Development and Validation of a Gene-Targeted dCAPS Marker for Marker-Assisted Selection of Low-Alkaloid Content in Seeds of Narrow-Leafed Lupin (Lupinus angustifolius L.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material for Marker Development and Validation
- CO—wild and primitive populations originating from places of natural distribution—60 accessions;
- LR—landraces—two accessions;
- XD—breeding lines (created by man as an effect of selection after crossings or induced mutations (excluding cultivars))—57 accessions;
- CV—past and present registered cultivars—76 accessions;
- unknown classification—seven accessions.
2.2. Alkaloid Extraction and Analysis
2.3. DNA Isolation
2.4. Amplification and Sequencing
2.5. RNA Isolation and QA Gene Expression Analyses to Confirm Marker Results
3. Results
3.1. Overview of AP2/ERF Transcription Factor DNA Sequence and Development of a Novel, Diagnostic Marker
3.2. Variation of Seed QA Content among Representatives of NLL Collection, Incorporated in Marker Validation
3.3. iuc_RAP2-7 Marker Validation in NLL Collection
3.4. Assessment of DNA Sequence of Ambiguous Accessions
3.5. RAP2-7, LDC, LaAT, and LaCAO Gene Expression Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wink, M.; Meißner, C.; Witte, L. Patterns of quinolizidine alkaloids in 56 species of the genus Lupinus. Phytochemistry 1995, 38, 139–153. [Google Scholar] [CrossRef]
- Cowling, W.A.; Huyghe, C.; Święcicki, W. Lupin Breeding. In Lupins as Crop Plants: Biology, Production, and Utilization; Gladstones, J.S., Atkins, C.A., Hamblin, J., Eds.; CAB International: Wallingford, UK, 1998; pp. 93–120. [Google Scholar]
- Kamel, K.A.; Święcicki, W.; Kaczmarek, Z.; Barzyk, P. Quantitative and qualitative content of alkaloids in seeds of a narrow-leafed lupin (Lupinus angustifolius L.) collection. Genet. Resour. Crop. Evol. 2016, 63, 711–719. [Google Scholar] [CrossRef]
- Kroc, M.; Rybiński, W.; Wilczura, P.; Kamel, K.; Kaczmarek, Z.; Barzyk, P.; Święcicki, W. Quantitative and qualitative analysis of alkaloids composition in the seeds of a white lupin (Lupinus albus L.) collection. Genet. Resour. Crop. Evol. 2017, 64, 1853–1860. [Google Scholar] [CrossRef]
- Saito, K.; Koike, Y.; Suzuki, H.; Murakoshi, I. Biogenetic implication of lupin alkaloid biosynthesis in bitter and sweet forms of Lupinus luteus and L. albus. Phytochemistry 1993, 34, 1041–1044. [Google Scholar] [CrossRef]
- Suzuki, H.; Murakoshi, I.; Saito, K. A novel O-tigloyltransferase for alkaloid biosynthesis in plants. Purification, characterization, and distribution in Lupinus plants. J. Biol. Chem. 1994, 269, 15853–15860. [Google Scholar] [PubMed]
- Hirai, M.Y.; Suzuki, H.; Yamazaki, M.; Saito, K. Biochemical and partial molecular characterization of bitter and sweet forms of Lupinus angustifolius, an experimental model for study of molecular regulation of quinolizidine alkaloid biosynthesis. Chem. Pharm. Bull. 2000, 48, 1458–1461. [Google Scholar] [CrossRef][Green Version]
- Okada, T.; Hirai, M.Y.; Suzuki, H.; Yamazaki, M.; Saito, K. Molecular Characterization of a Novel Quinolizidine Alkaloid O-Tigloyltransferase: cDNA Cloning, Catalytic Activity of Recombinant Protein and Expression Analysis in Lupinus Plants. Plant. Cell Physiol. 2005, 46, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Bunsupa, S.; Okada, T.; Saito, K.; Yamazaki, M. An acyltransferase-like gene obtained by differential gene expression profiles of quinolizidine alkaloid-producing and nonproducing cultivars of Lupinus angustifolius. Plant. Biotechnol. 2011, 28, 89–94. [Google Scholar] [CrossRef]
- Bunsupa, S.; Katayama, K.; Ikeura, E.; Oikawa, A.; Toyooka, K.; Saito, K.; Yamazaki, M. Lysine decarboxylase catalyzes the first step of quinolizidine alkaloid biosynthesis and coevolved with alkaloid production in leguminosae. Plant. Cell 2012, 24, 1202–1216. [Google Scholar] [CrossRef]
- Yang, T.; Nagy, I.; Mancinotti, D.; Otterbach, S.L.; Andersen, T.B.; Motawia, M.S.; Asp, T.; Geu-Flores, F. Transcript profiling of a bitter variety of narrow-leafed lupin to discover alkaloid biosynthetic genes. J. Exp. Bot. 2017, 68, 5527–5537. [Google Scholar] [CrossRef]
- Frick, K.M.; Foley, R.C.; Kamphuis, L.G.; Siddique, K.H.M.; Garg, G.; Singh, K.B. Characterization of the genetic factors affecting quinolizidine alkaloid biosynthesis and its response to abiotic stress in narrow-leafed lupin (Lupinus angustifolius L.). Plant. Cell Environ. 2018, 23, 13172. [Google Scholar] [CrossRef] [PubMed]
- Kroc, M.; Koczyk, G.; Kamel, K.A.; Czepiel, K.; Fedorowicz-Strońska, O.; Krajewski, P.; Kosińska, J.; Podkowiński, J.; Wilczura, P.; Święcicki, W. Transcriptome-derived investigation of biosynthesis of quinolizidine alkaloids in narrow-leafed lupin (Lupinus angustifolius L.) highlights candidate genes linked to iucundus locus. Sci. Rep. 2019, 9, 2231. [Google Scholar] [CrossRef] [PubMed]
- Hondelmann, W. The lupin—ancient and modern crop plant. Theor. Appl. Genet. 1984, 68, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cowling, W.A.; Buirchell, B.J.; Tapia, M.E. Lupin. Lupinus L. Promoting the Conservation and Use of Underutilized and Neglected Crops.; International Board for Plant Genetic Resources (IBPGR): Rome, Italy, 1998; Volume 23, p. 105. [Google Scholar]
- Li, X.; Yang, H.; Buirchell, B.; Yan, G. Development of a DNA marker tightly linked to low-alkaloid gene iucundus in narrow-leafed lupin (Lupinus angustifolius L.) for marker-assisted selection. Crop. Pasture Sci. 2011, 62, 218–224. [Google Scholar] [CrossRef]
- Bohra, A.; Pandey, M.K.; Jha, U.C.; Singh, B.; Singh, I.P.; Datta, D.; Chaturvedi, S.K.; Nadarajan, N.; Varshney, R.K. Genomics-assisted breeding in four major pulse crops of developing countries: present status and prospects. Theor. Appl. Genet. 2014, 127, 1263–1291. [Google Scholar] [CrossRef] [PubMed]
- Kage, U.; Kumar, A.; Dhokane, D.; Karre, S.; Kushalappa, A.C. Functional molecular markers for crop improvement. Crit. Rev. Biotechnol. 2016, 36, 917–930. [Google Scholar] [CrossRef] [PubMed]
- Varshney, R.K.; Kudapa, H.; Pazhamala, L.; Chitikineni, A.; Thudi, M.; Bohra, A.; Gaur, P.M.; Janila, P.; Fikre, A.; Kimurto, P.; et al. Translational Genomics in Agriculture: Some Examples in Grain Legumes. Crit. Rev. Plant. Sci. 2015, 34, 169–194. [Google Scholar] [CrossRef]
- Andersen, J.R.; Lubberstedt, T. Functional markers in plants. Trends Plant. Sci. 2003, 8, 554–560. [Google Scholar] [CrossRef]
- Gupta, P.K.; Rustgi, S. Molecular markers from the transcribed/expressed region of the genome in higher plants. Funct. Integr. Genom. 2004, 4, 139–162. [Google Scholar] [CrossRef]
- Gupta, P.K.; Balyan, H.S.; Varshney, R.K.; Gill, K.S. Development and use of molecular markers for crop improvement. Plant. Breed. 2013, 132, 431–432. [Google Scholar] [CrossRef]
- Poczai, P.; Varga, I.; Laos, M.; Cseh, A.; Bell, N.; Valkonen, J.P.T.; Hyvönen, J. Advances in plant gene-targeted and functional markers: a review. Plant. Methods 2013, 9, 6. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.; Renshaw, D.; Luckett, D.; Clements, J.; Yan, G.; Adhikari, K.; Buirchell, B.; Sweetingham, M.; Yang, H. Development of a sequence-specific PCR marker linked to the gene “pauper” conferring low-alkaloids in white lupin (Lupinus albus L.) for marker assisted selection. Mol. Breed. 2009, 23, 153–161. [Google Scholar] [CrossRef]
- Hane, J.K.; Ming, Y.; Kamphuis, L.G.; Nelson, M.N.; Garg, G.; Atkins, C.A.; Bayer, P.E.; Bravo, A.; Bringans, S.; Cannon, S.; et al. A comprehensive draft genome sequence for lupin (Lupinus angustifolius), an emerging health food: insights into plant–microbe interactions and legume evolution. Plant. Biotechnol. J. 2017, 15, 318–330. [Google Scholar] [CrossRef] [PubMed]
- Tellmann, G. The E-Method: a highly accurate technique for gene-expression analysis. Nat. Methods 2006, 3, i–ii. [Google Scholar] [CrossRef]
- Neff, M.M.; Neff, J.D.; Chory, J.; Pepper, A.E. dCAPS, a simple technique for the genetic analysis of single nucleotide polymorphisms: experimental applications in Arabidopsis thaliana genetics. Plant J. 1998, 14, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Shirasawa, K.; Isobe, S.; Tabata, S.; Sato, S.; Hirakawa, H.; Ohyama, A.; Fukuoka, H.; Aoki, K.; Rothan, C. Genome-Wide SNP Genotyping to Infer the Effects on Gene Functions in Tomato. DNA Res. 2013, 20, 221–233. [Google Scholar] [CrossRef]
- Schaefer, C.; Rost, B. Predict impact of single amino acid change upon protein structure. BMC Genom. 2012, 13 (Suppl. 4), S4. [Google Scholar] [CrossRef]
- Shanthirabalan, S.; Chomilier, J.; Carpentier, M. Structural effects of point mutations in proteins. Proteins Struct. Funct. Bioinform. 2018, 86, 853–867. [Google Scholar] [CrossRef]
- Yang, H.; Jian, J.; Li, X.; Renshaw, D.; Clements, J.; Sweetingham, M.W.; Tan, C.; Li, C. Application of whole genome re-sequencing data in the development of diagnostic DNA markers tightly linked to a disease-resistance locus for marker-assisted selection in lupin (Lupinus angustifolius). BMC Genom. 2015, 16, 1–17. [Google Scholar] [CrossRef]
- Gremigni, P.; Hamblin, J.; Harris, D.; Cowling, W.A. The interaction of phosphorus and potassium with seed alkaloid concentrations, yield and mineral content in narrow-leafed lupin (Lupinus angustifolius L.). Plant. Soil 2003, 253, 413–427. [Google Scholar] [CrossRef]
- Jansen, G.; Jürgens, H.U.; Ordon, F. Effects of temperature on the alkaloid content of seeds of Lupinus angustifolius cultivars. J. Agron. Crop. Sci. 2009, 195, 172–177. [Google Scholar] [CrossRef]
- Li, L.; Tacke, E.; Hofferbert, H.R.; Lubeck, J.; Strahwald, J.; Draffehn, A.M.; Walkemeier, B.; Gebhardt, C. Validation of candidate gene markers for marker-assisted selection of potato cultivars with improved tuber quality. Theor. Appl. Genet. 2013, 126, 1039–1052. [Google Scholar] [CrossRef] [PubMed]
- Nie, X.; Sutherland, D.; Dickison, V.; Singh, M.; Murphy, A.M.; de Koeyer, D. Development and Validation of High-Resolution Melting Markers Derived from Rysto STS Markers for High-Throughput Marker-Assisted Selection of Potato Carrying Rysto. Phytopathology 2016, 106, 1366–1375. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Collard, B.C.Y.; Mackill, D.J. Marker-assisted selection: an approach for precision plant breeding in the twenty-first century. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2008, 363, 557–572. [Google Scholar] [CrossRef]
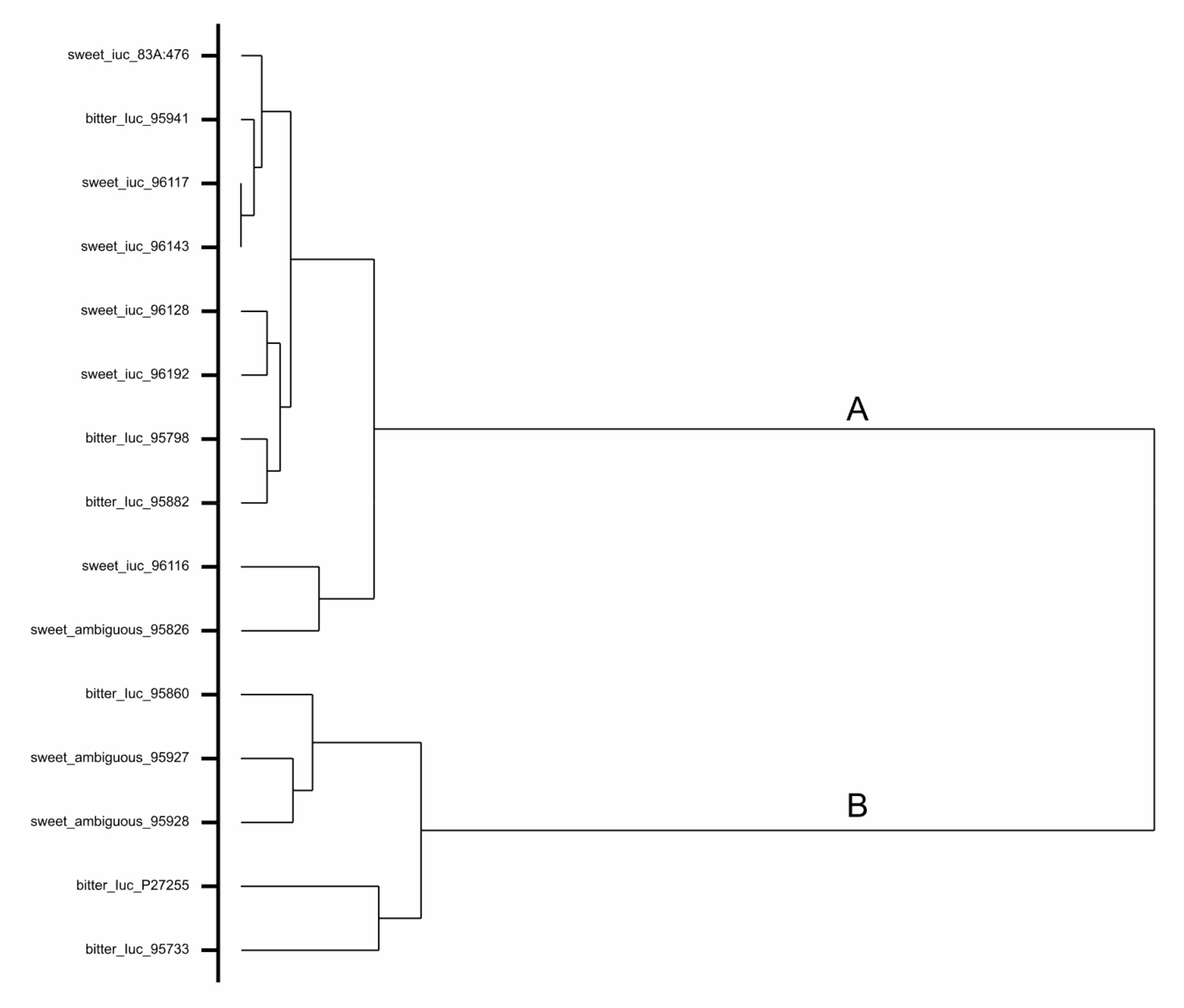

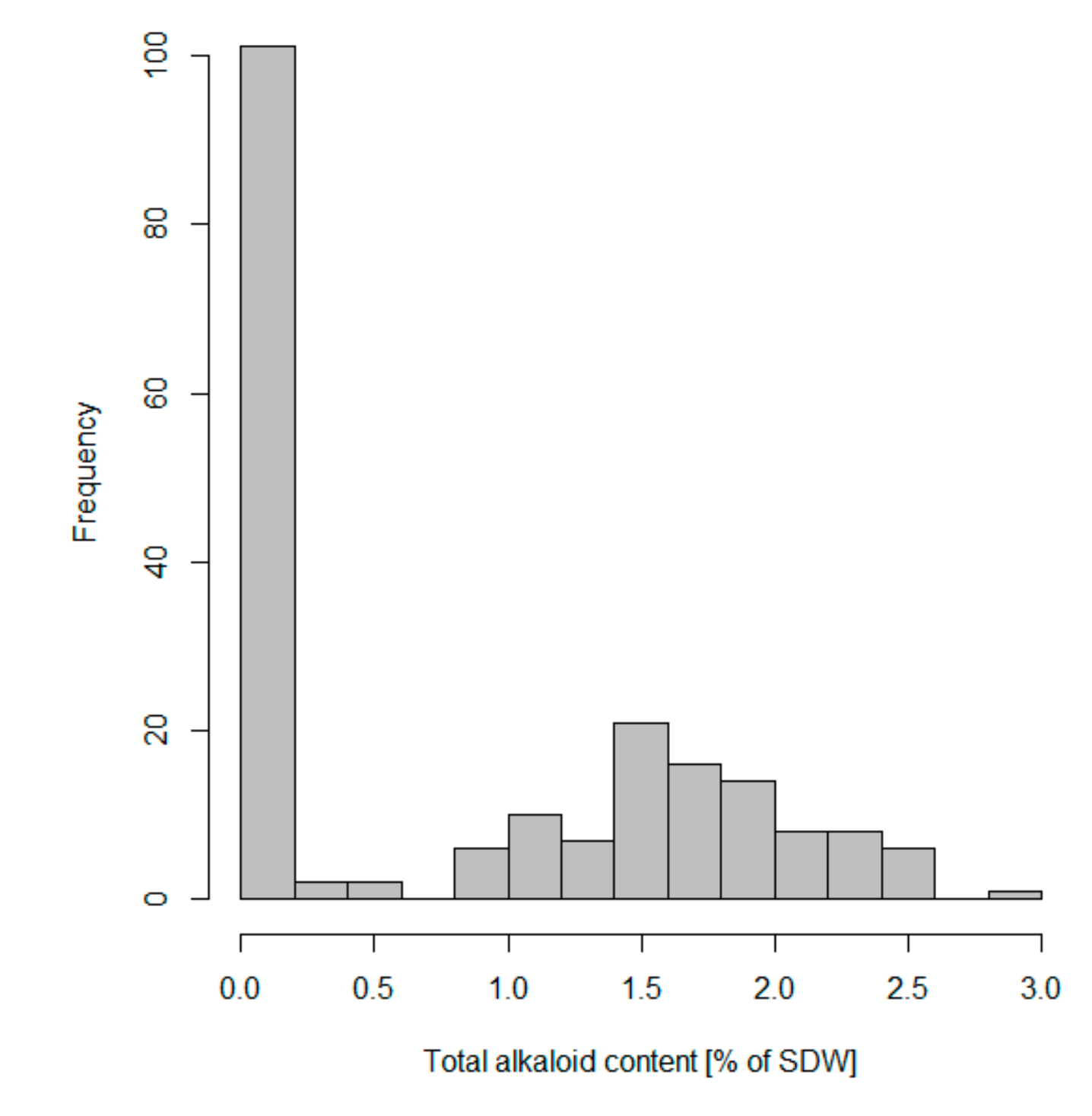
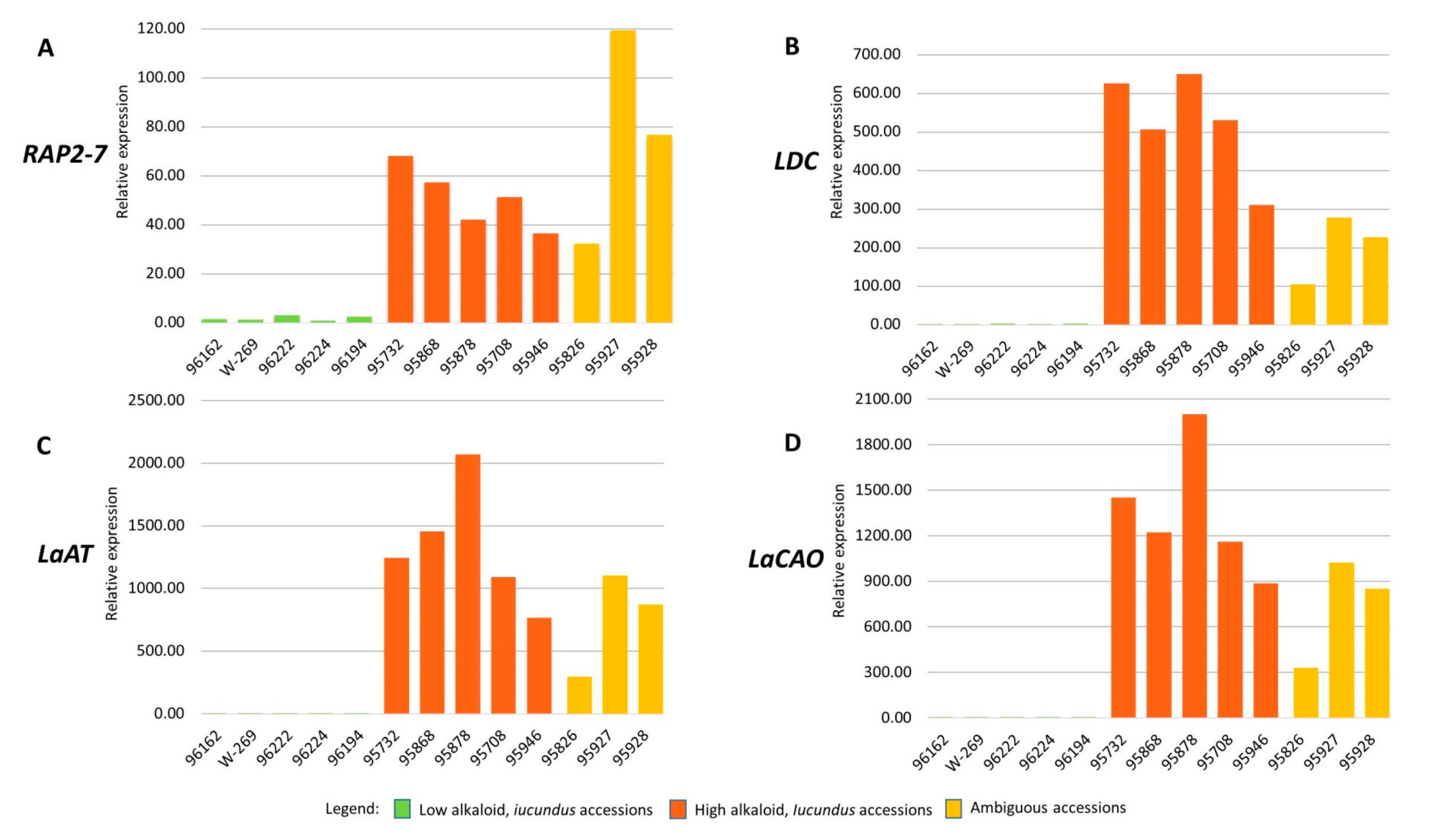
| Catalogue No. a | Accession Name | Total Alkaloid Content (% of Seed Dry Weight) | Lupanine (%) | 13-Hydroxylupanine (%) | Angustifoline (%) | Isolupanine (%) | GC Analysis | RAP2-7 DNA Sequence Assessment and Marker Development | qPCR Analysis |
|---|---|---|---|---|---|---|---|---|---|
| low-alkaloid accessions | |||||||||
| 96143 | Danja b | 0.0022 | 45.45 | 40.91 | 13.64 | - | Kamel et al. (2016) | + | |
| 96117 | Yandee b | 0.0029 | 31.03 | 48.28 | 13.79 | 6.90 | + | ||
| 96116 | IIIyarie b | 0.0032 | 31.25 | 50.00 | 12.50 | 6.25 | + | ||
| 96192 | Bordako b | 0.0157 | 52.87 | 33.76 | 7.64 | 5.73 | + | ||
| – | 83A:476, DxW maternal line b | 0.0829 | 65.36 | 21.34 | 11.65 | 1.65 | Kroc et al. (2019) | + | |
| 96128 | Stadolishchienskij L-569 b | 0.0004 | 22.26 | 60.86 | 16.81 | - | + | + | |
| – | W-269 b | 0.0010 | 46.31 | 43.89 | 4.06 | 5.75 | + | ||
| 96162 | Gunguru b | 0.0127 | 50.23 | 37.83 | 9.95 | 2.00 | + | ||
| 96224 | W-226B b | 0.0146 | 69.64 | 26.31 | 1.49 | 2.56 | + | ||
| 96222 | W-197 b | 0.0250 | 71.02 | 21.94 | 1.80 | 5.25 | + | ||
| 96194 | Borweta b | 0.0299 | 73.06 | 17.58 | 5.10 | 4.25 | + | ||
| 95826 | Brianskij-35 b | 0.1205 | 31.78 | 37.18 | 15.52 | 15.52 | Kamel et al. (2016) | ||
| Brianskij-35 c | 0.0851 | 38.16 | 26.09 | 19.81 | 15.93 | present study | + | ||
| 95927 | Brianskij-123 b | 0.1895 | 24.22 | 41.85 | 19.53 | 14.41 | Kamel et al. (2016) | ||
| Brianskij-123 c | 0.1160 | 42.03 | 20.55 | 20.06 | 17.36 | present study | + | ||
| 95928 | Brianskij-237/83 b | 0.1200 | 28.67 | 38.67 | 17.17 | 15.50 | Kamel et al. (2016) | + | |
| high-alkaloid accessions | |||||||||
| 95941 | Azuro b | 1.3809 | 56.24 | 26.73 | 15.55 | 1.48 | Kamel et al. (2016) | + | |
| 95798 | Bitter Blaue Lupine b | 1.8431 | 52.46 | 27.94 | 18.22 | 1.37 | + | ||
| 95882 | Population-29 b | 2.1170 | 46.09 | 30.71 | 20.67 | 2.52 | + | ||
| 95860 | Petakli Tikwa b | 2.2960 | 21.41 | 50.71 | 27.52 | 0.36 | + | ||
| 95733 | Population B-530/7 b | 2.3009 | 56.44 | 25.23 | 18.16 | 0.17 | + | ||
| – | P27255, DxW paternal line b | 2.7272 | 67.75 | 16.40 | 13.69 | 2.16 | Kroc et al. (2019) | + | |
| 95946 | Morsico Pop.1100 b | 2.4305 | 43.07 | 33.96 | 22.80 | 0.17 | + | ||
| 95878 | Population-4 b | 2.4975 | 12.76 | 62.16 | 24.81 | 0.26 | + | ||
| 95868 | Population-22746 b | 2.5024 | 9.82 | 65.03 | 24.90 | 0.25 | + | ||
| 95732 | Population B-529/79 b | 2.5124 | 58.52 | 25.68 | 15.57 | 0.19 | + | ||
| 95708 | Badajoz-3 b | 2.8751 | 13.63 | 66.45 | 19.64 | 0.29 | + | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kroc, M.; Czepiel, K.; Wilczura, P.; Mokrzycka, M.; Święcicki, W. Development and Validation of a Gene-Targeted dCAPS Marker for Marker-Assisted Selection of Low-Alkaloid Content in Seeds of Narrow-Leafed Lupin (Lupinus angustifolius L.). Genes 2019, 10, 428. https://doi.org/10.3390/genes10060428
Kroc M, Czepiel K, Wilczura P, Mokrzycka M, Święcicki W. Development and Validation of a Gene-Targeted dCAPS Marker for Marker-Assisted Selection of Low-Alkaloid Content in Seeds of Narrow-Leafed Lupin (Lupinus angustifolius L.). Genes. 2019; 10(6):428. https://doi.org/10.3390/genes10060428
Chicago/Turabian StyleKroc, Magdalena, Katarzyna Czepiel, Paulina Wilczura, Monika Mokrzycka, and Wojciech Święcicki. 2019. "Development and Validation of a Gene-Targeted dCAPS Marker for Marker-Assisted Selection of Low-Alkaloid Content in Seeds of Narrow-Leafed Lupin (Lupinus angustifolius L.)" Genes 10, no. 6: 428. https://doi.org/10.3390/genes10060428
APA StyleKroc, M., Czepiel, K., Wilczura, P., Mokrzycka, M., & Święcicki, W. (2019). Development and Validation of a Gene-Targeted dCAPS Marker for Marker-Assisted Selection of Low-Alkaloid Content in Seeds of Narrow-Leafed Lupin (Lupinus angustifolius L.). Genes, 10(6), 428. https://doi.org/10.3390/genes10060428





