Enhancing Salt Tolerance of Plants: From Metabolic Reprogramming to Exogenous Chemical Treatments and Molecular Approaches
Abstract
1. Introduction
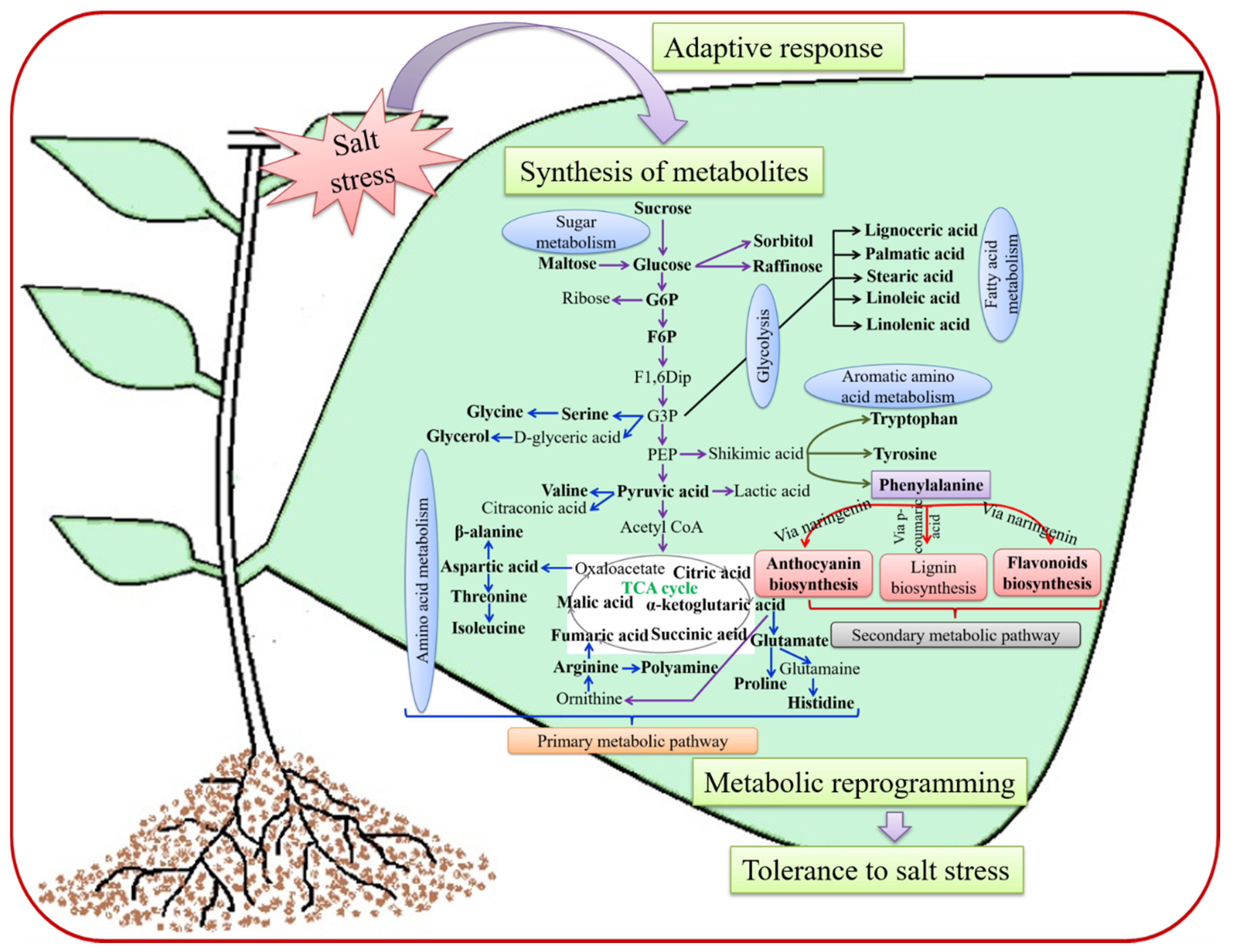
2. Metabolic Reprogramming in Plants Responding to SS
2.1. Primary Metabolites and Their Response to SS
2.1.1. Carbohydrates: Compatible Solute Accumulation under SS
2.1.2. Amino Acid Production and Their Involvement in Plants under SS
2.2. Secondary Metabolites and Response to SS
2.2.1. Alkaloids: As Stimulants under SS
2.2.2. Terpene Production and Response to SS
2.2.3. Phenolics: Potential Antioxidants under SS
2.2.4. Dimethylsulfonium Compounds: An Important Osmoprotectant
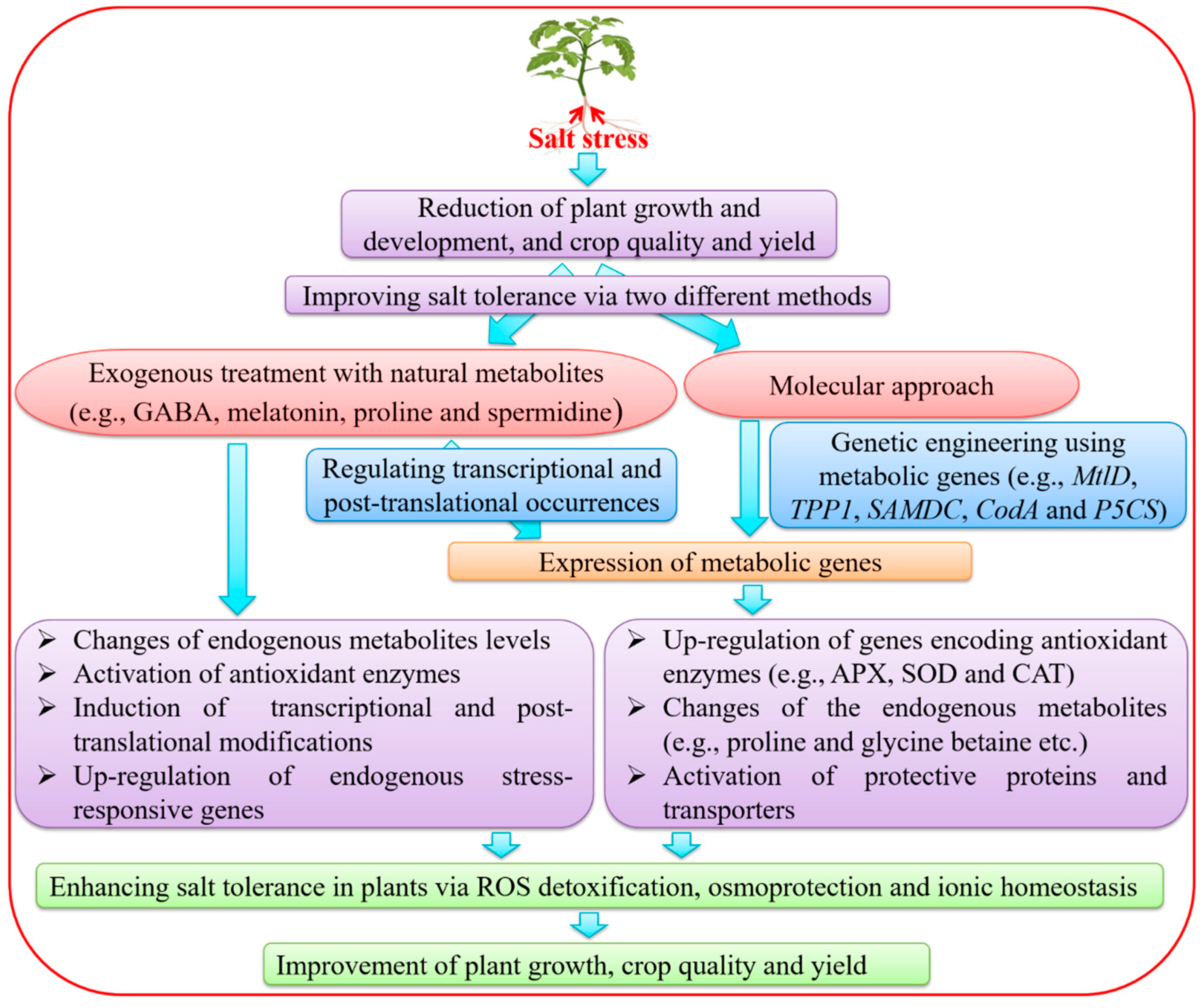
3. Improvement of SS Tolerance by Exogenous Treatments with Metabolites
4. Genetic Engineering of Metabolic Genes for the Improvement of Salt Tolerance
5. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ASs | Abiotic stresses |
| AAs | Amino acids |
| ALA | 5-aminolevulinic acid |
| APX | Ascorbate peroxidase |
| CAT | Catalase |
| DMSP | Dimethylsulfoniopropionate |
| DMSO | Dimethylsulfoxide |
| ESs | Environmental stresses |
| GB | Glycine betaine |
| GABA | γ-amino butyric acid |
| MDA | Malondialdehyde |
| PMs | Primary metabolites |
| PA | Polyamines |
| Put | Putrescine |
| ROS | Reactive oxygen species |
| SMs | Secondary metabolites |
| SS | Salt stress |
| SOD | Superoxide dismutase |
| Spd | Spermidine |
| Spm | Spermine |
References
- Kumari, A.; Das, P.; Parida, A.K.; Agarwal, P.K. Proteomics, metabolomics, and ionomics perspectives of salinity tolerance in halophytes. Front. Plant Sci. 2015, 6, 537. [Google Scholar] [CrossRef]
- Esfahani, M.N.; Kusano, M.; Nguyen, K.H.; Watanabe, Y.; Van Ha, C.; Saito, K.; Sulieman, S.; Herrera-Estrella, L.; Tran, L.S.P. Adaptation of the symbiotic Mesorhizobium–chickpea relationship to phosphate deficiency relies on reprogramming of whole-plant metabolism. Proc. Natl. Acad. Sci. USA 2016, 113, 4610–4619. [Google Scholar] [CrossRef]
- Anwar, A.; Kim, J.K. Transgenic breeding approaches for improving abiotic stress tolerance: Recent progress and future perspectives. Int. J. Mol. Sci. 2020, 21, 2695. [Google Scholar] [CrossRef]
- Kumar, K.; Mosa, K.A. Ion transporters: A decisive component of salt stress tolerance in plants. In Managing Salt Tolerance in Plants: Molecular and Genomic Perspectives; Wani, S.H., Hossai, M.A., Eds.; CRC Press: Boca Raton, FL, USA, 2015; pp. 373–390. [Google Scholar]
- Valifard, M.; Mohsenzadeh, S.; Kholdebarin, B. Salinity effects on phenolic content and antioxidant activity of Salvia macrosiphon. Iran. J. Sci. Technol. A 2017, 41, 295–300. [Google Scholar] [CrossRef]
- Lenntech. Salinity Hazard. 2018. Available online: https://www.lenntech.com/applications/irrigation/salinity/salinity-hazard-irrigation.htm#ixzz5Q8KINkqb (accessed on 16 June 2019).
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, P.; Kumar, R. Soil salinity: A serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi J. Biol. Sci. 2015, 22, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Patel, M.K.; Mishra, A.; Jha, B. Physio-biochemical composition and untargeted metabolomics of cumin (Cuminum cyminum L.) make it promising functional food and help in mitigating salinity stress. PLoS ONE 2015, 10, e0144469. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Zhang, H.; Song, C.; Zhu, J.K.; Shabala, S. Mechanisms of plant responses and adaptation to soil salinity. Innovation 2020, 1, 100017. [Google Scholar] [CrossRef]
- Shulaev, V.; Cortes, D.; Miller, G.; Mittler, R. Metabolomics for plant stress response. Physiol. Plant. 2008, 132, 199–208. [Google Scholar] [CrossRef]
- Zhu, J.K. Abiotic stress signaling and responses in plants. Cell 2016, 167, 313–324. [Google Scholar] [CrossRef]
- Ezquer, I.; Salameh, I.; Colombo, L.; Kalaitzis, P. Plant cell walls tackling climate change: Biotechnological strategies to improve crop adaptations and photosynthesis in response to global warming. Plants 2020, 9, 212. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.A.; Iqbal, M.; Rasheed, R.; Hussain, I.; Riaz, M.; Arif, M.S. Environmental stress and secondary metabolites in plants. In Plant Metabolites and Regulation Under Environmental Stress; Ahmad, P., Ahanger, M.A., Singh, V.P., Tripathi, D.K., Alam, P., Alyemeni, M.N., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 153–167. [Google Scholar]
- Savvides, A.; Ali, S.; Tester, M.; Fotopoulos, V. Chemical priming of plants against multiple abiotic stresses: Mission possible? Trends Plant Sci. 2016, 21, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.C.; Lin, K.H.; Ho, S.L.; Chiang, C.M.; Yang, C.M. Enhancing the abiotic stress tolerance of plants: From chemical treatment to biotechnological approaches. Physiol. Plant. 2018, 164, 452–466. [Google Scholar] [CrossRef] [PubMed]
- Reguera, M.; Peleg, Z.; Blumwald, E. Targeting metabolic pathways for genetic engineering abiotic stress-tolerance in crops. BBA-Gene Regul. Mech. 2012, 1819, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Ishaku, G.A.; Tizhe, D.T.; Bamanga, R.A.; Afolabi, E.T. Biotechnology and drought stress tolerance in plants. Asian J. Plant Sci. 2020, 34–46. [Google Scholar] [CrossRef]
- Kusano, M.; Fukushima, A.; Redestig, H.; Saito, K. Metabolomic approaches toward understanding nitrogen metabolism in plants. J. Exp. Bot. 2011, 62, 1439–1453. [Google Scholar] [CrossRef]
- Seki, M.; Kamei, A.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Molecular responses to drought, salinity and frost: Common and different paths for plant protection. Curr. Opin. Biotechnol. 2003, 14, 194–199. [Google Scholar] [CrossRef]
- Krasensky, J.; Jonak, C. Drought, salt, and temperature stress-induced metabolic rearrangements and regulatory networks. J. Exp. Bot. 2012, 63, 1593–1608. [Google Scholar] [CrossRef]
- Singh, A.K.; Dhanapal, S.; Yadav, B.S. The dynamic responses of plant physiology and metabolism during environmental stress progression. Mol. Biol. Rep. 2020, 47, 1459–1470. [Google Scholar] [CrossRef]
- Arbona, V.; Iglesias, D.J.; Talon, M.; Gomez-Cadenas, A. Plant phenotype demarcation using nontargeted LC–MS and GC–MS metabolite profiling. J. Agric. Food Chem. 2009, 57, 7338–7347. [Google Scholar] [CrossRef]
- Roessner, U.; Luedemann, A.; Brust, D.; Fiehn, O.; Linke, T.; Willmitzer, L.; Fernie, A.R. Metabolic profiling allows comprehensive phenotyping of genetically or environmentally modified plant systems. Plant Cell 2001, 13, 11–29. [Google Scholar] [CrossRef] [PubMed]
- Llanes, A.; Andrade, A.; Alemano, S.; Luna, V. Metabolomic approach to understand plant adaptations to water and salt stress. In Plant Metabolites and Regulation Under Environmental Stress; Ahmad, P., Ahanger, M.A., Singh, V.P., Tripathi, D.K., Alam, P., Alyemeni, M.N., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 133–144. [Google Scholar]
- Roberts, L.D.; Souza, A.L.; Gerszten, R.E.; Clish, C.B. Targeted metabolomics. Curr. Protoc. Mol. Biol. 2012, 98, 30. [Google Scholar] [CrossRef] [PubMed]
- Van Meulebroek, L.; Hanssens, J.; Steppe, K.; Vanhaecke, L. Metabolic fingerprinting to assess the impact of salinity on carotenoid content in developing tomato fruits. Int. J. Mol. Sci. 2016, 17, 821. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.; Patel, M.K.; Jha, B. Non–targeted metabolomics and scavenging activity of reactive oxygen species reveal the potential of Salicornia brachiata as a functional food. J. Funct. Foods 2015, 13, 21–31. [Google Scholar] [CrossRef]
- Cusido, R.M.; Onrubia, M.; Sabater-Jara, A.B.; Moyano, E.; Bonfill, M.; Goossens, A.; Palazon, J. A rational approach to improving the biotechnological production of taxanes in plant cell cultures of Taxus spp. Biotechnol. Adv. 2014, 32, 1157–1167. [Google Scholar] [CrossRef]
- Pal, S.; Zhao, J.; Khan, A.; Yadav, N.S.; Batushansky, A.; Barak, S.; Rewald, B.; Fait, A.; Lazarovitch, N.; Rachmilevitch, S. Paclobutrazol induces tolerance in tomato to deficit irrigation through diversified effects on plant morphology, physiology and metabolism. Sci. Rep. 2016, 6, 39321. [Google Scholar] [CrossRef]
- Zagorchev, L.; Seal, C.E.; Kranner, I.; Odjakova, M. A central role for thiols in plant tolerance to abiotic stress. Int. J. Mol. Sci. 2013, 14, 7405–7432. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Kumar, R.; Bohra, A.; Pandey, A.K.; Pandey, M.K.; Kumar, A. Metabolomics for plant improvement: Status and prospects. Front. Plant Sci. 2017, 8, 1302. [Google Scholar] [CrossRef]
- Gupta, B.; Huang, B. Mechanism of salinity tolerance in plants: Physiological, biochemical, and molecular characterization. Int. J. Genomics 2014, 2014, 701596. [Google Scholar] [CrossRef]
- Ahanger, M.A.; Gul, F.; Ahmad, P.; Akram, N.A. Environmental stresses and metabolomics-deciphering the role of stress responsive metabolites. In Plant Metabolites and Regulation Under Environmental Stress; Ahmad, P., Ahanger, M.A., Singh, V.P., Tripathi, D.K., Alam, P., Alyemeni, M.N., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 53–67. [Google Scholar]
- Slama, I.; Abdelly, C.; Bouchereau, A.; Flowers, T.; Savoure, A. Diversity, distribution and roles of osmoprotective compounds accumulated in halophytes under abiotic stress. Ann. Bot. 2015, 115, 433–447. [Google Scholar] [CrossRef] [PubMed]
- Patton, A.J.; Cunningham, S.M.; Volenec, J.J.; Reicher, Z.J. Differences in freeze tolerance of zoysiagrasses: II. Carbohydrate and proline accumulation. Crop Sci. 2007, 47, 2170–2181. [Google Scholar] [CrossRef]
- Yang, L.; Zhao, X.; Zhu, H.; Paul, M.; Zu, Y.; Tang, Z. Exogenous trehalose largely alleviates ionic unbalance, ROS burst, and PCD occurrence induced by high salinity in Arabidopsis seedlings. Front. Plant Sci. 2014, 5, 570. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa, A.; Yabuta, Y.; Shigeoka, S. Galactinol and raffinose constitute a novel function to protect plants from oxidative damage. Plant Physiol. 2008, 147, 1251–1263. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa-Yokoi, A.; Yabuta, Y.; Shigeoka, S. The contribution of carbohydrates including raffinose family oligosaccharides and sugar alcohols to protection of plant cells from oxidative damage. Plant Signal Behav. 2008, 3, 1016–1018. [Google Scholar] [CrossRef] [PubMed]
- Rosa, M.; Prado, C.; Podazza, G.; Interdonato, R.; González, J.A.; Hilal, M.; Prado, F.E. Soluble sugars: Metabolism, sensing and abiotic stress: A complex network in the life of plants. Plant Signal Behav. 2009, 4, 388–393. [Google Scholar] [CrossRef]
- Coello, P.; Hey, S.J.; Halford, N.G. The sucrose non-fermenting-1-related (SnRK) family of protein kinases: Potential for manipulation to improve stress tolerance and increase yield. J. Exp. Bot. 2010, 62, 883–893. [Google Scholar] [CrossRef]
- Purcell, P.C.; Smith, A.M.; Halford, N.G. Antisense expression of a sucrose non-fermenting-1-related protein kinase sequence in potato results in decreased expression of sucrose synthase in tubers and loss of sucrose-inducibility of sucrose synthase transcripts in leaves. Plant J. 1998, 14, 195–202. [Google Scholar] [CrossRef]
- Wu, D.; Cai, S.; Chen, M.; Ye, L.; Chen, Z.; Zhang, H.; Dai, F.; Wu, F.; Zhang, G. Tissue metabolic responses to salt stress in wild and cultivated barley. PLoS ONE 2013, 8, e55431. [Google Scholar] [CrossRef]
- Dias, D.A.; Hill, C.B.; Jayasinghe, N.S.; Atieno, J.; Sutton, T.; Roessner, U. Quantitative profiling of polar primary metabolites of two chickpea cultivars with contrasting responses to salinity. J. Chromatogr. B 2015, 1000, 1–13. [Google Scholar] [CrossRef]
- Bendaly, A.; Messedi, D.; Smaoui, A.; Ksouri, R.; Bouchereau, A.; Abdelly, C. Physiological and leaf metabolome changes in the xerohalophyte species Atriplex halimus induced by salinity. Plant Physiol. Biochem. 2016, 103, 208–218. [Google Scholar] [CrossRef] [PubMed]
- Jorge, T.F.; Duro, N.; Da Costa, M.; Florian, A.; Ramalho, J.C.; Ribeiro-Barros, A.I.; Fernie, A.R.; António, C. GC-TOF-MS analysis reveals salt stress-responsive primary metabolites in Casuarina glauca tissues. Metabolomics 2017, 13, 95. [Google Scholar] [CrossRef]
- Hossain, M.S.; Persicke, M.; ElSayed, A.I.; Kalinowski, J.; Dietz, K.J. Metabolite profiling at the cellular and subcellular level reveals metabolites associated with salinity tolerance in sugar beet. J. Exp. Bot. 2017, 68, 5961–5976. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Lutz, A.; Hill, C.B.; Callahan, D.L.; Roessner, U. A quantitative profiling method of phytohormones and other metabolites applied to barley roots subjected to salinity stress. Front. Plant Sci. 2017, 7. [Google Scholar] [CrossRef]
- Rai, V.K. Role of amino acids in plant responses to stresses. Biol. Plant. 2002, 45, 481–487. [Google Scholar] [CrossRef]
- Lu, Y.; Lam, H.; Pi, E.; Zhan, Q.; Tsai, S.; Wang, C.; Kwan, Y.; Ngai, S. Comparative metabolomics in Glycine max and Glycine soja under salt stress to reveal the phenotypes of their offspring. J. Agric. Food Chem. 2013, 61, 8711–8721. [Google Scholar] [CrossRef]
- Sobhanian, H.; Motamed, N.; Jazii, F.R.; Nakamura, T.; Komatsu, S. Salt stress induced differential proteome and metabolome response in the shoots of Aeluropus lagopoides (Poaceae), a halophyte C4 plant. J. Proteome Res. 2010, 9, 2882–2897. [Google Scholar] [CrossRef]
- Pang, Q.; Zhang, A.; Zang, W.; Wei, L.; Yan, X. Integrated proteomics and metabolomics for dissecting the mechanism of global responses to salt and alkali stress in Suaeda corniculata. Plant Soil 2016, 402, 379–394. [Google Scholar] [CrossRef]
- Xie, Z.; Wang, C.; Zhu, S.; Wang, W.; Xu, J.; Zhao, X. Characterizing the metabolites related to rice salt tolerance with introgression lines exhibiting contrasting performances in response to saline conditions. Plant Growth Regul. 2020, 92, 157–167. [Google Scholar] [CrossRef]
- Liu, B.; Peng, X.; Han, L.; Hou, L.; Li, B. Effects of exogenous spermidine on root metabolism of cucumber seedlings under salt stress by GC-MS. Agronomy 2020, 10, 459. [Google Scholar] [CrossRef]
- Chen, Z.; Cuin, T.A.; Zhou, M.; Twomey, A.; Naidu, B.P.; Shabala, S. Compatible solute accumulation and stress-mitigating effects in barley genotypes contrasting in their salt tolerance. J. Exp. Bot. 2007, 58, 4245–4255. [Google Scholar] [CrossRef] [PubMed]
- Gavaghan, C.L.; Li, J.V.; Hadfield, S.T.; Hole, S.; Nicholson, J.K.; Wilson, I.D.; Howe, P.W.; Stanley, P.D.; Holmes, E. Application of NMR-based metabolomics to the investigation of salt stress in maize (Zea mays). Phytochem. Anal. 2011, 22, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.B.; Jha, D.; Bacic, A.; Tester, M.; Roessner, U. Characterization of ion contents and metabolic responses to salt stress of different Arabidopsis AtHKT1; 1 genotypes and their parental strains. Mol. Plant. 2013, 6, 350–368. [Google Scholar] [CrossRef] [PubMed]
- Gong, Q.; Li, P.; Ma, S.; Indu Rupassara, S.; Bohnert, H.J. Salinity stress adaptation competence in the extremophile Thellungiella halophila in comparison with its relative Arabidopsis thaliana. Plant J. 2005, 44, 826–839. [Google Scholar] [CrossRef] [PubMed]
- Gagneul, D.; Aïnouche, A.; Duhazé, C.; Lugan, R.; Larher, F.R.; Bouchereau, A. A reassessment of the function of the so-called compatible solutes in the halophytic Plumbaginaceae Limonium latifolium. Plant Physiol. 2007, 144, 1598–1611. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Du, Y.; Chen, S.; Tang, H. Dynamic metabonomic responses of tobacco (Nicotiana tabacum) plants to salt stress. J. Proteome Res. 2011, 10, 1904–1914. [Google Scholar] [CrossRef]
- Martinez, V.; Mestre, T.C.; Rubio, F.; Girones-Vilaplana, A.; Moreno, D.A.; Mittler, R.; Rivero, R.M. Accumulation of flavonols over hydroxycinnamic acids favors oxidative damage protection under abiotic stress. Front. Plant Sci. 2016, 7, 838. [Google Scholar] [CrossRef]
- Kováčik, J.; Klejdus, B.; Hedbavny, J.; Bačkor, M. Salicylic acid alleviates NaCl-induced changes in the metabolism of Matricaria chamomilla plants. Ecotoxicology 2009, 18, 544–554. [Google Scholar] [CrossRef]
- Lim, J.H.; Park, K.J.; Kim, B.K.; Jeong, J.W.; Kim, H.J. Effect of salinity stress on phenolic compounds and carotenoids in buckwheat (Fagopyrum esculentum M.) sprout. Food Chem. 2012, 135, 1065–1070. [Google Scholar] [CrossRef]
- Sanchez, D.H.; Pieckenstain, F.L.; Escaray, F.; Erban, A.; Kraemer, U.T.E.; Udvardi, M.K.; Kopka, J. Comparative ionomics and metabolomics in extremophile and glycophytic Lotus species under salt stress challenge the metabolic pre–adaptation hypothesis. Plant Cell Environ. 2011, 34, 605–617. [Google Scholar] [CrossRef]
- Ni, J.; Yang, X.; Zhu, J.; Liu, Z.; Ni, Y.; Wu, H.; Zhang, H.; Liu, T. Salinity-induced metabolic profile changes in Nitraria tangutorum Bobr. suspension cells. Plant Cell Tiss. Org. Cult. 2015, 122, 239–248. [Google Scholar] [CrossRef]
- Petridis, A.; Therios, I.; Samouris, G.; Tananaki, C. Salinity-induced changes in phenolic compounds in leaves and roots of four olive cultivars (Olea europaea L.) and their relationship to antioxidant activity. Environ. Exp. Bot. 2012, 79, 37–43. [Google Scholar] [CrossRef]
- Tounekti, T.; Vadel, A.M.; Ennajeh, M.; Khemira, H.; Munné-Bosch, S. Ionic interactions and salinity affect monoterpene and phenolic diterpene composition in rosemary (Rosmarinus officinalis). J. Soil Sci. Plant Nutr. 2011, 174, 504–514. [Google Scholar] [CrossRef]
- Valifard, M.; Mohsenzadeh, S.; Kholdebarin, B.; Rowshan, V.; Niazi, A.; Moghadam, A. Effect of salt stress on terpenoid biosynthesis in Salvia mirzayanii: From gene to metabolite. J. Hortic. Sci. Biotechnol. 2019, 94, 389–399. [Google Scholar] [CrossRef]
- Behr, J.H.; Bouchereau, A.; Berardocco, S.; Seal, C.E.; Flowers, T.J.; Zörb, C. Metabolic and physiological adjustment of Suaeda maritima to combined salinity and hypoxia. Ann. Bot. 2017, 119, 965–976. [Google Scholar]
- Akula, R.; Ravishankar, G.A. Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal Behav. 2011, 6, 1720–1731. [Google Scholar] [CrossRef] [PubMed]
- Verma, N.; Shukla, S. Impact of various factors responsible for fluctuation in plant secondary metabolites. J. Appl. Res. Med. Aromat. Plants 2015, 2, 105–113. [Google Scholar] [CrossRef]
- Kusano, M.; Yang, Z.; Okazaki, Y.; Nakabayashi, R.; Fukushima, A.; Saito, K. Using metabolomic approaches to explore chemical diversity in rice. Mol. Plant. 2015, 8, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Yang, C.; Wei, Y.; Ma, Q.; Yang, L.; Chen, X. Genomics grand for diversified plant secondary metabolites. Plant Divers. Resour. 2011, 33, 53–64. [Google Scholar]
- Wink, M. Modes of action of herbal medicines and plant secondary metabolites. Medicines 2015, 2, 251–286. [Google Scholar] [CrossRef]
- Ziegler, J.; Facchini, P.J. Alkaloid biosynthesis: Metabolism and trafficking. Annu. Rev. Plant Biol. 2008, 59, 735–769. [Google Scholar] [CrossRef] [PubMed]
- Sytar, O.; Mbarki, S.; Zivcak, M.; Brestic, M. The involvement of different secondary metabolites in salinity tolerance of crops. In Salinity Responses and Tolerance in Plants; Kumar, V., Wani, S.H., Suprasanna, P., Tran, L.S.P., Eds.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 21–48. [Google Scholar]
- Sachan, N.; Rogers, D.T.; Yun, K.Y.; Littleton, J.M.; Falcone, D.L. Reactive oxygen species regulate alkaloid metabolism in undifferentiated N. tabacum cells. Plant Cell Rep. 2010, 29, 437. [Google Scholar] [CrossRef] [PubMed]
- Anitha, S.; Kumari, B.R. Reserpine accumulation in NaCl treated calli of Rauvolfia tetraphylla L. Sci. Asia 2006, 32, 417–419. [Google Scholar] [CrossRef]
- Jaleel, C.A.; Manivannan, P.; Sankar, B.; Kishorekumar, A.; Panneerselvam, R. Calcium chloride effects on salinity-induced oxidative stress, proline metabolism and indole alkaloid accumulation in Catharanthus roseus. C. R. Biol. 2007, 330, 674–683. [Google Scholar] [CrossRef]
- Osman, M.E.; Elfeky, S.S.; El Soud, K.A.; Hasan, A.M. Response of Catharanthus roseus shoots to salinity and drought in relation to vincristine alkaloid content. Asian J. Plant Sci. 2007, 6, 1223–1228. [Google Scholar] [CrossRef][Green Version]
- Benjamin, J.J.; Lucini, L.; Jothiramshekar, S.; Parida, A. Metabolomic insights into the mechanisms underlying tolerance to salinity in different halophytes. Plant Physiol. Biochem. 2019, 135, 528–545. [Google Scholar] [CrossRef]
- Mosadegh, H.; Trivellini, A.; Ferrante, A.; Lucchesini, M.; Vernieri, P.; Mensuali, A. Applications of UV-B lighting to enhance phenolic accumulation of sweet basil. Sci. Hortic. 2018, 229, 107–116. [Google Scholar] [CrossRef]
- Han-Chen, Z.; Ferdinand, S.L.; Xing-Kai, Y.; Yan, Z.; Shu, W. Analysis of terpene synthase family genes in Camellia sinensis with an emphasis on abiotic stress conditions. Sci. Rep. 2020, 10, 1–13. [Google Scholar]
- Kopaczyk, J.M.; Warguła, J.; Jelonek, T. The variability of terpenes in conifers under developmental and environmental stimuli. Environ. Exp. Bot. 2020, 25, 104197. [Google Scholar] [CrossRef]
- Dahham, S.S.; Tabana, Y.M.; Iqbal, M.A.; Ahamed, M.B.; Ezzat, M.O.; Majid, A.S.; Majid, A.M. The anticancer, antioxidant and antimicrobial properties of the sesquiterpene β-caryophyllene from the essential oil of Aquilaria crassna. Molecules 2015, 20, 11808–11829. [Google Scholar] [CrossRef]
- Porres-MartÃnez, M.; Carretero, M.E.; Gómez-Serranillos, M.P. In vitro neuroprotective potential of the monoterpenes α-pinene and 1, 8-cineole against H2O2-induced oxidative stress in PC12 cells. Zeitschrift für Naturforschung C 2016, 71, 191–199. [Google Scholar]
- Karray-Bouraoui, N.; Rabhi, M.; Neffati, M.; Baldan, B.; Ranieri, A.; Marzouk, B.; Lachaâl, M.; Smaoui, A. Salt effect on yield and composition of shoot essential oil and trichome morphology and density on leaves of Mentha pulegium. Ind. Crop. Prod. 2009, 30, 338–343. [Google Scholar] [CrossRef]
- Cheynier, V.; Comte, G.; Davies, K.M.; Lattanzio, V.; Martens, S. Plant phenolics: Recent advances on their biosynthesis, genetics, and ecophysiology. Plant Physiol. Biochem. 2013, 72, 1–20. [Google Scholar] [CrossRef]
- Mierziak, J.; Kostyn, K.; Kulma, A. Flavonoids as important molecules of plant interactions with the environment. Molecules 2014, 19, 16240–16265. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Gould, K.S.; Lister, C. Flavonoid functions in plants. In Flavonoids. Chemistry, Biochemistry, and Applications; Andersen, Ø.M., Markham, K.R., Eds.; CRC Press: Boca Raton, FL, USA, 2006; pp. 397–441. [Google Scholar]
- Minh, L.T.; Khang, D.T.; Ha, P.T.T.; Tuyen, P.T.; Minh, T.N.; Quan, N.V.; Xuan, T.D. Effects of salinity stress on growth and phenolics of rice (Oryza sativa L.). Int. Lett. Nat. Sci. 2016, 57, 1–10. [Google Scholar] [CrossRef]
- Tattini, M.; Galardi, C.; Pinelli, P.; Massai, R.; Remorini, D.; Agati, G. Differential accumulation of flavonoids and hydroxycinnamates in leaves of Ligustrum vulgare under excess light and drought stress. New Phytol. 2004, 163, 547–561. [Google Scholar] [CrossRef]
- Olsen, K.M.; Slimestad, R.; Lea, U.S.; Brede, C.; Lovdal, T.; Ruoff, P.; Verheul, M.; Lillo, C. Temperature and nitrogen effects on regulators and products of the flavonoid pathway: Experimental and kinetic model studies. Plant Cell Environ. 2009, 32, 286–299. [Google Scholar] [CrossRef]
- Agati, G.; Biricolti, S.; Guidi, L.; Ferrini, F.; Fini, A.; Tattini, M. The biosynthesis of flavonoids is enhanced similarly by UV radiation and root zone salinity in L. vulgare leaves. J. Plant Physiol. 2011, 168, 204–212. [Google Scholar] [CrossRef]
- Di Ferdinando, M.; Brunetti, C.; Fini, A.; Tattini, M. Flavonoids as antioxidants in plants under abiotic stresses. In Abiotic Stress Responses in Plants; Ahmad, P., Prasad, M., Eds.; Springer: New York, NY, USA, 2012; pp. 159–179. [Google Scholar]
- Xu, Z.; Zhou, J.; Ren, T.; Du, H.; Liu, H.; Li, Y.; Zhang, C. Salt stress decreases seedling growth and development but increases quercetin and kaempferol content in Apocynum venetum. Plant Biol. 2020, 22, 813–821. [Google Scholar] [CrossRef]
- Oliveira, D.M.; Mota, T.R.; Salatta, F.V.; Sinzker, R.C.; Končitíková, R.; Kopečný, D.; Simister, R.; Silva, M.; Goeminne, G.; Morreel, K.; et al. Cell wall remodeling under salt stress: Insights into changes in polysaccharides, feruloylation, lignification, and phenolic metabolism in maize. Plant Cell Environ. 2020. [Google Scholar] [CrossRef] [PubMed]
- Pi, E.; Zhu, C.; Fan, W.; Huang, Y.; Qu, L.; Li, Y.; Zhao, Q.; Ding, F.; Qiu, L.; Wang, H.; et al. Quantitative phosphoproteomic and metabolomic analyses reveal GmMYB173 optimizes flavonoid metabolism in soybean under salt stress. Mol. Cell. Proteom. 2018, 17, 1209–1224. [Google Scholar] [CrossRef] [PubMed]
- Hussain Wani, S.; Brajendra Singh, N.; Haribhushan, A.; Iqbal Mir, J. Compatible solute engineering in plants for abiotic stress tolerance-role of glycine betaine. Curr. Genom. 2013, 14, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Hoque, M.A.; Banu, M.N.A.; Nakamura, Y.; Shimoishi, Y.; Murata, Y. Proline and glycine betaine enhance antioxidant defense and methylglyoxal detoxification systems and reduce NaCl–induced damage in cultured tobacco cells. J. Plant Physiol. 2007, 165, 813–824. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.H.; Murata, N. Glycine betaine protects plants against abiotic stress: Mechanisms and biotechnological applications. Plant Cell Environ. 2011, 34, 1–20. [Google Scholar] [CrossRef]
- Ashraf, M.F.M.R.; Foolad, M.R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Woodrow, P.; Ciarmiello, L.F.; Annunziata, M.G.; Pacifico, S.; Iannuzzi, F.; Mirto, A.; D’Amelia, L.; Dell’Aversana, E.; Piccolella, S.; Fuggi, A.; et al. Durum wheat seedling responses to simultaneous high light and salinity involve a fine reconfiguration of amino acids and carbohydrate metabolism. Physiol. Plant. 2017, 159, 290–312. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Alam, M.; Rahman, A.; Hasanuzzaman, M.; Nahar, K.; Fujita, M. Exogenous proline and glycine betaine mediated upregulation of antioxidant defense and glyoxalase systems provides better protection against salt-induced oxidative stress in two rice (Oryza sativa L.) varieties. Biomed Res. Int. 2014, 2014, 17. [Google Scholar] [CrossRef]
- Hibino, T.; Meng, Y.L.; Kawamitsu, Y.; Uehara, N.; Matsuda, N.; Tanaka, Y.; Ishikawa, H.; Baba, S.; Takabe, T.; Wada, K.; et al. Molecular cloning and functional characterization of two kinds of betaine-aldehyde dehydrogenase in betaine-accumulating mangrove Avicennia marina (Forsk.) Vierh. Plant Mol. Biol. 2001, 45, 353–363. [Google Scholar] [CrossRef]
- Patterson, J.H.; Newbigin, E.D.; Tester, M.; Bacic, A.; Roessner, U. Metabolic responses to salt stress of barley (Hordeum vulgare L.) cultivars, Sahara and Clipper, which differ in salinity tolerance. J. Exp. Bot. 2009, 60, 4089–4103. [Google Scholar]
- Hu, L.; Hu, T.; Zhang, X.; Pang, H.; Fu, J. Exogenous glycine betaine ameliorates the adverse effect of salt stress on perennial ryegrass. J. Am. Soc. Hortic. Sci. 2012, 137, 38–46. [Google Scholar] [CrossRef]
- Borges, A.V.; Champenois, W. Preservation protocol for dimethylsulfoniopropionate and dimethylsulfoxide analysis in plant material of the Mediterranean seagrass Posidonia oceanica, and reevaluation of dimethylsulfoniopropionate leaf content. Aquat. Bot. 2017, 143, 8–10. [Google Scholar] [CrossRef]
- McParland, E.L.; Wright, A.; Art, K.; He, M.; Levine, N.M. Evidence for contrasting roles of dimethylsulfoniopropionate production in Emiliania huxleyi and Thalassiosira oceanica. New Phytol. 2020, 226, 396–409. [Google Scholar] [CrossRef] [PubMed]
- Wittek, B.; Carnat, G.; Tison, J.L.; Gypens, N. Response of dimethylsulfoniopropionate (DMSP) and dimethylsulfoxide (DMSO) cell quotas to salinity and temperature shifts in the sea-ice diatom Fragilariopsis cylindrus. Polar Biol. 2020, 43, 483–494. [Google Scholar] [CrossRef]
- Dickson, D.M.J.; Kirst, G.O. The role of β-dimethylsulphoniopropionate, glycine betaine and homarine in the osmoacclimation of Platymonas subcordiformis. Planta 1986, 167, 536–543. [Google Scholar] [CrossRef]
- Sunda, W.; Kieber, D.J.; Kiene, R.P.; Huntsman, S. An antioxidant function for DMSP and DMS in marine algae. Nature 2002, 418, 317–320. [Google Scholar] [CrossRef]
- Irani, S.; Todd, C.D. Exogenous allantoin increases Arabidopsis seedlings tolerance to NaCl stress and regulates expression of oxidative stress response genes. J. Plant Physiol. 2018, 221, 43–50. [Google Scholar] [CrossRef]
- Li, H.; Chang, J.; Chen, H.; Wang, Z.; Gu, X.; Wei, C.; Zhang, Y.; Ma, J.; Yang, J.; Zhang, X. Exogenous melatonin confers salt stress tolerance to watermelon by improving photosynthesis and redox homeostasis. Front. Plant Sci. 2017, 8, 295. [Google Scholar] [CrossRef]
- Ke, Q.; Ye, J.; Wang, B.; Ren, J.; Yin, L.; Deng, X.; Wang, S. Melatonin mitigates salt stress in wheat seedlings by modulating polyamine metabolism. Front. Plant Sci. 2018, 9, 914. [Google Scholar] [CrossRef]
- Siddiqui, M.H.; Alamri, S.; Al-Khaishany, M.Y.; Khan, M.N.; Al-Amri, A.; Ali, H.M.; Alaraidh, I.A.; Alsahli, A.A. Exogenous melatonin counteracts NaCl-Induced damage by regulating the antioxidant system, proline and carbohydrates metabolism in tomato seedlings. Int. J. Mol. Sci. 2019, 20, 353. [Google Scholar] [CrossRef]
- Nounjan, N.; Theerakulpisut, P. Effects of exogenous proline and trehalose on physiological responses in rice seedlings during salt-stress and after recovery. Plant Soil Environ. 2012, 58, 309–315. [Google Scholar] [CrossRef]
- Sheteiwy, M.S.; An, J.; Yin, M.; Jia, X.; Guan, Y.; He, F.; Hu, J. Cold plasma treatment and exogenous salicylic acid priming enhances salinity tolerance of Oryza sativa seedlings. Protoplasma 2019, 256, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Roychoudhury, A.; Basu, S.; Sengupta, D.N. Amelioration of salinity stress by exogenously applied spermidine or spermine in three varieties of indica rice differing in their level of salt tolerance. J. Plant Physiol. 2011, 168, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Demiral, T.; Türkan, I. Exogenous glycine betaine affects growth and proline accumulation and retards senescence in two rice cultivars under NaCl stress. Environ. Exp. Bot. 2006, 56, 72–79. [Google Scholar] [CrossRef]
- Bajwa, A.A.; Farooq, M.; Nawaz, A. Seed priming with sorghum extracts and benzyl aminopurine improves the tolerance against salt stress in wheat (Triticum aestivum L.). Physiol. Mol. Biol. Plants 2018, 24, 239–249. [Google Scholar] [CrossRef]
- Avalbaev, A.; Yuldashev, R.; Fedorova, K.; Somov, K.; Vysotskaya, L.; Allagulova, C.; Shakirova, F. Exogenous methyl jasmonate regulates cytokinin content by modulating cytokinin oxidase activity in wheat seedlings under salinity. J. Plant Physiol. 2016, 191, 101–110. [Google Scholar] [CrossRef]
- Li, S.; Jin, H.; Zhang, Q. The effect of exogenous spermidine concentration on polyamine metabolism and salt tolerance in zoysiagrass (Zoysia japonica Steud) subjected to short-term salinity stress. Front. Plant Sci. 2016, 7, 1221. [Google Scholar] [CrossRef]
- Xiong, J.L.; Wang, H.C.; Tan, X.Y.; Zhang, C.L.; Naeem, M.S. 5-aminolevulinic acid improves salt tolerance mediated by regulation of tetrapyrrole and proline metabolism in Brassica napus L. seedlings under NaCl stress. Plant Physiol. Biochem. 2018, 124, 88–99. [Google Scholar] [CrossRef]
- Jin, X.; Liu, T.; Xu, J.; Gao, Z.; Hu, X. Exogenous GABA enhances muskmelon tolerance to salinity-alkalinity stress by regulating redox balance and chlorophyll biosynthesis. BMC Plant Biol. 2019, 19, 48. [Google Scholar] [CrossRef]
- Zhou, Y.; Diao, M.; Chen, X.; Cui, J.; Pang, S.; Li, Y.; Hou, C.; Liu, H.Y. Application of exogenous glutathione confers salinity stress tolerance in tomato seedlings by modulating ions homeostasis and polyamine metabolism. Sci. Hortic. 2019, 250, 45–58. [Google Scholar] [CrossRef]
- Rouphael, Y.; Raimondi, G.; Lucini, L.; Carillo, P.; Kyriacou, M.C.; Colla, G.; Pannico, A.; El-Nakhel, C.; De Pascale, S. Physiological and metabolic responses triggered by omeprazole improve tomato plant tolerance to NaCl stress. Front. Plant Sci. 2018, 9, 249. [Google Scholar] [CrossRef] [PubMed]
- Shaki, F.; Ebrahimzadeh Maboud, H.; Niknam, V. Penconazole alleviates salt-induced damage in safflower (Carthamus tinctorius L.) plants. J. Plant Interact. 2018, 13, 420–427. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhong, M.; Shu, S.; Du, N.; He, L.; Yuan, L.; Sun, J.; Guo, S. Effects of exogenous putrescine on leaf anatomy and carbohydrate metabolism in cucumber (Cucumis sativus L.) under salt stress. J. Plant Growth Regul. 2015, 34, 451–464. [Google Scholar] [CrossRef]
- Zhang, H.J.; Zhang, N.A.; Yang, R.C.; Wang, L.; Sun, Q.Q.; Li, D.B.; Cao, Y.Y.; Weeda, S.; Zhao, B.; Ren, S.; et al. Melatonin promotes seed germination under high salinity by regulating antioxidant systems, ABA and GA4 interaction in cucumber (Cucumis sativus L.). J. Pineal Res. 2014, 57, 269–279. [Google Scholar] [CrossRef]
- Arora, D.; Bhatla, S.C. Melatonin and nitric oxide regulate sunflower seedling growth under salt stress accompanying differential expression of Cu/Zn SOD and Mn SOD. Free Radic. Biol. Med. 2017, 106, 315–328. [Google Scholar] [CrossRef]
- Li, X.; Yu, B.; Cui, Y.; Yin, Y. Melatonin application confers enhanced salt tolerance by regulating Na+ and Cl− accumulation in rice. Plant Growth Regul. 2017, 83, 441–454. [Google Scholar] [CrossRef]
- Zheng, X.; Tan, D.X.; Allan, A.C.; Zuo, B.; Zhao, Y.; Reiter, R.J.; Wang, L.; Wang, Z.; Guo, Y.; Zhou, J.; et al. Chloroplastic biosynthesis of melatonin and its involvement in protection of plants from salt stress. Sci. Rep. 2017, 7, 41236. [Google Scholar] [CrossRef]
- Chen, Y.E.; Mao, J.J.; Sun, L.Q.; Huang, B.; Ding, C.B.; Gu, Y.; Liao, J.Q.; Hu, C.; Zhang, Z.W.; Yuan, S.; et al. Exogenous melatonin enhances salt stress tolerance in maize seedlings by improving antioxidant and photosynthetic capacity. Physiol. Plant. 2018, 164, 349–363. [Google Scholar] [CrossRef]
- Zhao, G.; Zhao, Y.; Yu, X.; Kiprotich, F.; Han, H.; Guan, R.; Wang, R.; Shen, W. Nitric oxide is required for melatonin-enhanced tolerance against salinity stress in rapeseed (Brassica napus L.) seedlings. Int. J. Mol. Sci. 2018, 19, 1912. [Google Scholar] [CrossRef]
- de Freitas, P.A.F.; de Souza Miranda, R.; Marques, E.C.; Prisco, J.T.; Gomes-Filho, E. Salt tolerance induced by exogenous proline in maize is related to low oxidative damage and favorable ionic homeostasis. J. Plant Growth Regul. 2018, 37, 911–924. [Google Scholar] [CrossRef]
- de Freitas, P.A.F.; de Carvalho, H.H.; Costa, J.H.; de Souza Miranda, R.; da Cruz Saraiva, K.D.; de Oliveira, F.D.B.; Coelho, D.G.; Prisco, J.T.; Gomes-Filho, E. Salt acclimation in sorghum plants by exogenous proline: Physiological and biochemical changes and regulation of proline metabolism. Plant Cell Rep. 2019, 38, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Pál, M.; Szalai, G.; Janda, T. Speculation: Polyamines are important in abiotic stress signaling. Plant Sci. 2015, 237, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Shao, Q.; Yin, L.; Younis, A.; Zheng, B. Polyamine function in plants: Metabolism, regulation on development, and roles in abiotic stress responses. Front. Plant Sci. 2019, 9, 1945. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Yu, H.M.; Meng, X.F.; Lin, J.S.; Li, Y.J.; Hou, B.K. Ectopic expression of glycosyltransferase UGT 76E11 increases flavonoid accumulation and enhances abiotic stress tolerance in Arabidopsis. Plant Biol. 2018, 20, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Chen, S.; Xie, L.; Lu, Z.; Liu, M.; Han, X.; Qiao, G.; Jiang, J.; Zhuo, R.; Qiu, W.; et al. Overexpression of cysteine protease gene from Salix matsudana enhances salt tolerance in transgenic Arabidopsis. Environ. Exp. Bot. 2018, 147, 53–62. [Google Scholar] [CrossRef]
- Karthikeyan, A.; Pandian, S.K.; Ramesh, M. Transgenic indica rice cv. ADT 43 expressing a Δ1-pyrroline-5-carboxylate synthetase (P5CS) gene from Vigna aconitifolia demonstrates salt tolerance. Plant Cell Tiss. Org. Cult. 2011, 107, 383–395. [Google Scholar] [CrossRef]
- Hmida-Sayari, A.; Gargouri-Bouzid, R.; Bidani, A.; Jaoua, L.; Savouré, A.; Jaoua, S. Overexpression of Δ1-pyrroline-5-carboxylate synthetase increases proline production and confers salt tolerance in transgenic potato plants. Plant Sci. 2005, 169, 746–752. [Google Scholar] [CrossRef]
- Kumar, V.; Shriram, V.; Kishor, P.K.; Jawali, N.; Shitole, M.G. Enhanced proline accumulation and salt stress tolerance of transgenic indica rice by over-expressing P5CSF129A gene. Plant Biotechnol. Rep. 2010, 4, 37–48. [Google Scholar] [CrossRef]
- Espasandin, F.D.; Calzadilla, P.I.; Maiale, S.J.; Ruiz, O.A.; Sansberro, P.A. Overexpression of the arginine decarboxylase gene improves tolerance to salt stress in Lotus tenuis plants. J. Plant Growth Regul. 2018, 37, 156–165. [Google Scholar] [CrossRef]
- Capell, T.; Bassie, L.; Christou, P. Modulation of the polyamine biosynthetic pathway in transgenic rice confers tolerance to drought stress. Proc. Natl. Acad. Sci. USA 2004, 101, 9909–9914. [Google Scholar] [CrossRef]
- He, C.; Yang, A.; Zhang, W.; Gao, Q.; Zhang, J. Improved salt tolerance of transgenic wheat by introducing betA gene for glycine betaine synthesis. Plant Cell Tiss. Org. Cult. 2010, 101, 65–78. [Google Scholar] [CrossRef]
- Zhang, H.; Dong, H.; Li, W.; Sun, Y.; Chen, S.; Kong, X. Increased glycine betaine synthesis and salinity tolerance in AhCMO transgenic cotton lines. Mol. Breed. 2009, 23, 289–298. [Google Scholar] [CrossRef]
- Roy, M.; Wu, R. Overexpression of S-adenosylmethionine decarboxylase gene in rice increases polyamine level and enhances sodium chloride-stress tolerance. Plant Sci. 2002, 163, 987–992. [Google Scholar] [CrossRef]
- Jang, I.C.; Oh, S.J.; Seo, J.S.; Choi, W.B.; Song, S.I.; Kim, C.H.; Kim, Y.S.; Seo, H.S.; Do Choi, Y.; Nahm, B.H.; et al. Expression of a bifunctional fusion of the Escherichia coli genes for trehalose-6-phosphate synthase and trehalose-6-phosphate phosphatase in transgenic rice plants increases trehalose accumulation and abiotic stress tolerance without stunting growth. Plant Physiol. 2003, 131, 516–524. [Google Scholar] [CrossRef]
- Garg, A.K.; Kim, J.K.; Owens, T.G.; Ranwala, A.P.; Do Choi, Y.; Kochian, L.V.; Wu, R.J. Trehalose accumulation in rice plants confers high tolerance levels to different abiotic stresses. Proc. Natl. Acad. Sci. USA 2002, 99, 15898–15903. [Google Scholar] [CrossRef]
- Li, H.W.; Zang, B.S.; Deng, X.W.; Wang, X.P. Overexpression of the trehalose-6-phosphate synthase gene OsTPS1 enhances abiotic stress tolerance in rice. Planta 2011, 234, 1007–1018. [Google Scholar] [CrossRef]
- Abebe, T.; Guenzi, A.C.; Martin, B.; Cushman, J.C. Tolerance of mannitol-accumulating transgenic wheat to water stress and salinity. Plant Physiol. 2003, 131, 1748–1755. [Google Scholar] [CrossRef]
- Saibi, W.; Feki, K.; Mahmoud, R.B.; Brini, F. Durum wheat dehydrin (DHN-5) confers salinity tolerance to transgenic Arabidopsis plants through the regulation of proline metabolism and ROS scavenging system. Planta 2015, 242, 1187–1194. [Google Scholar] [CrossRef]
- Li, C.; Liu, S.; Yao, X.; Wang, J.; Wang, T.; Zhang, Z.; Zhang, P.; Chen, K. PnF3H, a flavanone 3-hydroxylase from the Antarctic moss Pohlia nutans, confers tolerance to salt stress and ABA treatment in transgenic Arabidopsis. Plant Growth Regul. 2017, 83, 489–500. [Google Scholar] [CrossRef]
- Gao, M.; Sakamoto, A.; Miura, K.; Murata, N.; Sugiura, A.; Tao, R. Transformation of Japanese persimmon (Diospyros kaki Thunb.) with a bacterial gene for choline oxidase. Mol. Breed. 2000, 6, 501–510. [Google Scholar] [CrossRef]
- Gao, M.; Tao, R.; Miura, K.; Dandekar, A.M.; Sugiura, A. Transformation of Japanese persimmon (Diospyros kaki Thunb.) with apple cDNA encoding NADP-dependent sorbitol-6-phosphate dehydrogenase. Plant Sci. 2001, 160, 837–845. [Google Scholar] [CrossRef]
- Ali, A.; Ali, Q.; Iqbal, M.S.; Nasir, I.A.; Wang, X. Salt tolerance of potato genetically engineered with the Atriplex canescens BADH gene. Biol. Plant. 2020, 64, 271–279. [Google Scholar] [CrossRef]
- Rezaei Qusheh Bolagh, F.; Solouki, A.; Tohidfar, M.; Zare Mehrjerdi, M.; Izadi-Darbandi, A.; Vahdati, K. Agrobacterium-mediated transformation of Persian walnut using BADH gene for salt and drought tolerance. J. Hortic. Sci. Biotechnol. 2020, 1–10. [Google Scholar] [CrossRef]
- Wen, X.P.; Pang, X.M.; Matsuda, N.; Kita, M.; Inoue, H.; Hao, Y.J.; Honda, C.; Moriguchi, T. Over-expression of the apple spermidine synthase gene in pear confers multiple abiotic stress tolerance by altering polyamine titers. Transgenic Res. 2008, 17, 251–263. [Google Scholar] [CrossRef]
- Zhai, H.; Wang, F.; Si, Z.; Huo, J.; Xing, L.; An, Y.; He, S.; Liu, Q. A myo-inositol-1-phosphate synthase gene, IbMIPS1, enhances salt and drought tolerance and stem nematode resistance in transgenic sweet potato. Plant Biotechnol. J. 2016, 14, 592–602. [Google Scholar] [CrossRef]
- Hu, L.; Zhou, K.; Liu, Y.; Yang, S.; Zhang, J.; Gong, X.; Ma, F. Overexpression of MdMIPS1 enhances salt tolerance by improving osmosis, ion balance, and antioxidant activity in transgenic apple. Plant Sci. 2020, 301, 110654. [Google Scholar] [CrossRef]
- Zhao, M.; Liu, H.; Deng, Z.; Chen, J.; Yang, H.; Li, H.; Xia, Z.; Li, D. Molecular cloning and characterization of S-adenosylmethionine decarboxylase gene in rubber tree (Hevea brasiliensis). Physiol. Mol. Biol. Plants 2017, 23, 281–290. [Google Scholar] [CrossRef]
- Walia, H.; Wilson, C.; Condamine, P.; Liu, X.; Ismail, A.M.; Zeng, L.; Wanamaker, S.I.; Mandal, J.; Xu, J.; Cui, X.; et al. Comparative transcriptional profiling of two contrasting rice genotypes under salinity stress during the vegetative growth stage. Plant Physiol. 2005, 139, 822–835. [Google Scholar] [CrossRef]
- Hussain, S.S.; Raza, H.; Afzal, I.; Kayani, M.A. Transgenic plants for abiotic stress tolerance: Current status. Arch. Agron. Soil Sci. 2012, 58, 693–721. [Google Scholar] [CrossRef]
- Kumar, K.; Gambhir, G.; Dass, A.; Tripathi, A.K.; Singh, A.; Jha, A.K.; Yadava, P.; Choudhary, M.; Rakshit, S. Genetically modified crops: Current status and future prospects. Planta 2020, 251, 1–27. [Google Scholar] [CrossRef]
| Plant Species | Tissue | Methods of Analysis | Up-Regulated Metabolites during Salt Stress | Down-Regulated Metabolites during Salt Stress | References |
|---|---|---|---|---|---|
| Aeluropus Lagopoides | Shoots | CE-MS | AA: Alanine, asparagine, lysine, glutamine, arginine, glycine, proline, histidine, phenylalanine, serine, valine, tyrosine and cytosine OM: Adenine, adenosine and adenosine 5′-monophosphate | OM: Citrate, aconitate, 2-oxoglutarate, succinate, fumarate, ribulose-5-phosphate, ribose-5- phosphate and glucose-6-phosphate | [52] |
| Hordeum. vulgare | Leaves and roots | HPLC | In roots: CH: Sorbitol, mannitol and pinitol In leaves: AA: Proline OM: Glycine betaine | In roots: OM: Glycine betaine (undetectable) In leaves: CH: Sorbitol, mannitol and pinitol | [56] |
| H. spontaneum, H. vulgare | Leaves and roots | GC-MS | In roots: CH: Sucrose, trehalose, turanose, inositol, mannitol and xylitol AA: Proline OM: Citric acid and isocitric acid In leaves: CH: Isomaltose, raffinose and glucose AA: Asparagine, glycine, isoleucine, leucine, proline and serine OM: Pipecolic acid, glucose-6-phospate, fructose-6-phospate, 3-phosphoglyceric acid, pyruvate, uracil, diethylphosphate and putrescine | In roots: AA: Asparagine, β-alanine and valine OM: Fructose-6- phospate, glucose-6- phosphate, 3-phosphoglyceric acid, α-ketoglutaric acid, ascorbic acid, gluconic acid, orotic acid, pyroglutamic acid and threonic acid In leaves: CH: Sucrose, maltose, mannitol, glycerol and inositol FA: Palmitic acid OM: Citric acid, α-ketoglutaric acid, fumaric acid, malic acid, succinic acid, glucosan, menthol, 4-aminobutyrate, ascorbic acid, benzoic acid, pyroglutamic acid and threonic acid | [44] |
| Zea mays | Shoots and roots | 1H-NMR | In shoots: CH: Sucrose AA: Alanine, glutamate and asparagine OM: Glycine betaine In roots: CH: Sucrose AA: Alanine OM: γ-amino-N-butyric acid, malic acid and succinate | In shoots: CH: Glucose OM: Citrate, malic acid, 2-oxoglutarate and trans-aconitic acid In roots: CH: Glucose OM: Acetoacetate | [57] |
| Arabidopsis thaliana | Roots and shoots | GC-MS | CH: Galactose, sucrose, trehalose, gentiobiose, melibiose, xylitol, galactinol, galactonate and threonate AA: Threonine, β-alanine, 5-oxoproline, glutamic acid, asparagine and glutamine OM: Glycerol-3-phosphate and myo-inositol-1-phosphate | CH: Fructose, erythritol and xylitol AA: Isoleucine and proline OM: Sinapinate, urea, succinate, citrate, aconitate, fumarate, malate, glycerate, propanoate and butyrate | [58] |
| A. thaliana | Whole Plant | GC-HP5890 | CH: Sucrose, fructose, sorbose, raffinose and inositol AA: Glycine, proline serine, glutamic acid and threonine OM: Citric acid, galactinol, malic acid and phosphoric acid | CH: Glucose, glycerol, maltose and trehalose AA: Aspartic acid OM: Fumaric acid and succinic acid | [59] |
| Lepidium latifolium | Shoots and roots | HPLC and 1H-NMR | In shoots: CH: Sucrose, fructose, glucose, meso-inositol and chiro-inositol AA: Proline In roots: CH: Sucrose, fructose, glucose, meso-inositol and chiro-inositol AA: Proline and β-alanine OM: Choline-O-sulfate and β-alanine betaine | In shoots: AA: β-Alanine, glutamic acid and glutamine OM: Choline, choline-O-sulfate, β-alanine betaine, malate and citrate In roots: AA: Glutamic acid and glutamine OM: Choline, malate and citrate | [60] |
| Thellungiella halophila | Whole plant | GC-HP5890 | CH: Fructose, sorbose, galactinol, glucose, glycerol, inositol, raffinose and trehalose AA: Aspartic acid glutamic acid, proline, glycine, serine and threonine | CH: Sucrose and maltose FA: palmitic acid and stearic acid OM: Citric acid, fumaric acid, malic acid and phosphoric acid | [59] |
| Cicer arietinum | Flower and pod tissues | GC-QqQ-MS and LC-MS | CH: Gentiobiose, fructose, sucrose and erythritol AA: Arginine, glutamic acid, glycine, histidine, homoserine, hydroxyproline, isoleucine, leucine, lysine, methionine, proline, threonine, tryptophan and valine OM: Pipecolate, isocitrate, cis-aconitate, citrate, fumarate, malate, citrate and 2-oxoglutarate | CH: Arabinose, erythritol and inositol AA: Cysteine OM: Putrescine and GABA | [45] |
| Glycine max, G. soja | Leaves | GC-MS and LC-FT/MS | CH: Lactitol and maltitol FA: Linolenic acid OM: Abscisic acid and caffeic acid | CH: Sucrose AA: Alanine OM: Glutathione | [51] |
| Nicotiana tabacum | Aerial Part | 1H-NMR | CH: Glucose, fructose, sucrose and myo-inositol AA: Glutamine, proline, asparagine, valine, isoleucine, phenylalanine, tryptophan and tyrosine OM: Succinate, nicotine, formate and allantoin | AA: Aspartate and alanine OM: Malate, γ-amino-n-butyrate, choline, ethanolamine, hypoxanthine, dimethylamine, N-methylnicotinamide, uracil and uridine | [61] |
| Solanum lycopersicon | Leaves | UHPLC-QTOF-MS | SM: 1,3-dicaffeoylquinic acid, 1-feruoyl-5-caffeoylquinic acid, 3-caffeoyl-1-5 quinolactone and quercetin-3-hexodide | SM: Kaempferol, dihydrokaempferol, kaempferol 3-O-glucoside, naringenin, naringenin chalcone and quercetin-3-rutenoside | [62] |
| Cuminum. cyminum | Shoots | GC-MS and HPLC | AA: Isoleucine, glycine, proline, leucine and glutamate FA: Myristic acid, pentadeconoic acid, heptadecanoic acid, oleic acid, cis-11,14,17-eicosadienoic acid, heneicosanoic acid, palmitic acid, stearic acid and cis-11,14-eicosadienoic acid | FA: α-linolenic acid, cis-11-eicosadienoic acid, behenic acid, tricosanoic acid and linoleic acid | [9] |
| Matricaria chamomilla | Leaves and roots | HPLC | In rosette leaves: AA: Alanine, proline and tyrosine OM: Salicylic acid SM: protocatechuic, p-hydroxybenzoic ferulic acid, o-coumaric and p-coumaric In roots: AA: Alanine and proline SM: Chlorogenic, caffeic acid and p-coumaric | In rosette leaves: AA: Glutamic acid, serine, cysteine and lysine SM: Salicylic acid, chlorogenic acid and caffeic acid In roots: AA: Aspartic acid, glycine and phenylalanine SM: Benzoic acid, protocatechuic, protocatechuic, p-hydroxybenzoic aldehyde and o-coumaric | [63] |
| Fagopyrum esculentum | Sprouts | UFLC | SM: Isoorientin, orientin, rutin and vitexin | [64] | |
| Lotus sp. | Shoots | GC/EI-TOF-MS | CH: Sucrose, myo-inositol and fructose AA: Proline, threonine, serine, glycine and phenylalanine OM: Glycerophosphoglycerol | OM: Citric acid, malic acid, succinic, fumaric, erythronic, glycolic and aconitic acid | [65] |
| Nitraria tangutorum | Cell suspensions | GC-MS | AA: Alanine, valine, serine, proline and asparagine FA: Hexadecanoic acid, octadecanoic acid and 9,12-octadecadienoic acid OM: Galactofuranose and succinate SM: Sitosterol | OM: Malic acid and acetamide | [66] |
| Olea europaea | Leaves and roots | HPLC | SM: Oleuropein | SM: Hydroxytyrosol | [67] |
| Rosmarinus officinalis | Leaves | HPLC | SM: Cineole and camphor | SM: Nopol, α-terpineol, borneol and camphene | [68] |
| Salvia mirzayanii | Leaves | HGC-MS | OM: α-terpinyl acetate, 1,8-cineole and linalyl acetate | OM: Bicyclogermacrene | [69] |
| Salicornia brachiata | Shoots | GC-MS, HPLC | AA: Proline, valine, isoleucine, leucine, cysteine, methionine and tyrosine FA: Tridecanoic acid, heptadecanoic acid, stearic acid, oleic acid, linoleic acid, α-linolenic acid, arachidic acid, heneicosanoic acid and lignoceric acid | AA: Glycine, arginine and serine OM: Myristoleic acid, pentadecanoic acid, palmitic acid and palmitoleic acid | [28] |
| Atriplex. halimus | Seedling | GC-FID and UPLC | CH: Saccharose and myo-inositol AA: Alanine, proline, arginine threonine, glycine, valine, leucine, phenylalanine and tryptophan | CH: Malate | [46] |
| Suaeda corniculata | Leaves | 1H-NMR | OM: Betaine | CH: Sucrose, glucose and fructose AA: Valine, glycine, alanine, leucine, isoleucine, glutamine, glutamate, aspartate and threonine OM: Malate, succinate, 2-oxoglutarate, fumarate, dimethylamine and choline | [53] |
| Suaeda maritima | Shoots and roots | GC-FID, UPLC and 1H-NMR | CH: Sucrose AA: Proline, valine, glutamine and glycine OM: Betaine, citrate and glycerate | CH: Glucose, fructose and myo-nositol AA: Serine, glutamate isoleucine and threonine OM: GABA | [70] |
| Metabolites | Plant Species | Stress Tolerance | References |
|---|---|---|---|
| Proline and trehalose | Oryza sativa | Salt stress | [119] |
| Salicylic acid | O. sativa | Salt stress | [120] |
| Spermidine or spermine | O. sativa | Salt stress | [121] |
| Glycine betaine | O. sativa | Salt stress | [122] |
| Sorghum extracts and benzyl aminopurine | Triticum aestivum | Salt stress | [123] |
| Methyl jasmonate | T. aestivum | Salt stress | [124] |
| Spermidine | Zoysia japonica | Salt stress | [125] |
| 5-aminolevulinic acid | Brassica napus | Salt stress | [126] |
| Allantoin | Arabidopsis thaliana | Salt stress | [115] |
| γ-aminobutyrate | Cucumis melo | Saline- alkaline stress | [127] |
| Glutathione | Solanum lycopersicum | Salt stress | [128] |
| Omeprazole | S. lycopersicum | Salt stress | [129] |
| Penconazole | Carthamus tinctorius | Salt stress | [130] |
| Putrescine | C. sativus | Salt stress | [131] |
| Melatonin | Citrullus lanatus; T. aestivum; S. lycopersicum; C. sativus; Helianthus annuus; O. sativa; A. thaliana; Zea mays; B. napus | Salt stress | [116,117,118,132,133,134,135,136,137] |
| Proline | Z. mays; Sorghum bicolor | Salt stress | [138,139] |
| Gene | Locus ID | Source | Transgenic Plants | Stress Tolerance | References |
|---|---|---|---|---|---|
| ∆1-pyrroline-5-carboxylate synthetase (P5CS) | VIRPYRR | Vigna aconitifolia | Oryza sativa | Salt stress | [144] |
| ∆1-pyrroline-5-carboxylate synthetase (P5CSF129A) | P5CS_VIGAC | V. aconitifolia | O. sativa | Salt stress | [146] |
| S-adenosylmethionine decarboxylase (SAMDC) | CAA58762 | Tritordeum | O. sativa | Salt stress | [151] |
| Trehalose-6-phosphate synthase (TPS) and trehalose-6-phosphate phosphatase (TPP) | EU070413 and NC_002695 | Escherichia coli | O. sativa | Salt stress | [152] |
| Trehalose-6-phosphate synthase (TPS; otsA) and Trehalose-6-phosphate phosphatase (TPP; otsB) | NC_000913 | E. coli | O. sativa | Salinity, cold (10 °C) and dehydration | [153] |
| Trehalose-6-phosphate synthase1 | HM050424 | O. sativa | O. sativa | Salinity and polyethylene glycol | [154] |
| Choline dehydrogenase (betA) | NC_000913 | E. coli | Triticum aestivum | Salt stress | [149] |
| Mannitol-1-phosphate dehydrogenase (mtlD) | EFF7369098 | E. coli | T. aestivum | Salinity and polyethylene glycol | [155] |
| Cysteine protease (SmCP) | KC715825 | Salix matsudana | Arabidopsis | Salt stress | [143] |
| Dehydrin (DHN-5) | CAY85463 | T. aestivum | Arabidopsis | Salt stress | [156] |
| Flavanone 3-hydroxylase (PnF3H) | MK036761 | Pohlia nutans | Arabidopsis | Salinity and oxidative stress | [157] |
| Choline oxidase (codA) | AY304485 | Arthrobacter. globiformis | Diospyros kaki | Salt stress | [158] |
| Sorbitol-6-phosphate dehydrogenase (S6PDH) | NM_001294028 | Malus domestica | D. kaki | Salt stress | [159] |
| Arginine decarboxylase (ADC) | BQ739966 | Avena sativa | Lotus tenuis | Salt stress | [147] |
| Choline monooxygenase (AhCMO) | AF270651 | Atriplex hortensis | Gossypium hirsutum | Salt stress | [150] |
| Betaine aldehyde dehydrogenase (BADH) | JF776157 | A. canescens | Solanum tuberosum | Salt stress | [160] |
| Betaine aldehyde dehydrogenase (BADH) | FJ595952 | Spinacia oleracea | Juglans regia | salinity and drought | [161] |
| Spermidine synthase (MdSPDS1) | LOC103451952 | M. domestica | Pyrus communis | Salt, osmotic and copper stresses | [162] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patel, M.K.; Kumar, M.; Li, W.; Luo, Y.; Burritt, D.J.; Alkan, N.; Tran, L.-S.P. Enhancing Salt Tolerance of Plants: From Metabolic Reprogramming to Exogenous Chemical Treatments and Molecular Approaches. Cells 2020, 9, 2492. https://doi.org/10.3390/cells9112492
Patel MK, Kumar M, Li W, Luo Y, Burritt DJ, Alkan N, Tran L-SP. Enhancing Salt Tolerance of Plants: From Metabolic Reprogramming to Exogenous Chemical Treatments and Molecular Approaches. Cells. 2020; 9(11):2492. https://doi.org/10.3390/cells9112492
Chicago/Turabian StylePatel, Manish Kumar, Manoj Kumar, Weiqiang Li, Yin Luo, David J. Burritt, Noam Alkan, and Lam-Son Phan Tran. 2020. "Enhancing Salt Tolerance of Plants: From Metabolic Reprogramming to Exogenous Chemical Treatments and Molecular Approaches" Cells 9, no. 11: 2492. https://doi.org/10.3390/cells9112492
APA StylePatel, M. K., Kumar, M., Li, W., Luo, Y., Burritt, D. J., Alkan, N., & Tran, L.-S. P. (2020). Enhancing Salt Tolerance of Plants: From Metabolic Reprogramming to Exogenous Chemical Treatments and Molecular Approaches. Cells, 9(11), 2492. https://doi.org/10.3390/cells9112492










