Massive Loss of Olfactory Receptors But Not Trace Amine-Associated Receptors in the World’s Deepest-Living Fish (Pseudoliparis swirei)
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source and Sequence Data Mining
2.2. Phylogenetic and Orthologous Group Analysis
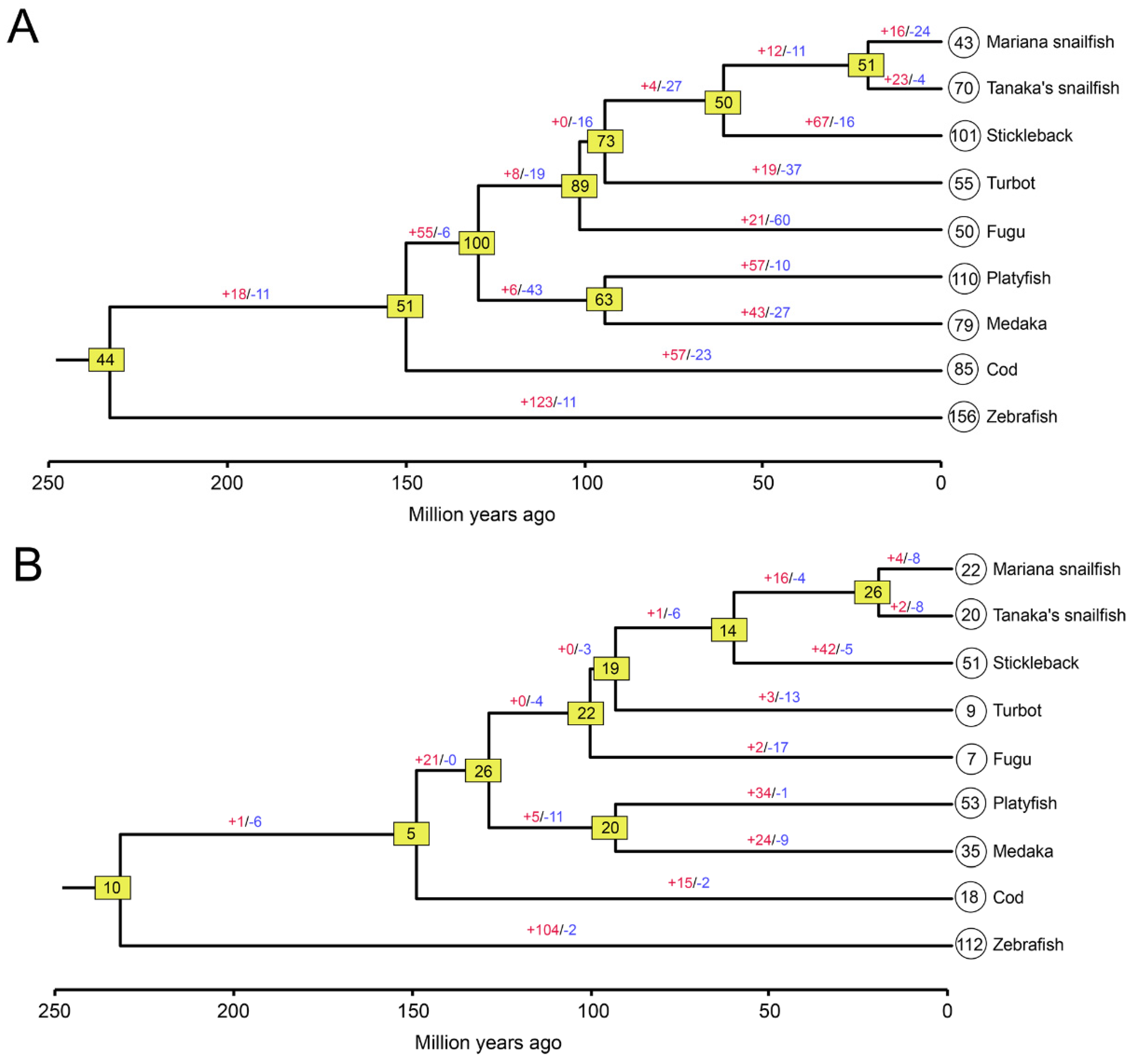
2.3. Estimation of Gene Gain and Loss Events
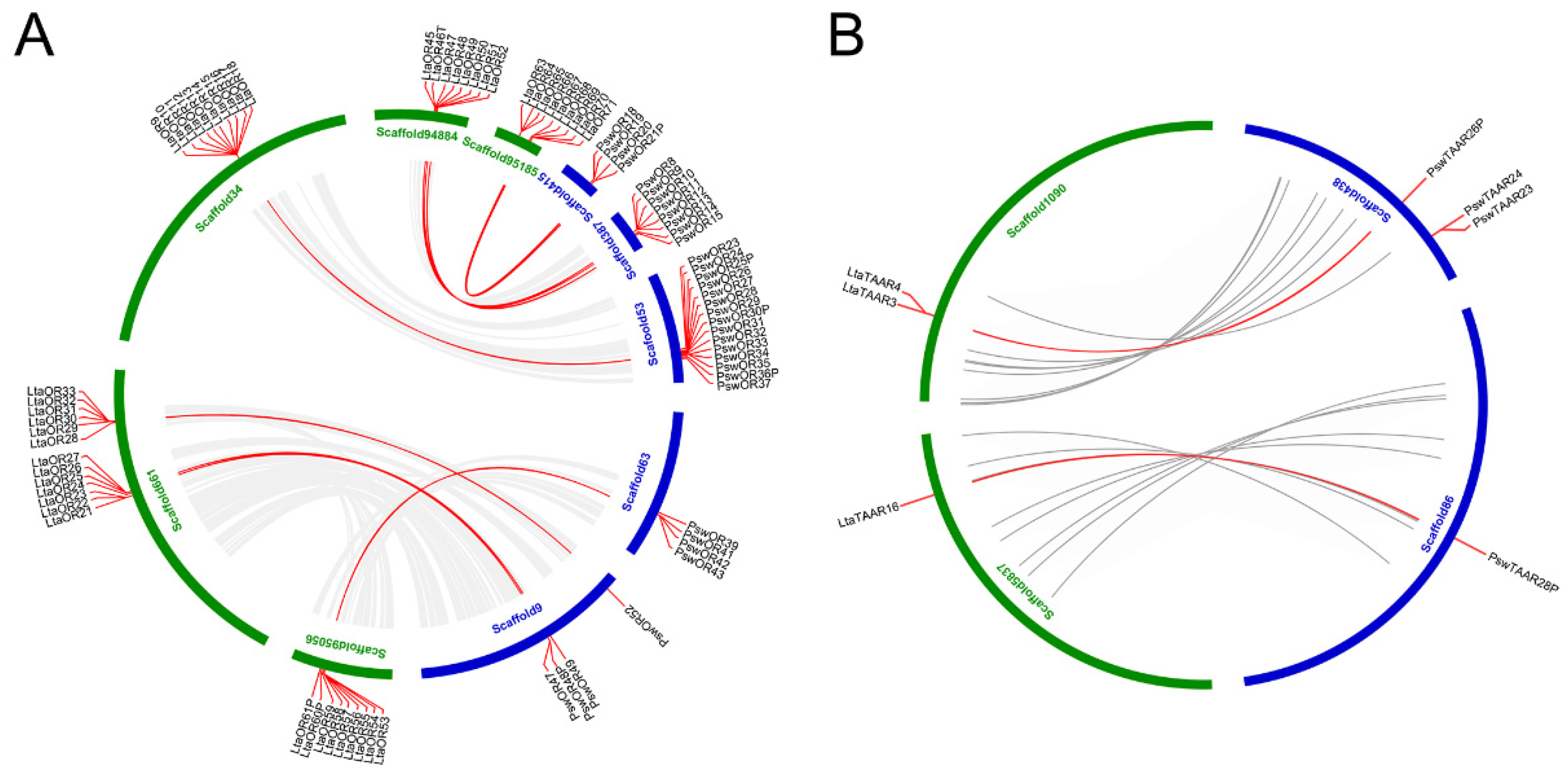
2.4. Chromosomal Distribution and Gene Duplication
2.5. Selection Analysis
3. Results
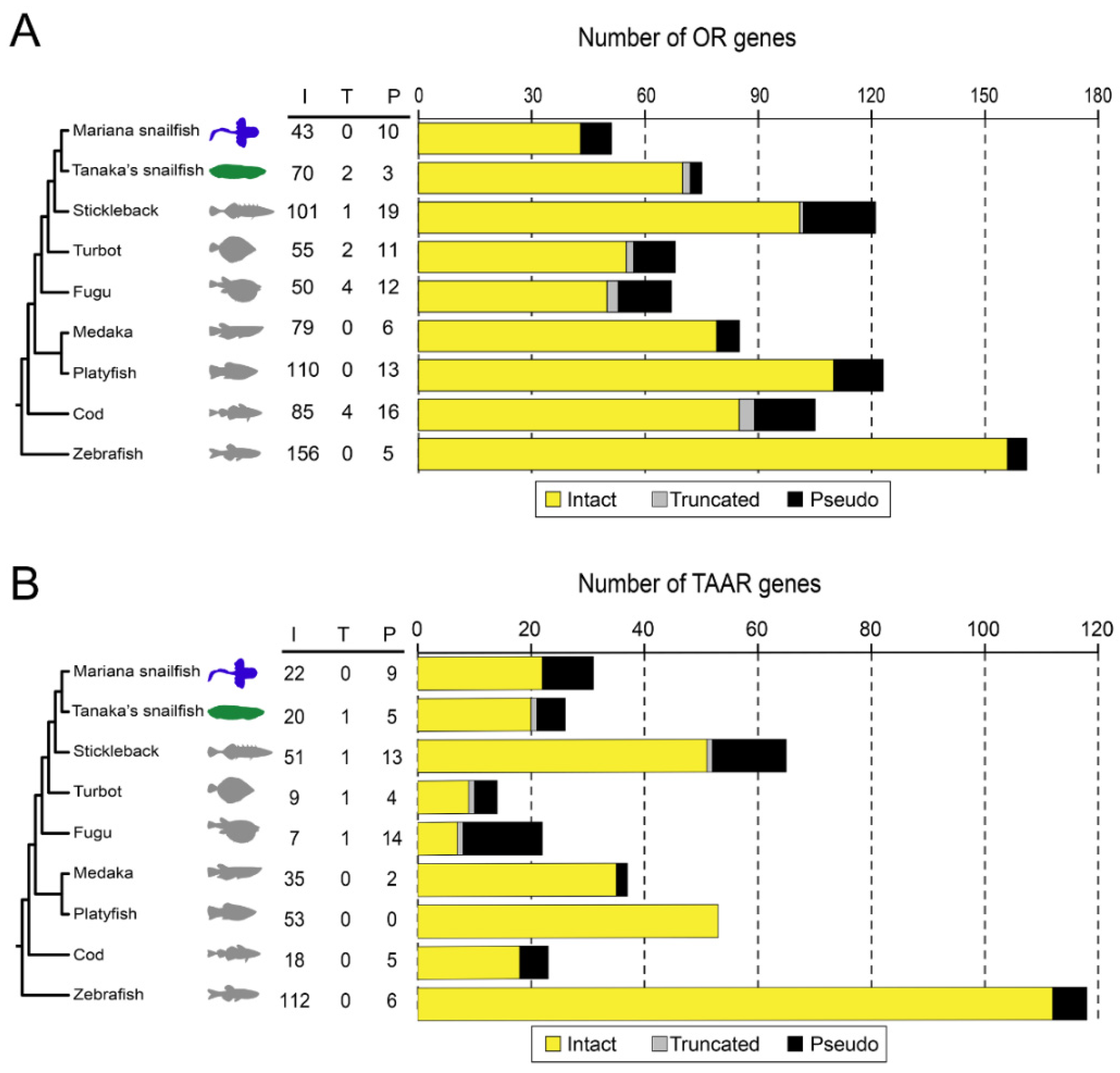
3.1. Substantially Reduced Number of OR Genes in the Mariana Snailfish Caused by Pseudogenization
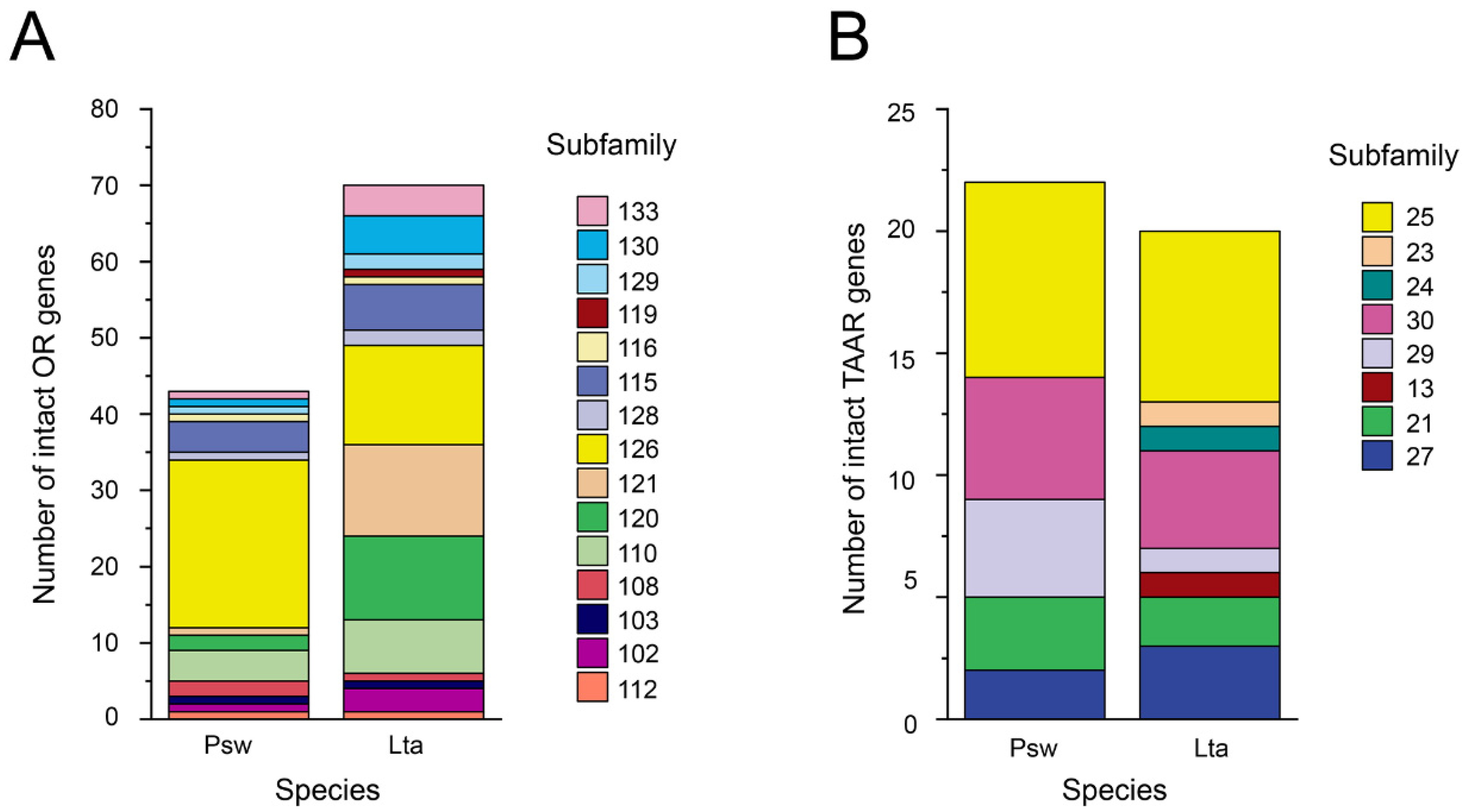
3.2. An Overall Decreased Size of OR Gene Families and a Reduced Number of TAAR Gene Families in the Mariana Snailfish Compared to Tanaka’s Snailfish
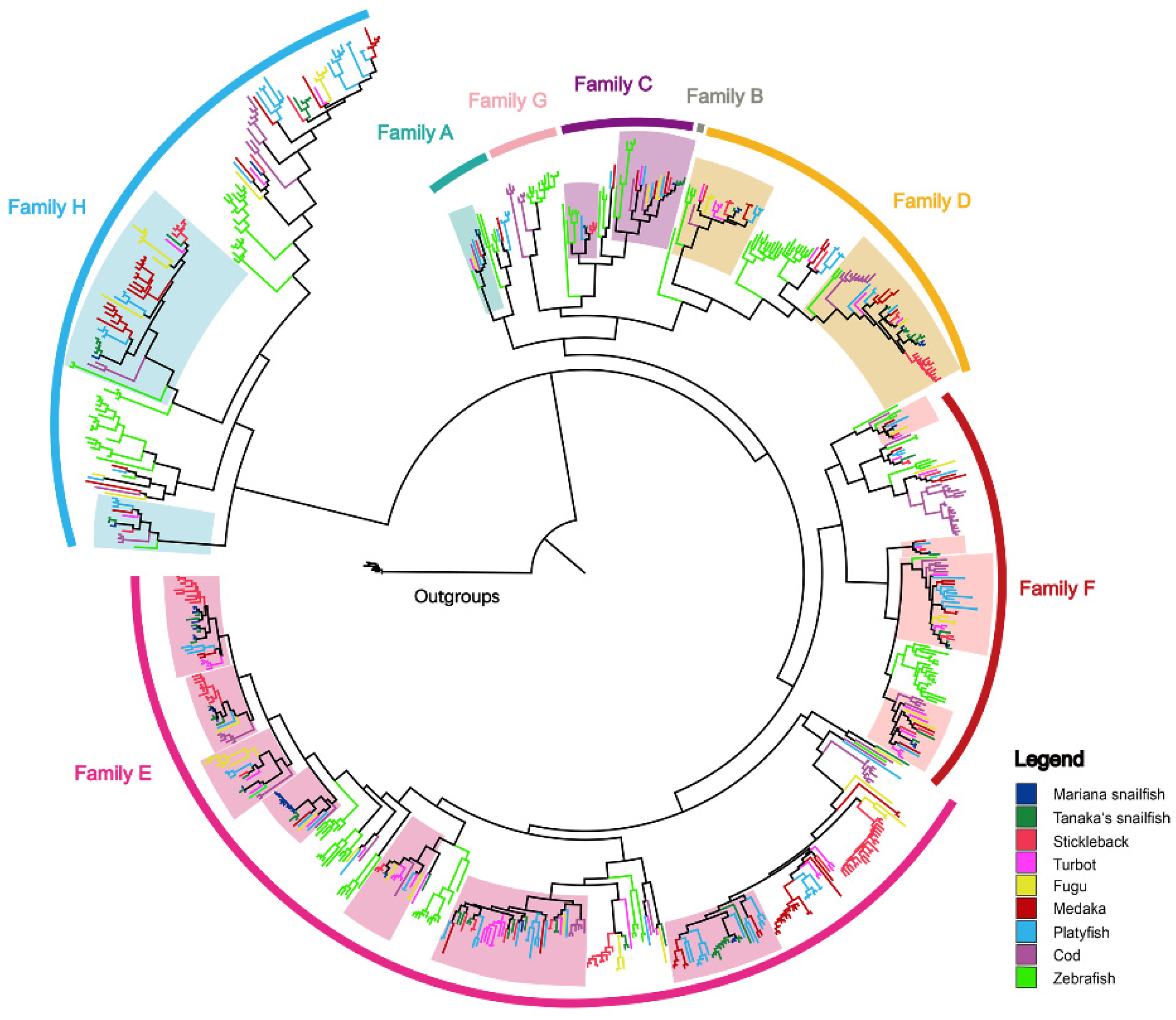
3.3. Highly Dynamic Gene Birth and Death Shaped the OR and TAAR Evolution
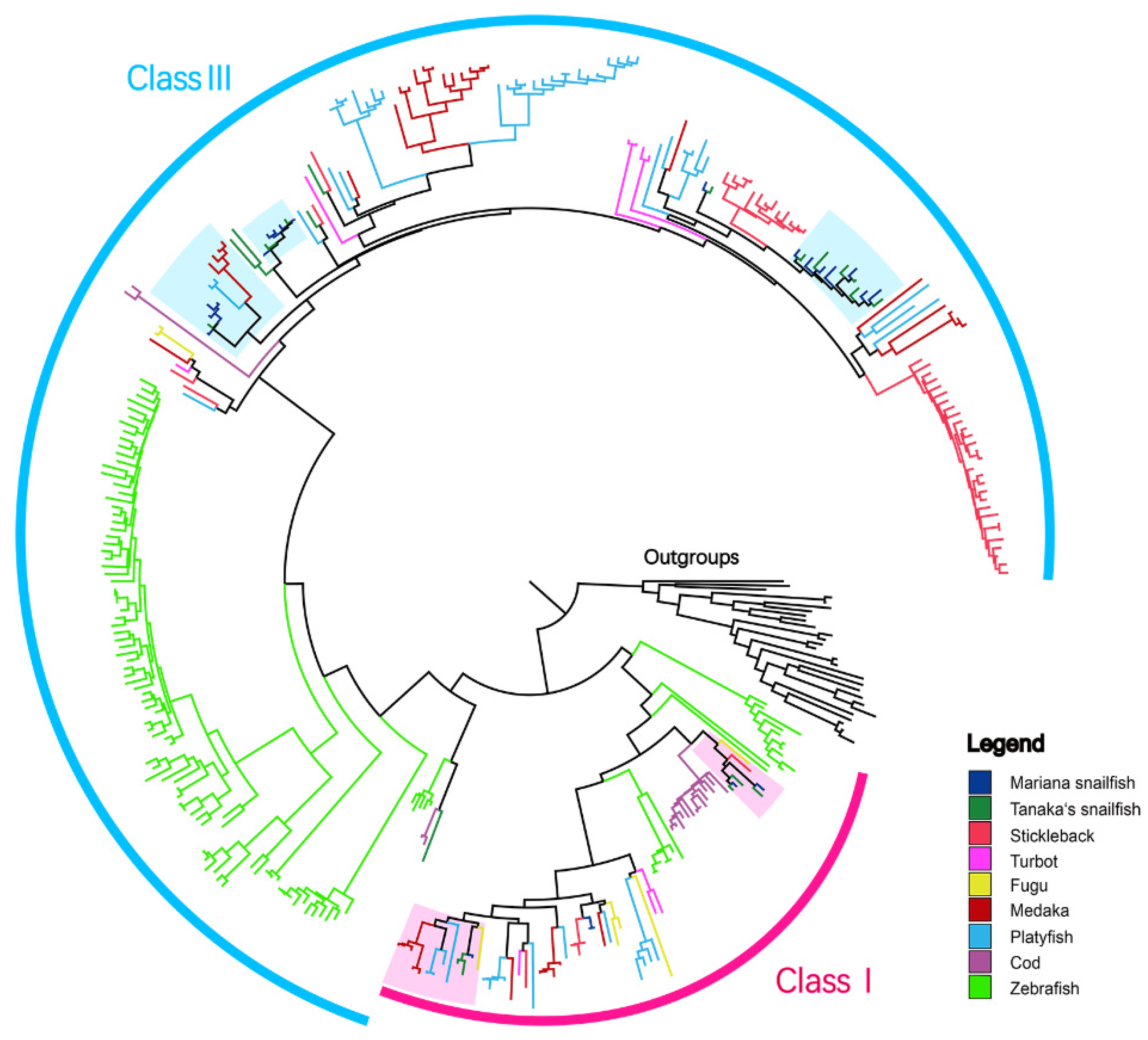
3.4. Genomic Organization Reveals More Orthologous Regions in ORs than in TAARs
3.5. Collinear Analysis Reveals Orthologous Pairs Formed Prior to the Divergence of the Mariana Snailfish and Tanaka’s Snailfish
3.6. Strong Relaxed Selective Strength in ORs But Slightly Enhanced Selective Strength in TAARs of the Mariana Snailfish
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Miyasaka, N.; Arganda-Carreras, I.; Wakisaka, N.; Masuda, M.; Sumbul, U.; Seung, H.S.; Yoshihara, Y. Olfactory projectome in the zebrafish forebrain revealed by genetic single-neuron labelling. Nat. Commun. 2014, 5, 3639. [Google Scholar] [CrossRef]
- Niimura, Y.; Matsui, A.; Touhara, K. Extreme expansion of the olfactory receptor gene repertoire in African elephants and evolutionary dynamics of orthologous gene groups in 13 placental mammals. Genome Res. 2014, 24, 1485–1496. [Google Scholar] [CrossRef]
- Niimura, Y. Olfactory Receptor Multigene Family in Vertebrates: From the Viewpoint of Evolutionary Genomics. Curr. Genom. 2012, 13, 103–114. [Google Scholar] [CrossRef]
- Chess, A.; Simon, I.; Cedar, H.; Axel, R. Allelic Inactivation Regulates Olfactory Receptor Gene-Expression. Cell 1994, 78, 823–834. [Google Scholar] [CrossRef]
- Yabuki, Y.; Koide, T.; Miyasaka, N.; Wakisaka, N.; Masuda, M.; Ohkura, M.; Nakai, J.; Tsuge, K.; Tsuchiya, S.; Sugimoto, Y.; et al. Olfactory receptor for prostaglandin F2α mediates male fish courtship behavior. Nat. Neurosci. 2016, 19, 897–904. [Google Scholar] [CrossRef]
- Lindemann, L.; Hoener, M.C. A renaissance in trace amines inspired by a novel GPCR family. Trends Pharmacol. Sci. 2005, 26, 274–281. [Google Scholar] [CrossRef]
- Hussain, A.; Saraiva, L.R.; Korsching, S.I. Positive Darwinian selection and the birth of an olfactory receptor clade in teleosts. Proc. Natl. Acad. Sci. USA 2009, 106, 4313–4318. [Google Scholar] [CrossRef]
- Li, Q.; Tachie-Baffour, Y.; Liu, Z.; Baldwin, M.W.; Kruse, A.C.; Liberles, S.D. Non-classical amine recognition evolved in a large clade of olfactory receptors. eLife 2015, 4, e10441. [Google Scholar] [CrossRef]
- Nei, M.; Niimura, Y.; Nozawa, M. The evolution of animal chemosensory receptor gene repertoires: Roles of chance and necessity. Nat. Rev. Genet. 2008, 9, 951–963. [Google Scholar] [CrossRef]
- Niimura, Y. On the origin and evolution of vertebrate olfactory receptor genes: Comparative genome analysis among 23 chordate species. Genome Biol. Evol. 2009, 1, 34–44. [Google Scholar] [CrossRef]
- Hughes, G.M.; Boston, E.S.M.; Finarelli, J.A.; Murphy, W.J.; Higgins, D.G.; Teeling, E.C. The Birth and Death of Olfactory Receptor Gene Families in Mammalian Niche Adaptation. Mol. Biol. Evol. 2018, 35, 1390–1406. [Google Scholar] [CrossRef] [PubMed]
- Niimura, Y.; Matsui, A.; Touhara, K. Acceleration of Olfactory Receptor Gene Loss in Primate Evolution: Possible Link to Anatomical Change in Sensory Systems and Dietary Transition. Mol. Biol. Evol. 2018, 35, 1437–1450. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Yang, Z.; Maldonado, E.; Li, C.; Zhang, G.; Gilbert, M.T.; Jarvis, E.D.; O’Brien, S.J.; Johnson, W.E.; Antunes, A. Olfactory Receptor Subgenomes Linked with Broad Ecological Adaptations in Sauropsida. Mol. Biol. Evol. 2015, 32, 2832–2843. [Google Scholar] [CrossRef]
- Goldman-Huertas, B.; Mitchell, R.F.; Lapoint, R.T.; Faucher, C.P.; Hildebrand, J.G.; Whiteman, N.K. Evolution of herbivory in Drosophilidae linked to loss of behaviors, antennal responses, odorant receptors, and ancestral diet. Proc. Natl. Acad. Sci. USA 2015, 112, 3026–3031. [Google Scholar] [CrossRef]
- Slone, J.D.; Pask, G.M.; Ferguson, S.T.; Millar, J.G.; Berger, S.L.; Reinberg, D.; Liebig, J.; Ray, A.; Zwiebel, L.J. Functional characterization of odorant receptors in the ponerine ant, Harpegnathos saltator. Proc. Natl. Acad. Sci. USA 2017, 114, 8586–8591. [Google Scholar] [CrossRef]
- Pellegrino, M.; Steinbach, N.; Stensmyr, M.C.; Hansson, B.S.; Vosshall, L.B. A natural polymorphism alters odour and DEET sensitivity in an insect odorant receptor. Nature 2011, 478, 511–514. [Google Scholar] [CrossRef]
- Jamieson, A.J.; Fujii, T.; Mayor, D.J.; Solan, M.; Priede, I.G. Hadal trenches: The ecology of the deepest places on Earth. Trends Ecol. Evol. 2010, 25, 190–197. [Google Scholar] [CrossRef]
- Gerringer, M.E.; Andrews, A.H.; Huss, G.R.; Nagashima, K.; Popp, B.N.; Linley, T.D.; Gallo, N.D.; Clark, M.R.; Jamieson, A.J.; Drazen, J.C. Life history of abyssal and hadal fishes from otolith growth zones and oxygen isotopic compositions. Deep Sea Res. Part I 2018, 132, 37–50. [Google Scholar] [CrossRef]
- Shen, Y.J.; Dai, W.; Gao, Z.M.; Yan, G.Y.; Gan, X.N.; He, S.P. Molecular phylogeny and divergence time estimates using the mitochondrial genome for the hadal snailfish from the Mariana trench. Sci. Bull. 2017, 62, 1106–1108. [Google Scholar] [CrossRef]
- Wang, K.; Shen, Y.; Yang, Y.; Gan, X.; Liu, G.; Hu, K.; Li, Y.; Gao, Z.; Zhu, L.; Yan, G.; et al. Morphology and genome of a snailfish from the Mariana Trench provide insights into deep-sea adaptation. Nat. Ecol. Evol. 2019, 3, 823–833. [Google Scholar] [CrossRef]
- Linley, T.D.; Stewart, A.L.; McMillan, P.J.; Clark, M.R.; Gerringer, M.E.; Drazen, J.C.; Fujii, T.; Jamieson, A.J. Bait attending fishes of the abyssal zone and hadal boundary: Community structure, functional groups and species distribution in the Kermadec, New Hebrides and Mariana trenches. Deep Sea Res. Part I 2017, 121, 38–53. [Google Scholar] [CrossRef]
- Fujita, T.; Kitagawa, D.; Okuyama, Y.; Ishito, Y.; Inada, T.; Jin, Y. Diets of the Demersal Fishes on the Shelf Off Iwate, Northern Japan. Mar. Biol. 1995, 123, 219–233. [Google Scholar] [CrossRef]
- Blin, M.; Tine, E.; Meister, L.; Elipot, Y.; Bibliowicz, J.; Espinasa, L.; Retaux, S. Developmental evolution and developmental plasticity of the olfactory epithelium and olfactory skills in Mexican cavefish. Dev. Biol. 2018, 441, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Shiriagin, V.; Korsching, S.I. Massive Expansion of Bitter Taste Receptors in Blind Cavefish, Astyanax mexicanus. Chem. Senses 2019, 44, 23–32. [Google Scholar] [CrossRef]
- Lisney, T.J.; Wagner, H.J.; Collin, S.P. Ontogenetic Shifts in the Number of Axons in the Olfactory Tract and Optic Nerve in Two Species of Deep-Sea Grenadier Fish (Gadiformes: Macrouridae: Coryphaenoides). Front. Ecol. Evol. 2018, 6, 168. [Google Scholar] [CrossRef]
- Johnson, M.; Zaretskaya, I.; Raytselis, Y.; Merezhuk, Y.; McGinnis, S.; Madden, T.L. NCBI BLAST: A better web interface. Nucleic Acids Res. 2008, 36, 5–9. [Google Scholar] [CrossRef]
- Flicek, P.; Amode, M.R.; Barrell, D.; Beal, K.; Billis, K.; Brent, S.; Carvalho-Silva, D.; Clapham, P.; Coates, G.; Fitzgerald, S.; et al. Ensembl 2014. Nucleic Acids Res. 2014, 42, 749–755. [Google Scholar] [CrossRef]
- Birney, E.; Clamp, M.; Durbin, R. GeneWise and genomewise. Genome Res. 2004, 14, 988–995. [Google Scholar] [CrossRef]
- Krogh, A.; Larsson, B.; von Heijne, G.; Sonnhammer, E.L. Predicting transmembrane protein topology with a hidden Markov model: Application to complete genomes. J. Mol. Biol. 2001, 305, 567–580. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT Multiple Sequence Alignment Software Version 7: Improvements in Performance and Usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Waterhouse, A.M.; Procter, J.B.; Martin, D.M.A.; Clamp, M.; Barton, G.J. Jalview Version 2-a multiple sequence alignment editor and analysis workbench. Bioinformatics 2009, 25, 1189–1191. [Google Scholar] [CrossRef] [PubMed]
- Dereeper, A.; Audic, S.; Claverie, J.M.; Blanc, G. BLAST-EXPLORER helps you building datasets for phylogenetic analysis. BMC Evol. Biol. 2010, 10, 8. [Google Scholar] [CrossRef] [PubMed]
- Dereeper, A.; Guignon, V.; Blanc, G.; Audic, S.; Buffet, S.; Chevenet, F.; Dufayard, J.F.; Guindon, S.; Lefort, V.; Lescot, M.; et al. Phylogeny.fr: Robust phylogenetic analysis for the non-specialist. Nucleic Acids Res. 2008, 36, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Dufayard, J.F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New Algorithms and Methods to Estimate Maximum-Likelihood Phylogenies: Assessing the Performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef]
- Stover, B.C.; Muller, K.F. TreeGraph 2: Combining and visualizing evidence from different phylogenetic analyses. BMC Bioinform. 2010, 11, 7. [Google Scholar] [CrossRef]
- Alioto, T.S.; Ngai, J. The repertoire of olfactory C family G protein-coupled receptors in zebrafish: Candidate chemosensory receptors for amino acids. BMC Genom. 2006, 7, 309. [Google Scholar] [CrossRef]
- Darby, C.A.; Stolzer, M.; Ropp, P.J.; Barker, D.; Durand, D. Xenolog classification. Bioinformatics 2017, 33, 640–649. [Google Scholar] [CrossRef]
- Figueras, A.; Robledo, D.; Corvelo, A.; Hermida, M.; Pereiro, P.; Rubiolo, J.A.; Gomez-Garrido, J.; Carrete, L.; Bello, X.; Gut, M.; et al. Whole genome sequencing of turbot (Scophthalmus maximus; Pleuronectiformes): A fish adapted to demersal life. DNA Res. 2016, 23, 181–192. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, H.; Debarry, J.D.; Tan, X.; Li, J.; Wang, X.; Lee, T.H.; Jin, H.; Marler, B.; Guo, H.; et al. MCScanX: A toolkit for detection and evolutionary analysis of gene synteny and collinearity. Nucleic Acids Res. 2012, 40, e49. [Google Scholar] [CrossRef]
- Chen, C.; Xia, R.; Chen, H.; He, Y. TBtools, a Toolkit for Biologists integrating various HTS-data handling tools with a user-friendly interface. bioRxiv 2018, 289660. [Google Scholar] [CrossRef]
- Xu, L.; Dong, Z.; Fang, L.; Luo, Y.; Wei, Z.; Guo, H.; Zhang, G.; Gu, Y.Q.; Coleman-Derr, D.; Xia, Q.; et al. OrthoVenn2: A web server for whole-genome comparison and annotation of orthologous clusters across multiple species. Nucleic Acids Res. 2019, 47, W53–W258. [Google Scholar] [CrossRef] [PubMed]
- Pond, S.L.; Frost, S.D.; Muse, S.V.J.B. HyPhy: Hypothesis testing using phylogenies. Bioinformatics 2005, 21, 676–679. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.D.; Wertheim, J.O.; Weaver, S.; Murrell, B.; Scheffler, K.; Kosakovsky Pond, S.L. Less is more: An adaptive branch-site random effects model for efficient detection of episodic diversifying selection. Mol. Biol. Evol. 2015, 32, 1342–1353. [Google Scholar] [CrossRef] [PubMed]
- Wertheim, J.O.; Murrell, B.; Smith, M.D.; Kosakovsky Pond, S.L.; Scheffler, K. RELAX: Detecting relaxed selection in a phylogenetic framework. Mol. Biol. Evol. 2015, 32, 820–832. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Gojobori, T. A method for detecting positive selection at single amino acid sites. Mol. Biol. Evol. 1999, 16, 1315–1328. [Google Scholar] [CrossRef]
- Delport, W.; Poon, A.F.; Frost, S.D.; Kosakovsky Pond, S.L. Datamonkey 2010: A suite of phylogenetic analysis tools for evolutionary biology. Bioinformatics 2010, 26, 2455–2457. [Google Scholar] [CrossRef]
- Yohe, L.R.; Brand, P.; Handling editor: Rebecca, F. Evolutionary ecology of chemosensation and its role in sensory drive. Curr. Zool. 2018, 64, 525–533. [Google Scholar] [CrossRef]
- Bear, D.M.; Lassance, J.M.; Hoekstra, H.E.; Datta, S.R. The Evolving Neural and Genetic Architecture of Vertebrate Olfaction. Curr. Biol. 2016, 26, R1039–R1049. [Google Scholar] [CrossRef]
- Kishida, T.; Kubota, S.; Shirayama, Y.; Fukami, H. The olfactory receptor gene repertoires in secondary-adapted marine vertebrates: Evidence for reduction of the functional proportions in cetaceans. Biol. Lett. 2007, 3, 428–430. [Google Scholar] [CrossRef]
- Blankenship, L.E.; Levin, L.A. Extreme food webs: Foraging strategies and diets of scavenging amphipods from the ocean’s deepest 5 kilometers. Limnol. Oceanogr. 2007, 52, 1685–1697. [Google Scholar] [CrossRef]
- Kajiya, K.; Inaki, K.; Tanaka, M.; Haga, T.; Kataoka, H.; Touhara, K. Molecular bases of odor discrimination: Reconstitution of olfactory receptors that recognize overlapping sets of odorants. J. Neurosci. 2001, 21, 6018–6025. [Google Scholar] [CrossRef] [PubMed]
- Lv, L.Y.; Liang, X.F.; He, S. Genome-Wide Identification and Characterization of Olfactory Receptor Genes in Chinese Perch, Siniperca chuatsi. Genes 2019, 10, 178. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.H.; Zhang, X.M.; Firestein, S. Comparative genomics of odorant and pheromone receptor genes in rodents. Genomics 2007, 89, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.T.; Lee, K.; Choi, H.; Choi, M.K.; Le, M.T.; Song, N.; Kim, J.H.; Seo, H.G.; Oh, J.W.; Lee, K.; et al. The complete swine olfactory subgenome: Expansion of the olfactory gene repertoire in the pig genome. BMC Genom. 2012, 13. [Google Scholar] [CrossRef]
- Malnic, B.; Godfrey, P.A.; Buck, L.B. The human olfactory receptor gene family. Proc. Natl. Acad. Sci. USA 2004, 101, 2584–2589. [Google Scholar] [CrossRef]
- Gao, S.; Liu, S.; Yao, J.; Li, N.; Yuan, Z.; Zhou, T.; Li, Q.; Liu, Z. Genomic organization and evolution of olfactory receptors and trace amine-associated receptors in channel catfish, Ictalurus punctatus. Biochim. Biophys. Acta. Gen. Subj. 2017, 1861, 644–651. [Google Scholar] [CrossRef]
- Tessarolo, J.A.; Tabesh, M.J.; Nesbitt, M.; Davidson, W.S. Genomic organization and evolution of the trace amine-associated receptor (TAAR) repertoire in Atlantic salmon (Salmo salar). Genes Genomes Genet. 2014, 4, 1135–1141. [Google Scholar] [CrossRef]
- Brand, P.; Ramirez, S.R. The Evolutionary Dynamics of the Odorant Receptor Gene Family in Corbiculate Bees. Genome Biol. Evol. 2017, 9, 2023–2036. [Google Scholar] [CrossRef]
- Kuehn, M.; Welsch, H.; Zahnert, T.; Hummel, T. Changes of pressure and humidity affect olfactory function. Eur. Arch. Oto-Rhino-Laryngol. 2008, 265, 299–302. [Google Scholar] [CrossRef]
- Jamieson, A.; Malkocs, T.; Piertney, S. Bioaccumulation of persistent organic pollutants in the deepest ocean fauna. Nat. Ecol. Evol. 2017, 1, 0051. [Google Scholar] [CrossRef]
| Gene Family | Gene Subfamily | Orthologous Groups | Selection Intensity k | p-Value |
|---|---|---|---|---|
| OR | ||||
| Family A | OR112 | OGn3 | 0.58 | <0.001 * |
| Family C | OR102 | OGn5 | 0.10 | 0.568 |
| OR103 | OGn4 | 1.63 | 0.500 | |
| Family D | OR108 | OGn6 | 0.46 | 0.456 |
| OR110 | OGn7 | 1.37 | 0.084 | |
| Family E | OR120 | OGn12 | 0 | 0.134 |
| OR121 | OGn13 | 0.51 | 0.042 * | |
| OR126 | OGn15 | 0.80 | 0.283 | |
| OGn16 | 1.00 | 1.000 | ||
| OGn17 | 1.25 | 0.415 | ||
| OGn18 | 0.19 | 1.000 | ||
| OR127 | OGn14 | 0.62 | 0.260 | |
| Family F | OR115 | OGn9 | 0.22 | 0.592 |
| OGn10 | 0.11 | 0.013 * | ||
| OGn11 | 2.17 | 0.108 | ||
| OR116 | OGn8 | 0.31 | 0.007 * | |
| Family H | OR129 | OGn1 | 0 | 0.049 * |
| OR130 | OGn2 | 0 | 0.009 * | |
| TAAR | ||||
| Class I | 21 | OGn2 | 8.05 | 0.102 |
| 27 | OGn1 | 4.57 | 0.020 * | |
| Class III | 29 | OGn3 | 0.73 | 0.076 |
| 30 | OGn4 | 8.24 | 0.177 | |
| 25 | OGn5 | 0.82 | 0.266 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, H.; Du, K.; Gan, X.; Yang, L.; He, S. Massive Loss of Olfactory Receptors But Not Trace Amine-Associated Receptors in the World’s Deepest-Living Fish (Pseudoliparis swirei). Genes 2019, 10, 910. https://doi.org/10.3390/genes10110910
Jiang H, Du K, Gan X, Yang L, He S. Massive Loss of Olfactory Receptors But Not Trace Amine-Associated Receptors in the World’s Deepest-Living Fish (Pseudoliparis swirei). Genes. 2019; 10(11):910. https://doi.org/10.3390/genes10110910
Chicago/Turabian StyleJiang, Haifeng, Kang Du, Xiaoni Gan, Liandong Yang, and Shunping He. 2019. "Massive Loss of Olfactory Receptors But Not Trace Amine-Associated Receptors in the World’s Deepest-Living Fish (Pseudoliparis swirei)" Genes 10, no. 11: 910. https://doi.org/10.3390/genes10110910
APA StyleJiang, H., Du, K., Gan, X., Yang, L., & He, S. (2019). Massive Loss of Olfactory Receptors But Not Trace Amine-Associated Receptors in the World’s Deepest-Living Fish (Pseudoliparis swirei). Genes, 10(11), 910. https://doi.org/10.3390/genes10110910







