Neural Crest-Derived Chondrocytes Isolation for Tissue Engineering in Regenerative Medicine
Abstract
1. Introduction
2. Materials and Methods
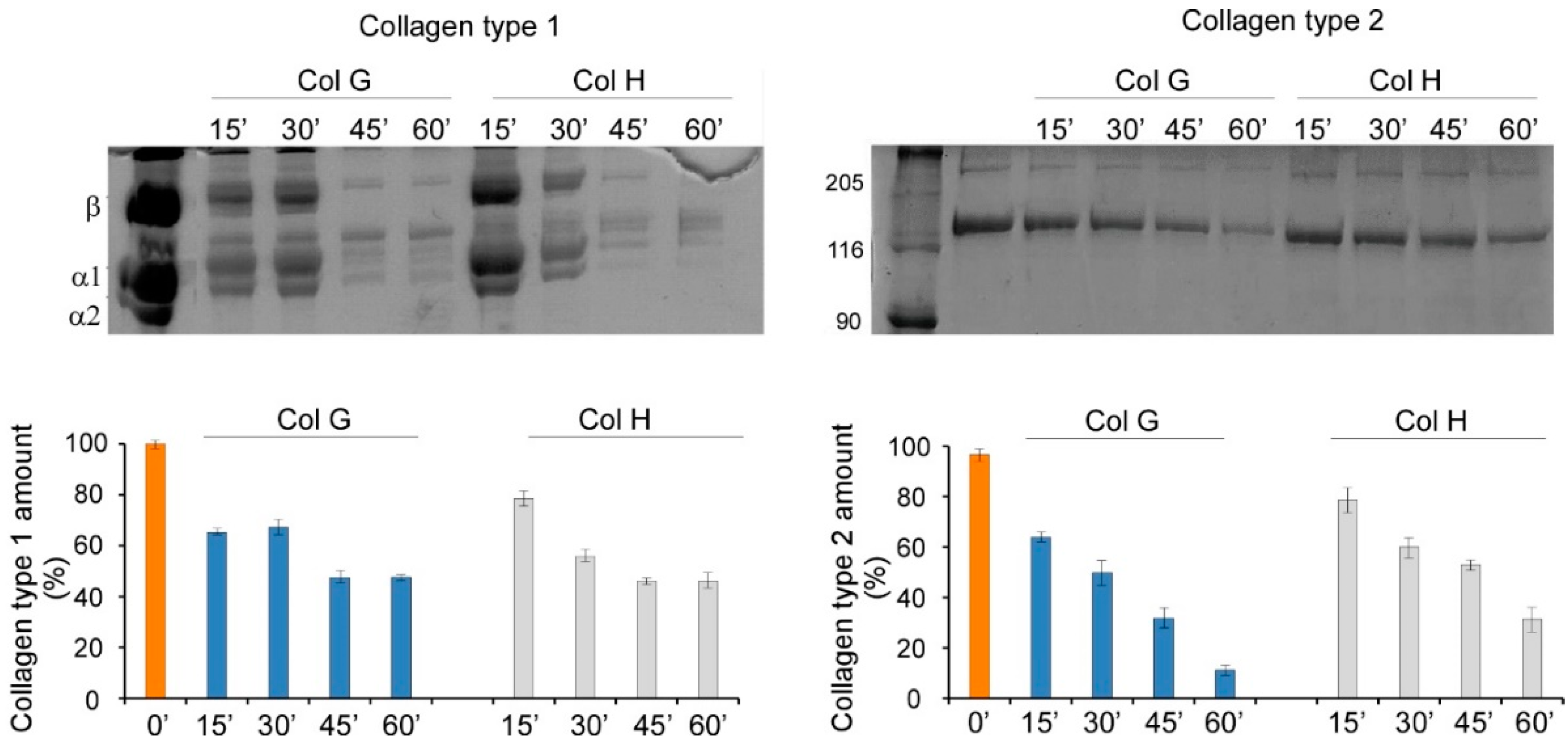
2.1. Determination of the Enzymatic Activity of Recombinant ColG and ColH
2.2. Digestion of Collagen and Cartilage Tissue
2.3. Isolation of Nasal Septal Chondrocytes from Bovine Tissue and Cell Culture Monolayer Expansion
2.4. SDS Electrophoresis
2.5. RNA Isolation and cDNA Synthesis
2.6. RT-qPCR
3. Results and Discussions
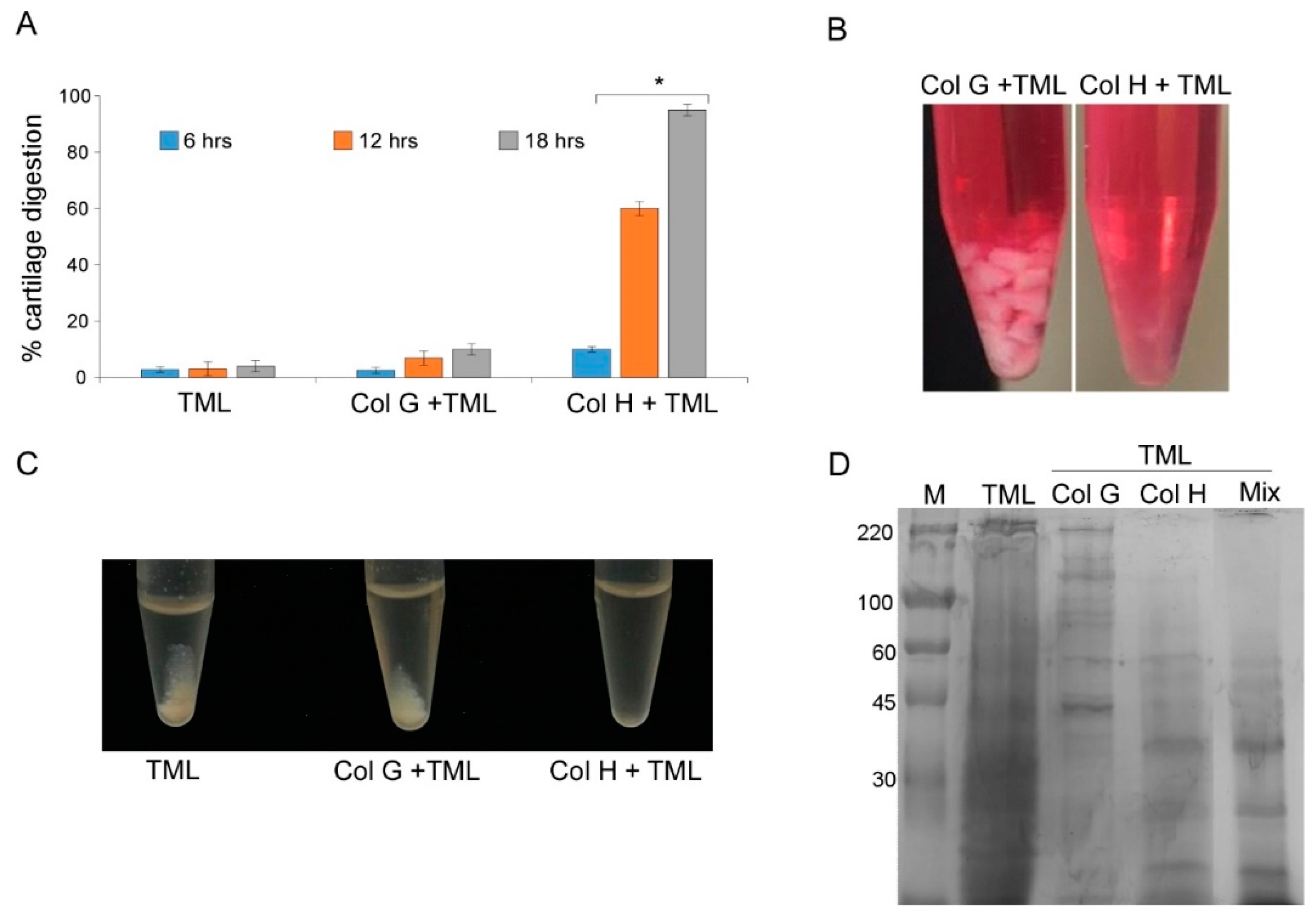
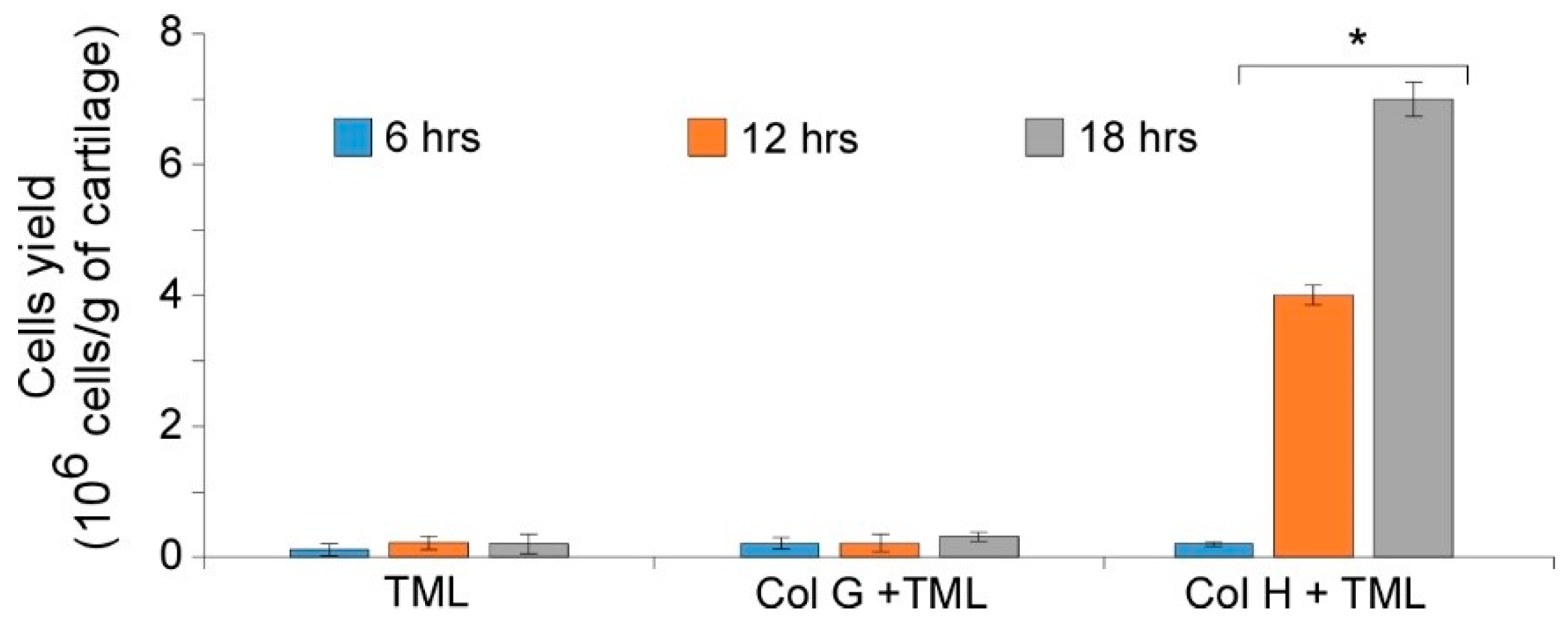
3.1. Cartilage Digestion Using Recombinant Collagenases G and H
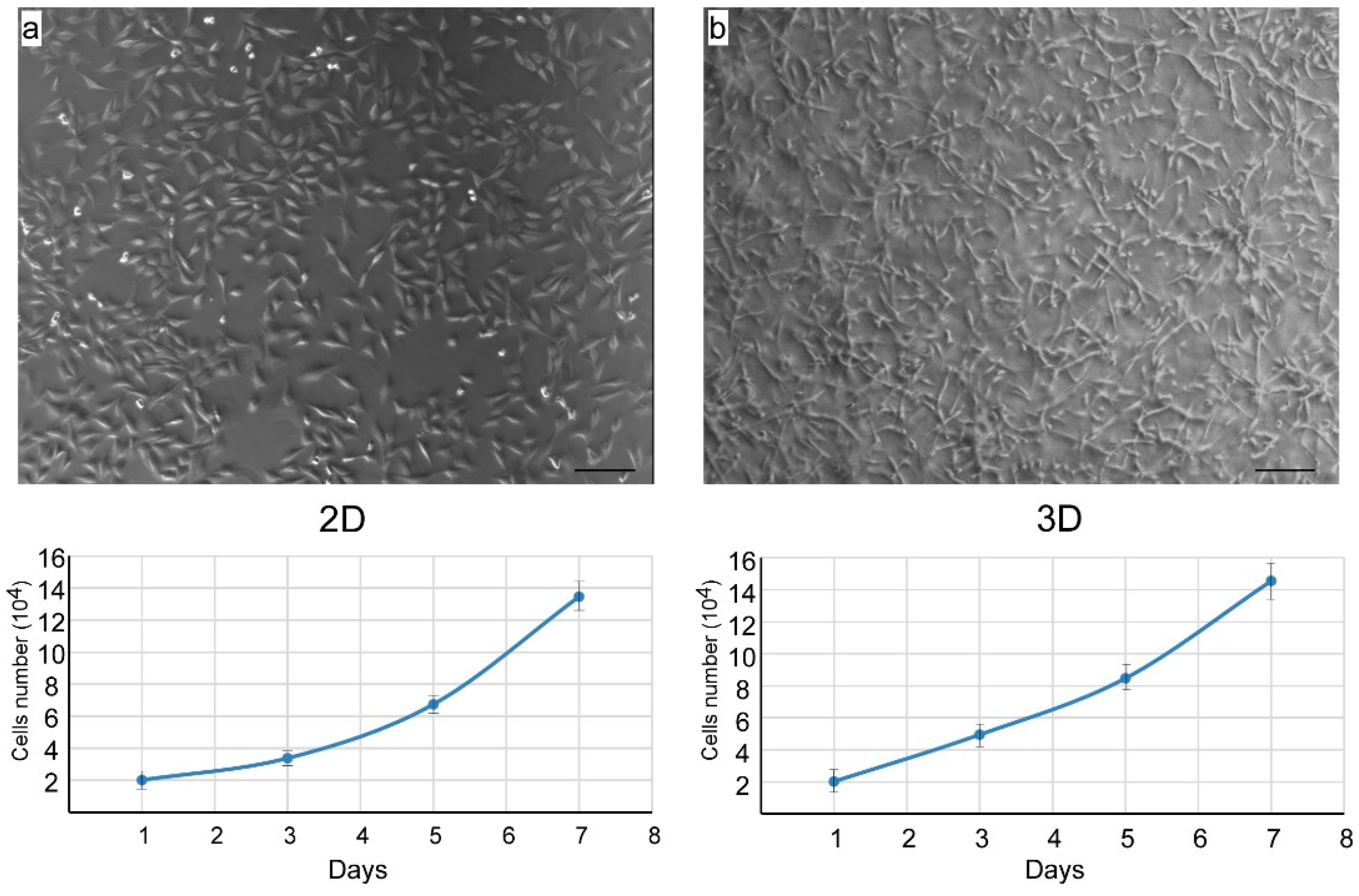
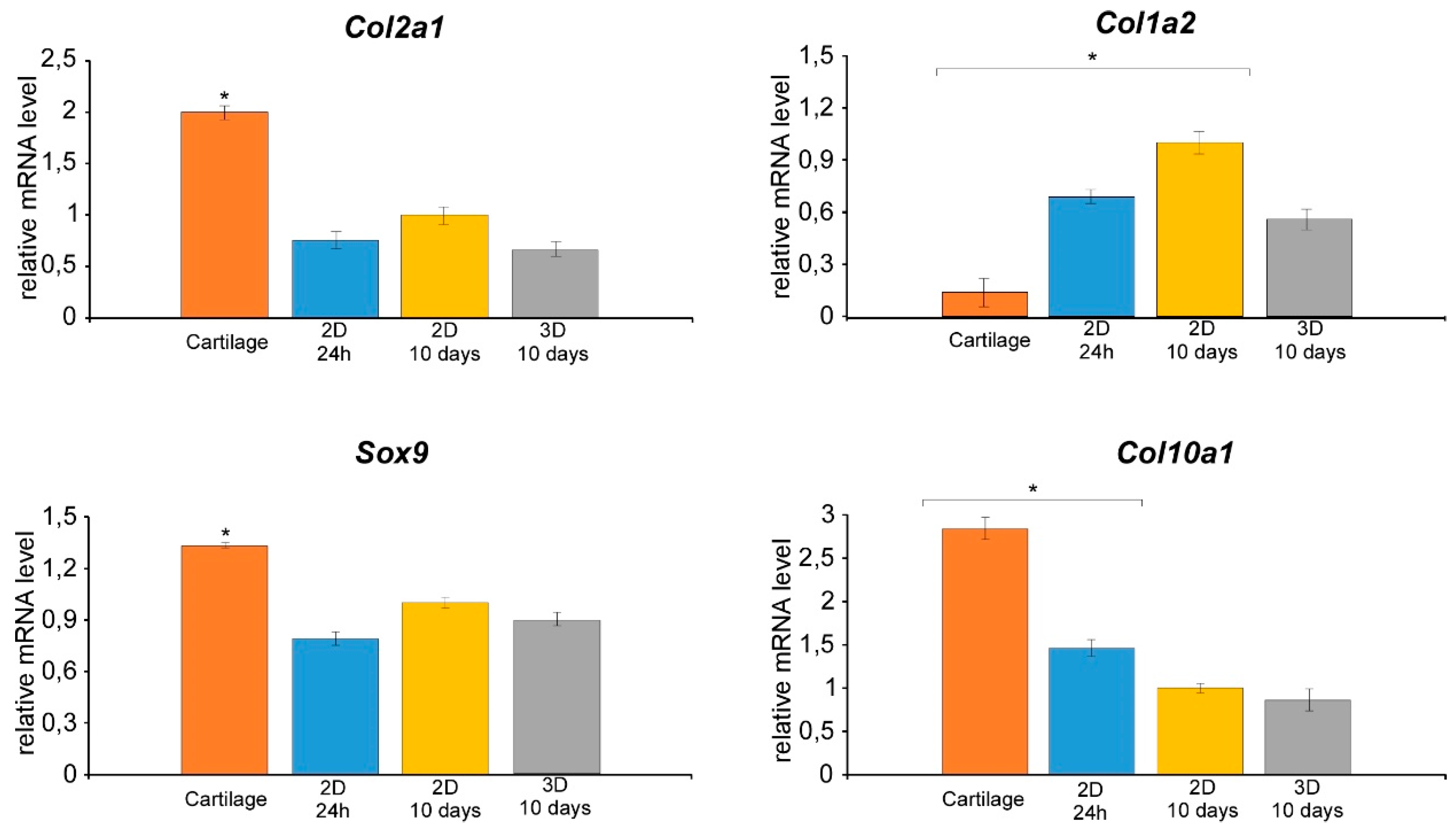
3.2. Isolation of Nasal Septal Chondrocytes and their characterization
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mao, A.S.; Mooney, D.J. Regenerative medicine: Current therapies and future directions. Proc. Natl. Acad. Sci. USA 2015, 112, 14452–14459. [Google Scholar] [CrossRef] [PubMed]
- Mumme, M.; Barbero, A.; Miot, S.; Wixmerten, A.; Feliciano, S.; Wolf, F.; Asnaghi, A.M.; Baumhoer, D.; Bieri, O.; Kretzschmar, M.; et al. Nasal chondrocyte-based engineered autologous cartilage tissue for repair of articular cartilage defects: An observational first-in-human trial. Lancet 2016, 388, 1985–1994. [Google Scholar] [CrossRef]
- Brittberg, M.; Lindahl, A.; Nilsson, A.; Ohlsson, C.; Isaksson, O.; Peterson, L. Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N. Engl. J. Med. 1994, 331, 889–895. [Google Scholar] [CrossRef] [PubMed]
- Oehme, D.; Goldschlager, T.; Ghosh, P.; Rosenfeld, J.; Jenkin, G. Cell-Based Therapies Used to Treat Lumbar Degenerative Disc Disease: A Systematic Review of Animal Studies and Human Clinical Trials. Stem Cells Int. 2015, 2015, 946031. [Google Scholar] [CrossRef]
- Smith, L.J.; Silverman, L.; Sakai, D.; Le Maitre, C.L.; Mauck, R.L.; Malhotra, N.R.; Buckley, C.T. Advancing cell therapies for intervertebral disc regeneration from the lab to the clinic: Recommendations of the ORS spine section. JOR Spine 2018, 1, e1036. [Google Scholar] [CrossRef]
- Martín, A.R.; Patel, J.M.; Zlotnick, H.M. Emerging therapies for cartilage regeneration in currently excluded ‘red knee’ populations. NPJ Regen. Med. 2019, 4, 1–11. [Google Scholar] [CrossRef]
- Vedicherla, S.; Buckley, C.T. Rapid chondrocyte isolation for tissue engineering applications: The effects of enzyme concentration and temporal exposure on the matrix forming capacity of nasal derived chondrocytes. BioMed Res. Int. 2017, 2017, 2395138. [Google Scholar] [CrossRef]
- Chang, A.A.; Reuther, M.S.; Briggs, K.K.; Schumacher, B.L.; Williams, G.M.; Corr, M.; Sah, R.L.; Watson, D. In vivo implantation of tissue-engineered human nasal septal neocartilage constructs: A pilot study. Otolaryngol. Head Neck Surg. 2012, 146, 46–52. [Google Scholar] [CrossRef]
- Pelttari, K.; Mumme, M.; Barbero, A.; Martin, I. Nasal chondrocytes as a neural crest-derived cell source for regenerative medicine. Curr. Opin. Biotechnol. 2017, 47, 1–6. [Google Scholar] [CrossRef]
- Pelttari, K.; Pippenger, B.; Mumme, M.; Feliciano, S.; Scotti, C.; Mainil-Varlet, P.; Procino, A.; Rechenberg, B.; Schwamborn, T.; Jakob, M.; et al. Adult human neural crest–derived cells for articular cartilage repair. Sci. Transl. Med. 2014, 6, 119. [Google Scholar] [CrossRef]
- Sarah, I.; Lepage, M.; Sharma, R.; Dukoff, D.; Stalker, L.; LaMarre, J.; Koch, T. Gene Expression Profile Is Different between Intact and Enzymatically Digested Equine Articular Cartilage. Cartilage 2019. [Google Scholar] [CrossRef]
- Miyazaki, Y.; Murayama, K.; Fathi, I.; Imura, T.; Yamagata, Y.; Watanabe, K. Strategy towards tailored donor tissue-specific pancreatic islet isolation. PLoS ONE 2019, 14, e0216136. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, O.; Jung, C.M.; Minami, J.; Katayama, S.; Nishi, N.; Okabe, A. A study of the collagen-binding domain of a 116-kDa Clostridium histolyticum collagenase. J. Biol. Chem. 1998, 273, 3643–3648. [Google Scholar] [CrossRef] [PubMed]
- Caviness, P.; Bauer, R.; Tanaka, K.; Janowska, K.; Roeser, J.R.; Harter, D.; Sanders, J.; Ruth, C.; Matsushita, O.; Sakon, J. Ca2+ induced orientation of tandem collagen binding domains from clostridial collagenase ColG permits two opposing functions of collagen fibril formation and retardation. FEBS J. 2018, 285, 3254–3269. [Google Scholar] [CrossRef] [PubMed]
- Philominathan, S.T.L.; Koide, T.; Hamada, K.; Yasui, H.; Seifert, S.; Matsushita, O.; Sakon, J. Unidirectional binding of Clostridial collagenase to triple helical substrates. J. Biol. Chem. 2009, 284, 10868–11087. [Google Scholar] [CrossRef]
- Eckhard, U.; Schonauer, E.; Nuss, D.; Brandstetter, H. Structure of collagenase G reveals a chew-and and digest mechanism of bacterial collagenolysis. Nat. Struct. Mol. Biol. 2011, 18, 1109–1114. [Google Scholar] [CrossRef]
- McCarthy, R.C.; Spurlin, B.; Wright, M.J.; Breite, A.G.; Sturdevant, L.K.; Dwulet, C.S.; Dwulet, F.E. Development and characterization of a collagen degradation assay to assess purified collagenase used in islet isolation. Transplant. Proc. 2008, 40, 339–342. [Google Scholar] [CrossRef]
- Salamone, M.; Seidita, G.; Cuttitta, A.; Rigogliuso, S.; Mazzola, S.; Bertuzzi, F.; Ghersi, G. A new method to value efficiency of enzyme blends for pancreatic tissue digestion. Transplant. Proc. 2010, 42, 2043–2048. [Google Scholar] [CrossRef]
- Salamone, M.; Cuttitta, A.; Bertuzzi, F.; Ricordi, C.; Ghersi, G.; Seidita, G. Biochemical Comparison Between Clostridium Hystoliticum Collagenases G and H obtained by DNA Recombinant and Extractive Procedures. Chem. Eng. Trans. 2012, 27, 259–264. [Google Scholar]
- Campora, S.; Sanjuan Alberte, P.; Bruno, C.; Ghersi, G. Isolation of Adult Rat Cardiomyocytes Using Recombinant Collagenases. Chem. Eng. Trans. 2018, 64, 25–30. [Google Scholar]
- Salamone, M.; Nicosia, A.; Bennici, C.; Quatrini, P.; Catania, V.; Mazzola, S.; Ghersi, G.; Cuttitta, A. Comprehensive Analysis of a Vibrio parahaemolyticus Strain Extracellular Serine Protease VpSP37. PLoS ONE 2015, 10, e0126349. [Google Scholar] [CrossRef] [PubMed]
- Salamone, M.; Saladino, S.; Pampalone, M.; Campora, S.; Ghersi, G. Tissue Dissociation and Primary Cells Isolation Using Recombinant Collagenases Class I and II. Chem. Eng. Trans. 2014, 38, 247–252. [Google Scholar]
- Volpe, L.; Salamone, M.; Giardina, A.; Ghersi, G. Optimization of a Biotechnological Process for Production and Purification of Two Recombinant Proteins: Col G and Col H. Chem. Eng. Trans. 2016, 49, 61–66. [Google Scholar]
- Moore, S.; Stein, W. Photometric Ninhydrin Method for Use in the Chromatography of Amino Acids. J. Biol. Chem. 1948, 176, 367–388. [Google Scholar]
- Ref, W.; Grassmann, A.; Nordwig, Z. Physiol. Chemie 1960, 322, 267. [Google Scholar]
- Laemmli, U.K.; Mölbert, E.; Showe, M.; Kellenberger, E. Form-determining function of the genes required for the assembly of the head of bacteriophage T4. J. Mol. Biol. 1970, 49, 99–113. [Google Scholar] [CrossRef]
- Pratta, M.A.; Yao, W.; Decicco, C.; Tortorella, M.D.; Liu, R.Q.; Copeland, R.A.; Magolda, R.; Newton, R.C.; Trzaskos, J.M.; Arner, E.C. Aggrecan protects cartilage collagen from proteolytic cleavage. J. Biol. Chem. 2003, 278, 45539–45545. [Google Scholar] [CrossRef]
- Miao, Z.; Lu, Z.; Huayu, W.; Liu, H.; Li, M.; Lei, D.; Zheng, L.; Zhao, J. Collagen, agarose, alginate, and Matrigel hydrogels as cell substrates for culture of chondrocytes in vitro: A comparative study. J. Cell Biochem. 2018, 119, 7924–7933. [Google Scholar] [CrossRef]
- Jin, G.; Kim, H.-W. Efficacy of collagen and alginate hydrogels for the prevention of rat chondrocyte dedifferentiation. J. Tissue Eng. 2018, 9, 2041731418802438. [Google Scholar] [CrossRef]
- Marshall, O.J.; Harley, V.R. Molecular mechanisms of SOX9 action. Mol. Genet. Metab. 2000, 71, 455–462. [Google Scholar] [CrossRef]
- Tew, S.R.; Clegg, P.D. Analysis of post transcriptional regulation ofSOX9 mRNA during in vitro chondrogenesis. Tissue Eng. Part A 2011, 17, 1801–1807. [Google Scholar] [CrossRef] [PubMed]
- Tsuchimochi, K.; Otero, M.; Dragomir, C.L.; Plumb, D.A.; Zerbini, L.F.; Libermann, T.A. GADD45beta enhances Col10a1 transcription via the MTK1/MKK3/6/p38 axis and activation of C/EBPbeta-TAD4 in terminally differentiating chondrocytes. J. Biol. Chem. 2010, 285, 8395–8407. [Google Scholar] [CrossRef] [PubMed]
- Angele, P.; Kujat, R.; Nerlich, M.; Yoo, J.; Goldberg, V.; Johnstone, B. Engineering of osteochondral tissue with bone marrow mesenchymal progenitor cells in a derivatized hyaluronan-gelatin composite sponge. Tissue Eng. 1999, 5, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Benya, P.D.; Shaffer, J.D. Dedifferentiated chondrocytes reexpress the differentiated collagen phenotype when cultured in agarose gels. Cell 1982, 30, 215–224.36. [Google Scholar] [CrossRef]
- Aulthouse, A.L.; Beck, M.; Griffey, E.; Sanford, J.; Arden, K.; Machado, M.A.; Horton, W.A. Expression of the human chondrocyte phenotype in vitro. Vitro Cell Dev. Biol. 1989, 25, 659–668. [Google Scholar] [CrossRef]
- Häuselmann, H.J.; Fernandes, R.J.; Mok, S.S.; Schmid, T.M.; Block, J.A.; Aydelotte, M.B.; Kuettner, K.E.; Thonar, E.J. Phenotypic stability of bovine articular chondrocytes after long-term culture in alginate beads. J. Cell Sci. 1994, 107, 17–27. [Google Scholar]
- Kolettas, E.; Buluwela, L.; Bayliss, M.T.; Muir, H.I. Expression of cartilage-specific molecules is retained on long-term culture of human articular chondrocytes. J. Cell Sci. 1995, 108, 1991–1999. [Google Scholar]
| Exp1 | Exp2 | Exp3 | |
|---|---|---|---|
| ColG | 2.5 U/mL | - | 2.5 U/mL |
| ColH | - | 30 U/mL | 30 U/mL |
| TML | 25 µg/mL | 25 µg/mL | 25 µg/mL |
| Primers | Sequences (5′–3′) | Accession Number |
|---|---|---|
| GAPDH | ATCTCGCTCCTGGAAGATG a TCGGAGTGAACGGATTCG b | NM_001034034 |
| Actin β | TGGGCATGGAATCCTG a GGCGCGATGATCTTGAT b | NM_173979 |
| 18S | TTCGATGGTAGTCGCTGTGC a TTGGATGTGGTAGCCGTTTC b | NR 036642 |
| Col1A2 | GGATGGTCACCCTGGAAAAC a CCCCTAATGCCCTTGAAGC b | NM_174520 |
| Col2A1 | TGATCGTGGTGACAAAGGTG a ATCTGGGCAGCAAAGTTTCC b | NM_001001135 |
| Sox9 | ACGCAGATTCCCAAGACAC a GGTTTCCAGTCCAGTTTCG b | XM_014478986 |
| Col10A1 | CTGGAGTGGGGAAAAGAGG a TGCCTTCTGGTCCTTGTTC b | NM_174634 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salamone, M.; Rigogliuso, S.; Nicosia, A.; Tagliavia, M.; Campora, S.; Cinà, P.; Bruno, C.; Ghersi, G. Neural Crest-Derived Chondrocytes Isolation for Tissue Engineering in Regenerative Medicine. Cells 2020, 9, 962. https://doi.org/10.3390/cells9040962
Salamone M, Rigogliuso S, Nicosia A, Tagliavia M, Campora S, Cinà P, Bruno C, Ghersi G. Neural Crest-Derived Chondrocytes Isolation for Tissue Engineering in Regenerative Medicine. Cells. 2020; 9(4):962. https://doi.org/10.3390/cells9040962
Chicago/Turabian StyleSalamone, Monica, Salvatrice Rigogliuso, Aldo Nicosia, Marcello Tagliavia, Simona Campora, Paolo Cinà, Carmelo Bruno, and Giulio Ghersi. 2020. "Neural Crest-Derived Chondrocytes Isolation for Tissue Engineering in Regenerative Medicine" Cells 9, no. 4: 962. https://doi.org/10.3390/cells9040962
APA StyleSalamone, M., Rigogliuso, S., Nicosia, A., Tagliavia, M., Campora, S., Cinà, P., Bruno, C., & Ghersi, G. (2020). Neural Crest-Derived Chondrocytes Isolation for Tissue Engineering in Regenerative Medicine. Cells, 9(4), 962. https://doi.org/10.3390/cells9040962






