Genome-Wide Identification and Characterization of Xyloglucan Endotransglycosylase/Hydrolase in Ananas comosus during Development
Abstract
1. Introduction
2. Materials and Methods
2.1. Dataset Compilation and Identification of the XTH Gene Family
2.2. Multiple Sequence Alignment and Identification of Motifs
2.3. Phylogenetic Tree Analysis and Nomenclature of XTHs
2.4. Gene Structure Analysis
2.5. Chromosomal Distribution and Gene Duplication
2.6. Calculation of Ka/Ks
2.7. Transcriptome Analysis and Gene Expansion Patterns
2.8. Experimental Validation of XTH Transcript Levels by RT-qPCR Analysis
3. Results
3.1. Identification and Characteristics of XTHs
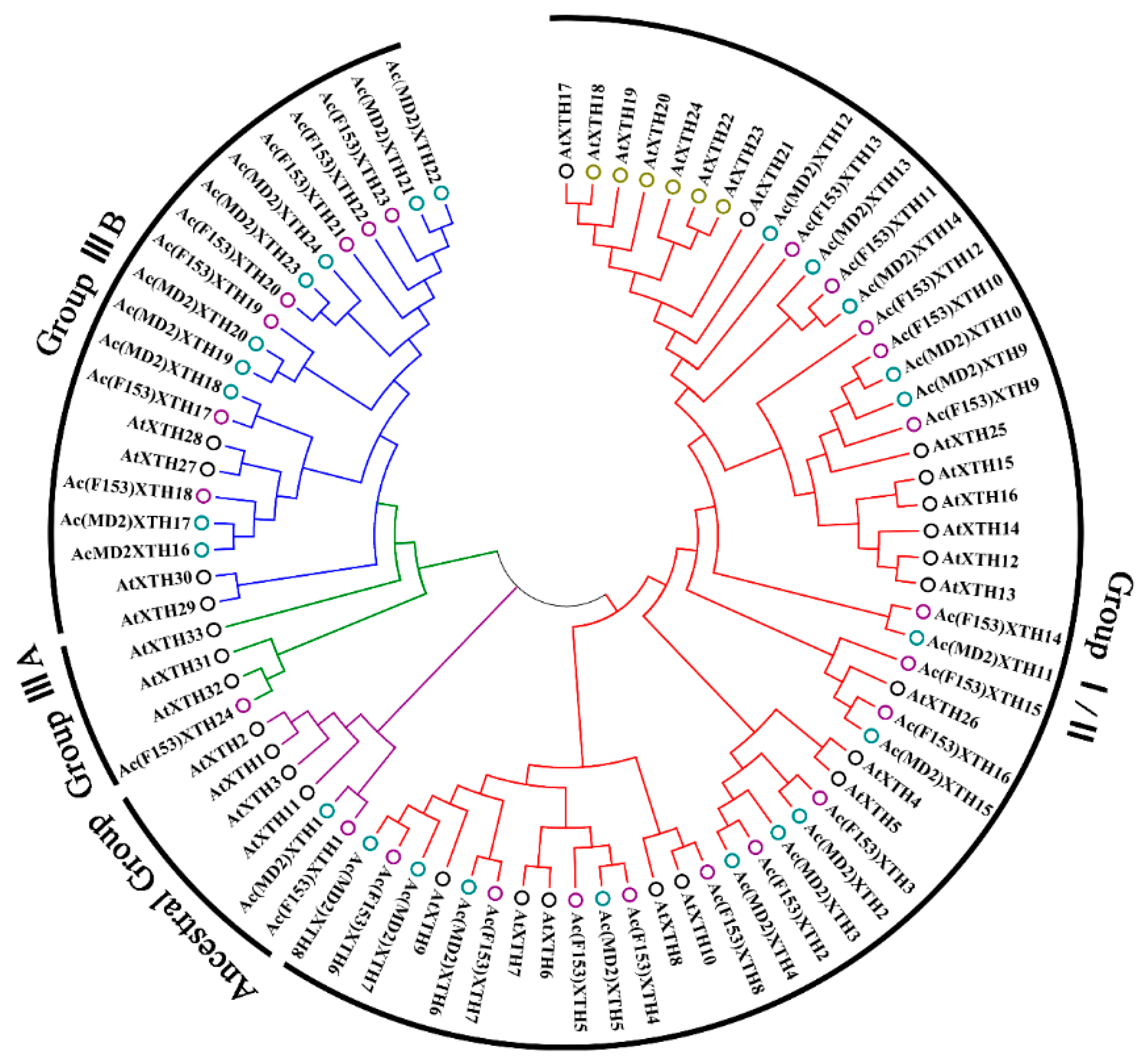
3.2. Phylogenetic Analysis and Classification of XTH Proteins
3.3. Sequence Alignment of XTHs
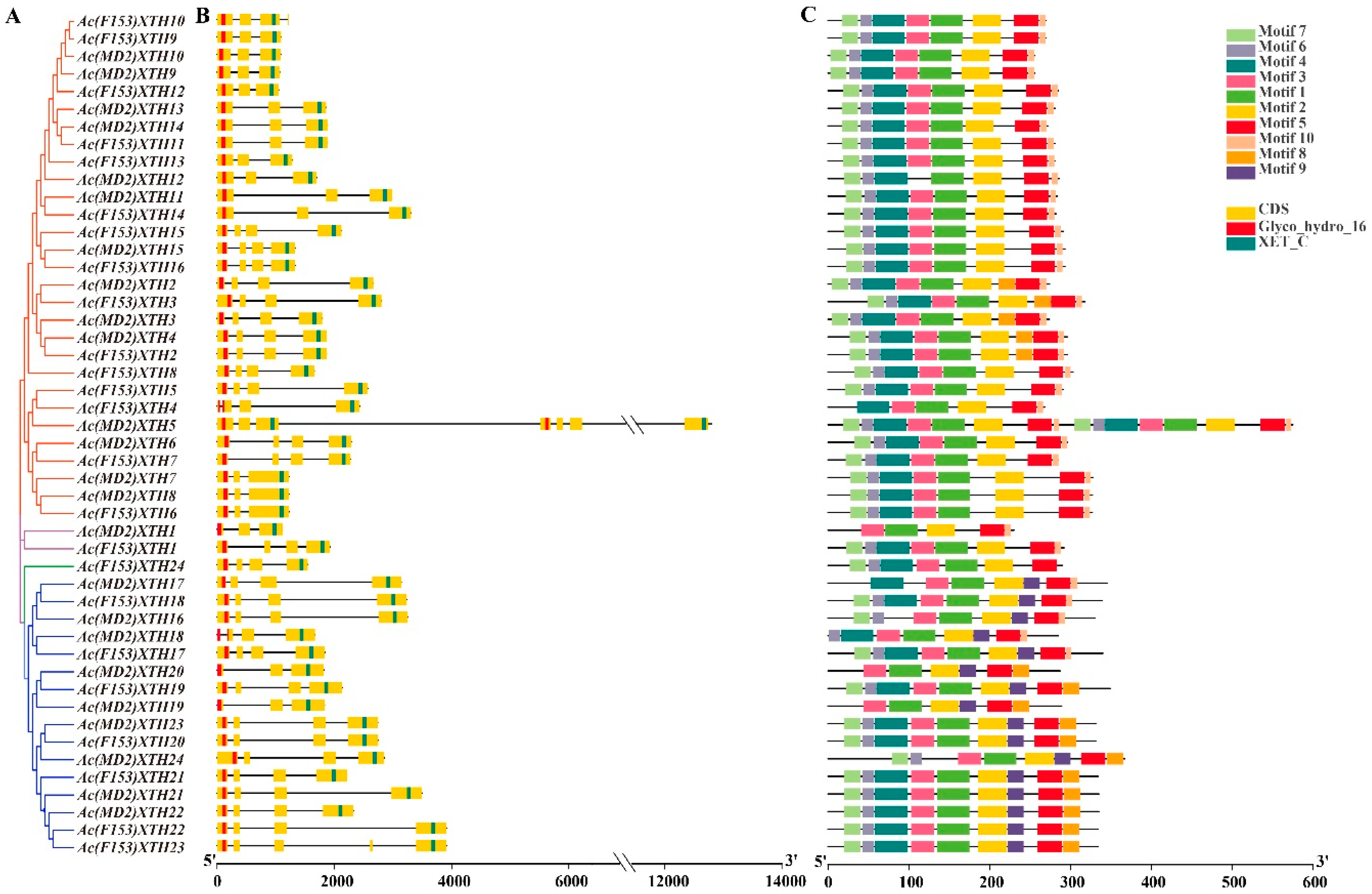
3.4. Gene Structure Analysis and the Pattern of the Motif in XTHs
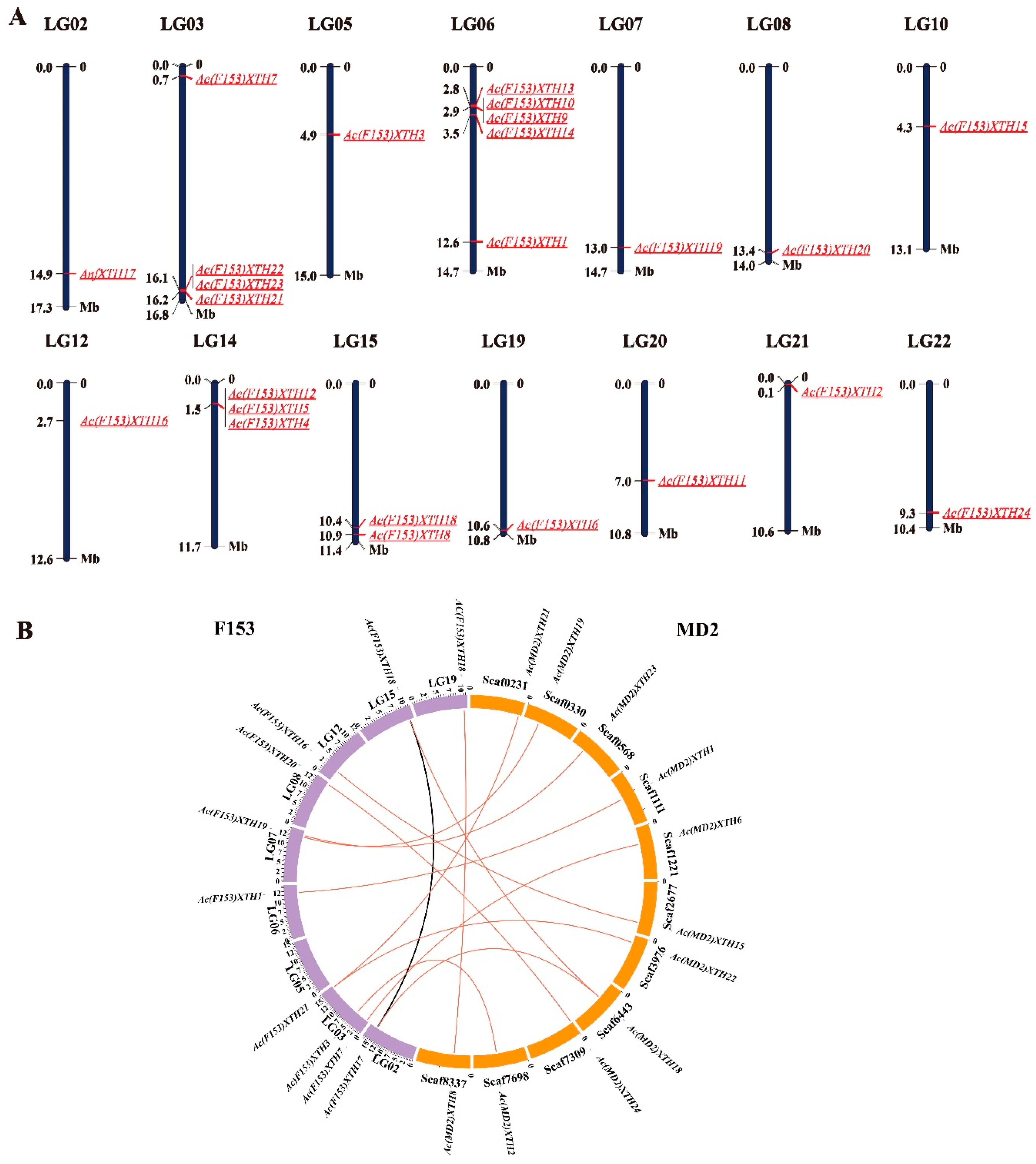
3.5. Chromosomal Distribution and Syntenic Analysis of XTH Genes
3.6. Ka/Ks Analysis of XTH Genes
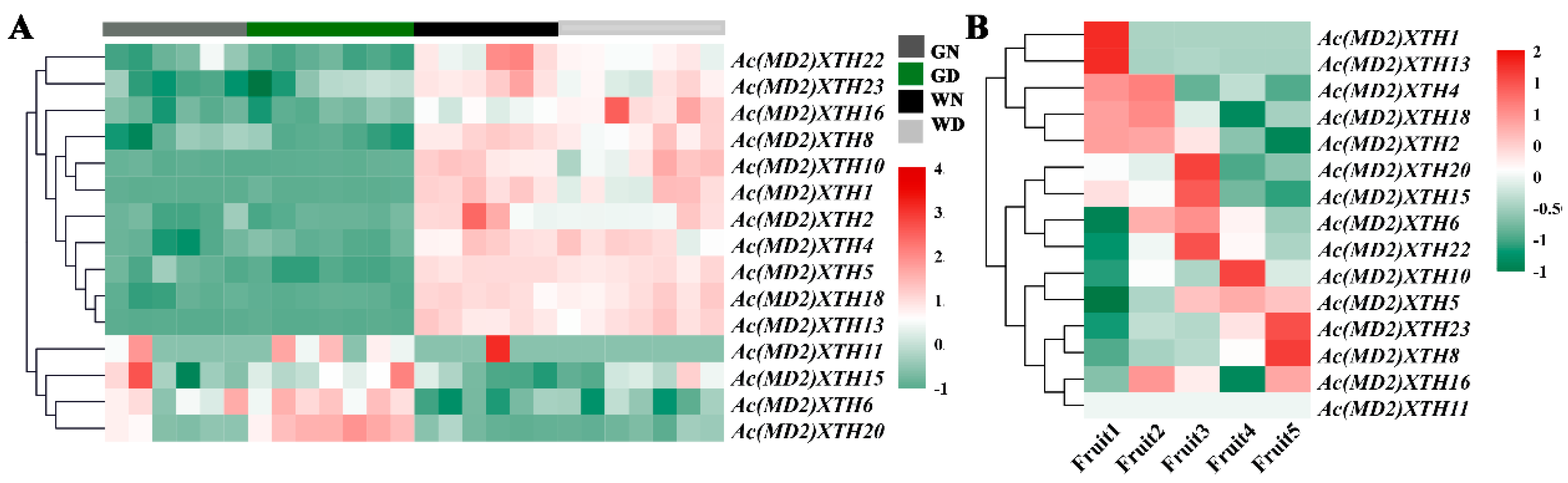
3.7. Differential Expression Profiles of XTHs during Development
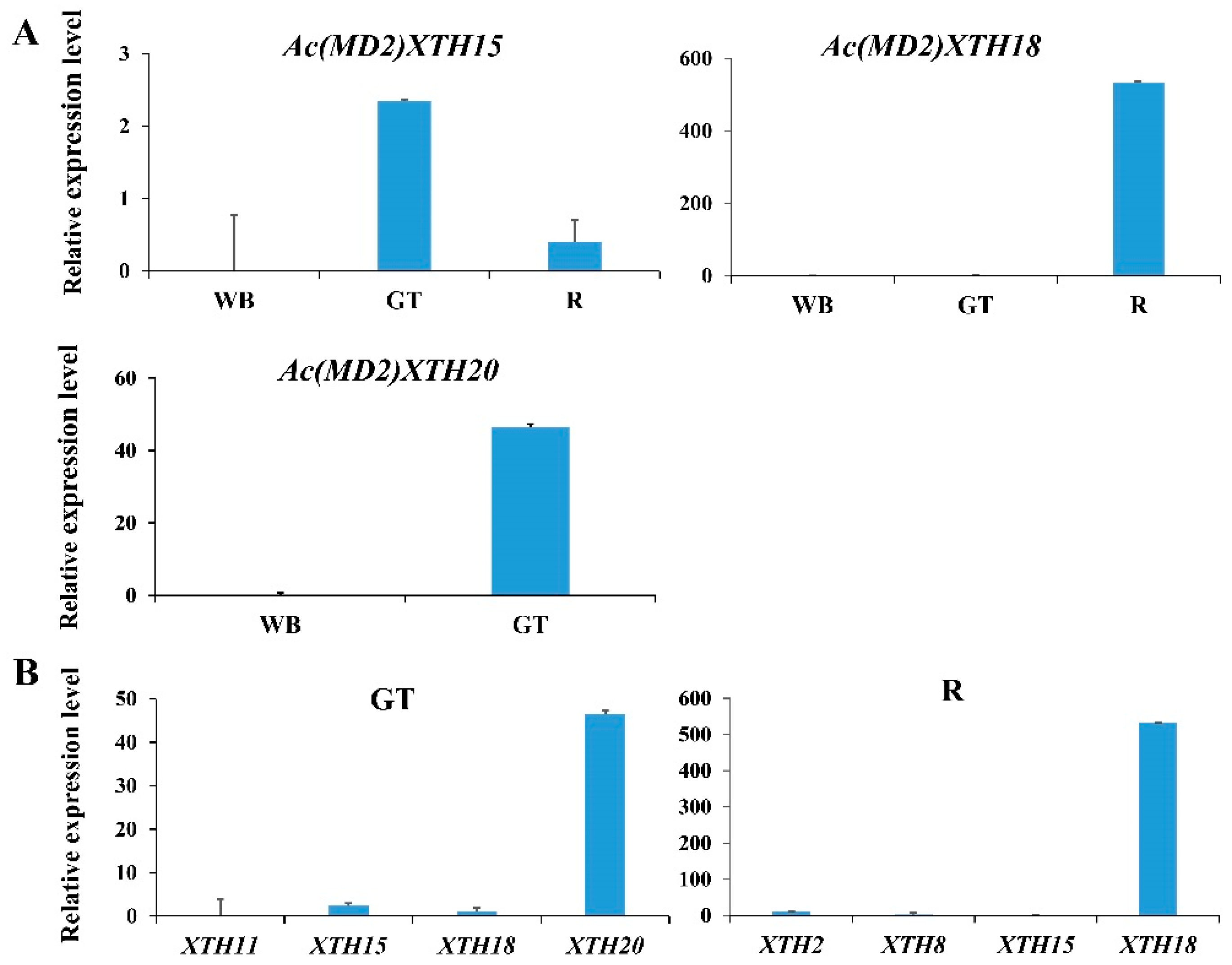
3.8. RT-qPCR Analysis of XTH Genes in Root and Leaf
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Detoni, A.M.; Carvalho, S.L.C.; Hoshino, A.T.; Pastorio, M.A.; Schmidt, M.A.H.; Menezes Junior, A.O.; Androcioli, H.G. First report of Chrysodeixis includens (Walker, [1858]) (Lepidotera: Noctuidae) Injurious to Pineapple (Ananas comosus L.) (Bromeliaceae) in Brazil. Braz. J. Biol. 2018, 78, 796–798. [Google Scholar] [CrossRef] [PubMed]
- Ming, R.; VanBuren, R.; Wai, C.M.; Tang, H.; Schatz, M.C.; Bowers, J.E.; Lyons, E.; Wang, M.L.; Chen, J.; Biggers, E.; et al. The pineapple genome and the evolution of CAM photosynthesis. Nat. Genet. 2015, 47, 1435–1442. [Google Scholar] [CrossRef] [PubMed]
- Žemlička, L.; Fodran, P.; Kolek, E.; Prónayová, N. Analysis of natural aroma and flavor of MD2 pineapple variety (Ananas comosus [L.] Merr.). Acta Chim. Slovaca 2013, 6, 123–128. [Google Scholar] [CrossRef]
- Iurlaro, A.; De Caroli, M.; Sabella, E.; De Pascali, M.; Rampino, P.; De Bellis, L.; Perrotta, C.; Dalessandro, G.; Piro, G.; Fry, S.C.; et al. Drought and Heat Differentially Affect XTH Expression and XET Activity and Action in 3-Day-Old Seedlings of Durum Wheat Cultivars with Different Stress Susceptibility. Front. Plant Sci. 2016, 7, 1686. [Google Scholar] [CrossRef] [PubMed]
- Miedes, E.; Lorences, E.P. Xyloglucan endotransglucosylase/hydrolases (XTHs) during tomato fruit growth and ripening. J. Plant Physiol. 2009, 166, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Eklof, J.M.; Brumer, H. The XTH gene family: An update on enzyme structure, function, and phylogeny in xyloglucan remodeling. Plant Physiol. 2010, 153, 456–466. [Google Scholar] [CrossRef] [PubMed]
- Fry, S.C.; Smith, R.C.; Renwick, K.F.; Martin, D.J.; Hodge, S.K.; Matthews, K.J. Xyloglucan endotransglycosylase, a new wall-loosening enzyme activity from plants. Biochem. J. 1992, 282, 821–828. [Google Scholar] [CrossRef] [PubMed]
- Strohmeier, M.; Hrmova, M.; Fischer, M.; Harvey, A.J.; Fincher, G.B.; Pleiss, J. Molecular modeling of family GH16 glycoside hydrolases: Potential roles for xyloglucan transglucosylases/hydrolases in cell wall modification in the poaceae. Protein Sci. 2004, 13, 3200–3213. [Google Scholar] [CrossRef] [PubMed]
- Rose, J.K.; Braam, J.; Fry, S.C.; Nishitani, K. The XTH family of enzymes involved in xyloglucan endotransglucosylation and endohydrolysis: Current perspectives and a new unifying nomenclature. Plant Cell Physiol. 2002, 43, 1421–1435. [Google Scholar] [CrossRef]
- Baumann, M.J.; Eklof, J.M.; Michel, G.; Kallas, A.M.; Teeri, T.T.; Czjzek, M.; Brumer, H., 3rd. Structural evidence for the evolution of xyloglucanase activity from xyloglucan endo-transglycosylases: Biological implications for cell wall metabolism. Plant Cell 2007, 19, 1947–1963. [Google Scholar] [CrossRef]
- Michailidis, G.; Argiriou, A.; Darzentas, N.; Tsaftaris, A. Analysis of xyloglucan endotransglycosylase/hydrolase (XTH) genes from allotetraploid (Gossypium hirsutum) cotton and its diploid progenitors expressed during fiber elongation. J. Plant Physiol. 2009, 166, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Johansson, P.; Denman, S.; Brumer, H.; Kallas, A.M.; Henriksson, H.; Bergfors, T.; Teeri, T.T.; Jones, T.A. Crystallization and preliminary X-ray analysis of a xyloglucan endotransglycosylase from Populus tremula x tremuloides. Acta Crystallogr. D Biol. Crystallogr. 2003, 59, 535–537. [Google Scholar] [CrossRef] [PubMed]
- Campbell, P.; Braam, J. Xyloglucan endotransglycosylases: Diversity of genes, enzymes and potential wall-modifying functions. Trends Plant Sci. 1999, 4, 361–366. [Google Scholar] [CrossRef]
- Kaewthai, N.; Gendre, D.; Eklof, J.M.; Ibatullin, F.M.; Ezcurra, I.; Bhalerao, R.P.; Brumer, H. Group III-A XTH genes of Arabidopsis encode predominant xyloglucan endohydrolases that are dispensable for normal growth. Plant Physiol. 2013, 161, 440–454. [Google Scholar] [CrossRef] [PubMed]
- Behar, H.; Graham, S.W.; Brumer, H. Comprehensive cross-genome survey and phylogeny of glycoside hydrolase family 16 members reveals the evolutionary origin of EG16 and XTH proteins in plant lineages. Plant J. 2018, 95, 1114–1128. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, R.; Nishitani, K. A comprehensive expression analysis of all members of a gene family encoding cell-wall enzymes allowed us to predict cis-regulatory regions involved in cell-wall construction in specific organs of Arabidopsis. Plant Cell Physiol. 2001, 42, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, R.; Rose, J.K.; Nishitani, K. A surprising diversity and abundance of xyloglucan endotransglucosylase/hydrolases in rice. Classification and expression analysis. Plant Physiol. 2004, 134, 1088–1099. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Xu, Z.; Ding, A.; Kong, Y. Genome-Wide Identification and Expression Profiling Analysis of the Xyloglucan Endotransglucosylase/Hydrolase Gene Family in Tobacco (Nicotiana tabacum L.). Genes 2018, 9, 273. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Valliyodan, B.; Prince, S.; Wan, J.; Nguyen, H.T. Characterization of the XTH Gene Family: New Insight to the Roles in Soybean Flooding Tolerance. Int. J. Mol. Sci. 2018, 19, 2705. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.M.; Liu, C.; Wu, F. Genome-Wide Identification, Characterization and Expression Analysis of Xyloglucan Endotransglucosylase/Hydrolase Genes Family in Barley (Hordeum vulgare). Molecules 2019, 24, 1935. [Google Scholar] [CrossRef]
- Thompson, J.E.; Fry, S.C. Restructuring of wall-bound xyloglucan by transglycosylation in living plant cells. Plant J. 2001, 26, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Harada, T.; Torii, Y.; Morita, S.; Onodera, R.; Hara, Y.; Yokoyama, R.; Nishitani, K.; Satoh, S. Cloning, characterization, and expression of xyloglucan endotransglucosylase/hydrolase and expansin genes associated with petal growth and development during carnation flower opening. J. Exp. Bot. 2011, 62, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Vissenberg, K.; Van Sandt, V.; Fry, S.C.; Verbelen, J.P. Xyloglucan endotransglucosylase action is high in the root elongation zone and in the trichoblasts of all vascular plants from Selaginella to Zea mays. J. Exp. Bot. 2003, 54, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, R.G.; Johnston, S.L.; Yauk, Y.-K.; Sharma, N.N.; Schröder, R. Analysis of xyloglucan endotransglucosylase/hydrolase (XTH) gene families in kiwifruit and apple. Postharvest Biol. Technol. 2009, 51, 149–157. [Google Scholar] [CrossRef]
- Opazo, M.C.; Figueroa, C.R.; Henriquez, J.; Herrera, R.; Bruno, C.; Valenzuela, P.D.; Moya-Leon, M.A. Characterization of two divergent cDNAs encoding xyloglucan endotransglycosylase/hydrolase (XTH) expressed in Fragaria chiloensis fruit. Plant Sci. 2010, 179, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.K.; Kim, J.E.; Park, J.A.; Eom, T.J.; Kim, W.T. Constitutive expression of abiotic stress-inducible hot pepper CaXTH3, which encodes a xyloglucan endotransglucosylase/hydrolase homolog, improves drought and salt tolerance in transgenic Arabidopsis plants. FEBS Lett. 2006, 580, 3136–3144. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.F.; Shi, Y.Z.; Lei, G.J.; Fry, S.C.; Zhang, B.C.; Zhou, Y.H.; Braam, J.; Jiang, T.; Xu, X.Y.; Mao, C.Z.; et al. XTH31, encoding an in vitro XEH/XET-active enzyme, regulates aluminum sensitivity by modulating in vivo XET action, cell wall xyloglucan content, and aluminum binding capacity in Arabidopsis. Plant Cell 2012, 24, 4731–4747. [Google Scholar] [CrossRef] [PubMed]
- Xuan, Y.; Zhou, Z.S.; Li, H.B.; Yang, Z.M. Identification of a group of XTHs genes responding to heavy metal mercury, salinity and drought stresses in Medicago truncatula. Ecotoxicol. Environ. Saf. 2016, 132, 153–163. [Google Scholar] [CrossRef]
- Sayers, E.W.; Agarwala, R.; Bolton, E.E.; Brister, J.R.; Canese, K.; Clark, K.; Connor, R.; Fiorini, N.; Funk, K.; Hefferon, T.; et al. Database resources of the National Center for Biotechnology Information. Nucleic Acids Res. 2019, 47, D23–D28. [Google Scholar] [CrossRef]
- Lamesch, P.; Berardini, T.Z.; Li, D.; Swarbreck, D.; Wilks, C.; Sasidharan, R.; Muller, R.; Dreher, K.; Alexander, D.L.; Garcia-Hernandez, M.; et al. The Arabidopsis Information Resource (TAIR): Improved gene annotation and new tools. Nucleic Acids Res. 2012, 40, D1202–D1210. [Google Scholar] [CrossRef]
- Potter, S.C.; Luciani, A.; Eddy, S.R.; Park, Y.; Lopez, R.; Finn, R.D. HMMER web server: 2018 update. Nucleic Acids Res. 2018, 46, W200–W204. [Google Scholar] [CrossRef] [PubMed]
- El-Gebali, S.; Mistry, J.; Bateman, A.; Eddy, S.R.; Luciani, A.; Potter, S.C.; Qureshi, M.; Richardson, L.J.; Salazar, G.A.; Smart, A.; et al. The Pfam protein families database in 2019. Nucleic Acids Res. 2019, 47, D427–D432. [Google Scholar] [CrossRef] [PubMed]
- Marchler-Bauer, A.; Bryant, S.H. CD-Search: Protein domain annotations on the fly. Nucleic Acids Res. 2004, 32, W327–W331. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, M.R.; Gasteiger, E.; Bairoch, A.; Sanchez, J.C.; Williams, K.L.; Appel, R.D.; Hochstrasser, D.F. Protein identification and analysis tools in the ExPASy server. Methods Mol. Biol. 1999, 112, 531–552. [Google Scholar] [PubMed]
- Petersen, T.N.; Brunak, S.; von Heijne, G.; Nielsen, H. SignalP 4.0: Discriminating signal peptides from transmembrane regions. Nat. Methods 2011, 8, 785–786. [Google Scholar] [CrossRef] [PubMed]
- Chou, K.-C.; Shen, H.-B. Cell-PLoc 2.0: An improved package of web-servers for predicting subcellular localization of proteins in various organisms. Nat. Sci. 2010, 2, 1090–1103. [Google Scholar] [CrossRef]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef]
- Bailey, T.L.; Johnson, J.; Grant, C.E.; Noble, W.S. The MEME Suite. Nucleic Acids Res. 2015, 43, W39–W49. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Henrissat, B.; Teeri, T.T.; Warren, R.A.J. A scheme for designating enzymes that hydrolyse the polysaccharides in the cell walls of plants. FEBS Lett. 1998, 425, 352–354. [Google Scholar] [CrossRef]
- Hu, B.; Jin, J.; Guo, A.Y.; Zhang, H.; Luo, J.; Gao, G. GSDS 2.0: An upgraded gene feature visualization server. Bioinformatics 2015, 31, 1296–1297. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Xia, R.; Chen, H.; He, Y. TBtools, a Toolkit for Biologists integrating various biological data handling tools with a user-friendly interface. BioRxiv 2018, 289660. [Google Scholar]
- Voorrips, R.E. MapChart: Software for the graphical presentation of linkage maps and QTLs. J. Hered. 2002, 93, 77–78. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tang, H.; Debarry, J.D.; Tan, X.; Li, J.; Wang, X.; Lee, T.H.; Jin, H.; Marler, B.; Guo, H.; et al. MCScanX: A toolkit for detection and evolutionary analysis of gene synteny and collinearity. Nucleic Acids Res. 2012, 40, e49. [Google Scholar] [CrossRef] [PubMed]
- Krzywinski, M.; Schein, J.; Birol, I.; Connors, J.; Gascoyne, R.; Horsman, D.; Jones, S.J.; Marra, M.A. Circos: An information aesthetic for comparative genomics. Genome Res. 2009, 19, 1639–1645. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, Y.; Zhang, Z.; Zhu, J.; Yu, J. KaKs_Calculator 2.0: A toolkit incorporating γ-series methods and sliding window strategies. Genom. Proteom. Bioinform. 2010, 8, 77–80. [Google Scholar] [CrossRef]
- Xu, H.; Yu, Q.; Shi, Y.; Hua, X.; Tang, H.; Yang, L.; Ming, R.; Zhang, J. PGD: Pineapple Genomics Database. Hortic. Res. 2018, 5, 66. [Google Scholar] [CrossRef]
- McGregor, N.; Yin, V.; Tung, C.C.; Van Petegem, F.; Brumer, H. Crystallographic insight into the evolutionary origins of xyloglucan endotransglycosylases and endohydrolases. Plant J. 2017, 89, 651–670. [Google Scholar] [CrossRef]
- Kallas, A.M.; Piens, K.; Denman, S.E.; Henriksson, H.; Faldt, J.; Johansson, P.; Brumer, H.; Teeri, T.T. Enzymatic properties of native and deglycosylated hybrid aspen (Populus tremulaxtremuloides) xyloglucan endotransglycosylase 16A expressed in Pichia pastoris. Biochem. J. 2005, 390, 105–113. [Google Scholar] [CrossRef]
- Thiel, T.; Graner, A.; Waugh, R.; Grosse, I.; Close, T.J.; Stein, N. Evidence and evolutionary analysis of ancient whole-genome duplication in barley predating the divergence from rice. BMC Evol. Biol. 2009, 9, 209. [Google Scholar] [CrossRef]
- Vision, T.J.; Brown, D.G.; Tanksley, S.D. The origins of genomic duplications in Arabidopsis. Science 2000, 290, 2114–2117. [Google Scholar] [CrossRef] [PubMed]
- Mark, P.; Baumann, M.J.; Eklof, J.M.; Gullfot, F.; Michel, G.; Kallas, A.M.; Teeri, T.T.; Brumer, H.; Czjzek, M. Analysis of nasturtium TmNXG1 complexes by crystallography and molecular dynamics provides detailed insight into substrate recognition by family GH16 xyloglucan endo-transglycosylases and endo-hydrolases. Proteins 2009, 75, 820–836. [Google Scholar] [CrossRef] [PubMed]
- Weihofen, A.; Binns, K.; Lemberg, M.K.; Ashman, K.; Martoglio, B. Identification of signal peptide peptidase, a presenilin-type aspartic protease. Science 2002, 296, 2215–2218. [Google Scholar] [CrossRef]
- Saura-Valls, M.; Faure, R.; Ragas, S.; Piens, K.; Brumer, H.; Teeri, T.T.; Cottaz, S.; Driguez, H.; Planas, A. Kinetic analysis using low-molecular mass xyloglucan oligosaccharides defines the catalytic mechanism of a Populus xyloglucan endotransglycosylase. Biochem. J. 2006, 395, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Fanutti, C.; Gidley, M.J.; Reid, J.S. Action of a pure xyloglucan endo-transglycosylase (formerly called xyloglucan-specific endo-(1-4)-β-D-glucanase) from the cotyledons of germinated nasturtium seeds. Plant J. 1993, 3, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Tabuchi, A.; Mori, H.; Kamisaka, S.; Hoson, T. A new type of endo-xyloglucan transferase devoted to xyloglucan hydrolysis in the cell wall of azuki bean epicotyls. Plant Cell Physiol. 2001, 42, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Marburger, S.; Alexandrou, M.A.; Taggart, J.B.; Creer, S.; Carvalho, G.; Oliveira, C.; Taylor, M.I. Whole genome duplication and transposable element proliferation drive genome expansion in Corydoradinae catfishes. Proc. Biol. Sci. 2018, 285, 20172732. [Google Scholar] [CrossRef]
- Flavell, R.B.; O’Dell, M.; Hutchinson, J. Nucleotide sequence organization in plant chromosomes and evidence for sequence translocation during evolution. Cold Spring Harb. Symp. Quant. Biol. 1981, 45, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Bremer, K. Gondwanan evolution of the grass alliance of families (Poales). Evolution 2002, 56, 1374–1387. [Google Scholar] [CrossRef]
- Linder, H.; Rudall, P. Evolutionary History of Poales. Annu. Rev. Ecol. Evol. Syst. 2005, 36, 107–124. [Google Scholar] [CrossRef]
- Puranik, S.; Sahu, P.P.; Mandal, S.N.; B, V.S.; Parida, S.K.; Prasad, M. Comprehensive genome-wide survey, genomic constitution and expression profiling of the NAC transcription factor family in foxtail millet (Setaria italica L.). PLoS ONE 2013, 8, e64594. [Google Scholar] [CrossRef] [PubMed]
- Benderoth, M.; Textor, S.; Windsor, A.J.; Mitchell-Olds, T.; Gershenzon, J.; Kroymann, J. Positive selection driving diversification in plant secondary metabolism. Proc. Natl. Acad. Sci. USA 2006, 103, 9118–9123. [Google Scholar] [CrossRef] [PubMed]
- Rainha, N.; Medeiros, V.P.; Camara, M.; Faustino, H.; Leite, J.P.; Barreto Mdo, C.; Cruz, C.; Pacheco, C.A.; Ponte, D.; Bernardes da Silva, A. Plasticity of crassulacean acid metabolism at subtropical latitudes: A pineapple case study. Physiol. Plant. 2016, 156, 29–39. [Google Scholar] [CrossRef] [PubMed]
| Name | Transcript ID | Length | MW (kDa) | PI | SP | Catalytic Site | Subcellular Localization |
|---|---|---|---|---|---|---|---|
| Ac(F153)XTH1 | XP_020091206.1 | 292 | 32.78 | 5.35 | 20 | DELDFEFLG | Cell wall |
| Ac(F153)XTH2 | XP_020111283.1 | 296 | 34.17 | 5.98 | 20 | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(F153)XTH3 | XP_020087864.1 | 318 | 37.29 | 7.59 | − | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(F153)XTH4 | XP_020102739.1 | 268 | 30.71 | 5.96 | − | DELDFEFLG | Cell wall |
| Ac(F153)XTH5 | XP_020102738.1 | 291 | 33.33 | 6.06 | 24 | DELDFEFLG | Cell wall |
| Ac(F153)XTH6 | XP_020109096.1 | 327 | 35.98 | 5.07 | 24 | NEFDFEFLG | Cell wall |
| Ac(F153)XTH7 | XP_020084286.1 | 285 | 31.65 | 5.53 | 27 | DEVDFEFLG | Cell wall |
| Ac(F153)XTH8 | XP_020104828.1 | 303 | 35.62 | 8.81 | − | DEIDFEFLG | Cell wall |
| Ac(F153)XTH9 | XP_020089756.1 | 270 | 30.56 | 6.1 | 20 | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(F153)XTH10 | XP_020090359.1 | 270 | 30.63 | 6.43 | 20 | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(F153)XTH11 | XP_020110218.1 | 281 | 31.54 | 4.69 | 21 | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(F153)XTH12 | XP_020102740.1 | 285 | 31.58 | 4.75 | 24 | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(F153)XTH13 | XP_020091231.1 | 281 | 31.46 | 5.71 | 23 | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(F153)XTH14 | XP_020090869.1 | 282 | 32.15 | 5.41 | 25 | DEVDFEFLG | Cell wall |
| Ac(F153)XTH15 | XP_020097886.1 | 291 | 33.33 | 4.8 | 26 | DEIDYEFLG | Cell wall |
| Ac(F153)XTH16 | XP_020100605.1 | 293 | 32.89 | 9.04 | 20 | NEVDFEFLG | Cell wall |
| Ac(F153)XTH17 | XP_020106929.1 | 340 | 38.92 | 5.95 | 28 | DELDFEFLG | Cell wall |
| Ac(F153)XTH18 | XP_020104936.1 | 339 | 38.68 | 6.56 | 26 | DELDFEFLG | Cell wall |
| Ac(F153)XTH19 | XP_020092864.1 | 349 | 39.41 | 8.69 | 23 | DELDFEFLG | Cell wall |
| Ac(F153)XTH20 | XP_020094226.1 | 331 | 37.68 | 5.79 | 20 | DELDFEFLG | Cell wall |
| Ac(F153)XTH21 | XP_020085280.1 | 334 | 37.63 | 5.93 | 19 | DELDFEFLG | Cell wall |
| Ac(F153)XTH22 | XP_020085278.1 | 334 | 37.65 | 5.93 | 19 | DELDFEFLG | Cell wall |
| Ac(F153)XTH23 | XP_020085279.1 | 334 | 37.65 | 5.93 | 19 | DELDFEFLG | Cell wall |
| Ac(F153)XTH24 | XP_020112380.1 | 290 | 33.23 | 9.53 | 18 | DEVDIEFLG | Cell wall |
| Ac(MD2)XTH1 | OAY79161.1 | 230 | 25.99 | 5.36 | − | DELDFEFLG | Cell wall |
| Ac(MD2)XTH2 | OAY64709.1 | 274 | 32.15 | 7.63 | − | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(MD2)XTH3 | OAY72845.1 | 274 | 31.83 | 5.98 | − | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(MD2)XTH4 | OAY65283.1 | 296 | 34.17 | 5.98 | 20 | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(MD2)XTH5 | OAY76125.1 | 575 | 64.58 | 5.26 | 24 | DELDFEFLG | Cell wall Cytoplasm |
| Ac(MD2)XTH6 | OAY78767.1 | 296 | 32.87 | 5.7 | 38 | DEVDFEFLG | Cell wall |
| Ac(MD2)XTH7 | OAY70160.1 | 328 | 36.16 | 5 | 24 | NEFDFEFLG | Cell wall |
| Ac(MD2)XTH8 | OAY63484.1 | 327 | 35.98 | 5.07 | 24 | NEFDFEFLG | Cell wall |
| Ac(MD2)XTH9 | OAY62696.1 | 256 | 29.06 | 6.1 | − | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(MD2)XTH10 | OAY67076.1 | 256 | 29.06 | 6.1 | − | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(MD2)XTH11 | OAY62698.1 | 284 | 32.34 | 5.41 | 27 | DEVDFEFLG | Cell wall |
| Ac(MD2)XTH12 | OAY79036.1 | 286 | 32.28 | 5.97 | 25 | DEIDFEFLG | Cell wall |
| Ac(MD2)XTH13 | OAY76653.1 | 281 | 31.57 | 4.69 | 21 | DEIDFEFLG | Cell wall Cytoplasm |
| Ac(MD2)XTH14 | OAY71418.1 | 272 | 30.48 | 4.68 | 21 | DEIDFEFLG | Cell wall |
| Ac(MD2)XTH15 | OAY73488.1 | 293 | 32.89 | 9.04 | 20 | NEVDFEFLG | Cell wall |
| Ac(MD2)XTH16 | OAY70295.1 | 330 | 37.64 | 6.6 | 26 | DELDFEFLG | Cell wall |
| Ac(MD2)XTH17 | OAY81259.1 | 345 | 38.76 | 5.63 | − | DELDFEFLG | Cell wall |
| Ac(MD2)XTH18 | OAY66122.1 | 285 | 32.60 | 6.22 | − | DELDFEFLG | Cell wall |
| Ac(MD2)XTH19 | OAY83621.1 | 289 | 32.55 | 8.57 | − | DELDFEFLG | Cell wall |
| Ac(MD2)XTH20 | OAY70631.1 | 287 | 32.53 | 8.26 | − | DELDFEFLG | Cell wall |
| Ac(MD2)XTH21 | OAY84325.1 | 335 | 37.79 | 6.17 | 19 | DELDFEFLG | Cell wall |
| Ac(MD2)XTH22 | OAY70279.1 | 335 | 37.80 | 6.17 | 19 | DELDFEFLG | Cell wall |
| Ac(MD2)XTH23 | OAY81925.1 | 331 | 37.68 | 5.95 | 20 | DELDFEFLG | Cell wall |
| Ac(MD2)XTH24 | OAY65080.1 | 367 | 41.61 | 8.54 | − | DELDFEFLG | Cell wall |
| Collinear XTH Pairs | Ka | Ks | Ka/Ks | p-Value (Fisher) | Duplication Time (Mya) |
|---|---|---|---|---|---|
| Ac(F153)XTH7-Ac(MD2)XTH6 | 0.003077 | 0.03004 | 0.102442 | 0.003098 | 2.46 |
| Ac(F153)XTH21-Ac(MD2)XTH22 | 0.003969 | 0.024998 | 0.158756 | 0.008438 | 2.05 |
| Ac(F153)XTH21-Ac(MD2)XTH21 | 0.006628 | 0.029215 | 0.226877 | 0.011896 | 2.39 |
| Ac(F153)XTH3-Ac(MD2)XTH2 | 0.00155 | 0.011451 | 0.135322 | 0.107858 | 0.94 |
| Ac(F153)XTH1-Ac(MD2)XTH1 | 0.008336 | 0.010261 | 0.812454 | 0.377377 | 0.84 |
| Ac(F153)XTH19-Ac(MD2)XTH19 | 0.007705 | 0.019414 | 0.396873 | 0.150722 | 1.59 |
| Ac(F153)XTH7-Ac(MD2)XTH12 | 0.062047 | 0.071925 | 0.862666 | 0.630505 | 5.90 |
| Ac(F153)XTH20-Ac(MD2)XTH24 | 0.001324 | 0.008482 | 0.15611 | 0.130065 | 0.70 |
| Ac(F153)XTH16-Ac(MD2)XTH15 | NA | 0.005103 | 0 | NA | 0.41 |
| Ac(F153)XTH18-Ac(MD2)XTH18 | 0.265729 | 4.64275 | 0.057235 | 1.19E−40 | 380.55 |
| Ac(F153)XTH17-Ac(MD2)XTH18 | NA | NA | NA | NA | NA |
| Ac(F153)XTH7-Ac(F153)XTH18 | 0.311919 | 4.15239 | 0.075118 | 1.28E−41 | 340.36 |
| Ac(F153)XTH6-Ac(MD2)XTH18 | NA | NA | NA | NA | NA |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Q.; Li, H.; Yin, C.; Wang, X.; Jiang, Q.; Zhang, R.; Ge, F.; Chen, Y.; Yang, L. Genome-Wide Identification and Characterization of Xyloglucan Endotransglycosylase/Hydrolase in Ananas comosus during Development. Genes 2019, 10, 537. https://doi.org/10.3390/genes10070537
Li Q, Li H, Yin C, Wang X, Jiang Q, Zhang R, Ge F, Chen Y, Yang L. Genome-Wide Identification and Characterization of Xyloglucan Endotransglycosylase/Hydrolase in Ananas comosus during Development. Genes. 2019; 10(7):537. https://doi.org/10.3390/genes10070537
Chicago/Turabian StyleLi, Qingyun, Huayang Li, Chongyang Yin, Xiaotong Wang, Qing Jiang, Rui Zhang, Fangfang Ge, Yudong Chen, and Long Yang. 2019. "Genome-Wide Identification and Characterization of Xyloglucan Endotransglycosylase/Hydrolase in Ananas comosus during Development" Genes 10, no. 7: 537. https://doi.org/10.3390/genes10070537
APA StyleLi, Q., Li, H., Yin, C., Wang, X., Jiang, Q., Zhang, R., Ge, F., Chen, Y., & Yang, L. (2019). Genome-Wide Identification and Characterization of Xyloglucan Endotransglycosylase/Hydrolase in Ananas comosus during Development. Genes, 10(7), 537. https://doi.org/10.3390/genes10070537





