Noble Metals and Soft Bio-Inspired Nanoparticles in Retinal Diseases Treatment: A Perspective
Abstract
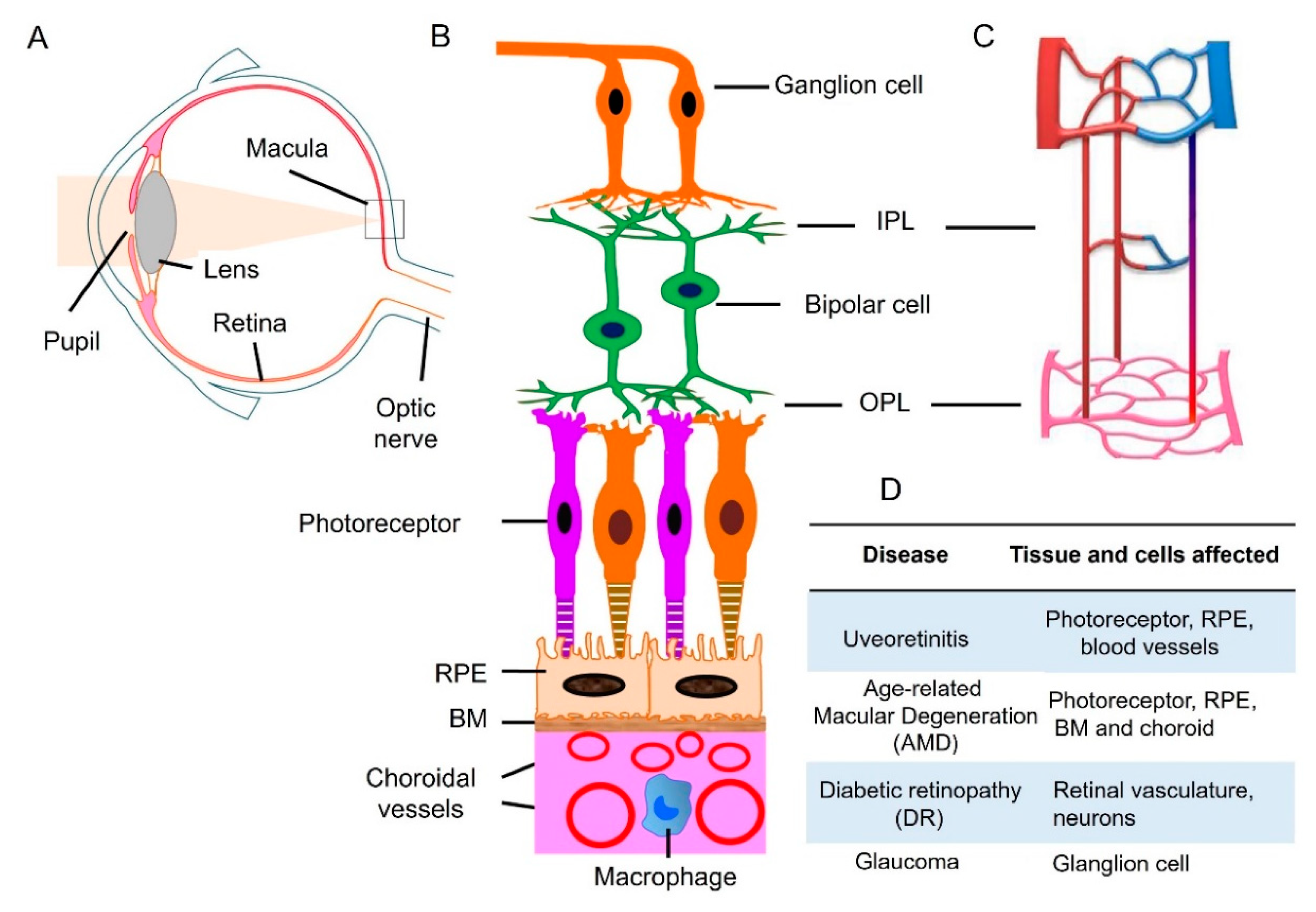
1. Introduction
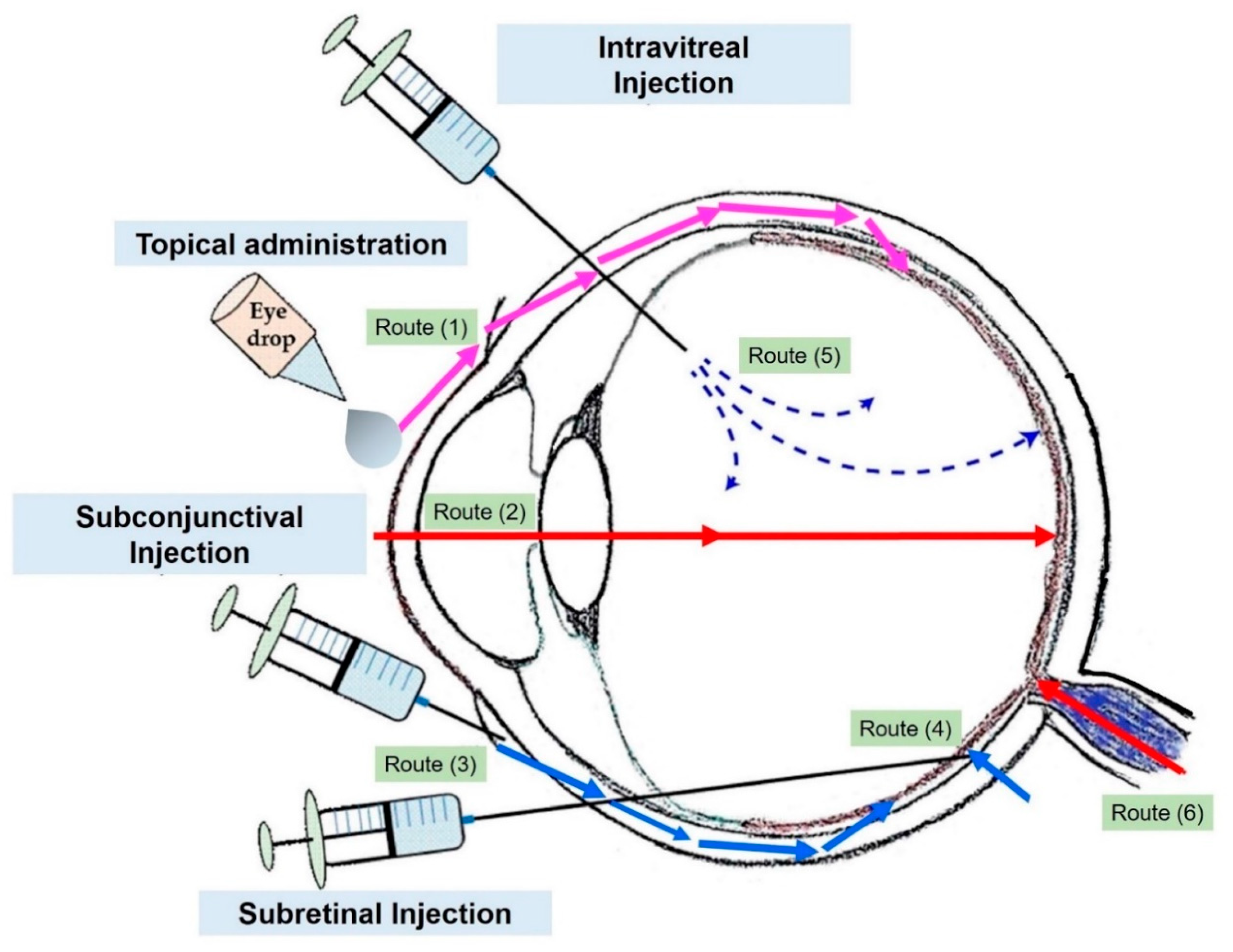
2. Drug Ocular Administrations
3. Physico-Chemical Properties of Nanomaterials
3.1. Noble Metal NPs: The Case of Gold (Au) and Silver (Ag)
3.2. Au and AgNPs: Safety Studies in In Vitro and In Vivo Retinal Models
3.2.1. AuNPs
3.2.2. AgNPs
4. Noble-Metal NPs as Therapy and Diagnostic for Retinal Disease
5. Bio-Inspired NPs for Ocular Drug Delivery
5.1. Liposomes as Drug Delivery Carrier
5.2. Niosomes for Drug and Gene Delivery
5.3. Hydrogels-Based Nanocarriers
5.3.1. Natural Hydrogels: Chitosan NPs (CHNPs)
5.3.2. Synthetic Hydrogel Based Nanocarriers
5.4. Active and Self-Propelling Nanoparticles: New Generation of Nanobots in Retinal Disease
6. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Davson, H. The Physiology of The Eye; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Stephen, R.; Wilkinson, C.; Schachat, A.; Hinton, D.; Wilkinson, C. Retina; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Günhan, E.; van der List, D.; Chalupa, L.M. Ectopic Photoreceptors and Cone Bipolar Cells in the Developing and Mature Retina. J. Neurosci. 2003, 15, 1383–1389. [Google Scholar] [CrossRef]
- Reichenbach, A.; Schnitzer, J.; Friedrich, A.; Brückner, G.; Schober, W. Development of the rabbit retina. Anat. Embryol. 1991, 183, 287–297. [Google Scholar] [CrossRef] [PubMed]
- Dilnawaz, F.S.S.K. Nanotechnology-Based Ophthalmic Drug Delivery System. In Focal Controlled Drug Delivery; Domb, A., Khan, W., Eds.; Advances in Delivery Science and Technology; Springer: Boston, MA, USA, 2014. [Google Scholar]
- Rakhi Dandonaa, L.D. Socioeconomic status and blindness. Br. J. Ophthalmol. 2001, 85, 1484–1488. [Google Scholar] [CrossRef] [PubMed]
- Yorston, D. The Retina Retinal Diseases and VISION 2020. Comm Eye Health 2003, 16, 19–20. [Google Scholar]
- Veleri, S.; Lazar, C.H.; Chang, B.; Sieving, P.A.; Banin, E.; Swaroop, A. Biology and therapy of inherited retinal degenerative disease: insights from mouse models. Dis. Models Mech. 2015, 8, 109–129. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Franco, Y.L.; Zhou, Y.; Chen, J. Nanotechnology in retinal drug delivery. Int. J. Ophthalmol. 2018, 11. [Google Scholar] [CrossRef]
- Gilbert, C.; Foster, A. Childhood blindness in the context of VISION 2020—The Right to Sight. Bull. World Health Organ. 2001, 79, 227–232. [Google Scholar]
- Jones, B.W.; Marc, R.E.; Pfeiffer, R.L. Retinal Degeneration, Remodeling and Plasticity. In Webvision: The Organization of the Retina and Visual System; Salt Lake City (UT): University of Utah Health Sciences Center, UT, USA, 2016. [Google Scholar]
- Rajappa, M.; Saxena, P.K.J. Ocular angiogenesis: Mechanisms and recent advances in therapy. Adv. Clin. Chem. 2010, 50, 103–121. [Google Scholar]
- Aiello, L.P.; Avery, R.L.; Arrigg, P.G.; Keyt, B.A.; Jampel, H.D.; Shah, S.T.; Pasquale, L.R.; Thieme, H.; Iwamoto, M.A.; Park, J.E.; et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopa-thy and other retinal disorders. N. Engl. J. Med. 1994, 331, 480–1487. [Google Scholar] [CrossRef]
- Weis, S.M.; Cheresh, D.A. Pathophysiological consequences of VEGF-induced vascular permeability. Nature 2005, 437, 497–504. [Google Scholar] [CrossRef]
- Kim, S.J. Novel Approaches for Retinal Drug and Gene Delivery. Transl. Vis. Sci. Technol. 2014, 3, 7. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bennett, J.; Chung, D.C.; Maguire, A. Gene delivery to the retina: From mouse to man. Methods Enzym. 2012, 507, 255–257. [Google Scholar]
- Xu, H.; Chen, M. Targeting the complement system for the management of retinal in fl ammatory and degenerative diseases. Eur. J. Pharmacol. 2016, 787, 94–104. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Coránguez, M.; Ramos, C.; Antonetti, D.A. The inner Blood-Retinal Barrier: Cellular Basis and Development. Vis. Res. 2017, 139, 123–137. [Google Scholar] [CrossRef]
- Gaudana, R.; Jwala, J.; Boddu, S.H.S.; Mitra, A.K. Recent Perspectives in Ocular Drug Delivery. Pharm. Res. 2009, 26, 1197–1216. [Google Scholar] [CrossRef]
- Kati-Sisko, V.; Hellinen, L. Expression, activity and pharmacokinetic impact of ocular transporters. Adv. Drug Deliv. Rev. 2018, 126, 3–22. [Google Scholar]
- Souto, E.B.; Dias-ferreira, J.; Ana, L.; Ettcheto, M.; Elena, S. Advanced Formulation Approaches for Ocular Drug Delivery: State-Of-The-Art and Recent Patents. Pharmaceutics 2019, 11, 460. [Google Scholar] [CrossRef]
- Le Bourlais, C.; Acar, L.; Zia, H.; Sado, P.A.; Needham, T.; Leverge, R. Ophthalmic drug delivery systems—Recent advances. Prog. Retin. Eye Res. 1998, 17, 33–58. [Google Scholar] [CrossRef]
- Sugrue, M.F. The pharmacology of antiglaucoma drugs. Pharmacol. Ther. 1989, 43, 91–1989. [Google Scholar] [CrossRef]
- Himawan, E.; Ekström, P.; Buzgo, M.; Gaillard, P.; Stefánsson, E.; Marigo, V.; Loftsson, T.; Paquet-Durand, F. Drug delivery to retinal photoreceptors. Drug Discov. Today 2019, 24, 1637–1643. [Google Scholar] [CrossRef]
- Marmor, M.F.; Akira Negi, D.M.M. Kinetics of macromolecules injected into the subretinal space. Exp. Eye Res. 1985, 40, 687–696. [Google Scholar] [CrossRef]
- Meyer, C.H.; Krohne, T.U.; Issa, P.C.; Liu, Z. Routes for Drug Delivery to the Eye and Retina: Intravitreal Injections. Retin. Pharmacother. Dev. Ophthalmol. 2016, 55, 63–70. [Google Scholar]
- Falavarjani, K.G.; Nguyen, Q.D. Adverse events and complications associated with intravitreal injection of anti-VEGF agents: A review of literature. Eye 2013, 27, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Raghava, S.; Hammond, M.K.U. Periocular routes for retinal drug delivery. Expert Opin. Drug Deliv. 2004, 1, 99–144. [Google Scholar] [CrossRef]
- Ghate, D.; Brooks, W.; McCarey, B.E.; Edelhauser, H.F. Pharmacokinetics of Intraocular Drug Delivery by Periocular Injections Using Ocular Fluorophotometry. Invest. Ophthalmol Vis. Sci. 2007, 48, 2230–2237. [Google Scholar] [CrossRef] [PubMed]
- Castellarinand, A.; Pieramici, D.J. Anterior segment complications following periocular and intraocular injections. Ophthalmol. Clin. N. Am. 2004, 17, 583–590. [Google Scholar] [CrossRef]
- Tsai, H.-H.; Wang, P.-Y.; Lin, I.-C.; Huang, H.; Liu, G.; Tseng, C.-L. Ocular Drug Delivery: Role of Degradable Polymeric Nanocarriers for Ophthalmic Application. Int. J. Mol. Sci. 2018, 19, 2830. [Google Scholar] [CrossRef]
- ISO, Int. Organ Stand Nanotechnologies Vocab Part 1 Core Terms ISO/TS 80004-12010. 2010. Available online: https://www.iso.org/standard/51240.html (accessed on 1 March 2020).
- ISO International Organization for Standardization. Technical Specification: Nanotechnologies Terminology and Definitions for Nanoobjects Nanoparticle, Nanofibre and Nanoplate. ISO/TS 80004-2:2008. 2008. Available online: https://www.iso.org/standard/44278.html (accessed on 1 March 2020).
- De Matteis, V.; Rinaldi, R. Toxicity Assessment in the Nanoparticle Era. In Cellular and Molecular Toxicology of Nanoparticles; Springer: Cham, Switzerland, 2018; pp. 1–19. [Google Scholar]
- Mu, Q.; Jiang, G.; Chen, L.; Zhou, H.; Fourches, D.; Tropsha, A.; Yan, B. Chemical Basis of Interactions Between Engineered Nanoparticles and Biological Systems. Chem. Rev. 2016, 7740–7781. [Google Scholar] [CrossRef]
- Bhatia, S. Nanoparticles Types, Classification, Characterization, Fabrication Methods and Drug Delivery Applications. In Natural Polymer Drug Delivery Systems; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Ruth Duncan, R.G. Nanomedicine(s) under the Microscope. Mol. Pharm. 2011, 8, 2101–2214. [Google Scholar] [CrossRef]
- Diebold, Y.; Calonge, M. Applications of nanoparticles in ophthalmology. Prog. Retin. Eye Res. 2010, 29, 596–609. [Google Scholar] [CrossRef]
- Farjo, K.M.; Ma, J. The potential of nanomedicine therapies to treat neovascular disease in the retina. J. Angiogenes Res. 2010, 2, 21. [Google Scholar] [CrossRef] [PubMed]
- JeffreyAdijanto, M.I.N. Nanoparticle-based technologies for retinal gene therapy. Eur. J. Pharm. Biopharm. 2015, 95, 353–367. [Google Scholar]
- Soto, F.; Chrostowski, R.; Soto, F. Frontiers of Medical Micro/Nanorobotics: In vivo Applications and Commercialization Perspectives Toward Clinical Uses. Front. Bioeng. Biotechnol. 2018, 6, 1–12. [Google Scholar] [CrossRef]
- Khlebtsovab, N.G.; Dykman, L.A. Optical properties and biomedical applications of plasmonic nanoparticles. J. Quant. Spectrosc. Radiat. Transf. 2010, 111, 1–35. [Google Scholar] [CrossRef]
- Anker, J.N.; Hall, W.P.; Lyandres, O.; Shah, N.C.; Zhao, J.V. Biosensing with plasmonic nanosensors. Nat. Mater. 2008, 7, 442–453. [Google Scholar] [CrossRef] [PubMed]
- Daniel, M.; Astruc, D. Gold Nanoparticles: Assembly, Supramolecular Chemistry, Quantum-Size-Related Properties, and Applications toward Biology, Catalysis, and Nanotechnology. Chem. Rev. 2004, 104, 293–346. [Google Scholar] [CrossRef]
- Asahi, T.; Uwada, T.; Masuhara, H. Single particle spectroscopic study on surface plasmon resonance probing local environmental conditions. Handai Nanophotonics 2006, 2, 219–228. [Google Scholar]
- Boken, J.; Khurana, P.; Thatai, S. Plasmonic nanoparticles and their analytical applications: A review. Appl. Spectrosc. Rev. 2017, 52, 774–820. [Google Scholar] [CrossRef]
- Loiseau, A.; Asila, V.; Boitel-aullen, G.; Lam, M. Silver-Based Plasmonic Nanoparticles for and Their Use in Biosensing. Biosens 2019, 2019, 9. [Google Scholar] [CrossRef]
- Rizzello, L.; Pompa, P.P. Nanosilver-based antibacterial drugs and devices: Mechanisms, methodological drawbacks, and guidelines. Chem. Soc. Rev. 2014, 43, 1501–1518. [Google Scholar] [CrossRef]
- Draslera, B.; Sayreb, P.; Günter, K.; Steinhäuserc, A.-R. In vitro approaches to assess the hazard of nanomaterials. NanoImpact 2018, 9, 51. [Google Scholar] [CrossRef]
- De Matteis, V. Exposure to Inorganic Nanoparticles: Routes of Entry, Immune Response, Biodistribution and In Vitro/In Vivo Toxicity Evaluation. Toxics 2017, 5, 29. [Google Scholar] [CrossRef] [PubMed]
- Naahidi, S.; Jafari, M.; Edalat, F.; Raymond, K.; Khademhosseini, A.C.P. Biocompatibility of engineered nanoparticles for drug delivery. J. Control. Release 2013, 10, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Turkevich, J.; Stevenson, P.C.; Hillier, J. A study of the nucleation and growth processes in the synthesis of colloidal gold. Faraday Soc. 1951, 11, 55–75. [Google Scholar] [CrossRef]
- Brust, M.; Walker, M.; Bethell, D.; Schiffrin, D.J. Synthesis of thiol-derivatised gold nanoparticles in a two-phaseliquid–liquid system. J. Chem. Soc. Chem. Commun. 1994, 7, 801–802. [Google Scholar] [CrossRef]
- Kumar, P.V.; Kala, S.M.J.; Prakash, K.S. Green synthesis of gold nanoparticles using Croton Caudatus Geisel leaf extract and their biological studies. Mater. Lett. 2019, 236, 19–22. [Google Scholar] [CrossRef]
- Molnár, Z.; Bódai, V.; Szakacs, G.; Erdélyi, B.; Fogarassy, Z. Green synthesis of gold nanoparticles by thermophilic filamentous fungi. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef]
- Giljohann, D.A.; Seferos, D.S.; Daniel, W.L.; Massich, M.D.; Patel, P.C. Gold Nanoparticles for Biology and Medicine. Angew. Chem. Int. Ed. Engl. 2010, 26, 3280–3294. [Google Scholar] [CrossRef]
- Orlando, A.; Colombo, M.; Prosperi, D.; Corsi, F.; Panariti, A.; Rivolta, I.; Masserini, M.; Cazzinga, E. Evaluation of gold nanoparticles biocompatibility: A multiparametric study on cultured endothelial cells and macrophages. J. Nanopart. Res. 2018, 18, 58. [Google Scholar] [CrossRef]
- Aghaie, T.; Jazayeri, M.H.; Manian, M.; Khani, l.; Erfani, M.; Rezayi, M.; Ferns, G.A.; Avan, A. Gold nanoparticle and polyethylene glycol in neural regeneration in the treatment of neurodegenerative diseases. J. Cell. Biochem. 2019, 120, 2749–2755. [Google Scholar] [CrossRef]
- Bailly, A.-L.; Correard, F.; Popov, A.; Gleb Tselikov, F.; Appay, R.; Al-Kattan, A.; Andrei, V.; Kabashin, D.B.; Esteve, M.-A. In vivo evaluation of safety, biodistribution and pharmacokinetics of lasersynthesized gold nanoparticles. Sci. Rep. 2019, 9, 12890. [Google Scholar] [CrossRef] [PubMed]
- Cassano, D.; Summa, M.; Pocoví-Martínez, S.; Mapanao, A.-K.; Catelani, T.; Bertorelli, R.; Voliani, V. Biodegradable Ultrasmall-in-Nano Gold Architectures: Mid-Period In Vivo Distribution and Excretion Assessment. Part. Part. Syst. Charact. 2019, 36, 1800464. [Google Scholar] [CrossRef]
- Khan, H.A.; Abdelhalim, M.A.K.; Alhomida, A.S.; Al-Ayed, M.S. Effects of Naked Gold Nanoparticles on Proinflammatory Cytokines mRNA Expression in Rat Liver and Kidney. Biomed. Res. Int. 2013, 2013, 590730. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.A.; Alamery, S.; Ibrahim, K.E.; El-Nagar, D.M.; Al-Harbi, N.; Rusop, M.; Alrokayan, S.H. Size and time-dependent induction of proinflammatory cytokines expression in brains of mice treated with gold nanoparticle. Saudi J. Biol. Sci. 2019, 26. [Google Scholar] [CrossRef]
- Karakoçak, B.B.; Raliya, R.; Davis, J.T.; Chavalmane, S.; Wang, W.N.; Ravi, N. Biocompatibility of gold nanoparticles in retinal pigment epithelial cell line. Toxicol. Vitr. 2016, 37, 61–69. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, J.H.; Kim, K.-W.; Kim, M.H. Intravenously administered gold nanoparticles pass through the blood–retinal barrier depending on the particle size, and induce no retinal toxicity. Nanotechnology 2009, 20, 505101. [Google Scholar] [CrossRef]
- Bakri, S.J.; Pulido, J.S.; Mukherjee, P.; Marler, R.J. Absence of histologic retinal toxicity of intravitreal nanogold in a rabbit model. Retin 2008, 2008, 147–149. [Google Scholar] [CrossRef]
- Söderstjerna, E.; Bauer, P.; Cedervall, T.; Abdshill, H.; Johansson, F.; Johansson, U.E. Silver and Gold Nanoparticles Exposure to In Vitro Cultured Retina—Studies on Nanoparticle Internalization, Apoptosis, Oxidative Stress, Glial- and Microglial Activity. PLoS ONE 2014, 9, e0105359. [Google Scholar] [CrossRef]
- Nakhlband, A. Impacts of nanomedicines in ocular pharmacotherapy. Bioimpacts 2011, 1, 7–22. [Google Scholar]
- Kittler, S.; Greulich Diendorf, M.; Köller, M.E. Toxicity of Silver Nanoparticles Increases during Storage Because of Slow Dissolution under Release of Silver Ions. Chem. Mater. 2010, 22, 4548–4554. [Google Scholar] [CrossRef]
- Yang, X.; Gondikas, A.P.; Marinakos, S.M.; Auffan, M.; Li, J.; Hsu-Kim, H. Mechanism of Silver Nanoparticle Toxicity Is Dependent on Dissolved Silver and Surface Coating in Caenorhabditis elegans. Environ. Sci. Technol. 2012, 46, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- De Matteis, V.; Malvindi, M.A.; Galeone, A.; Brunetti, V.; De Luca, E.; Kote, S.; Kshirsagar, P.; Sabella, S.; Bardi, G.; Pompa, P.P. Negligible particle-specific toxicity mechanism of silver nanoparticles: The role of Ag + ion release in the cytosol. Nanomedicine Nanotechnology. Biol. Med. 2015, 11, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, E.; Milani, M.; Fekri Aval, S.; Kouhi, M.; Akbarzadeh, A.; Tayefi Nasrabadi, H.; Nikasa, P.; Joo, S.W.; Hanifehpour, Y.; Nejati-Koshki, K.; et al. Silver nanoparticles: Synthesis methods, bio- applications and properties. Crit. Rev. Microbiol. 2016, 42, 173–180. [Google Scholar] [CrossRef]
- Zhang, T.; Song, Y.J.; Zhang, X.Y.; Wu, J.Y. Synthesis of silver nanostructures by multistep methods. Sensors (Switzerland) 2014, 14, 5860–5889. [Google Scholar] [CrossRef] [PubMed]
- Singh, C.; Kumar, J.; Kumar, P.; Singh Chauhan, B.; Tiwari, K.N.; Mishra, S.K.; Srikrishna, S.; Saini, R.; Nath, G.; Singth, J. Green synthesis of silver nanoparticles using aqueous leaf extract of Premna integrifolia (L.) rich in polyphenols and evaluation of their antioxidant, antibacterial and cytotoxic activity. Biotechnol. Biotechnol. Equip. 2019, 359–371. [Google Scholar] [CrossRef]
- Jun, Y.; Zi, Z.; Wanga, Y.; Zhaoa, G.; Liu, J.-X. Silver nanoparticles affect lens rather than retina development in zebrafish embryos. Ecotoxicol. Environ. Saf. 2018, 163, 279–288. [Google Scholar]
- Sriram, M.I.; Kalishwaralal, K.; Barathmanikanth, S. Size-based cytotoxicity of silver nanoparticles in bovine retinal endothelial cells. Nanosci. Methods 2012, 1, 56–77. [Google Scholar] [CrossRef]
- Kim, J.S.; Song, K.S.; Sung, J.H.; Ryu, H.R.; Choi, B.G.; Cho, H.S.; Lee, J.K. Genotoxicity, acute oral and dermal toxicity, eye and dermal irritation and corrosion and skin sensitisation evaluation of silver nanoparticles. Nanotoxicology 2013, 7, 953. [Google Scholar] [CrossRef]
- Maneewattanapinyo, P.; Banlunara, W.; Thammacharoen, C.; Ekgasit, S.; Kaewamatawong, T. An evaluation of acute toxicity of colloidal silver nanoparticles. J. Vet. Med. Sci. 2011, 73, 1417–1423. [Google Scholar] [CrossRef]
- De Matteis, V.; Cascione, M.; Cristina, C.; Rinaldi, R. Engineered Gold Nanoshells Killing Tumor Cells: New Perspectives. Curr. Pharm. Des. 2019, 25, 1477–1489. [Google Scholar] [CrossRef]
- Weissleder, R. A clearer vision for in vivo imaging. Nat. Biotechnol. 2001, 19, 316–317. [Google Scholar] [CrossRef] [PubMed]
- Song, H.B.; Wi, J.S.; Jo, D.H.; Kim, J.H.; Lee, S.W. Intraocular application of gold nanodisks optically tuned for optical coherence tomography: inhibitory effect on retinal neovascularization without unbearable toxicity. Nanomedicine 2017, 13, 1901–1911. [Google Scholar] [CrossRef] [PubMed]
- Gasparini, S.J.; Llonch, S.; Borsch, O. Transplantation of photoreceptors into the degenerative retina: Current state and future perspectives. Prog. Retin. Eye Res. 2019, 69, 1–37. [Google Scholar] [CrossRef]
- Chemla, Y.; Betzer, O.; Markus, A.; Farah, N.; Motiei, M. Gold nanoparticles for multimodal high-resolution imaging of transplanted cells for retinal replacement therapy. Nanomedicine 2019, 14, 1857–1871. [Google Scholar] [CrossRef]
- Kim, H.; Nguyen, V.; Manivasagan, P.; Kim, S.W.; Oh, J. Doxorubicin-fucoidan-gold nanoparticles composite for dualchemo-photothermal treatment on eye tumors. Oncotarget 2017, 8, 113719–113733. [Google Scholar] [CrossRef] [PubMed]
- Valluru, K.S.; Chinni, B.K.; Rao, N.A.; Bhatt, S.; Dogra, V.S. Basics and Clinical Applications of Photoacoustic Imaging. Ultrasound Clin. 2009, 4, 403–429. [Google Scholar] [CrossRef]
- Gordon, A.Y.; Lapierre-Landry, M.; Skala, M.C.; Penn, J.S. Photothermal Optical Coherence Tomography of AntiAngiogenic Treatment in the Mouse Retina Using Gold Nanorods as Contrast Agents. Transl. Vis. Sci. Technol. 2019, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.-M.; Hsiao, C.-Y.; Li, H.-J.; Fang, J.-Y.; Chang, D.-C.; Hung, C.-F. The Inhibitory Effects of Gold Nanoparticles on VEGF-A-Induced Cell Migration in Choroid-Retina Endothelial Cells. Int. J. Mol. Sci. 2020, 21, 109. [Google Scholar] [CrossRef]
- Gurunathanab, S.; Leeb, K.; Kalishwaralala, K.; Sheikpranbabua, S.; Vaidyanathana, R.; Eom, S.H. Antiangiogenic properties of silver nanoparticles. Biomaterials 2009, 30, 6341–6350. [Google Scholar] [CrossRef]
- Sheikpranbabu, S.; Kalishwaralal, K.; Venkataraman, D.; Eom, S.H.; Park, J.; Gurunathan, S. Silver nanoparticles inhibit VEGF-and IL-1β-induced vascular permeability via Src dependent pathway in porcine retinal endothelial cells. Nanobiotechnol. 2009, 7, 8. [Google Scholar] [CrossRef]
- Ngandeu Neubi, G.M.; Opoku-Damoah, Y.; Gu, X.; Han, Y.; Zhou, J.; Ding, Y. Bio-inspired drug delivery systems: an emerging platform for targeted cancer therapy. Biomater. Sci. 2018, 6, 958–973. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Kuang, H.; Zhang, W.; Aguilar, Z.P.; Wei, H.; Xu, H. Comparisons of the biodistribution and toxicological examinations after repeated intravenous administration of silver and gold nanoparticles in mice. Sci. Rep. 2017, 7, 3303. [Google Scholar] [CrossRef] [PubMed]
- Madamsetty, V.S.; Mukherjee, A.; Mukherjee, S. Recent Trends of the Bio-Inspired Nanoparticles in Cancer Theranostics. Front. Pharmacol. 2019, 10, 1264. [Google Scholar] [CrossRef] [PubMed]
- Birngruber, T.; Raml, R.; Gladdines, W.; Gatschelhofer, C.; Gander, E.; Ghosh, A.; Kroath, T.; Gaillard, P.J.; Pieber, T.R. Enhanced Doxorubicin Delivery to the Brain Administered Through Glutathione PEGylated Liposomal Doxorubicin (2B3-101) as Compared with Generic Caelyx,®/Doxil®—A Cerebral Open Flow Microperfusion Pilot Study. J. Pharm. Sci. 2014, 203, 1945–1948. [Google Scholar] [CrossRef]
- Trost, A.; Lange, S.; Schroedl, F.; Bruckner, D.; Motloch, K.A.; Bogner, B.; Kaser-Eichberger, A.; Strohmaier, C.; Runge, C.; Aigner, L.; et al. Brain and Retinal Pericytes: Origin, Function and Role. Front. Cell Neurosci. 2016, 4, 20. [Google Scholar] [CrossRef] [PubMed]
- Haque, S.; Shadab, M.D.; Alam, I.; Sahni, J.K.; Ali, J. Nanostructure-based drug delivery systems for brain targeting. Drug Dev. Ind. Pharm. 2012, 38, 387–411. [Google Scholar] [CrossRef] [PubMed]
- Diebold, Y.; Jarrín, M.; Sáez, V.; Carvalho, E.L.; Orea, M.; Calonge, M.; Seijo, B. Ocular drug delivery by liposome-chitosan nanoparticle complexes (LCS-NP). Biomaterials 2007, 28, 1553–1564. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; He, R.; Qian, J.; Guo, J.; Xue, K.; Yuan, Y. Immunology and Microbiology Treatment of Experimental Autoimmune Uveoretinitis with Intravitreal Injection of Tacrolimus (FK506) Encapsulated in Liposomes. Investig. Ophthalmol. Vis. Sci. 2010, 51, 3575–3582. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Zheng, M. Chrysophanol liposome preconditioning protects against cerebral ischemia–reperfusion injury by inhibiting oxidative stress and apoptosis in mice. Int. J. Pharmacol. 2014, 10, 55–68. [Google Scholar] [CrossRef]
- Choi, Y.H. Berberine hydrochloride protects C2C12 myoblast cells against oxidative stress-induced damage via induction of Nrf-2-mediated HO-1 expression. Drug Dev. Res. 2016, 77, 8. [Google Scholar] [CrossRef]
- Lai, S.; Wei, Y.; Wu, Q.; Zhou, K.; Liu, T.; Zhang, Y.; Jiang, N.; Xiao, W.; Chen, J.; Liu, Q.; et al. Liposomes for efFective drug delivery to the ocular posterior chamber. J. Nanobiotechnol. 2019, 17, 64. [Google Scholar] [CrossRef] [PubMed]
- Wolin, L.R.; Massopust, L.C. Characteristics of the ocular fundus in primates. J. Anat. 1967, 101, 693–699. [Google Scholar] [PubMed]
- Wang, J.-L.; Liu, Y.-L.; Li, Y.; Dai, W.-B.; Guo, Z.-M.; Wang, Z.-H.; Zhang, Q. EphA2 targeted doxorubicin stealth liposomes as a therapy system for choroidal neovascularization in rats. Ophthalmol. Vis. Sci. 2012, 53, 7348. [Google Scholar] [CrossRef] [PubMed]
- Ravar, F.; Saadat, E.; Gholami, M.; Dehghankelishadi, P.; Mahdavi, M.; Azami, S. Hyaluronic acid-coated liposomes for targeted delivery of paclitaxel, in-vitro characterization and in-vivo evaluation. J. Control. Release 2016, 10, 10–22. [Google Scholar] [CrossRef]
- Lee, J.; Goh, U.; Lee, H.J.; Kim, J.; Jeong, M. Effective retinal penetration of lipophilic and lipid-conjugated hydrophilic agents delivered by engineered liposomes. Mol. Pharm. 2017, 14, 423–430. [Google Scholar] [CrossRef]
- Hironaka, K.; Inokuchi, Y.; Fujisawa, T.; Shimazaki, H.; Akane, M.; Tozuka, Y.; Tsuruma, K.; Shimazawa, M.; Hara, H. Edaravone-loaded liposomes for retinal protection against oxidative stress-induced retinal damage. Eur. J. Pharm. Biopharm. 2011, 79, 119–125. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Kuwahara, T.; Watanabe, K. Antioxidant activity of 3-methyl-1-phenyl-2-pyrazolin-5-one. Redox Rep. 1996, 2, 333–338. [Google Scholar] [CrossRef]
- Vighi, E.; Trifunović, D.; Veiga-Crespo, P.; Rentsch, A.; Hoffmann, D.; Sahaboglu, A.; Strasser, T.; Kulkarni, M.; Bertolotti, E.; van den Heuvel, A.; et al. Combination of cGMP analogue and drug delivery system provides functional protection in hereditary retinal degeneration. Proc. Natl. Acad. Sci. USA 2018, 27, E2997–E3006. [Google Scholar] [CrossRef]
- Lucas, K.A.; Pitari, G.M.; Kazerounian, S.; Ruiz-Stewart, I.; Park, J.; Schulz, S.; Chepenik, K.P.; Waldman, S.A. Guanylyl cyclases and signaling by cyclic GMP. Pharmacol. Rev. 2000, 52, 375–414. [Google Scholar]
- Farber, D.B.; Lolley, R.N. Cyclic guanosine monophosphate: Elevation in degenerating photoreceptor cells of the C3H mouse retina. Science 1974, 186, 449–451. [Google Scholar] [CrossRef]
- Campbell, M.; Humphries, M.M.; Kiang, A.-S.; Nguyen, A.T.H.; Gobbo, O.L.; Tam, L.C.S.; Suzuki, M.; Hanrahan, F.; Ozaki, E.; Farrar, G.-J.; et al. Systemic low-molecular weight drug delivery to pre-selected neuronal regions. EMBO Mol. Med. 2011, 3, 235–245. [Google Scholar] [CrossRef]
- Davis, B.M.; Normando, E.M.; Guo, L.; Turner, L.A.; Nizari, S. Topical Delivery of Avastin to the Posterior Segment of the Eye In Vivo Using Annexin A5-associated Liposomes. Small 2014, 24, 1575–1584. [Google Scholar] [CrossRef] [PubMed]
- Platania, C.B.M.; Fisichella, V.; Fidilio, A.; Geraci, F.; Lazzara, F.; Leggio, G.M.; Salomone, S.; Drago, F.; Pignatello, R.; Caraci, F.; et al. Topical Ocular Delivery of TGF- β 1 to the Back of the Eye: Implications in Age-Related Neurodegenerative Diseases. Int. J. Mol. Sci. 2017, 18, 2076. [Google Scholar] [CrossRef] [PubMed]
- Boulyjenkov, V.; Berg, K. Gene Therapy: Promises, Problems and Prospects; Genes Resist. to Dis.; Springer: Berlin/Heidelberg, Germany, 2000. [Google Scholar]
- Ramamoorth, M.; Narvekar, A. Non viral vectors in gene therapy—An overview. J. Clin. Diagn. Res. 2015, 9, GE01. [Google Scholar] [CrossRef]
- Das, K.G.; Ram, A.R. Niosome ad a novel drug delivery system: A review. Appl. Pharm. 2013, 5, 1–7. [Google Scholar]
- Seleci, D.A.; Seleci, M.; Walter, J.G. Niosomes as Nanoparticular Drug Carriers: Fundamentals and Recent Applications. J. Nanomat. 2016, 2016, 13. [Google Scholar] [CrossRef]
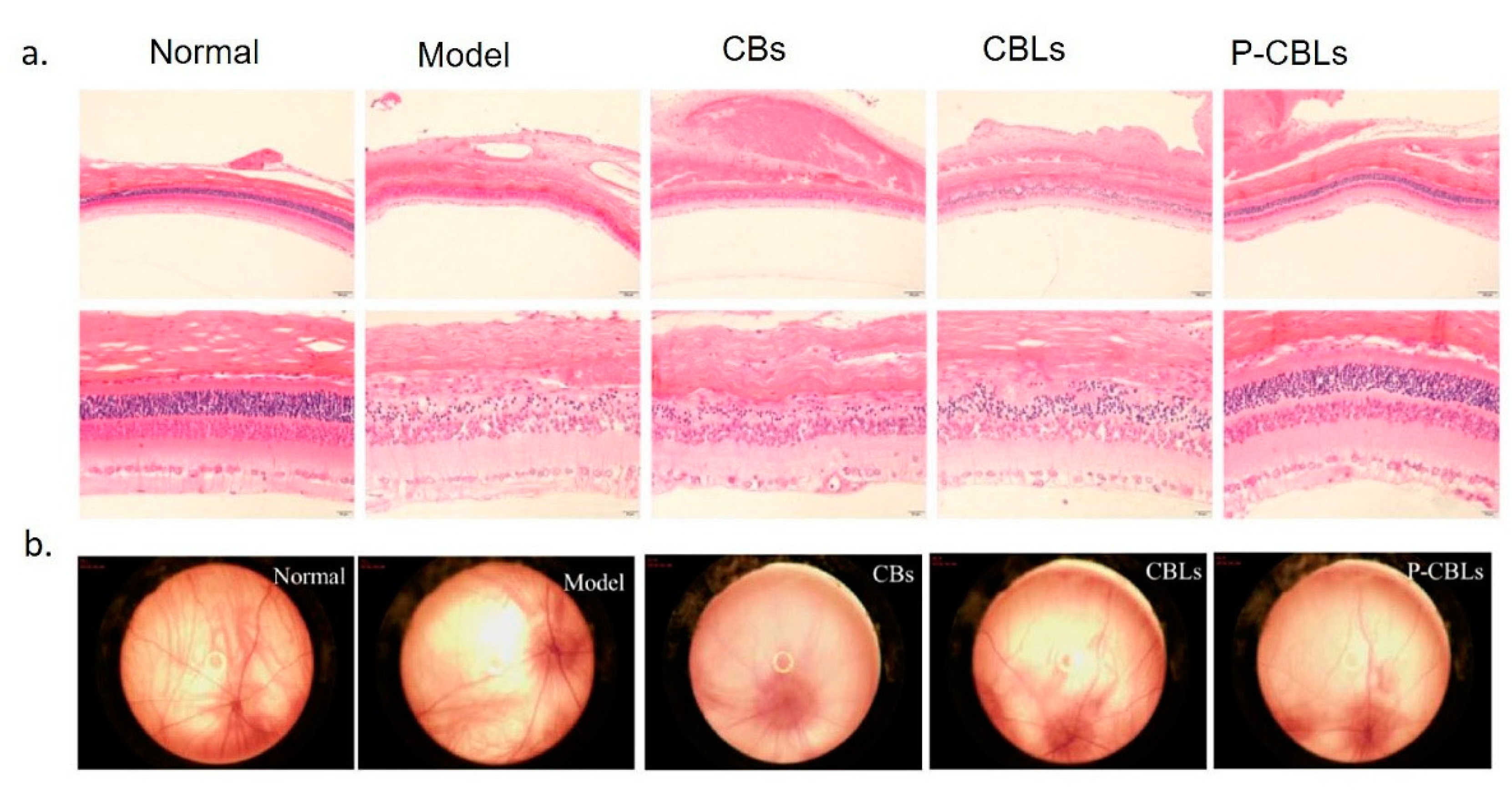
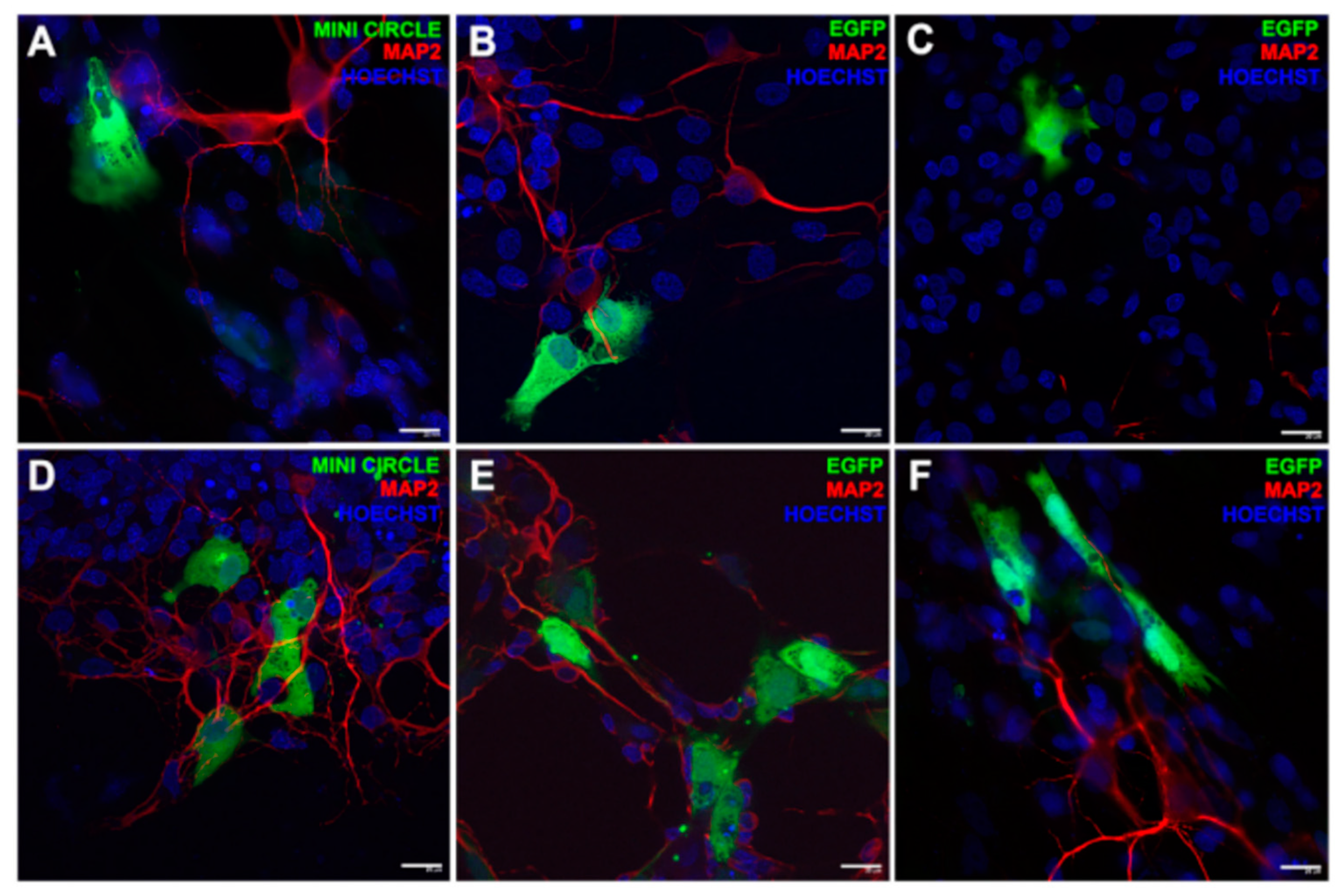
- Gallego, I.; Villate-Beitia, I.; Martínez-Navarrete, G.; Menéndez, M.; López-Méndez, T.; Soto-Sánchez, C.; Zárate, J.; Puras, G.; Fernández, E.; Pedraz, J.L. Non-viral vectors based on cationic niosomes and minicircle DNA technology enhance gene delivery efficiency for biomedical applications in retinal disorders. Nanomedicine. 2019, 17, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Mashal, M.; Attia, N.; Puras, G.; Martinez-Navarrete, G.; Fernandez, E.P. Retinal gene delivery enhancement by lycopene incorporation into cationic niosomes based on DOTMA and polysorbate 60. J. Control. Release 2017, 254, 55–64. [Google Scholar] [CrossRef]
- Guinedi, A.S.; Mortada, N.D.; Mansour, S. Preparation and evaluation of reverse-phase evaporation and multilamellar niosomes as ophthalmic carriers of acetazolamide. Int. J. Pharm. 2005, 306, 71–82. [Google Scholar] [CrossRef]
- Guglevaa, V.; Titevaa, S.; Rangelovb, S.; Momekova, D. Design and in vitro evaluation of doxycycline hyclate niosomes as a potential ocular delivery system. Int. J. Pharm. 2019, 5, 567. [Google Scholar] [CrossRef]
- Cintia, S.; De Paivaa Rosa, M.; Corralesa Arturo, L.; Villarreala William, J.; De-Quan, F.; Lia Michael, E.; Sternb Stephen, C. Corticosteroid and doxycycline suppress MMP-9 and inflammatory cytokine expression, MAPK activation in the corneal epithelium in experimental dry eye. Exp. Eye Res. 2006, 83, 526–535. [Google Scholar]
- Sapino, S.; Chirio, D.; Peira, E.; Abellán Rubio, E.; Brunella, V.; Jadhav, S.A.; Chindamo, G.; Gallarate, M. Ocular Drug Delivery: A Special Focus on the Thermosensitive Approach. Nanomaterials (Basel) 2019, 9, 884. [Google Scholar] [CrossRef] [PubMed]
- Hoare, T.R.; Kohane, D.S. Hydrogels in drug delivery: Progress and challenges. Polymer (Guildf) 2008, 49, 1993–2007. [Google Scholar] [CrossRef]
- Wang, K.; Han, Z. Injectable hydrogels for ophthalmic applications. J. Control. Release 2017, 268, 212–224. [Google Scholar] [CrossRef]
- Ferreira, N.N.; Ferreira, L.M.B.; Cardoso, V.M.O.; Boni, F.I.; Souza, A.L.R.; Gremião, M.P.D. Recent advances in smart hydrogels for biomedical applications: From self-assembly to functional approaches. Eur. Polym. J. 2018, 99, 117–133. [Google Scholar] [CrossRef]
- Elieh-Ali-Komi, D.; Hamblin, M.R. Chitin and Chitosan: Production and Application of Versatile Biomedical Nanomaterials. Int. J. Adv. Res. (Indore) 2016, 4, 411–427. [Google Scholar]
- ValérieDodane, V.D. Pharmaceutical applications of chitosan. Pharm. Sci. Technol. Today 1998, 1, 246–253. [Google Scholar]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Paliwal, R.; Paliwal, S.R.; Sulakhiya, K.; Kurmi, B.D.; Kenwat, R.; Mamgain, A. Chitosan-Based Nanocarriers for Ophthalmic Applications; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Nagarwal, R.C.; Kumar, R. Chitosan coated sodium alginate-chitosan nanoparticles loaded with 5-FU for ocular delivery: in vitro characterization and in vivo study in rabbit eye. Eur. J. Pharm. Sci. 2012, 20, 678–685. [Google Scholar] [CrossRef]
- Ameeduzzafar Imam, S.S.; Abbas Bukhari, S.N.; Ahmad, J. Formulation and optimization of levofloxacin loaded chitosan nanoparticle for ocular delivery: In-vitro characterization, ocular tolerance and antibacterial activity. Int. J. Biol. Macromol. 2018, 108, 650–659. [Google Scholar] [CrossRef]
- Andrei, G.; Peptu, C.A.; Popa, M.; Desbrieres, J.; Peptu, C.; Gardikiotis, F.; Costuleanu, M.; Costin, D.; Dupin, J.C.; Uhart, A. Formulation and evaluation of cefuroxim loaded submicron particles for ophthalmic delivery. Int. J. Pharm. 2015, 30, 16–29. [Google Scholar] [CrossRef]
- Kaskoos, R. Investigation of moxifloxacin loaded chitosan-dextran nanoparticles for topical instillation into eye: In-vitro and ex-vivo evaluation. Int. J. Pharm. Investig. 2014, 4, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Solanki, A.; Smalling, R.; Parola, A.H.; Nathan, I.; Kasher, R.; Pathak, Y.; Sutariya, V. Humanin Nanoparticles for Reducing Pathological Factors Characteristic of Age-related Macular Degeneration. Curr. Drug Deliv. 2019, 16, 226–232. [Google Scholar] [CrossRef]
- Cohen, A.; Lerner-Yardeni, J.; Meridor, D.; Kasher, R.; Nathan, I.; Parola, A. Humanin derivatives inhibit necrotic cell death in neurons. Mol. Med. 2015, 21, 505. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Ko, Y.; Chang, Y.; Huang, S. Thermosensitive chitosan-gelatin-based hydrogel containing curcumin- loaded nanoparticles and latanoprost as a dual-drug delivery system for glaucoma treatment. Exp. Eye Res. 2019, 179, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Radomska-leśniewska, D.M.; Osiecka-iwan, A.; Hyc, A.; Góźdź, A.; Dąbrowska, A.M.; Skopiński, P. Therapeutic potential of curcumin in eye diseases. Cent. Eur. J. Immunol. 2019, 44, 2–4. [Google Scholar] [CrossRef] [PubMed]
- Nathan Gooch, S.A.M.; Condie, R.; Michael, B.R.; Bonnie Archer, B.K.A.; Ocular, B. Drug Delivery for Glaucoma Management. Pharmaceutics 2012, 4, 197–211. [Google Scholar] [CrossRef]
- Michael, P.; Fautsch, D.H.J. Aqueous Humor Outflow: What Do We Know? Where Will It Lead Us? Investig. Ophthalmol. Vis. Sci. 2006, 47, 4181–4187. [Google Scholar]
- Silva, B.; Marto, J.; Braz, B.S.; Delgado, E.; Almeida, A.J.; Gonçalves, L. New nanoparticles for topical ocular delivery of erythropoietin. Int. J. Pharm. 2020, 119020. [Google Scholar] [CrossRef]
- Pandit, J.; Sultana, Y.; Aqil, M. Chitosan-coated PLGA nanoparticles of bevacizumab as novel drug delivery to target retina: optimization, characterization, and in vitro toxicity evaluation. Artif. Cells Nanomed. Biotechnol. 2016, 45, 1397–1407. [Google Scholar] [CrossRef]
- Mitra, R.N.; Han, Z.; Merwin, M.; Taai, M.; Al Conley, S.M. Synthesis and Characterization of Glycol Chitosan DNA Nanoparticles for Retinal Gene Delivery. ChemMedChem 2014, 9, 189–196. [Google Scholar] [CrossRef]
- Lee, J.-H.; Kim, H.-W. Emerging properties of hydrogels in tissue engineering. J. Tissue Eng. 2018, 9, 2041731418768285. [Google Scholar] [CrossRef] [PubMed]
- Chang, D.; Kinam Park, A. Hydrogels for sustained delivery ofbiologics to the back of the eye. Drug Discov. Today 2019, 24, 1470–1482. [Google Scholar] [CrossRef] [PubMed]
- Van Hovea, A.H.; Beltejar, M.G.; Benoit, D.S.W. Development and in vitro assessment of enzymatically-responsive poly(ethylene glycol) hydrogels for the delivery of therapeutic peptides. Biomaterials 2014, 35, 9719–9730. [Google Scholar] [CrossRef] [PubMed]
- Drapala, P.W.; Jiang, B.; Chiu, Y.-C.; Mieler, W.F.; Brey, E.M. The Effect of Glutathione as Chain Transfer Agent in PNIPAAm-Based Thermo-responsive Hydrogels for Controlled Release of Proteins. Pharm. Res. 2014, 31, 742–753. [Google Scholar] [CrossRef] [PubMed]
- Schultz, C.; Breaux, J.; Pharmd, J.S.; Douglas Morck, D.V.M. Drug delivery to the posterior segment of the eye through hydrogel contact lenses. Clin. Exp. Optom. 2011, 94, 212–218. [Google Scholar] [CrossRef]
- Kabiri, M.; Kamal, S.H.; Pawar, S.V.; Roy, P.R.; Derakhshandeh, M.; Kumar, U.; Hatzikiriakos, S.G.; Hossain, S.; Yadav, V.G. A stimulus-responsive, in situ-forming, nanoparticle-laden hydrogel for ocular drug delivery. Drug Deliv. Transl. Res. 2018, 8, 484–495. [Google Scholar] [CrossRef]
- Osswald, C.R.; Kang-Mieler, J.J. Controlled and Extended In Vitro Release of Bioactive Anti-Vascular Endothelial Growth Factors from a Microsphere-Hydrogel Drug Delivery System. Curr. Eye Res. 2016, 41, 1216–1222. [Google Scholar] [CrossRef]
- Agrahari, V.; Agrahari, V.; Hung, W.-T.; Christenson, L.K. Composite Nanoformulation Therapeutics for Long-Term Ocular Delivery of Macromolecules. Mol. Pharm. 2016, 13, 2912–2922. [Google Scholar] [CrossRef]
- Yang, H.; Tyagi, P.; Kadam, R.S.; Holden, C.A. Hybrid dendrimer hydrogel/PLGA nanoparticle platform sustains drug delivery for one week and antiglaucoma effects for four days following one-time topical administration. ACS Nano 2012, 25, 7595–7606. [Google Scholar] [CrossRef]
- Wadhams, G.H.; Armitage, J.P. Making sense of it all: Bacterial chemotaxis. Nat. Rev. Mol. Cell. Biol. 2004, 5, 1024–1037. [Google Scholar] [CrossRef] [PubMed]
- Micali, G.; Endres, R.G. Bacterial chemotaxis: information processing, thermodynamics, and behavior. Curr. Opin. Microbiol. 2016, 30, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Mei, Y.; Alexander, A. Solovev SS and OGS. Rolled-up nanotech on polymers: from basic perception to self-propelled catalytic microengines. Chem. Soc. Rev. 2011, 40, 2109–2119. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.S.; Philominathan, P. The physics of flagellar motion of E. coli during chemotaxis. Biophys. Rev. 2010, 2, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Troll, J.; Jeong, H.-H.; Wei, Q.; Stang, M.; Ziemssen, F.; Wang, Z.; Dong, M.; Schnichels, S.; Qui, T.; et al. A swarm of slippery micropropellers penetrates the vitreous body of the eye. Sci. Adv. 2018, 4, eaat4388. [Google Scholar] [CrossRef] [PubMed]
- Bohn, H.F.; Federle, W. Insect aquaplaning: Nepenthes pitcher plants capture prey with the peristome, a fully wettable water-lubricated anisotropic surface. Proc. Natl. Acad. Sci. USA 2004, 101, 14138–14143. [Google Scholar] [CrossRef]
- Wong, T.-S.; Kang, S.H.; Tang, S.K.Y.; Smythe, E.J.; Hatton, B.D.; Grinthal, A. Bioinspired self-repairing slippery surfaces with pressure-stable omniphobicity. Nature 2011, 477, 443–447. [Google Scholar] [CrossRef]
- Nistor, M.T.; Rusu, A.G. Nanorobots with Applications in Medicine; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Alhalafi, A.M. Applications of polymers in intraocular drug delivery systems. Oman J. Ophtalmol. 2017, 10, 3–8. [Google Scholar] [CrossRef]



© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Matteis, V.; Rizzello, L. Noble Metals and Soft Bio-Inspired Nanoparticles in Retinal Diseases Treatment: A Perspective. Cells 2020, 9, 679. https://doi.org/10.3390/cells9030679
De Matteis V, Rizzello L. Noble Metals and Soft Bio-Inspired Nanoparticles in Retinal Diseases Treatment: A Perspective. Cells. 2020; 9(3):679. https://doi.org/10.3390/cells9030679
Chicago/Turabian StyleDe Matteis, Valeria, and Loris Rizzello. 2020. "Noble Metals and Soft Bio-Inspired Nanoparticles in Retinal Diseases Treatment: A Perspective" Cells 9, no. 3: 679. https://doi.org/10.3390/cells9030679
APA StyleDe Matteis, V., & Rizzello, L. (2020). Noble Metals and Soft Bio-Inspired Nanoparticles in Retinal Diseases Treatment: A Perspective. Cells, 9(3), 679. https://doi.org/10.3390/cells9030679





