Genomic Instability in Fungal Plant Pathogens
Abstract
1. Introduction to Fungal Plant Pathogens
1.1. Fungi—A Threat to Agriculture and the Environment
1.2. Genetic Recombination in Fungal Plant Pathogens
2. Chromosome Plasticity—Implications in Plant Pathology
2.1. Aneuploidy and Copy-Number Variations (CNVs)
2.2. Structural Variations
2.3. Dynamics of Accessory Chromosomes in Fungal Plant Pathogens
3. Comparative Genomics Generate Hypotheses Regarding Causes of Genome Plasticity in Fungal Plant Pathogens
3.1. Genomic Analyses of the DNA Damage Response in Fungal Plant Pathogens
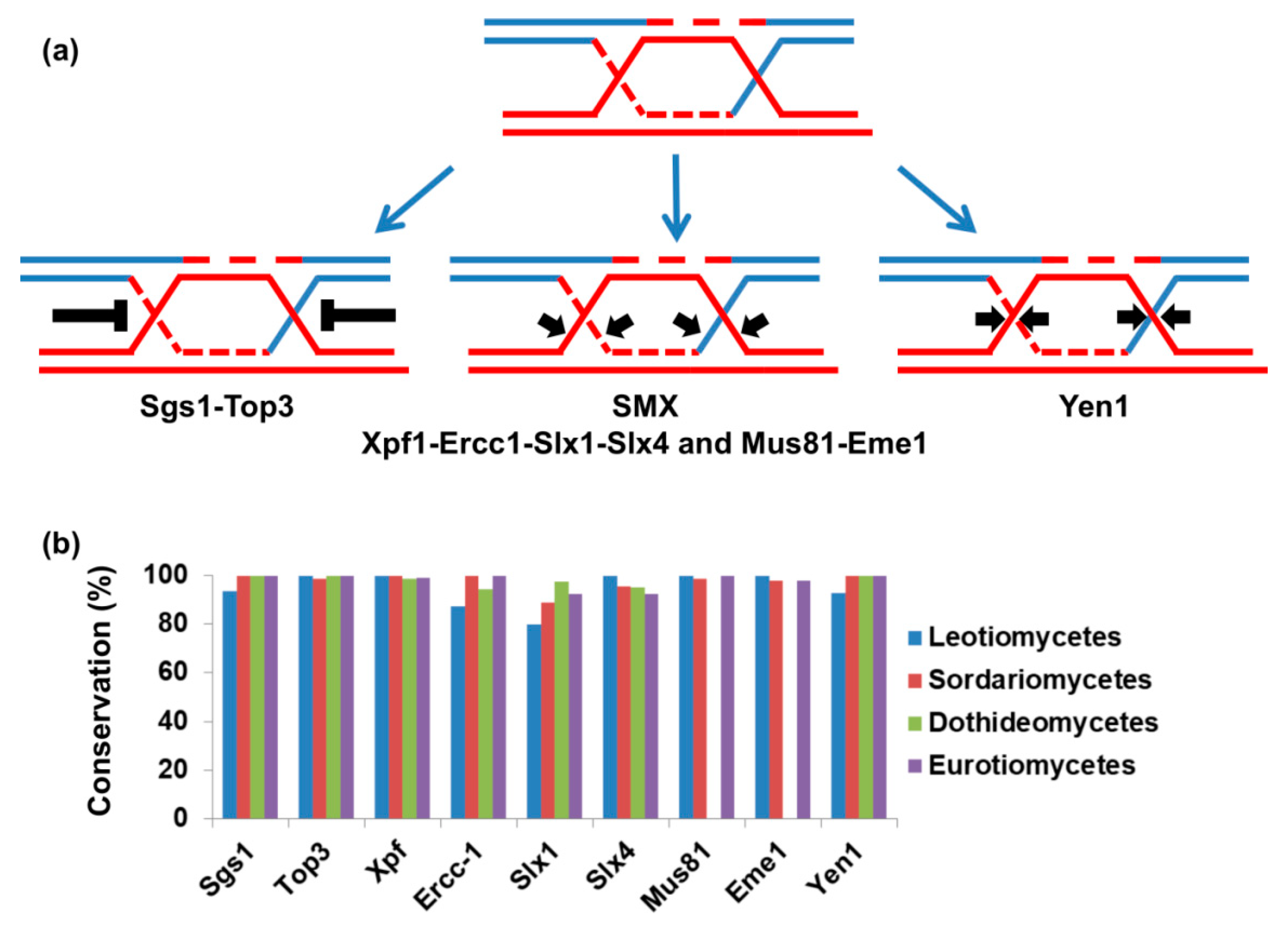
3.2. Resolution of Holliday Junctions
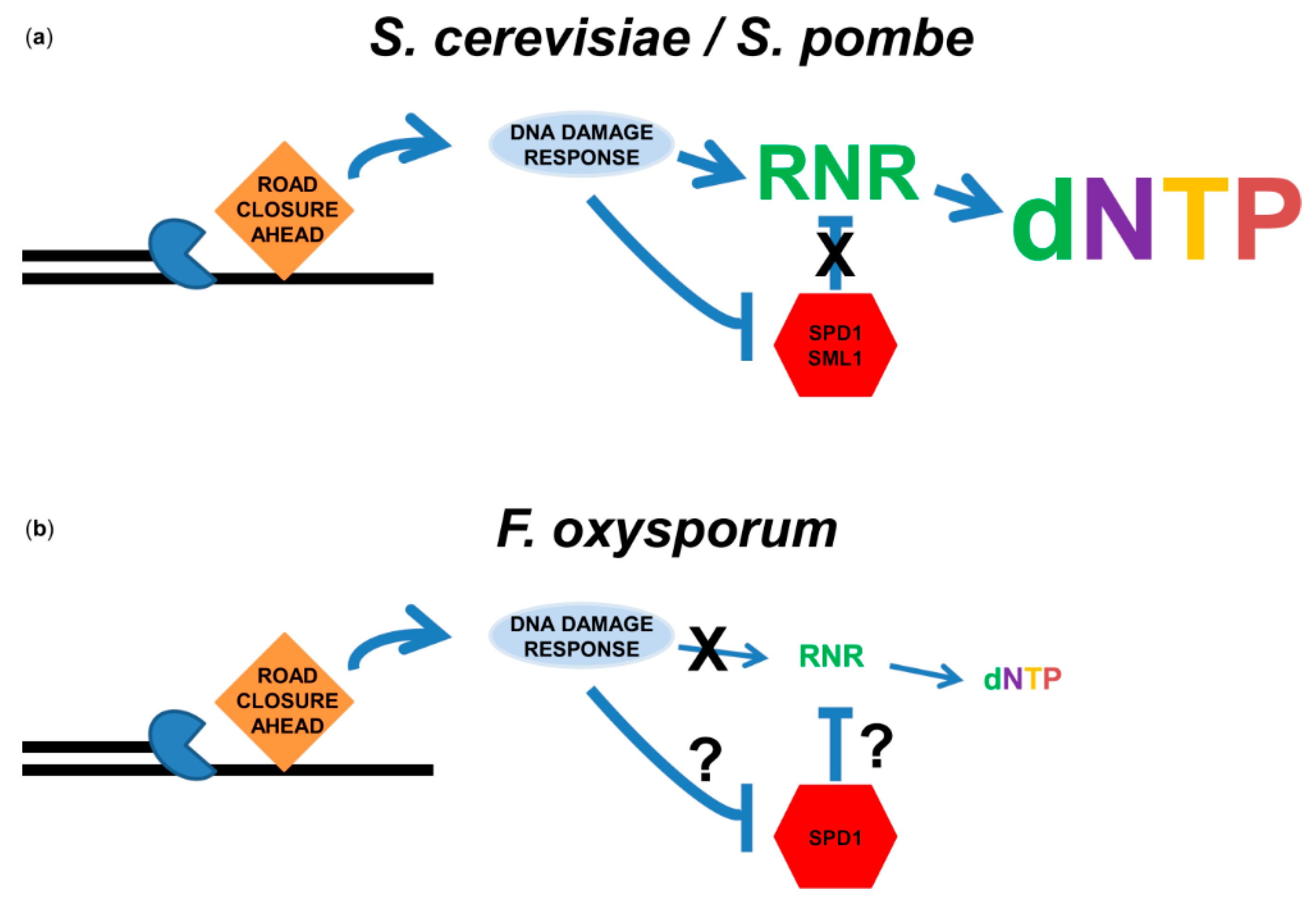
3.3. Deoxyribonucleotide Triphosphate (dNTP) Biosynthesis in Response to DNA Replication Stress
4. Conclusions
Funding
Conflicts of Interest
References
- Ainsworth, G.C. Introduction to the History of Plant Pathology; Cambridge University Press: Cambridge, UK, 1981. [Google Scholar]
- Nevo, D. Some diseases of agricultural crops and their control in the land of Israel during biblical, mishnaic and talmudic times. Phytoparasitica 1995, 23, 7–17. [Google Scholar] [CrossRef]
- Fortenbaugh, W. Theophrastus: His Psychological, Doxographical, and Scientific Writings; Taylor & Francis: Abingdon, UK, 2018. [Google Scholar]
- Ploetz, R.C. Fusarium wilt of banana. Phytopathology 2015, 105, 1512–1521. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.C.; Henk, D.A.; Briggs, C.J.; Brownstein, J.S.; Madoff, L.C.; McCraw, S.L.; Gurr, S.J. Emerging fungal threats to animal, plant and ecosystem health. Nature 2012, 484, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.T.; Croll, D.; Gladieux, P.; Soanes, D.M.; Persoons, A.; Bhattacharjee, P.; Hossain, M.S.; Gupta, D.R.; Rahman, M.M.; Mahboob, M.G.; et al. Emergence of wheat blast in Bangladesh was caused by a South American lineage of Magnaporthe oryzae. BMC Biol 2016, 14, 84. [Google Scholar] [CrossRef]
- Markets and Markets. Fungicides Market by Type (Chemical and Biological), Crop Type (Cereals & Grains, Oilseeds & Pulses, Fruits & Vegetables), Mode of Action (Contact & Systemic), Mode of Application, Form, and Region-Global Forecast to 2025; Markets and Markets: Pune, India, 2019. [Google Scholar]
- Nicolopoulou-Stamati, P.; Maipas, S.; Kotampasi, C.; Stamatis, P.; Hens, L. Chemical Pesticides and human health: The urgent need for a new concept in agriculture. Front.Public Health 2016, 4, 148. [Google Scholar] [CrossRef]
- Seeland, A.; Oehlmann, J.; Müller, R. Aquatic ecotoxicity of the fungicide pyrimethanil: Effect profile under optimal and thermal stress conditions. Environ. Pollut. 2012, 168, 161–169. [Google Scholar] [CrossRef]
- Korolev, N.; Mamiev, M.; Zahavi, T.; Elad, Y. Screening of Botrytis cinerea isolates from vineyards in Israel for resistance to fungicides. Eur. J. Plant Pathol. 2011, 129, 591–608. [Google Scholar] [CrossRef]
- Verweij, P.E.; Snelders, E.; Kema, G.H.J.; Mellado, E.; Melchers, W.J.G. Azole resistance in Aspergillus fumigatus: A side-effect of environmental fungicide use? Lancet Infect. Dis. 2009, 9, 789–795. [Google Scholar] [CrossRef]
- Garcia-Bastidas, F.; Ordonez, N.; Konkol, J.; Al-Qasim, M.; Naser, Z.; Abdelwali, M.; Salem, N.; Waalwijk, C.; Ploetz, R.C.; Kema, G.H.J. First Report of Fusarium oxysporum f. sp. cubense Tropical Race 4 associated with Panama disease of banana outside Southeast Asia. Plant Dis. 2014, 98, 694. [Google Scholar] [CrossRef]
- Dita, M.; Barquero, M.; Heck, D.; Mizubuti, E.S.G.; Staver, C.P. Fusarium Wilt of banana: Current knowledge on epidemiology and research needs toward sustainable disease management. Front Plant Sci. 2018, 9, 1468. [Google Scholar] [CrossRef]
- Singh, R.P.; Hodson, D.P.; Huerta-Espino, J.; Jin, Y.; Bhavani, S.; Njau, P.; Herrera-Foessel, S.; Singh, P.K.; Singh, S.; Govindan, V. The emergence of Ug99 races of the stem rust fungus is a threat to world wheat production. Annu. Rev. Phytopathol. 2011, 49, 465–481. [Google Scholar] [CrossRef] [PubMed]
- Drenth, A.; McTaggart, A.R.; Wingfield, B.D. Fungal clones win the battle, but recombination wins the war. IMA Fungus 2019, 10, 18. [Google Scholar] [CrossRef]
- McDonald, B.A.; Mundt, C.C. How knowledge of pathogen population biology informs management of Septoria Tritici blotch. Phytopathology 2016, 106, 948–955. [Google Scholar] [CrossRef]
- Stukenbrock, E.H. The Role of hybridization in the evolution and emergence of new fungal plant pathogens. Phytopathology 2016, 106, 104–112. [Google Scholar] [CrossRef]
- Lucas, J.A.; Hawkins, N.J.; Fraaije, B.A. The evolution of fungicide resistance. Adv. Appl. Microbiol. 2015, 90, 29–92. [Google Scholar] [CrossRef] [PubMed]
- Zhan, J.; Mundt, C.C.; McDonald, B.A. Sexual reproduction facilitates the adaptation of parasites to antagonistic host environments: Evidence from empirical study in the wheat-Mycosphaerella graminicola system. Int. J. Parasitol. 2007, 37, 861–870. [Google Scholar] [CrossRef] [PubMed]
- Wittenberg, A.H.; van der Lee, T.A.; Ben M’barek, S.; Ware, S.B.; Goodwin, S.B.; Kilian, A.; Visser, R.G.; Kema, G.H.; Schouten, H.J. Meiosis drives extraordinary genome plasticity in the haploid fungal plant pathogen Mycosphaerella Graminicola. PLoS ONE 2009, 4, e5863. [Google Scholar] [CrossRef]
- Goodwin, S.B.; Ben M’Barek, S.; Dhillon, B.; Wittenberg, A.H.J.; Crane, C.F.; Hane, J.K.; Foster, A.J.; Van der Lee, T.A.J.; Grimwood, J.; Aerts, A.; et al. Finished genome of the fungal wheat pathogen Mycosphaerella graminicola reveals dispensome structure, chromosome plasticity, and stealth pathogenesis. PLoS Genet. 2011, 7, e1002070. [Google Scholar] [CrossRef]
- Milgroom, M.G.; Sotirovski, K.; Risteski, M.; Brewer, M.T. Heterokaryons and parasexual recombinants of Cryphonectria parasitica in two clonal populations in southeastern Europe. Fungal Genet. Biol. 2009, 46, 849–854. [Google Scholar] [CrossRef]
- McGuire, I.C.; Davis, J.E.; Double, M.L.; MacDonald, W.L.; Rauscher, J.T.; McCawley, S.; Milgroom, M.G. Heterokaryon formation and parasexual recombination between vegetatively incompatible lineages in a population of the chestnut blight fungus, Cryphonectria parasitica. Mol. Ecol. 2005, 14, 3657–3669. [Google Scholar] [CrossRef]
- Ishikawa, F.H.; Souza, E.A.; Shoji, J.Y.; Connolly, L.; Freitag, M.; Read, N.D.; Roca, M.G. Heterokaryon incompatibility is suppressed following conidial anastomosis tube fusion in a fungal plant pathogen. PLoS ONE 2012, 7, e31175. [Google Scholar] [CrossRef] [PubMed]
- Varga, J.; Szigeti, G.; Baranyi, N.; Kocsubé, S.; O’Gorman, C.M.; Dyer, P.S. Aspergillus: Sex and recombination. Mycopathologia 2014, 178, 349–362. [Google Scholar] [CrossRef] [PubMed]
- Teunissen, H.A.; Verkooijen, J.; Cornelissen, B.J.; Haring, M.A. Genetic exchange of avirulence determinants and extensive karyotype rearrangements in parasexual recombinants of Fusarium oxysporum. Mol. Genet. Genom. 2002, 268, 298–310. [Google Scholar] [CrossRef] [PubMed]
- Berlin, A.; Samils, B.; Andersson, B. Multiple genotypes within aecial clusters in Puccinia graminis and Puccinia coronata: Improved understanding of the biology of cereal rust fungi. Fungal Biol. Biotechnol. 2017, 4, 3. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Upadhyaya, N.M.; Sperschneider, J.; Matny, O.; Nguyen-Phuc, H.; Mago, R.; Raley, C.; Miller, M.E.; Silverstein, K.A.T.; Henningsen, E.; et al. Emergence of the Ug99 lineage of the wheat stem rust pathogen through somatic hybridisation. Nat. Commun. 2019, 10, 5068. [Google Scholar] [CrossRef] [PubMed]
- Dunham, M.J.; Badrane, H.; Ferea, T.; Adams, J.; Brown, P.O.; Rosenzweig, F.; Botstein, D. Characteristic genome rearrangements in experimental evolution of Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 2002, 99, 16144–16149. [Google Scholar] [CrossRef]
- Gu, W.; Zhang, F.; Lupski, J.R. Mechanisms for human genomic rearrangements. Pathogenetics 2008, 1, 4. [Google Scholar] [CrossRef]
- Selmecki, A.; Forche, A.; Berman, J. Aneuploidy and isochromosome formation in drug-resistant. Candida Albicans Sci. 2006, 313, 367–370. [Google Scholar] [CrossRef]
- Ni, M.; Feretzaki, M.; Li, W.; Floyd-Averette, A.; Mieczkowski, P.; Dietrich, F.S.; Heitman, J. Unisexual and heterosexual meiotic reproduction generate aneuploidy and phenotypic diversity de novo in the yeast Cryptococcus neoformans. PLoS Biol. 2013, 11, e1001653. [Google Scholar] [CrossRef]
- Bennett, R.J.; Forche, A.; Berman, J. Rapid mechanisms for generating genome diversity: Whole ploidy shifts, aneuploidy, and loss of heterozygosity. Cold Spring Harb. Perspect. Med. 2014, 4. [Google Scholar] [CrossRef]
- Morrow, C.A.; Fraser, J.A. Ploidy variation as an adaptive mechanism in human pathogenic fungi. Semin. Cell Dev. Biol. 2013, 24, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Mulla, W.; Zhu, J.; Li, R. Yeast: A simple model system to study complex phenomena of aneuploidy. FEMS Microbiol. Rev. 2014, 38, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Bebber, D.P.; Gurr, S.J. Crop-destroying fungal and oomycete pathogens challenge food security. Fungal Genet Biol. 2015, 74, 62–64. [Google Scholar] [CrossRef] [PubMed]
- Kasuga, T.; Bui, M.; Bernhardt, E.; Swiecki, T.; Aram, K.; Cano, L.M.; Webber, J.; Brasier, C.; Press, C.; Grunwald, N.J.; et al. Host-induced aneuploidy and phenotypic diversification in the Sudden Oak Death pathogen Phytophthora ramorum. Bmc Genom. 2016, 17, 385 . [Google Scholar] [CrossRef] [PubMed]
- Van der Lee, T.; Testa, A.; Robold, A.; van ’t Klooster, J.; Govers, F. High-density genetic linkage maps of Phytophthora infestans reveal trisomic progeny and chromosomal rearrangements. Genetics 2004, 167, 1643–1661. [Google Scholar] [CrossRef]
- Elliott, M.; Yuzon, J.; C, M.M.; Tripathy, S.; Bui, M.; Chastagner, G.A.; Coats, K.; Rizzo, D.M.; Garbelotto, M.; Kasuga, T. Characterization of phenotypic variation and genome aberrations observed among Phytophthora ramorum isolates from diverse hosts. BMC Genom. 2018, 19, 320. [Google Scholar] [CrossRef]
- Bradshaw, R.E.; Sim, A.D.; Chettri, P.; Dupont, P.Y.; Guo, Y.; Hunziker, L.; McDougal, R.L.; Van der Nest, A.; Fourie, A.; Wheeler, D.; et al. Global population genomics of the forest pathogen Dothistroma septosporum reveal chromosome duplications in high dothistromin-producing strains. Mol. Plant Pathol. 2019, 20, 784–799. [Google Scholar] [CrossRef]
- Shain, L.; Franich, R.A. Induction of Dothistroma blight symptoms with dothistromin. Physiol. Plant Pathol. 1981, 19, 49–55. [Google Scholar] [CrossRef]
- Ferguson, L.R.; Parslow, M.I.; McLarin, J.A. Chromosome damage by dothistromin in human peripheral blood lymphocyte cultures: A comparison with aflatoxin B1. Mutat. Res.Genet. Toxicol. 1986, 170, 47–53. [Google Scholar] [CrossRef]
- Franich, R.A.; Carson, M.J.; Carson, S.D. Synthesis and accumulation of benzoic acid in Pinus radiata needles in response to tissue injury by dothistromin, and correlation with resistance of P. radiata families to Dothistroma pini. Physiol. Mol. Plant Pathol. 1986, 28, 267–286. [Google Scholar] [CrossRef]
- Jones, L.; Riaz, S.; Morales-Cruz, A.; Amrine, K.C.; McGuire, B.; Gubler, W.D.; Walker, M.A.; Cantu, D. Adaptive genomic structural variation in the grape powdery mildew pathogen, Erysiphe necator. BMC Genom. 2014, 15, 1081. [Google Scholar] [CrossRef]
- Hulvey, J.; Popko, J.T., Jr.; Sang, H.; Berg, A.; Jung, G. Overexpression of ShCYP51B and ShatrD in Sclerotinia homoeocarpa isolates exhibiting practical field resistance to a demethylation inhibitor fungicide. Appl. Environ. Microbiol. 2012, 78, 6674–6682. [Google Scholar] [CrossRef]
- Anderson, J.B.; Sirjusingh, C.; Parsons, A.B.; Boone, C.; Wickens, C.; Cowen, L.E.; Kohn, L.M. Mode of selection and experimental evolution of antifungal drug resistance in Saccharomyces cerevisiae. Genetics 2003, 163, 1287–1298. [Google Scholar]
- De Jonge, R.; Bolton, M.D.; Kombrink, A.; Van Den Berg, G.C.M.; Yadeta, K.A.; Thomma, B.P.H.J. Extensive chromosomal reshuffling drives evolution of virulence in an asexual pathogen. Genome Res. 2013, 23, 1271–1282. [Google Scholar] [CrossRef]
- Moller, M.; Habig, M.; Freitag, M.; Stukenbrock, E.H. Extraordinary genome instability and widespread chromosome rearrangements during vegetative growth. Genetics 2018, 210, 517–529. [Google Scholar] [CrossRef]
- Wang, Q.; Sun, M.; Zhang, Y.; Song, Z.; Zhang, S.; Zhang, Q.; Xu, J.-R.; Liu, H. Extensive chromosomal rearrangements and rapid evolution of novel effector superfamilies contribute to host adaptation and speciation in the basal ascomycetous fungi. Mol. Plant Pathol. 2020. [Google Scholar] [CrossRef]
- Argueso, J.L.; Westmoreland, J.; Mieczkowski, P.A.; Gawel, M.; Petes, T.D.; Resnick, M.A. Double-strand breaks associated with repetitive DNA can reshape the genome. Proc. Natl. Acad. Sci. USA 2008, 105, 11845–11850. [Google Scholar] [CrossRef]
- Scheifele, L.Z.; Cost, G.J.; Zupancic, M.L.; Caputo, E.M.; Boeke, J.D. Retrotransposon overdose and genome integrity. Proc. Natl. Acad. Sci. USA 2009, 106, 13927–13932. [Google Scholar] [CrossRef]
- Haber, J.E. Transpositions and translocations induced by site-specific double-strand breaks in budding yeast. DNA Repair (Amst.) 2006, 5, 998–1009. [Google Scholar] [CrossRef]
- Ma, L.J.; van der Does, H.C.; Borkovich, K.A.; Coleman, J.J.; Daboussi, M.J.; Di Pietro, A.; Dufresne, M.; Freitag, M.; Grabherr, M.; Henrissat, B.; et al. Comparative genomics reveals mobile pathogenicity chromosomes in Fusarium. Nature 2010, 464, 367–373. [Google Scholar] [CrossRef]
- Covert, S.F. Supernumerary chromosomes in filamentous fungi. Curr. Genet. 1998, 33, 311–319. [Google Scholar] [CrossRef]
- Han, Y.N.; Liu, X.G.; Benny, U.; Kistler, H.C.; VanEtten, H.D. Genes determining pathogenicity to pea are clustered on a supernumerary chromosome in the fungal plant pathogen Nectria haematococca. Plant J. 2001, 25, 305–314. [Google Scholar] [CrossRef]
- Akagi, Y.; Akamatsu, H.; Otani, H.; Kodama, M. Horizontal chromosome transfer, a mechanism for the evolution and differentiation of a plant-pathogenic fungus. Eukaryot. Cell 2009, 8, 1732–1738. [Google Scholar] [CrossRef]
- Dong, S.; Raffaele, S.; Kamoun, S. The two-speed genomes of filamentous pathogens: Waltz with plants. Curr. Opin. Genet. Dev. 2015, 35, 57–65. [Google Scholar] [CrossRef]
- Lievens, B.; Houterman, P.M.; Rep, M. Effector gene screening allows unambiguous identification of Fusarium oxysporum f. sp. lycopersici races and discrimination from other formae speciales. FEMS Microbiol. Lett. 2009, 300, 201–215. [Google Scholar] [CrossRef]
- Vlaardingerbroek, I.; Beerens, B.; Schmidt, S.M.; Cornelissen, B.J.; Rep, M. Dispensable chromosomes in Fusarium oxysporum f. sp. lycopersici. Mol. Plant Pathol. 2016, 17, 1455–1466. [Google Scholar] [CrossRef]
- Vlaardingerbroek, I.; Beerens, B.; Rose, L.; Fokkens, L.; Cornelissen, B.J.; Rep, M. Exchange of core chromosomes and horizontal transfer of lineage-specific chromosomes in Fusarium oxysporum. Environ. Microbiol. 2016, 18, 3702–3713. [Google Scholar] [CrossRef]
- Van Dam, P.; Fokkens, L.; Ayukawa, Y.; van der Gragt, M.; ter Horst, A.; Brankovics, B.; Houterman, P.M.; Arie, T.; Rep, M. A mobile pathogenicity chromosome in Fusarium oxysporum for infection of multiple cucurbit species. Sci. Rep. 2017, 7, 9042. [Google Scholar] [CrossRef]
- He, C.Z.; Rusu, A.G.; Poplawski, A.M.; Irwin, J.A.G.; Manners, J.M. Transfer of a supernumerary chromosome between vegetatively incompatible biotypes of the fungus Colletotrichum Gloeosporioides. Genet 1998, 150, 1459–1466. [Google Scholar]
- Mehrabi, R.; Mirzadi Gohari, A.; Kema, G.H.J. Karyotype variability in plant-pathogenic fungi. Annual. Rev. Phytopathol. 2017, 55, 483–503. [Google Scholar] [CrossRef]
- Fierro, F.; Martín, J.F. Molecular mechanisms of chromosomal rearrangement in fungi. Crit. Rev. Microbiol. 1999, 25, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Zeman, M.K.; Cimprich, K.A. Causes and consequences of replication stress. Nat. Cell. Biol. 2014, 16, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Helchowski, C.M.; Canman, C.E. The roles of DNA polymerase zeta and the Y family DNA polymerases in promoting or preventing genome instability. Mutat. Res. 2013, 743–744, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Ciccia, A.; Elledge, S.J. The DNA damage response: Making it safe to play with knives. Mol. Cell. 2010, 40, 179–204. [Google Scholar] [CrossRef]
- Serero, A.; Jubin, C.; Loeillet, S.; Legoix-Ne, P.; Nicolas, A.G. Mutational landscape of yeast mutator strains. Proc. Natl. Acad. Sci. USA 2014, 111, 1897–1902. [Google Scholar] [CrossRef]
- Wu, Y.; Suhasini, A.N.; Brosh, R.M., Jr. Welcome the family of FANCJ-like helicases to the block of genome stability maintenance proteins. Cell. Mol. Life Sci. 2009, 66, 1209–1222. [Google Scholar] [CrossRef]
- Degtyareva, N.P.; Chen, L.; Mieczkowski, P.; Petes, T.D.; Doetsch, P.W. Chronic oxidative DNA damage due to DNA repair defects causes chromosomal instability in Saccharomyces cerevisiae. Mol. Cell. Biol. 2008, 28, 5432–5445. [Google Scholar] [CrossRef]
- LeClerc, J.E.; Li, B.; Payne, W.L.; Cebula, T.A. High mutation frequencies among Escherichia coli and Salmonella pathogens. Science 1996, 274, 1208–1211. [Google Scholar] [CrossRef]
- Normark, B.H.; Normark, S. Evolution and spread of antibiotic resistance. J. Intern. Med. 2002, 252, 91–106. [Google Scholar] [CrossRef]
- Steenwyk, J.L.; Opulente, D.A.; Kominek, J.; Shen, X.X.; Zhou, X.; Labella, A.L.; Bradley, N.P.; Eichman, B.F.; Cadez, N.; Libkind, D.; et al. Extensive loss of cell-cycle and DNA repair genes in an ancient lineage of bipolar budding yeasts. PLoS Biol. 2019, 17, e3000255. [Google Scholar] [CrossRef]
- Milo, S.; Misgav, R.H.; Hazkani-Covo, E.; Covo, S. Limited DNA repair gene repertoire in Ascomycete yeast revealed by comparative genomics. Genome Biol. Evol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Gusa, A.; Jinks-Robertson, S. Mitotic recombination and adaptive genomic changes in human pathogenic fungi. Genes (Basel) 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Haber, J.E.; Ira, G.; Malkova, A.; Sugawara, N. Repairing a double-strand chromosome break by homologous recombination: Revisiting Robin Holliday’s model. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2004, 359, 79–86. [Google Scholar] [CrossRef]
- Mehta, A.; Haber, J.E. Sources of DNA double-strand breaks and models of recombinational DNA repair. Cold Spring Harb. Perspect. Biol. 2014, 6, a016428. [Google Scholar] [CrossRef]
- Cox, M.M.; Goodman, M.F.; Kreuzer, K.N.; Sherratt, D.J.; Sandler, S.J.; Marians, K.J. The importance of repairing stalled replication forks. Nature 2000, 404, 37–41. [Google Scholar] [CrossRef]
- Lambert, S.; Mizuno, K.; Blaisonneau, J.; Martineau, S.; Chanet, R.; Freon, K.; Murray, J.M.; Carr, A.M.; Baldacci, G. Homologous recombination restarts blocked replication forks at the expense of genome rearrangements by template exchange. Mol. Cell 2010, 39, 346–359. [Google Scholar] [CrossRef]
- Ahn, J.S.; Osman, F.; Whitby, M.C. Replication fork blockage by RTS1 at an ectopic site promotes recombination in fission yeast. EMBO J. 2005, 24, 2011–2023. [Google Scholar] [CrossRef]
- Iraqui, I.; Chekkal, Y.; Jmari, N.; Pietrobon, V.; Freon, K.; Costes, A.; Lambert, S.A. Recovery of arrested replication forks by homologous recombination is error-prone. PLoS Genet. 2012, 8, e1002976. [Google Scholar] [CrossRef]
- Fabre, F.; Chan, A.; Heyer, W.D.; Gangloff, S. Alternate pathways involving Sgs1/Top3, Mus81/Mms4, and Srs2 prevent formation of toxic recombination intermediates from single-stranded gaps created by DNA replication. Proc. Natl. Acad. Sci. USA 2002, 99, 16887–16892. [Google Scholar] [CrossRef]
- Magner, D.B.; Blankschien, M.D.; Lee, J.A.; Pennington, J.M.; Lupski, J.R.; Rosenberg, S.M. RecQ promotes toxic recombination in cells lacking recombination intermediate-removal proteins. Mol. Cell 2007, 26, 273–286. [Google Scholar] [CrossRef]
- West, S.C.; Chan, Y.W. Genome instability as a consequence of defects in the resolution of recombination intermediates. Cold Spring Harb. Symp. Quant. Biol. 2017, 82, 207–212. [Google Scholar] [CrossRef]
- El Ghamrasni, S.; Cardoso, R.; Halaby, M.J.; Zeegers, D.; Harding, S.; Kumareswaran, R.; Yavorska, T.; Chami, N.; Jurisicova, A.; Sanchez, O.; et al. Cooperation of Blm and Mus81 in development, fertility, genomic integrity and cancer suppression. Oncogene 2015, 34, 1780–1789. [Google Scholar] [CrossRef]
- Pardo, B.; Aguilera, A. Complex chromosomal rearrangements mediated by break-induced replication involve structure-selective endonucleases. PLoS Genet. 2012, 8, e1002979. [Google Scholar] [CrossRef]
- Wyatt, H.D.; West, S.C. Holliday junction resolvases. Cold Spring Harb. Perspect. Biol. 2014, 6, a023192. [Google Scholar] [CrossRef]
- Bizard, A.H.; Hickson, I.D. The dissolution of double Holliday junctions. Cold Spring Harb. Perspect. Biol. 2014, 6, a016477. [Google Scholar] [CrossRef]
- Wyatt, H.D.; Laister, R.C.; Martin, S.R.; Arrowsmith, C.H.; West, S.C. The SMX DNA repair tri-nuclease. Mol. Cell 2017, 65, 848–860.e11. [Google Scholar] [CrossRef]
- Chan, Y.W.; Fugger, K.; West, S.C. Unresolved recombination intermediates lead to ultra-fine anaphase bridges, chromosome breaks and aberrations. Nat. Cell Biol. 2018, 20, 92–103. [Google Scholar] [CrossRef]
- Ip, S.C.Y.; Rass, U.; Blanco, M.G.; Flynn, H.R.; Skehel, J.M.; West, S.C. Identification of Holliday junction resolvases from humans and yeast. Nature 2008, 456, 357–361. [Google Scholar] [CrossRef]
- Andersen, S.L.; Kuo, H.K.; Savukoski, D.; Brodsky, M.H.; Sekelsky, J. Three structure-selective endonucleases are essential in the absence of BLM helicase in Drosophila. PLoS Genet. 2011, 7, e1002315. [Google Scholar] [CrossRef]
- Munoz-Galvan, S.; Tous, C.; Blanco, M.G.; Schwartz, E.K.; Ehmsen, K.T.; West, S.C.; Heyer, W.D.; Aguilera, A. Distinct roles of Mus81, Yen1, Slx1-Slx4, and Rad1 nucleases in the repair of replication-born double-strand breaks by sister chromatid exchange. Mol. Cell. Biol. 2012, 32, 1592–1603. [Google Scholar] [CrossRef]
- Hanway, D.; Chin, J.K.; Xia, G.; Oshiro, G.; Winzeler, E.A.; Romesberg, F.E. Previously uncharacterized genes in the UV- and MMS-induced DNA damage response in yeast. Proc. Natl. Acad. Sci. USA 2002, 99, 10605–10610. [Google Scholar] [CrossRef] [PubMed]
- Boehm, E.W.A.; Ploetz, R.C.; Kistler, H.C. Statistical-analysis of electrophoretic karyotype variation among vegettative compatability groups of Fusarium oxysporum f.sp cubense. Mol. Plant-Microbe Interact. 1994, 7, 196–207. [Google Scholar] [CrossRef]
- Shahi, S.; Beerens, B.; Bosch, M.; Linmans, J.; Rep, M. Nuclear dynamics and genetic rearrangement in heterokaryotic colonies of Fusarium oxysporum. Fungal Genet. Biol. 2016, 91, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Rouse, J.; Jackson, S.P. Interfaces between the detection, signaling, and repair of DNA damage. Science 2002, 297, 547–551. [Google Scholar] [CrossRef]
- Milo-Cochavi, S.; Pareek, M.; Delulio, G.; Almog, Y.; Anand, G.; Ma, L.J.; Covo, S. The response to the DNA damaging agent methyl methanesulfonate in a fungal plant pathogen. Fungal Biol. 2019, 123, 408–422. [Google Scholar] [CrossRef]
- Elledge, S.J.; Zhou, Z.; Allen, J.B. Ribonucleotide reductase: Regulation, regulation, regulation. Trends Biochem. Sci. 1992, 17, 119–123. [Google Scholar] [CrossRef]
- Xu, H.; Faber, C.; Uchiki, T.; Fairman, J.W.; Racca, J.; Dealwis, C. Structures of eukaryotic ribonucleotide reductase I provide insights into dNTP regulation. Proc. Natl. Acad. Sci. USA 2006, 103, 4022–4027. [Google Scholar] [CrossRef]
- Guarino, E.; Salguero, I.; Kearsey, S.E. Cellular regulation of ribonucleotide reductase in eukaryotes. Semin. Cell Dev. Biol. 2014, 30, 97–103. [Google Scholar] [CrossRef]
- Elledge, S.J.; Davis, R.W. Two genes differentially regulated in the cell cycle and by DNA-damaging agents encode alternative regulatory subunits of ribonucleotide reductase. Genes Dev. 1990, 4, 740–751. [Google Scholar] [CrossRef]
- Tanaka, H.; Arakawa, H.; Yamaguchi, T.; Shiraishi, K.; Fukuda, S.; Matsui, K.; Takei, Y.; Nakamura, Y. A ribonucleotide reductase gene involved in a p53-dependent cell-cycle checkpoint for DNA damage. Nature 2000, 404, 42–49. [Google Scholar] [CrossRef]
- Jayachandran, G.; Fallon, A.M. The mosquito ribonucleotide reductase R2 gene: Ultraviolet light induces expression of a novel R2 variant with an internal amino acid deletion. Insect. Mol. Biol. 2004, 13, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Liu, Z. Arabidopsis ribonucleotide reductases are critical for cell cycle progression, DNA damage repair, and plant development. Plant Cell 2006, 18, 350–365. [Google Scholar] [CrossRef] [PubMed]
- Gon, S.; Napolitano, R.; Rocha, W.; Coulon, S.; Fuchs, R.P. Increase in dNTP pool size during the DNA damage response plays a key role in spontaneous and induced-mutagenesis in Escherichia coli. Proc. Natl. Acad. Sci. USA 2011, 108, 19311–19316. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Muller, E.G.; Rothstein, R. A suppressor of two essential checkpoint genes identifies a novel protein that negatively affects dNTP pools. Mol. Cell 1998, 2, 329–340. [Google Scholar] [CrossRef]
- Hakansson, P.; Dahl, L.; Chilkova, O.; Domkin, V.; Thelander, L. The Schizosaccharomyces pombe replication inhibitor Spd1 regulates ribonucleotide reductase activity and dNTPs by binding to the large Cdc22 subunit. J. Biol. Chem. 2006, 281, 1778–1783. [Google Scholar] [CrossRef]
- Zhao, X.; Chabes, A.; Domkin, V.; Thelander, L.; Rothstein, R. The ribonucleotide reductase inhibitor Sml1 is a new target of the Mec1/Rad53 kinase cascade during growth and in response to DNA damage. EMBO J. 2001, 20, 3544–3553. [Google Scholar] [CrossRef]
- Moss, J.; Tinline-Purvis, H.; Walker, C.A.; Folkes, L.K.; Stratford, M.R.; Hayles, J.; Hoe, K.L.; Kim, D.U.; Park, H.O.; Kearsey, S.E.; et al. Break-induced ATR and Ddb1-Cul4(Cdt)(2) ubiquitin ligase-dependent nucleotide synthesis promotes homologous recombination repair in fission yeast. Genes Dev. 2010, 24, 2705–2716. [Google Scholar] [CrossRef]
- Chabes, A.; Georgieva, B.; Domkin, V.; Zhao, X.; Rothstein, R.; Thelander, L. Survival of DNA damage in yeast directly depends on increased dNTP levels allowed by relaxed feedback inhibition of ribonucleotide reductase. Cell 2003, 112, 391–401. [Google Scholar] [CrossRef]
- Cohen, R.; Milo, S.; Sharma, S.; Savidor, A.; Covo, S. Ribonucleotide reductase from Fusarium oxysporum does not respond to DNA replication stress. DNA Repair (Amst.) 2019, 83, 102674. [Google Scholar] [CrossRef]
- Fasullo, M.; Tsaponina, O.; Sun, M.; Chabes, A. Elevated dNTP levels suppress hyper-recombination in Saccharomyces cerevisiae S-phase checkpoint mutants. Nucleic Acids Res. 2010, 38, 1195–1203. [Google Scholar] [CrossRef]
- Bester, A.C.; Roniger, M.; Oren, Y.S.; Im, M.M.; Sarni, D.; Chaoat, M.; Bensimon, A.; Zamir, G.; Shewach, D.S.; Kerem, B. Nucleotide deficiency promotes genomic instability in early stages of cancer development. Cell 2011, 145, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Galhardo, R.S.; Hastings, P.J.; Rosenberg, S.M. Mutation as a stress response and the regulation of evolvability. Crit. Rev. Biochem. Mol. Biol. 2007, 42, 399–435. [Google Scholar] [CrossRef] [PubMed]
- Ragheb, M.N.; Thomason, M.K.; Hsu, C.; Nugent, P.; Gage, J.; Samadpour, A.N.; Kariisa, A.; Merrikh, C.N.; Miller, S.I.; Sherman, D.R.; et al. Inhibiting the evolution of antibiotic resistance. Mol. Cell 2019, 73, 157–165.e5. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Covo, S. Genomic Instability in Fungal Plant Pathogens. Genes 2020, 11, 421. https://doi.org/10.3390/genes11040421
Covo S. Genomic Instability in Fungal Plant Pathogens. Genes. 2020; 11(4):421. https://doi.org/10.3390/genes11040421
Chicago/Turabian StyleCovo, Shay. 2020. "Genomic Instability in Fungal Plant Pathogens" Genes 11, no. 4: 421. https://doi.org/10.3390/genes11040421
APA StyleCovo, S. (2020). Genomic Instability in Fungal Plant Pathogens. Genes, 11(4), 421. https://doi.org/10.3390/genes11040421



