Human Genetic Adaptation to High Altitude: Evidence from the Andes
Abstract
:1. Introduction
2. Genetic Adaptation of Andean High-Altitude Populations
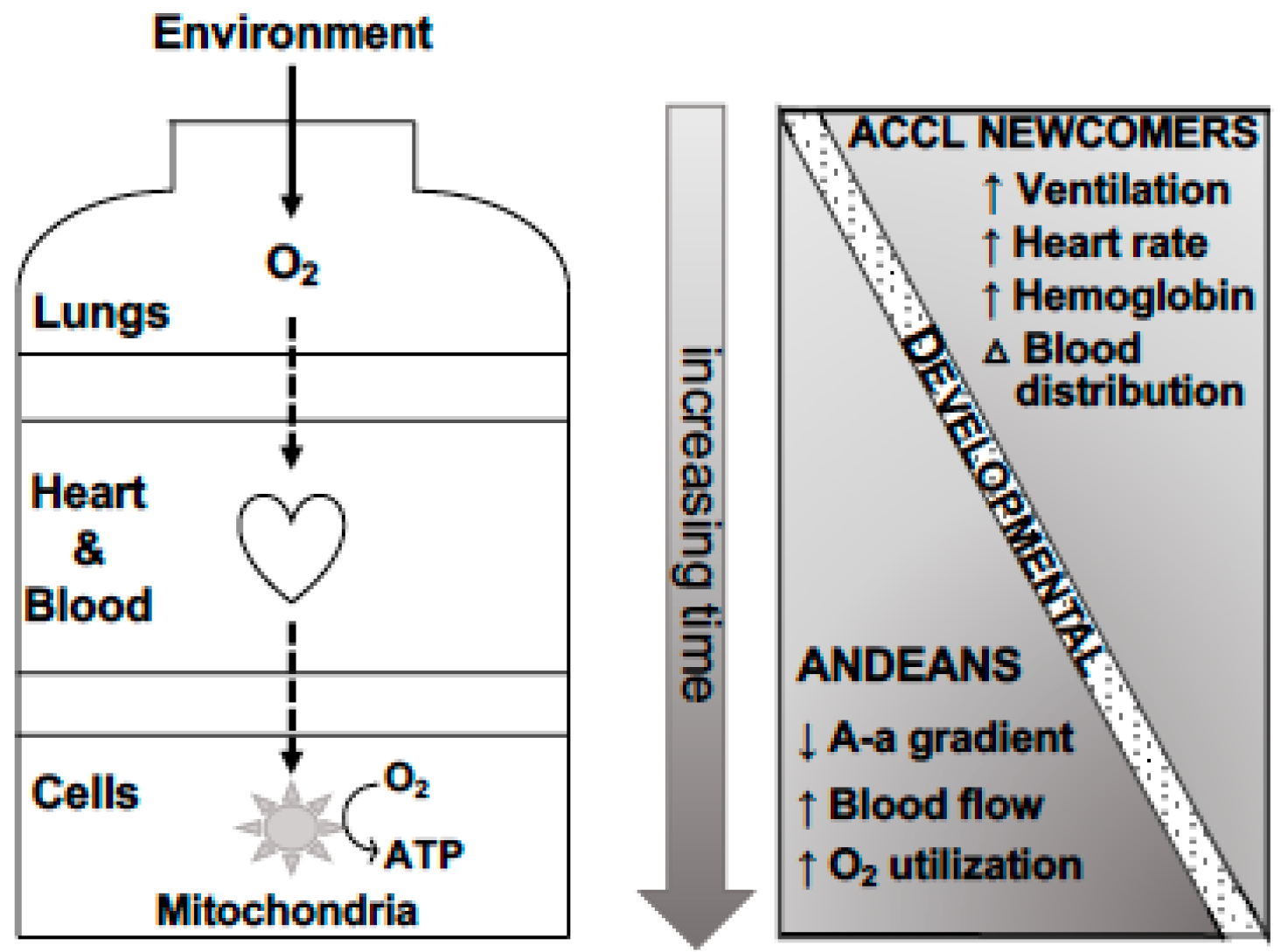
2.1. Physiologic Evidence of Genetic Adaptation to High Altitude
2.1.1. O2 Content
2.1.2. O2 Distribution
2.1.3. O2 Utilization
2.2. Genomic Evidence of Andean High Altitude Adaptation
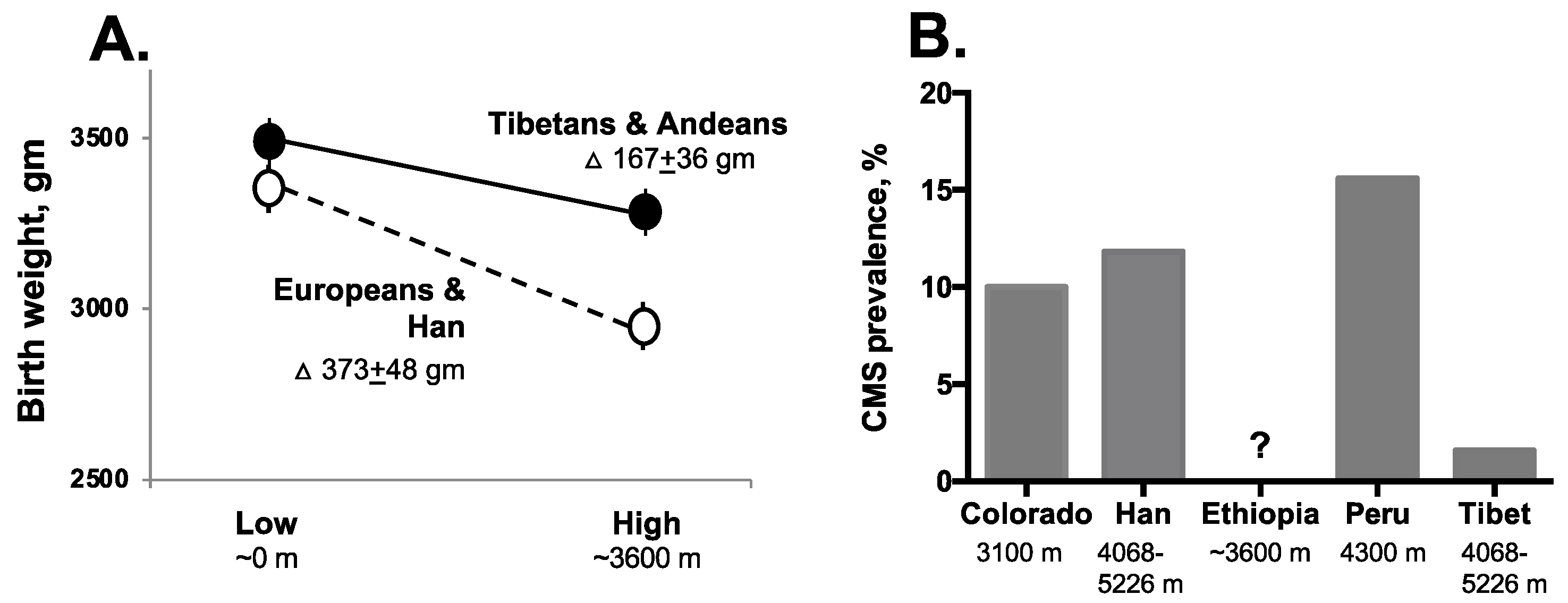
2.2.1. Perinatal Complications
2.2.2. Chronic Mountain Sickness
3. Speculation on the Role of Epigenetics for Andean High-Altitude Adaptation
3.1. Epigenetics and Transcriptional Responses to Hypoxia
3.2. Epigenetics and the Developmental Programming of Physiological Responses to Hypoxia
3.3. Inheritance of Epigenetic Marks
3.4. Querying Genomic-Epigenomic Interactions in High-Altitude Populations
4. Summary, Conclusions, and Directions for Future Work
Funding
Conflicts of Interest
References
- Gomez-Carballa, A.; Pardo-Seco, J.; Brandini, S.; Achilli, A.; Perego, U.A.; Coble, M.D.; Diegoli, T.M.; Alvarez-Iglesias, V.; Martinon-Torres, F.; Olivieri, A.; et al. The peopling of South America and the trans-Andean gene flow of the first settlers. Genome Res. 2018, 28, 767–779. [Google Scholar] [CrossRef] [PubMed]
- Harris, D.N.; Song, W.; Shetty, A.C.; Levano, K.S.; Caceres, O.; Padilla, C.; Borda, V.; Tarazona, D.; Trujillo, O.; Sanchez, C.; et al. Evolutionary genomic dynamics of Peruvians before, during, and after the Inca Empire. Proc. Natl. Acad. Sci. USA 2018, 115, E6526–E6535. [Google Scholar] [CrossRef] [PubMed]
- Dobzhansky, T. Adaptedness and fitness. In Population Biology and Evolution; Lewontin, R., Ed.; Syracuse University Press: Syracuse, NY, USA, 1968; p. 111. [Google Scholar]
- Frisancho, A.R.; Frisancho, H.G.; Milotich, M.; Brutsaert, T.; Albalak, R.; Spielvogel, H.; Villain, M.; Vargas, E.; Soria, R. Developmental, genetic, and environmental components of aerobic capacity at high altitude. Am. J. Phys. Anthropol. 1995, 96, 431–442. [Google Scholar] [CrossRef]
- Frisancho, A.R. Developmental responses to high altitude hypoxia. Am. J. Phys. Anthropol. 1970, 32, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Frisancho, A.R. Developmental adaptation: Where we go from here. Am. J. Hum. Biol. 2009, 21, 694–703. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.G. Human genetic adaptation to high altitude: Current status and future prospects. Quat. Int. 2017, 461, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, J.; Droma, T.; Sun, S.; Janes, C.; McCullough, R.E.; McCullough, R.G.; Cymerman, A.; Huang, S.Y.; Reeves, J.T.; Moore, L.G. Hypoxic ventilatory responsiveness in Tibetan compared with Han residents of 3,658 m. J. Appl. Physiol. 1993, 74, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Beall, C.M. Two routes to functional adaptation: Tibetan and Andean high-altitude natives. Proc. Natl. Acad. Sci. USA 2007, 104 (Suppl. 1), 8655–8660. [Google Scholar] [CrossRef]
- Brutsaert, T.D. Population genetic aspects and phenotypic plasticity of ventilatory responses in high altitude natives. Respir. Physiol. Neurobiol. 2007, 158, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Brutsaert, T.D.; Parra, E.J.; Shriver, M.D.; Gamboa, A.; Rivera-Ch, M.; Leon-Velarde, F. Ancestry explains the blunted ventilatory response to sustained hypoxia and lower exercise ventilation of Quechua altitude natives. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 289, R225–R234. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.G. Measuring high-altitude adaptation. J. Appl. Physiol. 2017, 123, 1371–1385. [Google Scholar] [CrossRef] [PubMed]
- Schoene, R.B.; Roach, R.C.; Lahiri, S.; Peters, R.M., Jr.; Hackett, P.H.; Santolaya, R. Increased diffusion capacity maintains arterial saturation during exercise in the Quechua Indians of Chilean Altiplano. Am. J. Hum. Biol. Off. J. Hum. Biol. Counc. 1990, 2, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, J.; Droma, T.; Sutton, J.R.; Groves, B.M.; McCullough, R.E.; McCullough, R.G.; Sun, S.; Moore, L.G. Smaller alveolar-arterial O2 gradients in Tibetan than Han residents of Lhasa (3658 m). Respir. Physiol. 1996, 103, 75–82. [Google Scholar] [CrossRef]
- Wagner, P.D.; Araoz, M.; Boushel, R.; Calbet, J.A.; Jessen, B.; Radegran, G.; Spielvogel, H.; Sondegaard, H.; Wagner, H.; Saltin, B. Pulmonary gas exchange and acid-base state at 5,260 m in high-altitude Bolivians and acclimatized lowlanders. J. Appl. Physiol. 2002, 92, 1393–1400. [Google Scholar] [CrossRef] [PubMed]
- Brutsaert, T.D.; Araoz, M.; Soria, R.; Spielvogel, H.; Haas, J.D. Higher arterial oxygen saturation during submaximal exercise in Bolivian Aymara compared to European sojourners and Europeans born and raised at high altitude. Am. J. Phys. Anthr. 2000, 113, 169–181. [Google Scholar] [CrossRef]
- DeGraff, A.C., Jr.; Grover, R.F.; Johnson, R.L., Jr.; Hammond, J.W., Jr.; Miller, J.M. Diffusing capacity of the lung in Caucasians native to 3,100 m. J. Appl. Physiol. 1970, 29, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.L., Jr.; Cassidy, S.S.; Grover, R.F.; Schutte, J.E.; Epstein, R.H. Functional capacities of lungs and thorax in beagles after prolonged residence at 3,100 m. J. Appl. Physiol. 1985, 59, 1773–1782. [Google Scholar] [CrossRef]
- Brutsaert, T.D.; Soria, R.; Caceres, E.; Spielvogel, H.; Haas, J.D. Effect of developmental and ancestral high altitude exposure on chest morphology and pulmonary function in Andean and European/North American natives. Am. J. Hum. Biol. Off. J. Hum. Biol. Counc. 1999, 11, 383–395. [Google Scholar] [CrossRef]
- Kiyamu, M.; Bigham, A.; Parra, E.; Leon-Velarde, F.; Rivera-Chira, M.; Brutsaert, T.D. Developmental and genetic components explain enhanced pulmonary volumes of female Peruvian Quechua. Am. J. Phys. Anthr. 2012, 148, 534–542. [Google Scholar] [CrossRef]
- Kiyamu, M.; León-Velarde, F.; Rivera-Chira, M.; Brutasert, T.D. Developmental effects determine submaximal arterial oxygen saturation in Peruvian Quechua. High Alt. Med. Biol. 2015, 16, 138–146. [Google Scholar] [CrossRef]
- Pomeroy, E.; Wells, J.C.; Stanojevic, S.; Miranda, J.J.; Moore, L.G.; Cole, T.J.; Stock, J.T. Surname-inferred Andean ancestry is associated with child stature and limb lengths at high altitude in Peru, but not at sea level. Am. J. Hum. Biol. 2015, 27, 798–806. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.G. Comparative human ventilatory adaptation to high altitude. Respir. Physiol. 2000, 121, 257–276. [Google Scholar] [CrossRef]
- Storz, J.F. Hemoglobin-oxygen affinity in high-altitude vertebrates: Is there evidence for an adaptive trend? J. Exp. Biol. 2016, 219, 3190–3203. [Google Scholar] [CrossRef] [PubMed]
- Winslow, R.M.; Chapman, K.W.; Gibson, C.C.; Samaja, M.; Monge, C.C.; Goldwasser, E.; Sherpa, M.; Blume, F.D.; Santolaya, R. Different hematologic responses to hypoxia in Sherpas and Quechua Indians. J. Appl. Physiol. 1989, 66, 1561–1569. [Google Scholar] [CrossRef] [PubMed]
- Bigham, A.W.; Kiyamu, M.; Leon-Velarde, F.; Parra, E.J.; Rivera-Ch, M.; Shriver, M.D.; Brutsaert, T.D. Angiotensin-converting enzyme genotype and arterial oxygen saturation at high altitude in Peruvian Quechua. High Alt. Med. Biol. 2008, 9, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Monge, C.; Bonavia, D.; León-Velarde, F.; Arregui, A. High altitude populations in Nepal and the Andes. In Hypoxia: The Adaptations; Sutton, J., Coates, G., Remmers, J., Eds.; B.C. Decker: Toronto, ON, Canada, 1990; pp. 53–58. [Google Scholar]
- Leon-Velarde, F.; Ramos, M.A.; Hernandez, J.A.; De Idiaquez, D.; Munoz, L.S.; Gaffo, A.; Cordova, S.; Durand, D.; Monge, C. The role of menopause in the development of chronic mountain sickness. Am. J. Physiol. 1997, 272, R90–R94. [Google Scholar] [CrossRef] [PubMed]
- Ballew, C.; Garruto, R.M.; Haas, J. High altitude hematology: Paradigm or enigma. In Human Population Biology; Little, M.A., Haas, J.D., Eds.; Oxford University Press: Oxford, UK, 1989; pp. 239–262. [Google Scholar]
- Rupert, J.L.; Hochachka, P.W. The evidence for hereditary factors contributing to high altitude adaptation in Andean natives: A review. High Alt. Med. Biol. 2001, 2, 235–256. [Google Scholar] [CrossRef]
- Vogel, J.A.; Hartley, L.H.; Cruz, J.C. Cardiac output during exercise in altitude natives at sea level and high altitude. J. Appl. Physiol. 1974, 36, 173–176. [Google Scholar] [CrossRef]
- Hochachka, P.W.; Clark, C.M.; Holden, J.E.; Stanley, C.; Ugurbil, K.; Menon, R.S. 31P magnetic resonance spectroscopy of the Sherpa heart: A phosphocreatine/adenosine triphosphate signature of metabolic defense against hypobaric hypoxia. Proc. Natl. Acad. Sci. USA 1996, 93, 1215–1220. [Google Scholar] [CrossRef]
- Sawka, M.N.; Young, A.J.; Rock, P.B.; Lyons, T.P.; Boushel, R.; Freund, B.J.; Muza, S.R.; Cymerman, A.; Dennis, R.C.; Pandolf, K.B.; et al. Altitude acclimatization and blood volume: Effects of exogenous erythrocyte volume expansion. J. Appl. Physiol. 1996, 81, 636–642. [Google Scholar] [CrossRef]
- Banchero, N.; Sime, F.; Peñaloza, D.; Cruz, J.; Gamboa, R.; Marticorena, E. Pulmonary pressure, cardiac output, and arterial oxygen saturation during exercise at high altitude and at sea level. Circulation 1966, 33, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jibaja, J.; Banchero, N.; Sime, F.; Peñaloza, D.; Gamboa, R.; Marticorena, E. Correlation between pulmonary artery pressure and level of altitude. Dis. Chest 1964, 46, 446–451. [Google Scholar] [CrossRef]
- Brutsaert, T.D. Do high-altitude natives have enhanced exercise performance at altitude? Appl. Physiol. Nutr. Metab. 2008, 33, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Soria, R.; Egger, M.; Scherrer, U.; Bender, N.; Rimoldi, S.F. Pulmonary artery pressure and arterial oxygen saturation in people living at high or low altitude: Systematic review and meta-analysis. J. Appl. Physiol. 2016, 121, 1151–1159. [Google Scholar] [CrossRef]
- Beall, C.M.; Decker, M.J.; Brittenham, G.M.; Kushner, I.; Gebremedhin, A.; Strohl, K.P. An Ethiopian pattern of human adaptation to high-altitude hypoxia. Proc. Natl. Acad. Sci. USA 2002, 99, 17215–17218. [Google Scholar] [CrossRef]
- Jansen, G.F.; Basnyat, B. Brain blood flow in Andean and Himalayan high-altitude populations: Evidence of different traits for the same environmental constraint. J. Cereb. Blood Flow Metab. 2011, 31, 706–714. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.R.; Mayhew, T.M.; Haas, J.D. The volumetric composition of human term placentae: Altitudinal, ethnic and sex differences in Bolivia. J. Anat. 1987, 152, 173–187. [Google Scholar] [PubMed]
- Groves, B.M.; Droma, T.; Sutton, J.R.; McCullough, R.G.; McCullough, R.E.; Zhuang, J.; Rapmund, G.; Sun, S.; Janes, C.; Moore, L.G. Minimal hypoxic pulmonary hypertension in normal Tibetans at 3,658 m. J. Appl. Physiol. 1993, 74, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.Y.; Sun, S.; Droma, T.; Zhuang, J.; Tao, J.X.; McCullough, R.G.; McCullough, R.E.; Micco, A.J.; Reeves, J.T.; Moore, L.G. Internal carotid arterial flow velocity during exercise in Tibetan and Han residents of Lhasa (3,658 m). J. Appl. Physiol. 1992, 73, 2638–2642. [Google Scholar] [CrossRef]
- Moore, L.G. Uterine blood flow as a determinant of feto-placental development. In The Placenta and Human Developmental Programming; Burton, G.J., Barker, D.J.P., Moffett, A., Thornburg, K.L., Eds.; Cambridge University Press: Cambridge, UK, 2011; pp. 126–146. [Google Scholar]
- Tymko, M.M.; Tremblay, J.C.; Hansen, A.B.; Howe, C.A.; Willie, C.K.; Stembridge, M.; Green, D.J.; Hoiland, R.L.; Subedi, P.; Anholm, J.D.; et al. The effect of alpha1-adrenergic blockade on post-exercise brachial artery flow-mediated dilatation at sea level and high altitude. J. Physiol. 2017, 595, 1671–1686. [Google Scholar] [CrossRef]
- Dhar, P.; Sharma, V.K.; Hota, K.B.; Das, S.K.; Hota, S.K.; Srivastava, R.B.; Singh, S.B. Autonomic cardiovascular responses in acclimatized lowlanders on prolonged stay at high altitude: A longitudinal follow up study. PLoS ONE 2014, 9, e84274. [Google Scholar] [CrossRef] [PubMed]
- Lundby, C.; Sander, M.; van Hall, G.; Saltin, B.; Calbet, J.A. Maximal exercise and muscle oxygen extraction in acclimatizing lowlanders and high altitude natives. J. Physiol. 2006, 573, 535–547. [Google Scholar] [CrossRef] [PubMed]
- Hoiland, R.L.; Bain, A.R.; Rieger, M.G.; Bailey, D.M.; Ainslie, P.N. Hypoxemia, oxygen content, and the regulation of cerebral blood flow. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2016, 310, R398–R413. [Google Scholar] [CrossRef] [PubMed]
- Norcliffe, L.J.; Rivera-Ch, M.; Claydon, V.E.; Moore, J.P.; Leon-Velarde, F.; Appenzeller, O.; Hainsworth, R. Cerebrovascular responses to hypoxia and hypocapnia in high-altitude dwellers. J. Physiol. 2005, 566, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.G.; Zamudio, S.; Zhuang, J.; Sun, S.; Droma, T. Oxygen transport in Tibetan women during pregnancy at 3,658 m. Am. J. Phys. Anthr. 2001, 114, 42–53. [Google Scholar] [CrossRef]
- Wilson, M.J.; Lopez, M.; Vargas, M.; Julian, C.; Tellez, W.; Rodriguez, A.; Bigham, A.; Armaza, J.F.; Niermeyer, S.; Shriver, M.; et al. Greater uterine artery blood flow during pregnancy in multigenerational (Andean) than shorter-term (European) high-altitude residents. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 293, R1313–R1324. [Google Scholar] [CrossRef] [PubMed]
- Julian, C.G.; Wilson, M.J.; Lopez, M.; Yamashiro, H.; Tellez, W.; Rodriguez, A.; Bigham, A.W.; Shriver, M.D.; Rodriguez, C.; Vargas, E.; et al. Augmented uterine artery blood flow and oxygen delivery protect Andeans from altitude-associated reductions in fetal growth. Am. J. Physiol. 2009, 296, R1564–R1575. [Google Scholar] [CrossRef]
- Julian, C.G.; Vargas, E.; Browne, V.A.; Wilson, M.J.; Bigham, A.W.; Rodriguez, C.; McCord, J.M.; Moore, L.G. Potential role for elevated maternal enzymatic antioxidant status in Andean protection against altitude-associated SGA. J. Matern. Fetal Neonatal Med. 2012, 25, 1233–1240. [Google Scholar] [CrossRef]
- Davila, R.D.; Julian, C.G.; Wilson, M.J.; Browne, V.A.; Rodriguez, C.; Bigham, A.W.; Shriver, M.D.; Vargas, E.; Moore, L.G. Do anti-angiogenic or angiogenic factors contribute to the protection of birth weight at high altitude afforded by Andean ancestry? Reprod. Sci. 2010, 17, 861–870. [Google Scholar] [CrossRef]
- Julian, C.G.; Hageman, J.L.; Wilson, M.J.; Vargas, E.; Moore, L.G. Lowland origin women raised at high altitude are not protected against lower uteroplacental O2 delivery during pregnancy or reduced birth weight. Am. J. Hum. Biol. 2011, 23, 509–516. [Google Scholar] [CrossRef]
- Charles, S.M.; Julian, C.G.; Vargas, E.; Moore, L.G. Higher estrogen levels during pregnancy in Andean than European residents of high altitude suggest differences in aromatase activity. J. Clin. Endocrinol. Metab. 2014, 99, 2908–2916. [Google Scholar] [CrossRef] [PubMed]
- Tissot van Patot, M.C.; Bendrick-Peart, J.; Beckey, V.E.; Serkova, N.; Zwerdlinger, L. Greater vascularity, lowered HIF-1/DNA binding, and elevated GSH as markers of adaptation to in vivo chronic hypoxia. Am. J. Physiol. Lung Cell. Mol. Physiol. 2004, 287, L525–L532. [Google Scholar] [CrossRef] [PubMed]
- Gassmann, N.N.; van Elteren, H.A.; Goos, T.G.; Morales, C.R.; Rivera-Ch, M.; Martin, D.S.; Cabala Peralta, P.; Passano Del Carpio, A.; Aranibar Machaca, S.; Huicho, L.; et al. Pregnancy at high altitude in the Andes leads to increased total vessel density in healthy newborns. J. Appl. Physiol. 2016, 121, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Holden, J.E.; Stone, C.K.; Clark, C.M.; Brown, W.D.; Nickles, R.J.; Stanley, C.; Hochachka, P.W. Enhanced cardiac metabolism of plasma glucose in high-altitude natives: Adaptation against chronic hypoxia. J. Appl. Physiol. 1995, 79, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Brooks, G.A.; Butterfield, G.E.; Wolfe, R.R.; Groves, B.M.; Mazzeo, R.S.; Sutton, J.R.; Wolfel, E.E.; Reeves, J.T. Increased dependence on blood glucose after acclimatization to 4,300 m. J. Appl. Physiol. 1991, 70, 919–927. [Google Scholar] [CrossRef]
- Braun, B.; Mawson, J.T.; Muza, S.R.; Dominick, S.B.; Brooks, G.A.; Horning, M.A.; Rock, P.B.; Moore, L.G.; Mazzeo, R.S.; Ezeji-Okoye, S.C.; et al. Women at altitude: Carbohydrate utilization during exercise at 4,300 m. J Appl Physiol 2000, 88, 246–256. [Google Scholar] [CrossRef]
- Woolcott, O.O.; Ader, M.; Bergman, R.N. Glucose homeostasis during short-term and prolonged exposure to high altitudes. Endocr. Rev. 2015, 36, 149–173. [Google Scholar] [CrossRef]
- Horscroft, J.A.; Kotwica, A.O.; Laner, V.; West, J.A.; Hennis, P.J.; Levett, D.Z.H.; Howard, D.J.; Fernandez, B.O.; Burgess, S.L.; Ament, S.; et al. Metabolic basis to Sherpa altitude adaptation. Proc. Natl. Acad. Sci. USA 2017, 114, 6382–6387. [Google Scholar] [CrossRef]
- Krampl, E.; Kametas, N.A.; Nowotny, P.; Roden, M.; Nicolaides, K.H. Glucose metabolism in pregnancy at high altitude. Diabetes Care 2001, 24, 817–822. [Google Scholar] [CrossRef]
- Zamudio, S.; Torricos, T.; Fik, E.; Oyala, M.; Echalar, L.; Pullockaran, J.; Tutino, E.; Martin, B.; Belliappa, S.; Balanza, E.; et al. Hypoglycemia and the origin of hypoxia-induced reduction in human fetal growth. PLoS ONE 2010, 5, e8551. [Google Scholar] [CrossRef]
- Semenza, G.L. Oxygen sensing, homeostasis, and disease. N. Engl. J. Med. 2011, 365, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.L.; Jiang, B.H.; Rue, E.A.; Semenza, G.L. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc. Natl. Acad. Sci. USA 1995, 92, 5510–5514. [Google Scholar] [CrossRef] [PubMed]
- Bruick, R.K.; McKnight, S.L. A conserved family of prolyl-4-hydroxylases that modify HIF. Science 2001, 294, 1337–1340. [Google Scholar] [CrossRef] [PubMed]
- Ivan, M.; Kondo, K.; Yang, H.; Kim, W.; Valiando, J.; Ohh, M.; Salic, A.; Asara, J.M.; Lane, W.S.; Kaelin, W.G., Jr. HIFα targeted for VHL-mediated destruction by proline hydroxylation: Implications for O2 sensing. Science 2001, 292, 464–468. [Google Scholar] [CrossRef] [PubMed]
- Jaakkola, P.; Mole, D.R.; Tian, Y.M.; Wilson, M.I.; Gielbert, J.; Gaskell, S.J.; von Kriegsheim, A.; Hebestreit, H.F.; Mukherji, M.; Schofield, C.J.; et al. Targeting of HIF-α to the von Hippel-Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science 2001, 292, 468–472. [Google Scholar] [CrossRef] [PubMed]
- Majmundar, A.J.; Wong, W.J.; Simon, M.C. Hypoxia-inducible factors and the response to hypoxic stress. Mol. Cell 2010, 40, 294–309. [Google Scholar] [CrossRef]
- Bigham, A.W.; Mao, X.; Mei, R.; Brutsaert, T.; Wilson, M.J.; Julian, C.G.; Parra, E.J.; Akey, J.M.; Moore, L.G.; Shriver, M.D. Identifying positive selection candidate loci for high-altitude adaptation in Andean populations. Hum. Genom. 2009, 4, 79–90. [Google Scholar]
- Jacovas, V.C.; Couto-Silva, C.M.; Nunes, K.; Lemes, R.B.; de Oliveira, M.Z.; Salzano, F.M.; Bortolini, M.C.; Hunemeier, T. Selection scan reveals three new loci related to high altitude adaptation in Native Andeans. Sci. Rep. 2018, 8, 12733. [Google Scholar] [CrossRef]
- Crawford, J.E.; Amaru, R.; Song, J.; Julian, C.G.; Racimo, F.; Cheng, J.Y.; Guo, X.; Yao, J.; Ambale-Venkatesh, B.; Lima, J.A.; et al. Natural selection on genes related to cardiovascular health in high-altitude adapted Andeans. Am. J. Hum. Genet. 2017, 101, 752–767. [Google Scholar] [CrossRef] [PubMed]
- Bigham, A.; Bauchet, M.; Pinto, D.; Mao, X.; Akey, J.M.; Mei, R.; Scherer, S.W.; Julian, C.G.; Wilson, M.J.; Lopez Herraez, D.; et al. Identifying signatures of natural selection in Tibetan and Andean populations using dense genome scan data. PLoS Genet. 2010, 6, e1001116. [Google Scholar] [CrossRef]
- Eichstaedt, C.A.; Antao, T.; Pagani, L.; Cardona, A.; Kivisild, T.; Mormina, M. The Andean adaptive toolkit to counteract high altitude maladaptation: Genome-wide and phenotypic analysis of the Collas. PLoS ONE 2014, 9, e93314. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.G.; Shriver, M.; Bemis, L.; Hickler, B.; Wilson, M.; Brutsaert, T.; Parra, E.; Vargas, E. Maternal adaptation to high-altitude pregnancy: An experiment of nature. Placenta 2004, 25, S60–S71. [Google Scholar] [CrossRef]
- Valverde, G.; Zhou, H.; Lippold, S.; de Filippo, C.; Tang, K.; Lopez Herraez, D.; Li, J.; Stoneking, M. A novel candidate region for genetic adaptation to high altitude in Andean populations. PLoS ONE 2015, 10, e0125444. [Google Scholar] [CrossRef] [PubMed]
- Jacovas, V.C.; Rovaris, D.L.; Perez, O.; de Azevedo, S.; Macedo, G.S.; Sandoval, J.R.; Salazar-Granara, A.; Villena, M.; Dugoujon, J.M.; Bisso-Machado, R.; et al. Genetic variations in the TP53 pathway in Native Americans strongly suggest adaptation to the high altitudes of the Andes. PLoS ONE 2015, 10, e0137823. [Google Scholar] [CrossRef] [PubMed]
- Rademaker, K.; Hodgins, G.; Moore, K.; Zarrillo, S.; Miller, C.; Bromley, G.R.; Leach, P.; Reid, D.A.; Alvarez, W.Y.; Sandweiss, D.H. Paleoindian settlement of the high-altitude Peruvian Andes. Science 2014, 346, 466–469. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.G. Human genetic adaptation to high altitude. High Alt. Med. Biol. 2001, 2, 257–279. [Google Scholar] [CrossRef] [PubMed]
- Niermeyer, S.; Andrade, M.M.; Vargas, E.; Moore, L.G. Neonatal oxygenation, pulmonary hypertension, and evolutionary adaptation to high altitude (2013 Grover Conference series). Pulm Circ. 2015, 5, 48–62. [Google Scholar] [CrossRef]
- Gonzales, G.F. Peruvian contributions to the study on human reproduction at high altitude: From the chronicles of the Spanish conquest to the present. Respir. Physiol. Neurobiol. 2007, 158, 172–179. [Google Scholar] [CrossRef]
- Julian, C.G.; Vargas, E.; Armaza, J.F.; Wilson, M.J.; Niermeyer, S.; Moore, L.G. High-altitude ancestry protects against hypoxia-associated reductions in fetal growth. Arch. Dis. Child Fetal Neonatal Ed. 2007, 92, F372–F377. [Google Scholar] [CrossRef]
- Bennett, A.; Sain, S.R.; Vargas, E.; Moore, L.G. Evidence that parent-of-origin affects birth-weight reductions at high altitude. Am. J. Hum. Biol. 2008, 20, 592–597. [Google Scholar] [CrossRef]
- Soria, R.; Julian, C.; Vargas, E.; Moore, L.; Giussani, D. Graduated effects of high-altitude hypoxia and highland ancestry on birth size. Pediatr. Res. 2013, 74, 633–638. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.G.; Young, D.; McCullough, R.E.; Droma, T.; Zamudio, S. Tibetan protection from intrauterine growth restriction (IUGR) and reproductive loss at high altitude. Am. J. Hum. Biol. 2001, 13, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Zamudio, S.; Postigo, L.; Illsley, N.P.; Rodriguez, C.; Heredia, G.; Brimacombe, M.; Echalar, L.; Torricos, T.; Tellez, W.; Maldonado, I.; et al. Maternal oxygen delivery is not related to altitude- and ancestry-associated differences in human fetal growth. J. Physiol. 2007, 582, 883–895. [Google Scholar] [CrossRef] [PubMed]
- Zamudio, S.; Palmer, S.K.; Droma, T.; Stamm, E.; Coffin, C.; Moore, L.G. Effect of altitude on uterine artery blood flow during normal pregnancy. J. Appl. Physiol. 1995, 79, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Julian, C.G.; Galan, H.L.; Wilson, M.J.; Desilva, W.; Cioffi-Ragan, D.; Schwartz, J.; Moore, L.G. Lower uterine artery blood flow and higher endothelin relative to nitric oxide metabolite levels are associated with reductions in birth weight at high altitude. Am. J. Physiol. 2008, 295, R906–R915. [Google Scholar] [CrossRef] [PubMed]
- Browne, V.A.; Toledo-Jaldin, L.; Davila, R.D.; Lopez, L.P.; Yamashiro, H.; Cioffi-Ragan, D.; Julian, C.G.; Wilson, M.J.; Bigham, A.W.; Shriver, M.D.; et al. High-end arteriolar resistance limits uterine artery blood flow and restricts fetal growth in preeclampsia and gestational hypertension at high altitude. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, R1221–R1229. [Google Scholar] [CrossRef] [PubMed]
- Mayhew, T.M.; Ohadike, C.; Baker, P.N.; Crocker, I.P.; Mitchell, C.; Ong, S.S. Stereological investigation of placental morphology in pregnancies complicated by pre-eclampsia with and without intrauterine growth restriction. Placenta 2003, 24, 219–226. [Google Scholar] [CrossRef]
- Bellamy, L.; Casas, J.P.; Hingorani, A.D.; Williams, D.J. Pre-eclampsia and risk of cardiovascular disease and cancer in later life: Systematic review and meta-analysis. BMJ 2007, 335, 974. [Google Scholar] [CrossRef]
- Irgens, H.U.; Reisaeter, L.; Irgens, L.M.; Lie, R.T. Long term mortality of mothers and fathers after pre-eclampsia: Population based cohort study. BMJ 2001, 323, 1213–1217. [Google Scholar] [CrossRef]
- Lykke, J.A.; Langhoff-Roos, J.; Sibai, B.M.; Funai, E.F.; Triche, E.W.; Paidas, M.J. Hypertensive pregnancy disorders and subsequent cardiovascular morbidity and type 2 diabetes mellitus in the mother. Hypertension 2009, 53, 944–951. [Google Scholar] [CrossRef]
- Roberts, J.M.; Hubel, C.A. Pregnancy: A screening test for later life cardiovascular disease. Womens Health Issues 2010, 20, 304–307. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.C.; Pell, J.P.; Walsh, D. Pregnancy complications and maternal risk of ischaemic heart disease: A retrospective cohort study of 129,290 births. Lancet 2001, 357, 2002–2006. [Google Scholar] [CrossRef]
- Wikstrom, A.K.; Haglund, B.; Olovsson, M.; Lindeberg, S.N. The risk of maternal ischaemic heart disease after gestational hypertensive disease. BJOG Int. J. Obstet. Gynaecol. 2005, 112, 1486–1491. [Google Scholar] [CrossRef]
- Palmer, S.K.; Moore, L.G.; Young, D.; Cregger, B.; Berman, J.C.; Zamudio, S. Altered blood pressure course during normal pregnancy and increased preeclampsia at high altitude (3100 meters) in Colorado. Am. J. Obs. Gynecol. 1999, 180, 1161–1168. [Google Scholar] [CrossRef]
- Keyes, L.E.; Armaza, J.F.; Niermeyer, S.; Vargas, E.; Young, D.A.; Moore, L.G. Intrauterine growth restriction, preeclampsia, and intrauterine mortality at high altitude in Bolivia. Pediatr. Res. 2003, 54, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.; Tudor, C.; Thorsten, V.R.; Craig, S.; Le, P.; Wright, L.L.; Varner, M.W. Maternal and neonatal outcomes of hospital vaginal deliveries in Tibet. Int. J. Gynaecol. Obstet. 2007, 98, 217–221. [Google Scholar] [CrossRef]
- Chaiworapongsa, T.; Chaemsaithong, P.; Korzeniewski, S.J.; Yeo, L.; Romero, R. Pre-eclampsia part 2: Prediction, prevention and management. Nat. Rev. Nephrol. 2014, 10, 531–540. [Google Scholar] [CrossRef] [PubMed]
- Bigham, A.W.; Julian, C.G.; Wilson, M.J.; Vargas, E.; Browne, V.A.; Shriver, M.D.; Moore, L.G. Maternal PRKAA1 and EDNRA genotypes are associated with birth weight, and PRKAA1 with uterine artery diameter and metabolic homeostasis at high altitude. Physiol. Genom. 2014, 46, 687–697. [Google Scholar] [CrossRef]
- Roos, S.; Powell, T.L.; Jansson, T. Placental mTOR links maternal nutrient availability to fetal growth. Biochem. Soc. Trans. 2009, 37, 295–298. [Google Scholar] [CrossRef]
- Yung, H.W.; Calabrese, S.; Hynx, D.; Hemmings, B.A.; Cetin, I.; Charnock-Jones, D.S.; Burton, G.J. Evidence of placental translation inhibition and endoplasmic reticulum stress in the etiology of human intrauterine growth restriction. Am. J. Pathol. 2008, 173, 451–462. [Google Scholar] [CrossRef]
- Yung, H.W.; Cox, M.; Tissot van Patot, M.; Burton, G.J. Evidence of endoplasmic reticulum stress and protein synthesis inhibition in the placenta of non-native women at high altitude. FASEB J. 2012, 26, 1970–1981. [Google Scholar] [CrossRef]
- Hill, C.M.; Baya, A.; Gavlak, J.; Carroll, A.; Heathcote, K.; Dimitriou, D.; L’Esperance, V.; Webster, R.; Holloway, J.; Virues-Ortega, J.; et al. Adaptation to life in the High Andes: nocturnal oxyhemoglobin saturation in early development. Sleep 2016, 39, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Monge, C.; Arregui, A.; Leon-Velarde, F. Pathophysiology and epidemiology of chronic mountain sickness. Int. J. Sports Med. 1992, 13, S79–S81. [Google Scholar] [CrossRef] [PubMed]
- Julian, C.G.; Vargas, E.; Gonzales, M.; Davila, R.D.; Ladenburger, A.; Reardon, L.; Schoo, C.; Powers, R.W.; Lee-Chiong, T.; Moore, L.G. Sleep-disordered breathing and oxidative stress in preclinical chronic mountain sickness (excessive erythrocytosis). Respir. Physiol. Neurobiol. 2013, 186, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Julian, C.G.; Gonzales, M.; Rodriguez, A.; Bellido, D.; Salmon, C.S.; Ladenburger, A.; Reardon, L.; Vargas, E.; Moore, L.G. Perinatal hypoxia increases susceptibility to high-altitude polycythemia and attendant pulmonary vascular dysfunction. Am. J. Physiol. Heart Circ. Physiol. 2015, 309, H565–H573. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.Y.; Li, W.; Li, Y.; Ge, R.L.; Cheng, Q.; Wang, S.; Zhao, G.; Wei, L.; Jin, Y.; Don, G. Epidemiology of chronic mountain sickness: Ten years’ study in Qinghai-TIbet. In Progress in Mountain Medicine and High Altitude Physiology; Ohno, H., Kobayashi, T., Masuyama, S., Nakashima, M., Eds.; Press Committee of the 3rd World Congress on Mountain Medicine and High Altitude Physiology: Matsumoto, Japan, 1998; pp. 120–125. [Google Scholar]
- Claydon, V.E.; Gulli, G.; Slessarev, M.; Appenzeller, O.; Zenebe, G.; Gebremedhin, A.; Hainsworth, R. Cerebrovascular responses to hypoxia and hypocapnia in Ethiopian high altitude dwellers. Stroke 2008, 39, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.G.; Niermeyer, S.; Vargas, E. Does chronic mountain sickness (CMS) have perinatal origins? Respir. Physiol. Neurobiol. 2007, 158, 180–189. [Google Scholar] [CrossRef]
- Bailey, D.M.; Rimoldi, S.F.; Rexhaj, E.; Pratali, L.; Salinas Salmon, C.; Villena, M.; McEneny, J.; Young, I.S.; Nicod, P.; Allemann, Y.; et al. Oxidative-nitrosative stress and systemic vascular function in highlanders with and without exaggerated hypoxemia. Chest 2013, 143, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Kryger, M.; Glas, R.; Jackson, D.; McCullough, R.E.; Scoggin, C.; Grover, R.F.; Weil, J.V. Impaired oxygenation during sleep in excessive polycythemia of high altitude: Improvement with respiratory stimulation. Sleep 1978, 1, 3–17. [Google Scholar] [CrossRef]
- Rexhaj, E.; Rimoldi, S.F.; Pratali, L.; Brenner, R.; Andries, D.; Soria, R.; Salinas, C.; Villena, M.; Romero, C.; Allemann, Y.; et al. Sleep-disordered breathing and vascular function in patients with chronic mountain sickness and healthy high-altitude dwellers. Chest 2016, 149, 991–998. [Google Scholar] [CrossRef]
- Reeves, J.T.; Weil, J.V. Chronic mountain sickness. A view from the crow’s nest. Adv. Exp. Med. Biol. 2001, 502, 419–437. [Google Scholar] [PubMed]
- Meadows, G.E.; O’Driscoll, D.M.; Simonds, A.K.; Morrell, M.J.; Corfield, D.R. Cerebral blood flow response to isocapnic hypoxia during slow-wave sleep and wakefulness. J. Appl. Physiol. 2004, 97, 1343–1348. [Google Scholar] [CrossRef] [PubMed]
- Rimoldi, S.F.; Rexhaj, E.; Pratali, L.; Bailey, D.M.; Hutter, D.; Faita, F.; Salmon, C.S.; Villena, M.; Nicod, P.; Allemann, Y.; et al. Systemic vascular dysfunction in patients with chronic mountain sickness. Chest 2012, 141, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Sartori, C.; Allemann, Y.; Trueb, L.; Delabays, A.; Nicod, P.; Scherrer, U. Augmented vasoreactivity in adult life associated with perinatal vascular insult. Lancet 1999, 353, 2205–2207. [Google Scholar] [CrossRef]
- Bigham, A.W.; Wilson, M.J.; Julian, C.G.; Kiyamu, M.; Vargas, E.; Leon-Velarde, F.; Rivera-Chira, M.; Rodriquez, C.; Browne, V.A.; Parra, E.; et al. Andean and Tibetan patterns of adaptation to high altitude. Am. J. Hum. Biol. 2013, 25, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Udpa, N.; Ronen, R.; Stobdan, T.; Liang, J.; Appenzeller, O.; Zhao, H.W.; Yin, Y.; Du, Y.; Guo, L.; et al. Whole-genome sequencing uncovers the genetic basis of chronic mountain sickness in Andean highlanders. Am. J. Hum. Genet. 2013, 93, 452–462. [Google Scholar] [CrossRef] [PubMed]
- Cole, A.M.; Petousi, N.; Cavalleri, G.L.; Robbins, P.A. Genetic variation in SENP1 and ANP32D as predictors of chronic mountain sickness. High Alt. Med. Biol. 2014, 15, 497–499. [Google Scholar] [CrossRef] [PubMed]
- Villafuerte, F.C. New genetic and physiological factors for excessive erythrocytosis and chronic mountain sickness. J. Appl. Physiol. 2015, 119, 1481–1486. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, C.; Bacalini, M.G.; Sazzini, M.; Pirazzini, C.; Franceschi, C.; Garagnani, P.; Luiselli, D. The epigenetic side of human adaptation: Hypotheses, evidences and theories. Ann. Hum. Biol. 2015, 42, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.A.; Baylin, S.B. The fundamental role of epigenetic events in cancer. Nat. Rev. Genet. 2002, 3, 415–428. [Google Scholar] [CrossRef]
- Robertson, K.D. DNA methylation, methyltransferases, and cancer. Oncogene 2001, 20, 3139–3155. [Google Scholar] [CrossRef] [PubMed]
- Illingworth, R.S.; Gruenewald-Schneider, U.; Webb, S.; Kerr, A.R.; James, K.D.; Turner, D.J.; Smith, C.; Harrison, D.J.; Andrews, R.; Bird, A.P. Orphan CpG islands identify numerous conserved promoters in the mammalian genome. PLoS Genet 2010, 6, e1001134. [Google Scholar] [CrossRef] [PubMed]
- Saxonov, S.; Berg, P.; Brutlag, D.L. A genome-wide analysis of CpG dinucleotides in the human genome distinguishes two distinct classes of promoters. Proc. Natl. Acad. Sci. USA 2006, 103, 1412–1417. [Google Scholar] [CrossRef] [PubMed]
- Feinberg, A.P. Phenotypic plasticity and the epigenetics of human disease. Nature 2007, 447, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.A. Functions of DNA methylation: Islands, start sites, gene bodies and beyond. Nat. Rev. Genet. 2012, 13, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Kulis, M.; Heath, S.; Bibikova, M.; Queiros, A.C.; Navarro, A.; Clot, G.; Martinez-Trillos, A.; Castellano, G.; Brun-Heath, I.; Pinyol, M.; et al. Epigenomic analysis detects widespread gene-body DNA hypomethylation in chronic lymphocytic leukemia. Nat. Genet. 2012, 44, 1236–1242. [Google Scholar] [CrossRef]
- Doi, A.; Park, I.H.; Wen, B.; Murakami, P.; Aryee, M.J.; Irizarry, R.; Herb, B.; Ladd-Acosta, C.; Rho, J.; Loewer, S.; et al. Differential methylation of tissue- and cancer-specific CpG island shores distinguishes human induced pluripotent stem cells, embryonic stem cells and fibroblasts. Nat. Genet. 2009, 41, 1350–1353. [Google Scholar] [CrossRef] [PubMed]
- Herman, J.G.; Latif, F.; Weng, Y.; Lerman, M.I.; Zbar, B.; Liu, S.; Samid, D.; Duan, D.S.; Gnarra, J.R.; Linehan, W.M.; et al. Silencing of the VHL tumor-suppressor gene by DNA methylation in renal carcinoma. Proc. Natl. Acad. Sci. USA 1994, 91, 9700–9704. [Google Scholar] [CrossRef]
- Lachance, G.; Uniacke, J.; Audas, T.E.; Holterman, C.E.; Franovic, A.; Payette, J.; Lee, S. DNMT3a epigenetic program regulates the HIF-2α oxygen-sensing pathway and the cellular response to hypoxia. Proc. Natl. Acad. Sci. USA 2014, 111, 7783–7788. [Google Scholar] [CrossRef]
- Yin, H.; Blanchard, K.L. DNA methylation represses the expression of the human erythropoietin gene by two different mechanisms. Blood 2000, 95, 111–119. [Google Scholar]
- Kallio, P.J.; Okamoto, K.; O’Brien, S.; Carrero, P.; Makino, Y.; Tanaka, H.; Poellinger, L. Signal transduction in hypoxic cells: Inducible nuclear translocation and recruitment of the CBP/p300 coactivator by the hypoxia-inducible factor-1alpha. EMBO J. 1998, 17, 6573–6586. [Google Scholar] [CrossRef] [PubMed]
- Wellmann, S.; Bettkober, M.; Zelmer, A.; Seeger, K.; Faigle, M.; Eltzschig, H.K.; Buhrer, C. Hypoxia upregulates the histone demethylase JMJD1A via HIF-1. Biochem. Biophys. Res. Commun. 2008, 372, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Watson, J.A.; Watson, C.J.; McCrohan, A.M.; Woodfine, K.; Tosetto, M.; McDaid, J.; Gallagher, E.; Betts, D.; Baugh, J.; O’Sullivan, J.; et al. Generation of an epigenetic signature by chronic hypoxia in prostate cells. Hum. Mol. Genet. 2009, 18, 3594–3604. [Google Scholar] [CrossRef] [PubMed]
- Bonasio, R.; Tu, S.; Reinberg, D. Molecular signals of epigenetic states. Science 2010, 330, 612–616. [Google Scholar] [CrossRef] [PubMed]
- Reik, W. Stability and flexibility of epigenetic gene regulation in mammalian development. Nature 2007, 447, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Khavari, D.A.; Sen, G.L.; Rinn, J.L. DNA methylation and epigenetic control of cellular differentiation. Cell Cycle 2010, 9, 3880–3883. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Xiao, Y.; Estrella, J.L.; Ducsay, C.A.; Gilbert, R.D.; Zhang, L. Effect of fetal hypoxia on heart susceptibility to ischemia and reperfusion injury in the adult rat. J. Soc. Gynecol. Investig. 2003, 10, 265–274. [Google Scholar] [CrossRef]
- Xue, Q.; Zhang, L. Prenatal hypoxia causes a sex-dependent increase in heart susceptibility to ischemia and reperfusion injury in adult male offspring: Role of protein kinase Cε. J. Pharmacol. Exp. Ther. 2009, 330, 624–632. [Google Scholar] [CrossRef]
- Patterson, A.J.; Chen, M.; Xue, Q.; Xiao, D.; Zhang, L. Chronic prenatal hypoxia induces epigenetic programming of PKCε gene repression in rat hearts. Circ. Res. 2010, 107, 365–373. [Google Scholar] [CrossRef]
- Rexhaj, E.; Bloch, J.; Jayet, P.Y.; Rimoldi, S.F.; Dessen, P.; Mathieu, C.; Tolsa, J.F.; Nicod, P.; Scherrer, U.; Sartori, C. Fetal programming of pulmonary vascular dysfunction in mice: Role of epigenetic mechanisms. Am. J. Physiol. Heart Circ. Physiol. 2011, 301, H247–H252. [Google Scholar] [CrossRef]
- Jayet, P.Y.; Rimoldi, S.F.; Stuber, T.; Salmon, C.S.; Hutter, D.; Rexhaj, E.; Thalmann, S.; Schwab, M.; Turini, P.; Sartori-Cucchia, C.; et al. Pulmonary and systemic vascular dysfunction in young offspring of mothers with preeclampsia. Circulation 2010, 122, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Smith, Z.D.; Chan, M.M.; Humm, K.C.; Karnik, R.; Mekhoubad, S.; Regev, A.; Eggan, K.; Meissner, A. DNA methylation dynamics of the human preimplantation embryo. Nature 2014, 511, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Roemer, I.; Reik, W.; Dean, W.; Klose, J. Epigenetic inheritance in the mouse. Curr. Biol. 1997, 7, 277–280. [Google Scholar] [CrossRef]
- Anway, M.D.; Cupp, A.S.; Uzumcu, M.; Skinner, M.K. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 2005, 308, 1466–1469. [Google Scholar] [CrossRef] [PubMed]
- Hitchins, M.P.; Ward, R.L. Constitutional (germline) MLH1 epimutation as an aetiological mechanism for hereditary non-polyposis colorectal cancer. J. Med. Genet. 2009, 46, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Yan, W.; Duan, E. Epigenetic inheritance of acquired traits through sperm RNAs and sperm RNA modifications. Nat. Rev. Genet. 2016, 17, 733–743. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, J.R.; van der Brug, M.P.; Hernandez, D.G.; Traynor, B.J.; Nalls, M.A.; Lai, S.L.; Arepalli, S.; Dillman, A.; Rafferty, I.P.; Troncoso, J.; et al. Abundant quantitative trait loci exist for DNA methylation and gene expression in human brain. PLoS Genet 2010, 6, e1000952. [Google Scholar] [CrossRef]
- Bell, J.T.; Pai, A.A.; Pickrell, J.K.; Gaffney, D.J.; Pique-Regi, R.; Degner, J.F.; Gilad, Y.; Pritchard, J.K. DNA methylation patterns associate with genetic and gene expression variation in HapMap cell lines. Genome Biol. 2011, 12, R10. [Google Scholar] [CrossRef]
- Moen, E.L.; Zhang, X.; Mu, W.; Delaney, S.M.; Wing, C.; McQuade, J.; Myers, J.; Godley, L.A.; Dolan, M.E.; Zhang, W. Genome-wide variation of cytosine modifications between European and African populations and the implications for complex traits. Genetics 2013, 194, 987–996. [Google Scholar] [CrossRef]
- Zhang, X.; Moen, E.L.; Liu, C.; Mu, W.; Gamazon, E.R.; Delaney, S.M.; Wing, C.; Godley, L.A.; Dolan, M.E.; Zhang, W. Linking the genetic architecture of cytosine modifications with human complex traits. Hum. Mol. Genet. 2014, 23, 5893–5905. [Google Scholar] [CrossRef]
- McClay, J.L.; Shabalin, A.A.; Dozmorov, M.G.; Adkins, D.E.; Kumar, G.; Nerella, S.; Clark, S.L.; Bergen, S.E.; Swedish Schizophrenia, C.; Hultman, C.M.; et al. High density methylation QTL analysis in human blood via next-generation sequencing of the methylated genomic DNA fraction. Genome Biol. 2015, 16, 291. [Google Scholar] [CrossRef] [PubMed]
- Zhi, D.; Aslibekyan, S.; Irvin, M.R.; Claas, S.A.; Borecki, I.B.; Ordovas, J.M.; Absher, D.M.; Arnett, D.K. SNPs located at CpG sites modulate genome-epigenome interaction. Epigenetics 2013, 8, 802–806. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.J.; Rupert, J.L. Hypoxia and environmental epigenetics. High Alt. Med. Biol. 2014, 15, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Scherrer, U.; Allemann, Y.; Rexhaj, E.; Rimoldi, S.F.; Sartori, C. Mechanisms and drug therapy of pulmonary hypertension at high altitude. High Alt. Med. Biol. 2013, 14, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Alkorta-Aranburu, G.; Beall, C.M.; Witonsky, D.B.; Gebremedhin, A.; Pritchard, J.K.; Di Rienzo, A. The genetic architecture of adaptations to high altitude in Ethiopia. PLoS Genet 2012, 8, e1003110. [Google Scholar] [CrossRef] [PubMed]
- Julian, C.G.; Pedersen, B.S.; Salmon, C.S.; Yang, I.V.; Gonzales, M.; Vargas, E.; Moore, L.G.; Schwartz, D.A. Unique DNA methylation patterns in offspring of hypertensive pregnancy. Clin. Transl. Sci. 2015, 8, 740–745. [Google Scholar] [CrossRef] [PubMed]
- Julian, C.G. Epigenomics and human adaptation to high altitude. J. Appl. Physiol. 2017, 123, 1362–1370. [Google Scholar] [CrossRef]
- Beall, C.M.; Cavalleri, G.L.; Deng, L.; Elston, R.C.; Gao, Y.; Knight, J.; Li, C.; Li, J.C.; Liang, Y.; McCormack, M.; et al. Natural selection on EPAS1 (HIF2α) associated with low hemoglobin concentration in Tibetan highlanders. Proc. Natl. Acad. Sci. USA 2010, 107, 11459–11464. [Google Scholar] [CrossRef]
- Minamishima, Y.A.; Moslehi, J.; Bardeesy, N.; Cullen, D.; Bronson, R.T.; Kaelin, W.G., Jr. Somatic inactivation of the PHD2 prolyl hydroxylase causes polycythemia and congestive heart failure. Blood 2008, 111, 3236–3244. [Google Scholar] [CrossRef]
- Takeda, K.; Aguila, H.L.; Parikh, N.S.; Li, X.; Lamothe, K.; Duan, L.J.; Takeda, H.; Lee, F.S.; Fong, G.H. Regulation of adult erythropoiesis by prolyl hydroxylase domain proteins. Blood 2008, 111, 3229–3235. [Google Scholar] [CrossRef]
- Percy, M.J.; Zhao, Q.; Flores, A.; Harrison, C.; Lappin, T.R.; Maxwell, P.H.; McMullin, M.F.; Lee, F.S. A family with erythrocytosis establishes a role for prolyl hydroxylase domain protein 2 in oxygen homeostasis. Proc. Natl. Acad. Sci. USA 2006, 103, 654–659. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, Y.; Ren, L.H.; Li, L.; Wang, Z.; Liu, S.S.; Li, S.Z.; Cao, T.S. Effects of race and sex on cerebral hemodynamics, oxygen delivery and blood flow distribution in response to high altitude. Sci. Rep. 2016, 6, 30500. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, D.L.; Le Lay, J.E.; Ruano, E.G.; Kaestner, K.H. TALE-mediated epigenetic suppression of CDKN2A increases replication in human fibroblasts. J. Clin. Investig. 2015, 125, 1998–2006. [Google Scholar] [CrossRef] [PubMed]
- Maeder, M.L.; Angstman, J.F.; Richardson, M.E.; Linder, S.J.; Cascio, V.M.; Tsai, S.Q.; Ho, Q.H.; Sander, J.D.; Reyon, D.; Bernstein, B.E.; et al. Targeted DNA demethylation and activation of endogenous genes using programmable TALE-TET1 fusion proteins. Nat. Biotechnol. 2013, 31, 1137–1142. [Google Scholar] [CrossRef] [PubMed]

| Variable | Acclimatized Newcomer | Andean | Andean versus Accl newcomer |
|---|---|---|---|
| PACO2, mmHg | 30 [8] | Higher [8,32] | ↑ω |
| A-a O2, mmHg | 7–11 [14,15] | Lower [4,15] | ↓ |
| SaO2, % | 92 [16,19,37] | Same [9,37,38,39,40] | ≅ |
| Hemoglobin, g/dL | 17.6 [16,19] | Same [9,17,21] | ≅ |
| CaO2, vol%11 | 21 [16,19] | Same [19] | ≅ |
| Ppa hypoxic response | Present | Intermediate [41] | ↓ |
| Brain blood flow velocity, cm/s | 27 [42] | 18% [39] | ↓ |
| Uterine artery blood flow, mL/min | 269 [43] | Higher [43] | ↑ |
| AS3MT [72] BRINP3 [73] CLC [72] DUOX2 [72] | EDNRA [71,74] EGLN1 [71,74] ELTD1 [75] ET-1 [76] FAM213A [77] | NOS2 [71,73,74] PRKAA1 [71,74] SFTPD [77] SP100 [72] | TBX5 [73] TMEM38B [72] TP53 pathway [78] VEGFB [75] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Julian, C.G.; Moore, L.G. Human Genetic Adaptation to High Altitude: Evidence from the Andes. Genes 2019, 10, 150. https://doi.org/10.3390/genes10020150
Julian CG, Moore LG. Human Genetic Adaptation to High Altitude: Evidence from the Andes. Genes. 2019; 10(2):150. https://doi.org/10.3390/genes10020150
Chicago/Turabian StyleJulian, Colleen G., and Lorna G. Moore. 2019. "Human Genetic Adaptation to High Altitude: Evidence from the Andes" Genes 10, no. 2: 150. https://doi.org/10.3390/genes10020150
APA StyleJulian, C. G., & Moore, L. G. (2019). Human Genetic Adaptation to High Altitude: Evidence from the Andes. Genes, 10(2), 150. https://doi.org/10.3390/genes10020150





