N-Formylated Peptide Induces Increased Expression of Both Formyl Peptide Receptor 2 (Fpr2) and Toll-Like Receptor 9 (TLR9) in Schwannoma Cells—An In Vitro Model for Early Inflammatory Profiling of Schwann Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture and fMLF Treatment
2.2. Western Blot Analysis
2.3. Statistical Analysis
3. Results
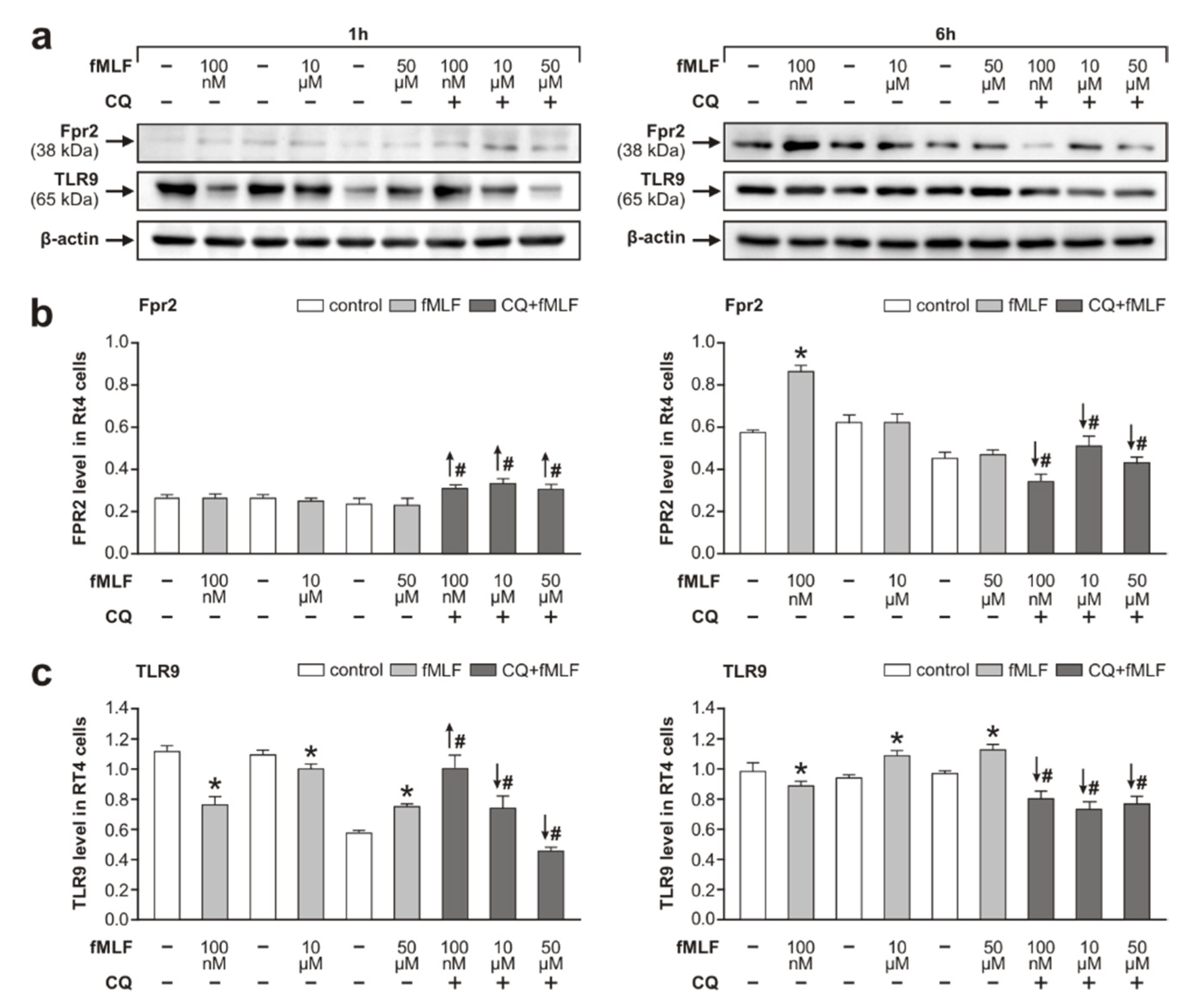
3.1. Fpr2 and TLR9 Protein Levels in RT4 Cells Following fMLF Stimulation
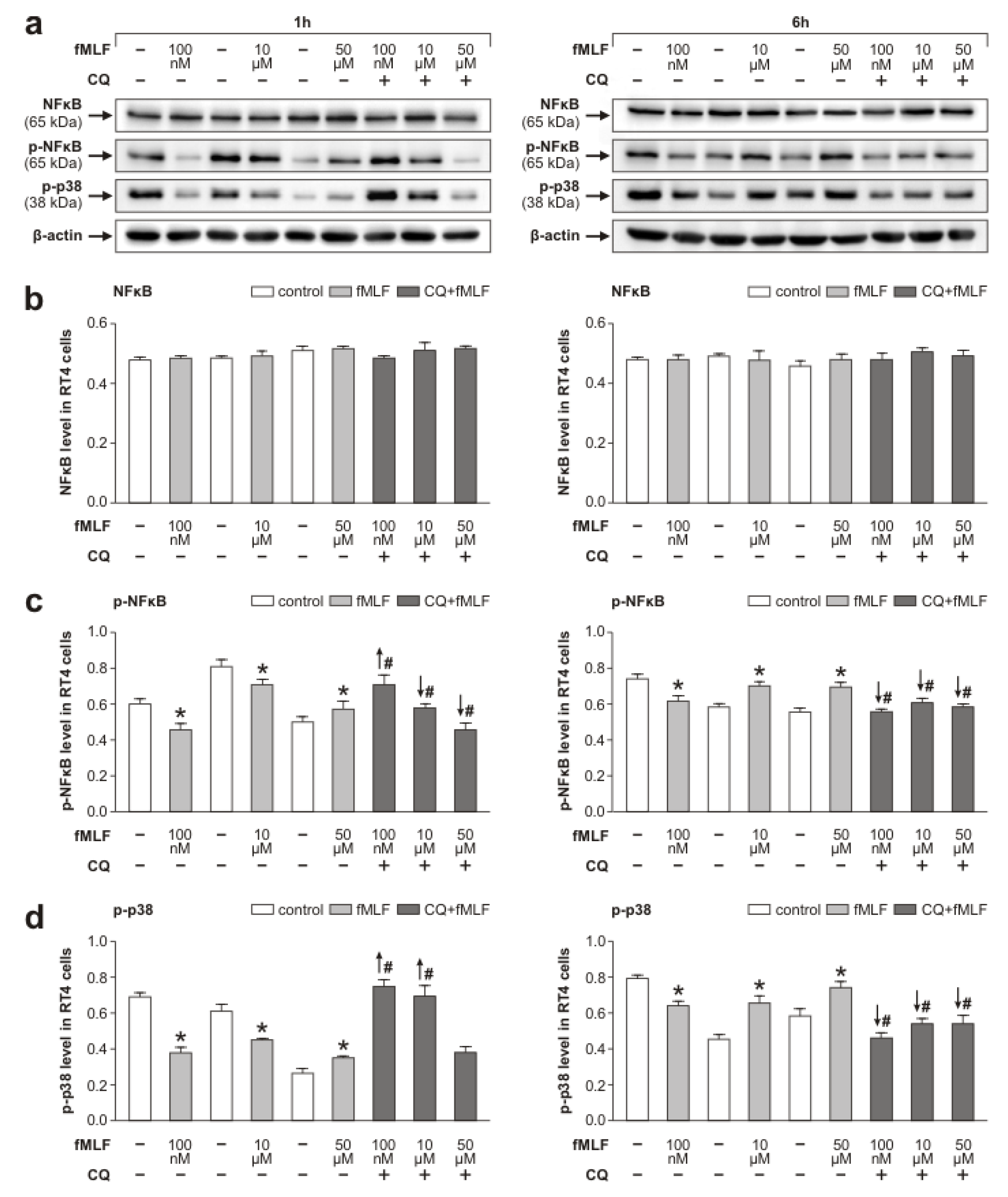
3.2. Fpr2 and TLR9 Molecular Signaling in RT4 Cells Following fMLF Stimulation
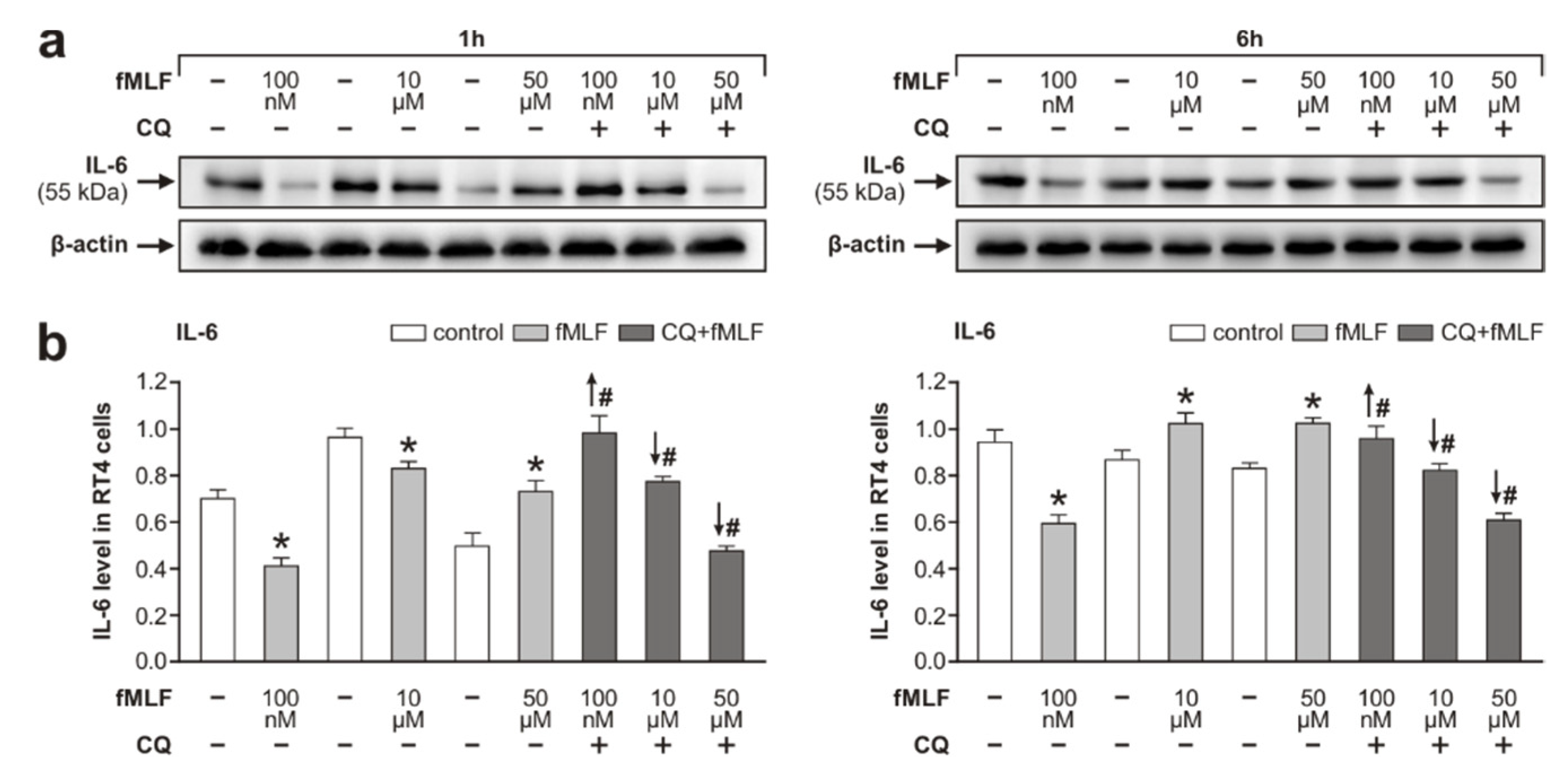
3.3. Inflammatory Profiling of RT4 Cells Following fMLF Stimulation
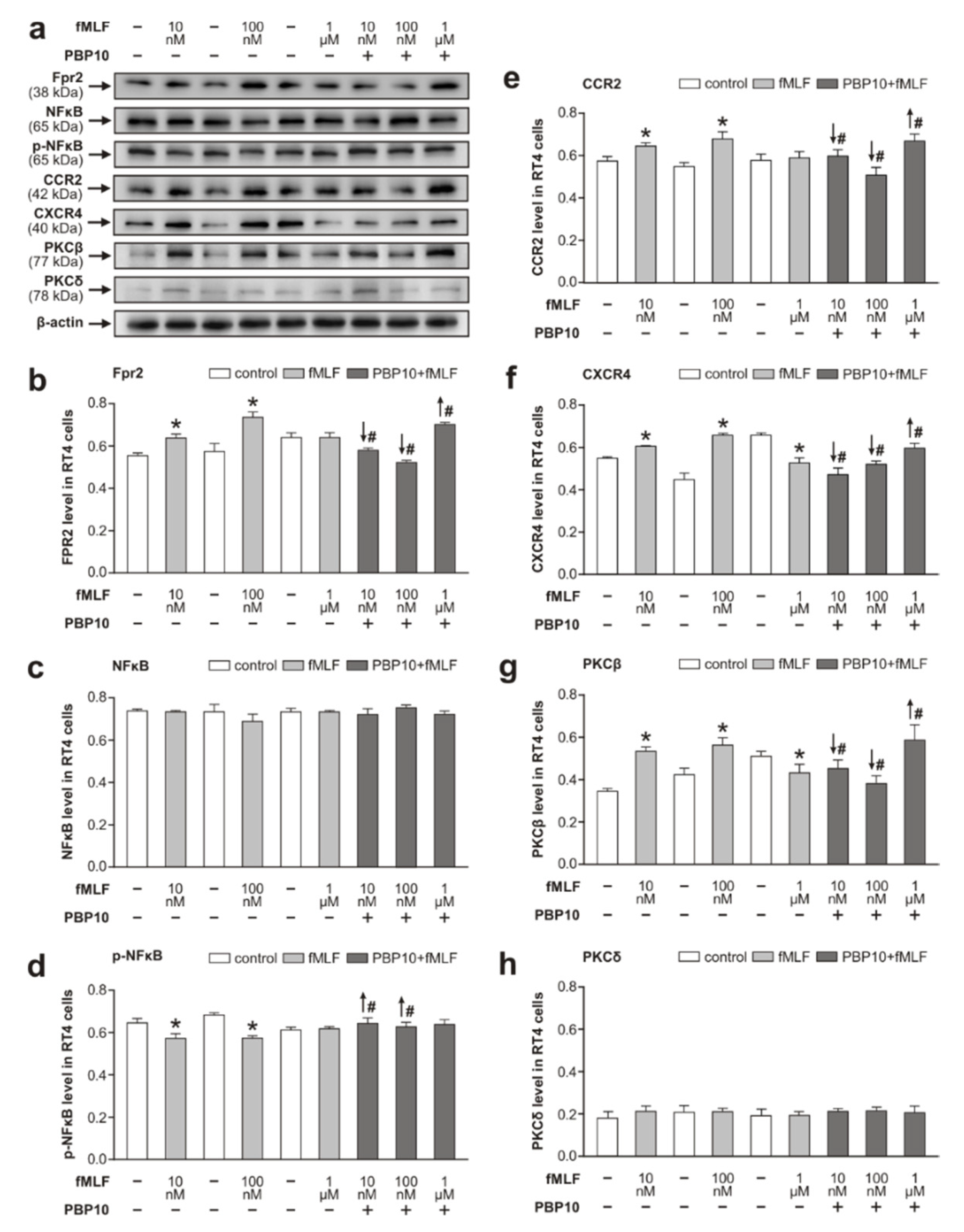
3.4. Effect of fMLF on Levels of Chemokine Receptor CCR2 and CXCR4
4. Discussion
4.1. Effects of fMLF Stimulation on Upregulation of Fpr2 and Modulation of TLR9 in RT4 Cells
- the upregulation of Fpr2 stimulated with 100 nM fMLF coincided with a simultaneous decrease in the activated form of TLR9,
- the level of Fpr2 increased in RT4 cells stimulated with fMLF for 1 h following TLR9 inhibition, and
- in contrast, fMLF stimulation for 6 h following inhibition of the active TLR9 form significantly reduced the level of Fpr2.
4.2. Cooperation of Fpr2 and Chemokine Receptors CCR2 and CXCR4 in RT4 Cells Following fMLF Stimulation
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jessen, K.R.; Mirsky, R. The repair Schwann cell and its function in regenerating nerves. J. Physiol. 2016, 594, 3521–3531. [Google Scholar] [CrossRef] [PubMed]
- Stoll, G.; Jander, S.; Myers, R.R. Degeneration and regeneration of the peripheral nervous system: From Augustus Waller’s observations to neuroinflammation. J. Peripher. Nerv. Syst. JPNS 2002, 7, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Rotshenker, S. Wallerian degeneration: The innate-immune response to traumatic nerve injury. J. Neuroinflamm. 2011, 8, 109. [Google Scholar] [CrossRef]
- Gaudet, A.D.; Popovich, P.G.; Ramer, M.S. Wallerian degeneration: Gaining perspective on inflammatory events after peripheral nerve injury. J. Neuroinflamm. 2011, 8, 110. [Google Scholar] [CrossRef] [PubMed]
- Dubový, P.; Jančálek, R.; Kubek, T. Role of inflammation and cytokines in peripheral nerve regeneration. Int. Rev. Neurobiol. 2013, 108, 173–206. [Google Scholar] [CrossRef] [PubMed]
- Dubový, P.; Klusáková, I.; Hradilová Svíženská, I. Inflammatory profiling of Schwann cells in contact with growing axons distal to nerve injury. BioMed. Res. Int. 2014, 2014, 691041. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Barrientos, S.A.; Martinez, N.W.; Yoo, S.; Jara, J.S.; Zamorano, S.; Hetz, C.; Twiss, J.L.; Alvarez, J.; Court, F.A. Axonal Degeneration Is Mediated by the Mitochondrial Permeability Transition Pore. J. Neurosci. 2011, 31, 966–978. [Google Scholar] [CrossRef]
- Wang, J.T.; Medress, Z.A.; Barres, B.A. Axon degeneration: Molecular mechanisms of a self-destruction pathway. J. Cell Biol. 2012, 196, 7–18. [Google Scholar] [CrossRef]
- Park, J.Y.; Jang, S.Y.; Shin, Y.K.; Koh, H.; Suh, D.J.; Shinji, T.; Araki, T.; Park, H.T. Mitochondrial swelling and microtubule depolymerization are associated with energy depletion in axon degeneration. Neuroscience 2013, 238, 258–269. [Google Scholar] [CrossRef]
- Freeman, M.R. Signaling mechanisms regulating Wallerian degeneration. Curr. Opin. Neurobiol. 2014, 27, 224–231. [Google Scholar] [CrossRef]
- Hirsiger, S.; Simmen, H.-P.; Werner, C.M.L.; Wanner, G.A.; Rittirsch, D. Danger signals activating the immune response after trauma. Mediat. Inflamm. 2012, 2012, 315941. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Raoof, M.; Chen, Y.; Sumi, Y.; Sursal, T.; Junger, W.; Brohi, K.; Itagaki, K.; Hauser, C.J. Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature 2010, 464, 104–107. [Google Scholar] [CrossRef] [PubMed]
- Shamash, S.; Reichert, F.; Rotshenker, S. The cytokine network of Wallerian degeneration: Tumor necrosis factor-alpha, interleukin-1alpha, and interleukin-1beta. J. Neurosci. Off. J. Soc. Neurosci. 2002, 22, 3052–3060. [Google Scholar] [CrossRef]
- Boivin, A.; Pineau, I.; Barrette, B.; Filali, M.; Vallières, N.; Rivest, S.; Lacroix, S. Toll-like receptor signaling is critical for Wallerian degeneration and functional recovery after peripheral nerve injury. J. Neurosci. 2007, 27, 12565–12576. [Google Scholar] [CrossRef] [PubMed]
- Goethals, S.; Ydens, E.; Timmerman, V.; Janssens, S. Toll-like receptor expression in the peripheral nerve. Glia 2010, 58, 1701–1709. [Google Scholar] [CrossRef] [PubMed]
- Latz, E.; Schoenemeyer, A.; Visintin, A.; Fitzgerald, K.A.; Monks, B.G.; Knetter, C.F.; Lien, E.; Nilsen, N.J.; Espevik, T.; Golenbock, D.T. TLR9 signals after translocating from the ER to CpG DNA in the lysosome. Nat. Immunol. 2004, 5, 190–198. [Google Scholar] [CrossRef]
- Raoof, M.; Zhang, Q.; Itagaki, K.; Hauser, C.J. Mitochondrial peptides are potent immune activators that activate human neutrophils via FPR-1. J. Trauma 2010, 68, 1328–1332. [Google Scholar] [CrossRef]
- Cui, Y. Up-Regulation of FPR2, a Chemotactic Receptor for Amyloid β 1–42 (Aβ42), in Murine Microglial Cells by TNFα. Neurobiol. Dis. 2002, 10, 366–377. [Google Scholar] [CrossRef][Green Version]
- Krysko, D.V.; Agostinis, P.; Krysko, O.; Garg, A.D.; Bachert, C.; Lambrecht, B.N.; Vandenabeele, P. Emerging role of damage-associated molecular patterns derived from mitochondria in inflammation. Trends Immunol. 2011, 32, 157–164. [Google Scholar] [CrossRef]
- Weiß, E.; Kretschmer, D. Formyl-Peptide Receptors in Infection, Inflammation, and Cancer. Trends Immunol. 2018, 39, 815–829. [Google Scholar] [CrossRef]
- Ye, R.D.; Boulay, F.; Wang, J.M.; Dahlgren, C.; Gerard, C.; Parmentier, M.; Serhan, C.N.; Murphy, P.M. International Union of Basic and Clinical Pharmacology. LXXIII. Nomenclature for the Formyl Peptide Receptor (FPR) Family. Pharmacol. Rev. 2009, 61, 119–161. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Hu, X.; Qian, L.; Yang, S.; Zhang, W.; Zhang, D.; Wu, X.; Fraser, A.; Wilson, B.; Flood, P.M.; et al. Formyl-methionyl-leucyl-phenylalanine–Induced Dopaminergic Neurotoxicity via Microglial Activation: A Mediator between Peripheral Infection and Neurodegeneration? Environ. Health Perspect. 2008, 116, 593–598. [Google Scholar] [CrossRef] [PubMed]
- He, H.-Q.; Ye, R.D. The Formyl Peptide Receptors: Diversity of Ligands and Mechanism for Recognition. Molecules 2017, 22, 455. [Google Scholar] [CrossRef]
- Porro, C.; Cianciulli, A.; Trotta, T.; Lofrumento, D.D.; Calvello, R.; Panaro, M.A. Formyl-methionyl-leucyl-phenylalanine Induces Apoptosis in Murine Neurons: Evidence for NO-Dependent Caspase-9 Activation. Biology 2019, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Hartt, J.K.; Barish, G.; Murphy, P.M.; Gao, J.L. N-formylpeptides induce two distinct concentration optima for mouse neutrophil chemotaxis by differential interaction with two N-formylpeptide receptor (FPR) subtypes. Molecular characterization of FPR2, a second mouse neutrophil FPR. J. Exp. Med. 1999, 190, 741–747. [Google Scholar] [CrossRef]
- Le, Y.; Oppenheim, J.J.; Wang, J.M. Pleiotropic roles of formyl peptide receptors. Cytokine Growth Factor Rev. 2001, 12, 91–105. [Google Scholar] [CrossRef]
- Cattaneo, F.; Guerra, G.; Ammendola, R. Expression and signaling of formyl-peptide receptors in the brain. Neurochem. Res. 2010, 35, 2018–2026. [Google Scholar] [CrossRef]
- Cattaneo, F.; Parisi, M.; Ammendola, R. Distinct Signaling Cascades Elicited by Different Formyl Peptide Receptor 2 (FPR2) Agonists. Int. J. Mol. Sci. 2013, 14, 7193–7230. [Google Scholar] [CrossRef]
- Hase, A.; Saito, F.; Yamada, H.; Arai, K.; Shimizu, T.; Matsumura, K. Characterization of glial cell line-derived neurotrophic factor family receptor alpha-1 in peripheral nerve Schwann cells. J. Neurochem. 2005, 95, 537–543. [Google Scholar] [CrossRef]
- Lee, H.K.; Seo, I.A.; Suh, D.J.; Hong, J.-I.; Yoo, Y.H.; Park, H.T. Interleukin-6 is required for the early induction of glial fibrillary acidic protein in Schwann cells during Wallerian degeneration. J. Neurochem. 2009, 108, 776–786. [Google Scholar] [CrossRef]
- Lamarca, A.; Gella, A.; Martiañez, T.; Segura, M.; Figueiro-Silva, J.; Grijota-Martinez, C.; Trullas, R.; Casals, N. Uridine 5′-Triphosphate Promotes In Vitro Schwannoma Cell Migration through Matrix Metalloproteinase-2 Activation. PLoS ONE 2014, 9, e98998. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Grovola, M.R.; Xie, H.; Coggins, G.E.; Duggan, P.; Hasan, R.; Huang, J.; Lin, D.W.; Song, C.; Witek, G.M.; et al. Comprehensive pharmacological profiling of neurofibromatosis cell lines. Am. J. Cancer Res. 2017, 7, 923–934. [Google Scholar] [PubMed]
- Rutz, M.; Metzger, J.; Gellert, T.; Luppa, P.; Lipford, G.B.; Wagner, H.; Bauer, S. Toll-like receptor 9 binds single-stranded CpG-DNA in a sequence- and pH-dependent manner. Eur. J. Immunol. 2004, 34, 2541–2550. [Google Scholar] [CrossRef] [PubMed]
- Ewald, S.E.; Lee, B.L.; Lau, L.; Wickliffe, K.E.; Shi, G.-P.; Chapman, H.A.; Barton, G.M. The ectodomain of Toll-like receptor 9 is cleaved to generate a functional receptor. Nature 2008, 456, 658–662. [Google Scholar] [CrossRef]
- Lacagnina, M.J.; Watkins, L.R.; Grace, P.M. Toll-like receptors and their role in persistent pain. Pharmacol. Ther. 2018, 184, 145–158. [Google Scholar] [CrossRef]
- Cussell, P.J.G.; Escalada, M.G.; Milton, N.G.N.; Paterson, A.W.J. The N-formyl peptide receptors: Contemporary roles in neuronal function and dysfunction. Neural Regen. Res. 2020, 15, 1191. [Google Scholar] [CrossRef]
- Xu, X.; Gera, N.; Li, H.; Yun, M.; Zhang, L.; Wang, Y.; Wang, Q.J.; Jin, T. GPCR-mediated PLCβγ/PKCβ/PKD signaling pathway regulates the cofilin phosphatase slingshot 2 in neutrophil chemotaxis. Mol. Biol. Cell 2015, 26, 874–886. [Google Scholar] [CrossRef]
- Rajarathnam, K.; Schnoor, M.; Richardson, R.M.; Rajagopal, S. How do chemokines navigate neutrophils to the target site: Dissecting the structural mechanisms and signaling pathways. Cell. Signal. 2019, 54, 69–80. [Google Scholar] [CrossRef]
- Gölz, G.; Uhlmann, L.; Lüdecke, D.; Markgraf, N.; Nitsch, R.; Hendrix, S. The cytokine/neurotrophin axis in peripheral axon outgrowth. Eur. J. Neurosci. 2006, 24, 2721–2730. [Google Scholar] [CrossRef]
- Klimaschewski, L.; Hausott, B.; Angelov, D.N. The Pros and Cons of Growth Factors and Cytokines in Peripheral Axon Regeneration. In Tissue Engineering of the Peripheral Nerve: Stem Cells and Regeneration Promoting Factors; Geuna, S., Perroteau, I., Tos, P., Battiston, B., Eds.; Elsevier Academic Press Inc.: San Diego, CA, USA, 2013; Volume 108, pp. 137–171. ISBN 978-0-12-410499-0. [Google Scholar]
- West, A.P.; Shadel, G.S. Mitochondrial DNA in innate immune responses and inflammatory pathology. Nat. Rev. Immunol. 2017, 17, 363–375. [Google Scholar] [CrossRef]
- Cui, Y.H.; Le, Y.Y.; Gong, W.H.; Proost, P.; Van Damme, J.; Murphy, W.J.; Wang, J.M. Bacterial lipopolysaccharide selectively up-regulates the function of the chemotactic peptide receptor formyl peptide receptor 2 in murine microglial cells. J. Immunol. 2002, 168, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.; Zhu, J.; Wang, X.; Tang, Y.; Zhou, P.; Hou, M.; Li, S. ANXA1 directs Schwann cells proliferation and migration to accelerate nerve regeneration through the FPR2/AMPK pathway. FASEB J. 2020, 34, 13993–14005. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Bao, Z.; Gong, W.; Tang, P.; Yoshimura, T.; Wang, J.M. Regulation of inflammation by members of the formyl-peptide receptor family. J. Autoimmun. 2017, 85, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Korimová, A.; Klusáková, I.; Hradilová-Svíženská, I.; Kohoutková, M.; Joukal, M.; Dubový, P. Mitochondrial Damage-Associated Molecular Patterns of Injured Axons Induce Outgrowth of Schwann Cell Processes. Front. Cell. Neurosci. 2018, 12, 457. [Google Scholar] [CrossRef]
- Hai, M.; Muja, N.; DeVries, G.H.; Quarles, R.H.; Patel, P.I. Comparative analysis of Schwann cell lines as model systems for myelin gene transcription studies. J. Neurosci. Res. 2002, 69, 497–508. [Google Scholar] [CrossRef]
- Geuna, S.; Raimondo, S.; Fregnan, F.; Haastert-Talini, K.; Grothe, C. In vitro models for peripheral nerve regeneration. Eur. J. Neurosci. 2016, 43, 287–296. [Google Scholar] [CrossRef]
- Filep, J.G.; Sekheri, M.; El Kebir, D. Targeting formyl peptide receptors to facilitate the resolution of inflammation. Eur. J. Pharmacol. 2018, 833, 339–348. [Google Scholar] [CrossRef]
- Kigerl, K.A.; de Rivero Vaccari, J.P.; Dietrich, W.D.; Popovich, P.G.; Keane, R.W. Pattern recognition receptors and central nervous system repair. Exp. Neurol. 2014, 258, 5–16. [Google Scholar] [CrossRef]
- Raabe, C.A.; Gröper, J.; Rescher, U. Biased perspectives on formyl peptide receptors. Biochim. Biophys. Acta BBA Mol. Cell Res. 2019, 1866, 305–316. [Google Scholar] [CrossRef]
- Riley, J.S.; Tait, S.W. Mitochondrial DNA in inflammation and immunity. EMBO Rep. 2020, 21, e49799. [Google Scholar] [CrossRef]
- Iribarren, P.; Chen, K.; Hu, J.; Gong, W.; Cho, E.H.; Lockert, S.; Uranchimeg, B.; Wang, J.M. CpG-containing oligodeoxynucleotide promotes microglial cell uptake of amyloid β 1–42 peptide by up-regulating the expression of the G-protein-coupled receptor mFPR2. FASEB J. 2005, 19, 2032–2034. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Huang, J.; Liu, Y.; Gong, W.; Cui, Y.; Wang, J.M. Synergy of TRIF-dependent TLR3 and MyD88-dependent TLR7 in up-regulating expression of mouse FPR2, a promiscuous G-protein-coupled receptor, in microglial cells. J. Neuroimmunol. 2009, 213, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Hacker, G.; Redecke, V.; Hacker, H. Activation of the immune system by bacterial CpG-DNA. Immunology 2002, 105, 245–251. [Google Scholar] [CrossRef]
- Takeshita, F.; Gursel, I.; Ishii, K.J.; Suzuki, K.; Gursel, M.; Klinman, D.M. Signal transduction pathways mediated by the interaction of CpG DNA with Toll-like receptor 9. Semin. Immunol. 2004, 16, 17–22. [Google Scholar] [CrossRef]
- Pineau, I.; Lacroix, S. Endogenous signals initiating inflammation in the injured nervous system. Glia 2009, 57, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Shih, R.-H.; Wang, C.-Y.; Yang, C.-M. NF-kappaB Signaling Pathways in Neurological Inflammation: A Mini Review. Front. Mol. Neurosci. 2015, 8, 77. [Google Scholar] [CrossRef]
- Dufton, N.; Hannon, R.; Brancaleone, V.; Dalli, J.; Patel, H.B.; Gray, M.; D’Acquisto, F.; Buckingham, J.C.; Perretti, M.; Flower, R.J. Anti-Inflammatory Role of the Murine Formyl-Peptide Receptor 2: Ligand-Specific Effects on Leukocyte Responses and Experimental Inflammation. J. Immunol. 2010, 184, 2611–2619. [Google Scholar] [CrossRef]
- Le, Y.; Murphy, P.M.; Wang, J.M. Formyl-peptide receptors revisited. Trends Immunol. 2002, 23, 541–548. [Google Scholar] [CrossRef]
- Migeotte, I.; Communi, D.; Parmentier, M. Formyl peptide receptors: A promiscuous subfamily of G protein-coupled receptors controlling immune responses. Cytokine Growth Factor Rev. 2006, 17, 501–519. [Google Scholar] [CrossRef]
- Hughes, C.E.; Nibbs, R.J.B. A guide to chemokines and their receptors. FEBS J. 2018, 285, 2944–2971. [Google Scholar] [CrossRef]
- Busillo, J.M.; Armando, S.; Sengupta, R.; Meucci, O.; Bouvier, M.; Benovic, J.L. Site-specific Phosphorylation of CXCR4 Is Dynamically Regulated by Multiple Kinases and Results in Differential Modulation of CXCR4 Signaling. J. Biol. Chem. 2010, 285, 7805–7817. [Google Scholar] [CrossRef] [PubMed]
- Cattaneo, F.; Russo, R.; Castaldo, M.; Chambery, A.; Zollo, C.; Esposito, G.; Pedone, P.V.; Ammendola, R. Phosphoproteomic analysis sheds light on intracellular signaling cascades triggered by Formyl-Peptide Receptor 2. Sci. Rep. 2019, 9, 17894. [Google Scholar] [CrossRef] [PubMed]
- Le, Y.; Li, B.; Gong, W.; Shen, W.; Hu, J.; Dunlop, N.M.; Oppenheim, J.J.; Wang, J.M. Novel pathophysiological role of classical chemotactic peptide receptors and their communications with chemokine receptors. Immunol. Rev. 2000, 177, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Jesaitis, A.J.; Klotz, K.-N. Cytoskeletal regulation of chemotactic receptors: Molecular complexation of N-formyl peptide receptors with G proteins and actin. Eur. J. Haematol. 2009, 51, 288–293. [Google Scholar] [CrossRef] [PubMed]
| fMLF-Treatment | 10 nM | 100 nM | 1 μM | 10 μM | 50 μM | |
|---|---|---|---|---|---|---|
| Fpr2 | 1 h | − | + | − | + | + |
| 6 h | + | + | + | + | + | |
| 1 μM CQ + 1 h | − | + | − | + | + | |
| 1 μM CQ + 6 h | − | + | − | + | + | |
| 1 μM PBP10 + 6 h | + | + | + | − | − | |
| TLR9 | 1 h | − | + | − | + | + |
| 6 h | − | + | − | + | + | |
| 1 μM CQ + 1 h | − | + | − | + | + | |
| 1 μM CQ + 6 h | − | + | − | + | + |
| Primary Antibody | Type of Antibody | Dilution | Cat. No/Producer |
|---|---|---|---|
| Fpr2 | polyclonal rabbit | 1:500 | NLS1878/Novus |
| TLR9 | monoclonal mouse | 1:1000 | NBP2-24729/Novus |
| CCR2 | polyclonal rabbit | 1:2000 | NBP1-48337/Novus |
| CXCR4 | polyclonal rabbit | 1:2000 | LS-C417098/LifeSpan |
| phospho-p38 | monoclonal mouse | 1:500 | 4511/Cell Signaling |
| NFκB | monoclonal rabbit | 1:1000 | 8242/Cell Signaling |
| phospho-NFκB | monoclonal rabbit | 1:1000 | 3033/Cell Signaling |
| IL-6 | polyclonal rabbit | 1:1000 | ARC0062/Invitrogen |
| PKCβ | polyclonal rabbit | 1:2000 | NBP2-19846/Novus |
| PKCδ | monoclonal mouse | 1:500 | Sc-8402/Santa Cruz |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Korimová, A.; Dubový, P. N-Formylated Peptide Induces Increased Expression of Both Formyl Peptide Receptor 2 (Fpr2) and Toll-Like Receptor 9 (TLR9) in Schwannoma Cells—An In Vitro Model for Early Inflammatory Profiling of Schwann Cells. Cells 2020, 9, 2661. https://doi.org/10.3390/cells9122661
Korimová A, Dubový P. N-Formylated Peptide Induces Increased Expression of Both Formyl Peptide Receptor 2 (Fpr2) and Toll-Like Receptor 9 (TLR9) in Schwannoma Cells—An In Vitro Model for Early Inflammatory Profiling of Schwann Cells. Cells. 2020; 9(12):2661. https://doi.org/10.3390/cells9122661
Chicago/Turabian StyleKorimová, Andrea, and Petr Dubový. 2020. "N-Formylated Peptide Induces Increased Expression of Both Formyl Peptide Receptor 2 (Fpr2) and Toll-Like Receptor 9 (TLR9) in Schwannoma Cells—An In Vitro Model for Early Inflammatory Profiling of Schwann Cells" Cells 9, no. 12: 2661. https://doi.org/10.3390/cells9122661
APA StyleKorimová, A., & Dubový, P. (2020). N-Formylated Peptide Induces Increased Expression of Both Formyl Peptide Receptor 2 (Fpr2) and Toll-Like Receptor 9 (TLR9) in Schwannoma Cells—An In Vitro Model for Early Inflammatory Profiling of Schwann Cells. Cells, 9(12), 2661. https://doi.org/10.3390/cells9122661





