High-Throughput Genotyping of Resilient Tomato Landraces to Detect Candidate Genes Involved in the Response to High Temperatures
Abstract
1. Introduction
2. Materials and Methods
2.1. Phenotypic Evaluation
2.2. Statistical Analysis
2.3. Genotyping Analysis
3. Results
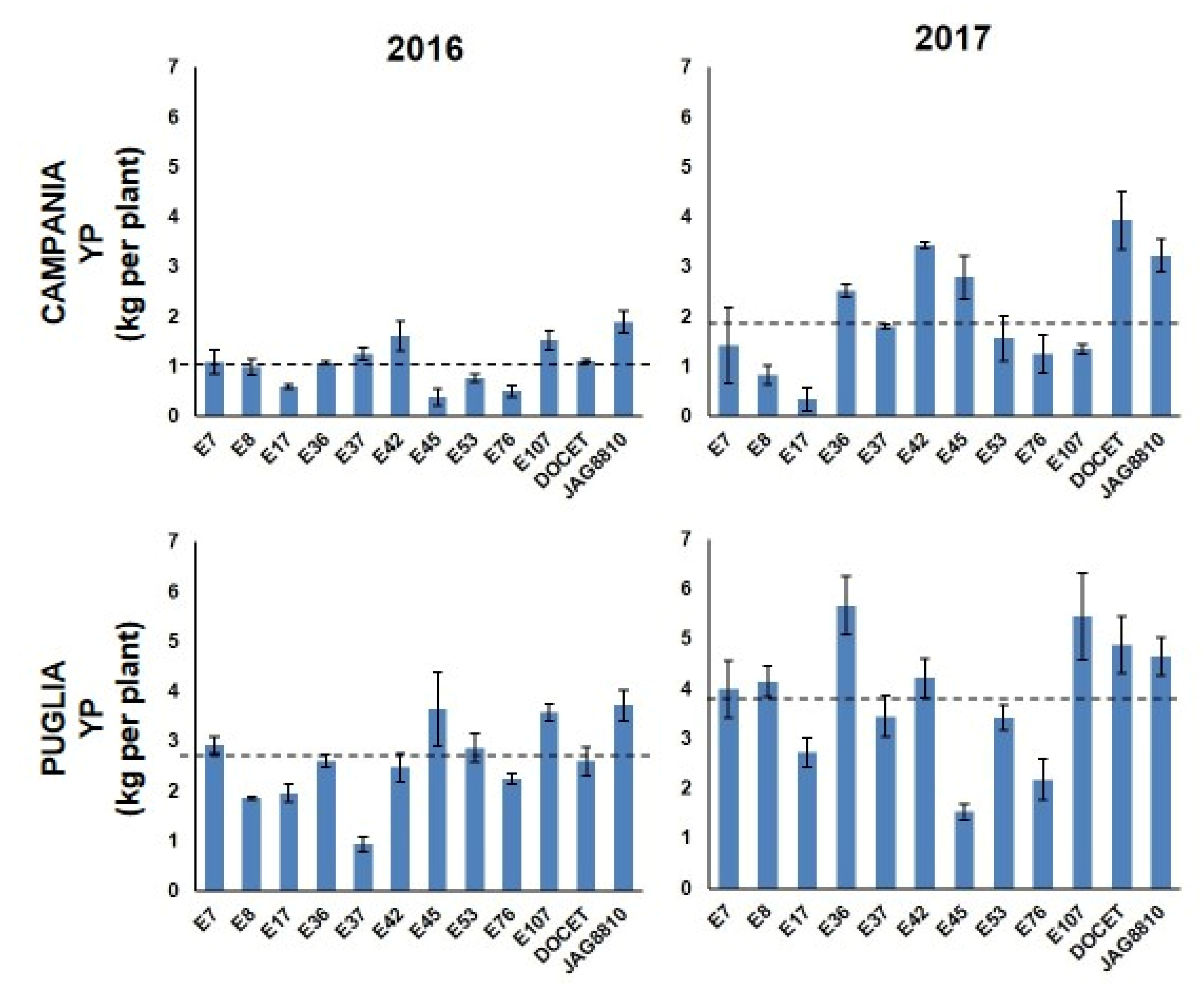
3.1. Phenotypic Analysis
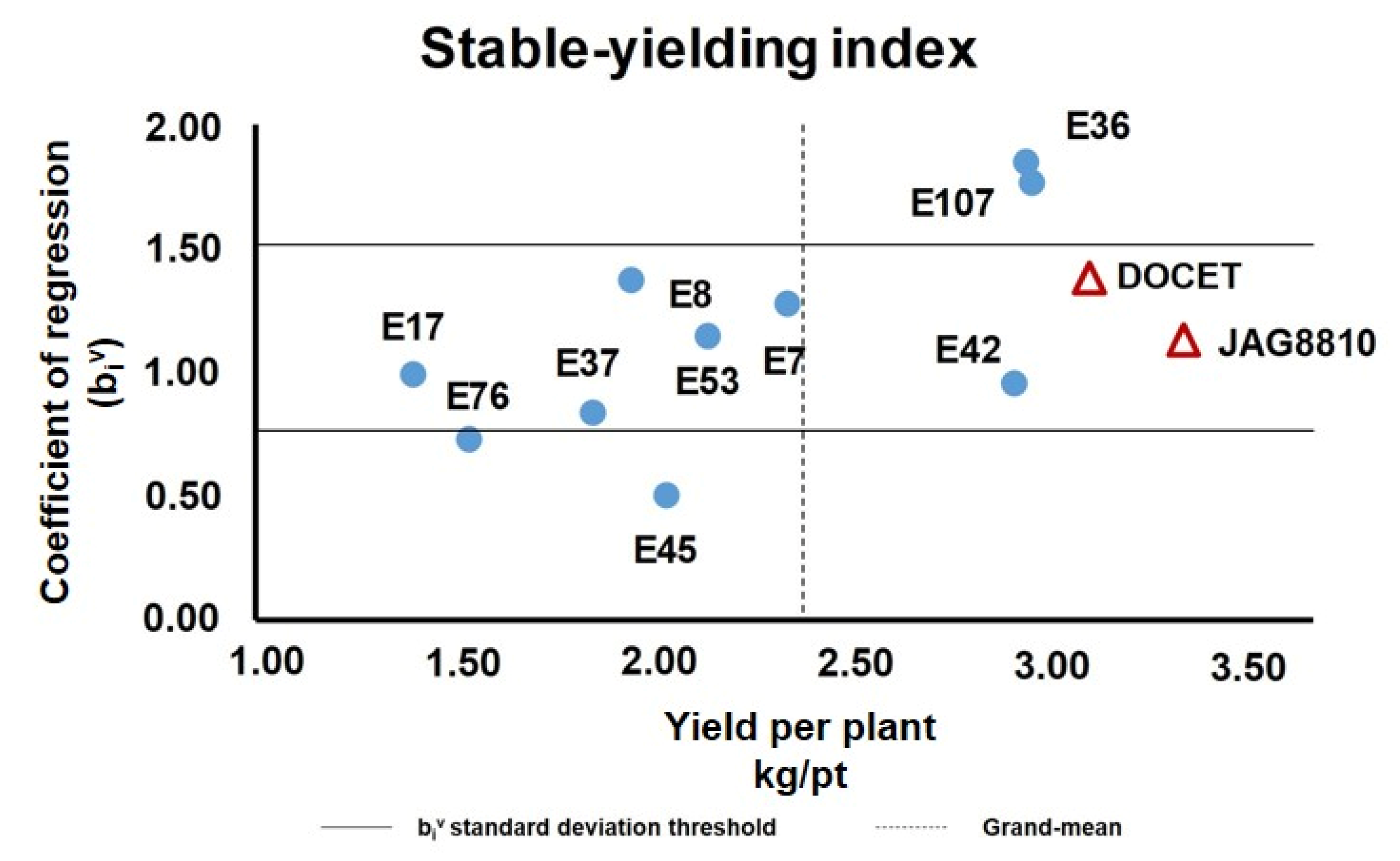
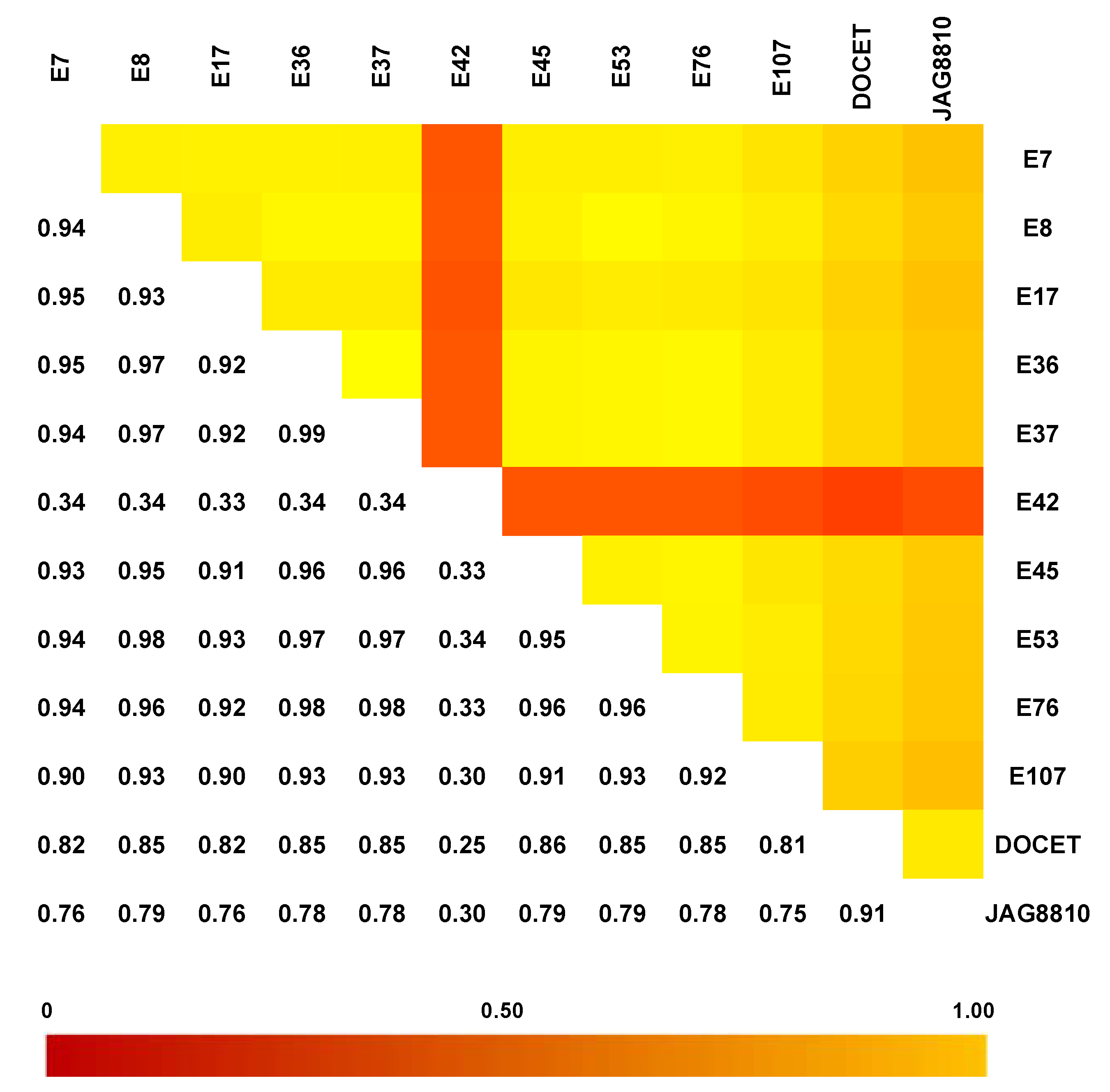
3.2. Selection of Stable-Yielding Genotypes
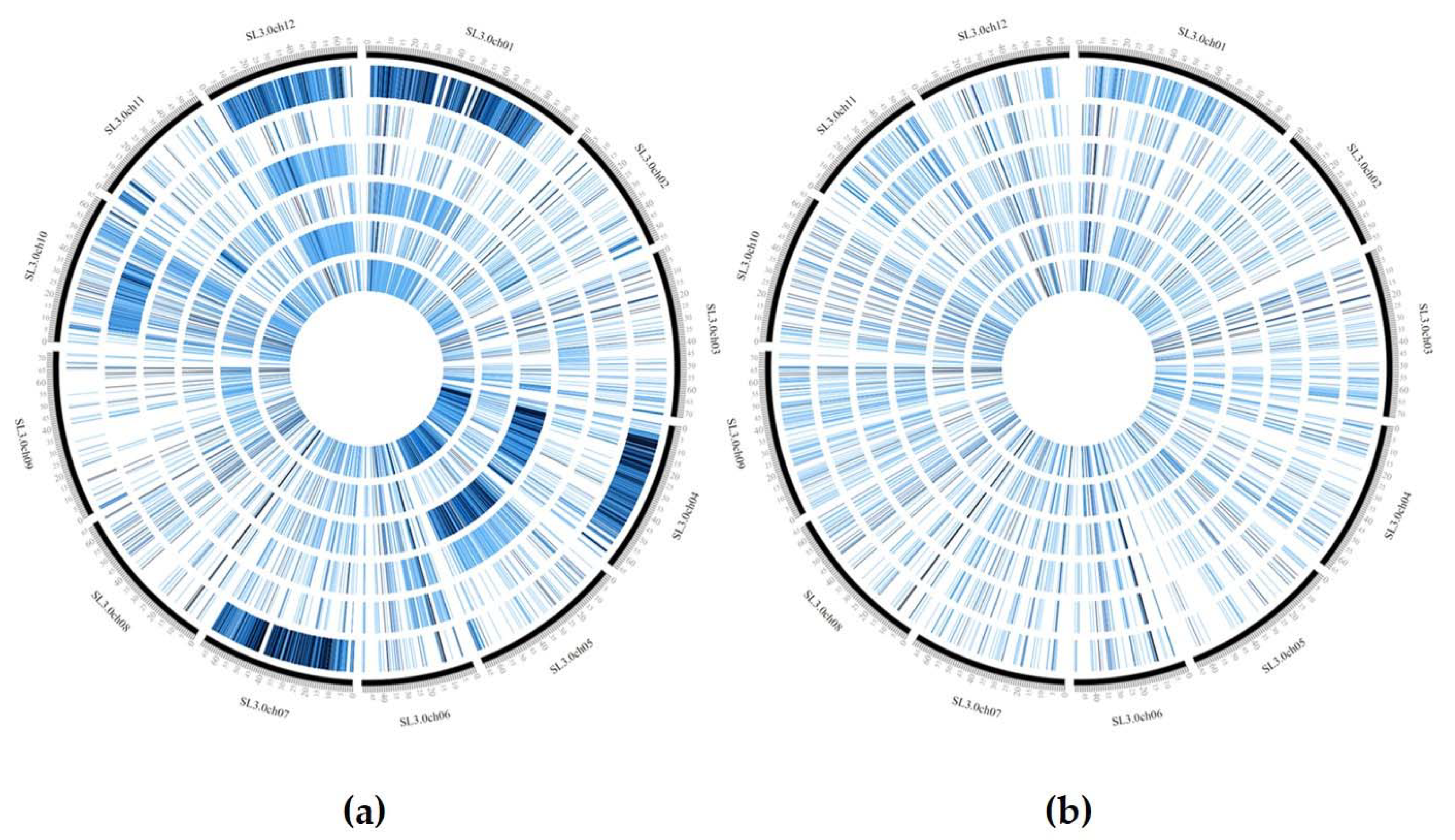
3.3. GBS Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ainsworth, E.A.; Ort, D.R. How Do We Improve Crop Production in a Warming World? Plant Physiol. 2010, 154, 526–530. [Google Scholar] [CrossRef] [PubMed]
- Gourdji, S.M.; Sibley, A.M.; Lobell, D.B. Global crop exposure to critical high temperatures in the reproductive period: Historical trends and future projections. Environ. Res. Lett. 2013, 8, 024041. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Alam, M.; Roychowdhury, R.; Fujita, M. Physiological, Biochemical, and Molecular Mechanisms of Heat Stress Tolerance in Plants. Int. J. Mol. Sci. 2013, 14, 9643–9684. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Peet, M.M.; Gardner, R.G. Altered flower retention and developmental patterns in nine tomato cultivars under elevated temperature. Sci. Hortic. 2004, 101, 95–101. [Google Scholar] [CrossRef]
- Warland, J.; McKeown, A.W.; McDonald, M.R. Impact of high air temperatures on Brassicacae crops in southern Ontario. Can. J. Plant Sci. 2006, 86, 1209–1215. [Google Scholar] [CrossRef]
- Lavell, A.; Oppenheimer, M.; Diop, C.; Hess, J.; Lempert, R.; Li, J.; Myeong, S. Managing the Risks of Extreme Events and Disasters to Advance Climate Change Adaptation; A Special Report of Working Groups I and II of the Intergovernmental Panel on Climate Change (IPCC); Intergovernmental Panel on Climate Change (IPCC): Geneva, Switzerland, 2012; pp. 25–64. [Google Scholar]
- Paupière, M.J.; Van Haperen, P.; Rieu, I.; Visser, R.G.F.; Tikunov, Y.M.; Bovy, A.G. Screening for pollen tolerance to high temperatures in tomato. Euphytica 2017, 213, 130. [Google Scholar] [CrossRef]
- Pingali, P.L. Green revolution: Impacts, limits, and the path ahead. Proc. Natl. Acad. Sci. USA 2012, 109, 12302–12308. [Google Scholar] [CrossRef]
- Massawe, F.; Mayes, S.; Cheng, A. Crop Diversity: An Unexploited Treasure Trove for Food Security. Trends Plant Sci. 2016, 21, 365–368. [Google Scholar] [CrossRef]
- Tieman, D.; Zhu, G.; Resende, M.F.R.; Lin, T.; Nguyen, C.; Bies, D.; Rambla, J.; Beltran, K.S.O.; Taylor, M.; Zhang, B.; et al. A chemical genetic roadmap to improved tomato flavor. Science 2017, 355, 391–394. [Google Scholar] [CrossRef]
- Stapley, J.; Reger, J.; Feulner, P.G.D.; Smadja, C.; Galindo, J.; Ekblom, R.; Bennison, C.; Ball, A.D.; Beckerman, A.P.; Slate, J. Adaptation genomics: The next generation. Trends Ecol. Evol. 2010, 25, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Tao, Y.; Zheng, Z.; Li, C.; Sweetingham, M.W.; Howieson, J. Application of next-generation sequencing for rapid marker development in molecular plant breeding: A case study on anthracnose disease resistance in Lupinus angustifolius L. BMC Genom. 2012, 13, 318. [Google Scholar] [CrossRef] [PubMed]
- Glaubitz, J.C.; Casstevens, T.M.; Lu, F.; Harriman, J.; Elshire, R.; Sun, Q.; Buckler, E.S. TASSEL-GBS: A High Capacity Genotyping by Sequencing Analysis Pipeline. PLoS ONE 2014, 9, e90346. [Google Scholar] [CrossRef] [PubMed]
- Davey, J.W.; Hohenlohe, P.A.; Etter, P.D.; Boone, J.Q.; Catchen, J.M.; Blaxter, M. Genome-wide genetic marker discovery and genotyping using next-generation sequencing. Nat. Rev. Genet. 2011, 12, 499–510. [Google Scholar] [CrossRef]
- Dodsworth, S.; Chase, M.W.; Sarkinen, T.; Knapp, S.; Leitch, A.R. Using genomic repeats for phylogenomics: A case study in wild tomatoes (Solanum section Lycopersicon: Solanaceae). Biol. J. Linn. Soc. 2015, 117, 96–105. [Google Scholar] [CrossRef]
- Ruggieri, V.; Francese, G.; Sacco, A.; D’Alessandro, A.; Rigano, M.M.; Parisi, M.; Milone, M.; Cardi, T.; Mennella, G.; Barone, A. An association mapping approach to identify favourable alleles for tomato fruit quality breeding. BMC Plant Biol. 2014, 14, 337. [Google Scholar] [CrossRef]
- Sacco, A.; Ruggieri, V.; Parisi, M.; Festa, G.; Rigano, M.M.; Picarella, M.E.; Mazzucato, A.; Barone, A. Exploring a Tomato Landraces Collection for Fruit-Related Traits by the Aid of a High-Throughput Genomic Platform. PLoS ONE 2015, 10, e0137139. [Google Scholar] [CrossRef]
- Ruggieri, V.; Calafiore, R.; Schettini, C.; Rigano, M.M.; Olivieri, F.; Frusciante, L.; Barone, A. Exploiting Genetic and Genomic Resources to Enhance Heat-Tolerance in Tomatoes. Agronomy 2019, 9, 22. [Google Scholar] [CrossRef]
- Eberhart, S.A.; Russell, W.A. Stability Parameters for Comparing Varieties 1. Crop. Sci. 1966, 6, 36–40. [Google Scholar] [CrossRef]
- Peterson, B.K.; Weber, J.N.; Kay, E.H.; Fisher, H.S.; Hoekstra, H.E. Double Digest RADseq: An Inexpensive Method for De Novo SNP Discovery and Genotyping in Model and Non-Model Species. PLoS ONE 2012, 7, e37135. [Google Scholar] [CrossRef]
- Catchen, J.; Hohenlohe, P.A.; Bassham, S.; Amores, A.; Cresko, W.A. Stacks: An analysis tool set for population genomics. Mol. Ecol. 2013, 22, 3124–3140. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- Drori, E.; Levy, D.; Smirin-Yosef, P.; Rahimi, O.; Salmon-Divon, M. CircosVCF: Circos visualization of whole-genome sequence variations stored in VCF files. Bioinformatics 2017, 33, 1392–1393. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Chow, C.C.; Tellier, L.C.A.M.; Vattikuti, S.; Purcell, S.M.; Lee, J.J. Second-generation PLINK: Rising to the challenge of larger and richer datasets. GigaScience 2015, 4, 7. [Google Scholar] [CrossRef] [PubMed]
- Jegadeesan, S.; Beery, A.; Altahan, L.; Meir, S.; Pressman, E.; Firon, N. Ethylene production and signaling in tomato (Solanum lycopersicum) pollen grains is responsive to heat stress conditions. Plant Reprod. 2018, 31, 367–383. [Google Scholar] [CrossRef] [PubMed]
- Marko, D.; El-Shershaby, A.; Carriero, F.; Summerer, S.; Petrozza, A.; Iannacone, R.; Schleiff, E.; Fragkostefanakis, S. Identification and Characterization of a Thermotolerant TILLING Allele of Heat Shock Binding Protein 1 in Tomato. Genes 2019, 10, 516. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, R.K.; Handa, A.K.; Mattoo, A.K. Transcript Abundance Patterns of 9-and 13-Lipoxygenase Subfamily Gene Members in Response to Abiotic Stresses (Heat, Cold, Drought or Salt) in Tomato (Solanum lycopersicum L.) Highlights Member-Specific Dynamics Relevant to Each Stress. Genes 2019, 10, 683. [Google Scholar] [CrossRef]
- Sato, S.; Kamiyama, M.; Iwata, T.; Makita, N.; Furukawa, H.; Ikeda, H. Moderate Increase of Mean Daily Temperature Adversely Affects Fruit Set of Lycopersicon esculentum by Disrupting Specific Physiological Processes in Male Reproductive Development. Ann. Bot. 2006, 97, 731–738. [Google Scholar] [CrossRef]
- Mesihovic, A.; Iannacone, R.; Firon, N.; Fragkostefanakis, S. Heat stress regimes for the investigation of pollen thermotolerance in crop plants. Plant Reprod. 2016, 29, 93–105. [Google Scholar] [CrossRef]
- Müller, F.; Rieu, I. Acclimation to high temperature during pollen development. Plant Reprod. 2016, 29, 107–118. [Google Scholar] [CrossRef]
- Xu, J.; Driedonks, N.; Rutten, M.J.M.; Vriezen, W.H.; De Boer, G.-J.; Rieu, I. Mapping quantitative trait loci for heat tolerance of reproductive traits in tomato (Solanum lycopersicum). Mol. Breed. 2017, 37, 114. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Wolters-Arts, M.; Mariani, C.; Huber, H.; Rieu, I. Heat stress affects vegetative and reproductive performance and trait correlations in tomato (Solanum lycopersicum). Euphytica 2017, 213, 156. [Google Scholar] [CrossRef]
- Araus, J.L.; Kefauver, S.; Zaman-Allah, M.; Olsen, M.S.; Cairns, J.E. Translating High-Throughput Phenotyping into Genetic Gain. Trends Plant Sci. 2018, 23, 451–466. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, J.; Inzé, D.; Nelissen, H.; Saibo, N. Source-Sink Regulation in Crops under Water Deficit. Trends Plant Sci. 2019, 24, 652–663. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Singh, S.K.; Srivastava, K. Stability Analysis in Tomato Inbreds and Their F1s for Yield and Quality Traits. Agric. Res. 2018, 8, 141–147. [Google Scholar] [CrossRef]
- Ayenan, M.A.T.; Danquah, A.; Hanson, P.; Ampomah-Dwamena, C.; Sodedji, F.A.K.; Asante, I.K.; Danquah, E.Y. Accelerating Breeding for Heat Tolerance in Tomato (Solanum lycopersicum L.): An Integrated Approach. Agronomy 2019, 9, 720. [Google Scholar] [CrossRef]
- Driedonks, N.; Wolters-Arts, M.; Huber, H.; De Boer, G.-J.; Vriezen, W.; Mariani, C.; Rieu, I. Exploring the natural variation for reproductive thermotolerance in wild tomato species. Euphytica 2018, 214, 67. [Google Scholar] [CrossRef]
- Gonzalo, M.J.; Li, Y.-C.; Chen, K.-Y.; Gil, D.; Montoro, T.; Náquera, I.; Baixauli, C.; Granell, A.; Monforte Gilabert, A.J. Genetic control of reproductive trraits in tomatoes under high temperature. Front. Plant Sci. 2020, 11, 326. [Google Scholar] [CrossRef]
- Wen, J.; Jiang, F.; Weng, Y.; Sun, M.; Shi, X.; Zhou, Y.; Yu, L.; Wu, Z. Identification of heat-tolerance QTLs and high-temperature stress-responsive genes through conventional QTL mapping, QTL-seq and RNA-seq in tomato. BMC Plant Biol. 2019, 19, 398–417. [Google Scholar] [CrossRef]
- Hernández-Bautista, A.; Lobato-Ortiz, R.; Cruz-Izquierdo, S.; García-Zavala, J.J.; Chávez-Servia, J.L.; Hernández-Leal, E.; Bonilla-Barrientos, O.; Gainza, F.; Opazo, I.; Munoz, C. Fruit size QTLs affect in a major proportion the yield in tomato. Chil. J. Agric. Res. 2015, 75, 402–409. [Google Scholar] [CrossRef]
- Zhang, S.; Yu, H.; Wang, K.; Zheng, Z.; Liu, L.; Xu, M.; Jiao, Z.; Li, R.; Liu, X.; Li, J.; et al. Detection of major loci associated with the variation of 18 important agronomic traits between Solanum pimpinellifolium and cultivated tomatoes. Plant J. 2018, 95, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Pfab, A.; Breindl, M.; Grasser, K.D. The Arabidopsis histone chaperone FACT is required for stress-induced expression of anthocyanin biosynthetic genes. Plant Mol. Biol. 2018, 96, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, S.; Tuteja, N. Cold, salinity and drought stresses: An overview. Arch. Biochem. Biophys. 2005, 444, 139–158. [Google Scholar] [CrossRef] [PubMed]
- Owttrim, G.W. RNA helicases and abiotic stress. Nucleic Acids Res. 2006, 34, 3220–3230. [Google Scholar] [CrossRef]
- Feng, C.Z.; Chen, Y.; Wang, C.; Kong, Y.H.; Wu, W.H.; Chen, Y.F. Arabidopsis RAV 1 transcription factor, phosphorylated by S n RK 2 kinases, regulates the expression of ABI 3, ABI 4, and ABI 5 during seed germination and early seedling development. Plant J. 2014, 80, 654–668. [Google Scholar] [CrossRef]
- Fu, M.; Kang, H.K.; Son, S.-H.; Kim, S.-K.; Nam, K.H. A Subset of Arabidopsis RAV Transcription Factors Modulates Drought and Salt Stress Responses Independent of ABA. Plant Cell Physiol. 2014, 55, 1892–1904. [Google Scholar] [CrossRef]
- Eom, S.; Lee, H.; Lee, J.; Wi, S.; Kim, S.K.; Hyun, T.K. Identification and Functional Prediction of Drought-Responsive Long Non-Coding RNA in Tomato. Agronomy 2019, 9, 629. [Google Scholar] [CrossRef]
- Xu, T.; Wang, Y.; Liu, X.; Lv, S.; Feng, C.; Qi, M.; Li, T. Small RNA and degradome sequencing reveals microRNAs and their targets involved in tomato pedicel abscission. Planta 2015, 242, 963–984. [Google Scholar] [CrossRef]
- Perrakis, A.; Bita, C.E.; Arhondakis, S.; Krokida, A.; Mekkaoui, K.; Denic, D.; Blazakis, K.; Kaloudas, D.; Kalaitzis, P. Suppression of a Prolyl 4 Hydroxylase Results in Delayed Abscission of Overripe Tomato Fruits. Front. Plant Sci. 2019, 10, 348. [Google Scholar] [CrossRef]
- Yu, C.; Cai, X.; Ye, Z.; Li, H. Genome-wide identification and expression profiling analysis of trihelix gene family in tomato. Biochem. Biophys. Res. Commun. 2015, 468, 653–659. [Google Scholar] [CrossRef]
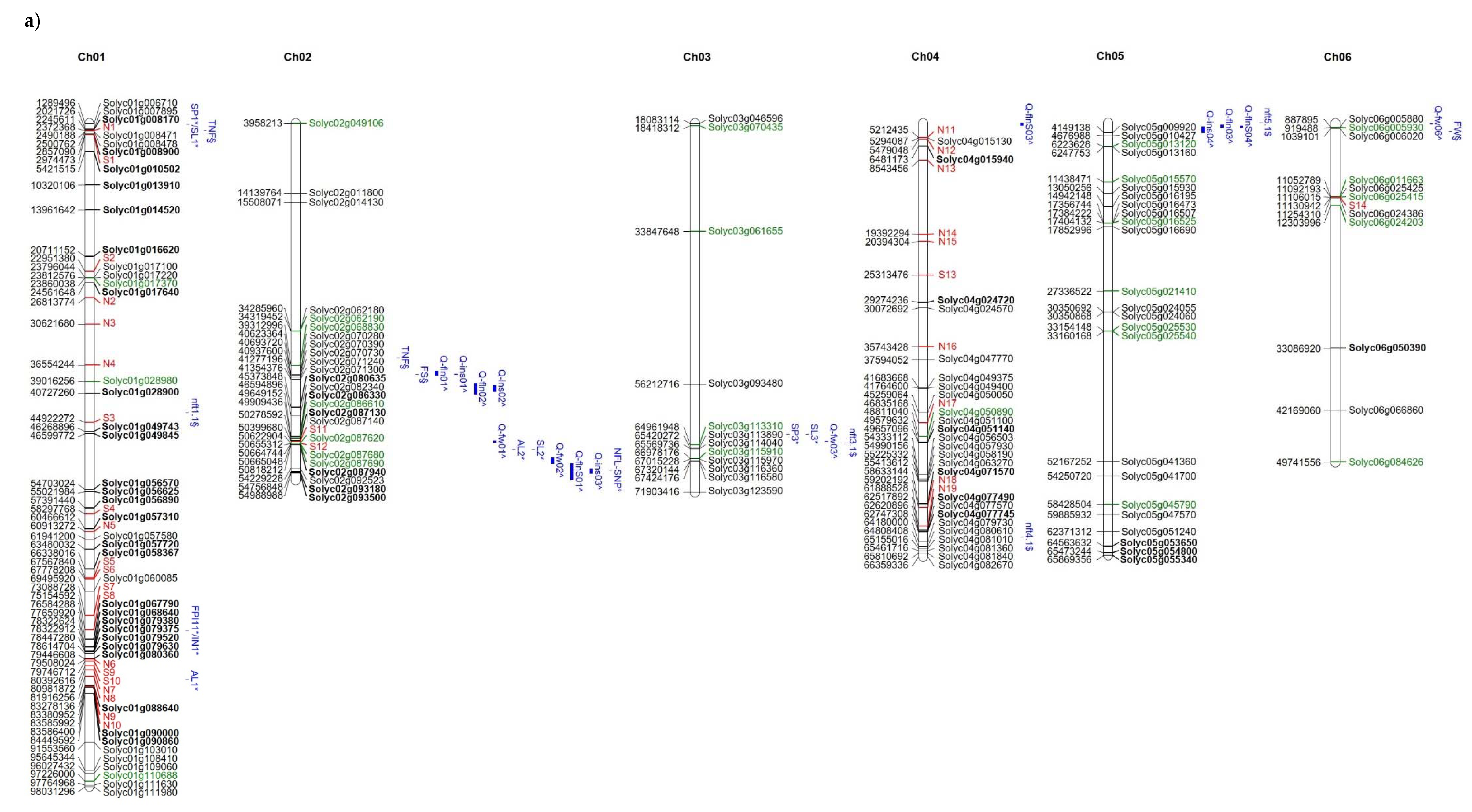
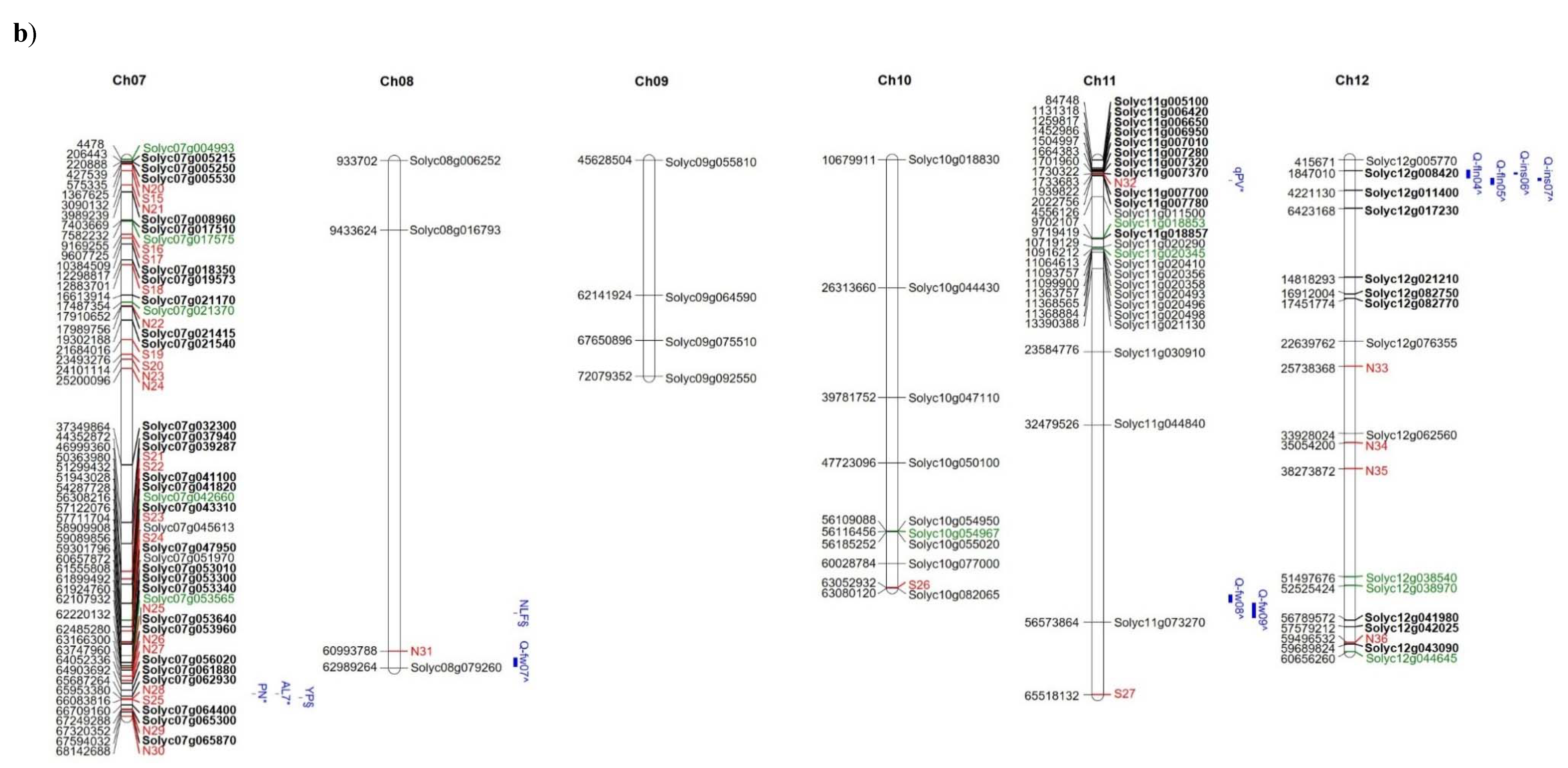
| Trait | Field | Controls | E7 | E8 | E17 | E36 | E37 | E42 | E45 | E53 | E76 | E107 | DOCET |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NFL | C2016 | D | * | ** | ** | * | |||||||
| J | * | ** | * | * | |||||||||
| C2017 | D | * | * | * | ** | ||||||||
| J | * | * | ** | ||||||||||
| P2016 | D | * | |||||||||||
| J | * | ** | *** | * | * | *** | * | ||||||
| P2017 | D | * | * | ** | * | ||||||||
| J | * | * | ** | * | |||||||||
| FS | C2016 | D | * | * | |||||||||
| J | * | ** | * | ** | |||||||||
| C2017 | D | * | |||||||||||
| J | ** | * | ** | ** | ** | ||||||||
| P2016 | D | ** | * | * | ** | * | ** | ** | |||||
| J | *** | * | * | ||||||||||
| P2017 | D | * | * | ||||||||||
| J | |||||||||||||
| TNF | C2016 | D | * | ** | * | ** | ** | * | * | ||||
| J | ** | * | ** | ** | * | ||||||||
| C2017 | D | * | ** | *** | * | * | |||||||
| J | *** | ** | ** | *** | * | ||||||||
| P2016 | D | ** | ** | * | ** | *** | * | ||||||
| J | ** | * | * | ** | *** | ||||||||
| P2017 | D | ** | ** | * | |||||||||
| J | ** | * | ** | ||||||||||
| FW | C2016 | D | * | ** | *** | * | * | *** | * | ** | |||
| J | *** | *** | ** | ** | *** | *** | ** | *** | |||||
| C2017 | D | *** | *** | *** | *** | *** | *** | *** | |||||
| J | * | * | * | * | * | ** | * | ||||||
| P2016 | D | *** | *** | *** | *** | *** | *** | * | *** | ||||
| J | ** | ** | ** | ** | ** | ** | * | ||||||
| P2017 | D | *** | ** | ||||||||||
| J | * | *** | ** | * | |||||||||
| YP | C2016 | D | *** | *** | *** | ** | |||||||
| J | * | ** | * | ** | ** | ** | * | ||||||
| C2017 | D | ** | ** | * | * | * | * | ||||||
| J | ** | *** | ** | *** | * | ** | *** | ||||||
| P2016 | D | ** | * | ||||||||||
| J | ** | ** | * | ** | * | * | |||||||
| P2017 | D | * | ** | * | |||||||||
| J | * | ** | * |
| Marker | E7 | E8 | E17 | E36 | E37 | E42 | E45 | E53 | E76 | E107 | DOCET | JAG8810 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SNP | 93 | 29 | 288 | 11 | 11 | 11,050 | 99 | 47 | 76 | 921 | 51 | 186 |
| InDel | 29 | 16 | 55 | 14 | 23 | 1640 | 41 | 36 | 20 | 83 | 57 | 99 |
| Type of Mutation | Total Mutations (no.) | Effect on Protein | Affected Genes (no.) | |||
|---|---|---|---|---|---|---|
| High (no.) | Moderate (no.) | Low (no.) | Modifier (no.) | |||
| SNP | 22,594 | 15 | 239 | 252 | 22,088 | 4124 |
| InDel | 3519 | 22 | 44 | 24 | 3429 | 1863 |
| Total | 26,113 | 37 | 283 | 276 | 25,517 | 5987 |
| Gene | Mutation | Position (SL3.0) | Mutated Genotypes | Predicted Effect | Protein Function |
|---|---|---|---|---|---|
| Solyc01g028980 | InDel | 39,016,299 | E42 | stop_gained | Gamma-tubulin complex component |
| SNP | 39,016,322 | E42 | stop_gained | ||
| Solyc02g049106 | SNP | 3,958,278 | E8, E17, E36, E42, E53, E76, E107, DOCET | stop_gained | Leucine-rich repeat protein kinase family protein |
| Solyc02g086610 | SNP | 49,909,435 | E42 | splice_acceptor_variant and intron_variant | Isocitrate dehydrogenase [NADP] |
| Solyc02g087620 | InDel | 50,622,904 | E42 | splice_donor_variant, intron_variant | Inositol hexakisphosphate and diphosphoinositol-pentakisphosphate kinase |
| Solyc02g087680 | InDel | 50,664,745 | E42 | frameshift_variant | FACT complex subunit SSRP1 |
| Solyc02g087690 | SNP | 50,665,049 | E42 | stop_lost, splice_region_variant | FACT complex subunit SSRP1 |
| Solyc03g070435 | InDel | 18,418,311 | E8, E53, E76, E107 | frameshift_variant | Alpha-mannosidase |
| Solyc03g061655 | InDel | 33,847,933 | E7, E8, E17, E36, E37, E42, E53 | frameshift_variant | Ribosomal protein S12 |
| Solyc03g113310 | InDel | 64,961,947 | DOCET, JAG88100 | frameshift_variant | Pseudouridine synthase family protein |
| Solyc03g115910 | InDel | 66,978,174 | E8, E53 | frameshift_variant | MADS-box transcription factor |
| Solyc04g050890 | SNP | 48,811,041 | E42 | stop_gained | DNA-directed RNA polymerase subunit alpha |
| Solyc05g013120 | SNP | 6,223,628 | E36, E37 | stop_gained | Ninja-family protein AFP1 |
| Solyc05g015570 | SNP | 11,438,471 | DOCET, JAG88100 | start_lost | UDP-glucose 6-dehydrogenase 1 |
| Solyc05g016525 | InDel | 17,404,131 | DOCET, JAG88100 | frameshift_variant | Core-2/I-branching beta-1 |
| Solyc05g021410 | InDel | 27,336,522 | E8, E36, E42, E45, E76, E107, DOCET | frameshift_variant | Histone-lysine N-methyltransferase SUVR5 |
| Solyc05g025530 | InDel | 33,154,148 | DOCET, JAG88100 | frameshift_variant | DNA-directed RNA polymerase subunit beta |
| Solyc05g025540 | SNP | 33,160,167 | DOCET, JAG88100 | stop_gained | Molybdenum cofactor sulfurase |
| Solyc05g045790 | SNP | 58,428,505 | DOCET, JAG88100 | splice_donor_variant, intron_variant | Cytochrome c oxidase subunit 2 |
| Solyc06g005930 | InDel | 919,488 | E7, E8, E42, E45, E53, E76, DOCET | frameshift_variant | Protein sensitivity to red light reduced 1 |
| Solyc06g011663 | SNP | 11,052,789 | E7, E8, E36, E37, E42, E45, E53, E76 | stop_gained | Beta glucosidase 25 |
| Solyc06g025415 | InDel | 11,106,015 | E7, E8, E17, E36, E37, E42, E45, E107, DOCET | frameshift_variant | Mediator of RNA polymerase II transcription subunit 14 |
| Solyc06g024203 | InDel | 12,303,996 | E8, E17, E36, E42, E53, E76, E107, JAG8810 | frameshift_variant, splice_region_variant | Peroxidase superfamily protein |
| Solyc06g084626 | InDel | 49,741,555 | E8, E45, E107, JAG8810 | frameshift_variant | 1-deoxy-D-xylulose 5-phosphate reductoisomerase |
| InDel | 49,741,558 | E17, E45, E107, DOCET, JAG8810 | frameshift_variant | ||
| Solyc07g004993 | InDel | 4478 | E17, E42, E76, DOCET | start_lost | Phosphatidylinositol N-acetyglucosaminlytransferase subunit P-like protein |
| Solyc07g017575 | InDel | 7,582,232 | E42 | stop_gained | Flavin-containing monooxygenase |
| Solyc07g021370 | SNP | 17,487,354 | E42 | stop_lost; splice_region_variant | DNA-directed DNA polymerase |
| Solyc07g042660 | SNP | 56,308,217 | E42 | splice_acceptor_variant&intron_variant | Helicase SNF2 domain-containing protein |
| Solyc07g053565 | InDel | 62,107,933 | E42 | frameshift_variant | NAD(P)H-quinone oxidoreductase subunit 2 |
| Solyc10g054967 | SNP | 56,116,455 | E17, E42 | stop_gained | Mediator of RNA polymerase II transcription subunit 20-like protein |
| Solyc11g018853 | InDel | 9,702,107 | E42, JAG8810 | frameshift_variant | Adenylate isopentenyltransferase |
| Solyc11g020345 | SNP | 10,916,212 | JAG8810 | splice_acceptor_variant and intron_variant | Small nuclear ribonucleoprotein family protein |
| Solyc12g038540 | InDel | 51,497,675 | E42 | frameshift_variant, splice_acceptor_variant, splice_region_variant, intron_variant | Transducin/WD40 repeat-like superfamily protein |
| Solyc12g038970 | InDel | 52,525,425 | E8, E36, E37, E45, E53, E76, E107, DOCET, JAG8810 | frameshift_variant | EMB1873 protein |
| Solyc12g044645 | InDel | 60,656,260 | E42 | frameshift_variant | AP2/B3 transcription factor family protein |
| Solyc12g044645 | SNP | 60,656,273 | E42 | stop_lost, splice_region_variant |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olivieri, F.; Calafiore, R.; Francesca, S.; Schettini, C.; Chiaiese, P.; Rigano, M.M.; Barone, A. High-Throughput Genotyping of Resilient Tomato Landraces to Detect Candidate Genes Involved in the Response to High Temperatures. Genes 2020, 11, 626. https://doi.org/10.3390/genes11060626
Olivieri F, Calafiore R, Francesca S, Schettini C, Chiaiese P, Rigano MM, Barone A. High-Throughput Genotyping of Resilient Tomato Landraces to Detect Candidate Genes Involved in the Response to High Temperatures. Genes. 2020; 11(6):626. https://doi.org/10.3390/genes11060626
Chicago/Turabian StyleOlivieri, Fabrizio, Roberta Calafiore, Silvana Francesca, Carlo Schettini, Pasquale Chiaiese, Maria Manuela Rigano, and Amalia Barone. 2020. "High-Throughput Genotyping of Resilient Tomato Landraces to Detect Candidate Genes Involved in the Response to High Temperatures" Genes 11, no. 6: 626. https://doi.org/10.3390/genes11060626
APA StyleOlivieri, F., Calafiore, R., Francesca, S., Schettini, C., Chiaiese, P., Rigano, M. M., & Barone, A. (2020). High-Throughput Genotyping of Resilient Tomato Landraces to Detect Candidate Genes Involved in the Response to High Temperatures. Genes, 11(6), 626. https://doi.org/10.3390/genes11060626






