Stress-Tolerant Yeasts: Opportunistic Pathogenicity Versus Biocontrol Potential
Abstract
1. Introduction
2. Materials and Methods
2.1. Yeast Strains and Culture Conditions
2.2. Growth under Oligotrophic Conditions and at 37 °C
2.3. Biofilm Quantification
2.4. Enzymatic Activities
2.5. Tolerance to H2O2 Oxidative Stress
2.6. Screening of Siderophore Production
2.7. Assimilation of Hydrocarbons
2.8. In Silico Analysis
3. Results
3.1. Growth under Oligotrophic Conditions and at 37 °C
3.2. Biofilm Quantification
3.3. Tolerance to H2O2 Oxidative Stress
3.4. Siderophore Production
3.5. Enzymatic Activities
3.6. Assimilation of Hydrocarbons
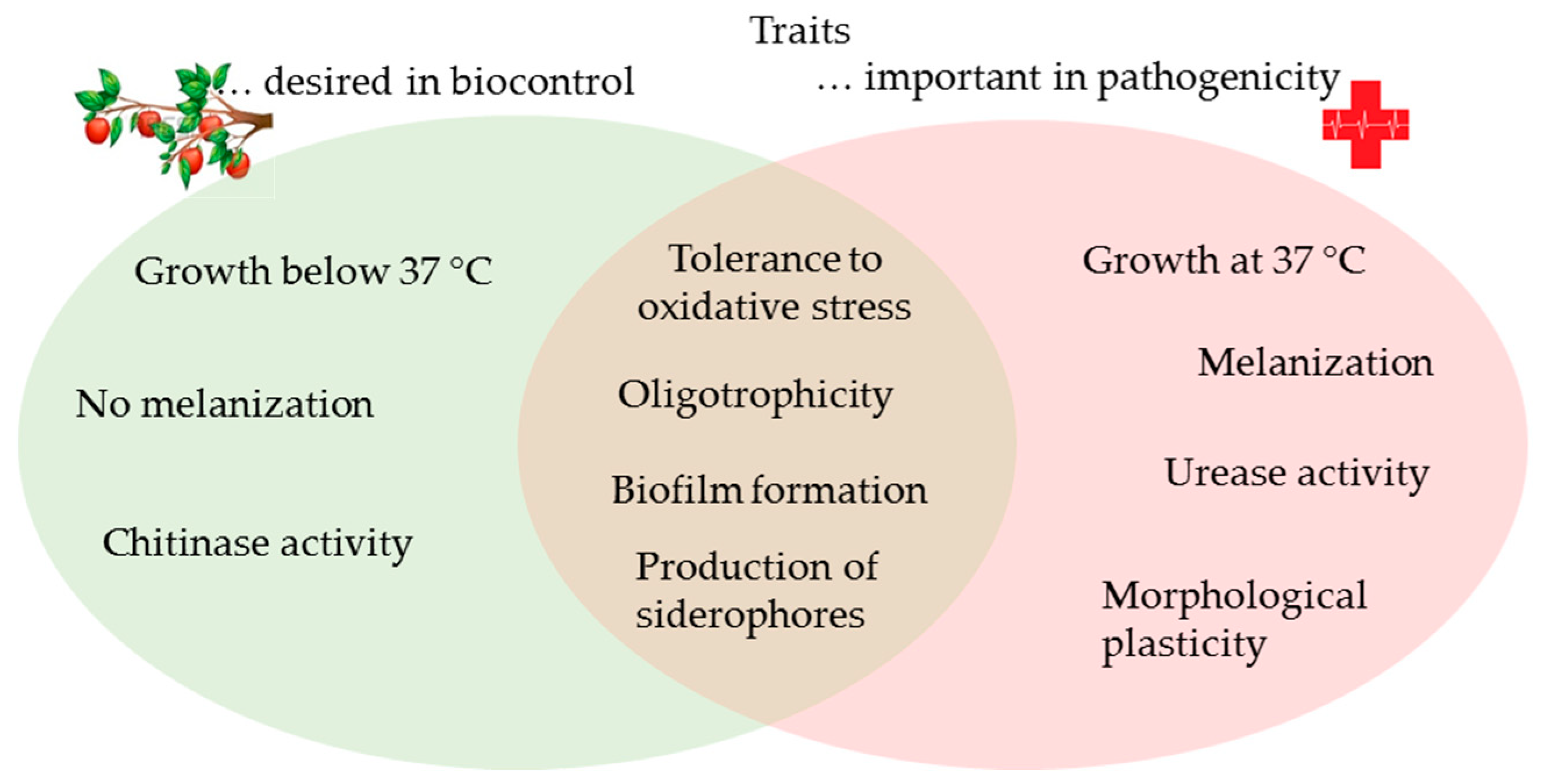
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sharma, R.R.; Singh, D.; Singh, R. Biological control of postharvest diseases of fruits and vegetables by microbial antagonists: A review. Biol. Control 2009, 50, 205–221. [Google Scholar] [CrossRef]
- Sui, Y.; Wisniewski, M.; Droby, S.; Liu, J. Responses of yeast biocontrol agents to environmental stress. Appl. Environ. Microbiol. 2015, 81, 2968–2975. [Google Scholar] [CrossRef] [PubMed]
- de Hoog, G.S.; Guarro, J.; Gené, J.; Figueras, M.J. Atlas of clinical fungi. In The Ultimate Benchtool for Diagnostics; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2015. [Google Scholar]
- Gostinčar, C.; Zajc, J.; Lenassi, M.; Plemenitaš, A.; de Hoog, S.; Al-Hatmi, A.M.S.; Gunde-Cimerman, N. Fungi between extremotolerance and opportunistic pathogenicity on humans. Fungal Divers. 2018, 93, 195–213. [Google Scholar] [CrossRef]
- Gostinčar, C.; Ohm, R.A.; Kogej, T.; Sonjak, S.; Turk, M.; Zajc, J.; Zalar, P.; Grube, M.; Sun, H.; Han, J.; et al. Genome sequencing of four Aureobasidium pullulans varieties: Biotechnological potential, stress tolerance, and description of new species. BMC Genom. 2014, 15, 549. [Google Scholar] [CrossRef] [PubMed]
- Novak Babič, M.; Zalar, P.; Zenko, B.; Schroers, H.J.; Džeroski, S.; Gunde-Cimerman, N. Candida and Fusarium species known as opportunistic human pathogens from customer-accessible parts of residential washing machines. Fungal Biol. 2015, 119, 95–113. [Google Scholar] [CrossRef] [PubMed]
- Chi, Z.M.; Wang, F.; Chi, Z.; Yue, L.X.; Liu, G.L.; Zhang, T. Bioproducts from Aureobasidium pullulans, a biotechnologically important yeast. Appl. Microbiol. Biotechnol. 2009, 82, 793–804. [Google Scholar] [CrossRef]
- Chan, G.F.; Puad, M.S.A.; Chin, C.F.; Rashid, N.A.A. Emergence of Aureobasidium pullulans as human fungal pathogen and molecular assay for future medical diagnosis. Folia Microbiol. 2011, 56, 459–467. [Google Scholar] [CrossRef]
- Gunde-Cimerman, N.; Zalar, P.; de Hoog, S.; Plemenitaš, A. Hypersaline waters in salterns—Natural ecological niches for halophilic black yeasts. FEMS Microbiol. Ecol. 2000, 32, 235–240. [Google Scholar]
- Shiomi, N.; Yasuda, T.; Inoue, Y.; Kusumoto, N.; Iwasaki, S.; Katsuda, T.; Katoh, S. Characteristics of neutralization of acids by newly isolated fungal cells. J. Biosci. Bioeng. 2004, 97, 54–58. [Google Scholar] [CrossRef]
- Zalar, P.; Gostinčar, C.; de Hoog, G.S.; Uršič, V.; Sudhadham, M.; Gunde-Cimerman, N. Redefinition of Aureobasidium pullulans and its varieties. Stud. Mycol. 2008, 61, 21–38. [Google Scholar] [CrossRef]
- Onofri, S. Antarctic Microfungi. In Enigmatic Microorganisms and Life in Extreme Environments; Seckbach, J., Ed.; Kluwer Academic: Dordrecht, The Netherlands, 1999; pp. 323–336. [Google Scholar]
- Zalar, P.; Novak, M.; De Hoog, G.S.; Gunde-Cimerman, N. Dishwashers—A man-made ecological niche accommodating human opportunistic fungal pathogens. Fungal Biol. 2011, 115, 997–1007. [Google Scholar] [CrossRef] [PubMed]
- Gümral, R.; Özhak-Baysan, B.; Tümgör, A.; Saraçlı, M.; Yıldıran, Ş.; Ilkit, M.; Zupančič, J.; Novak-Babič, M.; Gunde-Cimerman, N.; Zalar, P.; de Hoog, G.S. Dishwashers provide a selective extreme environment for human-opportunistic yeast-like fungi. Fungal Divers. 2015, 76, 1–9. [Google Scholar] [CrossRef]
- Nishimura, K.; Miyaji, M. Studies on a saprophyte of Exophiala dermatitidis isolated from a humidifier. Mycopathologia 1982, 77, 173–181. [Google Scholar] [CrossRef]
- Nishimura, K.; Miyaji, M.; Taguchi, H.; Tanaka, R. Fungi in bathwater and sludge of bathroom drainpipes. 1. Frequent isolation of Exophiala species. Mycopathologia 1987, 97, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Matos, T.; de Hoog, G.S.; de Boer, A.G.; de Crom, I.; Haase, G. High prevalence of the neurotrope Exophiala dermatitidis and related oligotrophic black yeasts in sauna facilities. Mycoses 2002, 45, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Lian, X.; de Hoog, G.S. Indoor wet cells harbour melanized agents of cutaneous infection. Med. Mycol. 2010, 48, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Heinrichs, G.; Hubner, I.; Schmidt, C.K.; de Hoog, G.S.; Haase, G. Analysis of black fungal biofilms occurring at domestic water taps (I): Compositional analysis using tag-encoded FLX amplicon pyrosequencing. Mycopathologia 2013, 175, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Novak Babič, M.; Zalar, P.; Ženko, B.; Džeroski, S.; Gunde-Cimerman, N. Yeasts and yeast-like fungi in tap water and groundwater, and their transmission to household appliances. Fungal Ecol. 2016, 20, 30–39. [Google Scholar] [CrossRef]
- Döğen, A.; Kaplan, E.; Ilkit, M.; de Hoog, G.S. Massive contamination of Exophiala dermatitidis and E. phaeomuriformis in railway stations in subtropical Turkey. Mycopathologia 2013, 175, 381–386. [Google Scholar] [CrossRef]
- Sudhadham, M.; Prakitsin, S.; Sivichai, S.; Chaiyarat, R.; Dorrestein, G.M.; Menken, S.B.J.; de Hoog, G.S. The neurotropic black yeast Exophiala dermatitidis has a possible origin in the tropical rain forest. Stud. Mycol. 2008, 61, 145–155. [Google Scholar] [CrossRef]
- Branda, E.; Turchetti, B.; Diolaiuti, G.; Pecci, M.; Smiraglia, C.; Buzzini, P. Yeast and yeast-like diversity in the southernmost glacier of Europe (Calderone Glacier, Apennines, Italy). FEMS Microbiol. Ecol. 2010, 72, 354–369. [Google Scholar] [CrossRef] [PubMed]
- Döğen, A.; Kaplan, E.; Öksüz, Z.; Serin, M.S.; Ilkit, M.; de Hoog, G.S. Dishwashers are a major source of human opportunistic yeast-like fungi in indoor environments in Mersin, Turkey. Med. Mycol. 2013, 51, 493–498. [Google Scholar] [CrossRef]
- Blasi, B.; Tafer, H.; Tesei, D.; Sterflinger, K. From glacier to sauna: RNA-Seq of the human pathogen black fungus Exophiala dermatitidis under varying temperature conditions exhibits common and novel fungal response. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Taj-Aldeen, S.J.; El Shafie, S.; Alsoub, H.; Eldeeb, Y.; de Hoog, G.S. Isolation of Exophiala dermatitidis from endotracheal aspirate of a cancer patient. Mycoses 2006, 49, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Butinar, L.; Spencer-Martins, I.; Gunde-Cimerman, N. Yeasts in high Arctic glaciers: The discovery of a new habitat for eukaryotic microorganisms. Antonie Van Leeuwenhoek 2007, 91, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Butinar, L.; Santos, S.; Spencer-Martins, I.; Oren, A.; Gunde-Cimerman, N. Yeast diversity in hypersaline habitats. FEMS Microbiol. Lett. 2005, 244, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Xin, G.; Glawe, D.; Doty, S.L. Characterization of three endophytic, indole-3-acetic acid-producing yeasts occurring in Populus trees. Mycol. Res. 2009, 113, 973–980. [Google Scholar] [CrossRef]
- Pitt, J.I.; Hocking, A.D. Fungi and Food Spoilage, 3rd ed.; Springer: Dordrecht, The Netherlands, 2009. [Google Scholar]
- De Siloniz, M.I.; Payo, E.M.; Callejo, M.A.; Marquina, D.; Peinado, J.M. Environmental adaptation factors of two yeasts isolated from the leachate of a uranium mineral heap. FEMS Microbiol. Lett. 2002, 210, 233–237. [Google Scholar] [CrossRef]
- Villegas, L.B.; Amoroso, M.J.; De Figueroa, L.I.C. Copper tolerant yeasts isolated from polluted area of Argentina. J. Basic Microbiol. 2005, 45, 381–391. [Google Scholar] [CrossRef]
- Zheng, C.L.; Zhou, J.T.; Wang, J.; Wang, J.; Qu, B.C. Isolation and characterization of a nitrobenzene degrading yeast strain from activated sludge. J. Hazard. Mater. 2008, 160, 194–199. [Google Scholar] [CrossRef]
- Russo, G.; Libkind, D.; Sampaio, J.P.; van Broock, M.R. Yeast diversity in the acidic Rio Agrio-Lake Caviahue volcanic environment (Patagonia, Argentina). FEMS Microbiol. Ecol. 2008, 65, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Bore, E.; Langsrud, S. Characterization of micro-organisms isolated from dairy industry after cleaning and fogging disinfection with alkyl amine and peracetic acid. J. Appl. Microbiol. 2005, 98, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Libkind, D.; Brizzio, S.; van Broock, M. Rhodotorula mucilaginosa, a carotenoid producing yeast strain from a Patagonian high-altitude lake. Folia Microbiol. 2004, 49, 19–25. [Google Scholar] [CrossRef]
- Brandão, L.R.; Libkind, D.; Vaz, A.B.M.; Santo, L.C.E.; Moline, M.; de Garcia, V.; van Broock, M.; Rosa, C.A. Yeasts from an oligotrophic lake in Patagonia (Argentina): Diversity, distribution and synthesis of photoprotective compounds and extracellular enzymes. FEMS Microbiol. Ecol. 2011, 76, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Gunde-Cimerman, N.; Butinar, L.; Sonjak, S.; Turk, M.; Uršič, V.; Zalar, P.; Plemenitaš, A. Halotolerant and halophilic fungi from coastal environments in the Arctics. In Adaptation to Life in High Salt Concentrations in Archaea, Bacteria, and Eukarya; Gunde-Cimerman, N., Oren, A., Plemenitaš, A., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 397–423. [Google Scholar]
- Li, R.P.; Zhang, H.Y.; Liu, W.M.; Zheng, X.D. Biocontrol of postharvest gray and blue mold decay of apples with Rhodotorula mucilaginosa and possible mechanisms of action. Int. J. Food Microbiol. 2011, 146, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Robiglio, A.; Sosa, M.C.; Lutz, M.C.; Lopes, C.A.; Sangorrin, M.P. Yeast biocontrol of fungal spoilage of pears stored at low temperature. Int. J. Food Microbiol. 2011, 147, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.Y.; Ge, L.L.; Chen, K.P.; Zhao, L.N.; Zhang, X.Y. Enhanced biocontrol activity of Rhodotorula mucilaginosa cultured in media containing chitosan against postharvest diseases in strawberries: Possible mechanisms underlying the effect. J. Agric. Food Chem. 2014, 62, 4214–4224. [Google Scholar] [CrossRef]
- Tuon, F.F.; Costa, S.F. Rhodotorula infection. A systematic review of 128 cases from literature. Rev. Iberoam. Micol. 2008, 25, 135–140. [Google Scholar] [CrossRef]
- Papon, N.; Savini, V.; Lanoue, A.; Simkin, A.J.; Creche, J.; Giglioli-Guivarc’h, N.; Clastre, M.; Courdavault, V.; Sibirny, A.A. Candida guilliermondii: Biotechnological applications, perspectives for biological control, emerging clinical importance and recent advances in genetics. Curr. Genet. 2013, 59, 73–90. [Google Scholar] [CrossRef]
- Zupančič, J.; Babič, M.N.; Zalar, P.; Gunde-Cimerman, N. The black yeast Exophiala dermatitidis and other selected opportunistic human fungal pathogens spread from dishwashers to kitchens. PLoS ONE 2016, 11, e0148166. [Google Scholar] [CrossRef]
- Savini, V.; Catavitello, C.; Onofrillo, D.; Masciarelli, G.; Astolfi, D.; Balbinot, A.; Febbo, F.; D’Amario, C.; D’Antonio, D. What do we know about Candida guilliermondii? A voyage throughout past and current literature about this emerging yeast. Mycoses 2010, 54, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Sui, Y.; Liu, J. Effect of glucose on thermotolerance and biocontrol efficacy of the antagonistic yeast Pichia guilliermondii. Biol. Control 2014, 74, 59–64. [Google Scholar] [CrossRef]
- Butinar, L.; Strmole, T.; Gunde-Cimerman, N. Relative incidence of ascomycetous yeasts in Arctic coastal environments. Antonie Van Leeuwenhoek 2011, 61, 832–843. [Google Scholar] [CrossRef]
- Capece, A.; Romano, P. “Pecorino di Filiano” cheese as a selective habitat for the yeast species, Debaryomyces hansenii. Int. J. Food Microbiol. 2009, 132, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Prista, C.; Loureiro-Dias, M.C.; Montiel, V.; Garcia, R.; Ramos, J. Mechanisms underlying the halotolerant way of Debaryomyces hansenii. FEMS Yeast Res. 2005, 5, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Gunde-Cimerman, N.; Frisvad, J.C.; Zalar, P.; Plemenitas, A. Halotolerant and halophilic fungi. In Biodiversity of Fungi: Their Role in Human Life; Deshmukh, S.K., Rai, M.K., Eds.; Oxford & IBH Publishing Co. Pvt. Ltd.: New Delhi, India, 2005; pp. 69–127. [Google Scholar]
- Martinez, J.L.; Luna, C.; Ramos, J. Proteomic changes in response to potassium starvation in the extremophilic yeast Debaryomyces hansenii. FEMS Yeast Res. 2012, 12, 651–661. [Google Scholar] [CrossRef] [PubMed]
- Taqarort, N.; Echairi, A.; Chaussod, R.; Nouaim, R.; Boubaker, H.; Benaoumar, A.A.; Boudyach, E. Screening and identification of epiphytic yeasts with potential for biological control of green mold of citrus fruits. World J. Microbiol. Biotechnol. 2008, 24, 3031–3038. [Google Scholar] [CrossRef]
- Desnos-Ollivier, M.; Ragon, M.; Robert, V.; Raoux, D.; Gantier, J.C.; Dromer, F. Debaryomyces hansenii (Candida famata), a rare human fungal pathogen often misidentified as Pichia guilliermondii (Candida guilliermondii). J. Clin. Microbiol. 2008, 46, 3237–3242. [Google Scholar] [CrossRef]
- Lopez-Martinez, R. Candidosis, a new challenge. Clin. Dermatol. 2010, 28, 178–184. [Google Scholar] [CrossRef]
- Spadaro, D.; Lore, A.; Garibaldi, A.; Gullino, M.L. A new strain of Metschnikowia fructicola for postharvest control of Penicillium expansum and patulin accumulation on four cultivars of apple. Postharvest Biol. Technol. 2013, 75, 1–8. [Google Scholar] [CrossRef]
- Kurtzman, C.P.; Droby, S. Metschnikowia fructicola, a new ascosporic yeast with potential for biocontrol of postharvest fruit rots. Syst. Appl. Microbiol. 2001, 24, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Piombo, E.; Sela, N.; Wisniewski, M.; Hoffmann, M.; Gullino, M.L.; Allard, M.W.; Levin, E.; Spadaro, D.; Droby, S. Genome sequence, assembly and characterization of two Metschnikowia fructicola strains used as biocontrol agents of postharvest diseases. Front. Microbiol. 2018, 9, 593. [Google Scholar] [CrossRef] [PubMed]
- Tkavc, R.; Matrosova, V.Y.; Grichenko, O.E.; Gostinčar, C.; Volpe, R.P.; Klimenkova, P.; Gaidamakova, E.K.; Zhou, C.E.; Stewart, B.J.; Lyman, M.G.; et al. Prospects for fungal bioremediation of acidic radioactive waste sites: Characterization and genome sequence of Rhodotorula taiwanensis MD1149. Front. Microbiol. 2018, 8, 2528. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wisniewski, M.; Droby, S.; Tian, S.; Hershkovitz, V.; Tworkoski, T. Effect of heat shock treatment on stress tolerance and biocontrol efficacy of Metschnikowia fructicola. FEMS Microbiol. Ecol. 2011, 76, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Ren, D.W.; Madsen, J.S.; Sørensen, S.J.; Burmølle, M. High prevalence of biofilm synergy among bacterial soil isolates in cocultures indicates bacterial interspecific cooperation. ISME J. 2015, 9, 81–89. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Dodman, R.L.; Reinke, J.R. A selective medium for determining the population of viable conidia of Cochliobolus sativus in soil. Aust. J. Agric. Res. 1982, 33, 287–291. [Google Scholar] [CrossRef]
- Paterson, R.R.M.; Bridge, P.D. Biochemical Techniques for Filamentous Fungi; CAB International: Wallingford, UK, 1994. [Google Scholar]
- Hankin, L.; Zucker, M.; Sands, D.C. Improved solid medium for the detection and enumeration of pectolytic bacteria. Appl. Microbiol. 1971, 22, 205–209. [Google Scholar]
- Hitha, P.K.; Girija, D. Isolation and screening of native microbial isolates for pectinase activity. Int. J. Sci. Res. 2014, 3, 632–634. [Google Scholar]
- Agrawal, T.; Kotasthane, A.S. Chitinolytic assay of indigenous Trichoderma isolates collected from different geographical locations of Chhattisgarh in Central India. Springerplus 2012, 1. [Google Scholar] [CrossRef]
- Lelliott, R.A.; Stead, D.E. Methods for the Diagnosis of Bacterial Diseases of Plants; Published on behalf of the British Society for Plant Pathology; Blackwell Scientific Publications: Oxford, UK, 1987; 216p. [Google Scholar]
- Brizzio, S.; Turchetti, B.; de García, V.; Libkind, D.; Buzzini, P.; van Broock, M. Extracellular enzymatic activities of basidiomycetous yeasts isolated from glacial and subglacial waters of northwest Patagonia (Argentina). Can. J. Microbiol. 2007, 53, 519–525. [Google Scholar] [CrossRef]
- de Garcia, V.; Zalar, P.; Brizzio, S.; Gunde-Cimerman, N.; van Broock, M. Cryptococcus species (Tremellales) from glacial biomes in the southern (Patagonia) and northern (Svalbard) hemispheres. FEMS Microbiol. Ecol. 2012, 82, 523–539. [Google Scholar] [CrossRef] [PubMed]
- Hankin, L.; Anagnostakis, S.L. The use of solid media for detection of enzyme production by fungi. Mycologia 1975, 67, 597–607. [Google Scholar] [CrossRef]
- Scott, J.A.; Untereiner, W.A. Determination of keratin degradation by fungi using keratin azure. Med. Mycol. 2004, 42, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Milagres, A.M.F.; Machuca, A.; Napoleão, D. Detection of siderophore production from several fungi and bacteria by a modification of chrome azurol S (CAS) agar plate assay. J. Microbiol. Methods 1999, 37, 1–6. [Google Scholar] [CrossRef]
- Schwyn, B.; Neilands, J.B. Universal chemical-assay for the detection and determination of siderophores. Anal. Biochem. 1987, 160, 47–56. [Google Scholar] [CrossRef]
- Satow, M.M.; Attili-Angelis, D.; de Hoog, G.S.; Angelis, D.F.; Vicente, V.A. Selective factors involved in oil flotation isolation of black yeasts from the environment. Stud. Mycol. 2008, 61, 157–163. [Google Scholar] [CrossRef]
- Prenafeta-Boldu, F.X.; Kuhn, A.; Luykx, D.M.A.M.; Anke, H.; van Groenestijn, J.W.; de Bont, J.A.M. Isolation and characterisation of fungi growing on volatile aromatic hydrocarbons as their sole carbon and energy source. Mycol. Res. 2001, 105, 477–484. [Google Scholar] [CrossRef]
- Dujon, B.; Sherman, D.; Fischer, G.; Durrens, P.; Casaregola, S.; Lafontaine, I.; De Montigny, J.; Marck, C.; Neuveglise, C.; Talla, E.; et al. Genome evolution in yeasts. Nature 2004, 430, 35–44. [Google Scholar] [CrossRef]
- Hershkovitz, V.; Sela, N.; Taha-Salaime, L.; Liu, J.; Rafael, G.; Kessler, C.; Aly, R.; Levy, M.; Wisniewski, M.; Droby, S. De-novo assembly and characterization of the transcriptome of Metschnikowia fructicola reveals differences in gene expression following interaction with Penicillium digitatum and grapefruit peel. BMC Genom. 2013, 14. [Google Scholar] [CrossRef]
- Ramirez, C.; Gonzalez, A. Rhodotorula grinbergsii sp. nov. isolated from decayed wood in the evergreen rainy Valdivian Forest of southern Chile. Mycopathologia 1984, 88, 51–53. [Google Scholar] [CrossRef]
- Butler, G.; Rasmussen, M.D.; Lin, M.F.; Santos, M.A.S.; Sakthikumar, S.; Munro, C.A.; Rheinbay, E.; Grabherr, M.; Forche, A.; Reedy, J.L.; et al. Evolution of pathogenicity and sexual reproduction in eight Candida genomes. Nature 2009, 459, 657–662. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Martinez, D.A.; Gujja, S.; Sykes, S.M.; Zeng, Q.; Szaniszlo, P.J.; Wang, Z.; Cuomo, C.A. Comparative genomic and transcriptomic analysis of Wangiella dermatitidis, a major cause of phaeohyphomycosis and a model black yeast human pathogen. G3 (Bethesda) 2014, 4, 561–578. [Google Scholar] [CrossRef] [PubMed]
- Petersen, T.N.; Brunak, S.; von Heijne, G.; Nielsen, H. SignalP 4.0: Discriminating signal peptides from transmembrane regions. Nat. Methods 2011, 8, 785–786. [Google Scholar] [CrossRef]
- Krogh, A.; Larsson, B.; von Heijne, G.; Sonnhammer, E.L.L. Predicting transmembrane protein topology with a hidden Markov model: Application to complete genomes. J. Mol. Biol. 2001, 305, 567–580. [Google Scholar] [CrossRef]
- Wei, T.; Simko, V. R Package “corrplot”: Visualization of a Correlation Matrix. 2017. Available online: https://github.com/taiyun/corrplot (accessed on 8 January 2018).
- Blin, K.; Medema, M.H.; Kazempour, D.; Fischbach, M.A.; Breitling, R.; Takano, E.; Weber, T. antiSMASH 2.0-a versatile platform for genome mining of secondary metabolite producers. Nucleic Acids Res. 2013, 41, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Cramer, R.A.; Stajich, J.E.; Yamanaka, Y.; Dietrich, F.S.; Steinbach, W.J.; Perfect, J.R. Phylogenomic analysis of non-ribosomal peptide synthetases in the genus Aspergillus. Gene 2006, 383, 24–32. [Google Scholar] [CrossRef]
- von Döhren, H. A survey of nonribosomal peptide synthetase (NRPS) genes in Aspergillus nidulans. Fungal Genet. Biol. 2009, 46, S45–S52. [Google Scholar] [CrossRef]
- Bushley, K.E.; Turgeon, B.G. Phylogenomics reveals subfamilies of fungal nonribosomal peptide synthetases and their evolutionary relationships. BMC Evol. Biol. 2010, 10. [Google Scholar] [CrossRef]
- Condon, B.J.; Leng, Y.Q.; Wu, D.L.; Bushley, K.E.; Ohm, R.A.; Otillar, R.; Martin, J.; Schackwitz, W.; Grimwood, J.; MohdZainudin, N.; et al. Comparative genome structure, secondary metabolite, and effector coding capacity across Cochliobolus pathogens. PLoS Genet. 2013, 9. [Google Scholar] [CrossRef]
- Gostinčar, C.; Gunde-Cimerman, N. Overview of oxidative stress response genes in selected halophilic fungi. Genes 2018, 9. [Google Scholar] [CrossRef]
- Katoh, K.; Kuma, K.; Toh, H.; Miyata, T. MAFFT version 5: Improvement in accuracy of multiple sequence alignment. Nucleic Acids Res. 2005, 33, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Dufayard, J.F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New algorithms and methods to estimate maximum-likelihood phylogenies: Assessing the performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Loftus, B.J.; Fung, E.; Roncaglia, P.; Rowley, D.; Amedeo, P.; Bruno, D.; Vamathevan, J.; Miranda, M.; Anderson, I.J.; Fraser, J.A.; et al. The genome of the basidiomycetous yeast and human pathogen Cryptococcus neoformans. Science 2005, 307, 1321–1324. [Google Scholar] [CrossRef] [PubMed]
- Blasi, B.; Tafer, H.; Kustor, C.; Poyntner, C.; Lopandic, K.; Sterflinger, K. Genomic and transcriptomic analysis of the toluene degrading black yeast Cladophialophora immunda. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Gostinčar, C.; Grube, M.; Gunde-Cimerman, N. Evolution of fungal pathogens in domestic environments? Fungal Biol. 2011, 115, 1008–1018. [Google Scholar] [CrossRef]
- Gostinčar, C.; Gunde-Cimerman, N.; Grube, M. Polyextremotolerance as the fungal answer to changing environments. In Microbial Evolution under Extreme Conditions; Bakermans, C., Ed.; de Gruyter: Berlin, Germany, 2015; pp. 185–208. [Google Scholar]
- Zhang, D.P.; Spadaro, D.; Garibaldi, A.; Gullino, M.L. Potential biocontrol activity of a strain of Pichia guilliermondii against grey mold of apples and its possible modes of action. Biol. Control 2011, 57, 193–201. [Google Scholar] [CrossRef]
- Mari, M.; Martini, C.; Spadoni, A.; Rouissi, W.; Bertolini, P. Biocontrol of apple postharvest decay by Aureobasidium pullulans. Postharvest Biol. Technol. 2012, 73, 56–62. [Google Scholar] [CrossRef]
- de Hoog, G.S.; Zalar, P.; Gerrits van den Ende, A.H.G.; Gunde-Cimerman, N. Relation of halotolerance to human-pathogenicity in the fungal tree of life: An overview of ecology and evolution under stress. In Adaptation to Life at High Salt Concentrations in Archaea, Bacteria, and Eukarya; Gunde-Cimerman, N., Oren, A., Plemenitaš, A., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 373–395. [Google Scholar]
- Casadevall, A.; Fang, F.C.; Pirofski, L.A. Microbial virulence as an emergent property: Consequences and opportunities. PLoS Path. 2011, 7. [Google Scholar] [CrossRef]
- Casadevall, A.; Pirofski, L.A. Accidental virulence, cryptic pathogenesis, Martians, lost hosts, and the pathogenicity of environmental microbes. Eukaryot. Cell 2007, 6. [Google Scholar] [CrossRef]
- Cairns, T.; Minuzzi, F.; Bignell, E. The host-infecting fungal transcriptome. FEMS Microbiol. Lett. 2010, 307, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Prenafeta-Boldu, F.X.; Summerbell, R.; de Hoog, G.S. Fungi growing on aromatic hydrocarbons: Biotechnology’s unexpected encounter with biohazard? FEMS Microbiol. Rev. 2006, 30, 109–130. [Google Scholar] [CrossRef] [PubMed]
- De Hoog, G.S. Evolution of black yeasts: Possible adaptation to the human host. Antonie Van Leeuwenhoek 1993, 63, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Robert, V.A.; Casadevall, A. Vertebrate endothermy restricts most fungi as potential pathogens. J. Infect. Dis. 2009, 200, 1623–1626. [Google Scholar] [CrossRef] [PubMed]
- Brunke, S.; Mogavero, S.; Kasper, L.; Hube, B. Virulence factors in fungal pathogens of man. Curr. Opin. Microbiol. 2016, 32, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Schaible, M.E.; Kaufmann, S.H.E. Iron and microbial infection. Nat. Rev. Microbiol. 2004, 2, 946–953. [Google Scholar] [CrossRef] [PubMed]
- Hamad, M. Antifungal immunotherapy and immunomodulation: A double-hitter approach to deal with invasive fungal infections. Scand. J. Immunol. 2008, 67, 533–543. [Google Scholar] [CrossRef]
- Neilands, J.B. Siderophores. Arch. Biochem. Biophys. 1993, 302, 1–3. [Google Scholar] [CrossRef]
- Johnson, L. Iron and siderophores in fungal-host interactions. Mycol. Res. 2008, 112, 170–183. [Google Scholar] [CrossRef]
- Chi, Z.; Wang, X.X.; Ma, Z.C.; Buzdar, M.A.; Chi, Z.M. The unique role of siderophore in marine-derived Aureobasidium pullulans HN6.2. BioMetals 2012, 25, 219–230. [Google Scholar] [CrossRef]
- Wang, W.L.; Chi, Z.M.; Chi, Z.; Li, J.; Wang, X.H. Siderophore production by the marine-derived Aureobasidium pullulans and its antimicrobial activity. Bioresour. Technol. 2009, 100, 2639–2641. [Google Scholar] [CrossRef] [PubMed]
- Fanning, S.; Mitchell, A.P. Fungal Biofilms. PLoS Path. 2012, 8, e1002585. [Google Scholar] [CrossRef] [PubMed]
- Inci, M.; Atalay, M.A.; Koc, A.N.; Yula, E.; Evirgen, O.; Durmaz, S.; Demir, G. Investigating virulence factors of clinical Candida isolates in relation to atmospheric conditions and genotype. Turk. J. Med. Sci. 2012, 42, 1476–1483. [Google Scholar] [CrossRef]
- Nunes, J.M.; Bizerra, F.C.; Ferreira, R.C.E.; Colombo, A.L. Molecular identification, antifungal susceptibility profile, and biofilm formation of clinical and environmental Rhodotorula species isolates. Antimicrob. Agents Chemother. 2013, 57, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Pandin, C.; Le Coq, D.; Canette, A.; Aymerich, S.; Briandet, R. Should the biofilm mode of life be taken into consideration for microbial biocontrol agents? Microb. Biotechnol. 2017, 10, 719–734. [Google Scholar] [CrossRef]
- Klein, M.N.; Kupper, K.C. Biofilm production by Aureobasidium pullulans improves biocontrol against sour rot in citrus. Food Microbiol. 2018, 69, 1–10. [Google Scholar] [CrossRef]
- Wachowska, U.; Głowacka, K.; Mikołajczyk, W.; Kucharska, K. Biofilm of Aureobasidium pullulans var. pullulans on winter wheat kernels and its effect on other microorganisms. Microbiology 2016, 85, 523–530. [Google Scholar] [CrossRef]
- Gori, K.; Knudsen, P.B.; Nielsen, K.F.; Arneborg, N.; Jespersen, L. Alcohol-based quorum sensing plays a role in adhesion and sliding motility of the yeast Debaryomyces hansenii. FEMS Yeast Res. 2011, 11, 643–652. [Google Scholar] [CrossRef]
- van Baarlen, P.; van Belkum, A.; Summerbell, R.C.; Crous, P.W.; Thomma, B.P.H.J. Molecular mechanisms of pathogenicity: How do pathogenic microorganisms develop cross-kingdom host jumps? FEMS Microbiol. Rev. 2007, 31, 239–277. [Google Scholar] [CrossRef]
- Kogej, T.; Wheeler, M.H.; Rizner, T.L.; Gunde-Cimerman, N. Evidence for 1,8-dihydroxynaphthalene melanin in three halophilic black yeasts grown under saline and non-saline conditions. FEMS Microbiol. Lett. 2004, 232, 203–209. [Google Scholar] [CrossRef]
- Zajc, J. Characterization of Yeast-Like Fungi Aureobasidium pullulans; National Institute of Biology: Ljubljana, Slovenia, Unpublished work; 2018. [Google Scholar]
- Breitenbach, M.; Weber, M.; Rinnerthaler, M.; Karl, T.; Breitenbach-Koller, L. Oxidative stress in fungi: Its function in signal transduction, interaction with plant hosts, and lignocellulose degradation. Biomolecules 2015, 5, 318–342. [Google Scholar] [CrossRef] [PubMed]
- Leal, S.M.; Vareechon, C.; Cowden, S.; Cobb, B.A.; Latge, J.P.; Momany, M.; Pearlman, E. Fungal antioxidant pathways promote survival against neutrophils during infection. J. Clin. Invest. 2012, 122, 2482–2498. [Google Scholar] [CrossRef] [PubMed]
- Ohm, R.A.; Feau, N.; Henrissat, B.; Schoch, C.L.; Horwitz, B.A.; Barry, K.W.; Condon, B.J.; Copeland, A.C.; Dhillon, B.; Glaser, F.; et al. Diverse lifestyles and strategies of plant pathogenesis encoded in the genomes of eighteen Dothideomycetes fungi. PLoS Pathogens 2012, 8, e1003037. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.T.; Liu, H.Q.; Wang, C.F.; Xu, J.R. Comparative analysis of fungal genomes reveals different plant cell wall degrading capacity in fungi. BMC Genom. 2013, 14. [Google Scholar] [CrossRef] [PubMed]
- Mahadevan, B.; Crawford, D.L. Properties of the chitinase of the antifungal biocontrol agent Streptomyces lydicus WYEC108. Enzyme Microb. Technol. 1997, 20, 489–493. [Google Scholar] [CrossRef]
- Cox, G.M.; Mukherjee, J.; Cole, G.T.; Casadevall, A.; Perfect, J.R. Urease as a virulence factor in experimental cryptococcosis. Infect. Immun. 2000, 68, 443–448. [Google Scholar] [CrossRef]
- Rutherford, J.C. The emerging role of urease as a general microbial virulence factor. PLoS Path. 2014, 10. [Google Scholar] [CrossRef] [PubMed]
- Isola, D.; Selbmann, L.; de Hoog, G.S.; Fenice, M.; Onofri, S.; Prenafeta-Boldu, F.X.; Zucconi, L. Isolation and screening of black fungi as degraders of volatile aromatic hydrocarbons. Mycopathologia 2013, 175, 369–379. [Google Scholar] [CrossRef]
- Chandran, P.; Das, N. Role of plasmid in diesel oil degradation by yeast species isolated from petroleum hydrocarbon-contaminated soil. Environ. Technol. 2012, 33, 645–652. [Google Scholar] [CrossRef]
- Seyedmousavi, S.; Badali, H.; Chlebicki, A.; Zhao, J.J.; Prenafeta-Boldu, F.X.; De Hoog, G.S. Exophiala sideris, a novel black yeast isolated from environments polluted with toxic alkyl benzenes and arsenic. Fungal Biol. 2011, 115, 1030–1037. [Google Scholar] [CrossRef]
- Zhao, J.J.; Zeng, J.S.; de Hoog, G.S.; Attili-Angelis, D.; Prenafeta-Boldu, F.X. Isolation and Identification of black yeasts by enrichment on atmospheres of monoaromatic hydrocarbons. Microb. Ecol. 2010, 60, 149–156. [Google Scholar] [CrossRef] [PubMed]
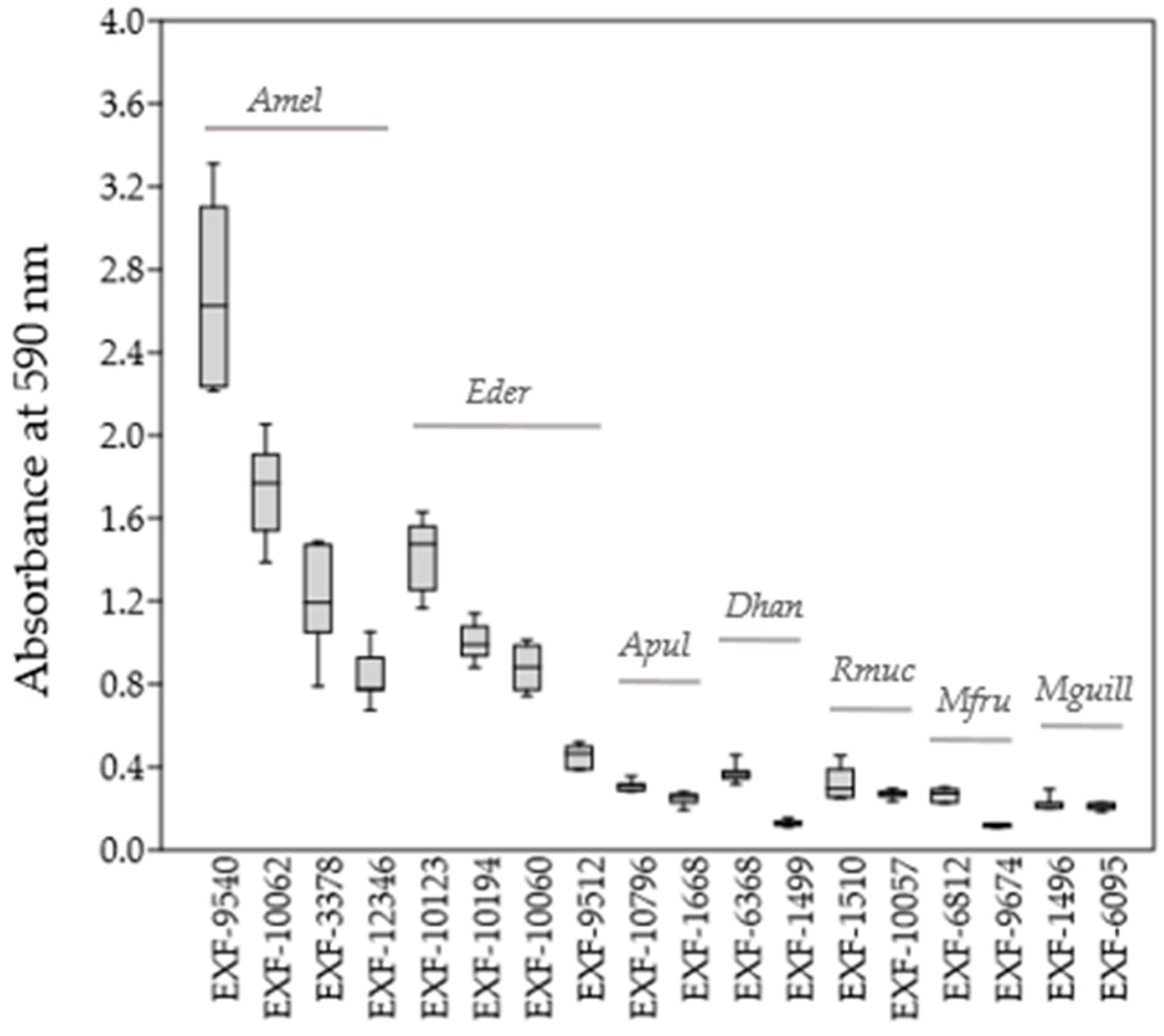
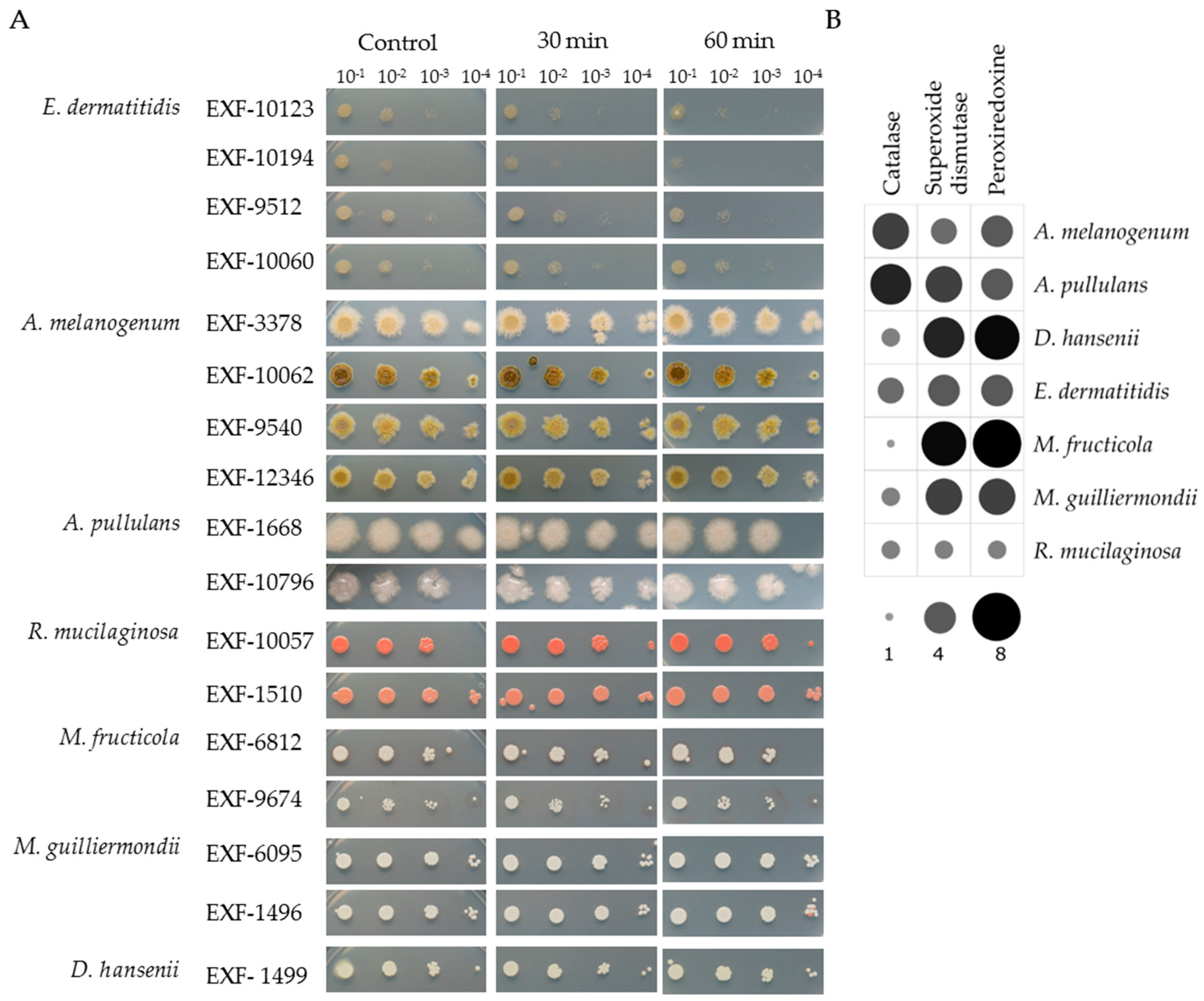
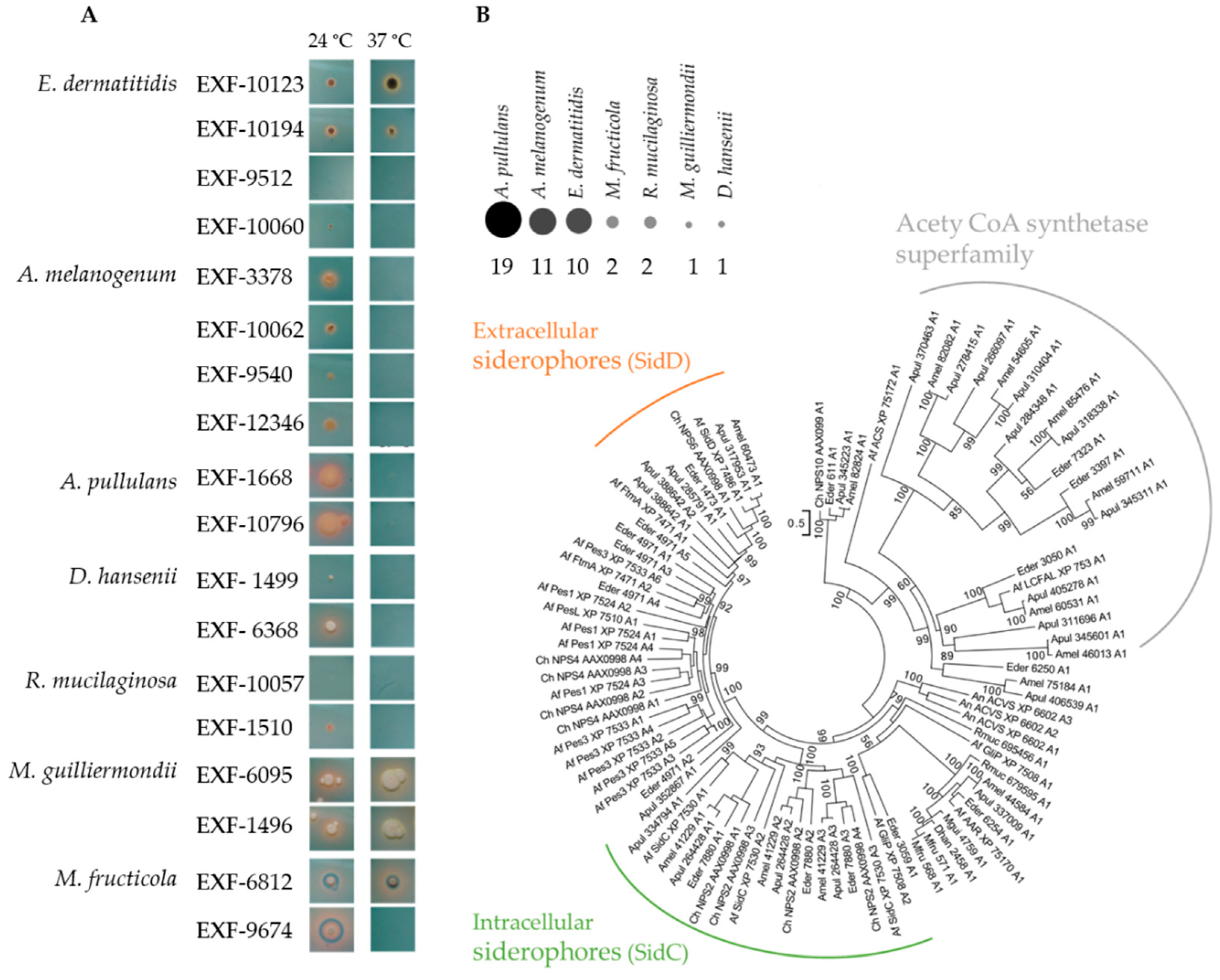
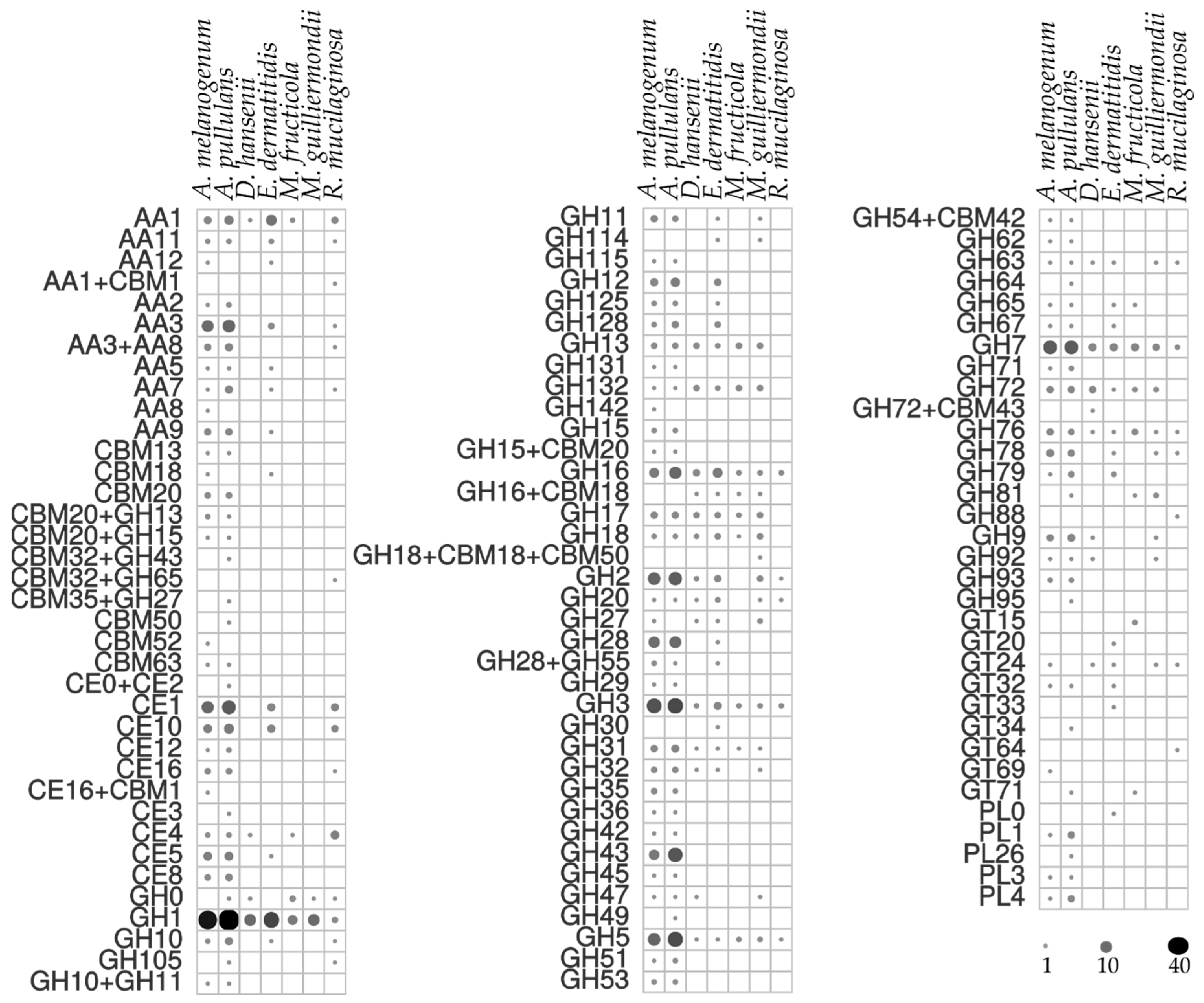
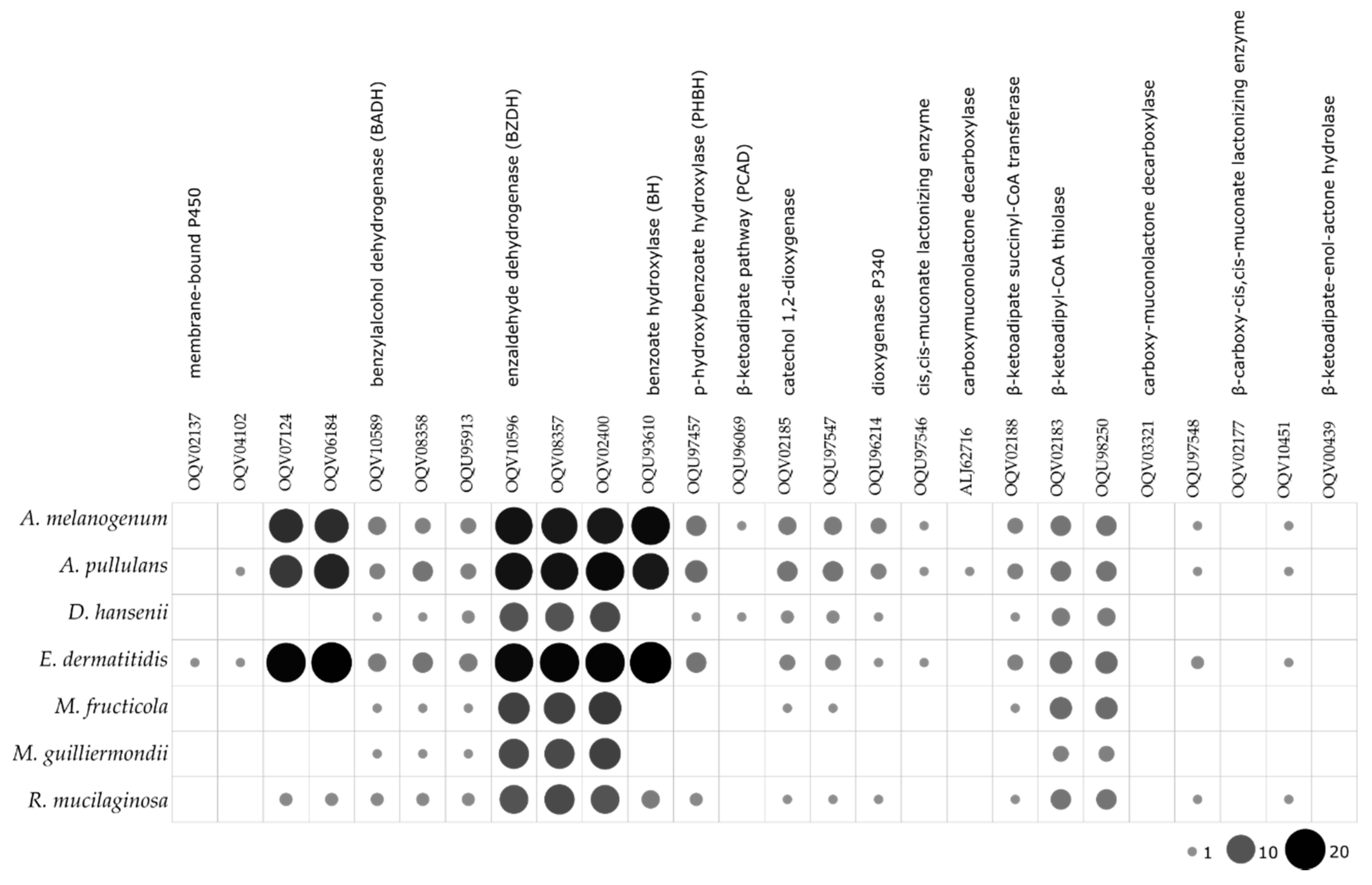
| Species | Accession Number | Source of Isolation |
|---|---|---|
| Exophiala dermatitidis | EXF-10123 (CBS 525.76) | Human |
| EXF-10194 | Patient with cystic fibrosis, Sweden | |
| EXF-9512 | Dishwasher rubber, Slovenia | |
| EXF-10060 | Tap water, Slovenia | |
| Aureobasidium melanogenum | EXF-3378 (CBS 110374) | Public fountain, Thailand |
| EXF-10062 | Tap water, Slovenia | |
| EXF-9540 | Dishwasher rubber, Slovenia | |
| EXF-12346 | Endoperitoneal fluid, India | |
| Aureobasidium pullulans | EXF-1668 | Glacial ice, Arctic |
| EXF-10796 | Grape surface, Slovenia | |
| Debaryomyces hansenii | EXF-1499 | Glacial ice, Arctic |
| EXF-6368 | Car fuel, Slovenia | |
| Rhodotorula mucilaginosa | EXF-10057 | Tap water, Slovenia |
| EXF-1510 | Glacial ice, Arctic | |
| Metschnikowia fructicola | EXF-6812 | Grape surface, Slovenia |
| EXF-9674 | Dishwasher drain, Slovenia | |
| Meyerozyma guilliermondii | EXF-6095 | Dishwasher rubber seal, Slovenia |
| EXF-1496 | Glacial ice, Arctic |
| Species | 24 °C | 37 °C | |||||
|---|---|---|---|---|---|---|---|
| Accession Number | YNB | 1/100 YNB | Agar | YNB | 1/100 YNB | Agar | |
| Exophiala dermatitidis | EXF-10123 | + | + | + | + | + | + |
| EXF-10194 | + | + | + | + | + | + | |
| EXF-9512 | + | + | + | + | + | + | |
| EXF-10060 | + | + | + | + | + | + | |
| Aureobasidium melanogenum | EXF-3378 | + | + | + | + | + | + |
| EXF-10062 | + | + | + | + | + | + | |
| EXF-9540 | + | + | + | + | + | + | |
| EXF-12346 | + | + | + | + | + | + | |
| Aureobasidium pullulans | EXF-1668 | + | + | + | - | - | - |
| EXF-10796 | + | + | + | - | - | - | |
| Debaryomyces hansenii | EXF-1499 | + | + | + | - | - | - |
| EXF-6368 | + | +/- | +/- | - | - | - | |
| Rhodotorula mucilaginosa | EXF-10057 | + | + | + | + | + | + |
| EXF-1510 | + | + | + | + | + | + | |
| Metchnikowia fructicola | EXF-6812 | + | + | + | + | + | - |
| EXF-9674 | + | + | + | +/- | +/- | - | |
| Meyerozyma guilliermondii | EXF-6095 | + | + | + | + | + | + |
| EXF-1496 | + | + | + | + | + | + | |
| Species | Strain | Enzymatic Activity | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amylases (EI) | Pectinase (EI) | Cellulase (EI) | Xylanase (EI) | Chitinase | β-Glucosidase | Esterase | Caseinase | Gelatinase | Keratinase | Urease | ||
| Exophiala dermatitidis | EXF-10123 | - | - | 1.25 ± 0.16 | 1.12 ± 0.21 | - | - | +/− | - | - | - | + |
| EXF-10194 | - | - | 1.48 ± 0.28 | +/− | - | - | +/− | - | - | - | +/− | |
| EXF-9512 | - | - | 1.45 ± 0.90 | 1.29 ± 0.14 | - | - | +/− | - | - | - | + | |
| EXF-10060 | - | - | 1.46 ± 0.11 | 1.22 ± 0.14 | - | - | + | - | - | - | + | |
| Aureobasidium melanogenum | EXF-3378 | - | +/− | 1.42 ± 0.27 | - | + | + | + | + | + | - | + |
| EXF-10062 | 1.30 ± 0.28 | 1.35 ± 0.30 | 1.31 ± 0.14 | - | + | + | + | + | + | - | + | |
| EXF-9540 | - | 1.23 ± 0.10 | 1.3 2± 0.2 | +/− | + | + | + | + | - | - | + | |
| EXF-12346 | - | 1.23 ± 0.20 | 1.42 ± 0.33 | 1.18 ± 0.27 | + | + | + | + | + | - | + | |
| Aureobasidium pullulans | EXF-1668 | 1.30 ± 0.08 | 1.38 ± 0.18 | 1.86 ± 0.67 | - | + | + | + | + | + | - | + |
| EXF-10796 | 1.19 ± 0.12 | 1.23 ± 0.06 | 1.56 ± 0.51 | - | + | + | + | + | + | - | + | |
| Debaryomyces hansenii | EXF-1499 | - | 1.33 ± 0.12 | 1.20 ± 0.00 | 1.25 ± 0.00 | - | - | +/− | - | + | - | - |
| EXF-6368 | - | - | 1.19 ± 0.17 | - | + | - | + | - | - | - | - | |
| Rhodotorula mucilaginosa | EXF-10057 | - | - | 1.31 ± 0.22 | 1.60 ± 0.44 | - | - | + | - | - | - | + |
| EXF-1510 | +/− | - | 1.29 ± 0.47 | +/− | - | - | + | - | - | - | + | |
| Metschnikowia fructicola | EXF-6812 | +/− | - | - | +/− | - | - | + | - | - | - | - |
| EXF-9674 | - | - | - | - | + | - | + | + | - | - | - | |
| Meyerozyma guilliermondii | EXF-6095 | - | - | - | - | + | - | + | - | - | - | - |
| EXF-1496 | - | - | +/− | - | + | - | + | - | - | - | - | |
| Species | Strain | Assimilation Activity | ||
|---|---|---|---|---|
| Toluene | n-Hexadecane | Mineral Oil | ||
| Exophiala dermatitidis | EXF-10123 | + | + | + |
| EXF-10194 | + | - | - | |
| EXF-9512 | + | - | - | |
| EXF-10060 | + | - | + | |
| Aureobasidium melanogenum | EXF-3378 | + | - | - |
| EXF-10062 | + | - | - | |
| EXF-9540 | + | - | + | |
| EXF-12346 | + | - | + | |
| Aureobasidium pullulans | EXF-1668 | + | - | - |
| EXF-10796 | + | - | - | |
| Debaryomyces hansenii | EXF-1499 | - | - | - |
| EXF-6368 | - | + | - | |
| Rhodotorula mucilaginosa | EXF-10057 | + | - | - |
| EXF-1510 | + | - | - | |
| Metschnikowia fructicola | EXF-6812 | - | + | - |
| EXF-9674 | - | + | - | |
| Meyerozyma guilliermondii | EXF-6095 | - | + | + |
| EXF-1496 | - | + | + | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zajc, J.; Gostinčar, C.; Černoša, A.; Gunde-Cimerman, N. Stress-Tolerant Yeasts: Opportunistic Pathogenicity Versus Biocontrol Potential. Genes 2019, 10, 42. https://doi.org/10.3390/genes10010042
Zajc J, Gostinčar C, Černoša A, Gunde-Cimerman N. Stress-Tolerant Yeasts: Opportunistic Pathogenicity Versus Biocontrol Potential. Genes. 2019; 10(1):42. https://doi.org/10.3390/genes10010042
Chicago/Turabian StyleZajc, Janja, Cene Gostinčar, Anja Černoša, and Nina Gunde-Cimerman. 2019. "Stress-Tolerant Yeasts: Opportunistic Pathogenicity Versus Biocontrol Potential" Genes 10, no. 1: 42. https://doi.org/10.3390/genes10010042
APA StyleZajc, J., Gostinčar, C., Černoša, A., & Gunde-Cimerman, N. (2019). Stress-Tolerant Yeasts: Opportunistic Pathogenicity Versus Biocontrol Potential. Genes, 10(1), 42. https://doi.org/10.3390/genes10010042







