Abstract
Euonymus microcarpus (Oliv.) Sprague, is a species of evergreen shrub of the genus Euonymus, family Celastraceae. Here, we extracted the genomic DNA from the leaves of E. microcarpus and constructed a paired-end library. The chloroplast genome of E. microcarpus was generated with the high-throughput sequencing by the illumina Hiseq X Ten platform and de novo assembly. The chloroplast genome had a quadripartite structure, containing a long single copy region with a size of 85,386 bp and a short single copy region with a size of 18,456 bp, separated by two inverted repeat regions of 26,850 bp. The chloroplast genome contained 133 genes identified in total, including 87 potential protein-coding genes, 38 transfer RNA genes, and eight ribosomal RNA genes. A total of 282 simple sequence repeats and 63 long repeats were found. Furthermore, the phylogenetic relationships inferred that E. microcarpus is sister to E. japonicus and E. schensianus. A comparison of the structure of the chloroplast genomes of eight Euonymus species suggests a nucleotide variability of the junction sites and a higher divergence of non-coding regions, compared to the coding regions. The original findings of the study serves as a good reference for chloroplast genome assembly and a valuable foundation for the genetic diversity and evolution of E. microcarpus.
1. Introduction
There are about 220 species in the genus Euonymus, half of which are distributed in China [1]. The species of the genus Euonymus have three fundamental characteristics. The first one is that most of the species in genus Euonymus have been used in traditional Chinese herbal medicine for treating tumors, thrombosis, diabetes, hyperlipidemia and inflammation because the herbal extracts contain numerous chemical components, including terpenoids, flavonoids, phenolic acids, lignans, cardenolides and alkaloids [2]. The second one is that most of the species in this genus displays a tolerance to the cold, drought, salt and wounding and are ideal ornamental shrubs or trees [3]. The last one is that the seed oil extracted from the plants in the genus Euonymus is composed of 3-acetyl-1,2-diacyl-sn-glycerols, which has a reduced viscosity and a lower crystallization temperature than that of the common vegetable oils and is extremely attractive for industrial purposes [4].
E. microcarpus (Oliv.) Sprague, an endemic species in China, distributed in Hubei, Shaanxi, Sichuan and Yunnan provinces, is a species of evergreen shrub of the genus Euonymus, family Celastraceae [5,6]. It grows in mixed forests along riversides and on hillsides, at an altitude of 350–1000 m [7]. As an extensively used herb in Chinese traditional medicine, the roots and stems of E. microcarpus have been used for relieving the rigidity of muscles, activating the collaterals and relieving rheumatism [7]. As an ideal ornamental plant species, E. microcarpus exhibits a tolerance to hard pruning [6].
Chloroplasts, the plastids in plants and green algae originated by the cyanobacteria endosymbiosis, are photosynthetic organelles producing energy for plant growth and development by photosynthesis and the oxygen-release processes [8,9]. More recent research has begun to uncover the novel roles of chloroplasts in plant growth and development. Melatonin produced in chloroplasts and mitochondria is important for plants under stress conditions [10]. The COP1LIKE protein acts as a repressor of photomorphogenesis and plays roles in the reproductive development and light-dependent pigment accumulation [11]. The chloroplast genomes (cpDNAs), maternally inherited in most plant species, are highly conserved in genes fundamental to plant life, gene content and gene order. Typically, cpDNAs are circular, containing a pair of inverted repeats (IRs), a large long single copy (LSC) region and a short single copy (SSC) region [12]. Most of the variations of the cpDNAs in size and structure, caused by plant evolution or special environmental adaptation, can be used to study the genetic diversity, population genetics and phylogenetic relationships among angiosperms [13].
The cpDNAs of several species of the genus Euonymus, such as E. phellomanus, E. fortunei and E. maackii, were sequenced by high-throughput sequencing and were assembled, annotated and published at present [14]. We collected wild E. microcarpus samples for the high-throughput cpDNA sequencing. The genomic features of the E. microcarpus cpDNA were determined, then the simple sequence repeats (SSRs) and the long repeats were obtained. The comparisons were made of the cpDNA sequences of E. microcarpus and 35 other species within the order Sapindales, then revealed their phylogenetic relationships.
In the present work, we report the first plastome sequence of E. microcarpus to guide the exploration of the use of the new genetic resources and the phylogenetic relationships within the order Sapindales. The findings of this paper provide a foundation for future genomic research on phylogenetic relationships and evolution within the genus Euonymus.
2. Materials and Methods
2.1. Plant materials, Library Construction, and cp Genome Sequencing and Assembly
Fresh and healthy leaves of E. microcarpus (Oliv.) Sprague were directly collected by Dr. Hongying Li from an individual at Qiuba, Luanchuan, Luoyang, Henan, China (34°03′58″ N, 111°66′72″ E) and were identified by the most outstanding feature of the colored fall berries that begin shrouded in pink to red capsules and open to orange arils. The voucher specimen (LY_2020_HAUST263) was chilled in a dry ice-ethanol bath, then deposited in a freezer in the Biological Breeding Institute (BBI), Taishan Academy of Forestry Sciences (TAFS), Taian, China (contact person: Dr. Wei Guo, wwwguoweinet@163.com). We extracted the genomic DNA with the CTAB method [15]. The sheared genomic DNA (fragment size ≤ 350 bp) was used for the DNA end repair, A-tailing, adapter ligation, and PCR amplification to construct the paired-end library. The Illumina Hiseq X Ten platform was then employed for sequencing.
Following the removal of the adapters and the low-quality reads with fastp (version 0.20.0), the read quality was improved and high-quality clean reads were obtained [16]. The E. microcarpus cpDNA was assembled using the SPAdes pipeline (http://bioinf.spbau.ru/SPAdes/) (accessed on 3 February 2020) with E. japonicus cpDNA as the reference (GenBank accession NC_028067).
2.2. Gene Annotation
Once the annotation was completed with the CpGAVAS pipeline, the cpDNA of E. microcarpus was deposited to GenBank data libraries under the following accession number ON611716 [17]. A map of the E. microcarpus cpDNA was created with OGDRAW (version v1.2) [18]. The program CodonW (version 1.4.4) (http://codonw.sourceforge.net) (accessed on 3 February 2020) was applied for the analyses of the relative synonymous codon usage (RSCU).
2.3. Repeats Identification
REPuter was applied to quantify and locate the forward and reverse complement repeats [19,20]. The MISA software (version 1.0) was used to identify the potential SSRs [21].
2.4. Phylogenetic Analysis
The alignment of the 36 cpDNAs was conducted with MAFFT (version 7.427) [22]. The phylogenetic trees with a maximum likelihood method were inferred by RAxML (version 8.2.10), using the ‘GTRGAMMA’ evolutionary model, using a rapid boot-strap algorithm of 1000 replications [22].
2.5. A Comparison of the Genomes of the Related Species
IRscope was used to visually display and compare the borders of the SSC, IRs and LSC among the eight Euonymus species [23]. The mVISTA with the Shuffle-LAGAN program was employed for the pairwise alignment with eight complete cp genomes, using the sequence annotation information of E. japonicus [24]. Once the sequences were aligned with the MAFFT (version 7.427), the pi values of the genes were computed using the CDS sequence alignments of the different genes with vcftools, and the Ka/Ks statistic, that is, the ratio of nonsynonymous (Ka) to synonymous (Ks) nucleotide substitution were calculated, based on the alignments with the KaKs_Calculator.
3. Results
3.1. Genome Characteristics of E. microcarpus
A total of 20,881,158 clean paired-end reads were yielded after discarding the adapters and low-quality reads, which comprised 6,264,347,400 bases, of which the percentage of Q30 was higher than 93.37% and the GC content was 37.55%. The cpDNA assembly of E. microcarpus, 157,542 bp in length, has a GC content of 37.22% and an average read coverage of 2321.42×. Compared with the cpDNA of E. szechuanensis, that of E. microcarpus shared conserved sequences and the synteny was identified (Table S1).
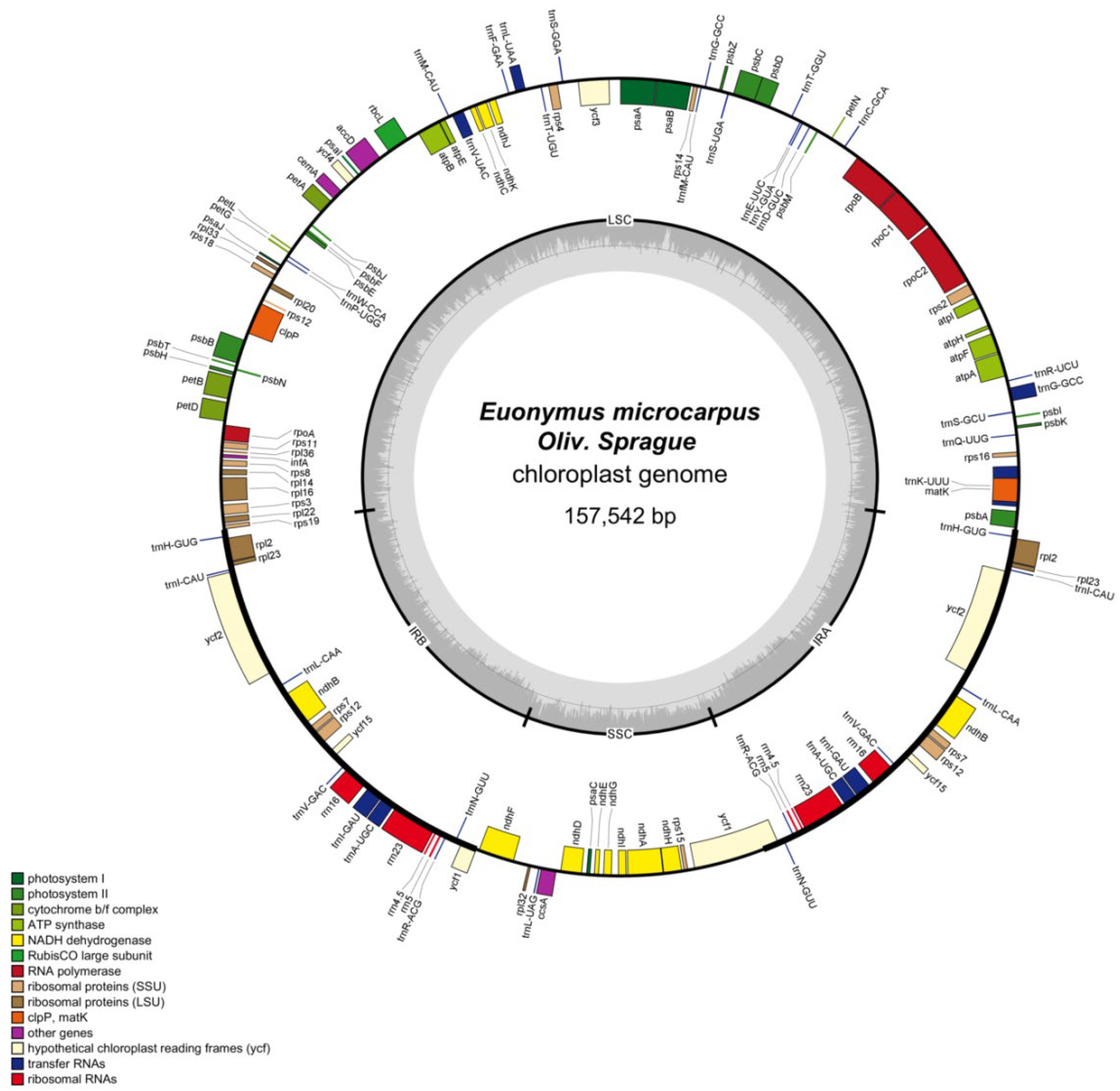
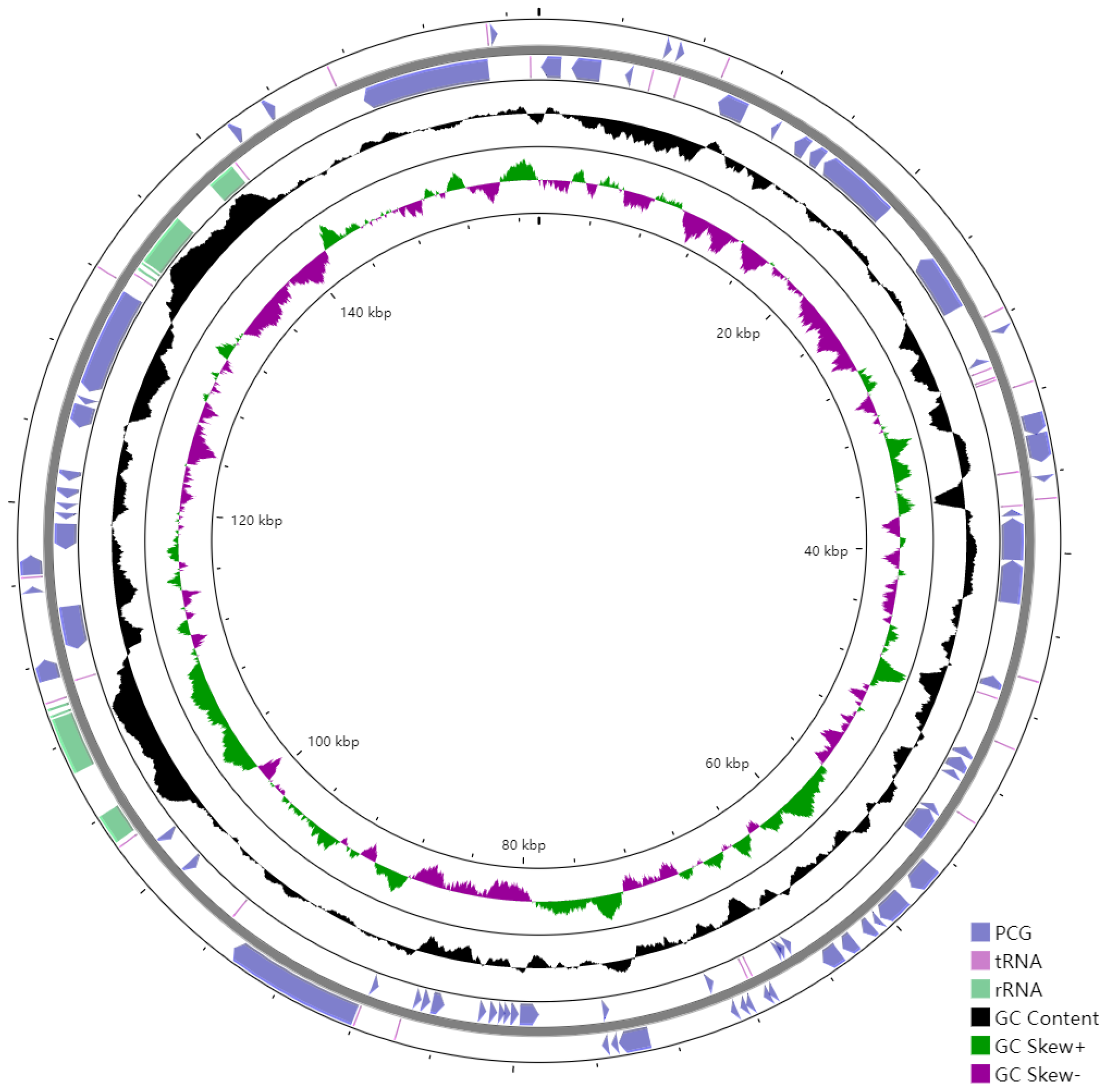
The cpDNA is a circular molecule displaying a characteristic quadripartite structure, containing a LSC region with a size of 85,386 bp and a SSC region with a size of 18,456 bp, separated by two IR regions of 26,850 bp (Figure 1). It was observed that the two IR regions display a considerably higher GC content than that of LSC and SSC regions (Figure 2).
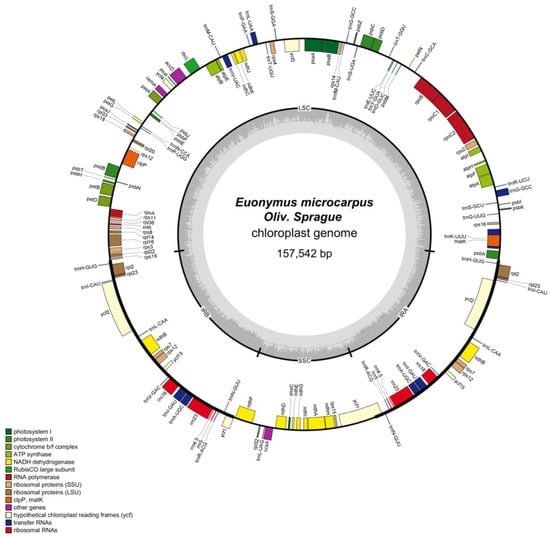
Figure 1.
Circular map of the E. microcarpus cpDNA. Genes shown outside and inside of the circle map are transcribed clockwise and counterclockwise, respectively. Different colors represent different functional genes. The dashed area in the inner circle corresponds to the GC content.
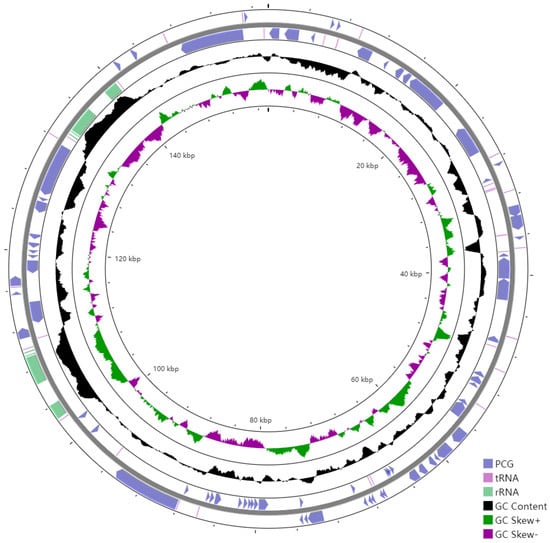
Figure 2.
GC content of the E. microcarpus cpDNA. From outer into inner, the first and second circles represent the CDSs of the protein coding genes (blue), rRNA operon (light purple), and tRNA information (reddish brown); the third circle represents the GC contents; the fourth circle represents the GC skew [(G − C)/(G + C); green, >0; purple, <0; and the fifth circle represents the coordinates of the cpDNA. The artificial start site is at 0 kbp.
The cpDNA was predicted to encode 87 potential protein-coding, 38 transfer RNA (tRNA), and eight ribosomal RNA (rRNA) genes (Table 1). In total, the duplication of 17 genes happened in the two IRs, including seven protein-coding genes, rpl2, rpl23, ycf2, ycf15, rps7, rps12 and ndhB (Table 1). Seventeen intron-containing genes were annotated, 11 of which were protein-coding genes, whereas the remaining six were tRNA genes (Table 2). Among the 11 protein-coding genes identified in this study, clpP, ycf3 and rps12 bear two introns, while the others bear only one. The rps12 gene was identified as trans-spliced with a single 5′-end at the LSC region while a repeated 3′-end exons was located in the IRs (Figure 1 and Table 2).

Table 1.
Annotated genes in the E. microcarpus cpDNA.

Table 2.
Genes with introns in the E. microcarpus cpDNA.
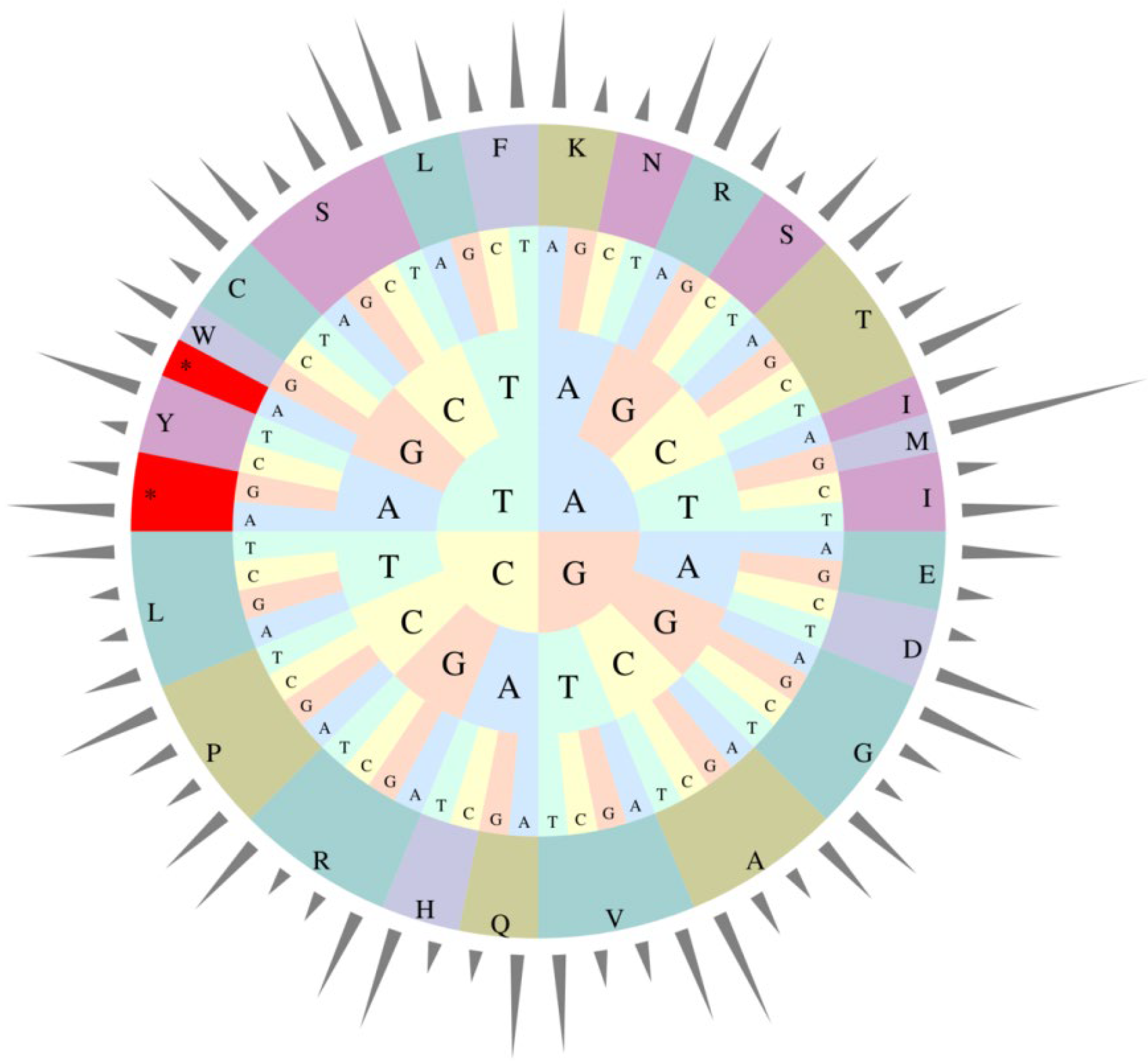
The relative occurrence of the synonymous codons in the coding sequences of E. microcarpus cpDNA was calculated using 26,883 codons. It suggests that the four most frequently used codons are AAA-K (1114), AUU-I (1093), GAA-E (1058) and UUU-F (982), accounting for 4.14%, 4.07%, 3.94% and 3.65% among all codons, respectively (Table S2 and Figure 3). Similar to the previous results for other angiosperms, most of the codons ending with A or T have RSCU values greater than 1, while, most of those ending with C or G have RSCU values of less than 1 [25,26,27].
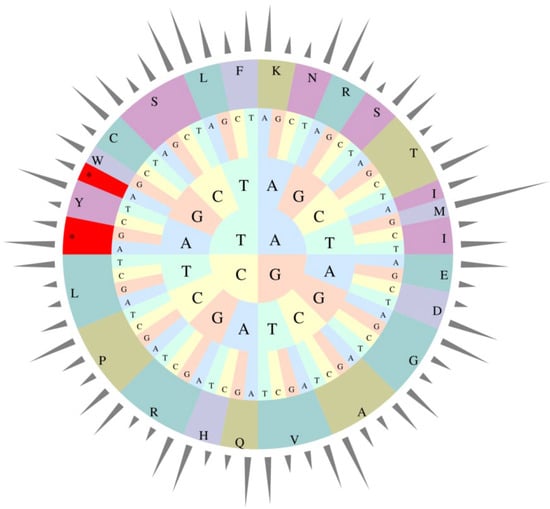
Figure 3.
Codon usage frequency of the E. microcarpus cpDNA.
3.2. Analysis of the Repeats
In the cpDNA of E. microcarpus, a total of 63 long repeats, ranging from 30 to 77 bp in length, were recognized, including 16 forward, 20 reverse, 6 complement and 21 palindrome repeats (Table S3). Furthermore, a total of 42, 29 and 9 long repeats were detected in the LSC, IRs and SSC regions, respectively.
In addition, 282 simple sequence repeats (SSRs) were detected in the cpDNA, among which 199, 10, 82, 8 and 4 were mono-, di-, tri-, tetra- and pentanucleotide repeats, respectively (Table S4). In all of the SSRs identified in this study, the mononucleotide SSRs were the most abundant, with an abundance of 70.57%.
3.3. Phylogenetic Analysis
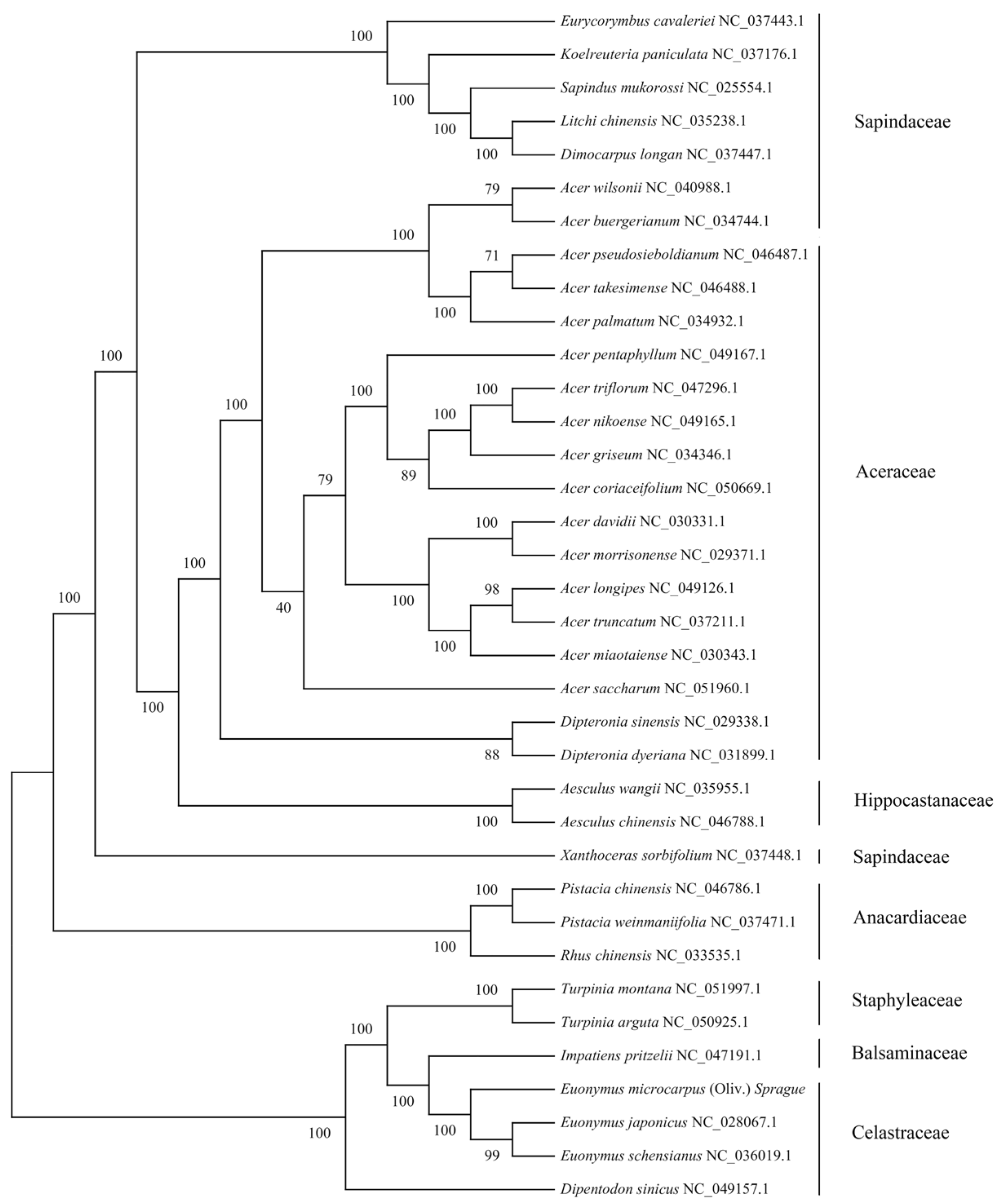
We selected cpDNAs of E. microcarpus and another 35 species within the family Sapindaceae, Aceraceae, Hippocastanaceae, Sapindaceae, Anacardiaceae, Staphyleaceae, Balsaminaceae and Celastraceae, to explore the genetic relationship between E. microcarpus and its relatives. Multiple alignments of all of the 36 cpDNAs was computed using MAFFT, then a maximum likelihood (ML) tree was determined using RAxML implementing the GTR-γ model. As shown in the Figure 4, all relationships were strongly supported by high bootstrap values ranging from 40 to 100.
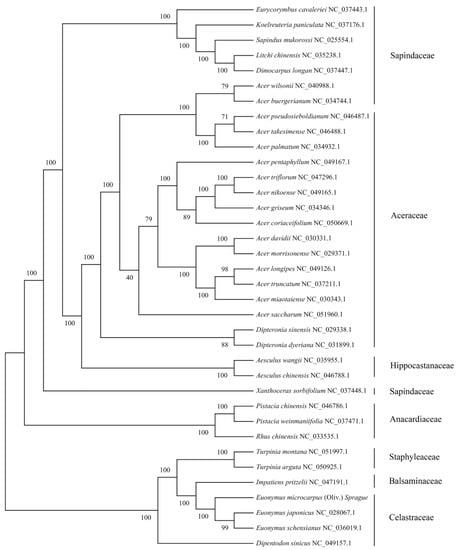
Figure 4.
Phylogenetic tree of 36 complete cpDNAs constructed using the maximum likelihood (ML). The numbers above the branches represent the ML bootstrap values.
The phylogenetic tree with most branches having high levels of support, shows two divergent clades. The first clade is formed by Sapindaceae, Aceraceae, Hippocastanaceae, Sapindaceae and Anacardiaceae, whereas the other is formed by Staphyleaceae, Balsaminaceae and Celastraceae. The phylogenetic analyses have established that three Euonymus species, Turpinia montana, Turpinia arguta, Impatiens pritzelii and Dipentodon sinicus, fell into the second clade. Dipentodon sinicus is located at the base of the clade. Subsequently, two Turpinia species and Impatiens pritzelii are separated. In addition, E. japonicus is sister to E. schensianus, and they are successively sister to E. microcarpus (Figure 4).
3.4. Comparative Analysis of the Euonymus cpDNA
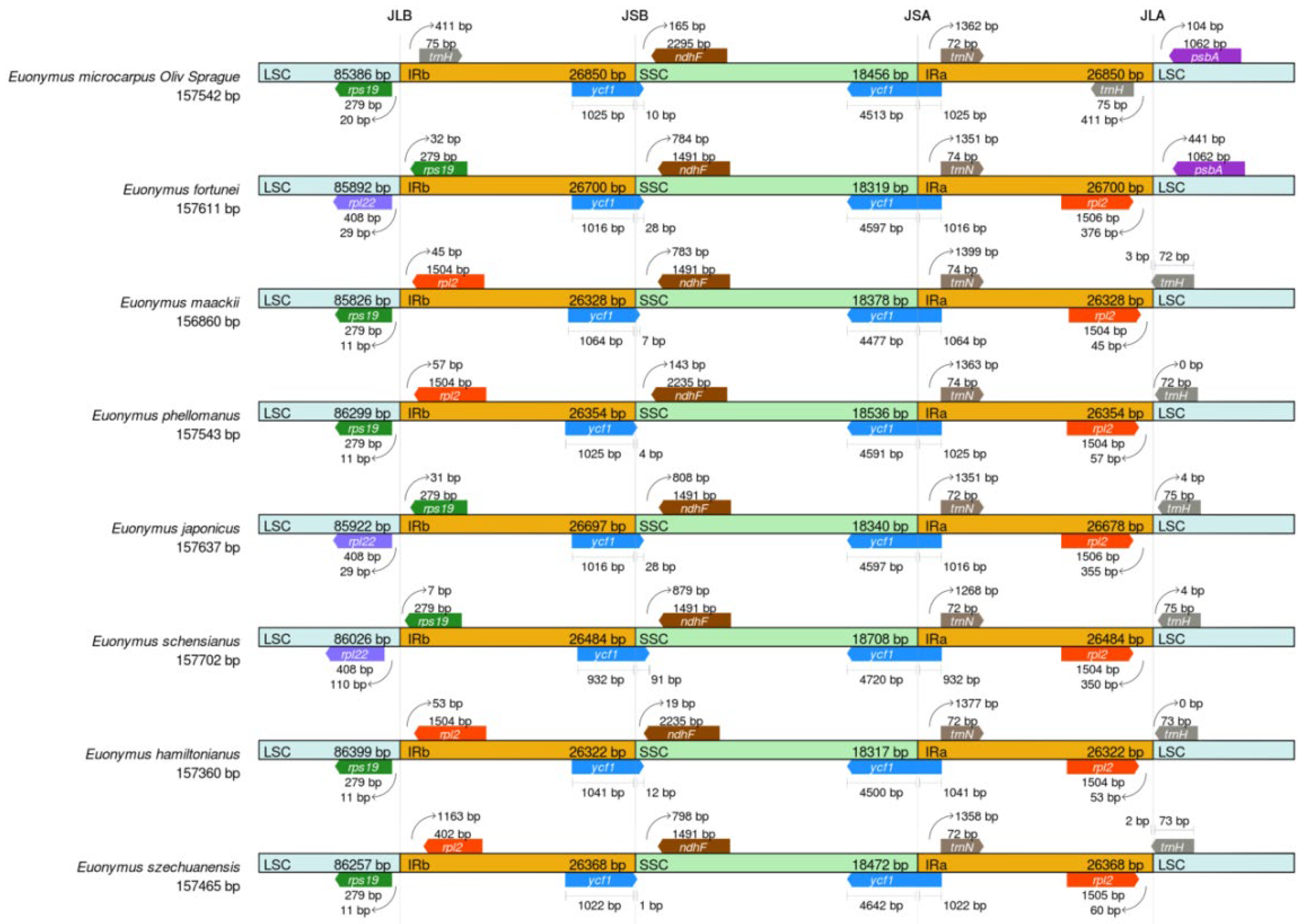
To advance our understanding of the cpDNAs of the genus Euonymus, further investigation was conducted to make critical comparisons of the IR/SSC and IR/LSC border positions in the eight selected Euonymus species, to access the degree of IR expansion or contraction among them. As shown in Figure 5, a large variability of the junction sites was observed in the eight cpDNAs, of which the LSC, IRa/b, and SSC regions have an average length of 86,001 bp, 26,513 bp and 18,441 bp, respectively.
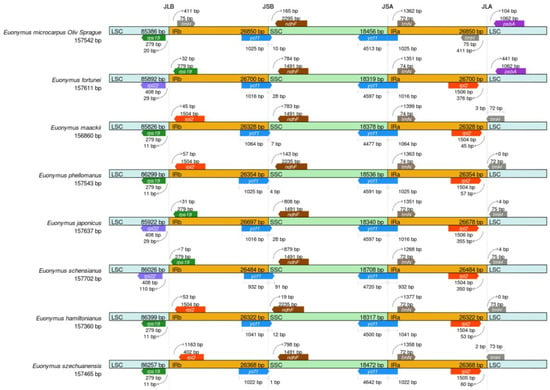
Figure 5.
Border comparisons of the eight selected cpDNAs.
The rps19 of E. microcarpus, E. maackii, E. phellomanus, E. hamiltonianus and E. szechuanensis, were located completely at the junction of the LSC. The trnH-GUG and psbA of E. microcarpus was located at the JLA junction. The trnH-GUG was completely found in the IRa region, while the psbA was completely located in the LSC region. The JLB junction of E. fortunei, E. japonicus and E. schensianus was positioned between the two genes, rpl22 and rps19. In the meantime, the gaps present in the gene rps19 in IRb from the JLB junction point were 32, 31 and 7 bp, respectively.
The trnH-GUG resided at the IRa and was 411 bp from the IRa/LSC border in E. microcarpus. In addition, the trnH-GUG and rpl2 of E. maackii, E. phellomanus, E. japonicus, E. schensianus, E. hamiltonianus and E. szechuanensis were positioned at the junction of JLA. In the midst of them, there was a consolidation of trnH-GUG into the IRa region 3 and 2 bp in the respective cpDNAs of E. maackii and E. szechuanensis. In the meantime, the trnH-GUG of E. phellomanus, E. japonicus, E. schensianus and E. hamiltonianus was totally positioned in the LSC region.
Furthermore, in the cpDNAs of all of the species, a complete copy of the functional ycf1 gene crossed JSA, while a truncated copy of ycf1 detected as a pseudogene fragment was located at JSB and very close to ndhF. The ycf1 fragments exhibited a 10-, 28-, 7-, 4-, 28-, 91-, 12- and 1-bp-long expansion from the IRb to the SSC regions in the cpDNAs of E. microcarpus, E. fortunei, E. maackii, E. phellomanus, E. japonicus, E. schensianus, E. hamiltonianus and E. szechuanensis, respectively. Notably, the cpDNAs of E. microcarpus, E. maackii, E. phellomanus, E. hamiltonianus and E. szechuanensis contained rps19 s in the LSC regions, whereas that of the other three species contained rpl22 in the LSC regions. All of the cpDNAs of all of the species possessed trnN in the IRa regions and the trnN were 1268–1399 bp away form the junction of JSA.
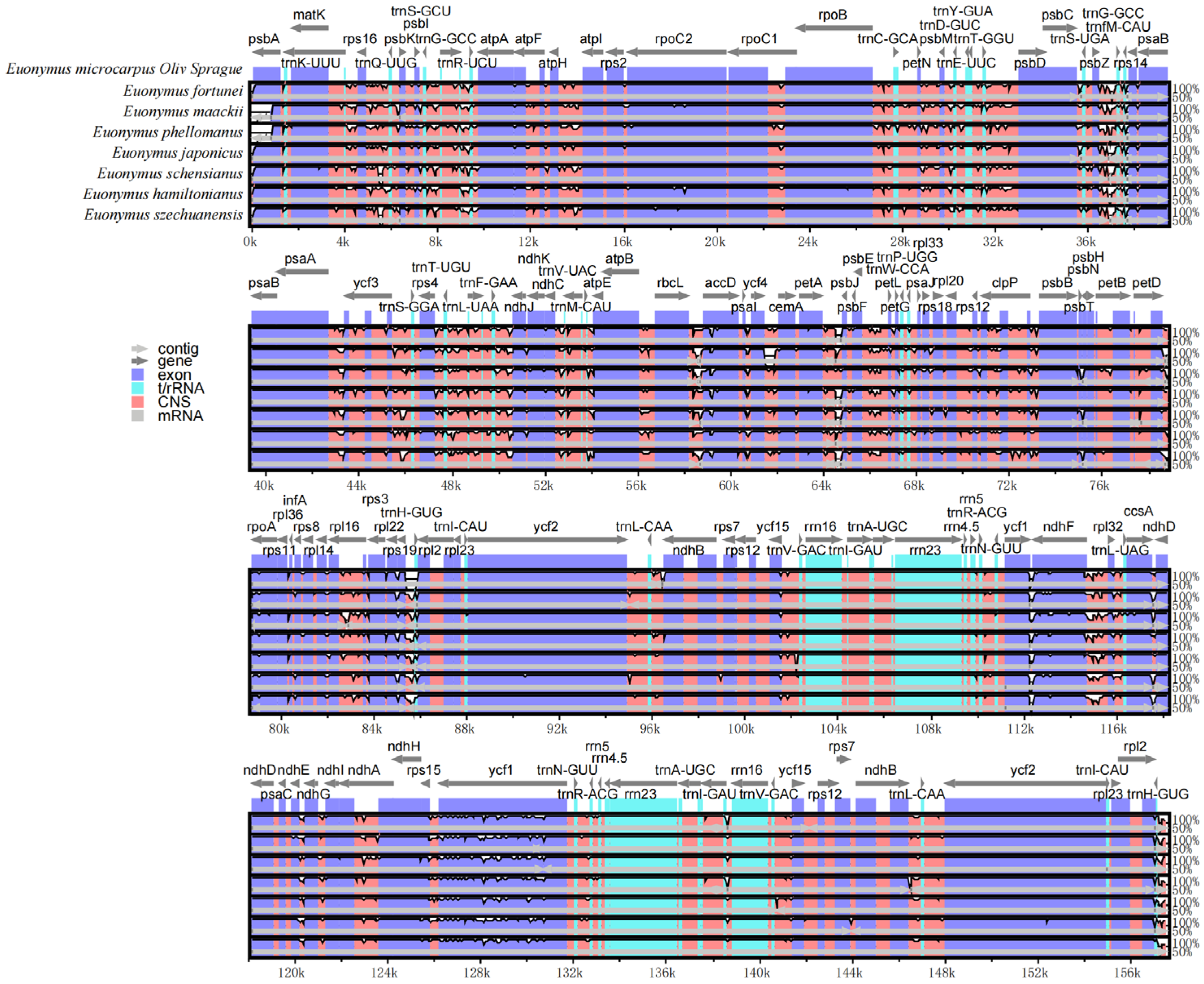
The overall organization of the cpDNAs among the selected eight species assessed using the cpDNA of E. microcarpus as reference, showed a similarity of the structure and gene order. In the meantime, the level of divergence of the non-coding regions between the species selected was higher than that of the coding regions and the same thing happened in the comparison between the single copy regions and IRs (Figure 6).
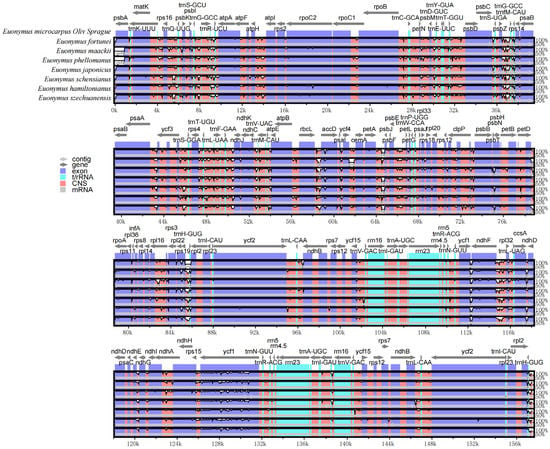
Figure 6.
Visualization of the alignment of E. microcarpus and seven related species. The vertical scale indicates the percent identity ranging from 50 to 100%. Gray arrows and thick black lines above the alignment indicate the gene orientation.
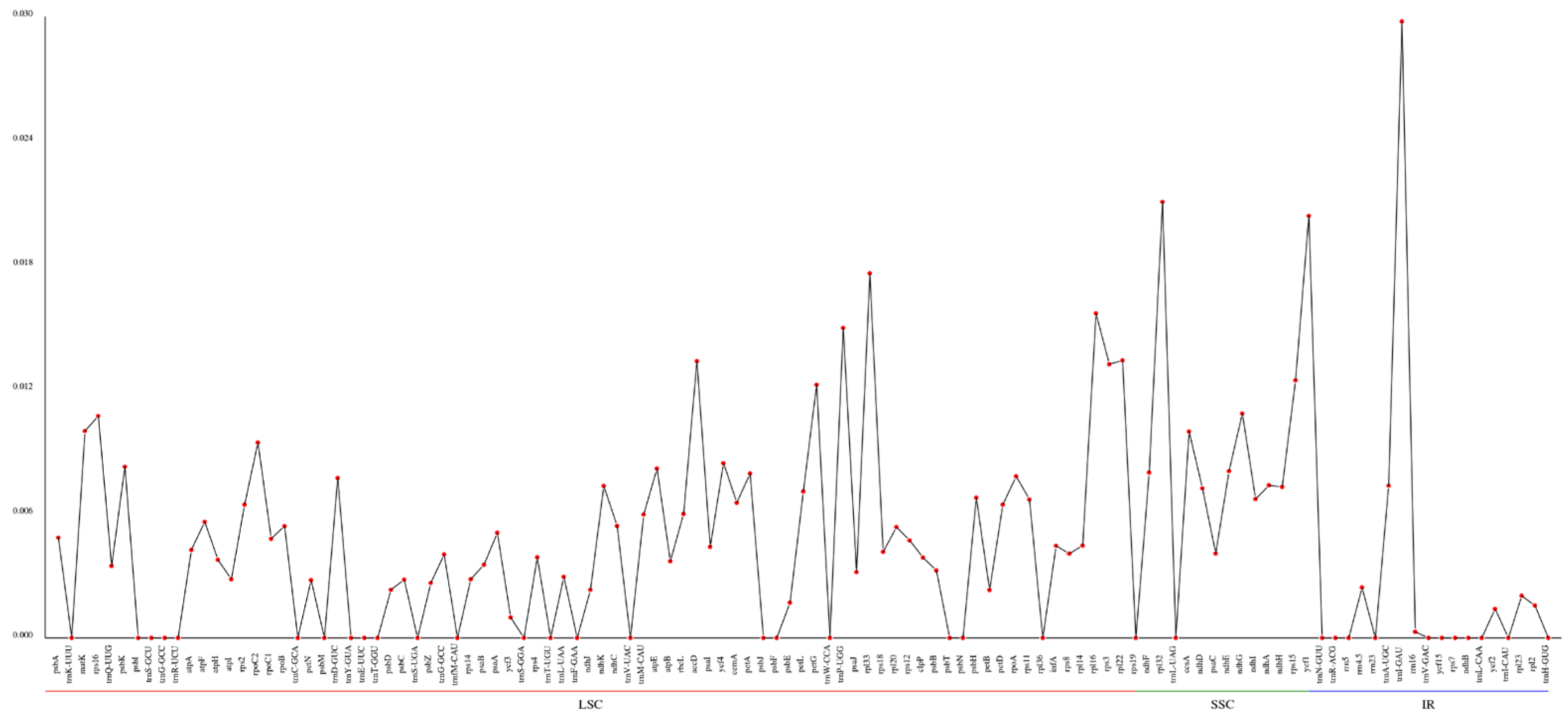
The nucleotide diversity (pi) values of the selected eight cpDNAs, calculated within the slide window, ranged from 0 to 0.02976, with an average of 0.00471 (Figure 7). According to the data, three regions were highly variable, IR.trnI-GAU, SSC.rpl32 and SSC.ycf1, with pi values higher than 0.02. The results showed low pi values, which suggests the cpDNA sequences are highly conserved at the sequence level throughout the genus.
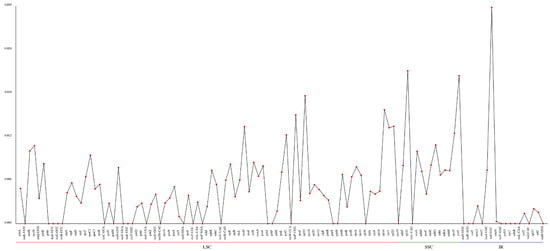
Figure 7.
Comparative analysis of the gene nucleotide variability (pi) values of eight Euonymus species. The X-axis and Y-axis show the genes and the pi values, respectively.
Between E. microcarpus and each of E. microcarpus, E. fortunei, E. maackii, E. phellomanus, E. japonicus, E. schensianus, E. hamiltonianus and E. szechuanensis, the substitution rates of synonymous (Ks) and nonsynonymous (Ka) were determined to test for the adaptive selection (Table S5). These results suggest that four genes of cpDNA of E. microcarpus, rpl16, ycf2-2, ycf1-2 and atpF with Ka/ks ratios as >1, underwent a positive selection in the comparisons of the E. microcarpus, versus the chosen species (Table S5). On the contrary, the remaining genes, with Ka/Ks < 1, underwent a negative selection, suggesting a slower evolution rate.
4. Discussion
The plant cpDNA carries an average of 120 genes, including genes involved in the gene expression and genes related to photosynthesis [28]. The precise cpDNAs of E. microcarpus, a circular molecule of 157,542 bp displaying a characteristic quadripartite structure as with other angiosperm cpDNAs (Figure 1), 87 potential protein-coding, 38 transfer RNA (tRNA), and eight ribosomal RNA (rRNA) genes (Table 1).
The GC content of the overall cpDNA is 37.22%, whereas that of the IR regions is higher than the LSC and SSC regions (Figure 2). The same thing happened in the cpDNAs of E. fortunei, E. maackii, E. phellomanus, E. japonicus, E. schensianus, E. hamiltonianus and E. szechuanensis, respectively [14,29]. The GC content is treated as an indicator of the local recombination rate and stability of the secondary structures [30].
The repeat sequences may raise the genetic diversity of the species and advance the cpDNA rearrangements. Generally, the cpSSRs, more easily obtained than the nuclear SSRs, are valuable genetic tools for addressing basic and applied plant biology questions, because of their codominant nature, high polymorphism and low substitution rate. Previous research detected 79 to 135 SSRs and 43 to 56 long repeats in the cpDNAs of six species of genus Euonymus, including E. Japonicus, E. maackii, E. phellomanus, E. schensianus, E. hamiltonianus and E. Fortunei [31], while the present study detected 282 SSRs and 63 long repeats in the cpDNA of E. microcarpus. Among the SSRs and the long repeats of the seven species of genus Euonymus, the mononucleotide SSRs were the majority, while the complement repeats were the minority. The SSR motifs could serve as potential molecular markers for the species identification, analysis of the population genetic and the genetic differentiation between individuals [31,32].
The phylogenetic analysis was conducted, based on the cpDNAs of 36 species within the order Sapindales. In the first clade, the cluster formed by three species within the family Anacardiaceae, including two species of the genus Pistacia and one species of the genus Rhus, diverged first, forming a sister relationship with a 100% bootstrap support. Then, Xanthoceras sorbifolium, belonging to the family Sapindaceae, diverged and formed a sister relationship to the family Sapindaceae, Aceraceae and Hippocastanaceae with a 100% bootstrap support. Having been cluster formed by the genus Acer and Dipteronia, the Aceraceae form a monophyletic cluster and is most closely related to the cluster of two Aesculus species. The results are consistent with current taxonomic classifications, showing the family Aceraceae and Hippocastanaceae, not distinct from the Sapindaceae, are in fact nested in the family Sapindaceae [33]. In the second clade, the phylogenetic tree fully supports that E. microcarpus is most closely associated to the cluster of E. japonicus and E. schensianus with a 100% bootstrap value. The genus Euonymus is monophyletic and is closely associated to Impatiens pritzelii and the cluster of two Turpinia species, successively, with high support scores. Dipentodon sinicus within Celastraceae, diverged first, forming a sister relationship to the family Staphyleaceae, Balsaminaceae and Celastraceae. This study showed that Dipentodon sinicus is not most closely related to three species of the genus Euonymus within Celastraceae. The systematic position of Dipentodon sinicus has been highly controversial since it was placed in the family Celastraceae [34]. Therefore, to better understand the phylogeny and evolution of the order Sapindales, more species are necessary. This study provides a valuable basis for future phylogenetic analyses of species within Celastraceae.
The modification of the cpDNA size could be driven by the important evolutionary events, expansion and shrinkage of the IR region, which occur frequently, lead to fluxes in the LSC/IR junctions and the initiation of pseudogenes, gene duplication or a reversion of the duplicated genes to a single copy [35]. Previous studies have found that the contraction and expansion of IRs affect the evolution rate of the protein-coding genes in the subfamily Pothoideae in the family Araceae [36]. In this study, we compared the cpDNAs of eight Euonymus species. In the cpDNAs of angiosperm, the borders between the LSC/SSC and IR regions are highly and relatively conserved [37]. It is clear that the rps19 of E. microcarpus, E. maackii, E. phellomanus, E. hamiltonianus and E. szechuanensis, were completely positioned in the LSC region at the junction of JLB, while, that of E. japonicus, E. schensianus and E. fortune, were located in the IRb region. It is obvious that the cpDNAs of E. japonicus, E. schensianus and E. fortunei showed expansions and contractions of the IRs and LSC, respectively, which is consistent with the findings of earlier reports [14]. The truncated copies of ycf1 were pseudogenized and observed at the junction of JSB in all of the eight selected Euonymus species. The results increase the current knowledge of the evolutionary patterns in the angiosperms.
5. Conclusions
In conclusion, the cpDNA of E. microcarpus, sequenced using an Illumina HiSeq X Ten system, displayed a typical quadripartite structure of most angiosperms. A large number of SSRs were detected, which could provide valuable information for designing exceedingly variable molecular markers for the population genetics and ecological and evolutional studies of E. microcarpus. Based on the cpDNAs of 36 species within the order Sapindales, the phylogenetic analysis showed a close relationship among E. microcarpus, E. japonicus and E. schensianus, suggesting that they are sister species. A comparison of the structure of the cpDNAs of eight Euonymus species was performed and it is obvious that there is a nucleotide variability of the junction sites and a higher divergence of non-coding regions, compared to coding regions. The obtained genetic resources and the results of the above analyses will facilitate future studies in the implementation of the effective conservation and management strategies, to help develop affordable and efficient genetic assays for the species identification of the genus Euonymus.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/genes13122352/s1, Table S1: Statistics of the synteny between the E. microcarpus and E. szechuanensis cpDNAs; Table S2: Codon usage of the E. microcarpus cpDNA from the RSCU tools; Table S3: Long repeat sequences in the E. microcarpus cpDNA; Table S4: Simple sequence repeats (SSRs) in the E. microcarpus cpDNA; Table S5: Ka/Ks ratios of the cp genes from E. microcarpus and seven related species.
Author Contributions
Conceptualization, H.L. and W.G.; methodology, H.L. and M.C.; software, Z.W.; validation, H.L. and Z.H.; formal analysis, W.Z. and W.G.; investigation, L.L. and W.G.; resources, H.L. and X.Z.; data curation, M.C. and Z.W.; writing—original draft preparation, H.L. and W.G.; writing—review and editing, H.L. and W.G.; visualization, Z.H. and X.Z.; supervision, H.L. and W.G.; project administration, W.G.; funding acquisition, X.Z and W.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China, grant number 32171701.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data used in our study have been submitted to the NCBI GenBank (accession number: ON611716). The related species and their GenBank accession numbers in this study are listed as follows: Eurycorymbus cavaleriei (NC_037443.1), Koelreuteria paniculata (NC_037176.1), Sapindus mukorossi (NC_025554.1), Litchi chinensis (NC_035238.1), Dimocarpus longan (NC_037447.1), Acer wilsonii (NC_040988.1), Acer buergerianum (NC_034744.1), Acer pseudosieboldianum (NC_046487.1), Acer takesimense (NC_046488.1), Acer palmatum (NC_034932.1), Acer pentaphyllum (NC_049167.1), Acer triflorum (NC_047296.1), Acer nikoense (NC_049165.1), Acer griseum (NC_034346.1), Acer coriaceifolium (NC_050669.1), Acer davidii (NC_030331.1), Acer morrisonense (NC_029371.1), Acer longipes (NC_049126.1), Acer truncatum (NC_037211.1), Acer miaotaiense (NC_030343.1), Acer saccharum (NC_051960.1), Dipteronia sinensis (NC_029338.1), Dipteronia dyeriana (NC_031899.1), Aesculus wangii (NC_035955.1), Aesculus chinensis (NC_046788.1), Xanthoceras sorbifolium (NC_037448.1), Pistacia chinensis (NC_046786.1), Pistacia weinmaniifolia (NC_037471.1), Rhus chinensis (NC_033535.1), Turpinia montana (NC_051997.1), Turpinia arguta (NC_050925.1), Impatiens pritzelii (NC_047191.1), Euonymus japonicus (NC_028067.1), Euonymus schensianus (NC_036019.1), Dipentodon sinicus (NC_049157.1).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lin, S.; Liu, Y.; Zhang, X.; Jia, S. Development and utilization of germplasm resources of Euonymus. Jilin Fores. Sci. Techno. 2009, 38, 17–19. [Google Scholar]
- Zhu, J.-X.; Qin, J.-J.; Chang, R.-J.; Zeng, Q.; Cheng, X.-R.; Zhang, F.; Jin, H.-Z.; Zhang, W.-D. Chemical Constituents of Plants from the Genus Euonymus. Chem. Biodivers. 2012, 9, 1055–1076. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Ding, Y.; Zhao, T.; Zhang, L. Research progress on cutting propagation of Euonymus plant. North. Hortic. 2015, 12, 193–197. [Google Scholar] [CrossRef]
- Durrett, T.P.; McClosky, D.D.; Tumaney, A.W.; Elzinga, D.A.; Ohlrogge, J.; Pollard, M. A Distinct DGAT with Sn -3 Acetyltransferase Activity That Synthesizes Unusual, Reduced-Viscosity Oils in Euonymus and Transgenic Seeds. Proc. Natl. Acad. Sci. USA 2010, 107, 9464–9469. [Google Scholar] [CrossRef] [PubMed]
- Qin, H.; Wang, Y.; Zhao, X.; Zhang, F. Euonymus microcarpus (Oliv.) Sprague—A New Recorded Species of Euonymus (Celastraceae) in Shanxi. J. Shanxi. Univ. 2016, 1, 20. [Google Scholar]
- Institute of Botany, Chinese Academy of Sciences. Iconographia Cormophytorum Sinicorum (Tomus II); Science Press: Beijing, China, 1994; p. 666. ISBN 9787030041319. [Google Scholar]
- Editorial Committee of Chinese Materia Medica of National Administration of Traditional Chinese Medicine. Chinese Materia Medica; Science and Technology Press: Shanghai, China, 1998; Volume 13, p. 189. ISBN 9787532375820. [Google Scholar]
- Jin, S.; Daniell, H. The Engineered Chloroplast Genome Just Got Smarter. Trends in Plant Sci. 2015, 20, 622–640. [Google Scholar] [CrossRef]
- Gao, L.-Z.; Liu, Y.-L.; Zhang, D.; Li, W.; Gao, J.; Liu, Y.; Li, K.; Shi, C.; Zhao, Y.; Zhao, Y.-J.; et al. Evolution of Oryza Chloroplast Genomes Promoted Adaptation to Diverse Ecological Habitats. Commun. Biol. 2019, 2, 278. [Google Scholar] [CrossRef]
- Khan, M.; Ali, S.; Manghwar, H.; Saqib, S.; Ullah, F.; Ayaz, A.; Zaman, W. Melatonin Function and Crosstalk with Other Phytohormones under Normal and Stressful Conditions. Genes 2022, 13, 1699. [Google Scholar] [CrossRef]
- Naeem, M.; Shahzad, K.; Saqib, S.; Shahzad, A.; Nasrullah; Younas, M.; Afridi, M.I. The Solanum Melongena COP1LIKE Manipulates Fruit Ripening and Flowering Time in Tomato (Solanum lycopersicum). Plant Growth Regul. 2022, 96, 369–382. [Google Scholar] [CrossRef]
- Mehmood, F.; Abdullah; Ubaid, Z.; Bao, Y.; Poczai, P.; Mirza, B. Comparative Plastomics of Ashwagandha (Withania, Solanaceae) and Identification of Mutational Hotspots for Barcoding Medicinal Plants. Plants 2020, 9, 752. [Google Scholar] [CrossRef]
- Daniell, H.; Lin, C.-S.; Yu, M.; Chang, W.-J. Chloroplast Genomes: Diversity, Evolution, and Applications in Genetic Engineering. Genome Biol. 2016, 17, 134. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Dong, Y.; Liu, Y.; Yu, X.; Yang, M.; Huang, Y. Comparative Analyses of Euonymus Chloroplast Genomes: Genetic Structure, Screening for Loci with Suitable Polymorphism, Positive Selection Genes, and Phylogenetic Relationships within Celastrineae. Front. Plant Sci. 2021, 11, 593984. [Google Scholar] [CrossRef] [PubMed]
- Doyle, J.J.; Doyle, J.L. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem. Bull. 1987, 19, 11–15. [Google Scholar]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. Fastp: An Ultra-Fast All-in-One FASTQ Preprocessor. Bioinformatics 2018, 34, i884–i890. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Shi, L.; Zhu, Y.; Chen, H.; Zhang, J.; Lin, X.; Guan, X. CpGAVAS, an Integrated Web Server for the Annotation, Visualization, Analysis, and GenBank Submission of Completely Sequenced Chloroplast Genome Sequences. BMC Genom. 2012, 13, 715. [Google Scholar] [CrossRef] [PubMed]
- Lohse, M.; Drechsel, O.; Kahlau, S.; Bock, R. OrganellarGenomeDRAW—A Suite of Tools for Generating Physical Maps of Plastid and Mitochondrial Genomes and Visualizing Expression Data Sets. Nucleic Acids Res. 2013, 41, W575–W581. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, S.; Choudhuri, J.V.; Ohlebusch, E.; Schleiermacher, C.; Stoye, J.; Giegerich, R. REPuter: The Manifold Applications of Repeat Analysis on a Genomic Scale. Nucleic Acids Res. 2001, 29, 4633–4642. [Google Scholar] [CrossRef]
- Kurtz, S.; Schleiermacher, C. REPuter: Fast Computation of Maximal Repeats in Complete Genomes. Bioinformatics 1999, 15, 426–427. [Google Scholar] [CrossRef]
- Beier, S.; Thiel, T.; Münch, T.; Scholz, U.; Mascher, M. MISA-Web: A Web Server for Microsatellite Prediction. Bioinformatics 2017, 33, 2583–2585. [Google Scholar] [CrossRef]
- Kuraku, S.; Zmasek, C.M.; Nishimura, O.; Katoh, K. ALeaves Facilitates On-Demand Exploration of Metazoan Gene Family Trees on MAFFT Sequence Alignment Server with Enhanced Interactivity. Nucleic Acids Res. 2013, 41, W22–W28. [Google Scholar] [CrossRef]
- Amiryousefi, A.; Hyvönen, J.; Poczai, P. IRscope: An Online Program to Visualize the Junction Sites of Chloroplast Genomes. Bioinformatics 2018, 34, 3030–3031. [Google Scholar] [CrossRef] [PubMed]
- Mayor, C.; Brudno, M.; Schwartz, J.R.; Poliakov, A.; Rubin, E.M.; Frazer, K.A.; Pachter, L.S.; Dubchak, I. VISTA: Visualizing Global DNA Sequence Alignments of Arbitrary Length. Bioinformatics 2000, 16, 1046–1047. [Google Scholar] [CrossRef]
- Nanjala, C.; Wanga, V.O.; Odago, W.; Mutinda, E.S.; Waswa, E.N.; Oulo, M.A.; Mkala, E.M.; Kuja, J.; Yang, J.-X.; Dong, X.; et al. Plastome Structure of 8 Calanthe s.l. Species (Orchidaceae): Comparative Genomics, Phylogenetic Analysis. BMC Plant Biol. 2022, 22, 387. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Ge, X.; Cano, A.; Salazar, B.G.M.; Deng, Y. Comparative Analysis of Chloroplast Genomes for Five Dicliptera Species (Acanthaceae): Molecular Structure, Phylogenetic Relationships, and Adaptive Evolution. PeerJ 2020, 8, e8450. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Ye, W.; Lu, R.; Xu, W.; Qiu, Y. Phylogenomic and Comparative Analyses of Complete Plastomes of Croomia and Stemona (Stemonaceae). Int. J. Mol. Sci. 2018, 19, 2383. [Google Scholar] [CrossRef] [PubMed]
- Jansen, R.K.; Raubeson, L.A.; Boore, J.L.; dePamphilis, C.W.; Chumley, T.W.; Haberle, R.C.; Wyman, S.K.; Alverson, A.J.; Peery, R.; Herman, S.J.; et al. Methods for Obtaining and Analyzing Whole Chloroplast Genome Sequences. Methods Enzymol. 2005, 395, 348–384. [Google Scholar] [CrossRef]
- Wang, X.; Li, H.; Zheng, M.; Jiang, J. The Complete Chloroplast Genome of Euonymus szechuanensis. Mitochondrial DNA Part B 2020, 5, 1130–1131. [Google Scholar] [CrossRef]
- Meunier, J.; Duret, L. Recombination Drives the Evolution of GC-Content in the Human Genome. Mol. Biol. Evol. 2004, 21, 984–990. [Google Scholar] [CrossRef]
- Pezoa, I.; Villacreses, J.; Rubilar, M.; Pizarro, C.; Galleguillos, M.J.; Ejsmentewicz, T.; Fonseca, B.; Espejo, J.; Polanco, V.; Sánchez, C. Generation of Chloroplast Molecular Markers to Differentiate Sophora toromiro and Its Hybrids as a First Approach to Its Reintroduction in Rapa Nui (Easter Island). Plants 2021, 10, 342. [Google Scholar] [CrossRef]
- Pereira, G.S.; Nunes, E.S.; Laperuta, L.D.C.; Braga, M.F.; Penha, H.A.; Diniz, A.L.; Munhoz, C.F.; Gazaffi, R.; Garcia, A.A.F.; Vieira, M.L.C. Molecular Polymorphism and Linkage Analysis in Sweet Passion Fruit, an Outcrossing Species. Ann. Appl. Biol. 2013, 162, 347–361. [Google Scholar] [CrossRef]
- Muller, J.; Leenhouts, P.W. A general survey of pollen types in Sapindaceae in relation to taxonomy. In The Evolutionary Significance of the Exine; Ferguson, I.K., Muller, J., Eds.; Academic Press: London, UK, 1976; Volume 1, pp. 407–445. ISBN 9780122536502. [Google Scholar]
- An, M.-T.; Cui, X.-Y.; Yang, J.-X.; Hu, G.-X. The Complete Chloroplast Genome of the Threatened Dipentodon sinicus (Dipentodontaceae). J. Genet. 2019, 98, 4. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Qian, J.; Li, X.; Sun, Z.; Xu, X.; Chen, S. Complete Chloroplast Genome of Medicinal Plant Lonicera Japonica: Genome Rearrangement, Intron Gain and Loss, and Implications for Phylogenetic Studies. Molecules 2017, 22, 249. [Google Scholar] [CrossRef] [PubMed]
- Abdullah; Henriquez, C.L.; Mehmood, F.; Carlsen, M.M.; Islam, M.; Waheed, M.T.; Poczai, P.; Croat, T.B.; Ahmed, I. Complete Chloroplast Genomes of Anthurium huixtlense and Pothos scandens (Pothoideae, Araceae): Unique Inverted Repeat Expansion and Contraction Affect Rate of Evolution. J. Mol. Evol. 2020, 88, 562–574. [Google Scholar] [CrossRef] [PubMed]
- Palmer, J.D. Comparative Organization of Chloroplast Genomes. Annu. Rev. Genet. 1985, 19, 325–354. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).