Schwann Cell Role in Selectivity of Nerve Regeneration
Abstract
:1. Introduction
2. Current Strategies to Improve Functional Recovery After Peripheral Nerve Injury
3. Preferential Motor Reinnervation
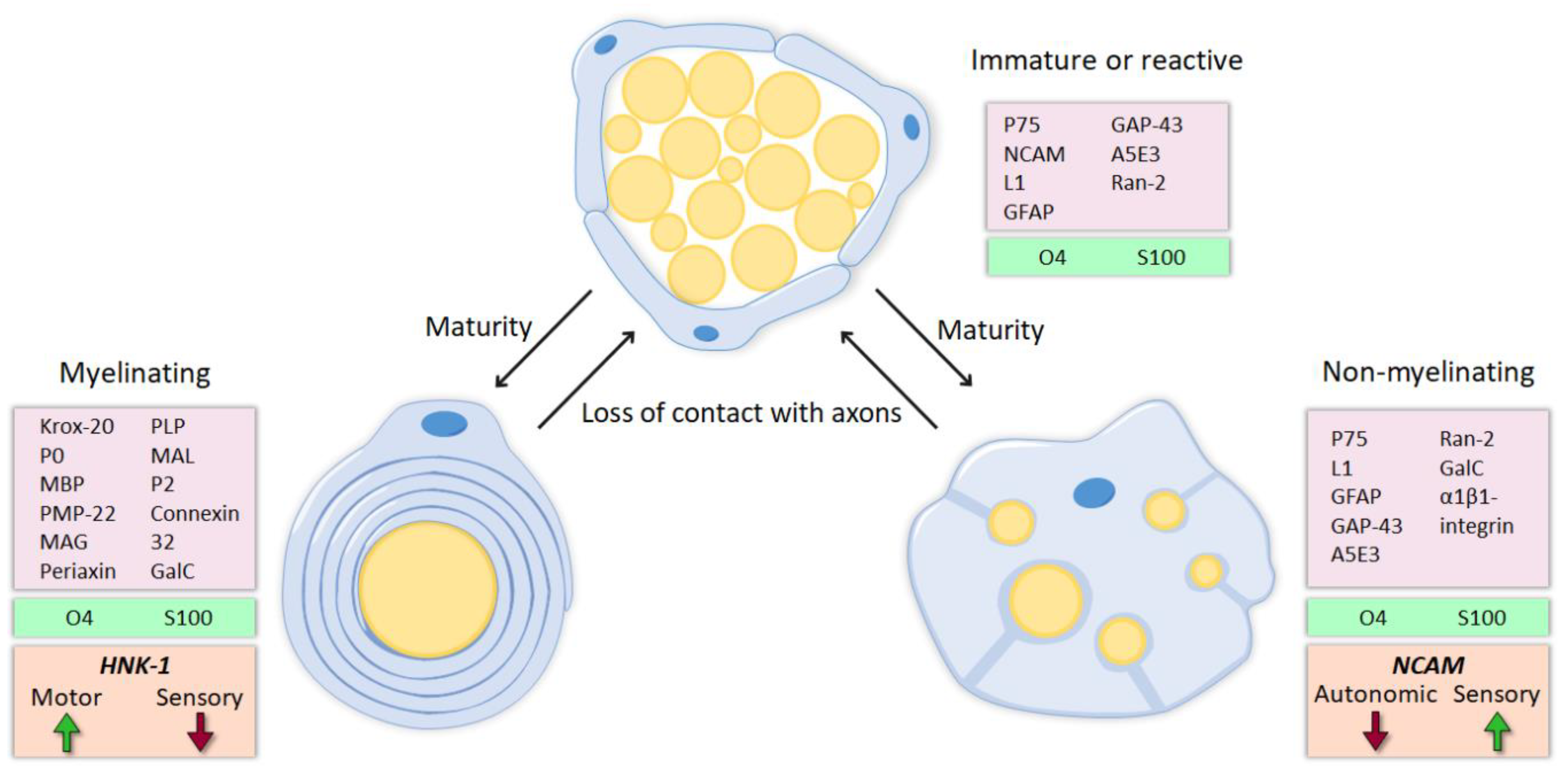
4. Motor and Sensory Schwann Cells
4.1. Molecular Identity
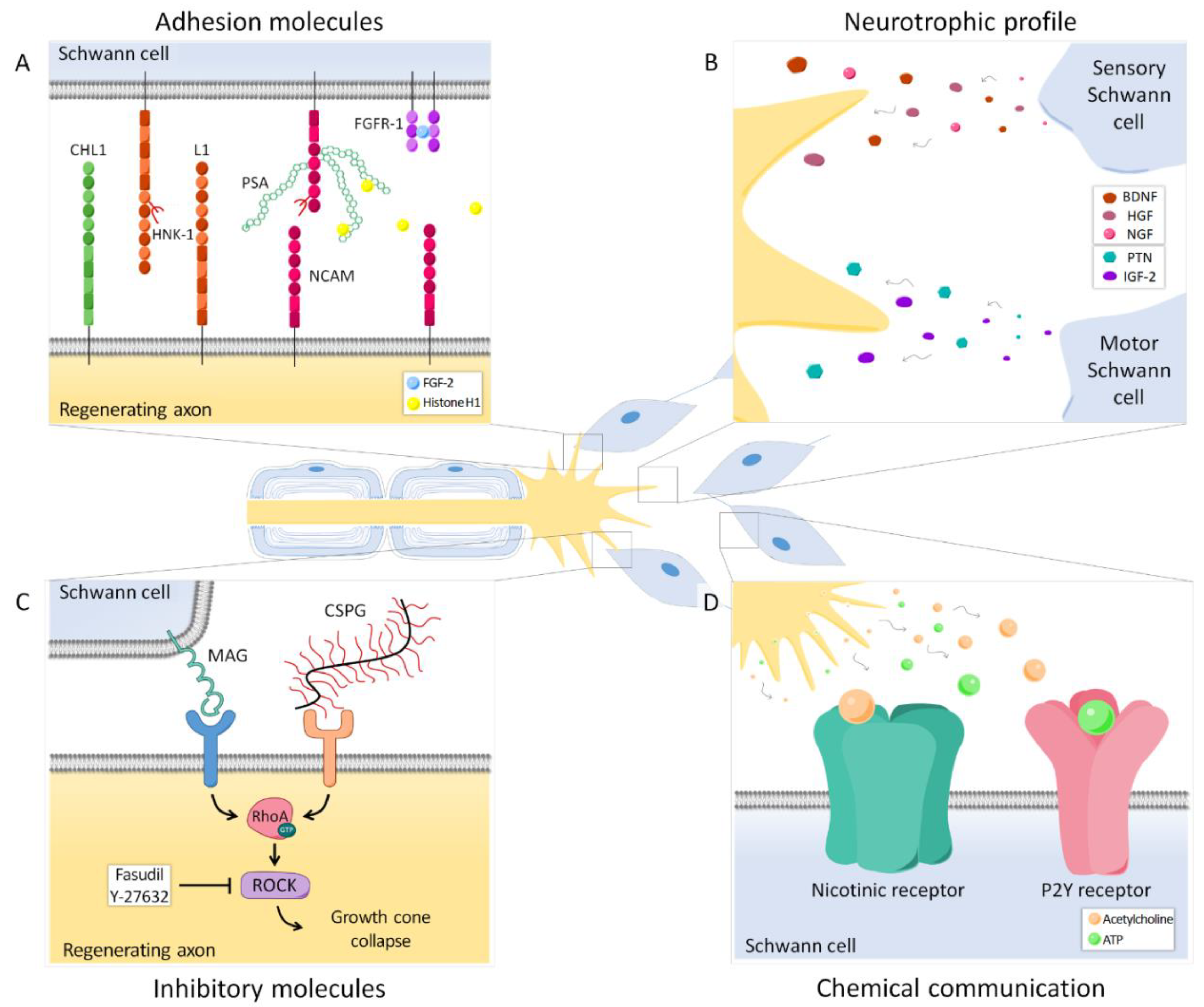
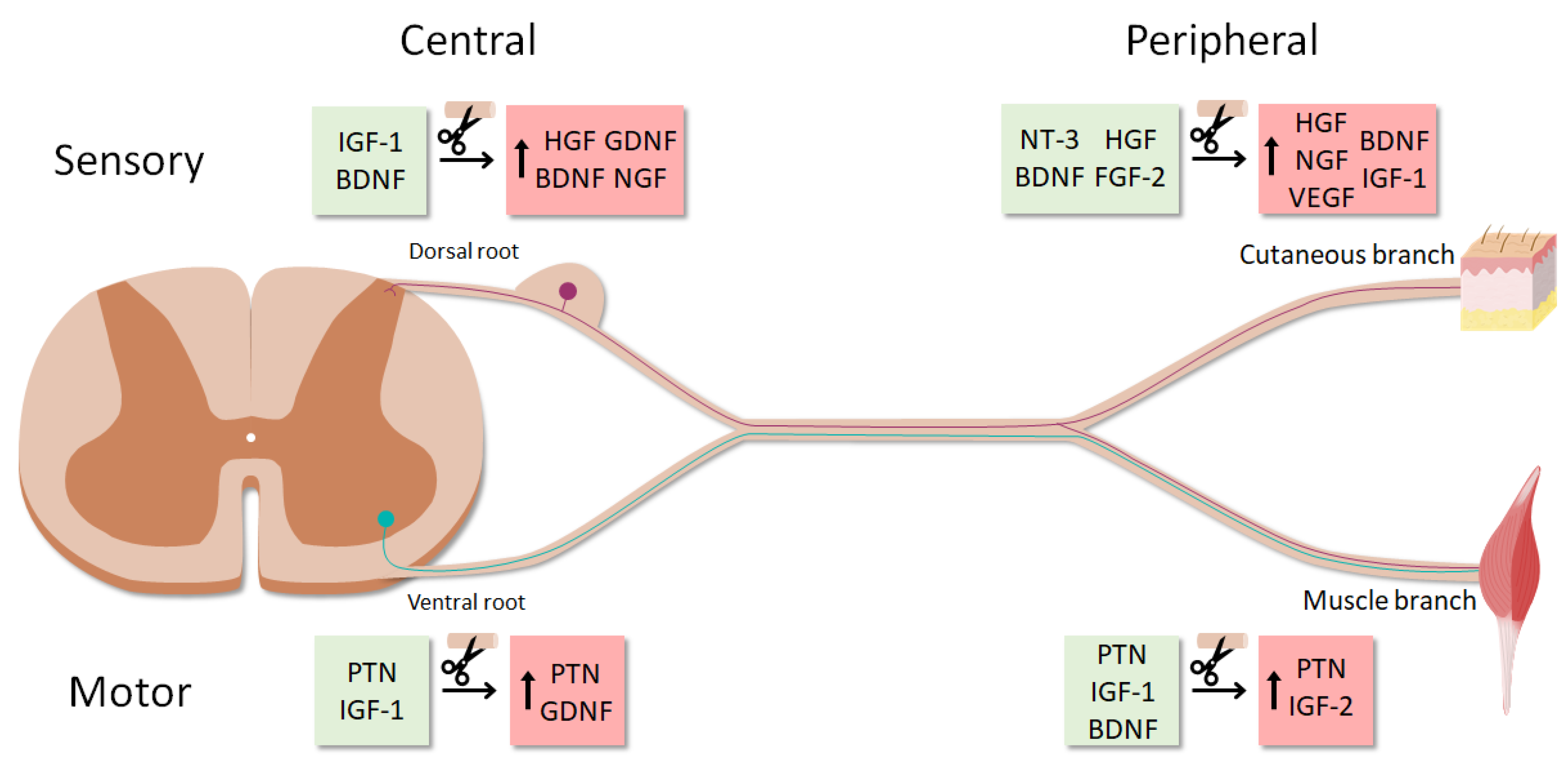
4.2. Neurotrophic Profile
4.3. Gene Expression
4.4. Phenotypic Memory
5. Adhesion Molecules
5.1. Polysialic Acid
5.2. Other Adhesion Molecules
6. Inhibitory Molecules
7. Electrical Stimulation
8. Conclusions
Funding
Conflicts of Interest
References
- Costigan, M.; Befort, K.; Karchewski, L.; Griffin, R.S.; D’Urso, D.; Allchorne, A.; Sitarski, J.; Mannion, J.W.; Pratt, R.E.; Woolf, C.J. Replicate high-density rat genome oligonucleotide microarrays reveal hundreds of regulated genes in the dorsal root ganglion after peripheral nerve injury. BMC Neurosci. 2002, 3, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Allodi, I.; Udina, E.; Navarro, X. Specificity of peripheral nerve regeneration: Interactions at the axon level. Prog. Neurobiol. 2012, 98, 16–37. [Google Scholar] [CrossRef] [PubMed]
- Jessen, K.R.; Mirsky, R. Signals that determine Schwann cell identity. J. Anat. 2002, 200, 367–376. [Google Scholar] [CrossRef] [PubMed]
- Jessen, K.R.; Mirsky, R. Negative regulation of myelination: Relevance for development, injury, and demyelinating disease. Glia 2008, 56, 1552–1565. [Google Scholar] [CrossRef]
- Jessen, K.R.; Mirsky, R. The repair Schwann cell and its function in regenerating nerves. J. Physiol. 2016, 594, 3521–3531. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jessen, K.R.; Mirsky, R. The origin and development of glial cells in peripheral nerves. Nat. Rev. Neurosci. 2005, 6, 671–682. [Google Scholar] [CrossRef]
- Michailov, G.V.; Sereda, M.W.; Brinkmann, B.G.; Fischer, T.M.; Haug, B.; Birchmeier, C.; Role, L.; Lai, C.; Schwab, M.H. Axonal Neuregulin-1 Regulates Myelin Sheath Thickness. Science 2004, 304, 700–704. [Google Scholar] [CrossRef] [Green Version]
- Bosse, F.; Hasenpusch-Theil, K.; Küry, P.; Müller, H.W. Gene expression profiling reveals that peripheral nerve regeneration is a consequence of both novel injury-dependent and reactivated developmental processes. J. Neurochem. 2006, 96, 1441–1457. [Google Scholar] [CrossRef]
- Arthur-Farraj, P.J.; Latouche, M.; Wilton, D.K.; Quintes, S.; Chabrol, E.; Banerjee, A.; Woodhoo, A.; Jenkins, B.; Rahman, M.; Turmaine, M.; et al. c-Jun Reprograms Schwann Cells of Injured Nerves to Generate a Repair Cell Essential for Regeneration. Neuron 2012, 75, 633–647. [Google Scholar] [CrossRef] [Green Version]
- Welleford, A.S.; Quintero, J.E.; El Seblani, N.; Blalock, E.; Gunewardena, S.; Shapiro, S.M.; Riordan, S.M.; Huettl, P.; Guduru, Z.; Stanford, J.A.; et al. RNA Sequencing of Human Peripheral Nerve in Response to Injury: Distinctive Analysis of the Nerve Repair Pathways. Cell Transplant. 2020, 29, 1–13. [Google Scholar] [CrossRef]
- Yi, S.; Zhang, H.; Gong, L.; Wu, J.; Zha, G.; Zhou, S.; Gu, X.; Yu, B. Deep Sequencing and Bioinformatic Analysis of Lesioned Sciatic Nerves after Crush Injury. PLoS ONE 2015, 10, e0143491. [Google Scholar] [CrossRef] [PubMed]
- Arthur-Farraj, P.J.; Morgan, C.C.; Adamowicz, M.; Gomez-Sanchez, J.A.; Fazal, S.V.; Beucher, A.; Razzaghi, B.; Mirsky, R.; Jessen, K.R.; Aitman, T.J. Changes in the Coding and Non-coding Transcriptome and DNA Methylome that Define the Schwann Cell Repair Phenotype after Nerve Injury. Cell Rep. 2017, 20, 2719–2734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stassart, R.M.; Fledrich, R.; Velanac, V.; Brinkmann, B.G.; Schwab, M.H.; Meijer, D.; Sereda, M.W.; Nave, K.-A. A role for Schwann cell-derived neuregulin-1 in remyelination. Nat. Neurosci. 2013, 16, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Brück, W. The role of macrophages in Wallerian degeneration. Brain Pathol. 1997, 7, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Sanchez, J.A.; Pilch, K.S.; Van Der Lans, M.; Fazal, S.V.; Benito, C.; Wagstaff, L.J.; Mirsky, R.; Jessen, K.R. After nerve injury, lineage tracing shows that myelin and Remak Schwann cells elongate extensively and branch to form repair Schwann cells, which shorten radically on remyelination. J. Neurosci. 2017, 37, 9086–9099. [Google Scholar] [CrossRef] [PubMed]
- Bodine-Fowler, S.C.; Meyer, R.S.; Moskovitz, A.; Abrams, R.; Botte, M.J. Inaccurate projection of rat soleus motoneurons: A comparison of nerve repair techniques. Muscle and Nerve 1997, 20, 29–37. [Google Scholar] [CrossRef]
- Valero-Cabré, A.; Navarro, X. Functional impact of axonal misdirection after peripheral nerve injuries followed by graft or tube repair. J. Neurotrauma 2002, 19, 1475–1485. [Google Scholar] [CrossRef]
- Chan, K.M.; Gordon, T.; Zochodne, D.W.; Power, H.A. Improving peripheral nerve regeneration: From molecular mechanisms to potential therapeutic targets. Exp. Neurol. 2014, 261, 826–835. [Google Scholar] [CrossRef]
- Mason, M.R.J.; Tannemaat, M.R.; Malessy, M.J.A.; Verhaagen, J. Gene Therapy for the Peripheral Nervous System: A Strategy to Repair the Injured Nerve? Curr. Gene Ther. 2011, 11, 75–89. [Google Scholar] [CrossRef]
- Udina, E.; Cobianchi, S.; Allodi, I.; Navarro, X. Effects of activity-dependent strategies on regeneration and plasticity after peripheral nerve injuries. Ann. Anat. 2011, 193, 347–353. [Google Scholar] [CrossRef]
- Duraikannu, A.; Krishnan, A.; Chandrasekhar, A.; Zochodne, D.W. Beyond Trophic Factors: Exploiting the Intrinsic Regenerative Properties of Adult Neurons. Front. Cell. Neurosci. 2019, 13, 1–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Navarro, X.; VerdÚ, E.; ButÍ, M. Comparison of regenerative and reinnervating capabilities of different functional types of nerve fibers. Exp. Neurol. 1994, 129, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.H.E.; Omura, T.; Cobos, E.J.; Latrémolière, A.; Ghasemlou, N.; Brenner, G.J.; Van Veen, E.; Barrett, L.; Sawada, T.; Gao, F.; et al. Accelerating axonal growth promotes motor recovery after peripheral nerve injury in mice. J. Clin. Investig. 2011, 121, 4332–4347. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abe, N.; Borson, S.H.; Gambello, M.J.; Wang, F.; Cavalli, V. Mammalian Target of Rapamycin (mTOR) activation increases axonal growth capacity of injured peripheral nerves. J. Biol. Chem. 2010, 285, 28034–28043. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cattin, A.L.; Burden, J.J.; Van Emmenis, L.; MacKenzie, F.E.; Hoving, J.J.A.; Garcia Calavia, N.; Guo, Y.; McLaughlin, M.; Rosenberg, L.H.; Quereda, V.; et al. Macrophage-Induced Blood Vessels Guide Schwann Cell-Mediated Regeneration of Peripheral Nerves. Cell 2015, 162, 1127–1139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Navarro, X.; Verdú, E. Cell transplants and artificial guides for nerve repair. In Brain Damage and Repair: From Molecular Research to Clinical Therapy; Herdegen, T., Delgado-García, J.M., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2004; pp. 451–471. ISBN 978-1-4020-1892-3. [Google Scholar]
- Pfister, B.J.; Gordon, T.; Loverde, J.R.; Kochar, A.S.; Mackinnon, S.E.; Kacy Cullen, D. Biomedical engineering strategies for peripheral nerve repair: Surgical applications, state of the art, and future challenges. Crit. Rev. Biomed. Eng. 2011, 39, 81–124. [Google Scholar] [CrossRef]
- Philips, C.; Cornelissen, M.; Carriel, V. Evaluation methods as quality control in the generation of decellularized peripheral nerve allografts. J. Neural Eng. 2018, 15, 021003. [Google Scholar] [CrossRef]
- Lundborg, G. A 25-year perspective of peripheral nerve surgery: Evolving neuroscientific concepts and clinical significance. J. Hand Surg. Am. 2000, 25, 391–414. [Google Scholar] [CrossRef]
- Hussain, G.; Wang, J.; Rasul, A.; Anwar, H.; Qasim, M.; Zafar, S.; Aziz, N.; Razzaq, A.; Hussain, R.; de Aguilar, J.L.G.; et al. Current status of therapeutic approaches against peripheral nerve injuries: A detailed story from injury to recovery. Int. J. Biol. Sci. 2020, 16, 116–134. [Google Scholar] [CrossRef] [Green Version]
- Sarker, M.D.; Naghieh, S.; McInnes, A.D.; Schreyer, D.J.; Chen, X. Regeneration of peripheral nerves by nerve guidance conduits: Influence of design, biopolymers, cells, growth factors, and physical stimuli. Prog. Neurobiol. 2018, 171, 125–150. [Google Scholar] [CrossRef]
- Li, R.; Li, D.; Zhang, H.; Wang, J.; Li, X.; Xiao, J. Growth factors-based therapeutic strategies and their underlying signaling mechanisms for peripheral nerve regeneration. Acta Pharmacol. Sin. 2020, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Han, G.H.; Peng, J.; Liu, P.; Ding, X.; Wei, S.; Lu, S.; Wang, Y. Therapeutic strategies for peripheral nerve injury: Decellularized nerve conduits and Schwann cell transplantation. Neural Regen. Res. 2019, 14, 1343–1351. [Google Scholar] [PubMed]
- Yousefi, F.; Lavi Arab, F.; Nikkhah, K.; Amiri, H.; Mahmoudi, M. Novel approaches using mesenchymal stem cells for curing peripheral nerve injuries. Life Sci. 2019, 221, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Brushart, T.M.E.; Seiler IV, W.A. Selective reinnervation of distal motor stumps by peripheral motor axons. Exp. Neurol. 1987, 97, 289–300. [Google Scholar] [CrossRef]
- Brushart, T. Preferential reinnervation of motor nerves by regenerating motor axons. J. Neurosci. 1988, 8, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Robinson, G.A.; Madison, R.D. Developmentally regulated changes in femoral nerve regeneration in the mouse and rat. Exp. Neurol. 2006, 197, 341–346. [Google Scholar] [CrossRef]
- Brushart, T.M.; Gerber, J.; Kessens, P.; Chen, Y.G.; Royall, R.M. Contributions of pathway and neuron to preferential motor reinnervation. J. Neurosci. 1998, 18, 8674–8681. [Google Scholar] [CrossRef]
- Robinson, G.A.; Madison, R.D. Preferential motor reinnervation in the mouse: Comparison of femoral nerve repair using a fibrin sealant or suture. Muscle Nerve 2003, 28, 227–231. [Google Scholar] [CrossRef]
- Uschold, T.; Robinson, G.A.; Madison, R.D. Motor neuron regeneration accuracy: Balancing trophic influences between pathways and end-organs. Exp. Neurol. 2007, 205, 250–256. [Google Scholar] [CrossRef]
- Robinson, G.A.; Madison, R.D. Manipulations of the mouse femoral nerve influence the accuracy of pathway reinnervation by motor neurons. Exp. Neurol. 2005, 192, 39–45. [Google Scholar] [CrossRef]
- Madison, R.D.; Sofroniew, M.V.; Robinson, G.A. Schwann cell influence on motor neuron regeneration accuracy. Neuroscience 2009, 163, 213–221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robinson, G.A.; Madison, R.D. Motor neurons can preferentially reinnervate cutaneous pathways. Exp. Neurol. 2004, 190, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Madison, R.D.; Robinson, G.A.; Chadaram, S.R. The specificity of motor neurone regeneration (preferential reinnervation). Acta Physiol. 2007, 189, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Madison, R.D.; McGee, C.; Rawson, R.; Robinson, G.A. Extracellular vesicles from a muscle cell line (C2C12) enhance cell survival and neurite outgrowth of a motor neuron cell line (NSC-34). J. Extracell. Vesicles 2014, 3, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, Y.; Maki, Y.; Yoshizu, T.; Tajima, T. Both stump area and volume of distal sensory nerve segments influence the regeneration of sensory axons in rats. Scand. J. Plast. Reconstr. Surg. Hand Surg. 1999, 33, 177–180. [Google Scholar]
- Robinson, G.A.; Madison, R.D. Influence of terminal nerve branch size on motor neuron regeneration accuracy. Exp. Neurol. 2009, 215, 228–235. [Google Scholar] [CrossRef]
- Tsubokawa, N.; Maki, Y.; Yoshizu, T.; Narisawa, H. Comparison of the neurotropic effects of motor and sensory Schwann cells during regeneration of peripheral nerves. Scand. J. Plast. Reconstr. Surg. Hand Surg. 1999, 33, 379–385. [Google Scholar]
- Brushart, T.M.E. Motor axons preferentially reinnervate motor pathways. J. Neurosci. 1993, 13, 2730–2738. [Google Scholar] [CrossRef]
- Redett, R.; Jari, R.; Crawford, T.; Chen, Y.G.; Rohde, C.; Brushart, T.M. Peripheral pathways regulate motoneuron collateral dynamics. J. Neurosci. 2005, 25, 9406–9412. [Google Scholar] [CrossRef]
- Witzel, C.; Rohde, C.; Brushart, T.M. Pathway sampling by regenerating peripheral axons. J. Comp. Neurol. 2005, 485, 183–190. [Google Scholar] [CrossRef]
- Smalheiser, N.R.; Crain, S.M. The possible role of “sibling neurite bias” in the coordination of neurite extension, branching, and survival. J. Neurobiol. 1984, 15, 517–529. [Google Scholar] [CrossRef] [PubMed]
- Martini, R.; Xin, Y.; Schmitz, B.; Schachner, M. The L2/HNK-1 Carbohydrate Epitope is Involved in the Preferential Outgrowth of Motor Neurons on Ventral Roots and Motor Nerves. Eur. J. Neurosci. 1992, 4, 628–639. [Google Scholar] [CrossRef] [PubMed]
- Martini, R.; Schachner, M.; Brushart, T. The L2/HNK-1 carbohydrate is preferentially expressed by previously motor axon-associated Schwann cells in reinnervated peripheral nerves. J. Neurosci. 1994, 14, 7180–7191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saito, H.; Nakao, Y.; Takayama, S.; Toyama, Y.; Asou, H. Specific expression of an HNK-1 carbohydrate epitope and NCAM on femoral nerve Schwann cells in mice. Neurosci. Res. 2005, 53, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Saito, H.; Kanje, M.; Dahlin, L.B. Crossed over repair of the femoral sensory and motor branches influences N-CAM. Neuroreport 2010, 21, 841–845. [Google Scholar] [CrossRef]
- Höke, A.; Redett, R.; Hameed, H.; Jari, R.; Zhou, C.; Li, Z.B.; Griffin, J.W.; Brushart, T.M. Schwann cells express motor and sensory phenotypes that regulate axon regeneration. J. Neurosci. 2006, 26, 9646–9655. [Google Scholar] [CrossRef] [Green Version]
- Brushart, T.M.; Aspalter, M.; Griffin, J.W.; Redett, R.; Hameed, H.; Zhou, C.; Wright, M.; Vyas, A.; Höke, A. Schwann cell phenotype is regulated by axon modality and central–peripheral location, and persists in vitro. Exp. Neurol. 2013, 247, 272–281. [Google Scholar] [CrossRef] [Green Version]
- Mi, R.; Chen, W.; Höke, A. Pleiotrophin is a neurotrophic factor for spinal motor neurons. Proc. Natl. Acad. Sci. USA 2007, 104, 4664–4669. [Google Scholar] [CrossRef] [Green Version]
- Fontenas, L.; Kucenas, S. Motor exit point (MEP) glia: Novel myelinating glia that bridge CNS and PNS myelin. Front. Cell. Neurosci. 2018, 12, 1–8. [Google Scholar] [CrossRef]
- Allodi, I.; Casals-Díaz, L.; Santos-Nogueira, E.; Gonzalez-Perez, F.; Navarro, X.; Udina, E. FGF-2 low molecular weight selectively promotes neuritogenesis of motor neurons in vitro. Mol. Neurobiol. 2013, 47, 770–781. [Google Scholar] [CrossRef]
- Santos, D.; Gonzalez-Perez, F.; Navarro, X.; Del Valle, J. Dose-dependent differential effect of neurotrophic factors on in vitro and in vivo regeneration of motor and sensory neurons. Neural Plast. 2016, 2016, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos, D.; González-Pérez, F.; Giudetti, G.; Micera, S.; Udina, E.; Del Valle, J.; Navarro, X. Preferential enhancement of sensory and motor axon regeneration by combining extracellular matrix components with neurotrophic factors. Int. J. Mol. Sci. 2017, 18, 65. [Google Scholar] [CrossRef] [PubMed]
- Del Valle, J.; Santos, D.; Delgado-Martínez, I.; de la Oliva, N.; Giudetti, G.; Micera, S.; Navarro, X. Segregation of motor and sensory axons regenerating through bicompartmental tubes by combining extracellular matrix components with neurotrophic factors. J. Tissue Eng. Regen. Med. 2018, 12, e1991–e2000. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, L.; Esch, F.S.; Marchionni, M.A.; Mudge, A.W. Control of Schwann cell survival and proliferation: Autocrine factors and neuregulins. Mol. Cell. Neurosci. 1998, 12, 141–156. [Google Scholar] [CrossRef] [PubMed]
- Jesuraj, N.J.; Nguyen, P.K.; Wood, M.D.; Moore, A.M.; Borschel, G.H.; Mackinnon, S.E.; Sakiyama-Elbert, S.E. Differential gene expression in motor and sensory Schwann cells in the rat femoral nerve. J. Neurosci. Res. 2012, 90, 96–104. [Google Scholar] [CrossRef] [Green Version]
- He, Q.; Man, L.; Ji, Y.; Ding, F. Comparison in the biological characteristics between primary cultured sensory and motor Schwann cells. Neurosci. Lett. 2012, 521, 57–61. [Google Scholar] [CrossRef]
- Kawamura, D.H.; Johnson, P.J.; Moore, A.M.; Magill, C.K.; Hunter, D.A.; Ray, W.Z.; Tung, T.H.H.; Mackinnon, S.E. Matching of motor-sensory modality in the rodent femoral nerve model shows no enhanced effect on peripheral nerve regeneration. Exp. Neurol. 2010, 223, 496–504. [Google Scholar] [CrossRef] [Green Version]
- Ali, S.A.; Rosko, A.J.; Hanks, J.E.; Stebbins, A.W.; Alkhalili, O.; Hogikyan, N.D.; Feldman, E.L.; Brenner, M.J. Effect of Motor versus Sensory Nerve Autografts on Regeneration and Functional Outcomes of Rat Facial Nerve Reconstruction. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Nichols, C.M.; Brenner, M.J.; Fox, I.K.; Tung, T.H.; Hunter, D.A.; Rickman, S.R.; Mackinnon, S.E. Effects of motor versus sensory nerve grafts on peripheral nerve regeneration. Exp. Neurol. 2004, 190, 347–355. [Google Scholar] [CrossRef]
- Brenner, M.J.; Hess, J.R.; Myckatyn, T.M.; Hayashi, A.; Hunter, D.A.; Mackinnon, S.E. Repair of motor nerve gaps with sensory nerve inhibits regeneration in rats. Laryngoscope 2006, 116, 1685–1692. [Google Scholar] [CrossRef] [Green Version]
- Lago, N.; Rodríguez, F.J.; Guzmán, M.S.; Jaramillo, J.; Navarro, X. Effects of motor and sensory nerve transplants on amount and specificity of sciatic nerve regeneration. J. Neurosci. Res. 2007, 85, 2800–2812. [Google Scholar] [CrossRef] [PubMed]
- Marquardt, L.M.; Sakiyama-Elbert, S.E. GDNF preconditioning can overcome Schwann cell phenotypic memory. Exp. Neurol. 2015, 265, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarhane, K.A.; Tuffaha, S.H.; Ibrahim, Z.; Cashman, C.R.; Krick, K.; Martin, R.; Broyles, J.M.; Cooney, D.S.; Lee, W.P.A.; Mi, R.; et al. Glial Cell Line–Derived Neurotrophic Factor and Chondroitinase Promote Axonal Regeneration in a Chronic Denervation Animal Model. Neurotherapeutics 2019, 16, 1283–1295. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Zhang, C.; Yu, Z.; Li, W.; Huang, Z.; Zhang, W. GDNF pretreatment overcomes Schwann cell phenotype mismatch to promote motor axon regeneration via sensory graft. Exp. Neurol. 2019, 318, 258–266. [Google Scholar] [CrossRef]
- Jesuraj, N.J.; Marquardt, L.M.; Kwasa, J.A.; Sakiyama-Elbert, S.E. Glial cell line-derived neurotrophic factor promotes increased phenotypic marker expression in femoral sensory and motor-derived Schwann cell cultures. Exp. Neurol. 2014, 257, 10–18. [Google Scholar] [CrossRef] [Green Version]
- Rutishauser, U. Polysialic acid in the plasticity of the developing and adult vertebrate nervous system. Nat. Rev. Neurosci. 2008, 9, 26–35. [Google Scholar] [CrossRef]
- Franz, C.K.; Rutishauser, U.; Rafuse, V.F. Polysialylated neural cell adhesion molecule is necessary for selective targeting of regenerating motor neurons. J. Neurosci. 2005, 25, 2081–2091. [Google Scholar] [CrossRef] [Green Version]
- Robinson, G.A.; Madison, R.D. Polysialic acid expression is not necessary for motor neuron target selectivity. Muscle Nerve 2013, 47, 364–371. [Google Scholar] [CrossRef] [Green Version]
- Franz, C.K.; Rutishauser, U.; Rafuse, V.F. Intrinsic neuronal properties control selective targeting of regenerating motoneurons. Brain 2008, 131, 1492–1505. [Google Scholar] [CrossRef] [Green Version]
- Allodi, I.; Mecollari, V.; González-Pérez, F.; Eggers, R.; Hoyng, S.; Verhaagen, J.; Navarro, X.; Udina, E. Schwann cells transduced with a lentiviral vector encoding Fgf-2 promote motor neuron regeneration following sciatic nerve injury. Glia 2014, 62, 1736–1746. [Google Scholar] [CrossRef]
- Mehanna, A.; Mishra, B.; Kurschat, N.; Schulze, C.; Bian, S.; Loers, G.; Irintchev, A.; Schachner, M. Polysialic acid glycomimetics promote myelination and functional recovery after peripheral nerve injury in mice. Brain 2009, 132, 1449–1462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mishra, B.; Von Der Ohe, M.; Schulze, C.; Bian, S.; Makhina, T.; Loers, G.; Kleene, R.; Schachner, M. Functional role of the interaction between polysialic acid and extracellular histone H1. J. Neurosci. 2010, 30, 12400–12413. [Google Scholar] [CrossRef] [PubMed]
- Simova, O.; Irintchev, A.; Mehanna, A.; Liu, J.; Dihné, M.; Bächle, D.; Sewald, N.; Loers, G.; Schachner, M. Carbohydrate mimics promote functional recovery after peripheral nerve repair. Ann. Neurol. 2006, 60, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Guseva, D.; Loers, G.; Schachner, M. Function-triggering antibodies to the adhesion molecule L1 enhance recovery after injury of the adult mouse femoral nerve. PLoS ONE 2014, 9, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guseva, D.; Angelov, D.N.; Irintchev, A.; Schachner, M. Ablation of adhesion molecule L1 in mice favours Schwann cell proliferation and functional recovery after peripheral nerve injury. Brain 2009, 132, 2180–2195. [Google Scholar] [CrossRef] [Green Version]
- Hillenbrand, R.; Molthagen, M.; Montag, D.; Schachner, M. The close homologue of the neural adhesion molecule L1 (CHL1): Patterns of expression and promotion of neurite outgrowth by heterophilic interactions. Eur. J. Neurosci. 1999, 11, 813–826. [Google Scholar] [CrossRef]
- Katic, J.; Loers, G.; Kleene, R.; Karl, N.; Schmidt, C.; Buck, F.; Zmijewski, J.W.; Jakovcevski, I.; Preissner, K.T.; Schachner, M. Interaction of the Cell Adhesion Molecule CHL1 with Vitronectin, Integrins, and the Plasminogen Activator Inhibitor-2 Promotes CHL1-Induced Neurite Outgrowth and Neuronal Migration. J. Neurosci. 2014, 34, 14606–14623. [Google Scholar] [CrossRef] [Green Version]
- Guseva, D.; Jakovcevski, I.; Irintchev, A.; Leshchyns’Ka, I.; Sytnyk, V.; Ponimaskin, E.; Schachner, M. Cell adhesion molecule close homolog of L1 (CHL1) guides the regrowth of regenerating motor axons and regulates synaptic coverage of motor neurons. Front. Mol. Neurosci. 2018, 11, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Vrbova, G.; Mehra, N.; Shanmuganathan, H.; Tyreman, N.; Schachner, M.; Gordon, T. Chemical communication between regenerating motor axons and Schwann cells in the growth pathway. Eur. J. Neurosci. 2009, 30, 366–375. [Google Scholar] [CrossRef]
- Torigoe, K.; Lundborg, G. Selective inhibition of early axonal regeneration by myelin-associated glycoprotein. Exp. Neurol. 1998, 150, 254–262. [Google Scholar] [CrossRef]
- Mukhopadhyay, G.; Doherty, P.; Walsh, F.S.; Crocker, P.R.; Filbin, M.T. A novel role for myelin-associated glycoprotein as an inhibitor of axonal regeneration. Neuron 1994, 13, 757–767. [Google Scholar] [CrossRef]
- Fruttiger, M.; Montag, D.; Schachner, M.; Martini, R. Crucial Role for the Myelin-associated Glycoprotein in the Maintenance of Axon-Myelin Integrity. Eur. J. Neurosci. 1995, 7, 511–515. [Google Scholar] [CrossRef] [PubMed]
- Mears, S.; Schachner, M.; Brushart, T.M. Antibodies to myelin-associated glycoprotein accelerate preferential motor reinnervation. J. Peripher. Nerv. Syst. 2003, 8, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Zuo, J.; Hernandez, Y.J.; Muir, D. Chondroitin sulfate proteoglycan with neurite-inhibiting activity is up-regulated following peripheral nerve injury. J. Neurobiol. 1998, 34, 41–54. [Google Scholar] [CrossRef]
- Zuo, J.; Neubauer, D.; Graham, J.; Krekoski, C.A.; Ferguson, T.A.; Muir, D. Regeneration of axons after nerve transection repair is enhanced by degradation of chondroitin sulfate proteoglycan. Exp. Neurol. 2002, 176, 221–228. [Google Scholar] [CrossRef]
- Niederöst, B.; Oertle, T.; Fritsche, J.; McKinney, R.A.; Bandtlow, C.E. Nogo-A and myelin-associated glycoprotein mediate neurite growth inhibition by antagonistic regulation of RhoA and Rac1. J. Neurosci. 2002, 22, 10368–10376. [Google Scholar] [CrossRef]
- Walker, B.A.; Ji, S.J.; Jaffrey, S.R. Intra-axonal translation of RhoA promotes axon growth inhibition by CSPG. J. Neurosci. 2012, 32, 14442–14447. [Google Scholar] [CrossRef] [Green Version]
- Joshi, A.R.; Bobylev, I.; Zhang, G.; Sheikh, K.A.; Lehmann, H.C. Inhibition of Rho-kinase differentially affects axon regeneration of peripheral motor and sensory nerves. Exp. Neurol. 2015, 263, 28–38. [Google Scholar] [CrossRef]
- Gordon, T.; English, A.W. Strategies to promote peripheral nerve regeneration: Electrical stimulation and/or exercise. Eur. J. Neurosci. 2016, 43, 336–350. [Google Scholar] [CrossRef] [Green Version]
- Al-Majed, A.A.; Neumann, C.M.; Brushart, T.M.; Gordon, T. Brief electrical stimulation promotes the speed and accuracy of motor axonal regeneration. J. Neurosci. 2000, 20, 2602–2608. [Google Scholar] [CrossRef]
- Brushart, T.M.; Jari, R.; Verge, V.; Rohde, C.; Gordon, T. Electrical stimulation restores the specificity of sensory axon regeneration. Exp. Neurol. 2005, 194, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Al-Majed, A.A.; Brushart, T.M.; Gordon, T. Electrical stimulation accelerates and increases expression of BDNF and trkB mRNA in regenerating rat femoral motoneurons. Eur. J. Neurosci. 2000, 12, 4381–4390. [Google Scholar] [PubMed]
- Wenjin, W.; Wenchao, L.; Hao, Z.; Feng, L.; Yan, W.; Wodong, S.; Xianqun, F.; Wenlong, D. Electrical Stimulation Promotes BDNF Expression in Spinal Cord Neurons Through Ca2+- and Erk-Dependent Signaling Pathways. Cell. Mol. Neurobiol. 2011, 31, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Eberhardt, K.A.; Irintchev, A.; Al-Majed, A.A.; Simova, O.; Brushart, T.M.; Gordon, T.; Schachner, M. BDNF/TrkB signaling regulates HNK-1 carbohydrate expression in regenerating motor nerves and promotes functional recovery after peripheral nerve repair. Exp. Neurol. 2006, 198, 500–510. [Google Scholar] [CrossRef] [PubMed]
- Geremia, N.M.; Gordon, T.; Brushart, T.M.; Al-Majed, A.A.; Verge, V.M.K. Electrical stimulation promotes sensory neuron regeneration and growth-associated gene expression. Exp. Neurol. 2007, 205, 347–359. [Google Scholar] [CrossRef]
- Al-Majed, A.A.; Siu, L.T.; Gordon, T. Electrical stimulation accelerates and enhances expression of regeneration-associated genes in regenerating rat femoral motoneurons. Cell. Mol. Neurobiol. 2004, 24, 379–402. [Google Scholar] [CrossRef]
- Brushart, T.M.; Hoffman, P.N.; Royall, R.M.; Murinson, B.B.; Witzel, C.; Gordon, T. Electrical stimulation promotes motoneuron regeneration without increasing its speed or conditioning the neuron. J. Neurosci. 2002, 22, 6631–6638. [Google Scholar] [CrossRef] [Green Version]
- Angelov, D.; Neiss, W.; Streppel, M.; Andermahr, J.; Mader, K.; Stennert, E. Nimodipine accelerates axonal sprouting after surgical repair of rat facial nerve. J. Neurosci. 1996, 16, 1041–1048. [Google Scholar] [CrossRef]
- Koppes, A.N.; Nordberg, A.L.; Paolillo, G.; Goodsell, N.; Darwish, H.; Zhang, L.; Thompson, D.M. Electrical Stimulation of Schwann Cells Promotes Sustained Increases in Neurite Outgrowth. Tissue Eng. Part A 2013, 20, 494–506. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.; Ye, Z.; Hu, X.; Lu, L.; Luo, Z. Electrical stimulation induces calcium-dependent release of NGF from cultured Schwann cells. Glia 2009, 58, 622–631. [Google Scholar] [CrossRef]
- Cobianchi, S.; Casals-Diaz, L.; Jaramillo, J.; Navarro, X. Differential effects of activity dependent treatments on axonal regeneration and neuropathic pain after peripheral nerve injury. Exp. Neurol. 2013, 240, 157–167. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bolívar, S.; Navarro, X.; Udina, E. Schwann Cell Role in Selectivity of Nerve Regeneration. Cells 2020, 9, 2131. https://doi.org/10.3390/cells9092131
Bolívar S, Navarro X, Udina E. Schwann Cell Role in Selectivity of Nerve Regeneration. Cells. 2020; 9(9):2131. https://doi.org/10.3390/cells9092131
Chicago/Turabian StyleBolívar, Sara, Xavier Navarro, and Esther Udina. 2020. "Schwann Cell Role in Selectivity of Nerve Regeneration" Cells 9, no. 9: 2131. https://doi.org/10.3390/cells9092131





