Birth, School, Work, Death, and Resurrection: The Life Stages and Dynamics of Transposable Element Proliferation
Abstract
1. Introduction
“And he that was dead came forth, bound hand and foot with graveclothes.”John 11:44
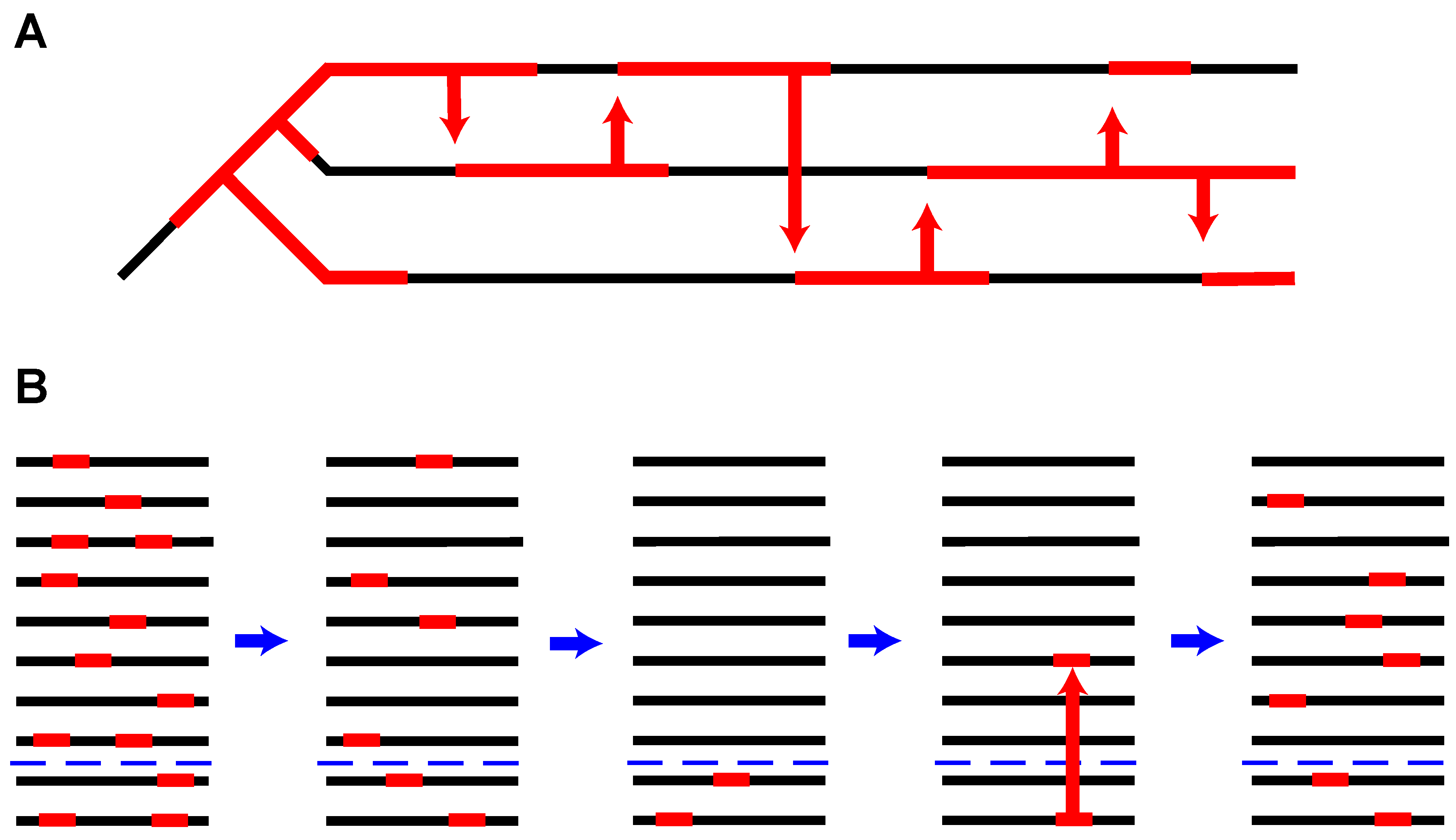
2. Long-Lasting Relationships
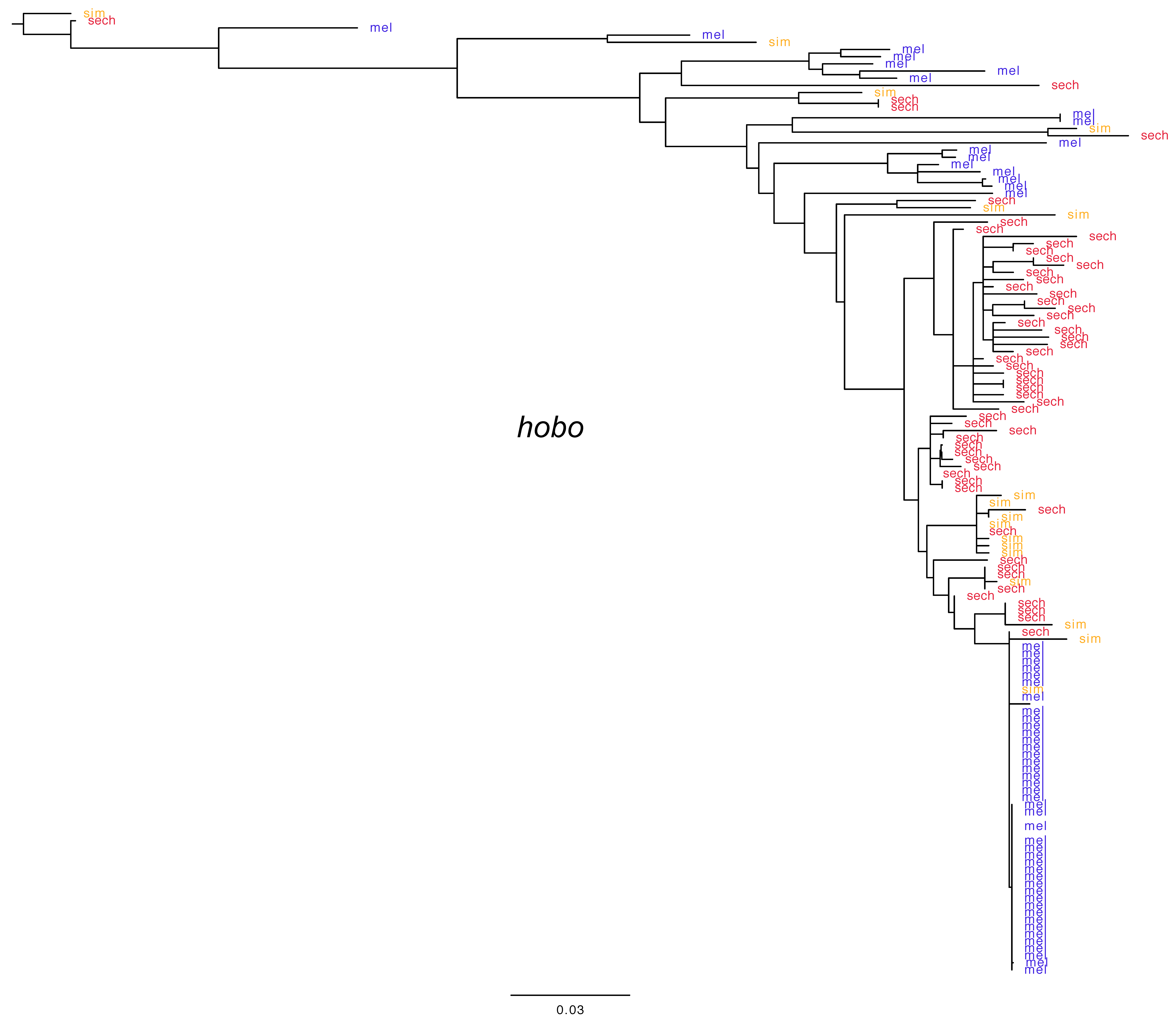
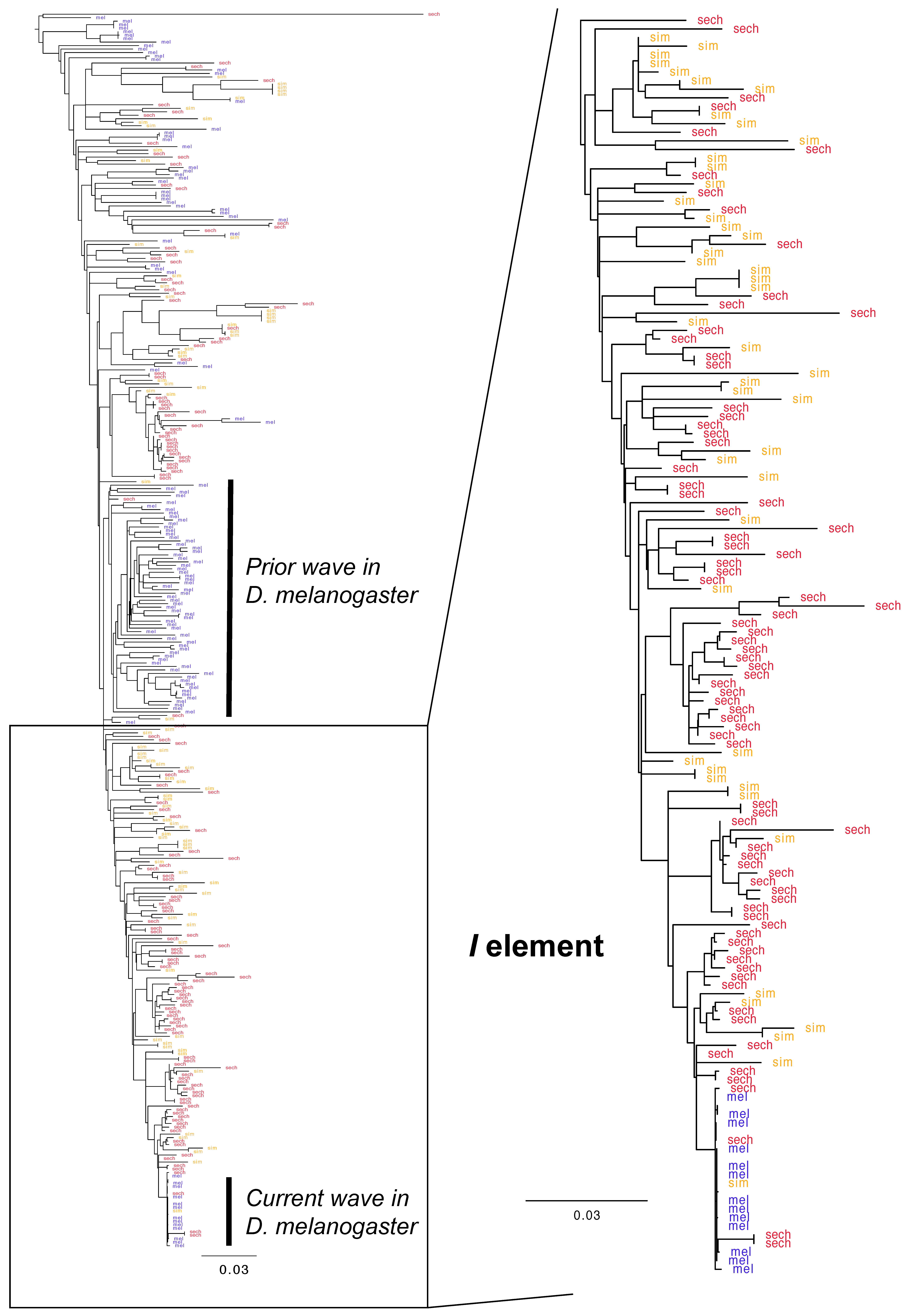
3. Horizontal Transfer: Fast, Cheap and Out of Control
“Based on our experience in building ground based mobile robots (legged and wheeled), we argue here for fast, cheap missions using large numbers of mass produced simple autonomous robots...”Brooks and Flynn. 1989. Fast, Cheap and Out of Control: A robot invasion of the solar system
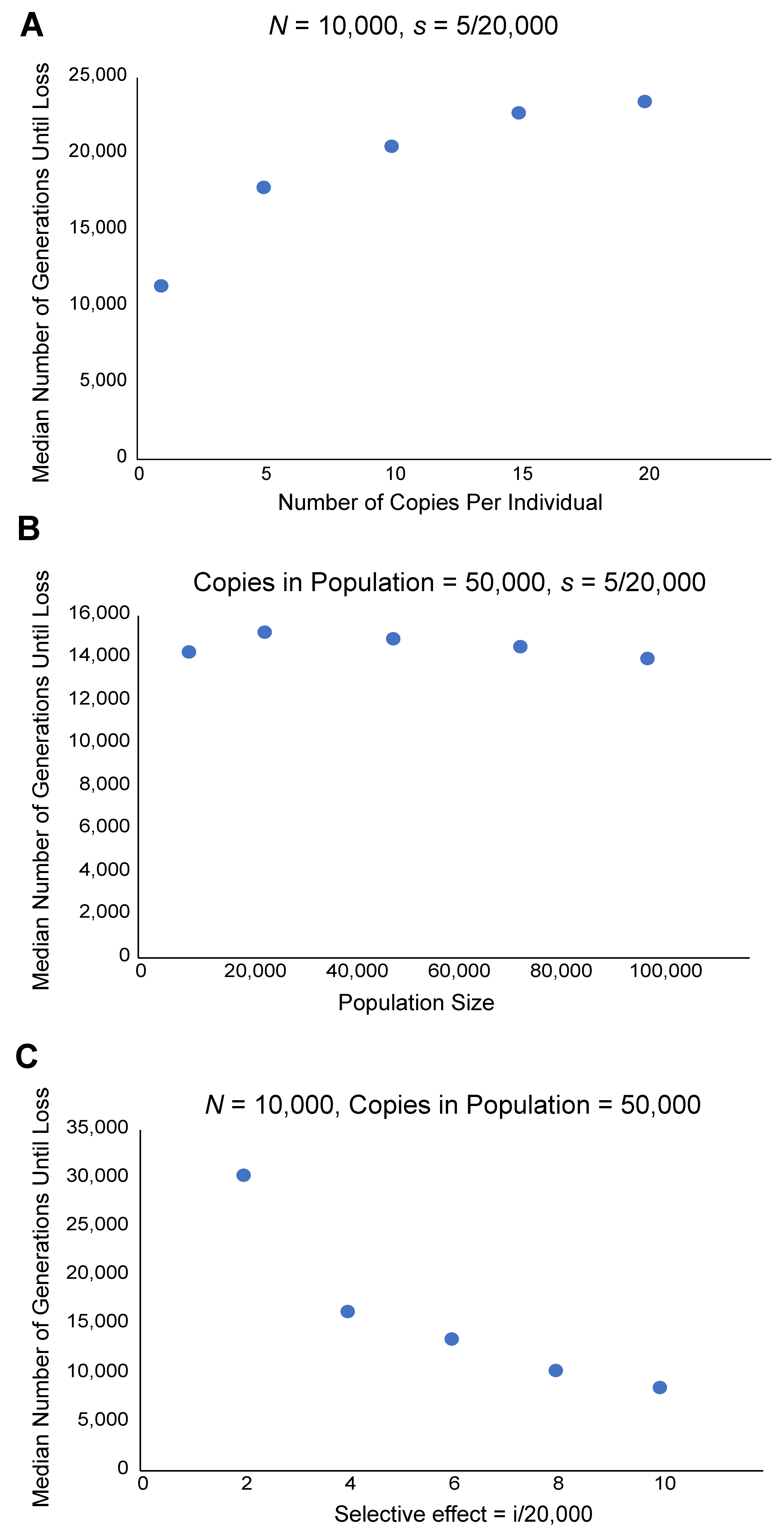
4. Extinction
5. Resurrection
Funding
Acknowledgments
Conflicts of Interest
References
- Hickey, D.A. Selfish DNA: A sexually-transmitted nuclear parasite. Genetics 1982, 101, 519–531. [Google Scholar] [PubMed]
- Charlesworth, B.; Sniegowski, P.; Stephan, W. The evolutionary dynamics of repetitive DNA in eukaryotes. Nature 1994, 371, 215–220. [Google Scholar] [CrossRef]
- Charlesworth, B.; Charlesworth, D. The population dynamics of transposable elements. Genet. Res. 1983, 42, 1–27. [Google Scholar] [CrossRef]
- Le Rouzic, A.; Capy, P. The first steps of transposable elements invasion: Parasitic strategy vs. genetic drift. Genetics 2005, 169, 1033–1043. [Google Scholar] [CrossRef]
- Song, M.J.; Schaack, S. Evolutionary Conflict between Mobile DNA and Host Genomes. Am. Nat. 2018, 192, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Venner, S.; Miele, V.; Terzian, C.; Biemont, C.; Daubin, V.; Feschotte, C.; Pontier, D. Ecological networks to unravel the routes to horizontal transposon transfers. PLoS Biol. 2017, 15, e2001536. [Google Scholar] [CrossRef] [PubMed]
- Venner, S.; Feschotte, C.; Biemont, C. Dynamics of transposable elements: Towards a community ecology of the genome. Trends Genet. TIG 2009, 25, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Kidwell, M.G.; Lisch, D.R. Perspective: Transposable elements, parasitic DNA, and genome evolution. Evolution 2001, 55, 1–24. [Google Scholar] [CrossRef]
- Hartl, D.L.; Lozovskaya, E.R.; Nurminsky, D.I.; Lohe, A.R. What restricts the activity of mariner-like transposable elements. Trends Genet. TIG 1997, 13, 197–201. [Google Scholar] [CrossRef]
- Kaplan, N.; Darden, T.; Langley, C.H. Evolution and extinction of transposable elements in Mendelian populations. Genetics 1985, 109, 459–480. [Google Scholar] [PubMed]
- Groth, S.B.; Blumenstiel, J.P. Horizontal Transfer Can Drive a Greater Transposable Element Load in Large Populations. J. Hered. 2017, 108, 36–44. [Google Scholar] [CrossRef]
- Casacuberta, E.; Pardue, M.L. Transposon telomeres are widely distributed in the Drosophila genus: TART elements in the virilis group. Proc. Natl. Acad. Sci. USA 2003, 100, 3363–3368. [Google Scholar] [CrossRef]
- Platt, R.N., 2nd; Vandewege, M.W.; Ray, D.A. Mammalian transposable elements and their impacts on genome evolution. Chromosome Res. 2018, 26, 25–43. [Google Scholar] [CrossRef] [PubMed]
- Boissinot, S.; Sookdeo, A. The Evolution of LINE-1 in Vertebrates. Genome Biol. Evol. 2016, 8, 3485–3507. [Google Scholar] [CrossRef] [PubMed]
- Ivancevic, A.M.; Kortschak, R.D.; Bertozzi, T.; Adelson, D.L. LINEs between Species: Evolutionary Dynamics of LINE-1 Retrotransposons across the Eukaryotic Tree of Life. Genome Biol. Evol. 2016, 8, 3301–3322. [Google Scholar] [CrossRef] [PubMed]
- Boissinot, S.; Chevret, P.; Furano, A.V. L1 (LINE-1) retrotransposon evolution and amplification in recent human history. Mol. Biol. Evol. 2000, 17, 915–928. [Google Scholar] [CrossRef] [PubMed]
- Lander, E.S.; Linton, L.M.; Birren, B.; Nusbaum, C.; Zody, M.C.; Baldwin, J.; Devon, K.; Dewar, K.; Doyle, M.; FitzHugh, W.; et al. Initial sequencing and analysis of the human genome. Nature 2001, 409, 860–921. [Google Scholar]
- Burton, F.H.; Loeb, D.D.; Voliva, C.F.; Martin, S.L.; Edgell, M.H.; Hutchison, C.A., 3rd. Conservation throughout mammalia and extensive protein-encoding capacity of the highly repeated DNA long interspersed sequence one. J. Mol. Biol. 1986, 187, 291–304. [Google Scholar] [CrossRef]
- Smit, A.F.; Toth, G.; Riggs, A.D.; Jurka, J. Ancestral, mammalian-wide subfamilies of LINE-1 repetitive sequences. J. Mol. Biol. 1995, 246, 401–417. [Google Scholar] [CrossRef]
- Ivancevic, A.M.; Kortschak, R.D.; Bertozzi, T.; Adelson, D.L. Horizontal transfer of BovB and L1 retrotransposons in eukaryotes. Genome Biol. 2018, 19, 85. [Google Scholar] [CrossRef]
- Furano, A.V. The biological properties and evolutionary dynamics of mammalian LINE-1 retrotransposons. Prog. Nucleic Acid Res. Mol. Biol. 2000, 64, 255–294. [Google Scholar] [PubMed]
- Khan, H.; Smit, A.; Boissinot, S. Molecular evolution and tempo of amplification of human LINE-1 retrotransposons since the origin of primates. Genome Res. 2006, 16, 78–87. [Google Scholar] [CrossRef]
- Boissinot, S.; Furano, A.V. Adaptive evolution in LINE-1 retrotransposons. Mol. Biol. Evol. 2001, 18, 2186–2194. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Goodier, J.L. Restricting retrotransposons: A review. Mob. DNA 2016, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Sookdeo, A.; Hepp, C.M.; McClure, M.A.; Boissinot, S. Revisiting the evolution of mouse LINE-1 in the genomic era. Mob. DNA 2013, 4, 3. [Google Scholar] [CrossRef] [PubMed]
- Hayward, B.E.; Zavanelli, M.; Furano, A.V. Recombination creates novel L1 (LINE-1) elements in Rattus norvegicus. Genetics 1997, 146, 641–654. [Google Scholar] [PubMed]
- Wincker, P.; Jubier-Maurin, V.; Roizes, G. Unrelated sequences at the 5’ end of mouse LINE-1 repeated elements define two distinct subfamilies. Nucleic Acids Res. 1987, 15, 8593–8606. [Google Scholar] [CrossRef]
- Jacobs, F.M.; Greenberg, D.; Nguyen, N.; Haeussler, M.; Ewing, A.D.; Katzman, S.; Paten, B.; Salama, S.R.; Haussler, D. An evolutionary arms race between KRAB zinc-finger genes ZNF91/93 and SVA/L1 retrotransposons. Nature 2014, 516, 242–245. [Google Scholar] [CrossRef]
- Furano, A.V.; Duvernell, D.D.; Boissinot, S. L1 (LINE-1) retrotransposon diversity differs dramatically between mammals and fish. Trends Genet. TIG 2004, 20, 9–14. [Google Scholar] [CrossRef]
- Novick, P.A.; Basta, H.; Floumanhaft, M.; McClure, M.A.; Boissinot, S. The evolutionary dynamics of autonomous non-LTR retrotransposons in the lizard Anolis carolinensis shows more similarity to fish than mammals. Mol. Biol. Evol. 2009, 26, 1811–1822. [Google Scholar] [CrossRef]
- Hellsten, U.; Harland, R.M.; Gilchrist, M.J.; Hendrix, D.; Jurka, J.; Kapitonov, V.; Ovcharenko, I.; Putnam, N.H.; Shu, S.; Taher, L.; et al. The genome of the Western clawed frog Xenopus tropicalis. Science 2010, 328, 633–636. [Google Scholar] [CrossRef]
- Duvernell, D.D.; Pryor, S.R.; Adams, S.M. Teleost fish genomes contain a diverse array of L1 retrotransposon lineages that exhibit a low copy number and high rate of turnover. J. Mol. Evol. 2004, 59, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Tollis, M.; Boissinot, S. Lizards and LINEs: Selection and demography affect the fate of L1 retrotransposons in the genome of the green anole (Anolis carolinensis). Genome Biol. Evol. 2013, 5, 1754–1768. [Google Scholar] [CrossRef]
- Xue, A.T.; Ruggiero, R.P.; Hickerson, M.J.; Boissinot, S. Differential Effect of Selection against LINE Retrotransposons among Vertebrates Inferred from Whole-Genome Data and Demographic Modeling. Genome Biol. Evol. 2018, 10, 1265–1281. [Google Scholar] [CrossRef]
- Petrov, D.A.; Aminetzach, Y.T.; Davis, J.C.; Bensasson, D.; Hirsh, A.E. Size matters: Non-LTR retrotransposable elements and ectopic recombination in Drosophila. Mol. Biol. Evol. 2003, 20, 880–892. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, E.A.; Huang, S.M.; Langley, C.H.; Judd, B.H. Chromosome rearrangement by ectopic recombination in Drosophila melanogaster: Genome structure and evolution. Genetics 1991, 129, 1085–1098. [Google Scholar]
- Lee, Y.C.; Langley, C.H. Long-term and short-term evolutionary impacts of transposable elements on Drosophila. Genetics 2012, 192, 1411–1432. [Google Scholar] [CrossRef] [PubMed]
- Charlesworth, B.; Langley, C.H.; Sniegowski, P.D. Transposable element distributions in Drosophila. Genetics 1997, 147, 1993–1995. [Google Scholar]
- Charlesworth, B.; Langley, C.H. The population genetics of Drosophila transposable elements. Annu. Rev. Genet. 1989, 23, 251–287. [Google Scholar] [CrossRef]
- Langley, C.H.; Montgomery, E.; Hudson, R.; Kaplan, N.; Charlesworth, B. On the role of unequal exchange in the containment of transposable element copy number. Genet. Res. 1988, 52, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Schaack, S.; Gilbert, C.; Feschotte, C. Promiscuous DNA: Horizontal transfer of transposable elements and why it matters for eukaryotic evolution. Trends Ecol. Evol. 2010, 25, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, C.; Feschotte, C. Horizontal acquisition of transposable elements and viral sequences: Patterns and consequences. Curr. Opin. Genet. Dev. 2018, 49, 15–24. [Google Scholar] [CrossRef]
- Hartl, D.L.; Lohe, A.R.; Lozovskaya, E.R. Modern thoughts on an ancyent marinere: Function, evolution, regulation. Annu. Rev. Genet. 1997, 31, 337–358. [Google Scholar] [CrossRef]
- Wallau, G.L.; Capy, P.; Loreto, E.; Le Rouzic, A.; Hua-Van, A. VHICA, a New Method to Discriminate between Vertical and Horizontal Transposon Transfer: Application to the Mariner Family within Drosophila. Mol. Biol. Evol. 2016, 33, 1094–1109. [Google Scholar] [CrossRef]
- Wallau, G.L.; Capy, P.; Loreto, E.; Hua-Van, A. Genomic landscape and evolutionary dynamics of mariner transposable elements within the Drosophila genus. BMC Genom. 2014, 15, 727. [Google Scholar] [CrossRef]
- Capy, P.; David, J.R.; Hartl, D.L. Evolution of the transposable element mariner in the Drosophila melanogaster species group. Genetica 1992, 86, 37–46. [Google Scholar] [CrossRef]
- Maruyama, K.; Hartl, D.L. Evolution of the transposable element mariner in Drosophila species. Genetics 1991, 128, 319–329. [Google Scholar] [PubMed]
- Lohe, A.R.; Moriyama, E.N.; Lidholm, D.A.; Hartl, D.L. Horizontal transmission, vertical inactivation, and stochastic loss of mariner-like transposable elements. Mol. Biol. Evol. 1995, 12, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Peccoud, J.; Loiseau, V.; Cordaux, R.; Gilbert, C. Massive horizontal transfer of transposable elements in insects. Proc. Natl. Acad. Sci. USA 2017, 114, 4721–4726. [Google Scholar] [CrossRef] [PubMed]
- Lampe, D.J.; Churchill, M.E.; Robertson, H.M. A purified mariner transposase is sufficient to mediate transposition in vitro. EMBO J. 1996, 15, 5470–5479. [Google Scholar] [CrossRef]
- Palazzo, A.; Caizzi, R.; Viggiano, L.; Marsano, R.M. Does the Promoter Constitute a Barrier in the Horizontal Transposon Transfer Process? Insight from Bari Transposons. Genome Biol. Evol. 2017, 9, 1637–1645. [Google Scholar] [CrossRef] [PubMed]
- Sookdeo, A.; Hepp, C.M.; Boissinot, S. Contrasted patterns of evolution of the LINE-1 retrotransposon in perissodactyls: The history of a LINE-1 extinction. Mob. DNA 2018, 9, 12. [Google Scholar] [CrossRef]
- Cantrell, M.A.; Scott, L.; Brown, C.J.; Martinez, A.R.; Wichman, H.A. Loss of LINE-1 activity in the megabats. Genetics 2008, 178, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Platt, R.N., 2nd; Ray, D.A. A non-LTR retroelement extinction in Spermophilus tridecemlineatus. Gene 2012, 500, 47–53. [Google Scholar] [CrossRef]
- Grahn, R.A.; Rinehart, T.A.; Cantrell, M.A.; Wichman, H.A. Extinction of LINE-1 activity coincident with a major mammalian radiation in rodents. Cytogenet. Genome Res. 2005, 110, 407–415. [Google Scholar] [CrossRef]
- Nuzhdin, S.V. Sure facts, speculations, and open questions about the evolution of transposable element copy number. Genetica 1999, 107, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Erickson, I.K.; Cantrell, M.A.; Scott, L.; Wichman, H.A. Retrofitting the genome: L1 extinction follows endogenous retroviral expansion in a group of muroid rodents. J. Virol. 2011, 85, 12315–12323. [Google Scholar] [CrossRef]
- Cantrell, M.A.; Ederer, M.M.; Erickson, I.K.; Swier, V.J.; Baker, R.J.; Wichman, H.A. MysTR: An endogenous retrovirus family in mammals that is undergoing recent amplifications to unprecedented copy numbers. J. Virol. 2005, 79, 14698–14707. [Google Scholar] [CrossRef]
- Deininger, P. Alu elements: Know the SINEs. Genome Biol. 2011, 12, 236. [Google Scholar] [CrossRef]
- Yang, L.; Wichman, H.A. Tracing the history of LINE and SINE extinction in sigmodontine rodents. bioRxiv 2018. [Google Scholar] [CrossRef]
- Hartl, D.L.; Lohe, A.R.; Lozovskaya, E.R. Regulation of the transposable element mariner. Genetica 1997, 100, 177–184. [Google Scholar] [CrossRef]
- Bingham, P.M.; Kidwell, M.G.; Rubin, G.M. The molecular basis of P-M dysgenesis—The role of the P-element, a P-Strain-Specific Transposon Family. Cell 1982, 29, 995–1004. [Google Scholar] [CrossRef]
- Bucheton, A.; Paro, R.; Sang, H.M.; Pelisson, A.; Finnegan, D.J. The molecular basis of the I-R hybrid dysgenesis syndrome in Drosophila melanogaster: Identification, cloning and properties of the I-Factor. Cell 1984, 38, 153–163. [Google Scholar] [CrossRef]
- Yannopoulos, G.; Stamatis, N.; Monastirioti, M.; Hatzopoulos, P.; Louis, C. hobo is responsible for the induction of hybrid dysgenesis by strains of Drosophila melanogater bearing the male recombination factor 23.5MRF. Cell 1987, 49, 487–495. [Google Scholar] [CrossRef]
- Blumenstiel, J.P.; Hartl, D.L. Evidence for maternally transmitted small interfering RNA in the repression of transposition in Drosophila virilis. Proc. Natl. Acad. Sci. USA 2005, 102, 15965–15970. [Google Scholar] [CrossRef]
- Brennecke, J.; Malone, C.D.; Aravin, A.A.; Sachidanandam, R.; Stark, A.; Hannon, G.J. An Epigenetic Role for Maternally Inherited piRNAs in Transposon Silencing. Science 2008, 322, 1387–1392. [Google Scholar] [CrossRef] [PubMed]
- Dorogova, N.V.; Bolobolova, E.U.; Zakharenko, L.P. Cellular aspects of gonadal atrophy in Drosophila P-M hybrid dysgenesis. Dev. Biol. 2017, 424, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Niki, Y.; Chigusa, S.I. Developmental analysis of gonadal sterility of P-M dysgenesis of Drosophila melanogaster. Jpn. J. Genet. 1986, 61, 147–157. [Google Scholar] [CrossRef]
- Daniels, S.B.; Peterson, K.R.; Strausbaugh, L.D.; Kidwell, M.G.; Chovnick, A. Evidence for horizontal transmission of the P transposable element between Drosophila species. Genetics 1990, 124, 339–355. [Google Scholar]
- Crow, J.F. The genesis of dysgenesis. Genetics 1988, 120, 315–318. [Google Scholar]
- Engels, W.R. Invasions of P elements. Genetics 1997, 145, 11–15. [Google Scholar]
- Crozatier, M.; Vaury, C.; Busseau, I.; Pelisson, A.; Bucheton, A. Structure and genomic organization of I elements involved in I-R hybrid dysgenesis in Drosophila melanogaster. Nucleic Acids Res. 1988, 16, 9199–9213. [Google Scholar] [CrossRef]
- Bucheton, A.; Simonelig, M.; Vaury, C.; Crozatier, M. Sequences similar to the I transposable element involved in I-R hybrid dysgenesis in D. melanogaster occur in other Drosophila species. Nature 1986, 322, 650–652. [Google Scholar] [CrossRef]
- Bucheton, A.; Vaury, C.; Chaboissier, M.C.; Abad, P.; Pelisson, A.; Simonelig, M. I elements and the Drosophila genome. Genetica 1992, 86, 175–190. [Google Scholar] [CrossRef]
- Vaury, C.; Abad, P.; Pelisson, A.; Lenoir, A.; Bucheton, A. Molecular characteristics of the heterochromatic I elements from a reactive strain of Drosophila melanogaster. J. Mol. Evol. 1990, 31, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Grentzinger, T.; Armenise, C.; Brun, C.; Mugat, B.; Serrano, V.; Pelisson, A.; Chambeyron, S. piRNA-mediated transgenerational inheritance of an acquired trait. Genome Res. 2012, 22, 1877–1888. [Google Scholar] [CrossRef] [PubMed]
- Periquet, G.; Hamelin, M.H.; Bigot, Y.; Lepissier, A. Geographical and historical patterns of the distribution of hobo elements in Drosophila melanogaster populations. J. Evol. Biol. 1989, 2, 223–229. [Google Scholar] [CrossRef]
- Boussy, I.A.; Itoh, M. Wanderings of hobo: A transposon in Drosophila melanogaster and its close relatives. Genetica 2004, 120, 125–136. [Google Scholar] [CrossRef]
- Ragagnin, G.T.; Bernardo, L.P.; Loreto, E.L. Unraveling the evolutionary scenario of the hobo element in populations of Drosophila melanogaster and D. simulans in South America using the TPE repeats as markers. Genet. Mol. Biol. 2016, 39, 145–150. [Google Scholar] [CrossRef]
- Boussy, I.A.; Daniels, S.B. hobo transposable elements in Drosophila melanogaster and D. simulans. Genet. Res. 1991, 58, 27–34. [Google Scholar] [CrossRef]
- Simmons, G.M. Horizontal transfer of hobo transposable elements within the Drosophila melanogaster species complex: Evidence from DNA sequencing. Mol. Biol. Evol. 1992, 9, 1050–1060. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lyozin, G.T.; Makarova, K.S.; Velikodvorskaja, V.V.; Zelentsova, H.S.; Khechumian, R.R.; Kidwell, M.G.; Koonin, E.V.; Evgen’ev, M.B. The structure and evolution of Penelope in the virilis species group of Drosophila: An ancient lineage of retroelements. J. Mol. Evol. 2001, 52, 445–456. [Google Scholar] [CrossRef]
- Evgen’ev, M.B. What happens when Penelope comes?: An unusual retroelement invades a host species genome exploring different strategies. Mob. Genet. Elem. 2013, 3, 397–408. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bartolome, C.; Bello, X.; Maside, X. Widespread evidence for horizontal transfer of transposable elements across Drosophila genomes. Genome Biol. 2009, 10, R22. [Google Scholar] [CrossRef] [PubMed]
- Hill, T.; Betancourt, A.J. Extensive exchange of transposable elements in the Drosophila pseudoobscura group. Mob. DNA 2018, 9, 20. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, C.; Schaack, S.; Pace, J.K., 2nd; Brindley, P.J.; Feschotte, C. A role for host-parasite interactions in the horizontal transfer of transposons across phyla. Nature 2010, 464, 1347–1350. [Google Scholar] [CrossRef]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blumenstiel, J.P. Birth, School, Work, Death, and Resurrection: The Life Stages and Dynamics of Transposable Element Proliferation. Genes 2019, 10, 336. https://doi.org/10.3390/genes10050336
Blumenstiel JP. Birth, School, Work, Death, and Resurrection: The Life Stages and Dynamics of Transposable Element Proliferation. Genes. 2019; 10(5):336. https://doi.org/10.3390/genes10050336
Chicago/Turabian StyleBlumenstiel, Justin P. 2019. "Birth, School, Work, Death, and Resurrection: The Life Stages and Dynamics of Transposable Element Proliferation" Genes 10, no. 5: 336. https://doi.org/10.3390/genes10050336
APA StyleBlumenstiel, J. P. (2019). Birth, School, Work, Death, and Resurrection: The Life Stages and Dynamics of Transposable Element Proliferation. Genes, 10(5), 336. https://doi.org/10.3390/genes10050336



