The Secret Garden of Neuronal circRNAs
Abstract
1. Introduction
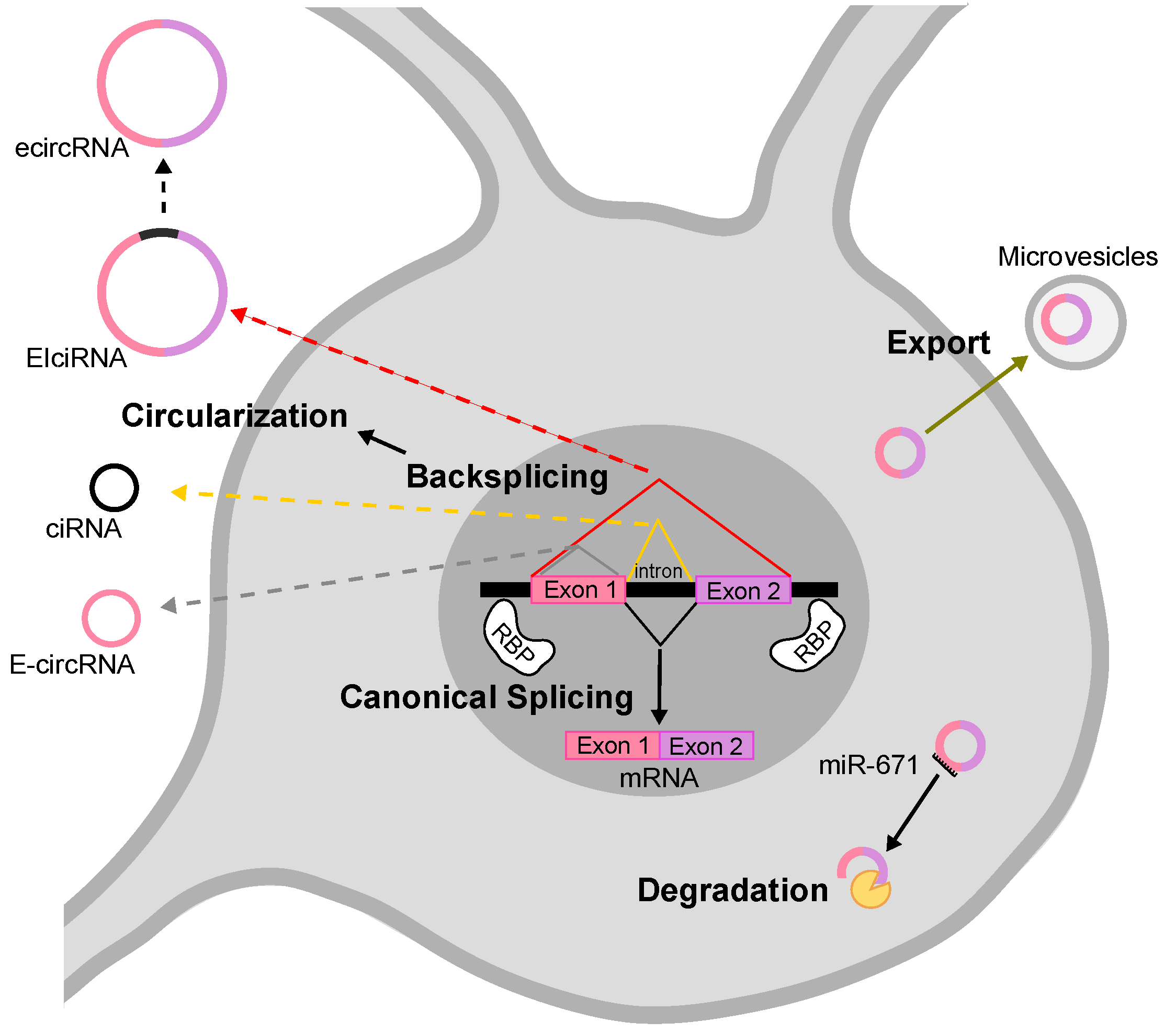
2. Biogenesis and Degradation of circRNAs
3. circRNA Known Functions
3.1. Regulation of Gene Transcription and Splicing
3.2. circRNAs as miRNA Sponges
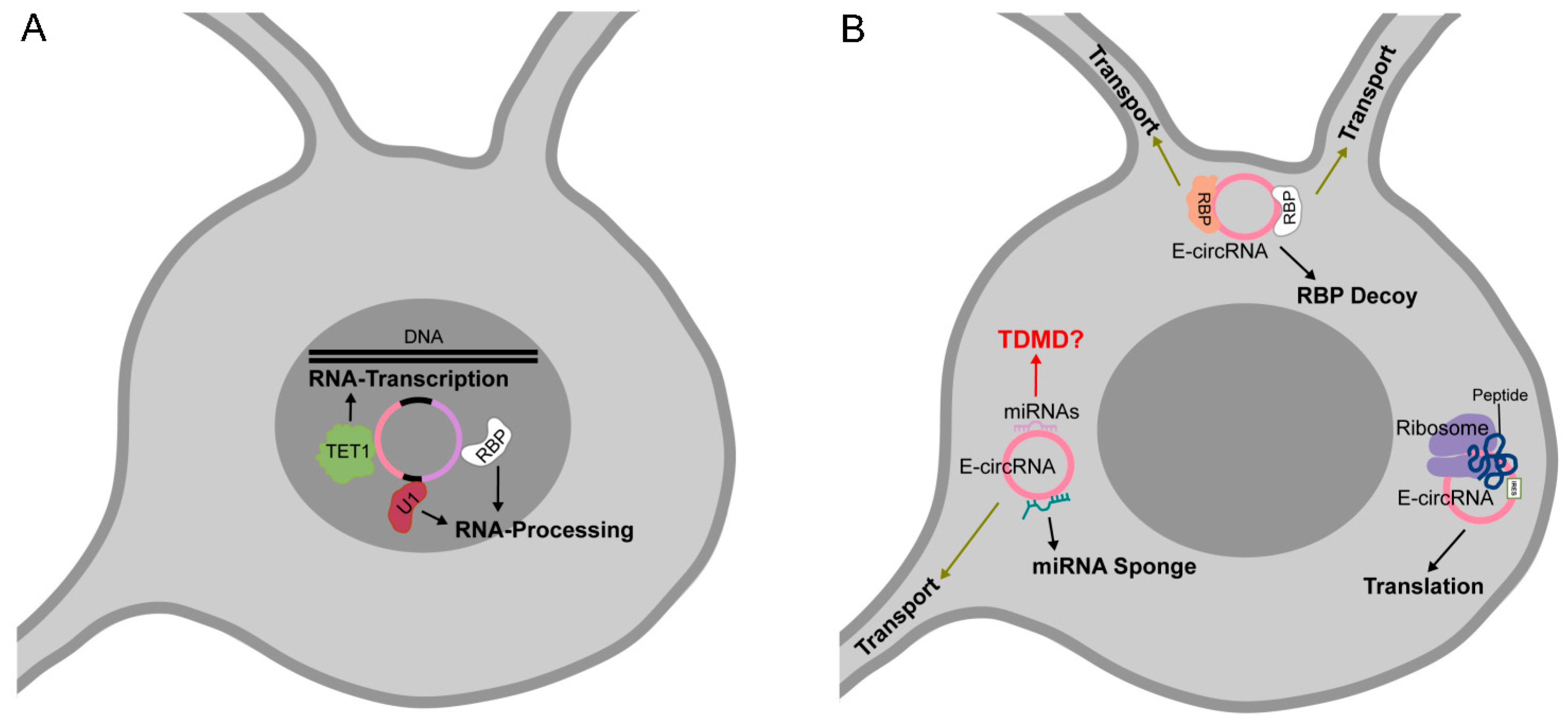
3.3. circRNAs as Protein Decoys, Scaffolds for Multiprotein Complex Assembly and Molecular Transporters
3.4. Translatable circRNAs
4. Technical Approaches of circRNA Analysis
4.1. Approaches for Functional Genetic Analysis
4.1.1. In Vitro Strategies for circRNA KD and KO
4.1.2. In Vivo Strategy for circRNA KD and KO
4.1.3. Overexpression Studies
5. Neuronal circRNAs
5.1. circRNAs in Brain Development and Homeostasis
5.2. circRNAs in Ageing
5.3. circRNAs in Neuronal Disorders
| Disease | Model System | circRNA (Validated) | circRNA Profile | Predicted Mechanism of Action | Putative Regulatory Network and Molecular Implication | Refs |
|---|---|---|---|---|---|---|
| SCZ | Human DLPFC | circGPR137B-3, circPPP2CA-3, circVCAN-2, circLONP2–6, circTOP1–10, circMYO9A-66, circHP1BP3–7 and circZNF236–2 down | Overall decrease of circRNA expression and complexity | miRNA sponge | circRNA-miRNA-mRNA network circRNA differential expression independent from host gene expression Decreased AS | [101] |
| SCZ and AFF | Human DLPFC | Identification of SNPs in circQTLs | Effects on circRNA biogenesis | [94] | ||
| SCZ and BPD | Human DLPFC, OFC and iPS-derived neurons | circHomer1 down in SCZ and AD; circCUL4A up in SCZ only; circADAM2 down in BPD only | Overall circRNA dysregulation | RBP decoy: circHomer1/HuD | Altered AS of synaptic genes and psychiatric disease associated genes | [75] |
| BPD | Human MFG | cNEBL and cEPHA3 up | No overall dysregulation of circRNA expression | Overall increased number of AS events and genes carrying AS | [109] | |
| ASD | Human FC, TC and CV | circARID1A up | 60 dysregulated circRNAs | miRNA sponge: circARID1A/miR-204-3p | circRNA-miRNA-mRNA network circRNA differential expression independent from host gene expression | [119] |
| ASD | Human FC, TC and CB | circZKSCAN1, circHIPK3 | ASD specific circRNA co-expression module in FC | miRNA sponge: CDR1as/miR-7 | CDR1as-miR-7-miR-671-Cyrano network. Percent of AS exons higher among circRNA-forming exons | [87] |
| ASD | Mouse HP from the BTBR T + tf/J ASD model | circCdh9, circCacna1c, circCacna1a, circHivep2, circCdc14b, circTrpc6, circCep112, circWdr49, circNcoa2 down; circRmst, circZcchc11, circMyrip up | 41 dysregulated circRNAs | circRNA differential expression independent from host gene expression | [92] | |
| AD | Human PC | circHOMER1 and circKCNN2 down | 33 dysregulated circRNAs | miRNA sponge | circRNA-miRNA-mRNA network circRNA differential expression independent from host gene expression | [120] |
| AD | Human HP CA1 and neo C from SAD patients | CDR1as down | miRNA sponge: CDR1as/miR-7 | CDR1as-miR-7-UBE2A mRNA network | [117,128] | |
| AD | Mouse brain from SAMP8 model | 235 dysregulated circRNAs | miRNA sponge | circRNA-miRNA-mRNA network | [115] | |
| AD | Rat HP from β1-42-induced AD model | 555 dysregulated circRNAs | miRNA sponge | circRNA-miRNA-mRNA network | [113] | |
| AD | Human HEK293 cells | 17 circRNAs derived from Aβ ORF of the APP gene | Peptide synthesis | Increases levels of Aβ and Aβ plaques | [112] | |
| PD | Mouse midbrain from MPTP-PD model and human MPP+-SH-SY-5Y cell PD model | circDLGAP4 down | miRNA sponge | circDLGAP4-miR-134-5p-CREB mRNA network | [116] | |
| PD | Human MPP+ SH-SY-5Y cellular PD model | circSNCA up | miRNA sponge | circSNCA-miR-7-SNCA mRNA network | [121] | |
| MMD | Human blood | 146 dysregulated circRNAs | miRNA sponge | circRNA-miRNA network | [117] | |
| MS | Human PBL | circ_0005402 and circ_0035560 down | 406 dysregulated circRNAs | New biomarkers | [108] | |
| ALS | Human PBMCs | circ_0023919, circ_0063411 and circ_0088036 | 425 dysregulated circRNAs | New biomarkers | [102] | |
| TL Epilepsy | Human TC | circEFCAB2, circSTK24, circVPS37C up; circDROSHA, circSTK17A, circUBQLN1, circCCT4, circUSP9 down | 442 dysregulated circRNAs | miRNA sponge | circRNA-miRNA-mRNA network prediction | [118] |
| TL Epilepsy | Human TC | circRNA-0067835 | 586 dysregulated circRNAs | miRNA sponge | circRNA-0067835-miR-155-FOXO3a mRNA network | [122] |
| Epilepsy | Mouse pilocarpine chronic epilepsy model | 43 dysregulated circRNAs | miRNA sponge | circRNA-miRNA-mRNA network | [123] |
6. circRNA New Functions
7. Perspectives and Conclusions
Can circRNAs Trigger TDMD?
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sänger, H.L.; Klotz, G.; Riesner, D.; Gross, H.J.; Kleinschmidt, A.K. Viroids are single-stranded covalently closed circular RNA molecules existing as highly base-paired rod-like structures. Proc. Natl. Acad. Sci. USA 1976, 73, 3852–3856. [Google Scholar] [CrossRef] [PubMed]
- Coca-Prados, M.; Hsu, M.T. Intracellular forms of simian virus 40 nucleoprotein complexes. II. Biochemical and electron microscopic analysis of simian virus 40 virion assembly. J. Virol. 1979, 31, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Nigro, J.M.; Cho, K.R.; Fearon, E.R.; Kern, S.E.; Ruppert, J.; Oliner, J.D.; Kinzler, K.W.; Vogelstein, B. Scrambled exons. Cell 1991, 64, 607–613. [Google Scholar] [CrossRef]
- Cocquerelle, C.; Daubersies, P.; Majérus, M.; Kerckaert, J.; Bailleul, B. Splicing with inverted order of exons occurs proximal to large introns. EMBO J. 1992, 11, 1095–1098. [Google Scholar] [CrossRef] [PubMed]
- Mironov, A.A.; Fickett, J.W.; Gelfand, M.S. Frequent Alternative Splicing of Human Genes. Genome Res. 1999, 9, 1288–1293. [Google Scholar] [CrossRef]
- Salzman, J.; Gawad, C.; Wang, P.L.; Lacayo, N.; Brown, P.O. Circular RNAs Are the Predominant Transcript Isoform from Hundreds of Human Genes in Diverse Cell Types. PLoS ONE 2012, 7, e30733. [Google Scholar] [CrossRef]
- Jeck, W.R.; Sorrentino, J.A.; Wang, K.; Slevin, M.K.; Burd, C.E.; Liu, J.; Marzluff, W.F.; Sharpless, N.E. Circular RNAs are abundant, conserved, and associated with ALU repeats. RNA 2012, 19, 141–157. [Google Scholar] [CrossRef]
- Memczak, S.; Jens, M.; Elefsinioti, A.; Torti, F.; Krueger, J.; Rybak, A.; Maier, L.; Mackowiak, S.D.; Gregersen, L.H.; Munschauer, M.; et al. Circular RNAs are a large class of animal RNAs with regulatory potency. Nature 2013, 495, 333–338. [Google Scholar] [CrossRef]
- Werfel, S.; Nothjunge, S.; Schwarzmayr, T.; Strom, T.-M.; Meitinger, T.; Engelhardt, S. Characterization of circular RNAs in human, mouse and rat hearts. J. Mol. Cell. Cardiol. 2016, 98, 103–107. [Google Scholar] [CrossRef]
- Wang, P.L.; Bao, Y.; Yee, M.-C.; Barrett, S.P.; Hogan, G.J.; Olsen, M.N.; Dinneny, J.R.; Brown, P.O.; Salzman, J. Circular RNA Is Expressed across the Eukaryotic Tree of Life. PLoS ONE 2014, 9, e90859. [Google Scholar] [CrossRef]
- Westholm, J.; Miura, P.; Olson, S.; Shenker, S.; Joseph, B.; Sanfilippo, P.; Celniker, S.E.; Graveley, B.R.; Lai, E.C. Genome-wide analysis of drosophila circular RNAs reveals their structural and sequence properties and age-dependent neural accumulation. Cell Rep. 2014, 9, 1966–1980. [Google Scholar] [CrossRef] [PubMed]
- Rybak-Wolf, A.; Stottmeister, C.; Glažar, P.; Jens, M.; Pino, N.; Giusti, S.; Hanan, M.; Behm, M.; Bartok, O.; Ashwal-Fluss, R.; et al. Circular RNAs in the Mammalian Brain Are Highly Abundant, Conserved, and Dynamically Expressed. Mol. Cell 2015, 58, 870–885. [Google Scholar] [CrossRef] [PubMed]
- You, X.; Vlatkovic, I.; Babic, A.; Will, T.; Epstein, I.; Tushev, G.; Akbalik, G.; Wang, M.; Glock, C.; Quedenau, C.; et al. Neural circular RNAs are derived from synaptic genes and regulated by development and plasticity. Nat. Neurosci. 2015, 18, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.J.; Huang, S.; Janitz, M. Changes in circular RNA expression patterns during human foetal brain development. Genomics 2019, 111, 753–758. [Google Scholar] [CrossRef]
- Gruner, H.; Cortés-López, M.; Cooper, D.; Bauer, M.; Miura, P. CircRNA accumulation in the aging mouse brain. Sci. Rep. 2016, 6, 38907. [Google Scholar] [CrossRef]
- Venø, M.T.; Hansen, T.B.; Venø, S.T.; Clausen, B.H.; Grebing, M.; Finsen, B.R.; Holm, I.E.; Kjems, J. Spatio-temporal regulation of circular RNA expression during porcine embryonic brain development. Genome Boil. 2015, 16, 245. [Google Scholar] [CrossRef]
- Salzman, J.; Chen, R.E.; Olsen, M.N.; Wang, P.L.; Brown, P.O. Cell-Type Specific Features of Circular RNA Expression. PLoS Genet. 2013, 9, e1003777. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X.-O.; Chen, T.; Xiang, J.-F.; Yin, Q.-F.; Xing, Y.-H.; Zhu, S.; Yang, L.; Chen, L.-L. Circular Intronic Long Noncoding RNAs. Mol. Cell 2013, 51, 792–806. [Google Scholar] [CrossRef]
- Chen, I.; Chen, C.-Y.; Chuang, T.-J. Biogenesis, identification, and function of exonic circular RNAs. Wiley Interdiscip. Rev. RNA 2015, 6, 563–579. [Google Scholar] [CrossRef]
- Rodríguez-Trelles, F.; Tarrio, R.; Ayala, F.J. Origins and Evolution of Spliceosomal Introns. Annu. Rev. Genet. 2006, 40, 47–76. [Google Scholar] [CrossRef]
- Li, Z.; Huang, C.; Bao, C.; Chen, L.; Lin, M.; Wang, X.; Zhong, G.; Yu, B.; Hu, W.; Dai, L.; et al. Exon-intron circular RNAs regulate transcription in the nucleus. Nat. Struct. Mol. Boil. 2015, 22, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Shen, T.; Han, M.; Wei, G.; Ni, T. An intriguing RNA species--perspectives of circularized RNA. Protein Cell 2015, 6, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.U.; Agarwal, V.; Guo, H.; Bartel, B. Expanded identification and characterization of mammalian circular RNAs. Genome Boil. 2014, 15, 1–14. [Google Scholar] [CrossRef]
- Liang, N.; Wilusz, J.E. Short intronic repeat sequences facilitate circular RNA production. Genes Dev. 2014, 28, 2233–2247. [Google Scholar] [CrossRef] [PubMed]
- Ashwal-Fluss, R.; Meyer, M.; Pamudurti, N.R.; Ivanov, A.; Bartok, O.; Hanan, M.; Evantal, N.; Memczak, S.; Rajewsky, N.; Kadener, S. circRNA Biogenesis Competes with Pre-mRNA Splicing. Mol. Cell 2014, 56, 55–66. [Google Scholar] [CrossRef]
- Conn, S.J.; Pillman, K.; Toubia, J.; Conn, V.M.; Salmanidis, M.; Phillips, C.; Roslan, S.; Schreiber, A.W.; Gregory, P.; Goodall, G.J. The RNA Binding Protein Quaking Regulates Formation of circRNAs. Cell 2015, 160, 1125–1134. [Google Scholar] [CrossRef]
- Ivanov, A.; Memczak, S.; Wyler, E.; Torti, F.; Porath, H.; Orejuela, M.R.; Piechotta, M.; Levanon, E.Y.; Landthaler, M.; Dieterich, C.; et al. Analysis of Intron Sequences Reveals Hallmarks of Circular RNA Biogenesis in Animals. Cell Rep. 2015, 10, 170–177. [Google Scholar] [CrossRef]
- Hansen, T.B.; Wiklund, E.D.; Bramsen, J.B.; Villadsen, S.B.; Statham, A.L.; Clark, S.J.; Kjems, J. miRNA-dependent gene silencing involving Ago2-mediated cleavage of a circular antisense RNA. EMBO J. 2011, 30, 4414–4422. [Google Scholar] [CrossRef]
- Piwecka, M.; Glažar, P.; Hernández-Miranda, L.R.; Memczak, S.; Wolf, S.A.; Rybak-Wolf, A.; Filipchyk, A.; Klironomos, F.; Jara, C.A.C.; Fenske, P.; et al. Loss of a mammalian circular RNA locus causes miRNA deregulation and affects brain function. Science 2017, 357, eaam8526. [Google Scholar] [CrossRef]
- Kleaveland, B.; Shi, C.Y.; Stefano, J.; Bartel, B. A Network of Noncoding Regulatory RNAs Acts in the Mammalian Brain. Cell 2018, 174, 350–362.e17. [Google Scholar] [CrossRef]
- Li, X.; Liu, C.-X.; Xue, W.; Zhang, Y.; Jiang, S.; Yin, Q.-F.; Wei, J.; Yao, R.; Yang, L.; Chen, L.-L. Coordinated circRNA Biogenesis and Function with NF90/NF110 in Viral Infection. Mol. Cell 2017, 67, 214–227.e7. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.-X.; Li, X.; Nan, F.; Jiang, S.; Gao, X.; Guo, S.-K.; Xue, W.; Cui, Y.; Dong, K.; Ding, H.; et al. Structure and Degradation of Circular RNAs Regulate PKR Activation in Innate Immunity. Cell 2019, 177, 865–880.e21. [Google Scholar] [CrossRef] [PubMed]
- Lasda, E.; Parker, R. Circular RNAs Co-Precipitate with Extracellular Vesicles: A Possible Mechanism for circRNA Clearance. PLoS ONE 2016, 11, e0148407. [Google Scholar] [CrossRef] [PubMed]
- Preußer, C.; Hung, L.-H.; Schneider, T.; Schreiner, S.; Hardt, M.; Moebus, A.; Santoso, S.; Bindereif, A. Selective release of circRNAs in platelet-derived extracellular vesicles. J. Extracell. Vesicles 2018, 7, 1424473. [Google Scholar] [CrossRef]
- Conn, V.M.; Hugouvieux, V.; Nayak, A.; Conos, S.A.; Capovilla, G.; Cildir, G.; Jourdain, A.; Tergaonkar, V.; Schmid, M.; Zubieta, C.; et al. A circRNA from SEPALLATA3 regulates splicing of its cognate mRNA through R-loop formation. Nat. Plants 2017, 3, 17053. [Google Scholar] [CrossRef]
- Chen, N.; Zhao, G.; Yan, X.; Lv, Z.; Yin, H.; Zhang, S.; Song, W.; Li, X.; Li, L.; Du, Z.; et al. A novel FLI1 exonic circular RNA promotes metastasis in breast cancer by coordinately regulating TET1 and DNMT1. Genome Boil. 2018, 19, 218. [Google Scholar] [CrossRef]
- Hansen, T.B.; I Jensen, T.; Clausen, B.H.; Bramsen, J.B.; Finsen, B.R.; Damgaard, C.K.; Kjems, J. Natural RNA circles function as efficient microRNA sponges. Nature 2013, 495, 384–388. [Google Scholar] [CrossRef]
- Xie, B.; Zhao, Z.; Liu, Q.; Wang, X.; Ma, Z.; Li, H. CircRNA has_circ_0078710 acts as the sponge of microRNA-31 involved in hepatocellular carcinoma progression. Gene 2018, 683, 253–261. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, X.; Hu, X.; Dai, L.; Fu, X.; Zhang, J.; Ao, Y. Circular RNA Related to the Chondrocyte ECM Regulates MMP13 Expression by Functioning as a MiR-136 ‘Sponge’ in Human Cartilage Degradation. Sci. Rep. 2016, 6, 22572. [Google Scholar] [CrossRef]
- Yu, C.-Y.; Li, T.-C.; Wu, Y.-Y.; Yeh, C.-H.; Chiang, W.; Chuang, C.-Y.; Kuo, H.-C. The circular RNA circBIRC6 participates in the molecular circuitry controlling human pluripotency. Nat. Commun. 2017, 8, 1149. [Google Scholar] [CrossRef]
- Zheng, Q.; Bao, C.; Guo, W.; Li, S.; Chen, J.; Chen, B.; Luo, Y.; Lyu, D.; Li, Y.; Shi, G.; et al. Circular RNA profiling reveals an abundant circHIPK3 that regulates cell growth by sponging multiple miRNAs. Nat. Commun. 2016, 7, 11215. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.-B.; Niu, Y.; Huang, G.-X.; Lu, J.-J.; Chen, A.; Zhu, L. Silencing of circRNA.2837 Plays a Protective Role in Sciatic Nerve Injury by Sponging the miR-34 Family via Regulating Neuronal Autophagy. Mol. Ther. Nucleic Acids 2018, 12, 718–729. [Google Scholar] [CrossRef] [PubMed]
- Van Rossum, D.; Verheijen, B.M.; Pasterkamp, R.J. Circular RNAs: Novel Regulators of Neuronal Development. Front. Mol. Neurosci. 2016, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Abdelmohsen, K.; Panda, A.C.; Munk, R.; Grammatikakis, I.; Dudekula, D.B.; De, S.; Kim, J.; Noh, J.H.; Kim, K.M.; Martindale, J.L.; et al. Identification of HuR target circular RNAs uncovers suppression of PABPN1 translation by CircPABPN1. RNA Boil. 2017, 14, 361–369. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, F.; Fang, E.; Xiao, W.; Mei, H.; Li, H.; Li, D.; Song, H.; Wang, J.; Hong, M.; et al. Circular RNA circAGO2 drives cancer progression through facilitating HuR-repressed functions of AGO2-miRNA complexes. Cell Death Differ. 2018, 26, 1346–1364. [Google Scholar] [CrossRef]
- Holdt, L.M.; Stahringer, A.; Sass, K.; Pichler, G.; Kulak, N.A.; Wilfert, W.; Kohlmaier, A.; Herbst, A.; Northoff, B.H.; Nicolaou, A.; et al. Circular non-coding RNA ANRIL modulates ribosomal RNA maturation and atherosclerosis in humans. Nat. Commun. 2016, 7, 12429. [Google Scholar] [CrossRef]
- Xia, P.; Wang, S.; Ye, B.; Du, Y.; Li, C.; Xiong, Z.; Qu, Y.; Fan, Z. A Circular RNA Protects Dormant Hematopoietic Stem Cells from DNA Sensor cGAS-Mediated Exhaustion. Immunity 2018, 48, 688–701.e7. [Google Scholar] [CrossRef]
- Du, W.W.; Fang, L.; Yang, W.; Wu, N.; Awan, F.M.; Yang, Z.; Yang, B.B. Induction of tumor apoptosis through a circular RNA enhancing Foxo3 activity. Cell Death Differ. 2016, 24, 357–370. [Google Scholar] [CrossRef]
- Du, W.W.; Yang, W.; Liu, E.; Yang, Z.; Dhaliwal, P.; Yang, B.B. Foxo3 circular RNA retards cell cycle progression via forming ternary complexes with p21 and CDK2. Nucleic Acids Res. 2016, 44, 2846–2858. [Google Scholar] [CrossRef]
- Du, W.W.; Yang, W.; Li, X.; Awan, F.M.; Yang, Z.; Fang, L.; Lyu, J.; Li, F.; Peng, C.; Krylov, S.N.; et al. A circular RNA circ-DNMT1 enhances breast cancer progression by activating autophagy. Oncogene 2018, 37, 5829–5842. [Google Scholar] [CrossRef]
- Zeng, Y.; Du, W.W.; Wu, Y.; Yang, Z.; Awan, F.M.; Li, X.; Yang, W.; Zhang, C.; Yang, Q.; Chen, Y.; et al. A Circular RNA Binds To and Activates AKT Phosphorylation and Nuclear Localization Reducing Apoptosis and Enhancing Cardiac Repair. Theranostics 2017, 7, 3842–3855. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Yuan, Z.; Du, K.Y.; Fang, L.; Lyu, J.; Zhang, C.; He, A.; Eshaghi, E.; Zeng, K.; Ma, J.; et al. Translation of yes-associated protein (YAP) was antagonized by its circular RNA via suppressing the assembly of the translation initiation machinery. Cell Death Differ. 2019, 26, 2758–2773. [Google Scholar] [CrossRef] [PubMed]
- Nan, A.; Chen, L.; Zhang, N.; Jia, Y.; Li, X.; Zhou, H.; Ling, Y.; Wang, Z.; Yang, C.; Liu, S.; et al. Circular RNA circNOL10 Inhibits Lung Cancer Development by Promoting SCLM1-Mediated Transcriptional Regulation of the Humanin Polypeptide Family. Adv. Sci. 2018, 6, 1800654. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zhao, F. Computational Strategies for Exploring Circular RNAs. Trends Genet. 2018, 34, 389–400. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Sarnow, P. Initiation of protein synthesis by the eukaryotic translational apparatus on circular RNAs. Science 1995, 268, 415–417. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Z. Efficient backsplicing produces translatable circular mRNAs. RNA 2014, 21, 172–179. [Google Scholar] [CrossRef]
- Wesselhoeft, R.A.; Kowalski, P.; Anderson, D.G. Engineering circular RNA for potent and stable translation in eukaryotic cells. Nat. Commun. 2018, 9, 2629. [Google Scholar] [CrossRef]
- Xia, X.; Li, X.; Li, F.; Wu, X.; Zhang, M.; Zhou, H.; Huang, N.; Yang, X.; Xiao, F.; Liu, D.; et al. A novel tumor suppressor protein encoded by circular AKT3 RNA inhibits glioblastoma tumorigenicity by competing with active phosphoinositide-dependent Kinase-1. Mol. Cancer 2019, 18, 131. [Google Scholar] [CrossRef]
- Chen, X.; Han, P.; Zhou, T.; Guo, X.; Song, X.; Li, Y. circRNADb: A comprehensive database for human circular RNAs with protein-coding annotations. Sci. Rep. 2016, 6, 34985. [Google Scholar] [CrossRef]
- Yang, Y.; Gao, X.; Zhang, M.; Yan, S.; Sun, C.; Xiao, F.; Huang, N.; Yang, X.; Zhao, K.; Zhou, H.; et al. Novel Role of FBXW7 Circular RNA in Repressing Glioma Tumorigenesis. J. Natl. Cancer Inst. 2017, 110, 304–315. [Google Scholar] [CrossRef]
- Pamudurti, N.R.; Bartok, O.; Jens, M.; Ashwal-Fluss, R.; Stottmeister, C.; Ruhe, L.; Hanan, M.; Wyler, E.; Perez-Hernandez, D.; Ramberger, E.; et al. Translation of CircRNAs. Mol. Cell 2017, 66, 9–21.e7. [Google Scholar] [CrossRef] [PubMed]
- Legnini, I.; Di Timoteo, G.; Rossi, F.; Morlando, M.; Briganti, F.; Sthandier, O.; Fatica, A.; Santini, T.; Andronache, A.; Wade, M.; et al. Circ-ZNF609 Is a Circular RNA that Can Be Translated and Functions in Myogenesis. Mol. Cell 2017, 66, 22–37.e9. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Fan, X.; Mao, M.; Song, X.; Wu, P.; Zhang, Y.; Jin, Y.; Yang, Y.; Chen, L.-L.; Wang, Y.; et al. Extensive translation of circular RNAs driven by N6-methyladenosine. Cell Res. 2017, 27, 626–641. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhao, K.; Xu, X.; Yang, Y.; Yan, S.; Wei, P.; Liu, H.; Xu, J.; Xiao, F.; Zhou, H.; et al. A peptide encoded by circular form of LINC-PINT suppresses oncogenic transcriptional elongation in glioblastoma. Nat. Commun. 2018, 9, 4475. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Huang, N.; Yang, X.; Luo, J.; Yan, S.; Xiao, F.; Chen, W.; Gao, X.; Zhao, K.; Zhou, H.; et al. A novel protein encoded by the circular form of the SHPRH gene suppresses glioma tumorigenesis. Oncogene 2018, 37, 1805–1814. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Molinie, B.; Daneshvar, K.; Pondick, J.V.; Wang, J.; Van Wittenberghe, N.O.; Xing, Y.; Giallourakis, C.C.; Mullen, A.C. Genome-Wide Maps of m6A circRNAs Identify Widespread and Cell-Type-Specific Methylation Patterns that Are Distinct from mRNAs. Cell Rep. 2017, 20, 2262–2276. [Google Scholar] [CrossRef]
- Bramsen, J.B.; Laursen, M.B.; Nielsen, A.F.; Hansen, T.B.; Bus, C.; Langkjær, N.; Babu, B.R.; Højland, T.; Abramov, M.; Van Aerschot, A.; et al. A large-scale chemical modification screen identifies design rules to generate siRNAs with high activity, high stability and low toxicity. Nucleic Acids Res. 2009, 37, 2867–2881. [Google Scholar] [CrossRef]
- Herrera-Carrillo, E.; Harwig, A.; Berkhout, B. Influence of the loop size and nucleotide composition on AgoshRNA biogenesis and activity. RNA Boil. 2017, 14, 1559–1569. [Google Scholar] [CrossRef]
- Xiao, M.-S.; Ai, Y.; Wilusz, J.E. Biogenesis and Functions of Circular RNAs Come into Focus. Trends Cell Boil. 2020, 30, 226–240. [Google Scholar] [CrossRef]
- Kristensen, L.S.; Andersen, M.S.; Stagsted, L.V.W.; Ebbesen, K.K.; Hansen, T.B.; Kjems, J. The biogenesis, biology and characterization of circular RNAs. Nat. Rev. Genet. 2019, 20, 675–691. [Google Scholar] [CrossRef]
- Patop, I.L.; Wüst, S.; Kadener, S. Past, present, and future of circ RNA s. EMBO J. 2019, 38, e100836. [Google Scholar] [CrossRef]
- Abudayyeh, O.O.; Gootenberg, J.S.; Essletzbichler, P.; Han, S.; Joung, J.; Belanto, J.J.; Verdine, V.; Cox, D.B.T.; Kellner, M.J.; Regev, A.; et al. RNA targeting with CRISPR–Cas13. Nature 2017, 550, 280–284. [Google Scholar] [CrossRef]
- Li, S.; Li, X.; Xue, W.; Zhang, L.; Cao, S.-M.; Lei, Y.-N.; Yang, L.-Z.; Guo, S.-K.; Zhang, J.-L.; Gao, X.; et al. Screening for functional circular RNAs using the CRISPR-Cas13 system. bioRxiv 2020, 1–48. [Google Scholar] [CrossRef]
- Pamudurti, N.R.; Patop, I.L.; Krishnamoorthy, A.; Ashwal-Fluss, R.; Bartok, O.; Kadener, S.; Reddy, N. An in vivo strategy for knockdown of circular RNAs. bioRxiv 2020. [Google Scholar] [CrossRef]
- Zimmerman, A.J.; Hafez, A.K.; Amoah, S.K.; Rodriguez, B.A.; Dell’Orco, M.; Lozano, E.; Hartley, B.J.; Alural, B.; LaLonde, J.; Chander, P.; et al. A psychiatric disease-related circular RNA controls synaptic gene expression and cognition. Mol. Psychiatry 2020, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Barrett, S.P.; Parker, K.R.; Horn, C.; Mata, M.; Salzman, J. ciRS-7 exonic sequence is embedded in a long non-coding RNA locus. PLoS Genet. 2017, 13, e1007114. [Google Scholar] [CrossRef] [PubMed]
- Barrett, S.; Salzman, J. Circular RNAs: Analysis, expression and potential functions. Development 2016, 143, 1838–1847. [Google Scholar] [CrossRef]
- Starke, S.; Jost, I.; Rossbach, O.; Schneider, T.; Schreiner, S.; Hung, L.-H.; Bindereif, A. Exon Circularization Requires Canonical Splice Signals. Cell Rep. 2015, 10, 103–111. [Google Scholar] [CrossRef]
- Kramer, M.C.; Liang, N.; Tatomer, D.C.; Gold, B.; March, Z.; Cherry, S.; Wilusz, J.E. Combinatorial control of Drosophila circular RNA expression by intronic repeats, hnRNPs, and SR proteins. Genes Dev. 2015, 29, 2168–2182. [Google Scholar] [CrossRef]
- Kelly, S.; Greenman, C.; Cook, P.R.; Papantonis, A. Exon Skipping Is Correlated with Exon Circularization. J. Mol. Boil. 2015, 427, 2414–2417. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, Z. Constructing GFP-Based Reporter to Study Back Splicing and Translation of Circular RNA. Methods Mol. Biol. 2018, 1724, 107–118. [Google Scholar] [PubMed]
- Raj, B.; Blencowe, B.J. Alternative Splicing in the Mammalian Nervous System: Recent Insights into Mechanisms and Functional Roles. Neuron 2015, 87, 14–27. [Google Scholar] [CrossRef] [PubMed]
- Deveson, I.W.; Brunck, M.E.; Blackburn, J.; Tseng, E.; Hon, T.; Clark, T.A.; Clark, M.B.; Crawford, J.; Dinger, M.; Nielsen, L.K.; et al. Universal Alternative Splicing of Noncoding Exons. Cell Syst. 2018, 6, 245–255.e5. [Google Scholar] [CrossRef]
- Pan, Q.; Shai, O.; Lee, L.J.; Frey, B.J.; Blencowe, B.J. Deep surveying of alternative splicing complexity in the human transcriptome by high-throughput sequencing. Nat. Genet. 2008, 40, 1413–1415. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.; Damoiseaux, R.; Chen, L.; Black, D.L. A broadly applicable high-throughput screening strategy identifies new regulators of Dlg4 (Psd-95) alternative splicing. Genome Res. 2013, 23, 998–1007. [Google Scholar] [CrossRef]
- Scotti, M.M.; Swanson, M. RNA mis-splicing in disease. Nat. Rev. Genet. 2015, 17, 19–32. [Google Scholar] [CrossRef]
- Gokool, A.; Anwar, F.; Voineagu, I. The Landscape of Circular RNA Expression in the Human Brain. Boil. Psychiatry 2019, 87, 294–304. [Google Scholar] [CrossRef]
- Szabó, L.; Morey, R.; Palpant, N.J.; Wang, P.L.; Afari, N.; Jiang, C.; Parast, M.M.; Murry, C.E.; Laurent, L.C.; Salzman, J. Statistically based splicing detection reveals neural enrichment and tissue-specific induction of circular RNA during human fetal development. Genome Boil. 2015, 16, 126. [Google Scholar] [CrossRef]
- Mahmoudi, E.; Cairns, M.J. Circular RNAs are temporospatially regulated throughout development and ageing in the rat. Sci. Rep. 2019, 9, 2564. [Google Scholar] [CrossRef]
- Mahmoudi, E.; Kiltschewskij, D.J.; Fitzsimmons, C.; Cairns, M.J. Depolarization-Associated CircRNA Regulate Neural Gene Expression and in Some Cases May Function as Templates for Translation. Cells 2019, 9, 25. [Google Scholar] [CrossRef]
- Cortés-López, M.; Gruner, M.; Cooper, D.A.; Gruner, H.N.; Voda, A.; Van Der Linden, A.M.; Miura, P. Global accumulation of circRNAs during aging in Caenorhabditis elegans. BMC Genom. 2018, 19, 1–12. [Google Scholar] [CrossRef]
- Gasparini, S.; Del Vecchio, G.; Gioiosa, S.; Flati, T.; Castrignano, T.; Legnini, I.; Licursi, V.; Ricceri, L.; Scattoni, M.L.; Rinaldi, A.; et al. Differential Expression of Hippocampal Circular RNAs in the BTBR Mouse Model for Autism Spectrum Disorder. Mol. Neurobiol. 2020, 57, 1–13. [Google Scholar] [CrossRef]
- Lee, E.C.S.; Elhassan, S.A.M.; Lim, G.P.L.; Kok, W.H.; Tan, S.W.; Leong, E.N.; Tan, S.H.; Chan, E.W.L.; Bhattamisra, S.K.; Rajendran, R.; et al. The roles of circular RNAs in human development and diseases. Biomed. Pharmacother. 2019, 111, 198–208. [Google Scholar] [CrossRef]
- Liu, Z.; Ran, Y.; Tao, C.; Li, S.; Chen, J.; Yang, E. Detection of circular RNA expression and related quantitative trait loci in the human dorsolateral prefrontal cortex. Genome Boil. 2019, 20, 99. [Google Scholar] [CrossRef] [PubMed]
- Charizanis, K.; Lee, K.-Y.; Batra, R.; Goodwin, M.; Zhang, C.; Yuan, Y.; Shiue, L.; Cline, M.; Scotti, M.M.; Xia, G.; et al. Muscleblind-like 2-mediated alternative splicing in the developing brain and dysregulation in myotonic dystrophy. Neuron 2012, 75, 437–450. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Gao, K.; Jankovic, J. The role of FUS gene variants in neurodegenerative diseases. Nat. Rev. Neurol. 2014, 10, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Gao, K.; Zheng, W.; Deng, X.; Xiong, W.; Song, Z.; Yang, Y.; Deng, H. Genetic analysis of the fused in sarcoma gene in Chinese Han patients with Parkinson’s disease. Park. Relat. Disord. 2014, 20, 119–121. [Google Scholar] [CrossRef] [PubMed]
- Lauriat, T.L.; Shiue, L.; Haroutunian, V.; Verbitsky, M.; Ares, M.; Ospina, L.; McInnes, L.A.; Ares, J.M. Developmental expression profile ofquaking, a candidate gene for schizophrenia, and its target genes in human prefrontal cortex and hippocampus shows regional specificity. J. Neurosci. Res. 2008, 86, 785–796. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Mangelsdorf, M.; Liu, J.; Zhu, L.; Wu, J.Y. RNA-binding proteins in neurological diseases. Sci. China Life Sci. 2014, 57, 432–444. [Google Scholar] [CrossRef]
- Errichelli, L.; Modigliani, S.D.; Laneve, P.; Colantoni, A.; Legnini, I.; Capauto, D.; Rosa, A.; De Santis, R.; Scarfò, R.; Peruzzi, G.; et al. FUS affects circular RNA expression in murine embryonic stem cell-derived motor neurons. Nat. Commun. 2017, 8, 14741. [Google Scholar] [CrossRef]
- Mahmoudi, E.; Fitzsimmons, C.; Geaghan, M.P.; Weickert, C.S.; Atkins, J.R.; Wang, X.; Cairns, M.J. Circular RNA biogenesis is decreased in postmortem cortical gray matter in schizophrenia and may alter the bioavailability of associated miRNA. Neuropsychopharmacology 2019, 44, 1043–1054. [Google Scholar] [CrossRef]
- Dolinar, A.; Koritnik, B.; Glavač, D.; Ravnik-Glavač, M. Circular RNAs as Potential Blood Biomarkers in Amyotrophic Lateral Sclerosis. Mol. Neurobiol. 2019, 56, 8052–8062. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.-T.; Zhou, J.; Dong, X.-L.; Bi, C.-W.; Jiang, R.; Dong, J.-F.; Tian, Y.; Yuan, H.-J.; Zhang, J. Circular Ribonucleic Acid Expression Alteration in Exosomes from the Brain Extracellular Space after Traumatic Brain Injury in Mice. J. Neurotrauma 2018, 35, 2056–2066. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zheng, Q.; Bao, C.; Li, S.; Guo, W.; Zhao, J.; Chen, D.; Gu, J.; He, X.; Huang, S. Circular RNA is enriched and stable in exosomes: A promising biomarker for cancer diagnosis. Cell Res. 2015, 25, 981–984. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, J.; Ma, J.; Sun, T.; Zhou, Q.; Wang, W.; Wang, G.; Wu, P.; Wang, H.; Jiang, L.; et al. Exosomal circRNAs: Biogenesis, effect and application in human diseases. Mol. Cancer 2019, 18, 116. [Google Scholar] [CrossRef] [PubMed]
- Saeedi, S.; Israel, S.; Nagy, C.; Turecki, G. The emerging role of exosomes in mental disorders. Transl. Psychiatry 2019, 9, 122. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Ren, M.; Li, H.; Yang, L.; Wang, X.; Yang, Q. Exosomal Circular RNA as a Biomarker Platform for the Early Diagnosis of Immune-Mediated Demyelinating Disease. Front. Genet. 2019, 10, 1–15. [Google Scholar] [CrossRef]
- Iparraguirre, L.; Muñoz-Culla, M.; Prada-Luengo, I.; Castillo-Triviño, T.; Olascoaga, J.; Otaegui, D. Circular RNA profiling reveals that circular RNAs from ANXA2 can be used as new biomarkers for multiple sclerosis. Hum. Mol. Genet. 2017, 26, 3564–3572. [Google Scholar] [CrossRef]
- Luykx, J.J.; Giuliani; Veldink, J.H.; Giuliani, F.; Giuliani, G. Coding and Non-Coding RNA Abnormalities in Bipolar Disorder. Genes 2019, 10, 946. [Google Scholar] [CrossRef]
- Glatt, S.J.; Cohen, O.S.; Faraone, S.V.; Tsuang, M.T. Dysfunctional gene splicing as a potential contributor to neuropsychiatric disorders. Am. J. Med Genet. Part B Neuropsychiatr. Genet. 2011, 156, 382–392. [Google Scholar] [CrossRef]
- Reble, E.; Dineen, A.; Barr, C.L. The contribution of alternative splicing to genetic risk for psychiatric disorders. Genes Brain Behav. 2017, 17, e12430. [Google Scholar] [CrossRef] [PubMed]
- Mo, D.; Li, X.; Raabe, C.A.; Rozhdestvensky, T.S.; Skryabin, B.V.; Brosius, J.; Cui, D. The role of Aβ circRNA in Alzheimer′s disease: Alternative mechanism of Aβ biogenesis from Aβ circRNA translation. bioRxiv 2018. [Google Scholar] [CrossRef]
- Wang, Z.; Xu, P.; Chen, B.; Zhang, Z.; Zhang, C.; Zhan, Q.; Huang, S.; Xia, Z.-A.; Peng, W. Identifying circRNA-associated-ceRNA networks in the hippocampus of Aβ1-42-induced Alzheimer’s disease-like rats using microarray analysis. Aging 2018, 10, 775–788. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Alexandrov, P.N.; Jaber, V.; Lukiw, W.J. Deficiency in the Ubiquitin Conjugating Enzyme UBE2A in Alzheimer’s Disease (AD) is Linked to Deficits in a Natural Circular miRNA-7 Sponge (circRNA; ciRS-7). Genes 2016, 7, 116. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhu, D.; Li, H.; Li, H.; Feng, C.; Zhang, W. Characterization of circRNA-Associated-ceRNA Networks in a Senescence-Accelerated Mouse Prone 8 Brain. Mol. Ther. 2017, 25, 2053–2061. [Google Scholar] [CrossRef]
- Feng, Z.; Zhang, L.; Wang, S.; Hong, Q. Circular RNA circDLGAP4 exerts neuroprotective effects via modulating miR-134-5p/CREB pathway in Parkinson’s disease. Biochem. Biophys. Res. Commun. 2020, 522, 388–394. [Google Scholar] [CrossRef]
- Zhao, M.; Gao, F.; Zhang, N.; Wang, S.; Zhang, Y.; Wang, R.; Zhao, J.-Z. Altered expression of circular RNAs in Moyamoya disease. J. Neurol. Sci. 2017, 381, 25–31. [Google Scholar] [CrossRef]
- Li, J.; Lin, H.; Sun, Z.; Kong, G.; Yan, X.; Wang, Y.; Wang, X.; Wen, Y.; Liu, X.; Zheng, H.; et al. High-Throughput Data of Circular RNA Profiles in Human Temporal Cortex Tissue Reveals Novel Insights into Temporal Lobe Epilepsy. Cell. Physiol. Biochem. 2018, 45, 677–691. [Google Scholar] [CrossRef]
- Chen, Y.-J.; Chen, C.-Y.; Mai, T.-L.; Chuang, C.-F.; Chen, Y.-C.; Gupta, S.K.; Yen, L.; Wang, Y.-D.; Chuang, T.-J. Genome-wide, integrative analysis of circular RNA dysregulation and the corresponding circular RNA-microRNA-mRNA regulatory axes in autism. Genome Res. 2020, 30, 375–391. [Google Scholar] [CrossRef]
- Dube, U.; Del Aguila, J.L.; Li, Z.; Budde, J.P.; Jiang, S.; Hsu, S.; Ibanez, L.; Fernandez, M.V.; Farias, F.; Norton, J.; et al. An atlas of cortical circular RNA expression in Alzheimer disease brains demonstrates clinical and pathological associations. Nat. Neurosci. 2019, 22, 1903–1912. [Google Scholar] [CrossRef]
- Sang, M.; Meng, L.; Sang, Y.; Liu, S.; Ding, P.; Ju, Y.; Liu, F.; Gu, L.; Lian, Y.; Li, J.; et al. Circular RNA ciRS-7 accelerates ESCC progression through acting as a miR-876-5p sponge to enhance MAGE-A family expression. Cancer Lett. 2018, 426, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Gong, G.-H.; An, F.-M.; Wang, Y.; Bian, M.; Wang, D.; Wei, C.-X. Comprehensive Circular RNA Profiling Reveals the Regulatory Role of the CircRNA-0067835/miR-155 Pathway in Temporal Lobe Epilepsy. Cell. Physiol. Biochem. 2018, 51, 1399–1409. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.-J.; Moon, J.; Jeon, D.; Kim, T.-J.; Yoo, J.-S.; Park, D.-K.; Lee, S.-T.; Jung, K.-H.; Park, K.-I.; Jung, K.-Y.; et al. Possible epigenetic regulatory effect of dysregulated circular RNAs in epilepsy. PLoS ONE 2018, 13, e0209829. [Google Scholar] [CrossRef] [PubMed]
- Xia, T.; Liao, Q.; Jiang, X.; Shao, Y.; Xiao, B.; Xi, Y.; Guo, J.-M. Long noncoding RNA associated-competing endogenous RNAs in gastric cancer. Sci. Rep. 2014, 4, 6088. [Google Scholar] [CrossRef] [PubMed]
- Salmena, L.; Poliseno, L.; Tay, Y.; Kats, L.M.; Pandolfi, P.P. A ceRNA Hypothesis: The Rosetta Stone of a Hidden RNA Language? Cell 2011, 146, 353–358. [Google Scholar] [CrossRef]
- Ebert, M.S.; Neilson, J.R.; Sharp, P.A. MicroRNA sponges: Competitive inhibitors of small RNAs in mammalian cells. Nat. Methods 2007, 4, 721–726. [Google Scholar] [CrossRef] [PubMed]
- Kartha, R.V.; Subramanian, S. Competing endogenous RNAs (ceRNAs): New entrants to the intricacies of gene regulation. Front. Genet. 2014, 5, 8. [Google Scholar] [CrossRef]
- Lukiw, W.J. Circular RNA (circRNA) in Alzheimer’s disease (AD). Front. Genet. 2013, 4. [Google Scholar] [CrossRef]
- Mortimer, S.A.; Kidwell, M.A.; A Doudna, J. Insights into RNA structure and function from genome-wide studies. Nat. Rev. Genet. 2014, 15, 469–479. [Google Scholar] [CrossRef]
- Lasda, E.; Parker, R. Circular RNAs: Diversity of form and function. RNA 2014, 20, 1829–1842. [Google Scholar] [CrossRef]
- Fischer, J.W.; Busa, V.F.; Shao, Y.; Leung, A.K.L. Structure-Mediated RNA Decay by UPF1 and G3BP1. Mol. Cell 2020, 78, 70–84.e6. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Schuman, E.M. Circular RNAs in Brain and Other Tissues: A Functional Enigma. Trends Neurosci. 2016, 39, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Pawlica, P.; Sheu-Gruttadauria, J.; Macrae, I.J.; Steitz, J.A. How Complementary Targets Expose the microRNA 3′ End for Tailing and Trimming during Target-Directed microRNA Degradation. Cold Spring Harb. Symp. Quant. Boil. 2019, 84, 179–183. [Google Scholar] [CrossRef]
- Cazalla, D.; Yario, T.; Steitz, J.A. Down-Regulation of a Host MicroRNA by a Herpesvirus saimiri Noncoding RNA. Science 2010, 328, 1563–1566. [Google Scholar] [CrossRef]
- Marcinowski, L.; Tanguy, M.; Krmpotic, A.; Rädle, B.; Lisnić, V.J.; Tuddenham, L.; Chane-Woon-Ming, B.; Ruzsics, Z.; Erhard, F.; Benkartek, C.; et al. Degradation of Cellular miR-27 by a Novel, Highly Abundant Viral Transcript Is Important for Efficient Virus Replication In Vivo. PLOS Pathog. 2012, 8, e1002510. [Google Scholar] [CrossRef] [PubMed]
- Ameres, S.L.; Hung, J.-H.; Xu, J.; Weng, Z.; Zamore, P.D. Target RNA-directed tailing and trimming purifies the sorting of endo-siRNAs between the two Drosophila Argonaute proteins. RNA 2010, 17, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Baccarini, A.; Chauhan, H.; Gardner, T.J.; Jayaprakash, A.D.; Sachidanandam, R.; Brown, B.D. Kinetic Analysis Reveals the Fate of a MicroRNA following Target Regulation in Mammalian Cells. Curr. Boil. 2011, 21, 369–376. [Google Scholar] [CrossRef] [PubMed]
- La Mata, M.; Gaidatzis, D.; Vitanescu, M.; Stadler, M.B.; Wentzel, C.; Scheiffele, P.; Filipowicz, W.; Großhans, H.; De La Mata, M. Potent degradation of neuronal mi RNA s induced by highly complementary targets. EMBO Rep. 2015, 16, 500–511. [Google Scholar] [CrossRef] [PubMed]
- Bitetti, A.; Mallory, A.C.; Golini, E.; Carrieri, C.; Gutiérrez, H.C.; Perlas, E.; Pérez-Rico, Y.A.; Tocchini-Valentini, G.P.; Enright, A.J.; Norton, W.H.J.; et al. MicroRNA degradation by a conserved target RNA regulates animal behavior. Nat. Struct. Mol. Boil. 2018, 25, 244–251. [Google Scholar] [CrossRef]
- Miranda, K.C.; Huynh, T.; Tay, Y.; Ang, Y.-S.; Tam, W.L.; Thomson, A.M.; Lim, B.; Rigoutsos, I. A Pattern-Based Method for the Identification of MicroRNA Binding Sites and Their Corresponding Heteroduplexes. Cell 2006, 126, 1203–1217. [Google Scholar] [CrossRef]
- Zok, T.; Antczak, M.; Zurkowski, M.; Popenda, M.; Błazewicz, J.; Adamiak, R.W.; Szachniuk, M. RNApdbee 2.0: Multifunctional tool for RNA structure annotation. Nucleic Acids Res. 2018, 46, W30–W35. [Google Scholar] [CrossRef] [PubMed]
- Glažar, P.; Papavasileiou, P.; Rajewsky, N. circBase: A database for circular RNAs. RNA 2014, 20, 1666–1670. [Google Scholar] [CrossRef] [PubMed]
- Mannironi, C.; Camon, J.; De Vito, F.; Biundo, A.; De Stefano, M.E.; Persiconi, I.; Bozzoni, I.; Fragapane, P.; Mele, A.; Presutti, C. Acute Stress Alters Amygdala microRNA miR-135a and miR-124 Expression: Inferences for Corticosteroid Dependent Stress Response. PLoS ONE 2013, 8, e73385. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Li, Z. miRNAs in synapse development and synaptic plasticity. Curr. Opin. Neurobiol. 2017, 45, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Bicker, S.; Lackinger, M.; Weis, K.; Schratt, G. MicroRNA-132, -134, and -138: A microRNA troika rules in neuronal dendrites. Cell. Mol. Life Sci. 2014, 71, 3987–4005. [Google Scholar] [CrossRef] [PubMed]
- Vo, N.K.; Cambronne, X.; Goodman, R.H. MicroRNA pathways in neural development and plasticity. Curr. Opin. Neurobiol. 2010, 20, 457–465. [Google Scholar] [CrossRef]
- Siegel, G.; Saba, R.; Schratt, G. microRNAs in neurons: Manifold regulatory roles at the synapse. Curr. Opin. Genet. Dev. 2011, 21, 491–497. [Google Scholar] [CrossRef]
- Wightman, F.F.; Giono, L.E.; Fededa, J.P.; De La Mata, M. Target RNAs Strike Back on MicroRNAs. Front. Genet. 2018, 9, 1–21. [Google Scholar] [CrossRef]
- Frances, H.B. The Secret Garden; F.A. Stokes: New York, NY, USA; Libr. Congr.: Washington, DC, USA, 1911. Available online: https://www.loc.gov/item/11021580/ (accessed on 8 July 2020).
| circRNA | Molecular Mechanism | Validation Method | Putative Physiological Function | Refs |
|---|---|---|---|---|
| ci-ankrd52 | Interacts with the RNA Pol II elongation complex | KD studies RNA/DNA double FISH RIP assay RNA pull-down assay | Promotes transcription of host gene | [18] |
| circEIF3J | Interacts with U1 snRNP and RNA Pol II | Nuclear Run-On assay ChIRP assay RIP assay RNA pull-down assay | Promotes transcription of host gene | [21] |
| circSEPALLATA3 | Forms R-loops | R-loop dot-blotting | Regulates splicing of cognate mRNA | [35] |
| FECR1 | Interacts with TET1 Binds to the DNMT1 promoter | RAT assay RNA ChIP assay OE studies | Promotes transcription of host gene by inducing DNA hypomethylation | [36] |
| CDR1as/ciRS-7 | miR-7 sponging | Ago PAR-CLIP Ago HITS-CLIP | Neuronal development | [8,37] |
| circHIPK3 | miR-24 sponging | Biotin-coupled miRNA pull-down assay RIP assay Luciferase reporter assay | Regulates cell growth | [41] |
| circBIRC6 | miR-34a and miR-145 sponging | Biotin-coupled miRNA pull-down assay RIP assay Luciferase reporter assay | Promotes pluripotency of human embryonic stem cells | [40] |
| circRNA.2837 | mir-34a sponging | OE and KD studies RIP assay Luciferase reporter assay | Regulates neuronal autophagy | [42] |
| circANRIL | PES1 decoy activity | λN-Peptide-mediated pull-down assay RIP assay | Impairs pre-rRNA processing and ribosome biogenesis, conferring atheroprotection | [47] |
| circPABPN1 | HuR decoy activity | RIP assay RNA pull-down assay | Suppresses PABPN1 mRNA translation reducing cellular proliferation | [44] |
| circ-DNMT1 | AUF1 and p53 nuclear translocation | Oligo blocking assay RIP assay RNA pull-down assay | Enhances in breast cancer cell proliferation, survival, autophagy and inhibits cell senescence | [51] |
| circAGO2 | HuR decoy activity and cytoplasmic translocation | EMSA RNA pull-down assay Luciferase assay | Enhances cancer progression | [46] |
| circ-Foxo3 | Promotes MDM2 and p53 interaction | KD studies RIP assay RNA pull-down assay | Enhances cell apoptosis | [49] |
| circ-Amotl1 | Promotes AKT1 and PDK1 interaction and nuclear translocation | Oligo blocking assay KD studies RIP assay RNA pull-down assay | Reduces cell apoptosis and promotes cardiac repair | [52] |
| cia-cGAS | Inhibits cGAS interaction with genomic DNA | RIP assay RNA pull-down assay FISH & IF assay | Contributes to the maintenance of long-term hematopoietic stem cells dormancy | [48] |
| circYAP | Silences Yap mRNA translation by inhibiting IF4G and PABP interaction | Cap-binding pull down assay IP-IB assays RIP assay RNA pull-down assay | Inhibits cancer progression | [53] |
| circNOL10 | Interacts with and inhibits SCML1 ubiquitination | RNA pull-down assay EMSA Luciferase assay | Reduces cell cycle progression and cell proliferation, and increases apoptosis | [45] |
| circMbl | Encodes circMbl1-peptide | Ribosome footprinting TRAP assay WB analysis | Putative synaptic functions | [61] |
| circ-ZNF609 | Encodes circ-ZNF609-derived protein | Sucrose gradient fractionation Flagged protein IP WB analysis | Regulates proliferation of myoblasts | [62] |
| circ-FBXW7 | Encodes the FBXW7-185aa protein | Luciferase assay Flagged protein WB analysis LC-MS analysis | Reduces proliferation and cell cycle progression of glioma cell lines | [60] |
| circPINTexon2 | Encodes the PINT87aa protein | RNC-seq IRES activity test Luciferase assay WB analysis LC-MS analysis | Reduces glioblastoma cell proliferation | [64] |
| circ-AKT3 | Encodes the AKT3-174aa protein | Luciferase assay MS followed by SDS-PAGE IF staining analysis | Reduces glioblastoma tumorigenicity | [58] |
| miRNA/circRNA | ΔG | Secondary Structure |
|---|---|---|
| miR-7/Cyrano ncRNA | −28.9 | 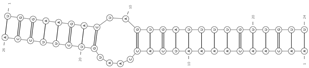 |
| mmu-miR-185-5p/mmu_circ_0001796—Zfp609 | −28.2 | 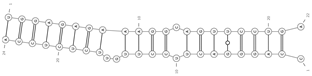 |
| mmu-miR-134-5p/mmu_circ_0005351—Pinx1 | −28.0 | 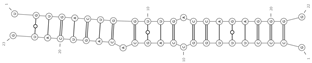 |
| mmu-miR-188-5p/mmu_circ_0000989—Spopl | −26.4 | 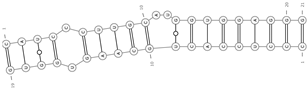 |
| mmu-miR-129-5p/mmu_circ_0005285—Myh6 | −25.5 | 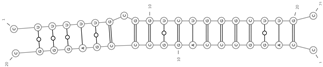 |
| mmu-miR-129-5p/mmu_circ_0004245—Sipa1l1 | −24.0 | 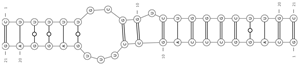 |
| mmu-miR-146a-5p/mmu_circ_0007086—Fbxo11 | −22.5 | 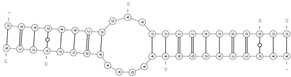 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gasparini, S.; Licursi, V.; Presutti, C.; Mannironi, C. The Secret Garden of Neuronal circRNAs. Cells 2020, 9, 1815. https://doi.org/10.3390/cells9081815
Gasparini S, Licursi V, Presutti C, Mannironi C. The Secret Garden of Neuronal circRNAs. Cells. 2020; 9(8):1815. https://doi.org/10.3390/cells9081815
Chicago/Turabian StyleGasparini, Silvia, Valerio Licursi, Carlo Presutti, and Cecilia Mannironi. 2020. "The Secret Garden of Neuronal circRNAs" Cells 9, no. 8: 1815. https://doi.org/10.3390/cells9081815
APA StyleGasparini, S., Licursi, V., Presutti, C., & Mannironi, C. (2020). The Secret Garden of Neuronal circRNAs. Cells, 9(8), 1815. https://doi.org/10.3390/cells9081815





