The State of Art of Regenerative Therapy in Cardiovascular Ischemic Disease: Biology, Signaling Pathways, and Epigenetics of Endothelial Progenitor Cells
Abstract
1. Endothelial Progenitor Cells (EPCs): Definition
2. Origin and Biological Significance of EPCs
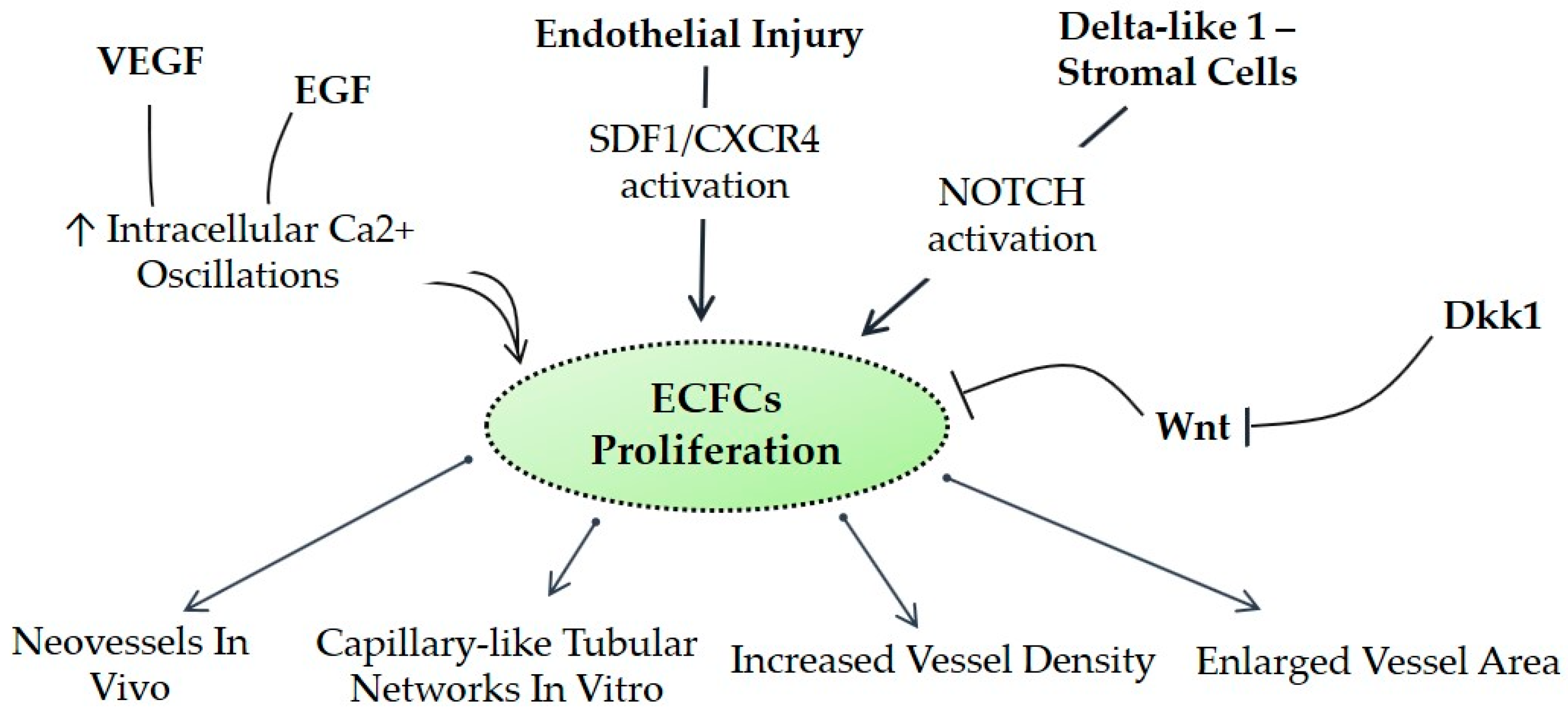
3. Signaling Pathways Driven by and Affecting EPCs
3.1. ECFCs and Calcium Homeostasis
3.2. NOTCH
3.3. VEGFR
3.4. WNT
3.5. CXCR4
4. Other Strategies in ECFC Modulation: Mesenchymal Stem Cells (MSCs) and Epigenetic
5. Myocardial Revascularization in Animal Models Using EPCs
6. Endothelial Progenitor Cells (EPCs) in Regenerative Medicine in Humans for Cardiovascular Disorders: State of the Art
7. Clinical Trials for Regenerative Medicine in Cardiovascular Ischemic Diseases Employing EPCs: Delivery Strategies
8. Limitations and Criticisms of Clinical Trials in the Field
Funding
Conflicts of Interest
Abbreviations
| ADSC | adipose-derived stromal cell |
| AMI | acute myocardial infarction |
| BM | bone marrow |
| BMEC | bone marrow endothelial cell |
| BMSC | bone marrow stem cell |
| BOEC | blood outgrowth endothelial cell |
| CABG | coronary artery bypass grafting |
| CAC | circulating angiogenic cell |
| CFU-Hill | colony-forming unit-Hill EPC |
| CPC | cardiac progenitor cell |
| CXCR4 | C-X-C chemokine receptor type 4 |
| CVD | cardiovascular disease |
| Dkk1 | dickkopf WNT signaling pathway inhibitor 1 |
| ECFC | endothelial colony-forming cell |
| EDTA | ethylenediaminetetraacetic acid |
| eEPC | early EPC |
| EGF | epidermal growth factor |
| EOC | endothelial outgrowth cell |
| EPC | endothelial progenitor cell |
| ER | endoplasmic reticulum |
| G-CSF | granulocyte-macrophage colony-stimulating factor |
| KDR | kinase insert domain receptor |
| lEPC | late EPC |
| HGF | hepatocyte growth factor |
| HPP | high proliferative potential |
| LV | left ventricle |
| LVEF | left ventricle ejection fraction |
| ICM | ischemic cardiomyopathy |
| MAC | myeloid angiogenic cell |
| MNC | mononuclear cell |
| MRI | magnetic resonance imaging |
| MSC | mesenchymal stem cell |
| NCX | Na+/Ca2+ exchanger |
| OEC | outgrowth endothelial cell |
| PAC | pro-angiogenic hematopoietic cell |
| PPCI | primary percutaneous intervention |
| SDF-1 | stromal cell-derived factor 1 |
| SERCA | sarco-endoplasmic reticulum Ca2+ ATPase |
| SHH | sonic hedgehog |
| TNF | tumor necrosis factor |
| VEGF-A | vascular endothelial growth factor-A |
| VEGFR | vascular endothelial growth factor receptor |
| VESC | vascular endothelial stem cell |
| vWF | von Willebrand factor |
| Wnt | wingless-type mouse mammary tumor virus integration site family |
References
- Reya, T.; Morrison, S.J.; Clarke, M.F.; Weissman, I.L. Stem cells, cancer, and cancer stem cells. Nature 2001, 414, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Sukmawati, D.; Tanaka, R. Introduction to next generation of endothelial progenitor cell therapy: A promise in vascular medicine. Am. J. Transl. Res. 2015, 7, 411–421. [Google Scholar] [PubMed]
- Basile, D.P.; Yoder, M.C. Circulating and tissue resident endothelial progenitor cells. J. Cell. Physiol. 2014, 229, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Barber, D.L.; Wherry, E.J.; Masopust, D.; Zhu, B.; Allison, J.P.; Sharpe, A.H.; Freeman, G.J.; Ahmed, R. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 2005, 439, 682–687. [Google Scholar] [CrossRef] [PubMed]
- Asahara, T.; Murohara, T.; Sullivan, A.; Silver, M.; Van Der Zee, R.; Li, T.; Witzenbichler, B.; Schatteman, G.; Isner, J.M. Isolation of Putative Progenitor Endothelial Cells for Angiogenesis. Science 1997, 275, 964–966. [Google Scholar] [CrossRef] [PubMed]
- Medina, R.J.; O’Neill, C.L.; O’Doherty, T.M.; Wilson, S.E.J.; Stitt, A.W. Endothelial Progenitors as Tools to Study Vascular Disease. Stem Cells Int. 2012, 2012, 1–5. [Google Scholar] [CrossRef]
- Peichev, M.; Naiyer, A.J.; Pereira, D.; Zhu, Z.; Lane, W.J.; Williams, M.; Oz, M.C.; Hicklin, D.J.; Witte, L.; Moore, M.A.S.; et al. Expression of VEGFR-2 and AC133 by circulating human CD34+ cells identifies a population of functional endothelial precursors. Blood 2000, 95, 952–958. [Google Scholar] [CrossRef]
- Wu, X.; Lensch, M.W.; Wylie-Sears, J.; Daley, G.Q.; Bischoff, J. Hemogenic Endothelial Progenitor Cells Isolated from Human Umbilical Cord Blood. Stem Cells 2007, 25, 2770–2776. [Google Scholar] [CrossRef]
- Medina, R.J.; O’Neill, C.L.; Sweeney, M.; Guduric-Fuchs, J.; Gardiner, T.A.; Simpson, D.A.; Stitt, A.W. Molecular analysis of endothelial progenitor cell (EPC) subtypes reveals two distinct cell populations with different identities. BMC Med. Genom. 2010, 3, 18. [Google Scholar] [CrossRef]
- Asahara, T.; Kawamoto, A.; Masuda, H. Concise Review: Circulating Endothelial Progenitor Cells for Vascular Medicine. Stem Cells 2011, 29, 1650–1655. [Google Scholar] [CrossRef]
- Medina, R.J.; Barber, C.L.; Sabatier, F.; Dignat-George, F.; Melero-Martin, J.M.; Khosrotehrani, K.; Ohneda, O.; Randi, A.M.; Chan, J.K.; Yamaguchi, T.; et al. Endothelial Progenitors: A Consensus Statement on Nomenclature. Stem Cells Transl. Med. 2017, 6, 1316–1320. [Google Scholar] [CrossRef] [PubMed]
- Prasain, N.; Meador, J.L.; Yoder, M.C. Phenotypic and functional characterization of endothelial colony forming cells derived from human umbilical cord blood. J. Vis. Exp. 2012, 62, e3872. [Google Scholar] [CrossRef]
- Medina, R.J.; O’Neill, C.L.; O’Doherty, T.M.; Knott, H.; Guduric-Fuchs, J.; Gardiner, T.A.; Stitt, A.W. Myeloid Angiogenic Cells Act as Alternative M2 Macrophages and Modulate Angiogenesis through Interleukin-8. Mol. Med. 2011, 17, 1045–1055. [Google Scholar] [CrossRef] [PubMed]
- Kanayasu-Toyoda, T.; Tanaka, T.; Kikuchi, Y.; Uchida, E.; Matsuyama, A.; Yamaguchi, T. Cell-surface MMP-9 protein is a novel functional marker to identify and separate pro-angiogenic cells from early endothelial progenitor cells derived from CD133+ cells. Stem Cells 2016, 34, 1251–1262. [Google Scholar] [CrossRef] [PubMed]
- Urbich, C.; Heeschen, C.; Aicher, A.; Dernbach, E.; Zeiher, A.M.; Dimmeler, S. Relevance of Monocytic Features for Neovascularization Capacity of Circulating Endothelial Progenitor Cells. Circulation 2003, 108, 2511–2516. [Google Scholar] [CrossRef]
- Ingram, D.A.; Mead, L.E.; Tanaka, H.; Meade, V.; Fenoglio, A.; Mortell, K.; Pollok, K.; Ferkowicz, M.J.; Gilley, D.; Yoder, M.C. Identification of a novel hierarchy of endothelial progenitor cells using human peripheral and umbilical cord blood. Blood 2004, 104, 2752–2760. [Google Scholar] [CrossRef]
- Poitevin, S.; Cussac, D.; Leroyer, A.; Albinet, V.; Sarlon-Bartoli, G.; Guillet, B.; Hubert, L.; Andrieu, N.; Couderc, B.; Parini, A.; et al. Sphingosine kinase 1 expressed by endothelial colony-forming cells has a critical role in their revascularization activity. Cardiovasc. Res. 2014, 103, 121–130. [Google Scholar] [CrossRef]
- Hur, J.; Yoon, C.H.; Kim, H.-S.; Choi, J.-H.; Kang, H.-J.; Hwang, K.-K.; Oh, B.-H.; Lee, M.-M.; Park, Y.-B. Characterization of Two Types of Endothelial Progenitor Cells and Their Different Contributions to Neovasculogenesis. Arter. Thromb. Vasc. Biol. 2004, 24, 288–293. [Google Scholar] [CrossRef]
- Yoder, M.C.; Mead, L.E.; Prater, D.; Krier, T.R.; Mroueh, K.N.; Li, F.; Krasich, R.; Temm, C.J.; Prchal, J.T.; Ingram, D.A. Redefining endothelial progenitor cells via clonal analysis and hematopoietic stem/progenitor cell principals. Blood 2006, 109, 1801–1809. [Google Scholar] [CrossRef]
- Kanzler, I.; Tuchscheerer, N.; Steffens, G.; Simsekyilmaz, S.; Konschalla, S.; Kroh, A.; Simons, D.; Asare, Y.; Schober, A.; Bucala, R.; et al. Differential roles of angiogenic chemokines in endothelial progenitor cell-induced angiogenesis. Basic Res. Cardiol. 2012, 108, 1–14. [Google Scholar] [CrossRef]
- Minami, Y.; Nakajima, T.; Ikutomi, M.; Morita, T.; Komuro, I.; Sata, M.; Sahara, M. Angiogenic potential of early and late outgrowth endothelial progenitor cells is dependent on the time of emergence. Int. J. Cardiol. 2015, 186, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Richards, W.O.; Prutzman, K.B.; O’Hea, M.F.; Audia, J.P.; Alvarez, D.F. Bariatric surgery improves the circulating numbers and biological activity of late outgrowth endothelial progenitor cells. Surg. Obes. Relat. Dis. 2014, 10, 906–913. [Google Scholar] [CrossRef] [PubMed]
- Melero-Martin, J.M.; Khan, Z.A.; Picard, A.; Wu, X.; Paruchuri, S.; Bischoff, J. In Vivo vasculogenic potential of human blood-derived endothelial progenitor cells. Blood 2007, 109, 4761–4768. [Google Scholar] [CrossRef] [PubMed]
- Tura, O.; Skinner, E.M.; Barclay, G.R.; Samuel, K.; Gallagher, R.C.; Brittan, M.; Hadoke, P.W.; Newby, D.E.; Turner, M.L.; Mills, N.L. Late Outgrowth Endothelial Cells Resemble Mature Endothelial Cells and Are Not Derived from Bone Marrow. Stem Cells 2013, 31, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Asahara, T.; Masuda, H.; Takahashi, T.; Kalka, C.; Pastore, C.; Silver, M.; Kearne, M.; Magner, M.; Isner, J.M. Bone marrow origin of endothelial progenitor cells responsible for postnatal vasculogenesis in physiological and pathological neovascularization. Circ. Res. 1999, 85, 221–228. [Google Scholar] [CrossRef]
- Guerra, G.; Perrotta, F.; Testa, G. Circulating Endothelial Progenitor Cells Biology and Regenerative Medicine in Pulmonary Vascular Diseases. Curr. Pharm. Biotechnol. 2018, 19, 700–707. [Google Scholar] [CrossRef]
- Takahashi, T.; Kalka, C.; Masuda, H.; Chen, D.; Silver, M.; Kearney, M.; Magner, M.; Isner, J.M.; Asahara, T. Ischemia- and cytokine-induced mobilization of bone marrow-derived endothelial progenitor cells for neovascularization. Nat. Med. 1999, 5, 434–438. [Google Scholar] [CrossRef]
- Hubert, L.; Darbousset, R.; Panicot-Dubois, L.; Robert, S.; Sabatier, F.; Fallague, K.; Dignat-George, F.; Dubois, C. Neutrophils recruit and activate human endothelial colony-forming cells at the site of vessel injury via P-selectin glycoprotein ligand-1 and L-selectin. J. Thromb. Haemost. 2014, 12, 1170–1181. [Google Scholar] [CrossRef]
- Liu, X.; Cho, W.C. Precision medicine in immune checkpoint blockade therapy for non-small cell lung cancer. Clin. Transl. Med. 2017, 6, 7. [Google Scholar] [CrossRef]
- Kang, K.-T.; Lin, R.-Z.; Kuppermann, D.; Melero-Martin, J.M.; Bischoff, J. Endothelial colony forming cells and mesenchymal progenitor cells form blood vessels and increase blood flow in ischemic muscle. Sci. Rep. 2017, 7, 770. [Google Scholar] [CrossRef]
- Moubarik, C.; Guillet, B.; Youssef, B.; Codaccioni, J.-L.; Piercecchi, M.-D.; Sabatier, F.; Lionel, P.; Dou, L.; Foucault-Bertaud, A.; Velly, L.; et al. Transplanted Late Outgrowth Endothelial Progenitor Cells as Cell Therapy Product for Stroke. Stem Cell Rev. Rep. 2010, 7, 208–220. [Google Scholar] [CrossRef] [PubMed]
- Nigro, E.; Stiuso, P.; Matera, M.; Monaco, M.L.; Caraglia, M.; Maniscalco, M.; Perrotta, F.; Mazzarella, G.; Daniele, A.; Bianco, A. The anti-proliferative effects of adiponectin on human lung adenocarcinoma A549 cells and oxidative stress involvement. Pulm. Pharmacol. Ther. 2019, 55, 25–30. [Google Scholar] [CrossRef]
- Lin, R.-Z.; Moreno-Luna, R.; Li, D.; Jaminet, S.-C.; Greene, A.K.; Melero-Martin, J.M. Human endothelial colony-forming cells serve as trophic mediators for mesenchymal stem cell engraftment via paracrine signaling. Proc. Natl. Acad. Sci. USA 2014, 111, 10137–10142. [Google Scholar] [CrossRef] [PubMed]
- Caplan, A.I. Mesenchymal Stem Cells: Time to Change the Name! Stem Cells Transl. Med. 2017, 6, 1445–1451. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, T.; Naito, H.; Suehiro, J.-I.; Lin, Y.; Kawaji, H.; Iba, T.; Kouno, T.; Ishikawa-Kato, S.; Furuno, M.; Takara, K.; et al. CD157 Marks Tissue-Resident Endothelial Stem Cells with Homeostatic and Regenerative Properties. Cell Stem Cell 2018, 22, 384–397.e6. [Google Scholar] [CrossRef]
- Chavakis, E.; Carmona, G.; Urbich, C.; Göttig, S.; Henschler, R.; Penninger, J.M.; Zeiher, A.M.; Chavakis, T.; Dimmeler, S. Phosphatidylinositol-3-Kinase-γ Is Integral to Homing Functions of Progenitor Cells. Circ. Res. 2008, 102, 942–949. [Google Scholar] [CrossRef]
- Cooke, J.P.; Losordo, D.W. Modulating the Vascular Response to Limb Ischemia. Circ. Res. 2015, 116, 1561–1578. [Google Scholar] [CrossRef]
- Carmeliet, P.; Jain, R.K. Molecular mechanisms and clinical applications of angiogenesis. Nature 2011, 473, 298–307. [Google Scholar] [CrossRef]
- Herbert, S.P.; Stainier, D.Y. Molecular control of endothelial cell behaviour during blood vessel morphogenesis. Nat. Rev. Mol. Cell Biol. 2011, 12, 551–564. [Google Scholar] [CrossRef]
- Le Bras, A.; Vijayaraj, P.; Oettgen, P. Molecular mechanisms of endothelial differentiation. Vasc. Med. 2010, 15, 321–331. [Google Scholar] [CrossRef]
- Moccia, F.; Guerra, G. Ca2+ Signalling in Endothelial Progenitor Cells: Friend or Foe? J. Cell. Physiol. 2015, 231, 314–327. [Google Scholar] [CrossRef] [PubMed]
- Guerra, G.; Lucariello, A.; Perna, A.; Botta, L.; De Luca, A.; Moccia, F. The Role of Endothelial Ca2+ Signaling in Neurovascular Coupling: A View from the Lumen. Int. J. Mol. Sci. 2018, 19, 938. [Google Scholar] [CrossRef] [PubMed]
- Zuccolo, E.; Lim, D.; Kheder, D.A.; Perna, A.; Catarsi, P.; Botta, L.; Rosti, V.; Riboni, L.; Sancini, G.; Tanzi, F.; et al. Acetylcholine induces intracellular Ca2+ oscillations and nitric oxide release in mouse brain endothelial cells. Cell Calcium 2017, 66, 33–47. [Google Scholar] [CrossRef] [PubMed]
- Zuccolo, E.; Kheder, D.A.; Lim, D.; Perna, A.; Di Nezza, F.; Botta, L.; Scarpellino, G.; Negri, S.; Martinotti, S.; Soda, T.; et al. Glutamate triggers intracellular Ca2+ oscillations and nitric oxide release by inducing NAADP- and InsP3 -dependent Ca2+ release in mouse brain endothelial cells. J. Cell. Physiol. 2018, 234, 3538–3554. [Google Scholar] [CrossRef] [PubMed]
- Moccia, F.; Berra-Romani, R.; Tritto, S.; Signorelli, S.; Taglietti, V.; Tanzi, F. Epidermal growth factor induces intracellular Ca2+ oscillations in microvascular endothelial cells. J. Cell. Physiol. 2003, 194, 139–150. [Google Scholar] [CrossRef]
- Potenza, D.M.; Guerra, G.; Avanzato, D.; Poletto, V.; Pareek, S.; Guido, D.; Gallanti, A.; Rosti, V.; Munaron, L.; Tanzi, F.; et al. Hydrogen sulphide triggers VEGF-induced intracellular Ca2+ signals in human endothelial cells but not in their immature progenitors. Cell Calcium 2014, 56, 225–234. [Google Scholar] [CrossRef]
- Kim, H.; Huang, L.; Critser, P.J.; Yang, Z.; Chan, R.J.; Wang, L.; Carlesso, N.; Voytik-Harbin, S.L.; Bernstein, I.D.; Yoder, M.C. Notch ligand Delta-like 1 promotes in vivo vasculogenesis in human cord blood–derived endothelial colony forming cells. Cytotherapy 2015, 17, 579–592. [Google Scholar] [CrossRef]
- Kwon, S.M.; Eguchi, M.; Wada, M.; Iwami, Y.; Hozumi, K.; Iwaguro, H.; Masuda, H.; Kawamoto, A.; Asahara, T. Specific Jagged-1 Signal From Bone Marrow Microenvironment is Required for Endothelial Progenitor Cell Development for Neovascularization. Circulation 2008, 118, 157–165. [Google Scholar] [CrossRef]
- Patel, J.; Wong, H.Y.; Wang, W.; Alexis, J.; Shafiee, A.; Stevenson, A.J.; Gabrielli, B.; Fisk, N.M.; Khosrotehrani, K. Self-Renewal and High Proliferative Colony Forming Capacity of Late-Outgrowth Endothelial Progenitors Is Regulated by Cyclin-Dependent Kinase Inhibitors Driven by Notch Signaling. Stem Cells 2016, 34, 902–912. [Google Scholar] [CrossRef]
- Rumjahn, S.M.; Yokdang, N.; Baldwin, K.A.; Thai, J.; Buxton, I.L.O. Purinergic regulation of vascular endothelial growth factor signaling in angiogenesis. Br. J. Cancer 2009, 100, 1465–1470. [Google Scholar] [CrossRef]
- Dragoni, S.; Laforenza, U.; Bonetti, E.; Lodola, F.; Bottino, C.; Berra-Romani, R.; Bongio, G.C.; Cinelli, M.P.; Guerra, G.; Pedrazzoli, P.; et al. Vascular Endothelial Growth Factor Stimulates Endothelial Colony Forming Cells Proliferation and Tubulogenesis by Inducing Oscillations in Intracellular Ca2+ Concentration. Stem Cells 2011, 29, 1898–1907. [Google Scholar] [CrossRef] [PubMed]
- Willert, K.; Brown, J.D.; Danenberg, E.; Duncan, A.W.; Weissman, I.L.; Reya, T.; Yates, J.R.; Nusse, R. Wnt proteins are lipid-modified and can act as stem cell growth factors. Nature 2003, 423, 448–452. [Google Scholar] [CrossRef] [PubMed]
- Esposito, T.; Lucariello, A.; Hay, E.; Contieri, M.; Tammaro, P.; Varriale, B.; Guerra, G.; De Luca, A.; Perna, A. Effects of curcumin and its adjuvant on TPC1 thyroid cell line. Chem. Interact. 2019, 305, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Perna, A.; De Luca, A.; Adelfi, L.; Pasquale, T.; Varriale, B.; Esposito, T. Effects of different extracts of curcumin on TPC1 papillary thyroid cancer cell line. BMC Complement. Altern. Med. 2018, 18, 1–9. [Google Scholar] [CrossRef]
- Smadja, D.M.; D’Audigier, C.; Weiswald, L.-B.; Badoual, C.; Dangles-Marie, V.; Mauge, L.; Evrard, S.; Laurendeau, I.; Lallemand, F.; Germain, S.; et al. The Wnt Antagonist Dickkopf-1 Increases Endothelial Progenitor Cell Angiogenic Potential. Arter. Thromb. Vasc. Biol. 2010, 30, 2544–2552. [Google Scholar] [CrossRef]
- Hattori, K.; Heissig, B.; Tashiro, K.; Honjo, T.; Tateno, M.; Shieh, J.H.; Hackett, N.R.; Quitoriano, M.S.; Crystal, R.G.; Rafii, S.; et al. Plasma elevation of stromal cell-derived factor-1 induces mobilization of mature and immature hematopoietic progenitor and stem cells. Blood 2001, 97, 3354–3360. [Google Scholar] [CrossRef]
- Sun, J.; Li, Y.; Graziani, G.M.; Filion, L.; Allan, D.S. E-Selectin Mediated Adhesion and Migration of Endothelial Colony Forming Cells Is Enhanced by SDF-1α/CXCR4. Plos ONE 2013, 8, e60890. [Google Scholar] [CrossRef]
- Oh, B.J.; Kim, D.K.; Kim, B.J.; Yoon, K.-S.; Park, S.G.; Park, K.S.; Lee, M.-S.; Kim, K.-W.; Kim, J.H. Differences in donor CXCR4 expression levels are correlated with functional capacity and therapeutic outcome of angiogenic treatment with endothelial colony forming cells. Biochem. Biophys. Res. Commun. 2010, 398, 627–633. [Google Scholar] [CrossRef]
- Cheng, L.; Lu, H.; Mei, H.; Tan, M. Human Adipose Derived Mesenchymal Stromal Cells Show More Efficient Angiogeneic Promotion on EPCs Than Umbilical Cord and Endometrium. Blood 2014, 124, 2765. [Google Scholar] [CrossRef]
- Shafiee, A.; Patel, J.; Wong, H.Y.; Donovan, P.; Hutmacher, D.W.; Fisk, N.M.; Khosrotehrani, K. Priming of endothelial colony-forming cells in a mesenchymal niche improves engraftment and vasculogenic potential by initiating mesenchymal transition orchestrated by NOTCH signaling. FASEB J. 2016, 31, 610–624. [Google Scholar] [CrossRef]
- Sun, R.; Li, X.; Liu, M.; Zeng, Y.; Chen, S.; Zhang, P. Advances in stem cell therapy for cardiovascular disease (Review). Int. J. Mol. Med. 2016, 38, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-R.; Tan, F.-Q.; Zhu, J.-Q.; Ye, T.; Wang, C.-L.; Zhu, Y.-F.; Dahms, H.-U.; Jin, F.; Yang, W.-X. Detection of DNA damage caused by cryopreservation using a modified SCGE in large yellow croaker, Pseudosciaena crocea. Acta Biol. Hung. 2014, 65, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, Z.; Zhang, D.Y.; Zhu, J.; Zhang, T.; Wang, C. microRNA 126 Inhibits the Transition of Endothelial Progenitor Cells to Mesenchymal Cells via the PIK3R2-PI3K/Akt Signalling Pathway. Plos ONE 2013, 8, e83294. [Google Scholar] [CrossRef] [PubMed]
- Fraineau, S.; Palii, C.G.; Allan, D.S.; Brand, M. Epigenetic regulation of endothelial-cell-mediated vascular repair. FEBS J. 2015, 282, 1605–1629. [Google Scholar] [CrossRef] [PubMed]
- Fraineau, S.; Palii, C.G.; McNeill, B.; Ritso, M.; Shelley, W.C.; Prasain, N.; Chu, A.; Vion, E.; Rieck, K.; Nilufar, S.; et al. Epigenetic Activation of Pro-angiogenic Signaling Pathways in Human Endothelial Progenitors Increases Vasculogenesis. Stem Cell Rep. 2017, 9, 1573–1587. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, T.; Kamon, J.; Ito, Y.; Tsuchida, A.; Yokomizo, T.; Kita, S.; Sugiyama, T.; Miyagishi, M.; Hara, K.; Tsunoda, M.; et al. Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature 2003, 423, 762–769. [Google Scholar] [CrossRef]
- Yang, J.; Yamato, M.; Kohno, C.; Nishimoto, A.; Sekine, H.; Fukai, F.; Okano, T. Cell sheet engineering: Recreating tissues without biodegradable scaffolds. Biomaterials 2005, 26, 6415–6422. [Google Scholar] [CrossRef]
- Dergilev, K.V.; Tsokolaeva, Z.; Makarevich, P.; Beloglazova, I.B.; Zubkova, E.; Boldyreva, M.A.; Ratner, E.; Dyikanov, D.; Menshikov, M.; Ovchinnikov, A.; et al. C-Kit Cardiac Progenitor Cell Based Cell Sheet Improves Vascularization and Attenuates Cardiac Remodeling following Myocardial Infarction in Rats. BioMed Res. Int. 2018, 2018, 1–13. [Google Scholar] [CrossRef]
- Hamdi, H.; Planat-Bénard, V.; Bel, A.; Puymirat, E.; Geha, R.; Pidial, L.; Nematalla, H.; Bellamy, V.; Bouaziz, P.; Peyrard, S.; et al. Epicardial adipose stem cell sheets results in greater post-infarction survival than intramyocardial injections. Cardiovasc. Res. 2011, 91, 483–491. [Google Scholar] [CrossRef]
- Kobayashi, H.; Shimizu, T.; Yamato, M.; Tono, K.; Masuda, H.; Asahara, T.; Kasanuki, H.; Okano, T. Fibroblast sheets co-cultured with endothelial progenitor cells improve cardiac function of infarcted hearts. J. Artif. Organs 2008, 11, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Atluri, P.; Panlilio, C.M.; Liao, G.P.; Hiesinger, W.; Harris, D.A.; McCormick, R.C.; Cohen, J.E.; Jin, T.; Feng, W.; Levit, R.D.; et al. Acute Myocardial Rescue with Endogenous Endothelial Progenitor Cell Therapy. Hear. Lung Circ. 2010, 19, 644–654. [Google Scholar] [CrossRef]
- Kamata, S.; Miyagawa, S.; Fukushima, S.; Nakatani, S.; Kawamoto, A.; Saito, A.; Harada, A.; Shimizu, T.; Daimon, T.; Okano, T.; et al. Improvement of Cardiac Stem Cell Sheet Therapy for Chronic Ischemic Injury by Adding Endothelial Progenitor Cell Transplantation: Analysis of Layer-Specific Regional Cardiac Function. Cell Transplant. 2014, 23, 1305–1319. [Google Scholar] [CrossRef] [PubMed]
- Mathiyalagan, P.; Liang, Y.; Kim, D.; Misener, S.; Thorne, T.; Kamide, C.E.; Klyachko, E.; Losordo, U.W.; Hajjar, R.J.; Sahoo, S.; et al. Angiogenic Mechanisms of Human CD34+ Stem Cell Exosomes in the Repair of Ischemic Hindlimb. Circ. Res. 2017, 120, 1466–1476. [Google Scholar] [CrossRef] [PubMed]
- Qu, Q.; Bing, W.; Meng, X.; Xi, J.; Bai, X.; Liu, Q.; Guo, Y.; Zhao, X.; Bi, Y. Upregulation of miR-126-3p promotes human saphenous vein endothelial cell proliferation in vitro and prevents vein graft neointimal formation ex vivo and in vivo. Oncotarget 2017, 8, 106790–106806. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liu, Q.; Wang, N.; Xu, Y.; Kang, L.; Ren, Y.; Zhu, G. Transplantation of Endothelial Progenitor Cells Overexpressing miR-126-3p Improves Heart Function in Ischemic Cardiomyopathy. Circ. J. 2018, 82, 2332–2341. [Google Scholar] [CrossRef]
- Li, J.; Wang, P.; Chen, Z.; Yu, S.; Xu, H. Fenofibrate Ameliorates Oxidative Stress-Induced Retinal Microvascular Dysfunction in Diabetic Rats. Curr. Eye Res. 2018, 43, 1–9. [Google Scholar] [CrossRef]
- Mathers, C.D.; Loncar, D. Projections of Global Mortality and Burden of Disease from 2002 to 2030. PLoS Med. 2006, 3, e442. [Google Scholar] [CrossRef]
- Gimbrone, M.A.; García-Cardeña, G. Endothelial Cell Dysfunction and the Pathobiology of Atherosclerosis. Circ. Res. 2016, 118, 620–636. [Google Scholar] [CrossRef]
- Woodward, L.; Akoumianakis, I.; Antoniades, C. Unravelling the adiponectin paradox: Novel roles of adiponectin in the regulation of cardiovascular disease. Br. J. Pharmacol. 2016, 174, 4007–4020. [Google Scholar] [CrossRef]
- Nigro, E.; Perrotta, F.; Monaco, M.L.; Polito, R.; Pafundi, P.C.; Matera, M.G.; Daniele, A.; Bianco, A. Implications of the Adiponectin System in Non-Small Cell Lung Cancer Patients: A Case-Control Study. Biomolecules 2020, 10, 926. [Google Scholar] [CrossRef]
- Perrotta, F.; Nigro, E.; Mollica, M.; Costigliola, A.; D’Agnano, V.; Daniele, A.; Bianco, A.; Guerra, G. Pulmonary Hypertension and Obesity: Focus on Adiponectin. Int. J. Mol. Sci. 2019, 20, 912. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.M.; Zalos, G.; Halcox, J.P.; Schenke, W.H.; Waclawiw, M.A.; Quyyumi, A.A.; Finkel, T. Circulating endothelial progenitor cells, vascular function, and cardiovascular risk. N. Engl. J. Med. 2003, 348, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Werner, N.; Wassmann, S.; Ahlers, P.; Schiegl, T.; Kosiol, S.; Link, A.; Walenta, K.; Nickenig, G. Endothelial progenitor cells correlate with endothelial function in patients with coronary artery disease. Basic Res. Cardiol. 2007, 102, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Massa, M.; Rosti, V.; Ferrario, M.; Campanelli, R.; Ramajoli, I.; Rosso, R.; De-Ferrari, G.M.; Ferlini, M.; Goffredo, L.; Bertoletti, A.; et al. Increased circulating hematopoietic and endothelial progenitor cells in the early phase of acute myocardial infarction. Blood 2005, 105, 199–206. [Google Scholar] [CrossRef]
- Urbich, C.; Aicher, A.; Heeschen, C.; Dernbach, E.; Hofmann, W.K.; Zeiher, A.M.; Dimmeler, S. Soluble factors released by endothelial progenitor cells promote migration of endothelial cells and cardiac resident progenitor cells. J. Mol. Cell. Cardiol. 2005, 39, 733–742. [Google Scholar] [CrossRef]
- Steinhoff, G.; Nesteruk, J.; Wolfien, M.; Kundt, G.; Börgermann, J.; David, R.; Garbade, J.; Große, J.; Haverich, A.; PERFECT Trial Investigators Group; et al. Cardiac Function Improvement and Bone Marrow Response—Outcome Analysis of the Randomized PERFECT Phase III Clinical Trial of Intramyocardial CD133+ Application After Myocardial Infarction. EBioMedicine 2017, 22, 208–224. [Google Scholar] [CrossRef]
- Turan, R.G.; Bozdag-T, I.; Ortak, J.; Kische, S.; Akin, I.; Schneider, H.; Turan, C.H.; Rehders, T.C.; Rauchhaus, M.; Kleinfeldt, T.; et al. Improved Functional Activity of Bone Marrow Derived Circulating Progenitor Cells After Intra Coronary Freshly Isolated Bone Marrow Cells Transplantation in Patients with Ischemic Heart Disease. Stem Cell Rev. Rep. 2010, 7, 646–656. [Google Scholar] [CrossRef][Green Version]
- Kim, S.H.; Cho, J.H.; Lee, Y.H.; Lee, J.H.; Kim, S.S.; Kim, M.Y.; Lee, M.G.; Kang, W.Y.; Lee, K.S.; Ahn, Y.K.; et al. Improvement in Left Ventricular Function with Intracoronary Mesenchymal Stem Cell Therapy in a Patient with Anterior Wall ST-Segment Elevation Myocardial Infarction. Cardiovasc Drugs Ther. 2018, 32, 329–338. [Google Scholar] [CrossRef]
- Lee, F.-Y.; Chen, Y.-L.; Sung, P.-H.; Ma, M.-C.; Pei, S.-N.; Wu, C.-J.; Yang, C.-H.; Fu, M.; Ko, S.-F.; Leu, S.; et al. Intracoronary Transfusion of Circulation-Derived CD34+ Cells Improves Left Ventricular Function in Patients With End-Stage Diffuse Coronary Artery Disease Unsuitable for Coronary Intervention*. Crit. Care Med. 2015, 43, 2117–2132. [Google Scholar] [CrossRef]
- Quyyumi, A.; Kereiakes, D.; Shavelle, D.; Henry, T.; Denktas, A.; Abdel-Latif, A.; Toma, C.; Barsness, G.; Frohwein, S.; Schatz, R.; et al. One year follow-up results from preserve-ami: A randomized, double-blind, placebo controlled clinical trial of intracoronary infusion of autologous CD34+ cells in patients with left ventricular dysfunction post stemi. J. Am. Coll. Cardiol. 2015, 65, A1593. [Google Scholar] [CrossRef]
- Arbustini, E.; Narula, N.; Tavazzi, L.; Serio, A.; Grasso, M.; Favalli, V.; Bellazzi, R.; Tajik, J.A.; Bonow, R.O.; Fuster, V.; et al. The MOGE(S) Classification of Cardiomyopathy for Clinicians. J. Am. Coll. Cardiol. 2014, 64, 304–318. [Google Scholar] [CrossRef] [PubMed]
- Vrtovec, B.; Poglajen, G.; Lezaic, L.; Sever, M.; Domanoviç, D.; Cernelc, P.; Socan, A.; Schrepfer, S.; Torre-Amione, G.; Haddad, F.; et al. Effects of Intracoronary CD34+ Stem Cell Transplantation in Nonischemic Dilated Cardiomyopathy Patients. Circ. Res. 2013, 112, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Lezaic, L.; Socan, A.; Poglajen, G.; Peitl, P.K.; Sever, M.; Cukjati, M.; Černelč, P.; Wu, J.C.; Haddad, F.; Vrtovec, B. Intracoronary Transplantation of CD34+ Cells Is Associated With Improved Myocardial Perfusion in Patients With Nonischemic Dilated Cardiomyopathy. J. Card. Fail. 2015, 21, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Sant’Anna, R.T.; Kalil, R.A.K.; Neto, A.S.P.; Júnior, F.P.; Fracasso, J.; Sant’Anna, J.R.M.; Marques, M.; Markoski, M.; Prates, P.R.; Nardi, N.B.; et al. Global Contractility Increment in Nonischemic Dilated Cardiomyopathy after Free Wall-Only Intramyocardial Injection of Autologous Bone Marrow Mononuclear Cells: An Insight over Stem Cells Clinical Mechanism of Action. Cell Transplant. 2010, 19, 959–964. [Google Scholar] [CrossRef] [PubMed]
- Bartunek, J.; Vanderheyden, M.; Vandekerckhove, B.; Mansour, S.; De Bruyne, B.; De Bondt, P.; Van Haute, I.; Lootens, N.; Heyndrickx, G.; Wijns, W. Intracoronary injection of CD133-positive enriched bone marrow progenitor cells promotes cardiac recovery after recent myocardial infarction: Feasibility and safety. Circulation 2005, 112, I-178–I-183. [Google Scholar]
- Manginas, A.; Goussetis, E.; Koutelou, M.; Karatasakis, G.; Peristeri, I.; Theodorakos, A.; Leontiadis, E.; Plessas, N.; Theodosaki, M.; Graphakos, S.; et al. Pilot study to evaluate the safety and feasibility of intracoronary CD133+ and CD133− CD34+ cell therapy in patients with nonviable anterior myocardial infarction. Catheter. Cardiovasc. Interv. 2007, 69, 773–781. [Google Scholar] [CrossRef]
- Assmus, B.; Honold, J.; Schächinger, V.; Britten, M.B.; Fischer-Rasokat, U.; Lehmann, R.; Teupe, C.; Pistorius, K.; Martin, H.; Abolmaali, N.D.; et al. Transcoronary Transplantation of Progenitor Cells after Myocardial Infarction. New Engl. J. Med. 2006, 355, 1222–1232. [Google Scholar] [CrossRef]
- Salvatore, D.R.; Seeger, F.H.; Honold, J.; Fischer-Rasokat, U.; Lehmann, R.; Fichtlscherer, S.; Schächinger, V.; Dimmeler, S.; Zeiher, A.M.; Assmus, B. Procedural Safety and Predictors of Acute Outcome of Intracoronary Administration of Progenitor Cells in 775 Consecutive Procedures Performed for Acute Myocardial Infarction or Chronic Heart Failure. Circ. Cardiovasc. Interv. 2013, 6, 44–51. [Google Scholar] [CrossRef]
- Assmus, B.; Walter, D.H.; Seeger, F.H.; Leistner, D.; Steiner, J.; Ziegler, I.; Lutz, A.; Khaled, W.; Klotsche, J.; Tonn, T.; et al. Effect of Shock Wave–Facilitated Intracoronary Cell Therapy on LVEF in Patients With Chronic Heart Failure. JAMA 2013, 309, 1622–1631. [Google Scholar] [CrossRef]
- Choudry, F.; Hamshere, S.; Saunders, N.; Veerapen, J.; Bavnbek, K.; Knight, C.; Pellerin, D.; Locca, D.; Westwood, M.; Rakhit, R.; et al. A randomized double-blind control study of early intra-coronary autologous bone marrow cell infusion in acute myocardial infarction: The REGENERATE-AMI clinical trial. Eur. Hear. J. 2015, 37, 256–263. [Google Scholar] [CrossRef]
- Colombo, A.; Castellani, M.; Piccaluga, E.; Pusineri, E.; Palatresi, S.; Longari, V.; Canzi, C.; Sacchi, E.; Rossi, E.; Rech, R.; et al. Myocardial blood flow and infarct size after CD133+ cell injection in large myocardial infarction with good recanalization and poor reperfusion: Results from a randomized controlled trial. J. Cardiovasc. Med. 2011, 12, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Stamm, C.; Kleine, H.-D.; Choi, Y.-H.; Dunkelmann, S.; Lauffs, J.-A.; Lorenzen, B.; Dávid, Á.; Liebold, A.; Nienaber, C.; Zurakowski, D.; et al. Intramyocardial delivery of CD133+ bone marrow cells and coronary artery bypass grafting for chronic ischemic heart disease: Safety and efficacy studies. J. Thorac. Cardiovasc. Surg. 2007, 133, 717–725.e5. [Google Scholar] [CrossRef] [PubMed]
- Mathiasen, A.B.; Haack-Sørensen, M.; Jørgensen, E.; Kastrup, J. Autotransplantation of mesenchymal stromal cells from bone-marrow to heart in patients with severe stable coronary artery disease and refractory angina—Final 3-year follow-up. Int. J. Cardiol. 2013, 170, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Aoki, J.; Serruys, P.W.; Van Beusekom, H.; Ong, A.T.; McFadden, E.P.; Sianos, G.; Van Der Giessen, W.J.; Regar, E.; De Feyter, P.J.; Davis, H.R.; et al. Endothelial Progenitor Cell Capture by Stents Coated With Antibody Against CD34. J. Am. Coll. Cardiol. 2005, 45, 1574–1579. [Google Scholar] [CrossRef] [PubMed]
- Low, A.F.; Lee, C.-H.; Teo, S.-G.; Chan, M.Y.; Tay, E.; Lee, Y.-P.; Chong, E.; Co, M.; Hay, E.T.; Lim, Y.-T.; et al. Effectiveness and Safety of the Genous Endothelial Progenitor Cell-Capture Stent in Acute ST-Elevation Myocardial Infarction. Am. J. Cardiol. 2011, 108, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Wöhrle, J.; Birkemeyer, R.; Markovic, S.; Nguyen, T.-V.; Sinha, A.; Miljak, T.; Spiess, J.; Rottbauer, W.; Rittger, H. Prospective randomised trial evaluating a paclitaxel-coated balloon in patients treated with endothelial progenitor cell capturing stents for de novo coronary artery disease. Heart 2011, 97, 1338–1342. [Google Scholar] [CrossRef]
- Seeger, J.; Markovic, S.; Birkemeyer, R.; Rittger, H.; Jung, W.; Brachmann, J.; Rottbauer, W.; Wöhrle, J. Paclitaxel-coated balloon plus bare-metal stent for de-novo coronary artery disease. Coron. Artery Dis. 2016, 27, 84–88. [Google Scholar] [CrossRef]
- Woudstra, P.; Kalkman, D.N.; Beijk, M.A.; Klomp, M.; Damman, P.; Koch, K.T.; Henriques, J.P.; Baan, J.; Vis, M.M.; Piek, J.J.; et al. Five-year follow-up of the endothelial progenitor cell capturing stent versus the paxlitaxel-eluting stent in de novo coronary lesions with a high risk of coronary restenosis. Catheter. Cardiovasc. Interv. 2017, 91, 1212–1218. [Google Scholar] [CrossRef]
- Saito, S.; Krucoff, M.; Nakamura, S.; Mehran, R.; Maehara, A.; Al-Khalidi, H.R.; Rowland, S.M.; Tasissa, G.; Morrell, D.; Joseph, D.; et al. Japan-United States of America Harmonized Assessment by Randomized Multicentre Study of OrbusNEich’s Combo StEnt (Japan-USA HARMONEE) study: Primary results of the pivotal registration study of combined endothelial progenitor cell capture and drug-eluting stent in patients with ischaemic coronary disease and non-ST-elevation acute coronary syndrome. Eur. Hear. J. 2018, 39, 2460–2468. [Google Scholar] [CrossRef]
- Noiseux, N.; Mansour, S.; Weisel, R.; Stevens, L.-M.; Der Sarkissian, S.; Tsang, K.; Crean, A.; LaRose, E.; Li, S.-H.; Wintersperger, B.; et al. The IMPACT-CABG trial: A multicenter, randomized clinical trial of CD133+ stem cell therapy during coronary artery bypass grafting for ischemic cardiomyopathy. J. Thorac. Cardiovasc. Surg. 2016, 152, 1582–1588.e2. [Google Scholar] [CrossRef]
- Katarzyna, R. Adult Stem Cell Therapy for Cardiac Repair in Patients After Acute Myocardial Infarction Leading to Ischemic Heart Failure: An Overview of Evidence from the Recent Clinical Trials. Curr. Cardiol. Rev. 2017, 13, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Keighron, C.; Lyons, C.; Creane, M.; O’Brien, T.; Liew, A. Recent Advances in Endothelial Progenitor Cells Toward Their Use in Clinical Translation. Front. Med. 2018, 5, 354. [Google Scholar] [CrossRef]
- Yue, Y.; Wang, C.; Benedict, C.L.; Huang, G.; Truongcao, M.M.; Roy, R.; Cimini, M.; Garikipati, V.N.S.; Cheng, Z.; Koch, W.J.; et al. Interleukin-10 Deficiency Alters Endothelial Progenitor Cell–Derived Exosome Reparative Effect on Myocardial Repair via Integrin-Linked Kinase Enrichment. Circ. Res. 2020, 126, 315–329. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-P.; Lin, F.-Y.; Huang, P.-H.; Chen, Y.-L.; Chen, W.-C.; Chen, H.-Y.; Huang, Y.-C.; Liao, W.-L.; Huang, H.-C.; Liu, P.-L.; et al. Endothelial Progenitor Cell Dysfunction in Cardiovascular Diseases: Role of Reactive Oxygen Species and Inflammation. BioMed Res. Int. 2012, 2013, 1–10. [Google Scholar] [CrossRef] [PubMed]
- DeSouza, C.V.; Hamel, F.G.; Bidasee, K.; O’Connell, K. Role of Inflammation and Insulin Resistance in Endothelial Progenitor Cell Dysfunction. Diabetes 2011, 60, 1286–1294. [Google Scholar] [CrossRef] [PubMed]
- Krishnamurthy, P.; Thal, M.; Verma, S.; Hoxha, E.; Lambers, E.; Ramirez, V.; Qin, G.; Losordo, D.; Kishore, R. Interleukin-10 Deficiency Impairs Bone Marrow–Derived Endothelial Progenitor Cell Survival and Function in Ischemic Myocardium. Circ. Res. 2011, 109, 1280–1289. [Google Scholar] [CrossRef]
- Park, A.; Barrera-Ramirez, J.; Ranasinghe, I.; Pilon, S.; Sy, R.; Fergusson, D.A.; Allan, D.S. Use of Statins to Augment Progenitor Cell Function in Preclinical and Clinical Studies of Regenerative Therapy: A Systematic Review. Stem Cell Rev. Rep. 2016, 12, 327–339. [Google Scholar] [CrossRef]
- Pelliccia, F.; Pasceri, V.; Moretti, A.; Tanzilli, G.; Speciale, G.; Gaudio, C. Endothelial progenitor cells predict long-term outcome in patients with coronary artery disease: Ten-year follow-up of the PROCREATION extended study. Int. J. Cardiol. 2020. [Google Scholar] [CrossRef]
- Garikipati, V.N.S.; Kishore, R. Induced Pluripotent Stem Cells Derived Extracellular Vesicles: A Potential Therapy for Cardiac Repair. Circ. Res. 2018, 122, 197–198. [Google Scholar] [CrossRef]
- Li, R.; Liang, J.; He, Y.; Qin, J.; He, H.; Lee, S.; Pang, Z.; Wang, J. Sustained Release of Immunosuppressant by Nanoparticle-anchoring Hydrogel Scaffold Improved the Survival of Transplanted Stem Cells and Tissue Regeneration. Theranostics 2018, 8, 878–893. [Google Scholar] [CrossRef]
- Vranckx, J.J.; Hondt, M.D. Tissue engineering and surgery: From translational studies to human trials. Innov. Surg. Sci. 2017, 2, 189–202. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, C.L.; O’Doherty, M.T.; Wilson, S.E.; Rana, A.A.; Hirst, C.; Stitt, A.W.; Medina, R.J. Therapeutic revascularisation of ischaemic tissue: The opportunities and challenges for therapy using vascular stem/progenitor cells. Stem Cell Res. Ther. 2012, 3, 31–37. [Google Scholar] [CrossRef] [PubMed]
| Cell Type | Number of Patients Treated (Controls) | Safety Feasibility | Technique | Findings | Limitations | References |
|---|---|---|---|---|---|---|
| CD133+ BMSCs | 19 (16) | + | Intracoronary injection | ↑ ejection fraction * ↑ LVSP/LVESVI ↓ chordae shortening * ↓ MIBI perfusion defect * | ↑ incidence of coronary events at 4-month follow-up | [95] |
| CD133+ CD34+133− BMSCs | 12 (12) | + | Intracoronary injection | ↑ ejection fraction # ↑ myocardial perfusion ↓ ED and ES volume ↓ ventricular Remodelling - No apparent major adverse cardiac events |
| [96] |
| BMSCs | 42 (60) | + | Intracoronary injection + shock wave | ↑ LV ejection fraction ↓ NYHA class ↓ NT-proBNP |
| [99] |
| CD133 selected/CD34+ BMSCs | 20 (20) | + | Intramyocardial delivery | ↑ LV systolic function |
| [102] |
| CD133+, CD34+, CD45+ BMSCs | 19 (14) | + | Intramyocardial injection | ↑ LV systolic function | - Small number of patients | [110] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perrotta, F.; Perna, A.; Komici, K.; Nigro, E.; Mollica, M.; D’Agnano, V.; De Luca, A.; Guerra, G. The State of Art of Regenerative Therapy in Cardiovascular Ischemic Disease: Biology, Signaling Pathways, and Epigenetics of Endothelial Progenitor Cells. Cells 2020, 9, 1886. https://doi.org/10.3390/cells9081886
Perrotta F, Perna A, Komici K, Nigro E, Mollica M, D’Agnano V, De Luca A, Guerra G. The State of Art of Regenerative Therapy in Cardiovascular Ischemic Disease: Biology, Signaling Pathways, and Epigenetics of Endothelial Progenitor Cells. Cells. 2020; 9(8):1886. https://doi.org/10.3390/cells9081886
Chicago/Turabian StylePerrotta, Fabio, Angelica Perna, Klara Komici, Ersilia Nigro, Mariano Mollica, Vito D’Agnano, Antonio De Luca, and Germano Guerra. 2020. "The State of Art of Regenerative Therapy in Cardiovascular Ischemic Disease: Biology, Signaling Pathways, and Epigenetics of Endothelial Progenitor Cells" Cells 9, no. 8: 1886. https://doi.org/10.3390/cells9081886
APA StylePerrotta, F., Perna, A., Komici, K., Nigro, E., Mollica, M., D’Agnano, V., De Luca, A., & Guerra, G. (2020). The State of Art of Regenerative Therapy in Cardiovascular Ischemic Disease: Biology, Signaling Pathways, and Epigenetics of Endothelial Progenitor Cells. Cells, 9(8), 1886. https://doi.org/10.3390/cells9081886







