed]
- Normanno, N.; Carotenuto, P.; Roma, C.; Rachiglio, A.M.; Pasquale, R.; Franco, R.; Antinolfi, G.; Piantedosi, F.; Illiano, A.; Botti, G.; et al. Optimizing Response to Gefitinib in the Treatment of Non-Small-Cell Lung Cancer. Pharmgenom. Pers. Med. 2011, 4, 1–9. [Google Scholar] [CrossRef][Green Version]
- D’Onofrio, R.; Caputo, F.; Prampolini, F.; Spallanzani, A.; Gelsomino, F.; Bettelli, S.; Manfredini, S.; Reggiani Bonetti, L.; Carotenuto, P.; Bocconi, A.; et al. CtDNA-Guided Rechallenge with Anti-EGFR Therapy in RASwt Metastatic Colorectal Cancer: Evidence from Clinical Practice. Tumori J. 2022, 030089162211225. [Google Scholar] [CrossRef] [PubMed]
- Danilov, A.I.; Gomes-Leal, W.; Ahlenius, H.; Kokaia, Z.; Carlemalm, E.; Lindvall, O. Ultrastructural and Antigenic Properties of Neural Stem Cells and Their Progeny in Adult Rat Subventricular Zone. Glia 2009, 57, 136–152. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Du, H.; Wang, X.; Mei, C.; Sieck, G.C.; Qian, Q. Characterization of Primary Cilia in Human Airway Smooth Muscle Cells. Chest 2009, 136, 561–570. [Google Scholar] [CrossRef] [PubMed]
- Kasahara, K.; Aoki, H.; Kiyono, T.; Wang, S.; Kagiwada, H.; Yuge, M.; Tanaka, T.; Nishimura, Y.; Mizoguchi, A.; Goshima, N.; et al. EGF Receptor Kinase Suppresses Ciliogenesis through Activation of USP8 Deubiquitinase. Nat. Commun. 2018, 9, 758. [Google Scholar] [CrossRef]
- Kunova Bosakova, M.; Varecha, M.; Hampl, M.; Duran, I.; Nita, A.; Buchtova, M.; Dosedelova, H.; Machat, R.; Xie, Y.; Ni, Z.; et al. Regulation of Ciliary Function by Fibroblast Growth Factor Signaling Identifies FGFR3-Related Disorders Achondroplasia and Thanatophoric Dysplasia as Ciliopathies. Hum. Mol. Genet. 2018, 27, 1093–1105. [Google Scholar] [CrossRef]
- Salati, M.; Caputo, F.; Baldessari, C.; Carotenuto, P.; Messina, M.; Caramaschi, S.; Dominici, M.; Bonetti, L.R. The Evolving Role of FGFR2 Inhibitors in Intrahepatic Cholangiocarcinoma: From Molecular Biology to Clinical Targeting. Cancer Manag. Res. 2021, 13, 7747–7757. [Google Scholar] [CrossRef]
- Neugebauer, J.M.; Amack, J.D.; Peterson, A.G.; Bisgrove, B.W.; Yost, H.J. FGF Signalling during Embryo Development Regulates Cilia Length in Diverse Epithelia. Nature 2009, 458, 651–654. [Google Scholar] [CrossRef]
- Gencer, S.; Oleinik, N.; Kim, J.; Panneer Selvam, S.; de Palma, R.; Dany, M.; Nganga, R.; Thomas, R.J.; Senkal, C.E.; Howe, P.H.; et al. TGF-β Receptor I/II Trafficking and Signaling at Primary Cilia Are Inhibited by Ceramide to Attenuate Cell Migration and Tumor Metastasis. Sci. Signal. 2017, 10, eaam7464. [Google Scholar] [CrossRef]
- Clement, C.A.; Ajbro, K.D.; Koefoed, K.; Vestergaard, M.L.; Veland, I.R.; Henriques de Jesus, M.P.R.; Pedersen, L.B.; Benmerah, A.; Andersen, C.Y.; Larsen, L.A.; et al. TGF-β Signaling Is Associated with Endocytosis at the Pocket Region of the Primary Cilium. Cell Rep. 2013, 3, 1806–1814. [Google Scholar] [CrossRef] [PubMed]
- Zanconato, F.; Cordenonsi, M.; Piccolo, S. YAP/TAZ at the Roots of Cancer. Cancer Cell 2016, 29, 783–803. [Google Scholar] [CrossRef] [PubMed]
- Habbig, S.; Bartram, M.P.; Müller, R.U.; Schwarz, R.; Andriopoulos, N.; Chen, S.; Sägmüller, J.G.; Hoehne, M.; Burst, V.; Liebau, M.C.; et al. NPHP4, a Cilia-Associated Protein, Negatively Regulates the Hippo Pathway. J. Cell Biol. 2011, 193, 633–642. [Google Scholar] [CrossRef] [PubMed]
- Bowers, A.J.; Boylan, J.F. Nek8, a NIMA Family Kinase Member, Is Overexpressed in Primary Human Breast Tumors. Gene 2004, 328, 135–142. [Google Scholar] [CrossRef]
- Carter, H.; Samayoa, J.; Hruban, R.H.; Karchin, R. Prioritization of Driver Mutations in Pancreatic Cancer Using Cancer-Specific High-Throughput Annotation of Somatic Mutations (CHASM). Cancer Biol. Ther. 2010, 10, 582–587. [Google Scholar] [CrossRef]
- Wood, L.D.; Parsons, D.W.; Jones, S.; Lin, J.; Sjöblom, T.; Leary, R.J.; Shen, D.; Boca, S.M.; Barber, T.; Ptak, J.; et al. The Genomic Landscapes of Human Breast and Colorectal Cancers. Science 2007, 318, 1108–1113. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Guan, Y.; Chen, X.; Yang, J.; Cheng, Y. DNA Repair Pathways in Cancer Therapy and Resistance. Front. Pharmacol. 2021, 11, 629266. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.A.; Collis, S.J. Ciliogenesis and the DNA Damage Response: A Stressful Relationship. Cilia 2016, 5, 19. [Google Scholar] [CrossRef]
- Chen, T.-Y.; Huang, B.-M.; Tang, T.K.; Chao, Y.-Y.; Xiao, X.-Y.; Lee, P.-R.; Yang, L.-Y.; Wang, C.-Y. Genotoxic Stress-Activated DNA-PK-P53 Cascade and Autophagy Cooperatively Induce Ciliogenesis to Maintain the DNA Damage Response. Cell Death Differ. 2021, 28, 1865–1879. [Google Scholar] [CrossRef]
- Abramowicz, I.; Carpenter, G.; Alfieri, M.; Colnaghi, R.; Outwin, E.; Parent, P.; Thauvin-Robinet, C.; Iaconis, D.; Franco, B.; O’Driscoll, M. Oral-Facial-Digital Syndrome Type I Cells Exhibit Impaired DNA Repair; Unanticipated Consequences of Defective OFD1 Outside of the Cilia Network. Hum. Mol. Genet. 2016, 26, ddw364. [Google Scholar] [CrossRef]
- Morleo, M.; Vieira, H.L.A.; Pennekamp, P.; Palma, A.; Bento-Lopes, L.; Omran, H.; Lopes, S.S.; Barral, D.C.; Franco, B. Crosstalk between Cilia and Autophagy: Implication for Human Diseases. Autophagy 2022, 19, 24–43. [Google Scholar] [CrossRef] [PubMed]
- Morleo, M.; Brillante, S.; Formisano, U.; Ferrante, L.; Carbone, F.; Iaconis, D.; Palma, A.; Buonomo, V.; Maione, A.S.; Grumati, P.; et al. Regulation of Autophagosome Biogenesis by OFD1-mediated Selective Autophagy. EMBO J. 2021, 40, e105120. [Google Scholar] [CrossRef] [PubMed]
- Morleo, M.; Franco, B. The Autophagy-Cilia Axis: An Intricate Relationship. Cells 2019, 8, 905. [Google Scholar] [CrossRef] [PubMed]
- Singla, V.; Romaguera-Ros, M.; Garcia-Verdugo, J.M.; Reiter, J.F. Ofd1, a Human Disease Gene, Regulates the Length and Distal Structure of Centrioles. Dev. Cell 2010, 18, 410–424. [Google Scholar] [CrossRef]
- Ferrante, M.I.; Zullo, A.; Barra, A.; Bimonte, S.; Messaddeq, N.; Studer, M.; Dollé, P.; Franco, B. Oral-Facial-Digital Type I Protein Is Required for Primary Cilia Formation and Left-Right Axis Specification. Nat. Genet. 2006, 38, 112–117. [Google Scholar] [CrossRef]
- Tang, Z.; Lin, M.G.; Stowe, T.R.; Chen, S.; Zhu, M.; Stearns, T.; Franco, B.; Zhong, Q. Autophagy Promotes Primary Ciliogenesis by Removing OFD1 from Centriolar Satellites. Nature 2013, 502, 254–257. [Google Scholar] [CrossRef]
- Meng, H.; Yang, X.; Liu, R.; Bao, J.; Hou, Y.; Sun, J.; Miao, S.; Qu, G. The Relationship Between Human Papillomavirus, OFD1 and Primary Ciliogenesis in the Progression of Oropharyngeal Cancer: A Retrospective Cohort Study. Pharmgenom. Pers. Med. 2020, 13, 633–644. [Google Scholar] [CrossRef]
- Kojima, R.; Hassan, E.; Ozawa, F.; Yamada-Namikawa, C.; Ogawa, S.; Mase, S.; Goto, S.; Nishikawa, R.; Inagaki, H.; Kato, Y.; et al. Abnormal Accumulation of OFD1 in Endometrial Cancer with Poor Prognosis Inhibits Ciliogenesis. Oncol. Lett. 2022, 24, 214. [Google Scholar] [CrossRef]
- Finetti, F.; Cassioli, C.; Cianfanelli, V.; Onnis, A.; Paccagnini, E.; Kabanova, A.; Baldari, C.T. The Intraflagellar Transport Protein IFT20 Controls Lysosome Biogenesis by Regulating the Post-Golgi Transport of Acid Hydrolases. Cell Death Differ. 2020, 27, 310–328. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Koga, H.; Kawaguchi, Y.; Tang, W.; Wong, E.; Gao, Y.-S.; Pandey, U.B.; Kaushik, S.; Tresse, E.; Lu, J.; et al. HDAC6 Controls Autophagosome Maturation Essential for Ubiquitin-Selective Quality-Control Autophagy. EMBO J. 2010, 29, 969–980. [Google Scholar] [CrossRef]
- Yang, H.; Zhang, F.; Long, H.; Lin, Y.; Liao, J.; Xia, H.; Huang, K. IFT20 Mediates the Transport of Cell Migration Regulators from the Trans-Golgi Network to the Plasma Membrane in Breast Cancer Cells. Front. Cell Dev. Biol. 2021, 9, 632198. [Google Scholar] [CrossRef]
- Aldana-Masangkay, G.I.; Sakamoto, K.M. The Role of HDAC6 in Cancer. J. Biomed. Biotechnol. 2011, 2011, 875824. [Google Scholar] [CrossRef]
- Peixoto, E.; Jin, S.; Thelen, K.; Biswas, A.; Richard, S.; Morleo, M.; Mansini, A.; Holtorf, S.; Carbone, F.; Pastore, N.; et al. HDAC6-Dependent Ciliophagy Is Involved in Ciliary Loss and Cholangiocarcinoma Growth in Human Cells and Murine Models. Am. J. Physiol.-Gastrointest. Liver Physiol. 2020, 318, G1022–G1033. [Google Scholar] [CrossRef]
- Hezel, A.F.; Gurumurthy, S.; Granot, Z.; Swisa, A.; Chu, G.C.; Bailey, G.; Dor, Y.; Bardeesy, N.; DePinho, R.A. Pancreatic Lkb1 Deletion Leads to Acinar Polarity Defects and Cystic Neoplasms. Mol. Cell. Biol. 2008, 28, 2414–2425. [Google Scholar] [CrossRef] [PubMed]
- Pampliega, O.; Orhon, I.; Patel, B.; Sridhar, S.; Díaz-Carretero, A.; Beau, I.; Codogno, P.; Satir, B.H.; Satir, P.; Cuervo, A.M. Functional Interaction between Autophagy and Ciliogenesis. Nature 2013, 502, 194–200. [Google Scholar] [CrossRef]
- Ma, M. Cilia and Polycystic Kidney Disease. Semin. Cell Dev. Biol. 2021, 110, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Walker, R.V.; Maranto, A.; Palicharla, V.R.; Hwang, S.-H.; Mukhopadhyay, S.; Qian, F. Cilia-Localized Counterregulatory Signals as Drivers of Renal Cystogenesis. Front. Mol. Biosci. 2022, 9. [Google Scholar] [CrossRef]
- Seeger-Nukpezah, T.; Geynisman, D.M.; Nikonova, A.S.; Benzing, T.; Golemis, E.A. The Hallmarks of Cancer: Relevance to the Pathogenesis of Polycystic Kidney Disease. Nat. Rev. Nephrol. 2015, 11, 515–534. [Google Scholar] [CrossRef]
- Gargalionis, A.N.; Korkolopoulou, P.; Farmaki, E.; Piperi, C.; Dalagiorgou, G.; Adamopoulos, C.; Levidou, G.; Saetta, A.; Fragkou, P.; Tsioli, P.; et al. Polycystin-1 and Polycystin-2 Are Involved in the Acquisition of Aggressive Phenotypes in Colorectal Cancer. Int. J. Cancer 2015, 136, 1515–1527. [Google Scholar] [CrossRef]
- Zhang, K.; Ye, C.; Zhou, Q.; Zheng, R.; Lv, X.; Chen, Y.; Hu, Z.; Guo, H.; Zhang, Z.; Wang, Y.; et al. PKD1 Inhibits Cancer Cells Migration and Invasion via Wnt Signaling Pathwayin Vitro. Cell Biochem. Funct. 2007, 25, 767–774. [Google Scholar] [CrossRef]
- Bian, G.-H.; Cao, G.; Lv, X.-Y.; Li, Q.-W.; Sun, H.; Xiao, Y.; Ai, J.-Z.; Yang, Q.-T.; Duan, J.-J.; Wang, Y.-D.; et al. Down-Regulation of Pkd2 by SiRNAs Suppresses Cell–Cell Adhesion in the Mouse Melanoma Cells. Mol. Biol. Rep. 2010, 37, 2387–2395. [Google Scholar] [CrossRef] [PubMed]
- Keith, D.S.; Torres, V.E.; King, B.F.; Zincki, H.; Farrow, G.M. Renal Cell Carcinoma in Autosomal Dominant Polycystic Kidney Disease. J. Am. Soc. Nephrol. 1994, 4, 1661–1669. [Google Scholar] [CrossRef] [PubMed]
- Hemal, A.K.; Khaitan, A.; Singh, I.; Kumar, M. Renal Cell Carcinoma in Cases of Adult Polycystic Kidney Disease: Changing Diagnostic and Therapeutic Implications. Urol. Int. 2000, 64, 9–12. [Google Scholar] [CrossRef]
- Orskov, B.; Sorensen, V.R.; Feldt-Rasmussen, B.; Strandgaard, S. Changes in Causes of Death and Risk of Cancer in Danish Patients with Autosomal Dominant Polycystic Kidney Disease and End-Stage Renal Disease. Nephrol. Dial. Transplant. 2012, 27, 1607–1613. [Google Scholar] [CrossRef] [PubMed]
- Hajj, P.; Ferlicot, S.; Massoud, W.; Awad, A.; Hammoudi, Y.; Charpentier, B.; Durrbach, A.; Droupy, S.; Benoît, G. Prevalence of Renal Cell Carcinoma in Patients with Autosomal Dominant Polycystic Kidney Disease and Chronic Renal Failure. Urology 2009, 74, 631–634. [Google Scholar] [CrossRef]
- Wetmore, J.B.; Calvet, J.P.; Yu, A.S.L.; Lynch, C.F.; Wang, C.J.; Kasiske, B.L.; Engels, E.A. Polycystic Kidney Disease and Cancer after Renal Transplantation. J. Am. Soc. Nephrol. 2014, 25, 2335–2341. [Google Scholar] [CrossRef]
- Gradilone, S.A.; Pisarello, M.J.; LaRusso, N.F. Primary Cilia in Tumor Biology: The Primary Cilium as a Therapeutic Target in Cholangiocarcinoma. Curr. Drug Targets 2015, 18, 958–963. [Google Scholar] [CrossRef]
- Pant, K.; Peixoto, E.; Richard, S.; Biswas, A.; O’Sullivan, M.G.; Giama, N.; Ha, Y.; Yin, J.; Carotenuto, P.; Salati, M.; et al. Histone Deacetylase Sirtuin 1 Promotes Loss of Primary Cilia in Cholangiocarcinoma. Hepatology 2021, 74, 3235–3248. [Google Scholar] [CrossRef]
- Youn, Y.H.; Hou, S.; Wu, C.-C.; Kawauchi, D.; Orr, B.A.; Robinson, G.W.; Finkelstein, D.; Taketo, M.M.; Gilbertson, R.J.; Roussel, M.F.; et al. Primary Cilia Control Translation and the Cell Cycle in Medulloblastoma. Genes Dev. 2022, 36, 737–751. [Google Scholar] [CrossRef]
- Moser, J.J.; Fritzler, M.J.; Rattner, J.B. Primary Ciliogenesis Defects Are Associated with Human Astrocytoma/Glioblastoma Cells. BMC Cancer 2009, 9, 448. [Google Scholar] [CrossRef]
- Rajagopalan, S.; Singh, A.; Khiabanian, H. Cilium Expression Score Predicts Glioma Survival. Front. Genet. 2021, 12, 758391. [Google Scholar] [CrossRef]
- Li, M.; Zhang, J.; Zhou, H.; Xiang, R. Primary Cilia–Related Pathways Moderate the Development and Therapy Resistance of Glioblastoma. Front. Oncol. 2021, 11, 718995. [Google Scholar] [CrossRef]
- Bakshi, A.; Chaudhary, S.C.; Rana, M.; Elmets, C.A.; Athar, M. Basal Cell Carcinoma Pathogenesis and Therapy Involving Hedgehog Signaling and Beyond. Mol. Carcinog. 2017, 56, 2543–2557. [Google Scholar] [CrossRef]
- Kuonen, F.; Huskey, N.E.; Shankar, G.; Jaju, P.; Whitson, R.J.; Rieger, K.E.; Atwood, S.X.; Sarin, K.Y.; Oro, A.E. Loss of Primary Cilia Drives Switching from Hedgehog to Ras/MAPK Pathway in Resistant Basal Cell Carcinoma. J. Investig. Dermatol. 2019, 139, 1439–1448. [Google Scholar] [CrossRef]
- Kim, J.; Dabiri, S.; Seeley, E.S. Primary Cilium Depletion Typifies Cutaneous Melanoma In Situ and Malignant Melanoma. PLoS ONE 2011, 6, e27410. [Google Scholar] [CrossRef]
- Zingg, D.; Debbache, J.; Peña-Hernández, R.; Antunes, A.T.; Schaefer, S.M.; Cheng, P.F.; Zimmerli, D.; Haeusel, J.; Calçada, R.R.; Tuncer, E.; et al. EZH2-Mediated Primary Cilium Deconstruction Drives Metastatic Melanoma Formation. Cancer Cell 2018, 34, 69–84. [Google Scholar] [CrossRef]
- Carotenuto, P.; Romano, A.; Barbato, A.; Quadrano, P.; Brillante, S.; Volpe, M.; Ferrante, L.; Tammaro, R.; Morleo, M.; De Cegli, R.; et al. Targeting the MITF/APAF-1 Axis as Salvage Therapy for MAPK Inhibitors in Resistant Melanoma. Cell Rep. 2022, 41, 111601. [Google Scholar] [CrossRef]
- Uebel, A.; Kewitz-Hempel, S.; Willscher, E.; Gebhardt, K.; Sunderkötter, C.; Gerloff, D. Resistance to BRAF Inhibitors: EZH2 and Its Downstream Targets as Potential Therapeutic Options in Melanoma. Int. J. Mol. Sci. 2023, 24, 1963. [Google Scholar] [CrossRef]
- Paul, C.; Tang, R.; Longobardi, C.; Lattanzio, R.; Eguether, T.; Turali, H.; Bremond, J.; Maurizy, C.; Gabola, M.; Poupeau, S.; et al. Loss of Primary Cilia Promotes Inflammation and Carcinogenesis. EMBO Rep. 2022, 23, e55687. [Google Scholar] [CrossRef]
- Dvorak, J.; Hadzi Nikolov, D.; Dusek, L.; Filipova, A.; Richter, I.; Buka, D.; Ryska, A.; Mokry, J.; Filip, S.; Melichar, B.; et al. Prognostic Significance of the Frequency of Primary Cilia in Cells of Small Bowel and Colorectal Adenocarcinoma. J. BUON 2016, 21, 1233–1241. [Google Scholar]
- Sénicourt, B.; Boudjadi, S.; Carrier, J.C.; Beaulieu, J.-F. Neoexpression of a Functional Primary Cilium in Colorectal Cancer Cells. Heliyon 2016, 2, e00109. [Google Scholar] [CrossRef]
- Rocha, C.; Papon, L.; Cacheux, W.; Marques Sousa, P.; Lascano, V.; Tort, O.; Giordano, T.; Vacher, S.; Lemmers, B.; Mariani, P.; et al. Tubulin Glycylases Are Required for Primary Cilia, Control of Cell Proliferation and Tumor Development in Colon. EMBO J. 2014, 33, 2247–2260. [Google Scholar] [CrossRef] [PubMed]
- Previdi, M.C.; Carotenuto, P.; Zito, D.; Pandolfo, R.; Braconi, C. Noncoding RNAs as Novel Biomarkers in Pancreatic Cancer: What Do We Know? Future Oncology. 2017, 13, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Carotenuto, P.; Fassan, M.; Pandolfo, R.; Lampis, A.; Vicentini, C.; Cascione, L.; Paulus-Hock, V.; Boulter, L.; Guest, R.; Quagliata, L.; et al. Wnt Signalling Modulates Transcribed-Ultraconserved Regions in Hepatobiliary Cancers. Gut 2017, 66, 1268–1277. [Google Scholar] [CrossRef] [PubMed]
- Lampis, A.; Carotenuto, P.; Vlachogiannis, G.; Cascione, L.; Hedayat, S.; Burke, R.; Clarke, P.; Bosma, E.; Simbolo, M.; Scarpa, A.; et al. MIR21 Drives Resistance to Heat Shock Protein 90 Inhibition in Cholangiocarcinoma. Gastroenterology 2018, 154, 1066–1079. [Google Scholar] [CrossRef] [PubMed]
- Carotenuto, P.; Hedayat, S.; Fassan, M.; Cardinale, V.; Lampis, A.; Guzzardo, V.; Vicentini, C.; Scarpa, A.; Cascione, L.; Costantini, D.; et al. Modulation of Biliary Cancer Chemo-Resistance Through MicroRNA-Mediated Rewiring of the Expansion of CD133+ Cells. Hepatology 2020, 72. [Google Scholar] [CrossRef]
- Barbato, A.; Piscopo, F.; Salati, M.; Reggiani-Bonetti, L.; Franco, B.; Carotenuto, P. Micro-RNA in Cholangiocarcinoma: Implications for Diagnosis, Prognosis, and Therapy. J. Mol. Pathol. 2022, 3, 88–103. [Google Scholar] [CrossRef]
- Mansini, A.P.; Lorenzo Pisarello, M.J.; Thelen, K.M.; Cruz-Reyes, M.; Peixoto, E.; Jin, S.; Howard, B.N.; Trussoni, C.E.; Gajdos, G.B.; LaRusso, N.F.; et al. MicroRNA (MiR)-433 and MiR-22 Dysregulations Induce Histone-Deacetylase-6 Overexpression and Ciliary Loss in Cholangiocarcinoma. Hepatology 2018, 68, 561–573. [Google Scholar] [CrossRef] [PubMed]
- Seeley, E.S.; Carrière, C.; Goetze, T.; Longnecker, D.S.; Korc, M. Pancreatic Cancer and Precursor Pancreatic Intraepithelial Neoplasia Lesions Are Devoid of Primary Cilia. Cancer Res. 2009, 69, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Quilichini, E.; Fabre, M.; Dirami, T.; Stedman, A.; de Vas, M.; Ozguc, O.; Pasek, R.C.; Cereghini, S.; Morillon, L.; Guerra, C.; et al. Pancreatic Ductal Deletion of Hnf1b Disrupts Exocrine Homeostasis, Leads to Pancreatitis, and Facilitates Tumorigenesis. Cell Mol. Gastroenterol. Hepatol. 2019, 8, 487–511. [Google Scholar] [CrossRef]
- Carotenuto, P.; Roma, C.; Rachiglio, A.M.; Tatangelo, F.; Pinto, C.; Ciardiello, F.; Nappi, O.; Iaffaioli, V.; Botti, G.; Normanno, N. Detection of KRAS Mutations in Colorectal Carcinoma Patients with an Integrated PCR/Sequencing and Real-Time PCR Approach. Pharmacogenomics 2010, 11, 1169–1179. [Google Scholar] [CrossRef] [PubMed]
- Emoto, K.; Masugi, Y.; Yamazaki, K.; Effendi, K.; Tsujikawa, H.; Tanabe, M.; Kitagawa, Y.; Sakamoto, M. Presence of Primary Cilia in Cancer Cells Correlates with Prognosis of Pancreatic Ductal Adenocarcinoma. Hum. Pathol. 2014, 45, 817–825. [Google Scholar] [CrossRef] [PubMed]
- Schimmack, S.; Kneller, S.; Dadabaeva, N.; Bergmann, F.; Taylor, A.; Hackert, T.; Werner, J.; Strobel, O. Epithelial to Stromal Re-Distribution of Primary Cilia during Pancreatic Carcinogenesis. PLoS ONE 2016, 11, e0164231. [Google Scholar] [CrossRef]
- Ghidini, M.; Cascione, L.; Carotenuto, P.; Lampis, A.; Trevisani, F.; Previdi, M.C.; Hahne, J.C.; Said-Huntingford, I.; Raj, M.; Zerbi, A.; et al. Characterisation of the Immune-Related Transcriptome in Resected Biliary Tract Cancers. Eur. J. Cancer 2017, 86, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Nakazono, K.; Tokuda, M.; Mashima, Y.; Dynlacht, B.D.; Itoh, H. HDAC 2 Promotes Loss of Primary Cilia in Pancreatic Ductal Adenocarcinoma. EMBO Rep. 2017, 18, 334–343. [Google Scholar] [CrossRef]
- Deng, Y.-Z.; Cai, Z.; Shi, S.; Jiang, H.; Shang, Y.-R.; Ma, N.; Wang, J.-J.; Guan, D.-X.; Chen, T.-W.; Rong, Y.-F.; et al. Cilia Loss Sensitizes Cells to Transformation by Activating the Mevalonate Pathway. J. Exp. Med. 2018, 215, 177–195. [Google Scholar] [CrossRef]
- Menzl, I.; Lebeau, L.; Pandey, R.; Hassounah, N.B.; Li, F.W.; Nagle, R.; Weihs, K.; McDermott, K.M. Loss of Primary Cilia Occurs Early in Breast Cancer Development. Cilia 2014, 3, 7. [Google Scholar] [CrossRef]
- Yuan, K.; Frolova, N.; Xie, Y.; Wang, D.; Cook, L.; Kwon, Y.-J.; Steg, A.D.; Serra, R.; Frost, A.R. Primary Cilia Are Decreased in Breast Cancer: Analysis of a Collection of Human Breast Cancer Cell Lines and Tissues. J. Histochem. Cytochem. 2010, 58, 857–870. [Google Scholar] [CrossRef]
- Légaré, S.; Chabot, C.; Basik, M. SPEN, a New Player in Primary Cilia Formation and Cell Migration in Breast Cancer. Breast Cancer Res. 2017, 19, 104. [Google Scholar] [CrossRef]
- Hassounah, N.B.; Nagle, R.; Saboda, K.; Roe, D.J.; Dalkin, B.L.; McDermott, K.M. Primary Cilia Are Lost in Preinvasive and Invasive Prostate Cancer. PLoS ONE 2013, 8, e68521. [Google Scholar] [CrossRef]
- Basten, S.G.; Willekers, S.; Vermaat, J.S.; Slaats, G.G.; Voest, E.E.; van Diest, P.J.; Giles, R.H. Reduced Cilia Frequencies in Human Renal Cell Carcinomas versus Neighboring Parenchymal Tissue. Cilia 2013, 2, 2. [Google Scholar] [CrossRef] [PubMed]
- Nacchio, M.; Pisapia, P.; Pepe, F.; Russo, G.; Vigliar, E.; Porcelli, T.; Luongo, C.; Iaccarino, A.; Pagni, F.; Salvatore, D.; et al. Predictive Molecular Pathology in Metastatic Thyroid Cancer: The Role of RET Fusions. Expert. Rev. Endocrinol. Metab. 2022, 17, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Park, K.C.; Sul, H.J.; Hong, H.J.; Kim, K.-H.; Kero, J.; Shong, M. Loss of Primary Cilia Promotes Mitochondria-Dependent Apoptosis in Thyroid Cancer. Sci. Rep. 2021, 11, 4181. [Google Scholar] [CrossRef] [PubMed]
- Nita, A.; Abraham, S.P.; Krejci, P.; Bosakova, M. Oncogenic FGFR Fusions Produce Centrosome and Cilia Defects by Ectopic Signaling. Cells 2021, 10, 1445. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-M.; Su, F.; Kalyana-Sundaram, S.; Khazanov, N.; Ateeq, B.; Cao, X.; Lonigro, R.J.; Vats, P.; Wang, R.; Lin, S.-F.; et al. Identification of Targetable FGFR Gene Fusions in Diverse Cancers. Cancer Discov. 2013, 3, 636–647. [Google Scholar] [CrossRef] [PubMed]
- de Andrea, C.E.; Zhu, J.-F.; Jin, H.; Bovée, J.V.; Jones, K.B. Cell Cycle Deregulation and Mosaic Loss of Ext1 Drive Peripheral Chondrosarcomagenesis in the Mouse and Reveal an Intrinsic Cilia Deficiency. J. Pathol. 2015, 236, 210–218. [Google Scholar] [CrossRef]
- Xiang, W.; Guo, F.; Cheng, W.; Zhang, J.; Huang, J.; Wang, R.; Ma, Z.; Xu, K. HDAC6 Inhibition Suppresses Chondrosarcoma by Restoring the Expression of Primary Cilia. Oncol. Rep. 2017, 38, 229–236. [Google Scholar] [CrossRef]
- Remo, A.; Grillo, F.; Mastracci, L.; Simbolo, M.; Fassan, M.; Cecchini, M.P.; Miscio, G.; Sassano, A.; Parente, P.; Vanoli, A.; et al. Loss of Primary Cilia Potentiates BRAF/MAPK Pathway Activation in Rhabdoid Colorectal Carcinoma: A Series of 21 Cases Showing Ciliary Rootlet CoiledCoil (CROCC) Alterations. Genes 2023, 14, 984. [Google Scholar] [CrossRef]
- Pancione, M.; Di Blasi, A.; Sabatino, L.; Fucci, A.; Dalena, A.M.; Palombi, N.; Carotenuto, P.; Aquino, G.; Daniele, B.; Normanno, N.; et al. A Novel Case of Rhabdoid Colon Carcinoma Associated with a Positive CpG Island Methylator Phenotype and BRAF Mutation. Hum. Pathol. 2011, 42, 1047–1052. [Google Scholar] [CrossRef]
- Fu, W.; Asp, P.; Canter, B.; Dynlacht, B.D. Primary Cilia Control Hedgehog Signaling during Muscle Differentiation and Are Deregulated in Rhabdomyosarcoma. Proc. Natl. Acad. Sci. USA 2014, 111, 9151–9156. [Google Scholar] [CrossRef]
- Liu, X.; Shen, Q.; Yu, T.; Huang, H.; Zhang, Z.; Ding, J.; Tang, Y.; Xu, N.; Yue, S. Small GTPase Arl6 Controls RH30 Rhabdomyosarcoma Cell Growth through Ciliogenesis and Hedgehog Signaling. Cell Biosci. 2016, 6, 61. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Zhang, L.; Wei, Q.; Shao, A. O6-Methylguanine-DNA Methyltransferase (MGMT): Challenges and New Opportunities in Glioma Chemotherapy. Front. Oncol. 2020, 9, 1547. [Google Scholar] [CrossRef]
- Loskutov, Y.V.; Griffin, C.L.; Marinak, K.M.; Bobko, A.; Margaryan, N.V.; Geldenhuys, W.J.; Sarkaria, J.N.; Pugacheva, E.N. LPA Signaling Is Regulated through the Primary Cilium: A Novel Target in Glioblastoma. Oncogene 2018, 37, 1457–1471. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Roine, N.; Mäkelä, T.P. CCRK Depletion Inhibits Glioblastoma Cell Proliferation in a Cilium-dependent Manner. EMBO Rep. 2013, 14, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Caligiuri, M.; Becker, F.; Murthi, K.; Kaplan, F.; Dedier, S.; Kaufmann, C.; Machl, A.; Zybarth, G.; Richard, J.; Bockovich, N.; et al. A Proteome-Wide CDK/CRK-Specific Kinase Inhibitor Promotes Tumor Cell Death in the Absence of Cell Cycle Progression. Chem. Biol. 2005, 12, 1103–1115. [Google Scholar] [CrossRef] [PubMed]
- Mansini, A.P.; Peixoto, E.; Jin, S.; Richard, S.; Gradilone, S.A. The Chemosensory Function of Primary Cilia Regulates Cholangiocyte Migration, Invasion, and Tumor Growth. Hepatology 2019, 69, 1582–1598. [Google Scholar] [CrossRef]
- Sabanovic, B.; Giulietti, M.; Piva, F. Role of Primary Cilium in Pancreatic Ductal Adenocarcinoma (Review). Int. J. Oncol. 2020, 57, 1095–1102. [Google Scholar] [CrossRef]
- Do, P.A.; Lee, C.H. The Role of CDK5 in Tumours and Tumour Microenvironments. Cancers 2020, 13, 101. [Google Scholar] [CrossRef]
- Husson, H.; Moreno, S.; Smith, L.A.; Smith, M.M.; Russo, R.J.; Pitstick, R.; Sergeev, M.; Ledbetter, S.R.; Bukanov, N.O.; Lane, M.; et al. Reduction of Ciliary Length through Pharmacologic or Genetic Inhibition of CDK5 Attenuates Polycystic Kidney Disease in a Model of Nephronophthisis. Hum. Mol. Genet. 2016, 25, 2245–2255. [Google Scholar] [CrossRef]
- Wang, E.X.; Turner, J.S.; Brautigan, D.L.; Fu, Z. Modulation of Primary Cilia by Alvocidib Inhibition of CILK1. Int. J. Mol. Sci. 2022, 23, 8121. [Google Scholar] [CrossRef]
- Snedecor, E.R.; Sung, C.C.; Moncayo, A.; Rothstein, B.E.; Mockler, D.C.; Tonnesen, M.G.; Jones, E.C.; Fujita, M.; Clark, R.A.; Shroyer, K.R.; et al. Loss of Primary Cilia in Melanoma Cells Is Likely Independent of Proliferation and Cell Cycle Progression. J. Investig. Dermatol. 2015, 135, 1456–1458. [Google Scholar] [CrossRef]
- Spranger, S.; Bao, R.; Gajewski, T.F. Melanoma-Intrinsic β-Catenin Signalling Prevents Anti-Tumour Immunity. Nature 2015, 523, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Macias, R.I.R.; Banales, J.M.; Sangro, B.; Muntané, J.; Avila, M.A.; Lozano, E.; Perugorria, M.J.; Padillo, F.J.; Bujanda, L.; Marin, J.J.G. The Search for Novel Diagnostic and Prognostic Biomarkers in Cholangiocarcinoma. Biochim. Et. Biophys. Acta (BBA)-Mol. Basis Dis. 2018, 1864, 1468–1477. [Google Scholar] [CrossRef]
- Nobutani, K.; Shimono, Y.; Yoshida, M.; Mizutani, K.; Minami, A.; Kono, S.; Mukohara, T.; Yamasaki, T.; Itoh, T.; Takao, S.; et al. Absence of Primary Cilia in Cell Cycle-Arrested Human Breast Cancer Cells. Genes Cells 2014, 19, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Franco, B.; Clarke, P.; Carotenuto, P. Pemigatinib. Fibroblast Growth Factor Receptor Inhibitor, Treatment of Cholangiocarcinoma. Drugs Future 2019, 44, 923. [Google Scholar] [CrossRef]
- Schraml, P.; Frew, I.J.; Thoma, C.R.; Boysen, G.; Struckmann, K.; Krek, W.; Moch, H. Sporadic Clear Cell Renal Cell Carcinoma but Not the Papillary Type Is Characterized by Severely Reduced Frequency of Primary Cilia. Mod. Pathol. 2009, 22, 31–36. [Google Scholar] [CrossRef]
- Corrigan, M.A.; Ferradaes, T.M.; Riffault, M.; Hoey, D.A. Ciliotherapy Treatments to Enhance Biochemically- and Biophysically-Induced Mesenchymal Stem Cell Osteogenesis: A Comparison Study. Cell Mol. Bioeng. 2019, 12, 53–67. [Google Scholar] [CrossRef]
- Pala, R.; Mohieldin, A.M.; Sherpa, R.T.; Kathem, S.H.; Shamloo, K.; Luan, Z.; Zhou, J.; Zheng, J.-G.; Ahsan, A.; Nauli, S.M. Ciliotherapy: Remote Control of Primary Cilia Movement and Function by Magnetic Nanoparticles. ACS Nano 2019, 13, 3555–3572. [Google Scholar] [CrossRef]
- Cebotaru, L.; Liu, Q.; Yanda, M.K.; Boinot, C.; Outeda, P.; Huso, D.L.; Watnick, T.; Guggino, W.B.; Cebotaru, V. Inhibition of Histone Deacetylase 6 Activity Reduces Cyst Growth in Polycystic Kidney Disease. Kidney Int. 2016, 90, 90–99. [Google Scholar] [CrossRef]
- Kramann, R.; Fleig, S.V.; Schneider, R.K.; Fabian, S.L.; DiRocco, D.P.; Maarouf, O.; Wongboonsin, J.; Ikeda, Y.; Heckl, D.; Chang, S.L.; et al. Pharmacological GLI2 Inhibition Prevents Myofibroblast Cell-Cycle Progression and Reduces Kidney Fibrosis. J. Clin. Investig. 2015, 125, 2935–2951. [Google Scholar] [CrossRef]
- Woo, Y.M.; Bae, J.-B.; Oh, Y.-H.; Lee, Y.-G.; Lee, M.J.; Park, E.Y.; Choi, J.-K.; Lee, S.; Shin, Y.; Lyu, J.; et al. Genome-Wide Methylation Profiling of ADPKD Identified Epigenetically Regulated Genes Associated with Renal Cyst Development. Hum. Genet. 2014, 133, 281–297. [Google Scholar] [CrossRef]
- Ischenko, I.; Petrenko, O.; Hayman, M.J. A MEK/PI3K/HDAC Inhibitor Combination Therapy for KRAS Mutant Pancreatic Cancer Cells. Oncotarget 2015, 6, 15814–15827. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.A.; Willemarck, N.; Talebi, A.; Marchand, A.; Binda, M.M.; Dehairs, J.; Rueda-Rincon, N.; Daniels, V.W.; Bagadi, M.; Raj, D.B.T.G.; et al. Identification of Drugs That Restore Primary Cilium Expression in Cancer Cells. Oncotarget 2016, 7, 9975–9992. [Google Scholar] [CrossRef]
- Wu, V.M.; Chen, S.C.; Arkin, M.R.; Reiter, J.F. Small Molecule Inhibitors of Smoothened Ciliary Localization and Ciliogenesis. Proc. Natl. Acad. Sci. USA 2012, 109, 13644–13649. [Google Scholar] [CrossRef] [PubMed]
- Zalewska, M.; Siara, M.; Sajewicz, W. G Protein-Coupled Receptors: Abnormalities in Signal Transmission, Disease States and Pharmacotherapy. Acta Pol. Pharm. 2014, 71, 229–243. [Google Scholar] [PubMed]
- Thomsen, W.; Frazer, J.; Unett, D. Functional Assays for Screening GPCR Targets. Curr. Opin. Biotechnol. 2005, 16, 655–665. [Google Scholar] [CrossRef]
- Geoerger, B.; Aerts, I.; Casanova, M.; Chisholm, J.C.; Hargrave, D.R.; Leary, S.; Ashley, D.M.; Bouffet, E.; MacDonald, T.; Hurh, E.; et al. A Phase I/II Study of LDE225, a Smoothened (Smo) Antagonist, in Pediatric Patients with Recurrent Medulloblastoma (MB) or Other Solid Tumors. J. Clin. Oncol. 2012, 30, 9519. [Google Scholar] [CrossRef]
- Chen, J.K.; Taipale, J.; Young, K.E.; Maiti, T.; Beachy, P.A. Small Molecule Modulation of Smoothened Activity. Proc. Natl. Acad. Sci. USA 2002, 99, 14071–14076. [Google Scholar] [CrossRef]
- Büttner, A.; Seifert, K.; Cottin, T.; Sarli, V.; Tzagkaroulaki, L.; Scholz, S.; Giannis, A. Synthesis and Biological Evaluation of SANT-2 and Analogues as Inhibitors of the Hedgehog Signaling Pathway. Bioorg. Med. Chem. 2009, 17, 4943–4954. [Google Scholar] [CrossRef]
- Rodon, J.; Tawbi, H.A.; Thomas, A.L.; Stoller, R.G.; Turtschi, C.P.; Baselga, J.; Sarantopoulos, J.; Mahalingam, D.; Shou, Y.; Moles, M.A.; et al. A Phase I, Multicenter, Open-Label, First-in-Human, Dose-Escalation Study of the Oral Smoothened Inhibitor Sonidegib (LDE225) in Patients with Advanced Solid Tumors. Clin. Cancer Res. 2014, 20, 1900–1909. [Google Scholar] [CrossRef]
- LoRusso, P.M.; Rudin, C.M.; Reddy, J.C.; Tibes, R.; Weiss, G.J.; Borad, M.J.; Hann, C.L.; Brahmer, J.R.; Chang, I.; Darbonne, W.C.; et al. Phase I Trial of Hedgehog Pathway Inhibitor Vismodegib (GDC-0449) in Patients with Refractory, Locally Advanced or Metastatic Solid Tumors. Clin. Cancer Res. 2011, 17, 2502–2511. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Davidow, L.; Arvanites, A.C.; Blanchard, J.; Lam, K.; Xu, K.; Oza, V.; Yoo, J.W.; Ng, J.M.Y.; Curran, T.; et al. Glucocorticoid Compounds Modify Smoothened Localization and Hedgehog Pathway Activity. Chem. Biol. 2012, 19, 972–982. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Roberts, C.W.M. Targeting EZH2 in Cancer. Nat. Med. 2016, 22, 128–134. [Google Scholar] [CrossRef] [PubMed]

| Gene/Gene Signatures | Molecular Pathway | Type of Cancer | Clinical Implication | References |
|---|---|---|---|---|
| PKD1; PKD2 | Wnt; RTK; Mechanotransduction | Colorectal, Melanoma | Therapeutic Targeting | [118] |
| PTCH1; CTNNB1 | HH; Wnt | Medulloblastoma | Diagnostic biomarkers | [68] |
| SHH; PTCH1; SMO; GLI | HH | Glioblastoma | Therapeutic Targeting | [132] |
| MGMT | HH | Glioblastoma/Glioma | Drug Resistance | [132,172] |
| LPAR1 | GPCR | Glioblastoma | Therapeutic Targeting | [173] |
| CCRK | AR, Wnt, AKT, EZH2, and NF-κB, HH | Glioblastoma | Therapeutic Targeting | [174,175] |
| LRGUK, NSUN7, LRRC27, SPAG17, EFHB, IFT27, DZIP1L, FOLR1, RGS22, TEX9, GALNT3, and GLB1L | Cilium-Associated Genes | Glioma | Prognostic Biomarkers | [131] |
| EZH2 | Wnt/b-Catenin | Melanoma | Therapeutic Targeting; Diagnostic Biomarkers | [136] |
| PTCH, SMO; GLI1 | HH | Colorectal Cancer | Diagnostic and Predictive Biomarkers | [141] |
| IFT88 | HH; MAPK | Cholangiocarcinoma; Thyroid Cancers | Diagnostic Biomarker | [176] |
| HDAC6 | HH; MAPK | Cholangiocarcinoma; Chondrosarcoma | Therapeutic Targeting; Diagnostic, Predictive Biomarkers | [57,113] |
| SIRT1 | HH; AKT; IL6 | Cholangiocarcinoma | Therapeutic Targeting | [128] |
| HDAC2 | KRAS | Pancreatic Ductal Adenocarcinoma | Therapeutic Targeting | [155] |
| AURKA; INPP5E | Cilium-Associated Genes | Pancreatic Ductal Adenocarcinoma | Prognostic Biomarkers | [177] |
| SPEN | ERα | Breast Cancer | Prognostic, predictive Biomarkers | [159] |
| CDK5 | Cell-Cycle-Related | Several Cancers | Prognostic, predictive Biomarkers; Therapeutic targeting | [178,179] |
| CILK1 | HH | Several Cancers | Therapeutic targeting | [178,180] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carotenuto, P.; Gradilone, S.A.; Franco, B. Cilia and Cancer: From Molecular Genetics to Therapeutic Strategies. Genes 2023, 14, 1428. https://doi.org/10.3390/genes14071428
Carotenuto P, Gradilone SA, Franco B. Cilia and Cancer: From Molecular Genetics to Therapeutic Strategies. Genes. 2023; 14(7):1428. https://doi.org/10.3390/genes14071428
Chicago/Turabian StyleCarotenuto, Pietro, Sergio A. Gradilone, and Brunella Franco. 2023. "Cilia and Cancer: From Molecular Genetics to Therapeutic Strategies" Genes 14, no. 7: 1428. https://doi.org/10.3390/genes14071428
APA StyleCarotenuto, P., Gradilone, S. A., & Franco, B. (2023). Cilia and Cancer: From Molecular Genetics to Therapeutic Strategies. Genes, 14(7), 1428. https://doi.org/10.3390/genes14071428






