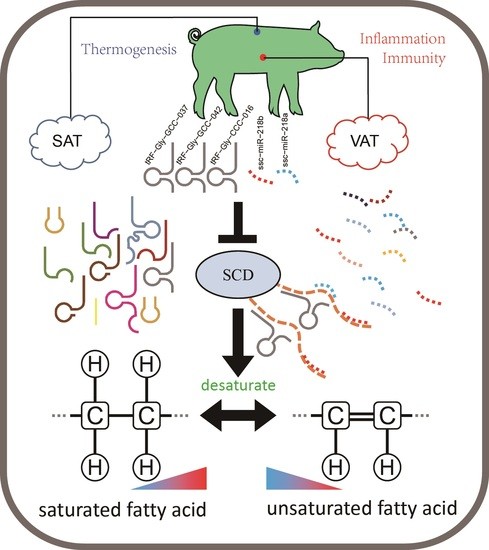
tsRNA Landscape and Potential Function Network in Subcutaneous and Visceral Pig Adipose Tissue
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Animals and Treatment
2.3. tsRNA-seq Data, miRNA Data, and mRNA Data
2.4. tsRNA-seq Data Analysis and Target Prediction
2.5. Tissue Section and Staining
2.6. Analysis of Fatty Acids
2.7. Statistical Analysis
3. Results
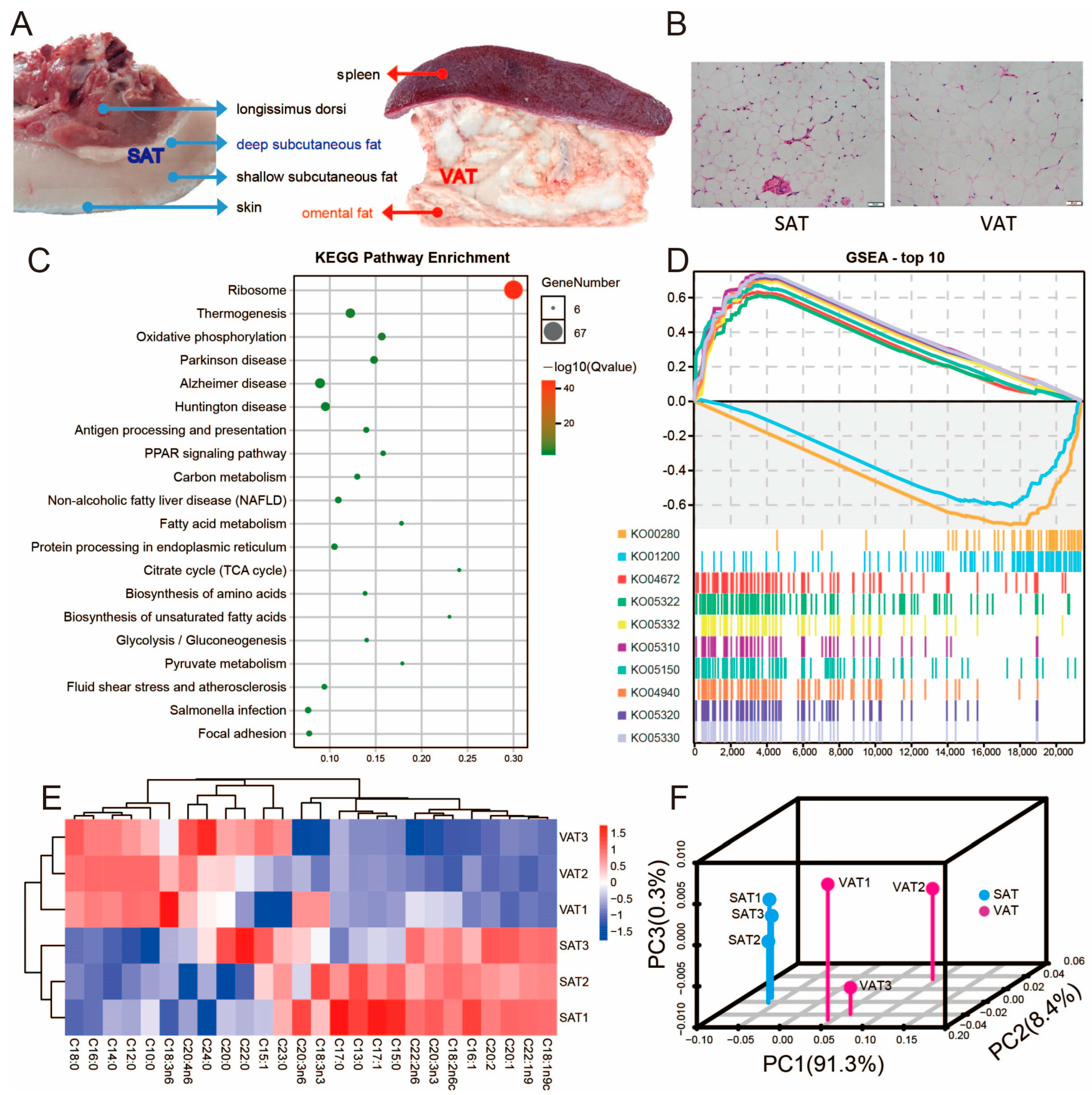
3.1. Morphological Characteristics of SAT and VAT in Pigs
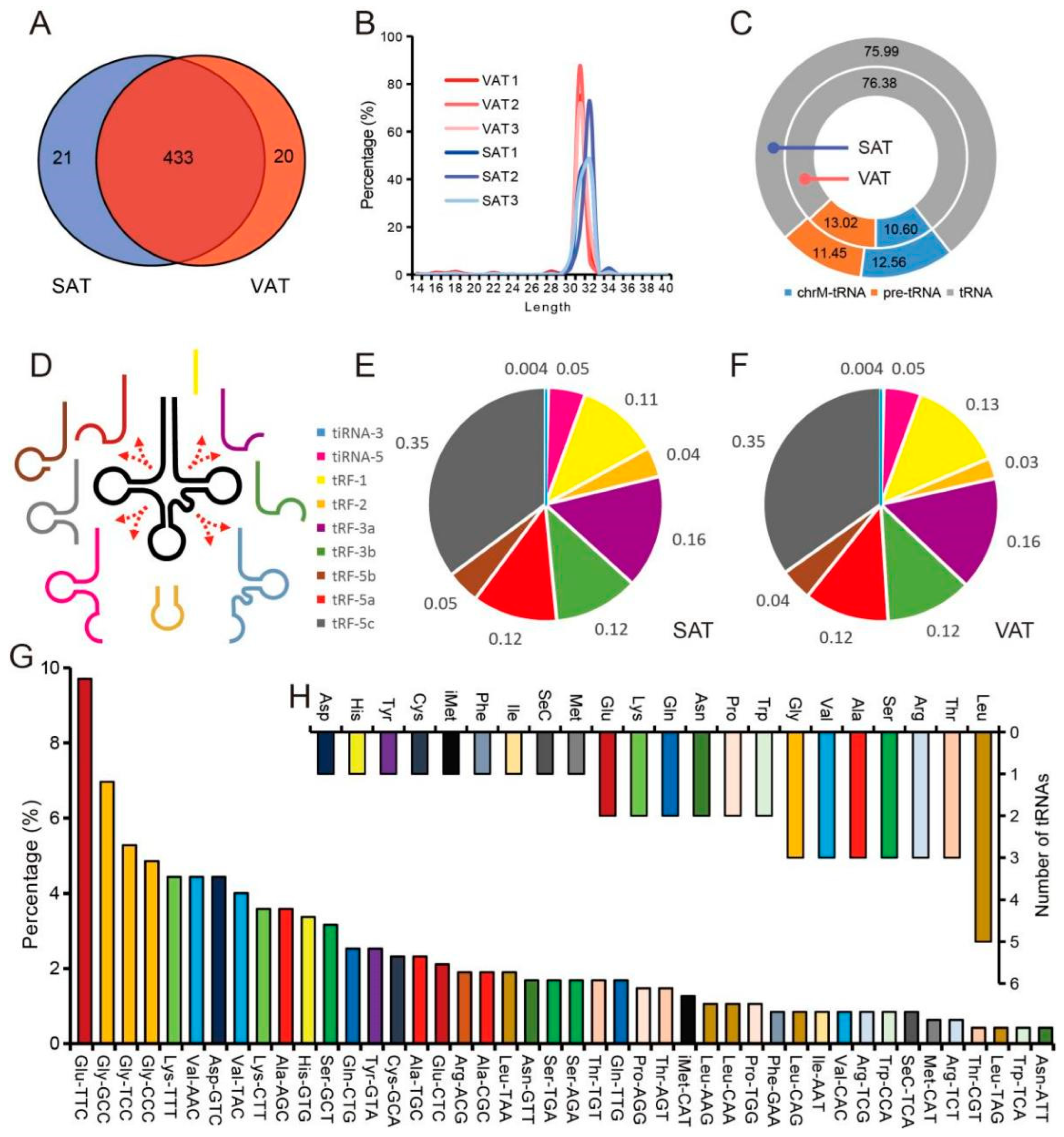
3.2. Characteristics of tsRNA in Pig Adipose Tissue
3.3. High Expression of tsRNA in Pig Adipose Tissue
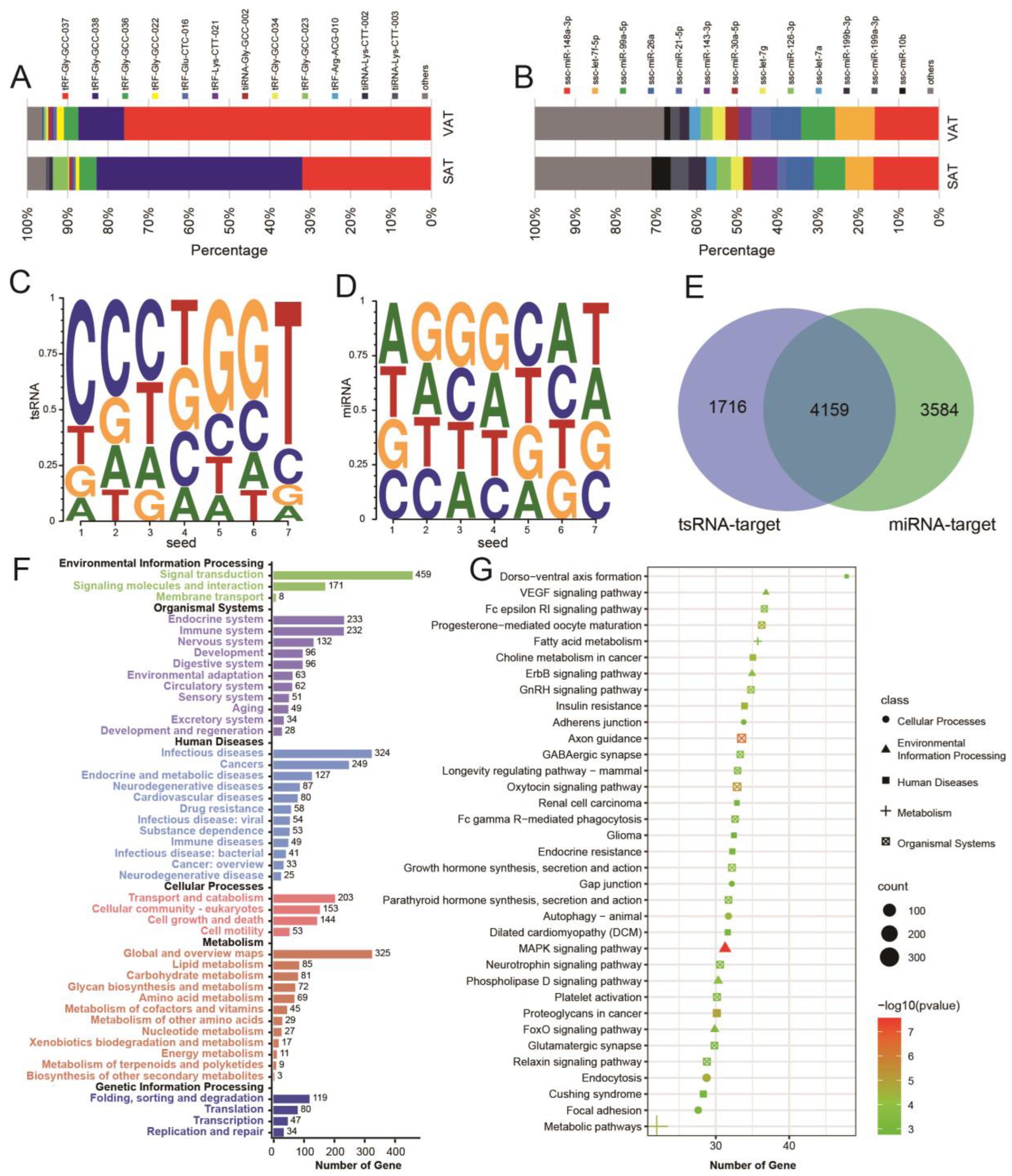
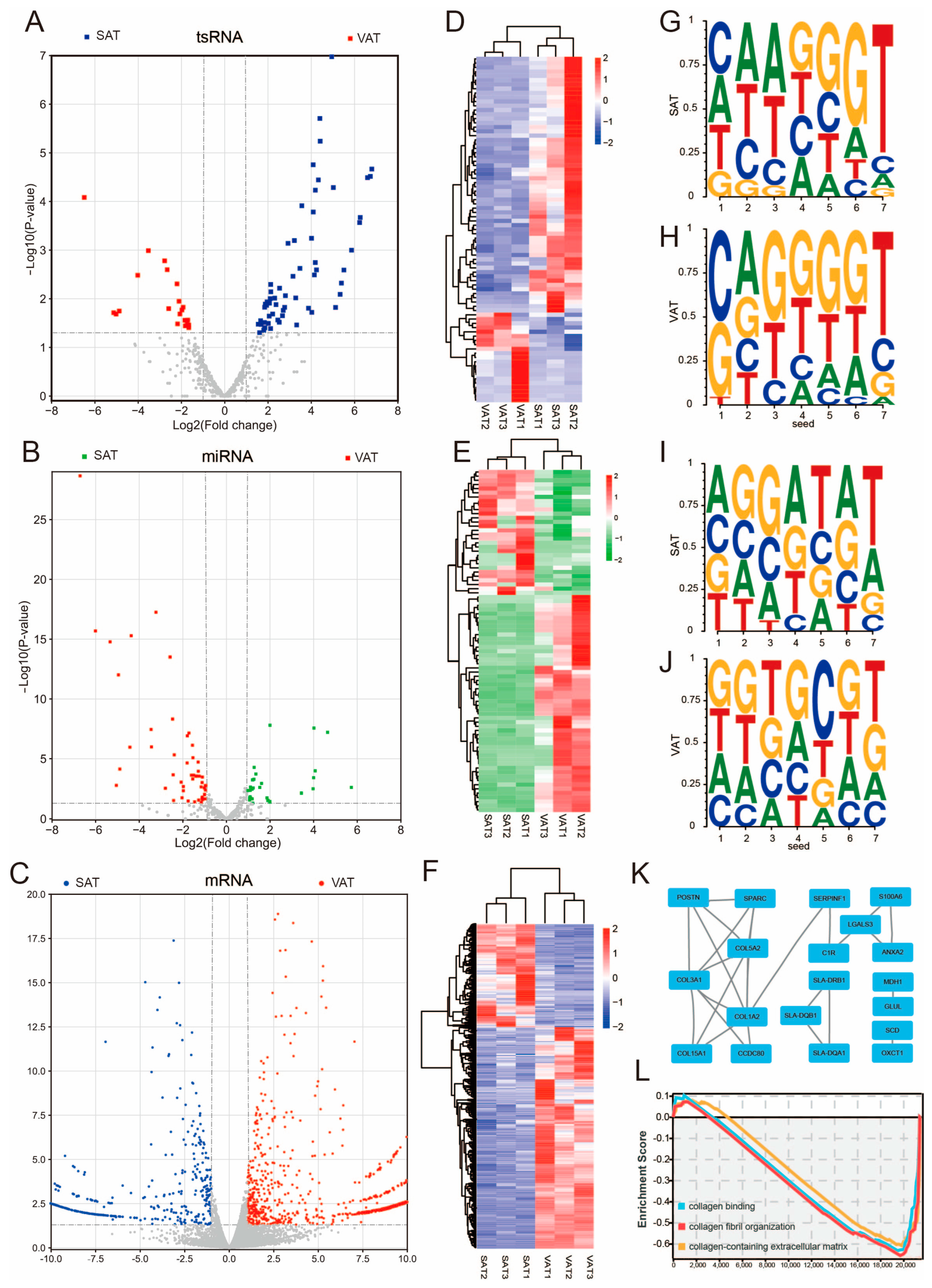
3.4. Differential Expression of tsRNAs, miRNAs, and mRNAs in Subcutaneous Adipose Tissue and Visceral Adipose Tissue
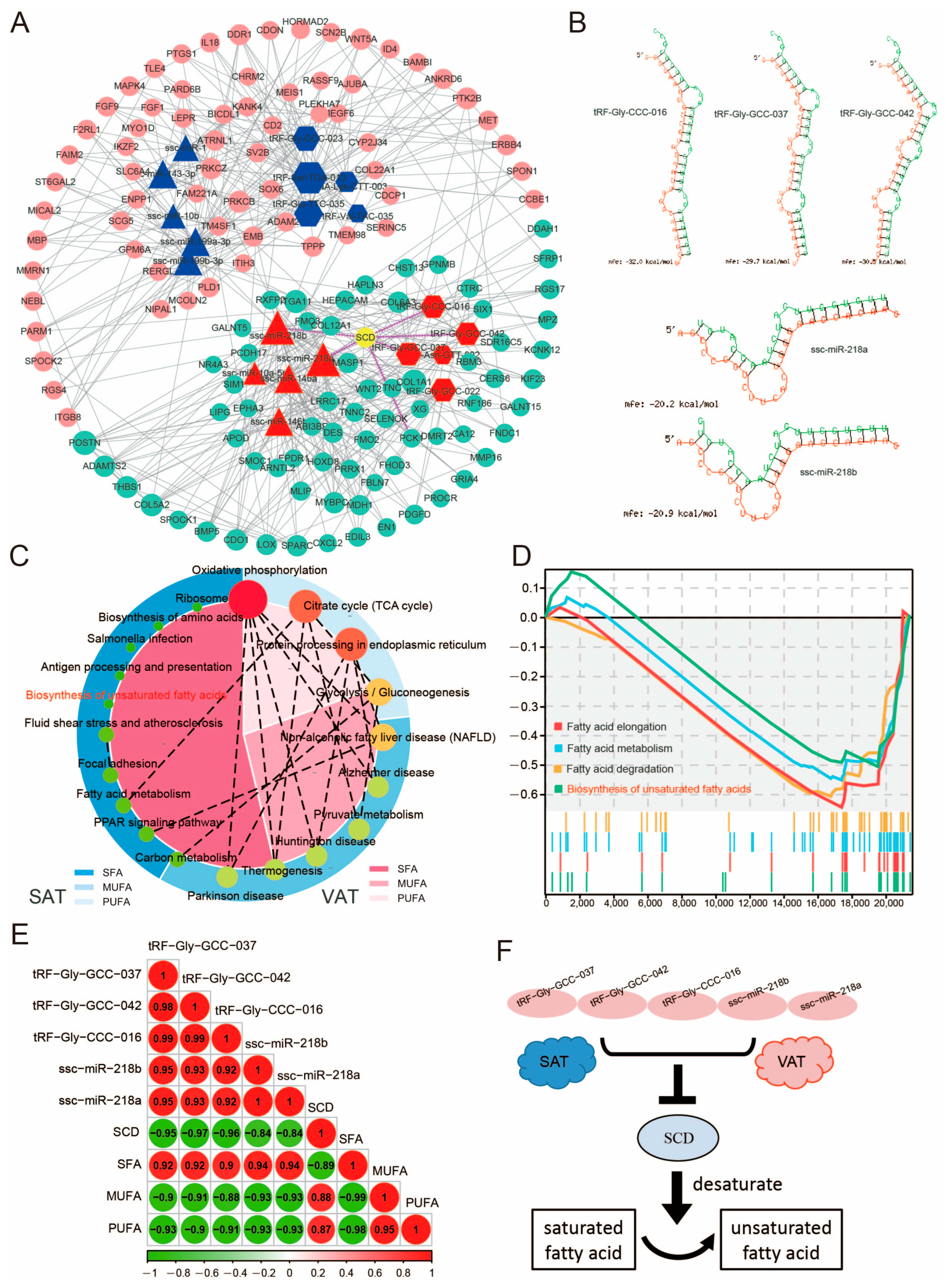
3.5. tsRNA/miRNA/mRNA Regulatory Network of Subcutaneous Adipose Tissue and Visceral Adipose Tissue
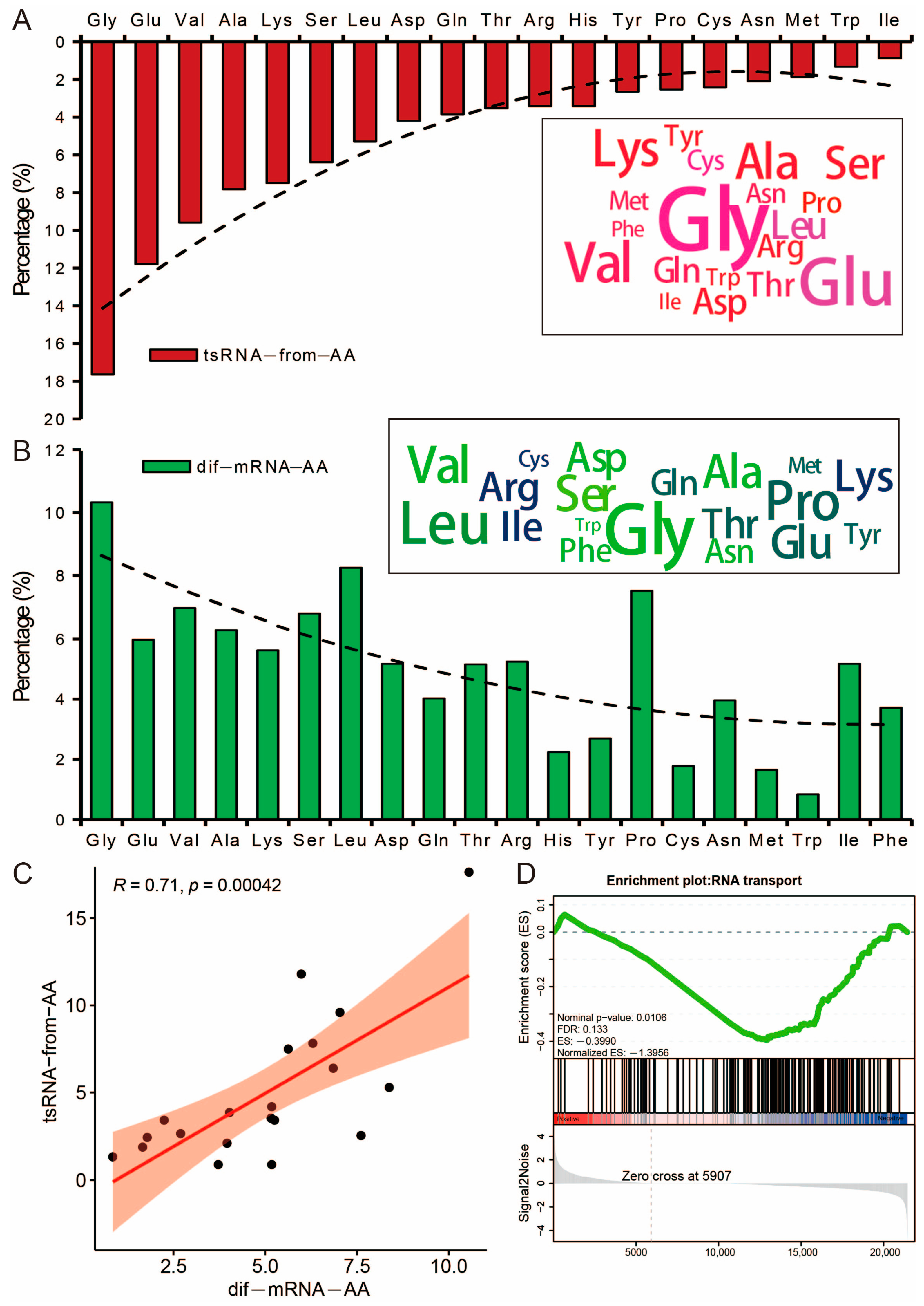
3.6. Potential Effects of tsRNA on the Amino Acid Transport Function of Its Host tRNA
3.7. Differential tsRNAs and miRNAs Are Involved in the Fatty Acid Metabolism Processes
4. Discussion
4.1. Expression Characteristics of tsRNAs in SAT and VAT
4.2. Potential Function of tsRNAs in Adipose Tissue
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lavie, C.J.; Laddu, D.; Arena, R.; Ortega, F.B.; Alpert, M.A.; Kushner, R.F. Reprint of: Healthy Weight and Obesity Prevention: JACC Health Promotion Series. J. Am. Coll. Cardiol. 2018, 72, 3027–3052. [Google Scholar] [CrossRef] [PubMed]
- Blüher, M. Obesity: Global epidemiology and pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, E.J.; LeRoith, D. Obesity and Diabetes: The Increased Risk of Cancer and Cancer-Related Mortality. Physiol. Rev. 2015, 95, 727–748. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Farias, M.; Fos-Domenech, J.; Serra, D.; Herrero, L.; Sánchez-Infantes, D. White adipose tissue dysfunction in obesity and aging. Biochem. Pharmacol. 2021, 192, 114723. [Google Scholar] [CrossRef] [PubMed]
- Lorente-Cebrián, S.; González-Muniesa, P.; Milagro, F.I.; Martínez, J.A. MicroRNAs and other non-coding RNAs in adipose tissue and obesity: Emerging roles as biomarkers and therapeutic targets. Clin. Sci. 2019, 133, 23–40. [Google Scholar] [CrossRef]
- Kurylowicz, A. microRNAs in Human Adipose Tissue Physiology and Dysfunction. Cells 2021, 10, 3342. [Google Scholar] [CrossRef]
- Squillaro, T.; Peluso, G.; Galderisi, U.; Di Bernardo, G. Long non-coding RNAs in regulation of adipogenesis and adipose tissue function. eLife 2020, 9, e59053. [Google Scholar] [CrossRef]
- Zhang, Y.; Tian, Z.; Ye, H.; Sun, X.; Zhang, H.; Sun, Y.; Mao, Y.; Yang, Z.; Li, M. Emerging functions of circular RNA in the regulation of adipocyte metabolism and obesity. Cell Death Discov. 2022, 8, 268. [Google Scholar] [CrossRef]
- Zong, T.; Yang, Y.; Zhao, H.; Li, L.; Liu, M.; Fu, X.; Tang, G.; Zhou, H.; Aung, L.H.H.; Li, P.; et al. tsRNAs: Novel small molecules from cell function and regulatory mechanism to therapeutic targets. Cell Prolif. 2021, 54, e12977. [Google Scholar] [CrossRef]
- Lee, Y.S.; Shibata, Y.; Malhotra, A.; Dutta, A. A novel class of small RNAs: tRNA-derived RNA fragments (tRFs). Genes Dev. 2009, 23, 2639–2649. [Google Scholar] [CrossRef]
- Bühler, M.; Spies, N.; Bartel, D.P.; Moazed, D. TRAMP-mediated RNA surveillance prevents spurious entry of RNAs into the Schizosaccharomyces pombe siRNA pathway. Nat. Struct. Mol. Biol. 2008, 15, 1015–1023. [Google Scholar] [CrossRef] [PubMed]
- Cole, C.; Sobala, A.; Lu, C.; Thatcher, S.R.; Bowman, A.; Brown, J.W.; Green, P.J.; Barton, G.J.; Hutvagner, G. Filtering of deep sequencing data reveals the existence of abundant Dicer-dependent small RNAs derived from tRNAs. RNA 2009, 15, 2147–2160. [Google Scholar] [CrossRef]
- Hsieh, L.C.; Lin, S.I.; Shih, A.C.; Chen, J.W.; Lin, W.Y.; Tseng, C.Y.; Li, W.H.; Chiou, T.J. Uncovering small RNA-mediated responses to phosphate deficiency in Arabidopsis by deep sequencing. Plant Physiol. 2009, 151, 2120–2132. [Google Scholar] [CrossRef]
- Burroughs, A.M.; Ando, Y.; de Hoon, M.J.; Tomaru, Y.; Suzuki, H.; Hayashizaki, Y.; Daub, C.O. Deep-sequencing of human Argonaute-associated small RNAs provides insight into miRNA sorting and reveals Argonaute association with RNA fragments of diverse origin. RNA Biol. 2011, 8, 158–177. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Anaya, J.; Mudunuri, S.B.; Dutta, A. Meta-analysis of tRNA derived RNA fragments reveals that they are evolutionarily conserved and associate with AGO proteins to recognize specific RNA targets. BMC Biol. 2014, 12, 78. [Google Scholar] [CrossRef] [PubMed]
- Haussecker, D.; Huang, Y.; Lau, A.; Parameswaran, P.; Fire, A.Z.; Kay, M.A. Human tRNA-derived small RNAs in the global regulation of RNA silencing. RNA 2010, 16, 673–695. [Google Scholar] [CrossRef]
- Thompson, D.M.; Parker, R. Stressing out over tRNA cleavage. Cell 2009, 138, 215–219. [Google Scholar] [CrossRef]
- Saikia, M.; Krokowski, D.; Guan, B.J.; Ivanov, P.; Parisien, M.; Hu, G.F.; Anderson, P.; Pan, T.; Hatzoglou, M. Genome-wide identification and quantitative analysis of cleaved tRNA fragments induced by cellular stress. J. Biol. Chem. 2012, 287, 42708–42725. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Hu, G.F. Emerging role of angiogenin in stress response and cell survival under adverse conditions. J. Cell. Physiol. 2012, 227, 2822–2826. [Google Scholar] [CrossRef]
- Maute, R.L.; Schneider, C.; Sumazin, P.; Holmes, A.; Califano, A.; Basso, K.; Dalla-Favera, R. tRNA-derived microRNA modulates proliferation and the DNA damage response and is down-regulated in B cell lymphoma. Proc. Natl. Acad. Sci. USA 2013, 110, 1404–1409. [Google Scholar] [CrossRef]
- Sharma, U.; Conine, C.C.; Shea, J.M.; Boskovic, A.; Derr, A.G.; Bing, X.Y.; Belleannee, C.; Kucukural, A.; Serra, R.W.; Sun, F.; et al. Biogenesis and function of tRNA fragments during sperm maturation and fertilization in mammals. Science 2016, 351, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Martinez, G. tRNA-derived small RNAs: New players in genome protection against retrotransposons. RNA Biol. 2018, 15, 170–175. [Google Scholar] [CrossRef] [PubMed]
- George, S.; Rafi, M.; Aldarmaki, M.; ElSiddig, M.; Al Nuaimi, M.; Amiri, K.M.A. tRNA derived small RNAs-Small players with big roles. Front. Genet. 2022, 13, 997780. [Google Scholar] [CrossRef]
- Han, J.; Tang, M.; Lu, C.; Shen, L.; She, J.; Wu, G. Subcutaneous, but not visceral, adipose tissue as a marker for prognosis in gastric cancer patients with cachexia. Clin. Nutr. 2021, 40, 5156–5161. [Google Scholar] [CrossRef] [PubMed]
- Mori, S.; Kiuchi, S.; Ouchi, A.; Hase, T.; Murase, T. Characteristic expression of extracellular matrix in subcutaneous adipose tissue development and adipogenesis; comparison with visceral adipose tissue. Int. J. Biol. Sci. 2014, 10, 825–833. [Google Scholar] [CrossRef]
- Zuriaga, M.A.; Fuster, J.J.; Farb, M.G.; MacLauchlan, S.; Bretón-Romero, R.; Karki, S.; Hess, D.T.; Apovian, C.M.; Hamburg, N.M.; Gokce, N.; et al. Activation of non-canonical WNT signaling in human visceral adipose tissue contributes to local and systemic inflammation. Sci. Rep. 2017, 7, 17326. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, H.; Tu, W.; Abbas Raza, S.H.; Cao, J.; Huang, J.; Wu, H.; Fan, C.; Wang, S.; Zhao, Y.; et al. Comparative Transcriptome Analysis Provides Insight into Spatio-Temporal Expression Characteristics and Genetic Regulatory Network in Postnatal Developing Subcutaneous and Visceral Fat of Bama Pig. Front. Genet. 2022, 13, 844833. [Google Scholar] [CrossRef]
- Hou, B.; Zhao, Y.; He, P.; Xu, C.; Ma, P.; Lam, S.M.; Li, B.; Gil, V.; Shui, G.; Qiang, G.; et al. Targeted lipidomics and transcriptomics profiling reveal the heterogeneity of visceral and subcutaneous white adipose tissue. Life Sci. 2020, 245, 117352. [Google Scholar] [CrossRef]
- Chen, M.; Zhang, F.; Chen, B.; Lau, C.; Xu, K.; Tong, T.; Huo, C.; Han, Q.; Su, T.; Kwan, H.Y. Omics approach to reveal the effects of obesity on the protein profiles of the exosomes derived from different adipose depots. Cell. Mol. Life Sci. CMLS 2022, 79, 570. [Google Scholar] [CrossRef]
- Lacedonia, D.; Tartaglia, N.; Scioscia, G.; Soccio, P.; Pavone, G.; Moriondo, G.; Gallo, C.; Foschino Barbaro, M.P.; Ambrosi, A. Different Expression of Micro-RNA in the Subcutaneous and Visceral Adipose Tissue of Obese Subjects. Rejuvenation Res. 2022, 25, 89–94. [Google Scholar] [CrossRef]
- Tan, Y.; Gan, M.; Shen, L.; Li, L.; Fan, Y.; Chen, Y.; Chen, L.; Niu, L.; Zhao, Y.; Jiang, A.; et al. Profiling and Functional Analysis of Long Noncoding RNAs and mRNAs during Porcine Skeletal Muscle Development. Int. J. Mol. Sci. 2021, 22, 503. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; Gan, M.; Wang, L.; Yang, Y.; Wang, J.; Chen, L.; Zhang, S.; Zhao, Y.; Niu, L.; Jiang, D.; et al. Differential Expression Analysis of tRNA-Derived Small RNAs from Subcutaneous Adipose Tissue of Obese and Lean Pigs. Animals 2022, 12, 3561. [Google Scholar] [CrossRef] [PubMed]
- Krüger, J.; Rehmsmeier, M. RNAhybrid: microRNA target prediction easy, fast and flexible. Nucleic Acids Res. 2006, 34, W451–W454. [Google Scholar] [CrossRef]
- Gan, M.; Ma, J.; Chen, L.; Zhang, S.; Niu, L.; Zhao, Y.; Li, X.; Pan, H.; Zhu, L.; Shen, L. Identification of tRNA-derived small RNAs and their potential roles in porcine skeletal muscle with intrauterine growth restriction. Front. Physiol. 2022, 13, 962278. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.P.; Lowe, T.M. GtRNAdb 2.0: An expanded database of transfer RNA genes identified in complete and draft genomes. Nucleic Acids Res. 2016, 44, D184–D189. [Google Scholar] [CrossRef] [PubMed]
- Lowe, T.M.; Chan, P.P. tRNAscan-SE On-line: Integrating search and context for analysis of transfer RNA genes. Nucleic Acids Res. 2016, 44, W54–W57. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Trapnell, C.; Pop, M.; Salzberg, S.L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 2009, 10, R25. [Google Scholar] [CrossRef]
- Gan, M.; Shen, L.; Fan, Y.; Guo, Z.; Liu, B.; Chen, L.; Tang, G.; Jiang, Y.; Li, X.; Zhang, S.; et al. High Altitude Adaptability and Meat Quality in Tibetan Pigs: A Reference for Local Pork Processing and Genetic Improvement. Animals 2019, 9, 1080. [Google Scholar] [CrossRef]
- Jensen, M.D. Adipose tissue and fatty acid metabolism in humans. J. R. Soc. Med. 2002, 95 (Suppl. 42), 3–7. [Google Scholar]
- Frayn, K.N.; Arner, P.; Yki-Järvinen, H. Fatty acid metabolism in adipose tissue, muscle and liver in health and disease. Essays Biochem. 2006, 42, 89–103. [Google Scholar] [CrossRef]
- Bai, Y.; McCoy, J.G.; Levin, E.J.; Sobrado, P.; Rajashankar, K.R.; Fox, B.G.; Zhou, M. X-ray structure of a mammalian stearoyl-CoA desaturase. Nature 2015, 524, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Tan, Z.; Gan, M.; Li, Q.; Chen, L.; Niu, L.; Jiang, D.; Zhao, Y.; Wang, J.; Li, X.; et al. tRNA-Derived Small Non-Coding RNAs as Novel Epigenetic Molecules Regulating Adipogenesis. Biomolecules 2019, 9, 274. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Wang, H.; Fan, T.; Chen, L.; Shi, Z.; Mi, J.; Huang, W.; Wang, R.; Hu, K. Potential Functions of the tRNA-Derived Fragment tRF-Gly-GCC Associated with Oxidative Stress in Radiation-Induced Lung Injury. Dose-Response Publ. Int. Hormesis Soc. 2022, 20, 15593258221128744. [Google Scholar] [CrossRef] [PubMed]
- Zwick, R.K.; Guerrero-Juarez, C.F.; Horsley, V.; Plikus, M.V. Anatomical, Physiological, and Functional Diversity of Adipose Tissue. Cell Metab. 2018, 27, 68–83. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Wang, Z.; Li, J. Progress on the protective effects of maternal fatty acid supplementation on infant asthma risk: A narrative review. Ann. Palliat. Med. 2021, 10, 2323–2330. [Google Scholar] [CrossRef]
- Wang, D.D.; Hu, F.B. Dietary Fat and Risk of Cardiovascular Disease: Recent Controversies and Advances. Annu. Rev. Nutr. 2017, 37, 423–446. [Google Scholar] [CrossRef]
- Kang, D.; Zhou, G.; Zhou, S.; Zeng, J.; Wang, X.; Jiang, Y.; Yang, Y.; Chen, Y. Comparative transcriptome analysis reveals potentially novel roles of Homeobox genes in adipose deposition in fat-tailed sheep. Sci. Rep. 2017, 7, 14491. [Google Scholar] [CrossRef]
- Chen, Z.; Cao, X.; Lu, Q.; Zhou, J.; Wang, Y.; Wu, Y.; Mao, Y.; Xu, H.; Yang, Z. circ01592 regulates unsaturated fatty acid metabolism through adsorbing miR-218 in bovine mammary epithelial cells. Food Funct. 2021, 12, 12047–12058. [Google Scholar] [CrossRef]
- Li, X.; Gan, Z.W.; Ding, Z.; Wu, Y.X.; Chen, X.Y.; Tian, H.M.; Liu, G.L.; Yang, Y.T.; Xie, L. Genetic Variants in the ELOVL5 but not ELOVL2 Gene Associated with Polyunsaturated Fatty Acids in Han Chinese Breast Milk. Biomed. Environ. Sci. BES 2017, 30, 64–67. [Google Scholar] [CrossRef]
- Shi, H.B.; Du, Y.; Zhang, C.H.; Sun, C.; He, Y.L.; Wu, Y.H.; Liu, J.X.; Luo, J.; Loor, J.J. Fatty acid elongase 5 (ELOVL5) alters the synthesis of long-chain unsaturated fatty acids in goat mammary epithelial cells. J. Dairy Sci. 2018, 101, 4586–4594. [Google Scholar] [CrossRef]
- Morais, S.; Monroig, O.; Zheng, X.; Leaver, M.J.; Tocher, D.R. Highly unsaturated fatty acid synthesis in Atlantic salmon: Characterization of ELOVL5- and ELOVL2-like elongases. Mar. Biotechnol. 2009, 11, 627–639. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Wan, D.; Zhang, Y.; Zhang, Y.; Long, C.; Chen, S.; He, L.; Tan, B.; Wu, X.; Yin, Y. Diurnal variations in polyunsaturated fatty acid contents and expression of genes involved in their de novo synthesis in pigs. Biochem. Biophys. Res. Commun. 2017, 483, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Renaville, B.; Bacciu, N.; Lanzoni, M.; Corazzin, M.; Piasentier, E. Polymorphism of fat metabolism genes as candidate markers for meat quality and production traits in heavy pigs. Meat Sci. 2015, 110, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, A.; Morgado, N. Trans fatty acid isomers in human health and in the food industry. Biol. Res. 1999, 32, 273–287. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Gu, H.; Liao, T.; Lei, Y.; Qiu, Y.; Chen, Q.; Chen, L.; Zhang, S.; Wang, J.; Hao, X.; et al. tsRNA Landscape and Potential Function Network in Subcutaneous and Visceral Pig Adipose Tissue. Genes 2023, 14, 782. https://doi.org/10.3390/genes14040782
Wang L, Gu H, Liao T, Lei Y, Qiu Y, Chen Q, Chen L, Zhang S, Wang J, Hao X, et al. tsRNA Landscape and Potential Function Network in Subcutaneous and Visceral Pig Adipose Tissue. Genes. 2023; 14(4):782. https://doi.org/10.3390/genes14040782
Chicago/Turabian StyleWang, Linghui, Hao Gu, Tianci Liao, Yuhang Lei, Yanhao Qiu, Qiuyang Chen, Lei Chen, Shunhua Zhang, Jinyong Wang, Xiaoxia Hao, and et al. 2023. "tsRNA Landscape and Potential Function Network in Subcutaneous and Visceral Pig Adipose Tissue" Genes 14, no. 4: 782. https://doi.org/10.3390/genes14040782
APA StyleWang, L., Gu, H., Liao, T., Lei, Y., Qiu, Y., Chen, Q., Chen, L., Zhang, S., Wang, J., Hao, X., Jiang, D., Zhao, Y., Niu, L., Li, X., Shen, L., Gan, M., & Zhu, L. (2023). tsRNA Landscape and Potential Function Network in Subcutaneous and Visceral Pig Adipose Tissue. Genes, 14(4), 782. https://doi.org/10.3390/genes14040782